Abstract
Background
The obesity paradox, where higher body mass index (BMI) is associated with improved survival, is observed in sepsis and cancer individually. However, its effect in patients with both conditions is unclear. The objective of this study is to evaluate the obesity paradox in intensive care unit (ICU) patients with sepsis and cancer and examine whether BMI’s impact on mortality varies across patient phenotypes.
Methods
Data from the Medical Information Mart for Intensive Care IV database were analyzed. Patients were categorized by BMI into underweight (<18.5 kg/m2, n = 173), normal weight (18.5–24.9 kg/m2, n = 1283), overweight (25–29.9 kg/m2, n = 1498), and obesity (≥30 kg/m2, n = 960). The primary outcome was 28-day mortality, with secondary outcomes including 6-month mortality, 1-year mortality, length of ICU stay, continuous renal replacement therapy usage, and invasive ventilation usage. Multivariable logistic regression and restricted cubic splines were used to explore the BMI-mortality relationship, and unsupervised clustering was performed to identify patient phenotypes.
Results
Among 3914 patients, obesity was associated with lower mortality. Clustering revealed four distinct phenotypes, with the protective effect of obesity being more evident in patients with lower Sequential Organ Failure Assessment (SOFA) scores (Cluster1, Cluster2 and Cluster3).
Conclusions
The obesity paradox is evident in both short-term outcome (28-day mortality) and long-term outcomes (6-month and 1-year mortality) among patients with sepsis and cancer, particularly in those presenting with lower disease severity. These findings highlight the need for personalized treatment approaches in this complex patient population.
Similar content being viewed by others
Background
Sepsis is a critical, life-threatening condition that arises from a dysregulated immune response to infection, often leading to multi-organ failure [1]. Globally, sepsis poses a significant health burden, with an estimated 50 million cases and 11 million deaths annually, accounting for approximately 20% of all global fatalities in 2017 [2]. In parallel, cancer remains a significant public health challenge in the 21st century, responsible for nearly one-sixth of deaths worldwide (16.8%) and contributing to 22.8% of mortality associated with noncommunicable diseases [3].
The coexistence of sepsis and cancer presents unique clinical challenges. Cancer treatments, including chemotherapy, radiation, surgery, and bone marrow transplantation, significantly elevate the risk of sepsis. Historical data from the 1990s indicated that cancer was implicated in 12% of sepsis admissions in the United States [4, 5]. Moreover, sepsis accounts for approximately 5% of all hospitalizations among cancer patients, with an in-hospital mortality rate 2.5 times higher than that of non-cancer-related sepsis [6,7,8]. Nearly 30% of cancer deaths are associated with sepsis [9].
Sepsis and cancer share significant pathophysiological features, primarily due to the host’s compromised ability to manage initial insults, whether from pathogen invasion in sepsis or malignant cell transformation in cancer. Both conditions lead to profound disruptions in immune homeostasis, affecting innate and adaptive immune responses [10]. This intersection raises critical questions regarding their mutual impact on disease progression and patient outcomes. Notably, cancer patients have a tenfold increased risk of developing sepsis compared to non-cancer patients, with substantial variability across different cancer types. This elevated risk is associated with increased mortality, with in-hospital mortality rates for cancer patients with sepsis or septic shock reported at 20% and 40%, respectively [11].
Various factors influence the prognosis of both sepsis and cancer, including age, sex, critical care interventions, comorbidities, and the severity of organ injury [12, 13]. Among these, obesity has recently emerged as a significant yet paradoxical influence on survival outcomes. Traditionally regarded as a risk factor for a wide range of health conditions, such as cardiovascular diseases, a higher body mass index (BMI) has unexpectedly been linked to reduced mortality in diseases like heart failure, coronary heart disease, atrial fibrillation, chronic obstructive pulmonary disease, diabetes, stroke, osteoporosis, and severe infections [14,15,16,17,18,19,20]. This counterintuitive phenomenon, known as the “obesity paradox,” has also been observed in sepsis and cancer [21,22,23,24,25,26,27,28,29]. However, the presence and implications of the obesity paradox in patients with both sepsis and cancer have not been thoroughly investigated.
This study aims to evaluate the presence of the obesity paradox in a substantial dataset of patients with sepsis and concomitant cancer admitted to the intensive care unit (ICU). Recognizing the importance of understanding sepsis heterogeneity for advancing precision medicine and providing more tailored treatments [30], we clustered the patients based on their clinical characteristics to determine whether the impact of BMI on patient mortality is consistent across different naturally occurring phenotypes.
Methods
Data source
This retrospective study utilized data extracted from the Medical Information Mart for Intensive Care IV (MIMIC IV) database [31], which includes follow-up data available for up to 1 year post-hospitalization. The authors, Yu Qi and Tuo Zhang have completed the Collaborative Institutional Training Initiative (CITI) examination and have authorized access to the database.
Patients
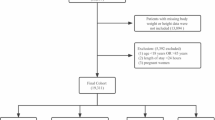
Sepsis cases were initially screened using ICD-9/10 codes, with subsequent confirmation requiring both suspected infection and SOFA score ≥2 according to Sepsis-3 criteria [32]. Exclusion criteria were as follows: (1) not the first ICU admission, (2) aged < 18 years, (3) known or suspected tuberculosis, (4) organ transplant recipients, (5) pregnancy, and (6) BMI < 10 or >100. The patient enrollment flowchart is presented in Fig. 1.
Flowchart of patient enrollment.
BMI groups
Height and weight data were extracted from the MIMIC IV database upon admission to the ICU, then the BMI values was calculated based on these measurements. Patients were categorized into four BMI groups based on their admission BMI: underweight (<18.5 kg/m²), normal (18.5–24.9 kg/m²), overweight (25–29.9 kg/m²), and obesity (≥30 kg/m²), according to World Health Organization (WHO) standards [33].
Data collection
Baseline characteristics were extracted from the MIMIC IV database using Structured Query Language (SQL), including age, sex, race, admission category, BMI, cancer site (top 20 cancer sites based on incidence, extracted using ICD-9 and ICD-10 codes) [34], SOFA score, SAPSII, Glasgow Coma Scale (GCS) score, Elixhauser comorbidities, diabetes, hypertension, time to antimicrobial therapy, and various laboratory tests (lactate, white blood cell count (WBC), monocyte count, neutrophil count, lymphocyte count, neutrophil-to-lymphocyte ratio (NLR), alanine transaminase (ALT), aspartate aminotransferase (AST), total bilirubin, blood urea nitrogen (BUN), creatinine, partial pressure of oxygen (SpO2), peripheral capillary oxygen saturation (PO2), international normalized ratio (INR), platelet count, glucose, sodium, chloride, bicarbonate, albumin), vital signs (heart rate, systolic blood pressure, respiratory rate, temperature), length of ICU stay (LOS), use of invasive ventilation (including tracheostomy), continuous renal replacement therapy (CRRT), and survival time. Median values of laboratory parameters and vital signs within the first 24 h of ICU admission were used in this study.
Primary and secondary outcomes
The primary outcome was 28-day mortality. Secondary outcomes included 6-month mortality, 1-year mortality, LOS, CRRT usage, and invasive ventilation usage.
Missing data imputation
Missing data were imputed using the K-Nearest Neighbors (KNN) method, implemented via the KNNImputer class from the scikit-learn package. Imputation was performed by considering the 10 nearest neighbors based on the Euclidean distance metric. To enable appropriate distance computation for categorical variables, one-hot encoding was applied to race, admission category, and cancer site prior to analysis. The distance metric was calculated based on the following standardized clinical parameters: age, sex, race, admission category, BMI, cancer site, SOFA score, SAPSII, GCS score, Elixhauser comorbidities, diabetes, hypertension, time to antimicrobial therapy, lactate, WBC, monocyte count, neutrophil count, lymphocyte count, NLR, ALT, AST, total bilirubin, BUN, creatinine, SpO2, PO2, INR, platelet count, glucose, sodium, chloride, bicarbonate, albumin, heart rate, systolic blood pressure, respiratory rate and temperature.
Survival analysis
Survival analysis was conducted to assess the association between BMI and mortality at 28 days, 6 months, and 1 year. Kaplan-Meier survival curves were generated and compared using the log-rank test. All analyses were performed using the survival and survminer packages in R.
Multivariable logistic regression
Collinearity among variables in the multivariable logistic regression was assessed by calculating the Variance Inflation Factor (VIF), with all VIFs being less than 10, indicating no significant collinearity. Multivariable logistic regression models were used to investigate the relationship between BMI categories and mortality outcomes at 28 days, 6 months, and 1 year. Variables with significant differences between groups (p < 0.05) were included in the analysis.
Restricted cubic splines
To evaluate the non-linear association between BMI and patient mortality, restricted cubic splines were utilized in multivariable logistic regression models using the rms package. Separate analyses were conducted for 28-day, 6-month, and 1-year mortality outcomes. BMI was modeled with three knots, adjusting for confounders. Odds ratios and 95% confidence intervals were calculated and visualized using ggplot2.
Patient clustering
Patients were clustered based on selected clinical and demographic variables, with the selection of 30 variables: age, sex, race, cancer site, GCS, Elixhauser comorbidities, lactate, WBC, monocyte count, neutrophil count, lymphocyte count, NLR, ALT, AST, total bilirubin, BUN, creatinine, PO2, SpO2, INR, platelet, glucose, sodium, chloride, bicarbonate, albumin, heart rate, respiratory rate, SBP, temperature [35].The clustering process involved a combination of Factor Analysis of Mixed Data (FAMD) and consensus clustering, chosen specifically because the dataset included both categorical and continuous variables. First, the FAMD function from the FactoMineR package was used to perform dimensionality reduction, retaining the first 20 dimensions. A scree plot was generated to visualize the percentage of variance explained by each dimension, and the first 10 principal components were extracted for clustering, as the scree plot indicated an elbow point at the 10th dimension. Consensus clustering was then performed using the ConsensusClusterPlus package with the following key parameters: maximum number of clusters (maxK) set to 10, 100 resampling iterations (reps), 80% of items sampled (pItem), 100% of features sampled (pFeature), Partitioning Around Medoids (PAM) algorithm, and Euclidean distance metric. Finally, Uniform Manifold Approximation and Projection (UMAP) was used for further dimensionality reduction and visualization of clustering results.
Statistical analysis
Categorical variables were summarized as frequencies and percentages, with differences between groups assessed using the chi-square test. Continuous variables, due to their skewed distributions, were presented as medians with interquartile ranges. The Mann-Whitney U-test was used for two-group comparisons, while the Kruskal-Wallis test was employed for multiple group comparisons. All statistical analyses were conducted using R software (version 4.4.1) and Python (version 3.10). A p-value less than 0.05 was considered statistically significant.
Results
Baseline patient characteristics
This study included a total of 3914 patients (Fig. 1). According to the WHO BMI classification [33], patients were categorized into four groups: underweight (n = 173, 4.4%), normal weight (n = 1283, 32.8%), overweight (n = 1498, 38.3%), and obesity (n = 960, 24.5%). Significant differences across BMI categories were noted in a wide array of variables, including age, sex, race, admission category, cancer site distribution, SOFA scores, Elixhauser comorbidities, diabetes, hypertension, and several laboratory parameters such as lactate, lymphocyte count, ALT, AST, total bilirubin, BUN, creatinine, PO2, SpO2, platelet count, glucose, sodium, chloride, bicarbonate, albumin, SBP, and temperature within the first 24 h of ICU admission (Table 1).
Clinical outcomes across different BMI categories
Patients classified as overweight had the shortest LOS and the lowest rates of invasive ventilation usage. In contrast, the use of CRRT increased progressively with higher BMI categories (Fig. 2A). Survival analysis revealed a decrease in 28-day, 6-month, and 1-year mortality rates with increasing BMI (Fig. 2B). Multivariable logistic regression analysis showed that obesity was independently associated with reduced mortality at 28 days, 6 months and 1 year (Fig. 2C and Table S1). Moreover, when BMI was treated as a continuous variable, restricted cubic spline analysis identified inflection points for 28-day, 6-month, and 1-year mortality at 26.3 kg/m² (Fig. 2D–F).
A Comparison of LOS, CRRT usage, and invasive ventilation usage among four BMI groups. B Kaplan–Meier survival curves stratified by the four BMI groups, illustrating survival probabilities over time. C Adjusted odds ratios for 28-day, 6-month, and 1-year mortality across the four BMI groups. D–F Restricted cubic splines showing the association between BMI and 28-day (D), 6-month (E), and 1-year (F) mortality after adjusting for confounders.
Heterogeneity analysis of sepsis patients with cancer
In subsequent analyses, we performed subgroup stratification of patients based on consensus clustering. Given the inclusion of both categorical and continuous variables, FAMD was first applied for dimensionality reduction. The scree plot identified an elbow at the 10th dimension (Fig. S1A), leading to the selection of the first ten dimensions for consensus clustering. The relative change in the area under the consensus cumulative distribution function (CDF) curve, with increasing clusters (k), showed minimal change beyond k = 4, supporting the choice of four clusters for final analysis (Fig. S1B–C). The final clustering results are depicted in Fig. 3A, with UMAP used for additional validation of cluster robustness (Fig. S1D). Significant differences were observed in the distribution of cancer sites across the clusters, with each cluster displaying distinct cancer site profiles. Cluster 1 was marked by a higher prevalence of brain and CNS cancer (11.79%), liver cancer (9.62%), and leukemia (8.11%). Cluster 2 had a predominance of lung cancer (40.70%), breast cancer (7.61%), and non-Hodgkin lymphoma (7.29%). Cluster 3 featured a diverse mix with leukemia (9.08%), non-Hodgkin lymphoma (8.62%), and lung cancer (8.40%). Cluster 4 comprised primarily liver cancer (23.68%), leukemia (14.31%), and pancreas cancer (12.60%) (Fig. 3B). As shown in Fig. 3C, Cluster 1 was associated with lower SOFA scores, while Cluster 4 had the highest SOFA scores. Comparison of clinical indicators across clusters revealed marked differences in parameters such as glucose levels, heart rate, and respiratory rate (Fig. 3D and Table 2). Survival analysis indicated significant variability in 28-day, 6-month, and 1-year mortality among the clusters, with Cluster 4 showing the highest mortality and Cluster 1 the lowest (Fig. 3F). Additionally, Cluster 2 exhibited the longest ICU stay. There was a notable increase in CRRT use from Cluster 1 to Cluster 4, while the utilization of invasive ventilation was lowest in Cluster 2 (Fig. 3G).
A Results of unsupervised consensus k clustering based on 30 variables. B Proportions of different cancer sites within each of the four clusters. C Distribution of the four clusters across categories of SOFA scores. D Heatmap showing the differences in clinical indicators among the four clusters. E Distribution of BMI categories within each patient cluster. F Kaplan–Meier survival curves stratified by the four clusters, illustrating survival probabilities over time. G Comparison of LOS, CRRT usage, and invasive ventilation usage among the four clusters.
Impact of BMI on mortality within clusters
After adjusting for confounders, obesity exhibit a protective effect on 28-day, 6-month and 1-year mortality in clusters (Cluster 1–2) with relatively lower SOFA scores. This protective association was attenuated in high SOFA score clusters (Cluster 3–4), where obesity showed no significant mortality benefit. However, overweight status in Cluster 3 emerged as an independent protective factor for 6-month mortality, persisting after full adjustment (Fig. 4A–D, Table S2–S5).
A–D Adjusted odds ratios for 28-day, 6-month, and 1-year mortality across the four BMI groups in cluster 1 (A), cluster 2 (B), cluster 3 (C), and cluster 4 (D).
Discussion
This study aligns with previous research demonstrating the protective effects of obesity in patients with either sepsis or cancer [21,22,23,24,25,26,27,28,29]. Our findings extend this observation to patients with concurrent sepsis and cancer, suggesting that obesity may confer a survival advantage in this population. By utilizing unsupervised clustering, we identified distinct patient phenotypes based on clinical characteristics and explored the relationship between BMI and prognosis across these subgroups.
In our analysis of clinical characteristics across the four BMI groups, we observed a trend toward increased disease severity with higher BMI levels. Previous studies have noted that patients with obesity often present with elevated baseline lactate levels compared to patients without obesity [36]. Our data corroborate these findings, showing that patients with obesity had higher lactate levels and SBP, which may indicate impaired organ perfusion. Additionally, elevated levels of ALT, AST, BUN, and creatinine in patients with obesity, coupled with more frequent use of CRRT, suggest more significant organ dysfunction in this group.
Consistent with prior studies in sepsis or cancer populations [21,22,23,24,25,26,27,28], our analysis indicates that, after adjusting for confounders, obesity provides a protective effect in patients with both sepsis and cancer. One possible explanation is that patients with obesity have substantial energy reserves, which may help them better endure the catabolic stress associated with sepsis and cancer [37]. This is supported by our finding that patients with obesity had higher albumin levels, indicating better nutritional status. Additionally, there may be a selection bias in care provision, with patients with obesity potentially receiving more intensive and aggressive interventions [28, 38]. Furthermore, the chronic low-grade inflammatory state often present in individuals with obesity might modulate the immune response, leading to a less severe acute inflammatory reaction [39, 40].
Our findings indicate that the protective effect of obesity is more evident in long-term outcomes, specifically 6-month and 1-year mortality. This also could be due to the less intense acute inflammatory response observed in patients with obesity, which could result in reduced lymphocyte apoptosis. Lymphocyte apoptosis is a critical factor in the development of immunosuppression in sepsis patients, often leading to secondary infections and negatively impacting long-term outcomes [30, 41, 42]. Our data show that patients with obesity had significantly higher lymphocyte counts and lower NLR, suggesting reduced lymphocyte apoptosis. This preservation of lymphocytes may contribute to improved long-term survival. Another possible explanation for the enhanced protective effect of obesity on long-term outcomes is the cumulative impact of cancer’s chronic burden, which may amplify the benefits of obesity during the extended recovery phase.
A key strength of this study is the evaluation of BMI’s impact on patient outcomes across different phenotypes. By employing unsupervised consensus clustering based on clinical characteristics, we stratified patients into four distinct clusters. Notably, in the most severely ill group, Cluster 4, we did not observe the obesity paradox. This lack of a protective effect in Cluster 4 could be due to the overwhelming severity of the illness, where the disease’s rapid progression likely outweighs the influence of any single factor, such as BMI [43]. Additionally, the relatively smaller sample size in Cluster 4 may have limited the statistical power to detect subtle protective effects of obesity on outcomes, if they exist.
This study does have limitations. First, as a single-center study, the generalizability of our findings may be limited, as the patient population and clinical practices may not fully reflect those of other centers. Second, we were unable to obtain detailed information on the specific cancer stages of the patients, limiting our ability to stratify analyses based on cancer staging, a critical factor influencing both sepsis severity and overall prognosis.
Conclusion
This study provides a comprehensive evaluation of the obesity paradox in a large cohort of patients with sepsis and concomitant cancer, offering novel insights into the complex interplay between BMI, sepsis, and cancer. Our findings indicate that higher BMI is associated with lower mortality. By employing unsupervised clustering, we identified distinct patient phenotypes, highlighting the variability in outcomes and resource utilization across different subgroups. These results contribute to a deeper understanding of the obesity paradox and underscore the importance of personalized treatment approaches in managing sepsis patients with cancer.
Data availability
The data underlying the present study are available from MIMIC IV database to approved researchers.
Code availability
The code generated in this study is available from the corresponding author upon reasonable request.
References
Cecconi M, Evans L, Levy M, Rhodes A. Sepsis and septic shock. Lancet. 2018;392:75–87.
Rudd KE, Johnson SC, Agesa KM, Shackelford KA, Tsoi D, Kievlan DR, et al. Global, regional, and national sepsis incidence and mortality, 1990–2017: analysis for the Global Burden of Disease Study. Lancet. 2020;395:200–11.
Bray F, Laversanne M, Weiderpass E, Soerjomataram I. The ever-increasing importance of cancer as a leading cause of premature death worldwide. Cancer. 2021;127:3029–30.
Williams MD, Braun LA, Cooper LM, Johnston J, Weiss RV, Qualy RL, et al. Hospitalized cancer patients with severe sepsis: analysis of incidence, mortality, and associated costs of care. Crit Care. 2004;8:R291–8.
Angus DC, Linde-Zwirble WT, Lidicker J, Clermont G, Carcillo J, Pinsky MR. Epidemiology of severe sepsis in the United States: analysis of incidence, outcome, and associated costs of care. Crit Care Med. 2001;29:1303–10.
Martin GS, Mannino DM, Eaton S, Moss M. The epidemiology of sepsis in the United States from 1979 through 2000. N Engl J Med. 2003;348:1546–54.
Torres VB, Azevedo LC, Silva UV, Caruso P, Torelly AP, Silva E, et al. Sepsis-associated outcomes in critically Ill patients with malignancies. Ann Am Thorac Soc. 2015;12:1185–92.
Rosolem MM, Rabello LS, Lisboa T, Caruso P, Costa RT, Leal JV, et al. Critically ill patients with cancer and sepsis: clinical course and prognostic factors. J Crit Care. 2012;27:301–7.
Danai PA, Moss M, Mannino DM, Martin GS. The epidemiology of sepsis in patients with malignancy. Chest. 2006;129:1432–40.
Hotchkiss RS, Moldawer LL. Parallels between cancer and infectious disease. N Engl J Med. 2014;371:380–3.
Lemiale V, Pons S, Mirouse A, Tudesq JJ, Hourmant Y, Mokart D, et al. Sepsis and septic shock in patients with malignancies: A Groupe de Recherche Respiratoire en Réanimation Onco-Hématologique Study. Crit Care Med. 2020;48:822–9.
Antequera A, Lopez-Alcalde J, Stallings E, Muriel A, Fernández Félix B, Del Campo R, et al. Sex as a prognostic factor for mortality in critically ill adults with sepsis: a systematic review and meta-analysis. BMJ Open. 2021;11:e048982.
Fathi M, Markazi-Moghaddam N, Ramezankhani A. A systematic review on risk factors associated with sepsis in patients admitted to intensive care units. Aust Crit Care. 2019;32:155–64.
Katta N, Loethen T, Lavie CJ, Alpert MA. Obesity and coronary heart disease: epidemiology, pathology, and coronary artery imaging. Curr Probl Cardiol. 2021;46:100655.
Alebna PL, Mehta A, Yehya A, daSilva-deAbreu A, Lavie CJ, Carbone S. Update on obesity, the obesity paradox, and obesity management in heart failure. Prog Cardiovasc Dis. 2024;82:34–42.
Tutor AW, Lavie CJ, Kachur S, Milani RV, Ventura HO. Updates on obesity and the obesity paradox in cardiovascular diseases. Prog Cardiovasc Dis. 2023;78:2–10.
Lavie CJ, Coursin DB, Long MT. The Obesity Paradox in Infections and Implications for COVID-19. Mayo Clin Proc. 2021;96:518–20.
Spelta F, Fratta Pasini AM, Cazzoletti L, Ferrari M. Body weight and mortality in COPD: focus on the obesity paradox. Eat Weight Disord. 2018;23:15–22.
Fassio A, Idolazzi L, Rossini M, Gatti D, Adami G, Giollo A, et al. The obesity paradox and osteoporosis. Eat Weight Disord. 2018;23:293–302.
Forlivesi S, Cappellari M, Bonetti B. Obesity paradox and stroke: a narrative review. Eat Weight Disord. 2021;26:417–23.
Oami T, Karasawa S, Shimada T, Nakada TA, Abe T, Ogura H, et al. Association between low body mass index and increased 28-day mortality of severe sepsis in Japanese cohorts. Sci Rep. 2021;11:1615.
Zhou Q, Wang M, Li S, Zhang J, Ma Q, Ding Y, et al. Impact of body mass index on survival of medical patients with sepsis: a prospective cohort study in a university hospital in China. BMJ Open. 2018;8:e021979.
Prescott HC, Chang VW, O’Brien JM Jr, Langa KM, Iwashyna TJ. Obesity and 1-year outcomes in older Americans with severe sepsis. Crit Care Med. 2014;42:1766–74.
Pepper DJ, Demirkale CY, Sun J, Rhee C, Fram D, Eichacker P, et al. Does obesity protect against death in sepsis? a retrospective cohort study of 55,038 adult patients. Crit Care Med. 2019;47:643–50.
Yeo HJ, Kim TH, Jang JH, Jeon K, Oh DK, Park MH, et al. Obesity paradox and functional outcomes in sepsis: a multicenter prospective study. Crit Care Med. 2023;51:742–52.
Sato T, Kudo D, Kushimoto S, Hasegawa M, Ito F, Yamanouchi S, et al. Associations between low body mass index and mortality in patients with sepsis: A retrospective analysis of a cohort study in Japan. PLoS One. 2021;16:e0252955.
Wurzinger B, Dünser MW, Wohlmuth C, Deutinger MC, Ulmer H, Torgersen C, et al. The association between body-mass index and patient outcome in septic shock: a retrospective cohort study. Wien Klin Wochenschr. 2010;122:31–6.
Zhang T, Li X, Meng Z, Fang W, Lian G, Ma W, et al. Obesity and septic patient outcomes: shaping the puzzle through age and sex perspectives. Clin Nutr. 2024;43:1013–20.
Lennon H, Sperrin M, Badrick E, Renehan AG. The obesity paradox in cancer: a review. Curr Oncol Rep. 2016;18:56.
van der Poll T, Shankar-Hari M, Wiersinga WJ. The immunology of sepsis. Immunity. 2021;54:2450–64.
Johnson A, Bulgarelli L, Pollard T, Horng S, Celi LA, Mark R MIMIC-IV (version 2.0) %J PhysioNet, 2022.
Singer M, Deutschman CS, Seymour CW, Shankar-Hari M, Annane D, Bauer M, et al. The Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3). JAMA. 2016;315:801–10.
Physical status: the use and interpretation of anthropometry. Report of a WHO Expert Committee. World Health Organ Tech Rep. Ser. 1995;854:1–452.
Bray F, Laversanne M, Sung H, Ferlay J, Siegel RL, Soerjomataram I, et al. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2024;74:229–63.
Seymour CW, Kennedy JN, Wang S, Chang CH, Elliott CF, Xu Z, et al. Derivation, validation, and potential treatment implications of novel clinical phenotypes for sepsis. JAMA. 2019;321:2003–17.
Jansson PA, Larsson A, Smith U, Lönnroth P. Lactate release from the subcutaneous tissue in lean and obese men. J Clin Invest. 1994;93:240–6.
Robinson MK, Mogensen KM, Casey JD, McKane CK, Moromizato T, Rawn JD, et al. The relationship among obesity, nutritional status, and mortality in the critically ill. Crit Care Med. 2015;43:87–100.
Arabi YM, Dara SI, Tamim HM, Rishu AH, Bouchama A, Khedr MK, et al. Clinical characteristics, sepsis interventions and outcomes in the obese patients with septic shock: an international multicenter cohort study. Crit Care. 2013;17:R72.
Ouchi N, Parker JL, Lugus JJ, Walsh K. Adipokines in inflammation and metabolic disease. Nat Rev Immunol. 2011;11:85–97.
Antonopoulos AS, Tousoulis D. The molecular mechanisms of obesity paradox. Cardiovasc Res. 2017;113:1074–86.
Zhang T, Lian G, Fang W, Tian L, Ma W, Zhang J, et al. Comprehensive single-cell analysis reveals novel anergic antigen-presenting cell subtypes in human sepsis. Front Immunol. 2023;14:1257572.
Pei F, Yao RQ, Ren C, Bahrami S, Billiar TR, Chaudry IH, et al. Expert consensus on the monitoring and treatment of sepsis-induced immunosuppression. Mil Med Res. 2022;9:74.
Jagan N, Morrow LE, Walters RW, Plambeck RW, Wallen TJ, Patel TM, et al. Sepsis and the obesity paradox: size matters in more than one way. Crit Care Med. 2020;48:e776–e782.
Funding
The study was supported by grant from the Shandong Provincial Medical and Health Science and Technology Development Program Project (No. 202105010228) and 2025 Heze Municipal Hospital Science and Technology Project (2025YN014).
Author information
Authors and Affiliations
Contributions
CHH: Methodology, Software, Visualization, Writing—original draft, Writing—review & editing. YQ: Data curation, Methodology, Software, Visualization, Writing—original draft, Writing—review & editing. TZ: Data curation, Methodology, Software, Visualization, Writing—original draft, Writing—review & editing. YL: Supervision, Writing—review & editing. JLW: Supervision, Writing—review & editing. WTL: Supervision, Writing—review & editing. JQL: Data curation, Methodology. XL: Funding acquisition, Supervision, Writing—review & editing.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
The MIMIC-IV database, a publicly available dataset, was utilized in this study. This database has been approved by the Institutional Review Board (IRB) of the Massachusetts Institute of Technology (IRB Protocol #2001P001699), with patient identities de-identified to ensure privacy and confidentiality. Consequently, the requirement for additional ethical approval and informed consent was waived for this manuscript.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Hou, C., Qi, Y., Zhang, T. et al. Evaluating the obesity paradox in patients with sepsis and cancer. Int J Obes 49, 1723–1732 (2025). https://doi.org/10.1038/s41366-025-01805-6
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41366-025-01805-6