Abstract
Cyclin-dependent kinase-like 5 (CDKL5) deficiency disorder (CDD) is a severe developmental and epileptic encephalopathy characterized by early onset drug-resistant seizures and later cognitive and social impairments. Existing therapies primarily involve antiseizure medications, which have sedative side effects and lack effective treatments for behavioral impairments. Potassium chloride cotransporter (KCC2) activity is regulated by phosphorylation and is a crucial component of the GABAergic inhibitory system. However, KCC2 dysfunction in CDD remains poorly understood. Here, to investigate potential KCC2 dysfunction, we used a constitutive Cdkl5 knockout mouse model of CDD. We used liquid chromatography coupled with tandem mass spectrometry and quantitative analysis to examine the unbiased phosphorylation of KCC2. We observed aberrant KCC2 phosphorylation and reduced expression, suggesting reduced KCC2 activity. Examining developmental KCC2 changes revealed significant alterations in key phosphorylation residues and decreased expression between postnatal days 14 and 21. Treatment with the KCC2 activator (OV350) between p10 and p21 saw a significant reduction in infantile spasms compared to vehicle-treated Cdkl5 knockout mice. Remarkably, when these mice were adults, the mice that received OV350 as pups had reduced seizure susceptibility and their cognitive and behavioral deficits were alleviated. These findings indicate that enhancing KCC2 function during a critical developmental window may be a promising therapeutic strategy for CDD and other developmental and epileptic encephalopathies.
Similar content being viewed by others
Introduction
Cyclin-dependent kinase-like 5 (Cdkl5) deficiency disorder (CDD) is a severe type of neurodevelopmental and epileptic encephalopathy (DEE) that affects 1 in 40,000–75,000 live births, and is one of the most common genetic forms of infantile epilepsy1. CDD is characterized by early onset treatment-resistant seizures beginning in early childhood, accompanied by severe neurodevelopment impairments and subsequent cognitive and social deficits throughout life2,3. Treatment-resistant seizures substantially raise the risk of injury or death4, highlighting the importance of understanding the underlying mechanisms and developing effective treatments. Current pharmacological therapies for CDD primarily focus on antiseizure medications (ASMs), such as the GABAergic enhancing neuroactive steroid, ganaxolone5. However, ASMs (especially when given as polytherapy) are associated with side effects such as somnolence, which limit the quality of life6,7. Unfortunately, there is no effective treatment for the cognitive or behavioral impairments associated with this disorder.
Most of the neuropathological changes and behavioral deficits that occur in human patients with CDD can be recapitulated in constitutive Cdkl5 knockout (KO) mouse models. Like human patients, loss of functional CDKL5 results in increased anxiety, depression and fear-related behavior, along with impairment in the acquisition and retention of spatial memory8,9,10. These Cdkl5 KO mice also show abnormal adult neurogenesis, reduced dendritic arborization and disruption in the organization of excitatory and inhibitory synapses11,12. Recently, a study showed a reduction in the protein levels of phosphorylated K+/Cl− cotransporter 2 (KCC2) in the cortex of neonatal Cdkl5 KO pups, which leads to the development of spontaneous recurrent seizures13.
CDKL5 is a serine/threonine protein kinase and is known to be essential for normal brain development. Postnatal developmental expression of CDKL5 has a similar timeline for the developmental activity of the potassium chloride cotransporter (KCC2). During this initial postnatal period, dramatic changes occur as major neuronal circuits are formed that lay down the initial pathways important for memory consolidation and sensory and behavioral processing14. KCC2 is the principal Cl−-extrusion mechanism used by developing and mature neurons in the central nervous system15. Its activity is a prerequisite for the efficacy of fast synaptic inhibition mediated by g-aminobutyric acid type A receptors (GABAAR), which are Cl−-permeable ligand-gated ion channels. The postnatal development of canonical hyperpolarizing GABAAR currents reflects the progressive decrease of intraneuronal Cl− levels, caused by the upregulation of KCC2 expression and subsequent activity16. The developmental appearance of hyperpolarizing GABAAR currents is determined by the phosphorylation status of KCC2, a process that facilitates its membrane trafficking and activity16,17. Deficits in KCC2 expression levels and activity have been detailed in patient and animal models of epilepsy18,19. Furthermore, we, and others, have demonstrated that KCC2 loss of function is strongly correlated with cognitive impairment (in Fragile X syndrome and Rett syndrome) and the development of pharmaco-resistant seizures that are insensitive to GABAAR-positive allosteric modulators such as benzodiazepines20,21,22,23,24.
The current understanding of whether KCC2 expression, phosphorylation and activity change during development in Cdkl5 KO pups remains unclear. Furthermore, it is crucial to investigate whether enhancing KCC2 activity during postnatal development, when neural circuits are maturing and the transition from excitation to inhibition occurs in immature neurons, could potentially normalize behavioral and cognitive deficits in Cdkl5 KO mice. To address this issue, we have developed a novel small-molecule activator, OV350, which potentiates KCC2 activity and effectively terminates pharmaco-resistant seizures in wild-type (WT) mice25. Here, we demonstrated the effects of ablating CDKL5 on the phosphorylation of KCC2 and its impact on the development of pharmaco-resistant seizures and cognitive and behavioral deficits. We observed that the pharmacological activation of KCC2 during postnatal development is an important time period for an intervention to improve adult sociability and cognition. We also showed that KCC2 activity during postnatal development reduces adult baseline electroencephalography (EEG) power and restores the ability of diazepam (DZ) to terminate intractable status epilepticus (SE) in Cdkl5 KO mice.
Materials and methods
Study design
This study tested whether early intervention with the KCC2 activator, OV350, during development would correct the phenotypes seen in adult Cdkl5 KO mice. We selected three phenotypes to test, which correspond to clinical phenotypes observed in CDD patients, namely, seizure/epileptic spasms, spatial memory impairments and behavioral deficiencies. All experiments included mice of both sexes and were conducted across multiple litters, with litters randomized to receive either vehicle (veh) or OV350 treatment. See Table 1 for the key resources.
Animals
The TUFTS University Institutional Animal Care and Use Committee approved all animal use. The animals were housed in temperature-controlled rooms on a 12-h day/night cycle. We purchased the Cdkl5 KO mice from The Jackson Laboratory (strain 021967) and used a mix of male and female Cdkl5 KO mice for our experiments. Our study design is focused on evaluating the therapeutic efficacy of KCC2 activation during a specific developmental window (postnatal days (p)10–21) within the Cdkl5−/− genotype. Therefore, we crossed homozygous females and hemizygous males to generate CDD mice (Table 1).
Drug preparation
OV350 was formulated with 6.25% DMSO and 93.75% (v/v) of 50% (w/v) captisol, and the veh was 6.25% DMSO and 93.75% (v/v) of 50% (w/v) captisol. Mice were injected with 50 mg/kg OV350, a dose which has previously been shown to reach a brain concentration of 676 nM within 4 h and is maintained for 8 h25.
Immunoblotting
Sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS–PAGE) was carried out as previously described26,27. Detailed methods are provided in the Supplementary Information.
Plasma membrane isolation
Plasma membranes were isolated as previously described26,27. Briefly, rapidly dissected cortical/hippocampal tissues were collected from seven 8–12-week-old male and female mice for each genotype in a starting buffer28. Detailed methods are provided in the Supplementary Information.
Immunoprecipitation
Protein G Dynabeads (Thermo Fisher) were washed and incubated overnight at 4 °C with KCC2 antibody or nonimmune mouse IgG. The beads were washed and crosslinked with dimethyl pimelimidate dissolved in triethanolamine for 30 min at room temperature. The beads were then eluted using nondenaturing soft elution buffer for BN–PAGE as outlined previously27,29.
Protein analysis by LC–MS/MS
Quantitative label-free proteomic analysis was conducted following the previously described method26,27. Gel bands of interest were excised and cut into 1-mm3 pieces. Subsequently, the peptides were extracted from gel bands, dried and stored at 4 °C before reconstitution in HPLC solvent. The samples were then loaded onto a nano-scale reverse-phase HPLC capillary column. The peptides were detected, isolated and fragmented to produce a tandem mass spectrum of specific fragment ions for each peptide.
Peptide/protein searches
The MS data in its raw form was processed as previously described26,27. Peptide sequences were identified by matching protein or translated nucleotide database sequences with the obtained fragmentation pattern using MSGF+26,27. Detailed methods are provided in the Supplementary Information.
Phosphopeptide proteomic analysis
To measure the impact of CDKL5 removal on global KCC2 phosphorylation, we used proteomics to measure the amount of phosphorylated peptides and unphosphorylated peptides and generate ratios of phosphorylation abundance for each known KCC2 phosphosite, as previously described26,27. Three experimental replicates were performed and a t-test was used to compare the proteomic data between the two genotypes. Detailed methods are provided in the Supplementary Information.
Whole-cell patch-clamp recordings
Whole-cell recordings from prefrontal cortex (PFC) neurons were performed in cortical coronal slices (350 μm) from p14–p21 mice. Recording electrodes (5–6 MΩ resistance) contained the following (in mM): 115 K-gluconate, 30 KCl, 10 HEPES, 1 MgCl2, 2 Na-ATP and 0.4 Na-GTP, pH 7.4. Slices were continuously perfused with oxygenated artificial cerebrospinal fluid-containing kynurenic acid (3 mM) and bumetanide (10 μM). A picospritzer pipette containing muscimol (5 μM), a GABAAR agonist, was used to activate GABAAR-mediated currents in cells held at voltages between −90 and −10 mV. Peak amplitude responses of the muscimol-activated current were plotted for each holding voltage and data were fitted by linear regression analysis. The reversal potential of GABA responses (EGABA) was obtained from the x-intercept value of the fit. Voltages were corrected offline with a liquid junction potential value of 13.2 mV. To measure the contribution of KCC2 activity to EGABA, measurements of EGABA were taken before and after a 5-min exposure to VU0463271 (10 μM), a selective inhibitor of KCC2.
Behavioral spasms
Neonatal pups, aged p10–p21, were removed from their dam and placed in a container on a heating pad for daily sessions lasting 30 min. Behavioral videos were analyzed for spontaneous high-amplitude spastic movements, low-amplitude movements, time spent on their sides and walking. Low-amplitude spasm-like events were categorized as muscle twitches and sudden tail movement between Cdkl5 KO and WT pups. Analyses were conducted blinded to the treatment and gender, and interrater reliability procedures were considered during the study. High-amplitude spastic movements were characterized by rapid extensions and flexions, involving two to four limbs, spine curving and stumbling, following previously criteria established30,31.
EEG surgeries and recording
Adult mice (8–9-weeks old) were subjected to EEG surgeries as previously described32. After the surgery, the mice recovered for 7 days in their home cages before experimentation. On the day of recording, the mice were connected to the pre-amps for recording. First, a 2-h-long baseline recording was obtained and then the mice received a 20 mg/kg kainate (KA) injection (intraperitoneal (i.p.)). Two hours later, the mice were administered with a single dose of 5 mg/kg DZ (i.p.) and were recorded for another hour to assess the effectiveness of postnatal OV350 treatment in adult Cdkl5 KO mice.
EEG analysis
To assess the potential impact of OV350 treatment on baseline EEG power, a 20-min silent period was analyzed, during which no muscular movement was detected based on the EMG channel. Mice received a single dose of 50 mg/kg OV350 or veh control for 12 consecutive days during the postnatal development period from postnatal day 10 to 21. To evaluate the effectiveness of OV350 in restoring the efficacy of DZ, a 40-min EEG signal was analyzed and compared to the veh group.
To identify and score seizures and SE, EEG recordings were examined to determine the onset of the first seizure and instances of SE33. To investigate the effectiveness of OV350 in preventing the onset of DZ-resistant SE, we compared the EEG epochs of 40 min following DZ administration across all three groups of mice.
Behavior
For all behavioral tests, WT mice were administered a veh only, while CDD mice were given injections of either a veh or OV350. Animals were randomly assigned to each group and all behavioral analyses were done while blinded to genotype. Interrater reliability procedures were considered during the study. After each experiment, the equipment was sanitized after each mouse using 70% ethanol, followed by Clidox. Both male and female mice were utilized for all experiments.
Barnes maze assay
Mice aged between 10 and 12 weeks were used for the assay. The maze used was a circular platform with a 1.5 m diameter containing 40 holes along the perimeter, each with a 2.5 cm diameter. An escape tunnel was positioned under one of these holes. Mice used natural spatial cues in the room to locate the escape hole under the maze. The test procedure involved placing the mice in the center of the maze. Afterward, the cage was lifted and the mice were given 3 min to explore the maze and find the escape hole. This test was repeated thrice daily with a 30-min interval between trials. The average duration for each day was considered for analysis. This protocol was followed for four consecutive days.
To assess short-term memory, the escape hole was removed on the fifth day and the mice were given 5 min to explore the arena. The time spent at each hole was measured and put into 45° bins, with each containing five holes. These bins were organized around the perimeter of the maze in a clockwise direction, with 0° representing the goal hole and the adjacent holes. The same assessment was repeated on day 12 after a week of no exposure to the maze for long-term memory. An overhead camera and Ethovision software were used to track the time spent in each area of the arena.
Three-chamber social interaction assay
At 13 weeks of age, mice were tested in a three-chamber setup, with each chamber measuring 40 cm × 40 cm. The test mice were allowed to explore the arena for 5 min for habituation. After this, an unfamiliar male or female mouse (8 weeks old) was placed under one of the cages and a dummy mouse was placed under the second cage, and the test mice were allowed to explore the arena for 10 min. The time spent in the chamber with the familiar versus the dummy mouse was also calculated. An overhead camera and Ethovision software were used to detect time spent in each region of the arena.
Statistical analysis
All data are presented as the mean ± s.e.m. The Shapiro–Wilk test was performed to find the normal distribution of datasets. Biochemistry data were analyzed using the Mann–Whitney test or t-test. Electrophysiology data were analyzed using a one-way analysis of variance (ANOVA) followed by Tukey’s multiple comparison. Infantile spasm data were analyzed using the Mann–Whitney test or t-test. Baseline EEG data were analyzed using the Mann–Whitney test or t-test. Seizure data were analyzed using a two-way ANOVA followed by Tukey’s multiple comparison. Behavioral data were analyzed using a two-way ANOVA to compare genotypes followed by Tukey’s multiple comparison, and a repeated measures ANOVA followed by Dunnett’s multiple comparison was used to analyze data obtained from individual mice across different trials. The data are presented in Supplementary Tables 1 and2 (including P values, test used, means, s.e.m. and the number of animals). P values <0.05 are considered statistically significant.
Results
CDKL5 loss is associated with aberrant KCC2 phosphorylation and expression
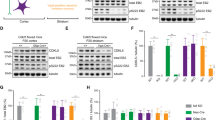
To confirm the association between CDKL5 and KCC2, we isolated the cortex and hippocampus and made purified plasma membrane preparations that were subjected to immunoprecipitation with KCC2 or control IgG antibodies. Precipitated material was immunoblotted with the CDKL5 antibody. In WT mice, CDKL5 is associated with KCC2 and is part of the KCC2 complex. CDKL5 was significantly reduced in the Cdkl5 KO mice (Fig. 1a). Next, we analyzed the phosphorylation status of KCC2 residues in both Cdkl5 KO and WT mice. Global phosphorylation analysis between the two groups was conducted using LC–MS/MS. Spectral searches identified phosphorylated serine (S) and threonine (T), which are crucial sites for post-translational modifications that influence the trafficking, plasma membrane expression and chloride extrusion capabilities of the KCC2 transporter. We estimated phosphorylation levels on these residues by comparing the quantities of phosphorylated versus dephosphorylated peptides. The S940 site, which is known to positively influence KCC2 activity, exhibited significantly lower phosphorylation levels in Cdkl5 KO mice compared to WT mice. By contrast, the T906 and T1007 sites, recognized for negatively regulating KCC2 activity, showed significantly higher phosphorylation levels in Cdkl5 KO mice than in WT mice. Furthermore, we found a notable reduction in phosphorylation on two novel residues, S932 and S1022, in Cdkl5 KO mice compared to WT mice (Fig. 1b). Cortical and hippocampal lysates prepared from WT or Cdkl5 KO mice (8–12 weeks old) were immunoblotted with antibodies against β-actin, CDKL5 and KCC2 and phospho-specific antibodies pS940, pT906 and pT1007. Compared to WT mice, there was a significant reduction in KCC2 expression in Cdkl5 KO mice (Fig. 1c,d). There was a concomitant reduction in the phosphorylation level of KCC2 at the serine 940 site (Fig. 1c,e). By contrast, phosphorylation is increased at T906 (Fig. 1c,f) and T1007 (Fig. 1c,g) sites in Cdkl5 KO mice. These results demonstrate that loss of CDKL5 results in a KCC2 phosphorylation phenotype that displays the hallmarks of an immature phenotype characterized by dysfunctional KCC2 activity.
a Immunoprecipitation of the KCC2 complex in plasma membrane preparations shows a strong association of CDKL5 with KCC2 in the WT mice when blotted with Cdkl5 antibody. CDKL5 is reduced in the KCC2 complex in Cdkl5 KO mice. b Spectra searches were performed on LC–MS/MS data obtained from purified KCC2 to identify phosphorylated residues. The S932, S940 and S1022 residue phosphorylation was significantly reduced in Cdkl5 KO mice compared to the WT mice. However, T906 and T1007 phosphorylation of KCC2 is increased in the Cdkl5 KO mice compared to the WT mice. c Forebrain lysates prepared from the WT or Cdkl5 KO mice were immunoblotted with β-actin, Cdkl5, KCC2 and phospho-specific antibodies against S940 (pS940), T906 (pT906) and T1007 (pT1007). d The graph shows a reduction in KCC2 expression in Cdkl5 KO mice compared to the WT mice. e S940 phosphorylation of KCC2 is reduced in Cdkl5 KO mice compared to the WT mice. f T906 phosphorylation of KCC2 is increased in the Cdkl5 KO mice compared to the WT mice. g KCC2 phosphorylation at T1007 is increased in Cdkl5 KO mice compared to the WT mice. The asterisk indicates the significance of the Cdkl5 KO mouse compared to WT. See Supplementary Table 1 for the statistical analysis.
CDKL5 loss orchestrates the alteration in postnatal regulation of KCC2 expression and phosphorylation
Phosphorylation of KCC2 at residues serine 940 (S940), threonine 1007 (T1007) and threonine 906 (T906) regulates KCC2 function in the adult brain, so we sought to examine the developmental profile of S940, T906 and T1007 phosphorylation. We measured total KCC2 expression and KCC2 S940, T906 and T1007 phosphorylation in whole-brain lysates at p0, p7, p14 and p21 and detected phosphorylation of this residue at each of these time points (Fig. 2a). Total KCC2 expression increased linearly over development until p14, followed by maintenance at this level as the brain neurons further matured by p21 in WT mice, with significantly lower expression at p14 and p21 in Cdkl5 KO pups compared to WT pups (Fig. 2b). We detected a significant decrease in S940 phosphorylation at p21 between WT and Cdkl5 KO pups (Fig. 2c). Phosphorylation of KCC2 at threonine residues 906 and 1007 decreases during development in WT mice, which contributes to the developmental upregulation of KCC2 function. Therefore, we also compared the phosphorylation status at these residues between WT and Cdkl5 KO pups. We observed significant increases in KCC2 phosphorylation at T1007 from p14 onward in Cdkl5 KO pups compared to WT pups (Fig. 2d). We also detected a significant increase in phosphorylation at T906 at p21 in Cdkl5 KO pups compared to the WT pups (Fig. 2e). An increase in phosphorylation in these threonine residues during development in Cdkl5 KO pups suggests a developmental delay in the maturation of KCC2.
a Western blotting was used to assess developmental changes in KCC2 expression and phosphorylation in WT and Cdkl5 KO pups at 0, 7, 14 and 21 postnatal days. Whole-brain lysates prepared from WT or Cdkl5 KO pups were immunoblotted with β-tubulin, CDKL5, KCC2 and phospho-specific antibodies against S940 (pS940), T906 (pT906) and T1007 (pT1007). b KCC2 expression progressively increased from p0 to p14 and stabilized by p21 in WT and Cdkl5 KO pups. However, KCC2 expression from p14 to p21 is significantly reduced in Cdkl5 KO mice compared to WT pups. c Relative to the levels of KCC2 expression, S940 phosphorylation is decreased from p0 to p21 in WT and Cdkl5 KO pups. S940 phosphorylation was significantly reduced at p21 in Cdkl5 KO pups compared to WT pups. d The graph shows a progressive reduction in T1007 phosphorylation over time in WT pups relative to KCC2 expression. Meanwhile, Cdkl5 ablation significantly increased phosphorylation at the T1007 site on KCC2 from p14 to p21 compared to WT pups. e T906 phosphorylation on KCC2 does not change over time from p0 to p21 during postnatal development in WT and Cdkl5 KO mice. However, the phosphorylation is significantly increased at the T906 site on KCC2 at p21 in Cdkl5 KO pups. The asterisk indicates the significance of the Cdkl5 KO mouse compared to WT on a particular day. See Supplementary Table 1 for the statistical analysis.
These results demonstrate that loss of CDKL5 results in a KCC2 phosphorylation phenotype that leads to decreased KCC2 function, which is strongly correlated with excessive neuronal excitation, seizure-like events, increased seizure susceptibility, and cognitive and behavioral impairments in adult mice34,35.
Cdkl5 KO pups show a population of neurons that have depolarized E GABA values
To examine KCC2 activity in neurons from Cdkl5 KO mice compared to WT mice, we employed a whole-cell patch-clamp Cl− loading assay35,36. By imposing an internal 32 mM Cl− load through the patch pipette, the subsequent measurement of EGABA reports the degree to which these neurons were able to extrude a fixed amount of Cl−. Without KCC2 activity, the Nernst equation dictates that neurons should have an EGABA value of −39 mV when loaded with 32 mM [Cl−]i. The WT neurons displayed an EGABA value of −57.3 ± 2.5 mV (n = 7), indicating the presence of a functional Cl− extrusion KCC2. PFC neurons from Cdkl5 KO mice displayed two populations, one that did not display any action potential at the cell’s resting membrane potential, similar to WT, and another population that displayed action potential firing at rest. The former population had an EGABA value of −61.2 ± 2.9 mV (n = 7). The latter had a depolarized EGABA value of −44.2 ± 3.6 mV (n = 4) that was significantly more depolarized compared to WT or the electrically silent Cdkl5 KO group of neurons (Supplementary Fig. 1a,b). Additional experiments were performed to validate the contribution of KCC2 activity to EGABA. We measured EGABA before and after a 5-min exposure to VU0463271 (10 μM), a selective inhibitor of KCC2. The neurons from the Cdkl5 KO mice that displayed an increased excitability had a significantly smaller depolarizing shift in EGABA (4.1 ± 2.5 mV, n = 6) compared to WT (11.1 ± 1.1 mV, n = 11) and the less excitable Cdkl5 KO neurons (14 ± 2 mV, n = 9; Supplementary Fig. 1c). This indicates that the population of neurons with depolarized EGABA has inadequate functional KCC2 to contend with the Cl− load.
Cdkl5 KO pups show intense infantile spasms
Dysfunction or mutations in KCC2 in mouse models and human patients often lead to infantile epileptic spasms37,38,39. Our proteomics data show a significant reduction in the expression of KCC2 and alterations in the phospho-profile of KCC2 during the postnatal developmental period, which may contribute to the infantile spasms in Cdkl5 KO mice.
Therefore, we next examined whether behavioral spasms occur in Cdkl5 KO pups during neonatal development. Since the onset of infantile spasms in humans is usually in the first year of life, we quantified behavioral spasms in Cdkl5 KO and WT littermate pups between p10 and p21. These ages represent a period of significant synapse development and brain growth in the respective species. Infantile spasms were defined as spontaneous high-amplitude, spastic movements, using the criteria developed for assessing infantile spasm-like phenotypes in other rodent models30,31.
The spasms typically occurred in a series and were separated by periods of low- and high-amplitude spasm-like events and behavioral arrest, often with the mouse remaining on its side (Fig. 3a,b). We first compared low-amplitude spasm-like events, such as muscle twitches and tail movement, between Cdkl5 KO and WT pups. The Cdkl5 KO pups exhibited significantly more low-amplitude spasm-like events than WT pups from p10 to p15 (Fig. 3c). No difference in low-amplitude events was observed from p16 to p21 between the two groups of mice. Next, we quantified the quantity of high-amplitude spasm-like events (clusters of rapid, full flexions, extensions, rapid movement of two to four limbs, sudden stumbling and spine curving) between Cdkl5 KO and WT pups. The number of high-amplitude spasms was significantly higher in Cdkl5 KO pups than in WT pups from p10 to p14 (Fig. 3d). No difference in high-amplitude events was observed from p15 to p21 between the two groups of mice. In addition, Cdkl5 KO pups spent more time on their side than WT pups from p10 to p14 (Fig. 3e) but not from p15 to p21. Also, Cdkl5 KO pups spent significantly less time walking during p10–p17 but started to walk at comparable levels to WT from p18 to p21 (Fig. 3f). Overall, Cdkl5 KO pups showed a delay in achieving important developmental milestones. These results suggest that infantile spasms in Cdkl5 KO mice may result from compromised KCC2 activity, potentially leading to improper neuronal circuit formation and contributing to abnormal behavior and developmental delays.
a The experimental timeline. b Representative example of a WT pup and Cdkl5 KO pups experiencing spontaneous high-amplitude spastic movements, including movement of two to all four limbs (1), rapid full flexion (2), extension of limbs (3) and and spine curving (4). c The number of low-amplitude spastic movements was increased in Cdkl5 KO pups compared to WT pups from postnatal day 10–15, based on scoring of the 30-min video recording of pup behavior. d High-amplitude movements were significantly more frequent in Cdkl5 KO pups from p10 to 14 than in WT pups. e Cdkl5 KO pups spent significantly more time on their sides, indicative of an abnormal behavioral state from p10 to 14 compared to WT pups. f Cdkl5 KO pups spent less time walking than WT pups from p10 to 17. g Cdkl5 KO pups treated with OV350 showed a reduced number of low-amplitude spastic movements compared to veh-treated KO pups on postnatal days 11–12. h High-amplitude movements were significantly less in OV350-treated Cdkl5 KO pups from p11 to 13 compared to veh-treated KO pups. i OV350-treated Cdkl5 KO pups spent significantly less time on their sides than veh-treated KO pups from p12 to 14. j Cdkl5 KO pups treated with OV350 spent more time walking than veh-treated KO pups from p12 to 15. The asterisk indicates the significant difference in infantile spasms and motor activities among different groups of mice. See Supplementary Table 1 for the statistical analysis.
Potentiating KCC2 activity during the postnatal developmental period reduces infantile spasms in Cdkl5 KO pups
We have previously demonstrated that enhancing KCC2 activity through genetic animal models or a novel small-molecule activator increases the seizure threshold and terminates ongoing status epilepticus16,25. We further investigated the effect of enhancing KCC2 activity during the critical development phase (p10–21) on infantile spasms. To test this hypothesis, we administered a daily dose of the KCC2 activator (OV350, 50 mg/kg i.p.) to Cdkl5 KO pups from p10 to p21, in comparison to age-matched, veh-treated Cdkl5 KO pups (Fig. 3a). Our observations revealed that OV350-treated Cdkl5 KO pups had significantly fewer low-amplitude spasm-like events than their veh-treated counterparts from p11 to p12 (Fig. 3g). In addition, KCC2 activation with OV350 decreased the occurrence of high-amplitude spasm-like events in Cdkl5 KO pups from p11 to p13 (Fig. 3h). The OV350-treated Cdkl5 KO pups also demonstrated significantly less time spent on their side than veh-treated KO pups from p12 to p14 (Fig. 3i) and more time walking than their veh-treated KO pups from p12 to p15 (Fig. 3j). These findings indicate that enhancing KCC2 activity during development can lessen infantile spasms in Cdkl5 KO pups and potentially support the formation of normal neuronal circuits during this critical time in development, which may improve deficits in neuronal excitability, sociability and spatial memory.
Adult Cdkl5 KO mice exhibit increased baseline EEG power, which is normalized by OV350 treatment during the postnatal developmental period
The postnatal change in KCC2 phosphorylation regulates the developmental GABA switch. Disruption to the timing of the switch (as would be expected in Cdkl5 KO mice with an immature KCC2 phosphorylation signature) will have long-lasting consequences for many developing brain circuits, altering their long-term excitability40,41. Here, we used EEG recordings to measure the baseline EEG signal in adult WT and Cdkl5 KO mice. We further examined the consequences of enhancing KCC2 activity during this critical developmental period on baseline EEG measurements. To address this hypothesis, we administered a daily dose of the KCC2 activator (OV350, 50 mg/kg i.p.) to Cdkl5 KO pups from p10 to p21, compared to veh-treated, age-matched, WT and Cdkl5 KO pups. After 4 weeks, EEG surgeries were performed. In the following week after recovering from the surgeries, a 2-h-long baseline recording was conducted to examine the difference in baseline EEG power between the WT-vehi, Cdkl5 KO-veh and Cdkl5 KO-OV350 mice (Fig. 4a). A 20-min-long EEG epoch recording was compared between all three groups. To quantify the difference, recordings were subjected to fast Fourier transformation (FFT) to convert the EEG signals from the time domain into the frequency domain, generating a power spectral density plot for frequencies between 0 and 100 Hz (Fig. 4b–d). Similar to the human CDD patients42, we observed that the total baseline EEG power of Cdkl5 KO-veh mice was significantly higher than that of WT-veh mice (Fig. 4e,f). To further find that which EEG frequency bands were contributing to an increase in total power, we also assessed the possible differences in the EEG frequency bands (delta, 0–4 Hz; theta, 4–8 Hz; alpha, 8–13 Hz; and beta, 13–30 Hz) and again, similar to the human CDD patients42 we observed a significant increase in power in the delta and theta frequency bands (Fig. 4g). Next, we compared whether KCC2 activation using OV350 during postnatal development reduces baseline EEG power in Cdkl5 KO mice. We observed that the total baseline EEG power of Cdkl5 KO-350 mice was significantly reduced compared to the KO-veh mice (Fig. 4h,i). Again, when we compared the power distribution across OV350 and veh-treated Cdkl5 KO mice, we observed that the Cdkl5 KO mice treated with OV350 while they were infants showed a significant reduction in power across the delta and theta frequency bands compared to the veh-treated Cdkl5 KO mice (Fig. 4j). These results suggest that the Cdkl5 KO mice may be more prone to seizures due to the higher baseline EEG power. Therefore, we compared the susceptibility to seizures and SE between the two groups of mice.
a The line diagram shows the experimental timeline. Two hours of baseline EEG recordings were performed after veh/OV350 (50 mg/kg i.p., arrow) administration during the postnatal development period (p10–p21). b Representative EEG trace and its spectrogram show the power distribution across different frequency bands from a veh-treated WT mouse. c The representative EEG trace is shown above, along with the power spectra from a veh-treated Cdkl5 KO mouse. d The representative EEG trace is shown above, along with the power spectra from an OV350-treated Cdkl5 KO mouse. e Baseline EEG recordings from WT-veh and KO-veh groups of mice were subjected to FFT, and a spectral plot is shown for frequencies between 0 and 100 Hz. The total EEG power was compared between the two groups of mice. Cdkl5 KO-veh mice had a higher baseline EEG power than WT-veh mice. f The graph shows a percentage increase in EEG power. g The EEG power is significantly increased across delta and theta frequency bands in Cdkl5 KO-veh mice compared to the WT-veh mice. h Mice treated with OV350 significantly reduced the EEG power compared to the veh-treated Cdkl5 KO mice. i The graph shows a percentage decrease in EEG power. j The EEG power is significantly decreased across the delta and theta frequency bands in Cdkl5 KO-350 mice compared to the KO-veh mice. The asterisk indicates the significant difference in EEG power between different groups of mice. See Supplementary Table 1 for the statistical analysis.
Cdkl5 KO mice are more susceptible to KA-induced seizures and status epilepticus
Since KCC2 phosphorylation influences seizure severity, Cdkl5 KO mice with altered KCC2 phosphorylation would be predicted to have increased duration of epileptiform activity, a decreased latency to the first epileptiform event and a decreased latency to onset of SE compared to WT mice. To address this hypothesis, we compared the susceptibility of KA-induced seizures in WT and Cdkl5 KO mice. A single dose of 20 mg/kg KA was administered, with recordings continuing for 2 h. After that, all three groups of mice received a saturating concentration of DZ (5 mg/kg i.p.), after which recordings were extended for an additional 1 h (Fig. 5a–d).
a The experimental timeline shows that WT pups were administered with the veh and Cdkl5 KO pups received either veh or OV350 (50 mg/kg i.p.) from p10 to p21 during the postnatal development period. Four weeks later, these mice underwent EEG surgeries. One week following the surgeries, on the day of the experiment, a 2 h baseline recording was conducted. Next, mice received a single injection of KA (20 mg/kg i.p., first arrow) to induce seizures and status epilepticus. Two hours after KA injection, mice were dosed (i.p.) with 5 mg/kg DZ, and EEG recordings were extended for 1 h. b Representative EEG trace and its spectrogram show power distribution across different frequency bands from a veh-treated WT mouse. c Representative EEG trace and spectrogram show power distribution across different frequency bands from a veh-treated Cdkl5 KO mouse. d A representative EEG trace is shown above in the power spectra of an OV350-treated Cdkl5 KO mouse. e The graph shows increased latency to the first seizure in Cdkl5 KO mice. The asterisk indicates the significant difference in latency to the first seizure among different groups of mice. f OV350-treated Cdkl5 KO mice took more time to get into KA-induced SE. The asterisk indicates the significant difference in latency to the SE among different groups of mice. g Veh-treated WT mice spent less time in epileptic events than the veh-treated Cdkl5 KO mice, and OV350 treatment did not reduce time spent in epileptic activity in the Cdkl5 KO mice. The asterisk indicates the significant difference in time spent in epileptic activity among different groups of mice. h EEG recordings of the epileptic activity from the veh- and OV350-treated mice were subjected to FFT, and a spectral plot is shown for frequencies between 0 and 100 Hz. The total EEG power was compared between all three groups of mice. OV350 treatment did not alter the EEG power in Cdkl5 KO mice. i EEG recordings of the post-DZ treatment period from all three groups of mice were subjected to FFT, and a spectral plot is shown for frequencies between 0 and 100 Hz. The total EEG power was compared. Mice treated with OV350 had significantly reduced EEG power compared to the veh-treated mice. The asterisk indicates the significant difference in change in post-DZ power among different groups of mice. See Supplementary Table 1 for the statistical analysis.
This model was chosen because of the similarities with patients having drug-resistant seizures as KA-induced seizures become refractory to benzodiazepines within minutes. The Cdkl5 KO mice took less time to have their first seizure (Fig. 5e) and developed SE faster than WT mice (Fig. 5f). In addition, the Cdkl5 KO mice spent more time in epileptic activity than the WT mice (Fig. 5g). However, we did not observe a difference in the EEG power of total epileptic activity (Fig. 5h). In both WT and Cdkl5 KO mice, the benzodiazepine, DZ, failed to suppress SE, as demonstrated by the lack of change in EEG power before and after DZ treatment (Fig. 5i).
Targeting KCC2 activity in infant Cdkl5 KO mice reduces seizure susceptibility and restores DZ efficacy in adult mice
We next explored if these seizure susceptibility issues observed in adult Cdkl5 KO mice could be negated by enhancing KCC2 function during development. Adult Cdkl5 KO mice that received OV350 treatment between p10 and p21 had a significantly increased latency to the first KA-induced seizure compared to veh-treated Cdkl5 KO mice (Fig. 5e). These mice also took a significantly longer time to develop status epilepticus than the veh-treated Cdkl5 KO mice (Fig. 5f). However, KCC2 activation did not reduce the time spent in epileptic activity and epileptic power in Cdkl5 KO mice (Fig. 5g,h). Consistent with previously published studies using the KA model, DZ did not modify EEG power in veh-treated mice. By contrast, in Cdkl5 KO mice treated with OV350 (50 mg/kg i.p.) between p10 and p21, DZ significantly reduced EEG power in adult mice (Fig. 5i).
OV350 treatment during postnatal development improves sociability in adult Cdkl5 KO mice
The Cdkl5 KO mice exhibit autistic-like behavioral abnormalities and perform poorly in social interaction tasks. Deficits in KCC2 expression and phosphorylation also promote autism-like behavior in mice14,43. However, increasing KCC2 activity during development improves sociability in mice16. Therefore, we investigated the impact of enhancing KCC2 activity using OV350 (50 mg/kg i.p.) during development (p10–p21) on social behavior in adult Cdkl5 KO mice (10–11 weeks) (Fig. 6a). We hypothesized that the abnormal social behavior in Cdkl5 KO mice is linked to altered KCC2 phosphorylation and reduced KCC2 activity, and potentiating KCC2 activity during development would rescue sociability in Cdkl5 KO mice. Sociability was assessed using a three-chamber social interaction test (Fig. 6b). First, sociability was examined by measuring time spent interacting with an unfamiliar (stranger) mouse. Consistent with the literature, Cdkl5 KO-veh mice spent significantly less time interacting with the stranger mouse than the WT-veh mice did, indicating that the Cdkl5-deficient mice have a reduced motivation for social interaction. However, the OV350-treated Cdkl5 KO mice spent more time interacting with the stranger mouse compared to the time Cdkl5 KO-veh mice spent, suggesting that potentiating KCC2 activity during development alleviates deficits in sociability in Cdkl5-deficient mice (Fig. 6c). Next, we measured how much time mice spend with a live mouse compared to a dummy mouse. Cdkl5 KO-veh-treated mice spent almost the same time with live and dummy mice. However, Cdkl5 KO-OV350 mice, like WT mice, spent more time with a live mouse (Fig. 6d). This finding suggests that KCC2 activation during development in Cdkl5 KO mice increases their preference for sociability as adult mice.
a The experimental timeline. The WT pups were administered a single dose of veh daily from p10 to p21. The Cdkl5 KO pups received a single dose of either vehor OV350 (50 mg/kg i.p.) daily from p10 to p21. We used a three-chamber social interaction assay to assess sociability and preference for social familiarity in the Cdkl5 KO mice treated with or without OV350. b A diagram of the sociability assay. Mice were allowed to explore either an unfamiliar mouse or a dummy mouse. c WT mice spent more time interacting with the stranger mouse than veh-treated Cdkl5 KO mice, and OV350-treated Cdkl5 KO mice spent more time interacting with the stranger mouse than veh-treated mice. The asterisk indicates the significance of the time spent with the stranger mouse. d The graph shows that both WT and OV350-treated Cdkl5 KO mice preferred interacting with the mouse versus the dummy mouse. The asterisk indicates the significance of the time spent with the dummy and the stranger mouse. See Supplementary Table 1 for the statistical analysis.
Administering OV350 during postnatal development improves spatial learning in Cdkl5 KO mice
Intellectual disabilities are a core feature of CDD and compromised KCC2 phosphorylation also impacts learning and memory. We performed a Barnes maze assay to examine spatial learning and memory in the Cdkl5 KO mice. We treated Cdkl5 KO pups with OV350 (50 mg/kg i.p.) or veh during development (p10–p21), and WT mice received veh injections only. We then examined the beneficial effects of increasing KCC2 activity during development on spatial memory in adult Cdkl5 KO mice (8–10 weeks old) (Fig. 7a,b). To assess the impact of treatment on learning, we recorded the latencies to enter the escape hole over 4 days of learning. Cdkl5 KO-veh mice performed poorly in escape latencies compared to the WT mice over the 4-day learning period, while the OV350-treated mice performed better than the veh-treated Cdkl5 KO mice but not as well as the WT mice (Supplementary Fig. 2). The WT mice showed a significant improvement over their day one escape latencies by day 2 (Fig. 7c). The Cdkl5 KO-veh showed an improvement in escape latencies by training day 4 (Fig. 7d). By contrast, the Cdkl5 KO-350 mice showed a significant improvement in escape latencies by training day 3 (Fig. 7e). This suggests that the rate of spatial learning was mildly improved in the OV350-treated Cdkl5 KO mice.
a The experimental timeline. The Cdkl5 KO pups received a single dose of either veh or OV350 (50 mg/kg i.p.) daily from p10 to p21. WT pups were administered a single dose of veh daily from p10 to p21. We used a Barnes maze assay to assess the rate of spatial learning in the WT-veh and Cdkl5 KO mice treated with or without OV350. b A cartoon illustration of the Barnes maze. c Latency to enter the goal hole was measured on days 1–4; the learning rate (day when there is a significant reduction in latency to goal compared to day 1) was comparable in WT mice. The asterisk indicates the significant difference in latency to the escape hole with reference to day 1. d The learning rate was comparable in veh-treated Cdkl5 KO mice from day 4. The asterisk indicates the significant difference in latency to the escape hole with reference to day 1. e The learning rate was comparable in OV350-treated Cdkl5 KO mice from day 3. The asterisk indicates the significant difference in latency to the escape hole with reference to day 1. f Spatial memory was assessed using a Barnes maze assay. After 4 days of training, the time spent at each hole was measured on day 5 and day 12 upon removal of the escape tunnel and data were binned into 45° groups. WT-veh mice spent more time in the goal area on day 5. Veh-treated Cdkl5 KO mice did not perform as well as the WT mice. OV350-treated (50 mg/kg i.p.) Cdkl5 KO mice performed considerably better. WT-veh and OV 350-treated Cdkl5 KO mice spent more time at the goal zone than the veh-treated Cdkl5 KO mice. g WT mice show enhanced specificity of the spatial memory on day 12 compared to the veh- and OV350-treated Cdkl5 KO mice. The WT mice spent more time at the goal than the other two groups of mice. *Indicates significant difference to time spent in the 0° region. See Supplementary Table 1 for the statistical analysis.
Potentiating KCC2 activity during postnatal development impacts spatial memory in Cdkl5 KO mice
Memory assessments were conducted over increasing periods after the learning portion of the Barnes maze assay. We assessed short-term and long-term memory on day 5 and day 12, respectively. To examine memory retention, we removed the escape chamber to measure the time spent in the space where the escape hole was initially located. Time spent at the escape hole differed between WT and Cdkl5 KO-veh mice on day 5, suggesting that the short-term memory was significantly impaired in the Cdkl5 KO-veh mice. Interestingly, OV350-treated Cdkl5 KO mice spent more time investigating the escape hole region than other regions in the maze on day 5 (Fig. 7f). By day 12, the Cdkl5 KO-veh mice could not differentiate between the escape and nonescape regions, but the WT mice showed long-term memory retention and spent more time in the escape hole region. While the OV350-treated Cdkl5 KO mice demonstrated a preference for the escape hole region compared to the nonescape hole region, they did not spend a significant time at the escape hole (Fig. 7g), suggesting that increasing KCC2 function during development improves short-term spatial memory retention as adults but has limited impact on long-term memory.
Discussion
Our findings suggest that direct activation of KCC2 during postnatal development in Cdkl5 KO mice normalizes baseline EEG power and enhances the effectiveness of DZ in terminating drug-induced SE. In addition, we observed that KCC2 activation during development alleviates sociability and cognitive deficits in Cdkl5 KO mice.
KCC2 functions by extruding intracellular chloride anions and is essential for the ontogenetic switch of GABAA-mediated responses from depolarizing to hyperpolarizing14. The expression and function of KCC2 play a crucial role in regulating neuronal excitability, and disruptions in KCC2 have been associated with the onset of infantile epilepsy44. Our findings indicate a significant decrease in KCC2 expression during postnatal development, as well as in adult Cdkl5 KO mice. This reduction in KCC2 expression may result from the activation of the BDNF–TrkB pathway in the Cdkl5 KO mice45. Notably, BDNF levels are elevated in Cdkl5 KO mice, which is known to negatively impact KCC2 expression46. It is well established that phosphorylation of KCC2 at the serine 940 site (pS940) is critical for stabilizing KCC2 on the neuronal membrane surface, enabling the extrusion of Cl− and maintaining KCC2 activity17. Consistent with an earlier report13, we observed a significant reduction in phosphorylation at the S940 site on KCC2 in the whole-brain and forebrain lysates from Cdkl5 KO mice. In addition, we noted a significant increase in phosphorylation at the threonine 1007 and 906 sites on KCC2 (pKCC2–T1007), which inhibits its transport function35. These findings are in line with the literature, indicating that the serine and threonine kinomes are altered in Cdkl5 KO mice10. This alteration may result from the inactivation of the mTOR signaling pathway10, which is essential for the phosphorylation of KCC247. Phosphorylation at these sites is necessary for the switch of GABAA-mediated excitation to inhibition during the second week of postnatal development. Our findings of altered phosphorylation suggest a potential delay in this critical developmental switch, which may contribute to hyperexcitability and infantile seizures in both human patients and animal models of CDD.
In the PFC of Cdkl5 KO mice, two populations of neurons were found, one that had an EGABA comparable to neurons from WT mice and a second population that had depolarized EGABA that was close to the predicted EGABA value for the Cl− loading conditions of the experiment, indicating that there was no active Cl− exclusion by KCC2 in that particular population. Owing to the limited circuit activity in ex vivo brain slices, the demand on KCC2 is low, hence the comparable EGABA values of WT and some Cdkl5 KO neurons. We have previously described in KCC2 S940A mice that KCC2 distribution and levels of Cl– extrusion were comparable to WT34. It was only when the brain slice was challenged with glutamate that KCC2 activity in S940A mice was unable to contend with the subsequent rise in [Cl−]i resulting from increased circuit and neuronal activity. Similarly, we predict that due to the KCC2 phosphorylation changes observed in Cdkl5 KO mice, there will be an increase in neuronal activity (particularly an increase in GABAergic interneuron activity), and KCC2 in Cdkl5 KO neurons will not be able to react to the rise in [Cl−]i. We have demonstrated the occurrence of epileptic-like high-amplitude spasms in Cdkl5 KO mice between the ages p10 and p14, which resolved thereafter. EGABA values were collected from p14–p21 mice; future studies will examine EGABA in younger mice to explore the possibility that a greater proportion of neurons in the age range of high-amplitude spasms (p10–p14) will have more depolarized EGABA values.
Neuronal and circuit hyperexcitability has been noted in EEG recordings from patients with CDD42 and EEG and brain slice recordings from conditional Cdkl5 knockout mice48, as well as multielectrode recordings of neurons from organoids from patients with CDD49. To investigate this phenomenon, we compared baseline EEG power in Cdkl5 KO mice to that of WT mice. Our analysis revealed a significant increase in baseline EEG power among the Cdkl5 KO mice. This is in line with EEG studies conducted on human patients42, where we observed elevated EEG power across the delta and theta frequency bands. These findings suggest that quantitative EEG measurements may serve as reliable biomarkers for diagnosing CDD and for the effective development of new treatments. Furthermore, we found that administering a KCC2 activator during postnatal development is sufficient to normalize EEG power levels in adult Cdkl5 KO mice. Currently, there is a lack of validated biomarkers to assess brain function and clinical severity in individuals with CDD, which complicates the objective evaluation of emerging treatments. Therefore, we propose the use of quantitative EEG parameters as objective measures of brain function and disease severity in future clinical trials for CDD.
In rodent studies, SE induced by KA is resistant to termination by DZ, modeling patients experiencing drug-resistant seizures32,50,51,52. Our research indicates that Cdkl5 KO mice exhibit a shorter time to first seizure and develop DZ-resistant SE more quickly than WT mice. Furthermore, the Cdkl5 KO mice tend to spend more time engaged in epileptic activity, potentially due to their elevated basal EEG power. Notably, we discovered that repeated administration of OV350 during postnatal development was sufficient in delaying the onset of DZ-resistant seizures and SE in adult Cdkl5 KO mice.
Major autistic-like phenotypes, such as social and communication deficits, are hallmark characteristics of CDD8,24,53,54. In line with existing literature, our findings indicate that Cdkl5 KO mice performed significantly worse in social interaction tasks than their WT counterparts. We observed an alleviation of sociability deficits when we enhanced KCC2 activity in Cdkl5 KO mice during postnatal development using OV350. However, activating KCC2 in adulthood did not lead to a reduction in these social deficits. This underscores the importance of early intervention during postnatal development as this period is crucial for forming neural circuits and the accumulation of KCC2, which facilitates the transition from excitation to inhibition in immature neurons.
Mice that lack CDKL5 exhibit several key characteristics associated with the neurological disorder, including impairments in hippocampal-dependent memory and deficits in motor coordination. While both young adult and middle-aged Cdkl5 KO mice demonstrate significant learning and memory deficits, they do not present motor impairments at an early age. Consistent with these observations, our studies revealed impairments in spatial learning and memory retention among Cdkl5 KO mice. We found that enhancing KCC2 activity during the postnatal development of these Cdkl5 KO mice led to long-term improvements in hippocampus-dependent spatial learning and memory retention. As a result, these mice performed better in learning the Barnes maze task and exhibited improved short-term memory. This aligns with our previous research, which indicated that the constitutive activation of KCC2 through the development of a transgenic mouse line enhanced spatial learning and memory. Recently, a study using CDD mouse models also found that re-expression of Cdkl5, while the mice were young (from 6 weeks), reverses many CDD-related phenotypes. Therefore, our findings reaffirm the notion that early interventions may be the most effective strategy to reduce cognitive deficits in Cdkl5 KO mice.
Several limitations warrant consideration before applying these findings to patient populations. First, we investigated only a single dose of OV350 (50 mg/kg), previously shown to reverse DZ-resistant seizures in adult mice25. Infants may need lower doses to achieve clinical efficacy. Second, we treated for 12 days during development (p10–p21), it remains to be seen whether a shorter time course of treatment would be equally effective. Third, the effect of OV350 on spontaneous seizures remains unknown. Owing to the absence of spontaneous seizures in older mice, we used KA-induced seizures, which respond to acute OV350 treatment25. We have observed infantile spasms in young Cdkl5 KO mice, and a previous study observed seizures recorded by EEG in p12 mice13. Future experiments are needed to determine the developmental stage at which these seizure events start and how long they last; this will determine the length and time course of treatment. Fourth, we focused on studying whether KCC2 activation via OV350 application during a specific developmental window (p10–p21) could reverse cognitive, social and epileptic phenotypes in CDD mice. In future experiments, an important control group would be WT pups treated with OV350 during the same developmental window (p10–p21). Including this control would allow us to assess whether KCC2 activation also influences behavior in adult WT mice. If WT mice showed no changes, it would have strengthened our findings that OV350 specifically rescues disease phenotypes in CDD mice.
Previous studies have implicated impaired chloride homeostasis in Rett syndrome, Fragile X syndrome and other DEEs, which contribute to network hyperexcitability23,55,56. Pharmacological enhancement of the KCC2 gene expression has been shown to have disease-modifying effects in Rett syndrome24. Here, we report that in addition to changes in expression, KCC2 phosphorylation is also altered in Cdkl5 KO mice during development in such a manner as to impair KCC2 activity. It remains to be determined whether similar alterations in KCC2 phosphorylation underlie the impaired transporter activity observed in Rett and Fragile X syndromes. Our data indicate that reduced KCC2 expression and phosphorylation changes in CDD present a critical challenge during the postnatal development period with therapeutic implications that differ from those observed in other DEEs. Specifically, we show that pharmacological activation of KCC2 during a defined postnatal window (p10–p21) not only suppresses epileptic spasms but also rescues cognitive and social behaviors in adult CDD mice. This suggests that early intervention targeting chloride extrusion can recalibrate circuit function before irreversible developmental deficits are established. While these results are promising, extrapolation to human CDD remains speculative and requires further studies. Human patients and the CDD mouse model have differences in developmental timing, as p10 mice are comparable to a 1-year-old child57, as well as variations in drug metabolism, safety profiles and patient variability that must be carefully addressed in future translational efforts. Overall, this study demonstrates that enhancing KCC2 function during infancy is a potential therapy for CDD and other developmental and epileptic encephalopathies.
References
Symonds, J. D. et al. Incidence and phenotypes of childhood-onset genetic epilepsies: a prospective population-based national cohort. Brain 142, 2303–2318 (2019).
D’Mello, S. R. Rett and Rett-related disorders: common mechanisms for shared symptoms? Exp. Biol. Med. 248, 2095–2108 (2023).
Leonard, H. et al. CDKL5 deficiency disorder: clinical features, diagnosis, and management. Lancet Neurol. 21, 563–576 (2022).
Laxer, K. D. et al. The consequences of refractory epilepsy and its treatment. Epilepsy Behav. 37, 59–70 (2014).
Knight, E. M. P. et al. Safety and efficacy of ganaxolone in patients with CDKL5 deficiency disorder: results from the double-blind phase of a randomised, placebo-controlled, phase 3 trial. Lancet Neurol. 21, 417–427 (2022).
Thomas, J. A., Ditchman, N. M., Guidotti Breting, L. & Narayanan, J. Quality of life in people with epilepsy: The associations of anti-seizure medications and biopsychosocial variables. Epilepsy Behav. 152, 109664 (2024).
Wong, K. et al. Caregiver perspective of benefits and side effects of anti-seizure medications in CDKL5 deficiency disorder from an international database. CNS Drugs 38, 719–732 (2024).
Okuda, K. et al. Comprehensive behavioral analysis of the Cdkl5 knockout mice revealed significant enhancement in anxiety- and fear-related behaviors and impairment in both acquisition and long-term retention of spatial reference memory. PLoS ONE 13, e0196587 (2018).
Terzic, B. et al. Temporal manipulation of Cdkl5 reveals essential postdevelopmental functions and reversible CDKL5 deficiency disorder-related deficits. J. Clin. Invest. 131, e143655 (2021).
Wang, I. T. et al. Loss of CDKL5 disrupts kinome profile and event-related potentials leading to autistic-like phenotypes in mice. Proc. Natl Acad. Sci. USA 109, 21516–21521 (2012).
Amendola, E. et al. Mapping pathological phenotypes in a mouse model of CDKL5 disorder. PLoS ONE 9, e91613 (2014).
Pizzo, R. et al. Lack of Cdkl5 disrupts the organization of excitatory and inhibitory synapses and parvalbumin interneurons in the primary visual cortex. Front. Cell Neurosci. 10, 261 (2016).
Liao, W. & Lee, K. Z. CDKL5-mediated developmental tuning of neuronal excitability and concomitant regulation of transcriptome. Hum. Mol. Genet. 32, 3276–3298 (2023).
Virtanen, M. A., Uvarov, P., Mavrovic, M., Poncer, J. C. & Kaila, K. The multifaceted roles of KCC2 in cortical development. Trends Neurosci. 44, 378–392 (2021).
Moore, Y. E., Kelley, M. R., Brandon, N. J., Deeb, T. Z. & Moss, S. J. Seizing control of KCC2: a new therapeutic target for epilepsy. Trends Neurosci. 40, 555–571 (2017).
Moore, Y. E. et al. Developmental regulation of KCC2 phosphorylation has long-term impacts on cognitive function. Front. Mol. Neurosci. 12, 173 (2019).
Lee, H. H. et al. Direct protein kinase C-dependent phosphorylation regulates the cell surface stability and activity of the potassium chloride cotransporter KCC2. J. Biol. Chem. 282, 29777–29784 (2007).
Kelley, M. R. et al. Compromising KCC2 transporter activity enhances the development of continuous seizure activity. Neuropharmacology 108, 103–110 (2016).
Sivakumaran, S. et al. Selective inhibition of KCC2 leads to hyperexcitability and epileptiform discharges in hippocampal slices and in vivo. J. Neurosci. 35, 8291–8296 (2015).
Deeb, T. Z., Maguire, J. & Moss, S. J. Possible alterations in GABAA receptor signaling that underlie benzodiazepine-resistant seizures. Epilepsia 53, 79–88 (2012).
Deshpande, L. S. et al. Development of pharmacoresistance to benzodiazepines but not cannabinoids in the hippocampal neuronal culture model of status epilepticus. Exp. Neurol. 204, 705–713 (2007).
Duarte, S. T. et al. Abnormal expression of cerebrospinal fluid cation chloride cotransporters in patients with Rett syndrome. PLoS ONE 8, e68851 (2013).
He, Q., Nomura, T., Xu, J. & Contractor, A. The developmental switch in GABA polarity is delayed in fragile X mice. J. Neurosci. 34, 446–450 (2014).
Tang, X. et al. Pharmacological enhancement of KCC2 gene expression exerts therapeutic effects on human Rett syndrome neurons and Mecp2 mutant mice. Sci. Transl. Med. 11, eaau0164 (2019).
Jarvis, R. et al. Direct activation of KCC2 arrests benzodiazepine refractory status epilepticus and limits the subsequent neuronal injury in mice. Cell Rep. Med. 4, 100957 (2023).
Choi, C. et al. Analyzing the mechanisms that facilitate the subtype-specific assembly of γ-aminobutyric acid type A receptors. Front. Mol. Neurosci. 15, 1017404 (2022).
Smalley, J. L. et al. Isolation and Characterization of multi-protein complexes enriched in the K–Cl co-transporter 2 from brain plasma membranes. Front. Mol. Neurosci. 13, 563091 (2020).
Smalley, J. L. et al. Spectrin-β 2 facilitates the selective accumulation of GABAA receptors at somatodendritic synapses. Commun. Biol. 6, 11 (2023).
Antrobus, R. & Borner, G. H. H. Improved elution conditions for native co-immunoprecipitation. PLoS ONE 6, e18218 (2011).
Pirone, A. et al. APC conditional knock-out mouse is a model of infantile spasms with elevated neuronal β-catenin levels, neonatal spasms, and chronic seizures. Neurobiol. Dis. 98, 149–157 (2017).
Price, M. G. et al. A triplet repeat expansion genetic mouse model of infantile spasms syndrome, Arx(GCG)10+7, with interneuronopathy, spasms in infancy, persistent seizures, and adult cognitive and behavioral impairment. J. Neurosci. 29, 8752–8763 (2009).
Colmers, P. L. W. et al. Sustained inhibition of GABA-AT by OV329 enhances neuronal inhibition and prevents development of benzodiazepine refractory seizures. eNeuro 11, ENEURO.0137-24.2024 (2024).
Arshad, M. N. & Naegele, J. R. Induction of temporal lobe epilepsy in mice with pilocarpine. Bio Protoc. 10, e3533 (2020).
Silayeva, L. et al. KCC2 activity is critical in limiting the onset and severity of status epilepticus. Proc. Natl Acad. Sci. USA 112, 3523–3528 (2015).
Moore, Y. E., Deeb, T. Z., Chadchankar, H., Brandon, N. J. & Moss, S. J. Potentiating KCC2 activity is sufficient to limit the onset and severity of seizures. Proc. Natl Acad. Sci. USA 115, 10166–10171 (2018).
Cho, N. et al. The brain-specific kinase LMTK3 regulates neuronal excitability by decreasing KCC2-dependent neuronal Cl− extrusion. iScience 27, 109512 (2024).
Baek, H. et al. Altered expression of KCC2 in GABAergic interneuron contributes prenatal stress-induced epileptic spasms in infant rat. Neurochem. Int. 97, 57–64 (2016).
Saitsu, H. et al. Impaired neuronal KCC2 function by biallelic SLC12A5 mutations in migrating focal seizures and severe developmental delay. Sci. Rep. 6, 30072 (2016).
Stödberg, T. et al. Mutations in SLC12A5 in epilepsy of infancy with migrating focal seizures. Nat. Commun. 6, 8038 (2015).
Furukawa, M. et al. Neonatal maternal separation delays the GABA excitatory-to-inhibitory functional switch by inhibiting KCC2 expression. Biochem. Biophys. Res. Commun. 3,1243–1249 (2017).
Reh, R. K. et al. Critical period regulation across multiple timescales. Proc. Natl. Acad. Sci. USA 117, 23242–23251 (2020).
Saby, J. N. et al. Electrophysiological biomarkers of brain function in CDKL5 deficiency disorder. Brain Commun. 4, fcac197 (2022).
Pisella, L. I. et al. Impaired regulation of KCC2 phosphorylation leads to neuronal network dysfunction and neurodevelopmental pathology. Sci. Signal. 12, eaay0300 (2019)
McMoneagle, E. et al. Neuronal K+–Cl− cotransporter KCC2 as a promising drug target for epilepsy treatment. Acta Pharmacol. Sin. 45, 1–22 (2024).
Hu, Z. et al. BDNF–TrkB signaling pathway-mediated microglial activation induces neuronal KCC2 downregulation contributing to dynamic allodynia following spared nerve injury. Mol. Pain 19, 17448069231185439 (2023).
Rivera, C. et al. BDNF-induced TrkB activation down-regulates the K+–Cl− cotransporter KCC2 and impairs neuronal Cl− extrusion. J. Cell Biol. 159, 747–752 (2002).
Huang, X., McMahon, J., Yang, J., Shin, D. & Huang, Y. Rapamycin down-regulates KCC2 expression and increases seizure susceptibility to convulsants in immature rats. Neuroscience 219, 33–47 (2012).
Wang, H. T. et al. CDKL5 deficiency in forebrain glutamatergic neurons results in recurrent spontaneous seizures. Epilepsia 62, 517–528 (2021).
Glass, M. R. et al. Excitatory cortical neurons from CDKL5 deficiency disorder patient-derived organoids show early hyperexcitability not identified in neurogenin2 induced neurons. Neurobiol. Dis. 215, 107093 (2025).
Bertoglio, D. et al. Kainic acid-induced post-status epilepticus models of temporal lobe epilepsy with diverging seizure phenotype and neuropathology. Front. Neurol. 8, 588 (2017).
Drysdale, N. D., Matthews, E., Schuetz, E., Pan, E. & McNamara, J. O. Intravenous kainic acid induces status epilepticus and late onset seizures in mice. Epilepsy Res. 178, 106816 (2021).
Sharma, A. K., Jordan, W. H., Reams, R. Y., Hall, D. G. & Snyder, P. W. Temporal profile of clinical signs and histopathologic changes in an F-344 rat model of kainic acid-induced mesial temporal lobe epilepsy. Toxicol. Pathol. 36, 932–943 (2008).
Jhang, C.-L., Huang, T.-N., Hsueh, Y.-P. & Liao, W. Mice lacking cyclin-dependent kinase-like 5 manifest autistic and ADHD-like behaviors. Hum. Mol. Genet. 26, 3922–3934 (2017).
Jhang, C. L., Lee, H. Y., Chen, J. C. & Liao, W. Dopaminergic loss of cyclin-dependent kinase-like 5 recapitulates methylphenidate-remediable hyperlocomotion in mouse model of CDKL5 deficiency disorder. Hum. Mol. Genet. 29, 2408–2419 (2020).
Contractor, A., Klyachko, V. A. & Portera-Cailliau, C. Altered neuronal and circuit excitability in Fragile X syndrome. Neuron 87, 699–715 (2015).
Tang, X. et al. KCC2 rescues functional deficits in human neurons derived from patients with Rett syndrome. Proc. Natl Acad. Sci. USA 113, 751–756 (2016).
Cottam, N. C. et al. From circuits to lifespan: translating mouse and human timelines with neuroimaging-based tractography. J. Neurosci. 45, e1429242025 (2025).
Schneider, A. C. et al. NIH Image to ImageJ: 25 years of image analysis. Nat. Methods 9, 671–675 (2012).
Acknowledgements
We thank S. Salar, P. Colmers and K. Abiraman for their technical support and for reviewing the manuscript. This work was supported by Ovid Therapeutics, Inc. This study also received support from the National Institutes of Health (NIH), National Institute of Neurological Disorders and Stroke grants NS081986 (to S.J.M.), NS101888 (to S.J.M.), NS103865 (to S.J.M.), NS111338 (to S.J.M.), NS108378 (to P.A.D. and S.J.M.), R21NS126914 (to P.A.D.), R21NS111338 (to P.A.D.) and R21NS111064 (to P.A.D.) and NIH National Institute of Mental Health grant MH097446 (to P.A.D.and S.J.M.).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
S.J.M. serves as a consultant for AstraZeneca, Ovid Therapeutics and Sage Therapeutics, relationships that are regulated by Tufts University. S.F.J.N. and S.J.M. hold equity in Ovid Therapeutics. T.N. and Z.Z. are employees of Ovid Therapeutics and hold equity. The other authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Arshad, M.N., Bope, C., Cho, N. et al. KCC2 activation during postnatal development alleviates long-term deficits in CDKL5-deficient mice. Exp Mol Med 58, 591–604 (2026). https://doi.org/10.1038/s12276-026-01670-x
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s12276-026-01670-x