Abstract
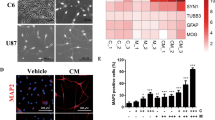
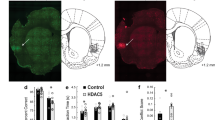
Histone deacetylases (HDACs) are promising therapeutic targets for neurological and psychiatric disorders that impact cognitive ability, but the relationship between various HDAC isoforms and cognitive improvement is poorly understood, particularly in mouse models of memory impairment. A goal shared by many is to develop HDAC inhibitors with increased isoform selectivity in order to reduce unwanted side effects, while retaining procognitive effects. However, studies addressing this tack at the molecular, cellular and behavioral level are limited. Therefore, we interrogated the biological effects of class I HDAC inhibitors with varying selectivity and assessed a subset of these compounds for their ability to regulate transcriptional activity, synaptic function and memory. The HDAC-1, -2, and -3 inhibitors, RGFP963 and RGFP968, were most effective at stimulating synaptogenesis, while the selective HDAC3 inhibitor, RGFP966, with known memory enhancing abilities, had minimal impact. Furthermore, RGFP963 increased hippocampal spine density, while HDAC3 inhibition was ineffective. Genome-wide gene expression analysis by RNA sequencing indicated that RGFP963 and RGFP966 induce largely distinct transcriptional profiles in the dorsal hippocampus of mature mice. The results of bioinformatic analyses were consistent with RGFP963 inducing a transcriptional program that enhances synaptic efficacy. Finally, RGFP963, but not RGFP966, rescued memory in a mouse model of Alzheimer’s Disease. Together, these studies suggest that the specific memory promoting properties of class I HDAC inhibitors may depend on isoform selectivity and that certain pathological brain states may be more receptive to HDAC inhibitors that improve network function by enhancing synapse efficacy.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Abel T, Zukin RS (2008). Epigenetic targets of HDAC inhibition in neurodegenerative and psychiatric disorders. Curr Opin Pharmacol 8: 57–64.
Akhtar MW, Raingo J, Nelson ED, Montgomery RL, Olson EN, Kavalali ET et al (2009). Histone deacetylases 1 and 2 form a developmental switch that controls excitatory synapse maturation and function. J Neurosci 29: 8288–8297.
Blank M, Dornelles AS, Werenicz A, Velho LA, Pinto DF, Fedi AC et al (2014). Basolateral amygdala activity is required for enhancement of memory consolidation produced by histone deacetylase inhibition in the hippocampus. Neurobiol Learn Mem 111: 1–8.
Bowers ME, Xia B, Carreiro S, Ressler KJ (2015). The Class I HDAC inhibitor RGFP963 enhances consolidation of cued fear extinction. Learn Mem 22: 225–231.
Broide RS, Redwine JM, Aftahi N, Young W, Bloom FE, Winrow CJ (2007). Distribution of histone deacetylases 1-11 in the rat brain. J Mol Neurosci 31: 47–58.
Day JJ, Sweatt JD (2011). Epigenetic mechanisms in cognition. Neuron 70: 813–829.
Fass DM, Reis SA, Ghosh B, Hennig KM, Joseph NF, Zhao WN, Nieland TJ et al (2013). Crebinostat: a novel cognitive enhancer that inhibits histone deacetylase activity and modulates chromatin-mediated neuroplasticity. Neuropharmacology 64: 81–96.
Feng G, Mellor RH, Bernstein M, Keller-Peck C, Nguyen QT, Wallace M et al (2000). Imaging neuronal subsets in transgenic mice expressing multiple spectral variants of GFP. Neuron 28: 41–51.
Fischer A, Sananbenesi F, Wang X, Dobbin M, Tsai LH (2007). Recovery of learning and memory is associated with chromatin remodelling. Nature 447: 178–182.
Graff J, Rei D, Guan JS, Wang WY, Seo J, Hennig KM et al (2012). An epigenetic blockade of cognitive functions in the neurodegenerating brain. Nature 483: 222–226.
Graff J, Tsai LH (2013). The potential of HDAC inhibitors as cognitive enhancers. Annu Rev Pharmacol Toxicol 53: 311–330.
Guan JS, Haggarty SJ, Giacometti E, Dannenberg JH, Joseph N, Gao J et al (2009). HDAC2 negatively regulates memory formation and synaptic plasticity. Nature 459: 55–60.
Haggarty SJ, Tsai LH (2011). Probing the role of HDACs and mechanisms of chromatin-mediated neuroplasticity. Neurobiol Learn Mem 96: 41–52.
Harms KJ, Tovar KR, Craig AM (2005). Synapse-specific regulation of AMPA receptor subunit composition by activity. J Neurosci 25: 6379–6388.
Hawk JD, Florian C, Abel T (2011). Post-training intrahippocampal inhibition of class I histone deacetylases enhances long-term object-location memory. Learn Mem 18: 367–370.
Herman D, Jenssen K, Burnett R, Soragni E, Perlman SL, Gottesfeld JM (2006). Histone deacetylase inhibitors reverse gene silencing in Friedreich's ataxia. Nat Chem Biol 2: 551–558.
Intlekofer KA, Berchtold NC, Malvaez M, Carlos AJ, McQuown SC, Cunningham MJ et al (2013). Exercise and sodium butyrate transform a subthreshold learning event into long-term memory via a brain-derived neurotrophic factor-dependent mechanism. Neuropsychopharmacology 38: 2027–2034.
Jankowsky JL, Xu G, Fromholt D, Gonzales V, Borchelt DR (2003). Environmental enrichment exacerbates amyloid plaque formation in a transgenic mouse model of Alzheimer disease. J Neuropathol Exp Neurol 62: 1220–1227.
Jia H, Kast RJ, Steffan JS, Thomas EA (2012). Selective histone deacetylase (HDAC) inhibition imparts beneficial effects in Huntington's disease mice: implications for the ubiquitin-proteasomal and autophagy systems. Hum Mol Genet 21: 5280–5293.
Kilgore M, Miller CA, Fass DM, Hennig KM, Haggarty SJ, Sweatt JD et al (2010). Inhibitors of class 1 histone deacetylases reverse contextual memory deficits in a mouse model of Alzheimer's disease. Neuropsychopharmacology 35: 870–880.
Knobloch M, Mansuy IM (2008). Dendritic spine loss and synaptic alterations in Alzheimer's disease. Mol Neurobiol 37: 73–82.
Levenson JM, O'Riordan KJ, Brown KD, Trinh MA, Molfese DL, Sweatt JD (2004). Regulation of histone acetylation during memory formation in the hippocampus. J Biol Chem 279: 40545–40559.
MacDonald JL, Roskams AJ (2008). Histone deacetylases 1 and 2 are expressed at distinct stages of neuro-glial development. Dev Dyn 237: 2256–2267.
Mahan AL, Mou L, Shah N, Hu JH, Worley PF, Ressler KJ (2012). Epigenetic modulation of Homer1a transcription regulation in amygdala and hippocampus with pavlovian fear conditioning. J Neurosci 32: 4651–4659.
Malvaez M, McQuown SC, Rogge GA, Astarabadi M, Jacques V, Carreiro S et al (2013). HDAC3-selective inhibitor enhances extinction of cocaine-seeking behavior in a persistent manner. Proc Natl Acad Sci USA 110: 2647–2652.
Malvaez M, Sanchis-Segura C, Vo D, Lattal KM, Wood MA (2010). Modulation of chromatin modification facilitates extinction of cocaine-induced conditioned place preference. Biol Psychiatry 67: 36–43.
McQuown SC, Barrett RM, Matheos DP, Post RJ, Rogge GA, Alenghat T et al (2011). HDAC3 is a critical negative regulator of long-term memory formation. J Neurosci 31: 764–774.
Mikaelsson MA, Miller CA (2011). The path to epigenetic treatment of memory disorders. Neurobiol Learn Mem 96: 13–18.
Monsey MS, Ota KT, Akingbade IF, Hong ES, Schafe GE (2011). Epigenetic alterations are critical for fear memory consolidation and synaptic plasticity in the lateral amygdala. PloS One 6: e19958.
Morris MJ, Mahgoub M, Na ES, Pranav H, Monteggia LM (2013). Loss of histone deacetylase 2 improves working memory and accelerates extinction learning. J Neurosci 33: 6401–6411.
Nistico R, Pignatelli M, Piccinin S, Mercuri NB, Collingridge G (2012). Targeting synaptic dysfunction in Alzheimer's disease therapy. Mol Neurobiol 46: 572–587.
Pozueta J, Lefort R, Shelanski ML (2013). Synaptic changes in Alzheimer's disease and its models. Neuroscience 251: 51–65.
Rai M, Soragni E, Jenssen K, Burnett R, Herman D, Coppola G et al (2008). HDAC inhibitors correct frataxin deficiency in a Friedreich ataxia mouse model. PloS One 3: e1958.
Ricobaraza A, Cuadrado-Tejedor M, Perez-Mediavilla A, Frechilla D, Del Rio J, Garcia-Osta A (2009). Phenylbutyrate ameliorates cognitive deficit and reduces tau pathology in an Alzheimer's disease mouse model. Neuropsychopharmacology 34: 1721–1732.
Rogge GA, Singh H, Dang R, Wood MA (2013). HDAC3 is a negative regulator of cocaine-context-associated memory formation. J Neurosci 33: 6623–6632.
Rudenko A, Tsai LH (2014). Epigenetic modifications in the nervous system and their impact upon cognitive impairments. Neuropharmacology 80C: 70–82.
Shi P, Scott MA, Ghosh B, Wan D, Wissner-Gross Z, Mazitschek R et al (2011). Synapse microarray identification of small molecules that enhance synaptogenesis. Nat Commun 2: 510.
Spires-Jones T, Knafo S (2012). Spines, plasticity, and cognition in Alzheimer's model mice. Neural Plast 2012: 319836.
Terry RD, Masliah E, Salmon DP, Butters N, DeTeresa R, Hill R et al (1991). Physical basis of cognitive alterations in Alzheimer's disease: synapse loss is the major correlate of cognitive impairment. Ann Neurol 30: 572–580.
Thomas EA, Coppola G, Desplats PA, Tang B, Soragni E, Burnett R et al (2008). The HDAC inhibitor 4b ameliorates the disease phenotype and transcriptional abnormalities in Huntington's disease transgenic mice. Proc Natl Acad Sci USA 105: 15564–15569.
Wagner FF, Zhang YL, Fass DM, Joseph N, Gale JP, Weiwer M et al (2015). Kinetically selective inhibitors of histone deacetylase 2 (HDAC2) as cognition enhancers. Chem Sci 6: 804–815.
Wells CE, Bhaskara S, Stengel KR, Zhao Y, Sirbu B, Chagot B et al (2013). Inhibition of histone deacetylase 3 causes replication stress in cutaneous T cell lymphoma. PLoS One 8: e68915.
Acknowledgements
We would like to thank Mohammad Fallahi of the Scripps Florida Bioinformatics Core for his assistance with the IPA analysis. This work was supported by grants to CAM from The National Institute on Drug Abuse (R01DA034116 and R03DA033499); to GR from The National Institute for Neurological Disorders and Stroke (R01NS064079 and R21NS082640), The National Institute for Mental Health (R01MH096847), and Repligen Corporation; and to JDS from the National Institute for Mental Health (R01MH57014).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Supplementary Information accompanies the paper on the Neuropsychopharmacology website
Rights and permissions
About this article
Cite this article
Rumbaugh, G., Daws, S., Ozkan, E. et al. Pharmacological Selectivity Within Class I Histone Deacetylases Predicts Effects on Synaptic Function and Memory Rescue. Neuropsychopharmacol 40, 2307–2316 (2015). https://doi.org/10.1038/npp.2015.93
Received:
Revised:
Accepted:
Published:
Issue date:
DOI: https://doi.org/10.1038/npp.2015.93
This article is cited by
-
Trichostatin A ameliorates Alzheimer’s disease-related pathology and cognitive deficits by increasing albumin expression and Aβ clearance in APP/PS1 mice
Alzheimer's Research & Therapy (2021)
-
How the epigenome integrates information and reshapes the synapse
Nature Reviews Neuroscience (2019)
-
The development prospection of HDAC inhibitors as a potential therapeutic direction in Alzheimer’s disease
Translational Neurodegeneration (2017)
-
Context and Auditory Fear are Differentially Regulated by HDAC3 Activity in the Lateral and Basal Subnuclei of the Amygdala
Neuropsychopharmacology (2017)
-
A First-in-Class Small-Molecule that Acts as a Dual Inhibitor of HDAC and PDE5 and that Rescues Hippocampal Synaptic Impairment in Alzheimer’s Disease Mice
Neuropsychopharmacology (2017)