Abstract
Taste G protein-coupled receptors (GPCRs), including sweet (TAS1Rs) and bitter (TAS2Rs) taste receptors, are essential for detecting nutrients and avoiding toxins, influencing dietary behavior, metabolism, and overall health. Beyond their role in taste perception, these receptors are widely expressed in extra-gustatory tissues, including the respiratory and gastrointestinal systems, where they regulate innate immune responses, hormone secretion, and energy balance. Dysfunctions or polymorphisms in TAS1Rs and TAS2Rs have been linked to various diseases such as asthma, type 2 diabetes, obesity, dental caries, periodontitis, and certain cancers. The structural features of these receptors, including their ligand-binding domains and signaling pathways, are central to their diverse functions. Recent studies also highlight their potential as therapeutic targets for managing conditions like metabolic syndrome and immune-related disorders. This review provides a detailed examination of the structural and functional dynamics of TAS1Rs and TAS2Rs, emphasizing their roles in disease mechanisms and exploring therapeutic strategies. While challenges remain in structural resolution and functional characterization, advancements in molecular modeling and pharmacological approaches shed light on their clinical potential. Understanding the tissue-specific roles and molecular mechanisms of taste GPCRs can pave the way for innovative treatments targeting these receptors, offering significant promise in addressing a range of health conditions.
Similar content being viewed by others
Introduction
Taste is a vital sensory function for human beings, and it plays a crucial role in determining dietary choices. It refers to the sensation elicited by taste receptors in taste buds upon receiving external stimuli, which subsequently generate taste signals and transmit them to the central nervous system. The taste system plays a crucial role in mammals’ ability to identify nutrient-rich foods for ingestion while avoiding potentially toxic or harmful substances, thereby holding significant importance for human beings and other mammalian species.1,2 Mammals primarily perceive tastes as sweet, bitter, sour, salty, and umami. Generally speaking, sweetness indicates energy-dense foods, umami aids in amino acid absorption, saltiness helps maintain electrolyte balance in daily diets, and sourness often signifies excessive acidity or food spoilage. Natural bitterness is commonly linked to the defense mechanisms against predation of animals and plants, whereas bacterial quorum-sensing molecules activating bitter taste receptors may contribute to bitterness in certain spoiled foods.
The first taste receptor gene TAS1R1 was discovered in 1999 by Hoon et al.3 when they sequenced the cDNA library of taste cells on the tongue of mice. Subsequently, the TAS1R2 gene was identified based on the TAS1R1 gene.3 In 2001, researchers identified TAS1R3, a gene related to the perception of sweet taste, on human chromosome 4, thus identifying the first potential sweet taste receptor.4,5,6,7,8,9 Currently, research on bitter, sweet and umami taste receptors in mammals is relatively well-established. Sweet, bitter, and umami perception are mediated by G-protein-coupled receptors (GPCRs) and their downstream second messenger signaling pathways, while salty or sour recognition mainly relies on ion channels. GPCRs are crucial proteins involved in cell signaling. The taste GPCRs can be categorized into two major families: type 1 taste receptors (TAS1Rs) and type 2 taste receptors (TAS2Rs). The sweet and umami taste receptors (TAS1Rs) belong to class C GPCRs and form heterodimers consisting of an extracellular domain and a seven transmembrane domain (7TM).3 On the other hand, bitter taste receptors (TAS2Rs) are generally classified as class A (rhodopsin-like) GPCRs.3 However, this assignment remains debated because TAS2Rs share limited sequence similarity with any other GPCR class, and some researchers supported that they should be classified separately as class T.10 In the phylogeny-based GRAFS framework proposed by Fredriksson and Schiöth, TAS2Rs were grouped with Frizzled receptors as a separate family (F).11 Therefore, the precise phylogenetic position of TAS2Rs remains ambiguous and requires further investigation. Despite their structural diversity, signaling from both TAS1Rs and TAS2Rs converges on downstream effectors including PLCβ2 and TRPM4/5.12,13
Taste GPCRs are not limited to the tongue (taste buds) but are also expressed in various tissues throughout the body, where they perform diverse physiological functions. For instance, TAS2Rs have been found in the respiratory system,14,15,16 gastrointestinal tract,17 urogenital tract,18,19 and periodontal tissues,20,21 suggesting a role in detecting harmful substances and regulating immune responses in these areas. TAS1Rs are expressed in the gastrointestinal tract, including the small and large intestines, where they likely participate in the regulation of nutrient absorption and hormone secretion.22,23 In the cardiovascular system, TAS2Rs are present in vascular smooth muscle and heart tissues, where they influence blood pressure regulation and cardiac function.24 Additionally, taste GPCRs in the brain and nervous system play roles in neurotransmission and the regulation of excitability and responsiveness.25,26 Thus, the widespread distribution of taste receptors throughout various tissues highlights their involvement in processes beyond traditional taste perception, such as immune regulation, endocrine signaling, and tissue responses to external stimuli.
The dysfunction of taste GPCRs may have implications for both health and disease. On the one hand, taste GPCRs in taste buds generate peripheral taste signals and contribute to taste perception, which inevitably affects human health by affecting food intake and diet choice. In extra-gustatory locations, taste GPCRs are expressed in a variety of cell types, including solitary chemosensory cells (SCCs)/tuft cells, fibroblasts, enteroendocrine cells, pancreatic β cells, immune cells, etc. These “ectopic” taste GPCRs contributes to various physiological functions, including innate immune regulation, hormone and metabolism regulation, and are intricately linked to the occurrence and progression of numerous diseases, thereby rendering them potential therapeutic targets.27,28 The in-depth investigation into the functional mechanism of taste GPCRs beyond the realm of taste perception and the experimental structural analysis of taste GPCRs serve as crucial prerequisites for comprehending the involvement of taste GPCRs in diseases and facilitating the development of efficacious pharmaceuticals, which constitute focal points within this review. Moreover, the scope of this review will be restricted to the sweet and bitter taste GPCRs, as there exists a plethora of comprehensive studies available on these two types of taste GPCRs in various diseases.
Signaling and structures of sweet and bitter taste GPCRs
Distribution of sweet and bitter taste signaling
While taste perception is limited to the sensory cells in the taste buds, taste receptors are also expressed and functional in various extra-gustatory tissues, where their signaling pathways differ.29 Although mediated by different GPCRs, sweet and bitter taste signaling share a prevalent pathway in taste buds (Fig. 1).30 Binding of tastants to sweet or bitter taste GPCRs activates the heterotrimeric G protein α-gustducin, leading to release of the β3γ13 subunit that activates phospholipase C-β2 (PLCβ2).31,32 PLCβ2 hydrolyzes phosphatidylinositol-4,5-bisphosphate (PIP2) into diacylglycerol (DAG) and inositol triphosphate (IP3), which binds to IP3 receptor type 3 (IP3R3) on endoplasmic reticulum, causing Ca2+ release.33,34 The increase in intracellular Ca2+ opens transient receptor potential channel M5 (TRPM5) and TRPM4 channels, resulting in Na+ influx that depolarizes the cell.13 In the scenario of taste perception, ATP is released by taste cells via voltage-gated channels CALHM1/CALHM3 in response to action potentials, serving as the primary neurotransmitter.35,36 This release stimulates purinergic receptors 2 and 3 (P2X2 and P2X3) on afferent cranial nerves, thereby activating the gustatory cortex.37 In addition to the above signaling pathway, sweet and bitter taste signaling is also regulated by cyclic adenosine monophosphate (cAMP).38 Early electrophysiological studies demonstrated that the application of cAMP analogs induces depolarization of taste bud cells by reducing K⁺ efflux through Protein Kinase A (PKA)-mediated phosphorylation of K⁺ channels.39 Both rat and hamster studies show that saccharin or sucrose depolarizes taste bud cells and elicits action potentials, and a permeable analog of cAMP can mimic this effect.40,41 Therefore, the cAMP–AC–PKA pathway may regulate sweet taste transduction in taste buds, although current evidence remains insufficient. Unlike sweet taste GPCRs, bitter stimuli cause the dissociation of Gα-gustducin, a transducin-family G protein that participates in taste signaling, including umami perception.42,43 The dissociated Gα-gustducin activates phosphodiesterase (PDE), leading to reduced intracellular cAMP levels and downstream modulation of PKA activity.44 Biochemical and rapid kinetic studies showed millisecond-scale reductions in cAMP in response to bitter tastants that were abolished by Gα-gustducin–specific antibodies but unaffected by inhibition of PLCβ2, demonstrating a Gα-gustducin–dependent regulation of cAMP levels.45 The reduction in cAMP levels affected PKA activity, thereby relieving inhibition of the PLCβ2-IP3 signaling pathway. Consequently, reduced PKA-mediated phosphorylation promotes the release of stored Ca2+ and neurotransmitters, thereby amplifying the bitter taste signal.46 Beyond stimulus-evoked regulation, basal regulation of cAMP-mediated signaling also appears to be important for maintaining taste cell responsiveness. Clapp et al. found that basal cAMP levels are elevated in taste buds from Gα-gustducin–deficient mice and that pharmacological inhibition of PKA restores Ca²⁺ responses to bitter stimuli.47 Based on these observations, they proposed a model in which Gα-gustducin is tonically active at rest, maintaining the low cAMP levels to ensure the key Ca²⁺ signaling effectors (PLCβ2 and IP₃R3) remain competent to trigger a Ca2+ response.47
Shared signaling pathway of sweet and bitter taste GPCRs. Left, Sweet taste GPCRs (TAS1R2/TAS1R3); Right, Bitter taste GPCRs (TAS2Rs). Sweet or bitter ligand binding to TAS1R2/TAS1R3 or TAS2Rs activates the taste G-protein gustducin (Gα/β/γ), which stimulates PLCβ2 to hydrolyze PIP2 into IP3 and DAG. IP3 gates IP3 receptors (IP3R) on the endoplasmic reticulum, leading to Ca²⁺ release and TRPM4/5 activation, which causes depolarization and action potential generation. Voltage-gated channels CALHM1/CALHM3 respond to action potentials and release adenosine-5’-triphosphate (ATP), serving as the primary neurotransmitter
The signaling components and pathways in extra-gustatory tissues partially overlap with the classic taste transduction cascade in taste buds. For example, in intestinal tuft cells, activation of TAS2Rs leads to dissociation of Gα-gustducin/Gβ1γ13 and/or Gαo/Gβ1γ13, where Gβ1γ13 engages a PLCβ2–IP3 pathway similar to that in taste buds. IP3 binds to and activates IP3R2, instead of IP3R3 found in taste buds, which exhibits a higher affinity for IP3 but a lower sensitivity to Ca2+. The release of Ca2+ activates TRPM5 only, causing Na⁺ influx and membrane depolarization, which ultimately promoting the release of IL-25 and triggering type 2 immune responses.48 However, the signal transduction pathways differ across various extra-gustatory tissues. In cystic fibrosis airway epithelial cells, upregulated Gαi suppresses AC activity and lowers intracellular cAMP levels, which in turn enhances Ca²⁺ signaling and TAS2R14-mediated innate immune responses.49 Moreover, TAS2Rs induce airway smooth muscle relaxation through four potential mechanisms, and the underlying signaling pathways are distinct.50,51,52,53 Therefore, the signaling components and pathways of sweet and bitter taste GPCRs in extra-gustatory tissues may depend on cell type and functional context, yet the precise mechanisms governing these pathways remain incompletely understood.
Protein structure of TAS1R2/TAS1R3
The Class C GPCR family is known for its unique domain composition and functional characteristics, primarily regulating physiological functions by binding to external chemical signals like neurotransmitters or metabolites. TAS1R2 and TAS1R3 detect sweet taste molecules through heterodimer formation, sharing three major domains: (1) the large extracellular Venus Flytrap Domain (VFT) responsible for initial ligand binding. (2) the Cysteine-Rich Domain (CRD) connecting VFT and transmembrane domain, facilitating signal transduction. (3) the classical seven-fold Transmembrane Domain (TMD), activating G-proteins and downstream signaling pathways.54 Other members of Class C GPCRs, such as metabotropic glutamate receptor (mGluR) and calcium-sensing receptor (CaSR), also possess the VFT, CRD, and TMD structure, serving as important references in studying sweet taste receptor structure.
Sweet taste ligands and their binding sites
The TAS1R2/TAS1R3 heterodimers exhibit the ability to bind various types of ligands, including small-molecule sweeteners, protein sweeteners, and allosteric regulatory molecules.55,56,57 Each type of ligand interacts with a specific binding site to initiate or modulate sweet taste signaling (Supplementary Table. 1). It has been demonstrated that small-molecule sweeteners, such as saccharides, primarily activate the receptor by binding to the VFT domain of TAS1R2. The VFT domain of TAS1R2 captures these molecules through two “splint-like” structures, leading to conformational changes and subsequent activation of downstream signaling cascades.57 These small-molecule sweeteners perfectly fit into the binding site of TAS1R2 due to their smaller size and simpler structure. For instance, sucrose, a natural carbohydrate, binds to the VFT region of both TAS1R2 and TAS1R3 with an EC50 value of 62 mmol/L.55 As peptide-based small molecule sweeteners, aspartame and neotame predominantly bind to the VFT region of TAS1R2 with EC50 values of 0.75 mmol/L and 5 mmol/L respectively.55,58 Saccharin and Acesulfame K are sulfonamide-based sweeteners that also interact with the VFT region of TAS1R2 at EC50 values of 0.19 mmol/L and 0.54 mmol/L respectively.55,58 In addition to VFT-targeting sweeteners, some small-molecule sweeteners have been reported to bind to the TMD of TAS1R2/TAS1R3, including cyclamate and perillartine. For instance, cyclamate functions as a partial agonist with species-dependent activation, binding to human TAS1R3 within a pocket formed by transmembrane helices 3, 5, and 6, as well as extracellular loop 2. This interaction induces a conformational shift in the helical bundle, facilitating G protein coupling and triggering a sweet taste response.59,60 Similarly, perillartine activates the receptor through direct interaction with the TMD of TAS1R2, bypassing the need for VFT engagement and modulating sweet taste in a species-specific manner.61,62
In contrast, protein-based sweeteners such as thaumatin typically bind to the VFT domain of TAS1R3 due to their substantial molecular weight. The VFT domain of TAS1R3 is relatively large and flexible, allowing it to accommodate these macromolecular ligands effectively. Existing literature suggests that the binding of protein-based sweeteners to TAS1R3 necessitates higher specificity, as they must interact with more intricate binding pockets in order to initiate signaling.63 These protein molecules provide a stronger activation signal by selectively binding to specific regions of TAS1R3, thereby enhancing the perception of sweetness. Protein sweeteners like thaumatin and monellin exhibit exceptionally high sweetness perception efficiency and target either the CRD or VFT region of TAS1R3 with very low EC50 values (0.005 mmol/L and 0.001 mmol/L respectively).64 The binding sites between these protein molecules and TAS1R3 are highly specific, resulting in intense and long-lasting perception of sweetness.
Allosteric modulators regulate the conformational transitions of the TAS1R2/TAS1R3 heterodimer, altering the intensity of sweet taste signal transmission, and can be categorized into sweet taste enhancers and inhibitors. Sweet taste enhancers primarily bind VFT domain of TAS1R2, stabilizing its active conformation. This enhances the affinity of sweet substances, such as sucrose or aspartame, reduces their EC50 values, and amplifies the sweet taste signal.65 Additionally, sweet taste enhancers interact with key residues in the VFT domain (e.g., K65, Y103, and D307), lowering the energy required for conformational closure, thereby increasing the sensitivity of the receptor and enhancing sweet perception. In contrast, sweet taste inhibitors, such as lactisole, typically act on the TMD of TAS1R3. By interacting with specific residues (e.g., A733 and R790), these inhibitors induce an inactive receptor conformation, thereby blocking signal transmission and suppressing sweet perception.57
Structure of human TAS1R2/TAS1R3
The structural resolution of TAS1R2/TAS1R3 faces several technical challenges, including low heterologous expression, the impact of receptor modification on stability, and difficulty in vitro extraction. These issues significantly limit detailed structural investigations of TAS1R2/TAS1R3 heterodimers, especially obtaining high-resolution crystal structures. Low heterologous expression is a major bottleneck for TAS1Rs research as it often results in insufficient protein production under laboratory conditions, making it difficult to obtain functional proteins for subsequent analysis.66,67 GPCRs require specific cellular environments and cofactors to maintain their native conformation and activity when expressed in heterologous systems, further complicating the expression and purification processes for TAS1Rs.68 Post-translational modifications like glycosylation or phosphorylation significantly influence the conformation and functionality of TAS1R2 and TAS1R3 receptors.69 Glycosylation not only aids proper protein folding and stability but also affects membrane localization and ligand binding ability.69 However, these modifications pose challenges in maintaining the active structure of the receptor during in vitro analysis. Additionally, modeling modifications such as phosphorylation that occur under specific cell signaling conditions prove challenging in vitro systems, limiting comprehensive investigation into receptor function. The complex structures of TAS1R2/TAS1R3 heterodimers make them prone to disassembly or loss of activity during extraction. Detergent treatment poses a significant risk to the transmembrane regions of GPCRs by potentially causing non-functional aggregation or structural disintegration.70 Furthermore, as the active structure of the TAS1Rs relies on cooperation with other molecules or cofactors, extracting the receptor alone may result in the loss of these crucial accessory components, further complicating efforts to obtain a complete and functional structure.68
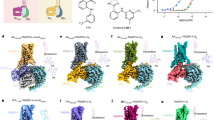
In 2025, three landmark studies have been published successively, deconstructing the structure of the sweet taste receptors TAS1R2/TAS1R3 for the first time. Zuker’s research team engineered an innovative dual-promoter baculoviral construct to co-express both subunits, successfully obtaining stable heterodimer formation while avoiding nonfunctional TAS1R2 homodimers during purification.71 Using single-particle cryo-EM, they characterized the agonist-bound TAS1R2/TAS1R3 heterodimers, including sucralose and aspartame. Consistent with predictions, sweet ligands bound exclusively to the VFT domain of TAS1R2 while TAS1R3 remained open and ligand-free, confirming the asymmetric architecture of the heterodimer. Mutation analysis identified key residues in the TAS1R2 binding pocket that govern ligand specificity and potency, linking structural contacts to functional activation. Beyond ligand recognition, the study traced conformational changes across domains: agonist-induced closure of the TAS1R2 VFT was transmitted through the CR linkers, where a notable kink in the TAS1R3 linker intercalated into the VFT dimer interface. This arrangement likely couples ligand binding to TMD rearrangements, creating a TM6–TM6 interface favorable for G protein engagement and intracellular signaling. Functionally, TAS1R2 emerges as the principal ligand-binding and G-protein-coupling subunit, whereas TAS1R3 contributes critical allosteric and structural roles at the VFT–CR interface and within the TMD dimer. Regrettably, the absence of the apo structure of the receptor restricted the characterization of structural differences between the inactive and active states of the heteromeric receptor. Nevertheless, this study established a structural and mechanistic framework that was supported by the subsequent two researchers.
Subsequently, Shi et al. resolved the human sweet taste receptor TAS1R2/TAS1R3 in both apo and sucralose-bound states using high-resolution cryo-EM, thereby addressing limitations of the Zuker team’s work.72 To achieve this, they employed an innovative fluorescent-protein fusion strategy that stabilized the full-length heterodimer, once again confirming its asymmetric structure. While reaffirming the ligand specificity of the TAS1R2 VFT, the study showed that ligand binding initiates a coordinated yet modest activation cascade. Upward movement of the TAS1R2 LB2 transmits force into the CRD, bending it and shifting the VFTs, while a TAS1R3 loop is displaced in the same direction. Together, these adjustments shorten the receptor’s vertical height and propagate subtle conformational changes into the TMD. At the TMD dimer interface, activation involves only minor TM6 rotations, primarily localized to the intracellular face of TAS1R2, consistent with the observations of Zuker et al. They also emphasized that TAS1R2/TAS1R3 transduces signals through small, asymmetric conformational changes, which are distinct from the large-scale rearrangements observed in other class C GPCRs.
Recently, Wang et al. further extended these findings through cryo-EM structures of apo and sweetener-bound receptors using both human/human and human/mouse heterodimers.73 This study determined the binding modes of sucralose and advantame in the TAS1R2 VFT pocket, thereby providing structural insights into receptor activation by natural sweet compounds and amino acid–based sweeteners. In addition, the authors captured two key events during activation that had not been reported previously. First, agonist binding induces clamshell closure in TAS1R2, accompanied by a stabilizing salt bridge that defines the compact state. Second, a previously undefined loose conformation was identified, in which the receptor adopts a more open, activation-prone state. This transition is driven by insertion of a TAS1R2 loop into the inter-subunit interface, displacing TAS1R3 and triggering separation of the VFT lower lobes. Notably, the TAS1R3 VFT clamshell also opens further in the loose state compared to the apo conformation. Together, these findings provide novel insights distinct from the previous two studies and suggest structure-guided strategies for designing receptor modulators, as sweeteners that stabilize the loose state may enhance perceived sweetness.
Protein structure of TAS2Rs
In humans, 26 TAS2Rs have been identified, while only the structures of TAS2R14 and TAS2R46 are determined.74 Notably, structural comparisons between these two receptors reveal substantial differences, particularly in the extracellular regions, suggesting that structural features identified in one TAS2R may not be generalizable across the family.75 This structural heterogeneity likely underlies the distinct ligand recognition profiles of individual TAS2Rs and may contribute to their tissue-specific expression patterns. Functionally, TAS2Rs are activated by an exceptionally broad spectrum of bitter ligands, including natural compounds (e.g., alkaloids, terpenoids, flavonoids), synthetic chemicals, toxic plant metabolites, and many clinically used drugs. This diversity reflects both the evolutionary role of TAS2Rs in detecting harmful substances and their emerging pharmacological relevance. BitterDB, a comprehensive resource for bitter-tasting molecules and their associated receptors, currently catalogs over 2 200 bitter compounds across 66 species and includes approximately 700 ligands with defined TAS2R associations (as of 2025).76 Given its depth and accessibility, detailed discussion of bitter ligands is omitted here.
Early structural studies of bitter taste GPCRs were mainly conducted through homology modeling, ligand docking experiments, and functional heterologous expression combined with in vitro mutagenesis.77 Brockhoff et al. apply point mutagenesis and function assay to identify key residues involved in ligand binding of TAS2R46. This strategy also enabled iterative refinement of the obtained structural models.78 Subsequently, Nowak et al. demonstrated that numerous mutations enhanced the potency and/or efficacy of specific agonists by generating point mutants of TAS2R14, revealing its broad ability to recognize diverse agonists.79 In addition to these studies, a series of early mutational analyses from the Chelikani laboratory provided functional insights into bitter taste receptor activation and regulation at a time when high-resolution structural information was not yet available.80,81,82,83 These studies proposed that interhelical and intracellular interaction networks stabilize TAS2Rs in an inactive conformation.80,82 Functional mutagenesis further showed that disruption of these interactions by selected point mutations can lead to constitutive receptor activity, manifested as detectable signaling output in the absence of ligand.80,81,82 Notably, mutations within the TM5–ICL3–TM6 region were found to be particularly prone to inducing constitutive activity, suggesting that these residues may impose conformational constraints on TM6 and raise the energetic barrier for activation.82 In addition, early mutational studies indicated an association between membrane cholesterol levels and the signaling strength of TAS2Rs.83 However, these predictions have significant limitations due to the low sequence homology with other GPCR families and the difficulty in obtaining experimental structures. In recent years, high-resolution structures of TAS2R46 and TAS2R14 have provided new insights for subsequent studies.84,85,86,87,88
The TAS2R46 receptor is widely acknowledged as a broad-spectrum bitter taste receptor with the ability to recognize various ligands (Supplementary Table. 1).84 Its structural analysis provides crucial insights into this characteristic.89 The ligand-binding pocket of TAS2R46 is described as relatively deep and exhibits significant structural flexibility, enabling it to accommodate and identify bitter molecules with diverse chemical properties. In the investigation of its cryo-EM structure, multiple polar and non-polar amino acid residues were observed surrounding the binding pocket of TAS2R46. These residues form stable interactions with ligand molecules through hydrogen bonding, van der Waals forces, and hydrophobic interactions.89 This diversity in binding modes greatly expands the range of ligand recognition for TAS2R46, allowing it to interact with bitter molecules of varying sizes, shapes, and chemical properties. To be specific, strychnine interacts with conserved amino acids within the TAS2R46 binding pocket through hydrogen bonding and hydrophobic interactions.89 These interactions induce rearrangements in the transmembrane helices, particularly helices 6 and 7, creating an optimal conformation for downstream G protein signaling.89 Functional analyses indicate that TAS2R46 primarily engages Gi/o-type G proteins, activating intracellular signaling pathways that regulate calcium ion release and trigger bitter taste signaling.89
The recent studies on TAS2R14 provide crucial insights into its structure, ligand binding, and signal transduction (Supplementary Table 1).85,86,87,88 TAS2R14 displays two distinct pockets, a conventional orthosteric binding site and a unique intracellular allosteric site, which are connected by an elongated cavity.85 Notably, Hu et al. reported that aristolochic acid (AA) bound to a third binding pocket of the TAS2R14-miniGs/gust complex, which is situated beneath pocket 2.86 Cholesterol occupies the orthosteric site, engaging in hydrophobic interactions with key residues like W893.32, critical for stabilizing receptor conformation. An intracellular binding site is targeted by ligands like compound 28.1 (Cpd 28.1) and flufenamic acid (FFA), forming interactions that enhance activation stability. Interestingly, cpd 28.1 was found at both intracellular and extracellular binding sites and to form more pronounced hydrophilic interactions with the orthosteric site than cholesterol.87 According to Peri et al., the detected cholesterol may result from hydrolysis of the excess cholesteryl hemisuccinate (CHS) used during protein purification, as their structural characterization using lower CHS concentrations indicated FFA binding at the orthosteric site as well.88 Upon ligand binding, conformational changes are observed in transmembrane helices, especially TM6, facilitating G-protein coupling. Cpd 28.1 and FFA act as both a direct agonist and a positive allosteric modulator, stabilizing the receptor’s active state and promoting interactions with Gα-gustducin and Gi1.88 This dual binding mechanism underscores the broad ligand recognition ability of TAS2R14 and its ability to signal via multiple pathways.
Collectively, these cryo-EM studies provide a structural basis for the functional hypotheses proposed by earlier mutational analyses of bitter taste GPCRs.84,85,86,87,88 The extensive networks of polar and hydrophobic interactions observed in TAS2R14 and TAS2R46 are compatible with the conformational restraint mechanisms proposed by the Chelikani laboratory.84,85,86,87,88 Moreover, the direct visualization of TM6 outward movement and formation of the intracellular G-protein–binding cavity highlights TM6 rearrangement as a common activation feature of bitter taste GPCRs.84 Notably, the identification of a cholesterol-binding pocket in TAS2R14 provides a structural rationale for earlier observations that membrane cholesterol modulates bitter taste receptor signaling.85,86,87 Thus, despite being derived from different TAS2R subtypes, the functional conclusions of early mutational studies are broadly supported by current structural evidence.
In-depth analysis of the amino acid sequences of TAS2R46 and TAS2R14 allowed identification of crucial residues that are closely related to ligand binding. These residues play a pivotal role in facilitating the ligand binding process and are often conserved amino acids responsible for establishing stable interactions with specific chemical groups. Unveiling the precise location and nature of these critical residues is beneficial for comprehending the specific ligand recognition mechanisms of TAS2Rs. Currently, several studies have reached a general consensus regarding the key residues of TAS2R14 binding sites.85,86,87,88 The orthosteric pocket is formed by an extensive network of hydrophobic residues, to which cholesterol,85,86 cpd28.1,87 and FFA88 can all bind. Notably, the highly conserved amino acid residue W893.32 forms CH-π interactions with the steroid nucleus of cholesterol, which is similar to the role of W883.32 in the activation of TAS2R46 by strychnine.85,86,89 Compared to cholesterol, cpd28.1 and FFA also interact with S652.60 and S176ECL2, whose critical role in receptor function has been confirmed through mutagenesis experiments.87,88 The allosteric binding site is formed by TM3, TM5–TM7, and the cytoplasmic end of the G protein α5 helix. S1945.54 and H2767.49 in this site form hydrogen bonds with several agonists, and mutations in these residues reduce or eliminate their potency and efficacy of cpd28.1,85,86,87 FFA,88 and AA.87 Additionally, L353H5.25 of Gαi1 and Gα-gust interact with the fluorine of cpd28.1 and Y1073.50 in TAS2R14, suggesting their potential role in enhancing the stability of the TAS2R14–G protein complex.85,86,87 Unlike TAS2R14, TAS2R46 has only one extracellular orthosteric binding site, where the horizontally placed indole ring of W883.32 serves as a landing platform for the strychnine molecule, establishing a π-π interaction with the benzene ring of strychnine.73 Kim et al. attempted to explore the intracellular binding site of TAS2R46 through structural superposition.85 Unfortunately, despite several conserved residues in both TAS2R46 and TAS2R14, there are still notable structural differences between them. The key residues of TAS2R14 undergo flipping, displacement, and replacement at the corresponding positions in TAS2R46.85 In summary, except for W3.32, the specific residues involved in the ligand-binding pockets of the two receptors are variable, suggesting that bitter taste GPCRs exhibit highly diversified binding modes for agonists.
Although the presence of cholesterol in the orthosteric pocket remains debated, the observation of cpd28.1 and FFA occupying this site suggests that dual-site binding in TAS2R14 is unlikely to be incidental.85,86,87,88 The cryo-EM structures of TAS2R14 consistently show stable conformations in which both binding pockets are occupied, suggesting that ligand binding contributes to receptor stability and activity. The ability of the orthosteric and allosteric sites to bind either identical or distinct ligands may enhance the responsiveness of TAS2R14 across varying environmental contexts and ligand concentrations, although this hypothesis requires further validation.75 The hydrophobic, elongated tunnel connecting two pockets resembles the lipid membrane-entry pathway described in lipid GPCRs.90,91 Kim et al. cautiously suggested that this tunnel might participate in cholesterol engagement, given the high concentration of cholesterol in the plasma membrane.85 Notably, cpd28.1, FFA, and AA all share the general features of small bitter tastants with hydrophobic aromatic scaffolds, which may facilitate membrane diffusion and promote access to the intracellular pocket.92,93 In contrast, the cryo-EM structure of TAS2R46 reveal a more defined ligand recognition pattern centered on residues W883.32 and E2657.3984. Many clinical drugs containing an aromatic moiety and a positively charged nitrogen interact with the two key residues through π–π stacking and salt-bridge formation.94 While, bitter tastants with a lactone substructure rely on hydrogen bonding between their carbonyl and hydroxyl groups and these residues.95 These structural studies on TAS2R14 and TAS2R46 provide frameworks for understanding the broad spectrum of bitter taste GPCRs and offer insights into how ligands from different origins adopt distinct binding modes.
The role of sweet and bitter taste GPCRs in diseases
The physiological functions of sweet and bitter taste GPCRs vary across different tissues and cell types (Fig. 2). The upstream signals and downstream functions of taste receptors are relatively clear in certain organs, tissues, and cell types. In some studies, researchers have discovered that specific cell types within particular tissues can express taste receptors. Their functions are often investigated through in vitro experiments involving exogenous ligand addition. However, the sources of endogenous stimulation for these taste receptors within the body remain unclear, thus, the physiological roles of taste receptors in these cells remain largely unknown. Currently, it is increasingly evident that based on their expression location and classification according to upstream stimulation patterns, sweet and bitter taste receptor functionalities primarily involve aspects such as taste perception, innate immunity, and metabolic regulation. Meanwhile, taste receptors may influence other conditions such as asthma, reproductive dysfunction, and cancers through additional mechanisms, however, further research is required to substantiate these claims (Supplementary Table 2).
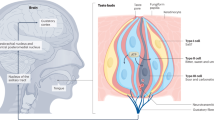
Distribution and expression of sweet and bitter taste GPCRs in diseases. The expression of sweet and bitter taste GPCRs in different body parts is related to various pathological conditions, including oral diseases (periodontitis, dental caries), respiratory diseases (CRS, asthma), metabolic disorders (T2D, obesity), urinary diseases (urinary tract infection), reproductive conditions (PTB, male infertility), neurological disorders (AD, PD), and various cancers. Specific receptor subtypes associated with each disease are indicated
Oral diseases
Dental caries
The primary cause of dental caries is the demineralization of tooth hard tissue resulting from acid production by oral microorganisms. Diet plays a significant role in the pathogenesis of dental caries by influencing the metabolic activities of oral microorganisms. Sweet and bitter taste GPCRs can exert an influence on dietary choices, modulate the host’s innate immune response, and impact the development of caries.
Single nucleotide polymorphisms (SNPs) in the genes encoding sweet taste GPCRs have been shown to alter sweet taste preference and sugar intake, thus affecting the risk of caries.96,97 Most studies reached the conclusion that individuals who hold an Ile191Val polymorphism in TAS1R2 consume fewer sugars and are at a lower risk of developing dental caries, while Ile homozygotes, whose carbohydrate intake is higher, showed more frequent high-risk caries experience.97,98,99,100 However, one study reported an opposing finding: children who carried the Val allele were more frequently affected by caries than those who carried the common Ile allele.101 Two SNP loci of TAS1R3 (rs307355 and rs35744813) have been reported to be associated with sweet taste sensitivity,102 and the rs307355 polymorphism has been claimed as an independent risk factor for dental caries.97 SNP rs307355 is located within the E-box transcription factor binding sites upstream of the TAS1R3 promoter, and the nucleotide variation at this site affects TAS1R3 expression by reducing promoter activity.102 In interpreting these findings, it is crucial to comprehend that the impact of taste receptor SNPs on diet and caries susceptibility is influenced by various factors such as ethnicity, age, and culture. Conversely, the correlation between taste receptor function and dental caries is evidently not fully elucidated by taste perception and dietary preference. A typical example is the finding regarding the TAS2R38 SNP and caries susceptibility.103,104
The function of TAS2R14 and TAS2R38 in gingival epithelial cells may be associated with the innate immune response triggered by cariogenic oral bacteria Streptococcus mutans, thereby impacting the host’s susceptibility to dental caries. An in vitro study was conducted to isolate and culture human gingival epithelial cells expressing different genotypes of TAS2R38.105 The results showed that TAS2R38 expression was significantly upregulated in PAV homozygous cells after S. mutans exposure, while no change was observed in AVI homozygous cells.105 The secretion of IL-1α and antibacterial peptide hBD-2 in gingival epithelial cells were also affected by exposure to S. mutans and/or TAS2R38 genotype.105 Although the evidence from this in vitro study is weak, some experimental results are difficult to explain, the results still suggest that TAS2R38 function in gingival epithelial cells may be involved in regulating the interaction between cariogenic bacteria and host innate immunity. Another study revealed that the quorum sensing molecule CSP-1 of S. mutans was capable of triggering the innate immune response in gingival epithelial cells through TAS2R14.106 TAS2R14 facilitated intracellular calcium influx and the secretion of pro-inflammatory cytokines (IL-8) upon stimulation by CSP-1 and attracted differentiated HL-60 immune cells.106 Intriguingly, treatment of gingival epithelial cells with CSP-1 inhibits the growth of Staphylococcus aureus through TAS2R14, by inducing hBD-2 secretion.106 This implies that S. mutans may exploit the bactericidal effect facilitated by bitter taste receptors to secure a competitive edge within the oral biofilm, representing one of the mechanisms through which S. mutans induces dental caries. Currently, only a limited number of in vitro cell-based studies suggest that bitter receptors can influence dental caries risk through innate immune regulation. More in-depth mechanistic exploration and animal experiments are necessary to obtain reliable conclusions.
Periodontitis
The initiation of periodontitis is associated with the dysbiosis of oral microecology caused by abnormal interaction between oral microflora and the host. Gingival SCCs, a cell type expressing bitter taste receptors and taste signal transduction elements, had been identified in the gingival epithelium of mice.107 Tas2r105 in gingival SCCs could be activated by gram-negative bacteria quorum sensing acyl-homoserine lactone (AHL) molecules. In mice lacking gingival SCCs (Pou2f3−/−) or taste signal transduction molecule (Gnat3−/−), there were disorders in secretion antimicrobial peptides (such as beta-defensin 3) in periodontal tissue, dysbiosis in periodontal microflora characterized by increased bacterial load, decreased diversity and increased pathogen NI1060, ultimately leading to more severe inflammation and alveolar bone resorption in a periodontitis model.107 These findings demonstrate that gingival SCCs act as immune sentinels in the periodontium, recognizing bacterial metabolites through bitter taste receptors and mediating host immune defense to maintain a balanced periodontal microecology. Bitter taste receptors can serve as key targets for regulating periodontal homeostasis. Topical treatment with exogenous bitter tastants could effectively boost the secretion of periodontal antimicrobial peptides mediated by bitter taste receptors and prevent excessive bacterial colonization within the periodontal microecology in animal experiments.107
By analysis of two single-cell RNA sequencing datasets from human periodontal tissues,108,109 we observed a significant expression of bitter taste receptors in periodontal fibroblasts.20 Subsequently, through in vitro culturing of human gingival fibroblasts, we successfully validated the presence of a wide range of bitter taste receptors.110 Our functional study revealed that activation of TAS2R16 could effectively counteract the NF-κB signaling pathway activated by LPS and subsequently reduce the expression levels of inflammatory factors such as IL-8, thereby mitigating chemotaxis of neutrophils.110 This particular function exhibited by TAS2R16 in gingival fibroblasts may play a crucial role in periodontal inflammation resolution, as partially demonstrated in our recently published animal study.111 Further investigations are warranted to elucidate the endogenous ligands capable of activating TAS2R16 or other functionally similar bitter taste receptors during periodontal inflammation resolution.
Pathogen infections
Taste GPCRs can function as chemoreceptors that not only detect chemicals known as tastants to initiate taste signaling but also recognize microorganisms such as bacteria, thereby activating downstream signaling cascades, contributing to innate host defense. Research on the functions of taste receptors related to innate immunity has primarily focused on a specific group of taste-like cells, commonly referred to as tuft cells in certain tissues (e.g. intestine and urinary tract) and SCCs in others (e.g. periodontal tissue and respiratory tract). Additionally, in other cell types such as epithelial cells and macrophages, the function of taste GPCRs is also associated with innate immunity. We will discuss recent advancements in understanding the roles of taste GPCRs in relation to pathogen infection across various organs and tissues.
Respiratory tract infections
In the upper respiratory tract, SCCs were observed in scattered locations such as the nasal respiratory epithelium, the entrance of the vomeronasal canal, and the larynx. Among these locations, SCCs of the nasal respiratory epithelium have been extensively studied. These cells primarily express TAS2Rs, with a relative low expression of TAS1Rs.112 Nasal SCCs were first identified in mice. Inhaled stimuli stimulate TAS2Rs on mouse nasal SCCs which then activate trigeminal nerves connected synaptically with them, resulting in changes in respiratory rate and triggering protective reflexes like apnea and sneezing,113 and releasing of antibacterial agents.14,113 Additionally, activation of SCCs leads to the release of neurotransmitters from trigeminal nerve terminals, which in turn induces mast cell degranulation and plasma extravasation. This process ultimately results in neurogenic inflammation within the nasal cavity.114
In the human sinuses, SCCs are present at a low density, approximately 1 in every 100 sinus epithelial cells.115 Despite their limited abundance, they play a crucial role as immune sentinels. In human primary sinonasal air-liquid interface cultures and tissue explants, Lee RJ et al. found that TAS2R activation in SCCs triggered calcium waves that propagate via gap junctions to the surrounding ciliated epithelium, stimulating the secretion of antimicrobial peptides. Conversely, activation of sweet taste receptor (TAS1R2 and TAS1R3) in SCC inhibits TAS2R-mediated calcium waves to maintain normal airway immune homeostasis, until depletion of glucose in the airway surface due to infection, which releases the inhibition of TAS1R-signaling on TAS2R-mediated immune response.15 Interestingly, common nasal colonizers can exploit this natural regulatory mechanism to evade immunity. Lee RJ et al. demonstrated that Staphylococcus-produced D-amino acids can activate SCC sweet taste receptors and inhibit TAS2R-mediated innate defense.116
In mice, SCCs are widely distributed throughout the lower respiratory tract, including the lungs.117 Tracheal SCCs express a diverse array of taste receptors, particularly TAS2Rs. When stimulated by AHL, these receptors can elevate intracellular Ca2+ levels, thereby promoting acetylcholine release and facilitating nerve impulse conduction along vagus nerve endings associated with tracheal SCCs. Consequently, this mechanism inhibits respiratory rate.118 In addition, thymic stromal lymphopoietin (TSLP) is highly expressed in tracheal SCCs in mice, indicating that SCC serves as a significant source of IL-25 and cysteine leukotriene within the trachea.117 This suggests that tracheal SCCs may play a crucial role in the pathogenesis and progression of chronic inflammatory diseases such as asthma and eosinophilic pneumonia. A recent study in mouse model also found SCCs (named tuft cells in the study) in the injured lung have been demonstrated to facilitate inflammation resolution and tissue repair in H1N1-infected lung.119 Although they did not establish a direct link between the taste GPCRs expressed by lung SCCs and their role in this process, they verified that G protein subunit Gγ13, a downstream protein of the taste GPCRs, is indispensable.119
Nasal ciliated epithelial cells have been demonstrated to express TAS2R4, TAS2R43, TAS2R38 and TAS2R46.16 The quorum sensing molecules N-butyryl-L-homoserine lactone (C4-HSL) and N-3-oxo-dodecanoyl-L-homoserine lactone (C12-HSL), which are produced by Pseudomonas aeruginosa, a common nasal pathogen, can activate TAS2R38 of nasal ciliated epithelial cells and mediate downstream innate immune responses.120 This includes promoting the release of antimicrobial peptides from neighboring cells115 and stimulating nitric oxide (NO) production by nasal epithelial cells to exhibit bactericidal effects, promotion of cilia movement and releasing of mucus to defend against pathogen infections.121 Takemoto K et al. discovered that TAS2R38 is associated with specific subtypes of chronic rhinosinusitis (CRS), and its expression is significantly reduced in nasal polyps from patients with eosinophilic CRS (ECRS) as well as ethmoid sinus mucosa from patients with non-eosinophilic CRS (non-ECRS) compared to healthy subjects.122 Oral and nasal fractional exhaled NO levels were notably decreased in non-ECRS patients, suggesting a complex involvement of TAS2R38 in ciliated cells related to specific CRS phenotypes.122 Furthermore, some studies have indicated that the polymorphism of TAS2R38 is linked to the severity of CRS. Functional (PAV) and non-functional (AVI) variants are observed for TAS2R38. Homozygous AVI/AVI individuals exhibit a diminished response to AHL compared to those homozygous PAV/PAV.120 This genetic variation also leads to increased susceptibility to gram-negative upper respiratory tract infections120 and a higher burden of biofilm formation.123
Intestinal parasitic infections
Intestinal tuft cells have been identified as mediators of type II immune responses that facilitate the expulsion of parasites.124 During parasitic infections, a positive feedback loop is established between intestinal tuft cells and type II innate lymphocytes (ILC2).125,126,127 Intestinal tuft cells secrete IL-25 to activate ILC2 and produce type II cytokines like IL-5 and IL-13, which subsequently promote the differentiation of tuft cells and further secretion of IL-25, thus forming a reinforcing positive feedback loop. Evidence from transcriptome analysis and functional experiments suggests that GPCRs play a pivotal role in the ability of tuft cells to receive signals from the environment and transmit them to functional outputs.17,128 The transcription analysis revealed the expression of taste GPCRs in tuft cells of various tissues, including mouse intestinal tuft cells.17 Taste GPCRs may serve as a crucial pathway for intestinal tuft cells to mediate type II immune responses.
Luo et al. discovered that mouse intestinal tuft cells expressed a diverse range of TAS2Rs, and infection with parasitic helminth Trichinella spiralis can induce tuft cell hyperplasia along with the increased expression of intestinal TAS2Rs.48 However, whether the tuft cell hyperplasia is driven by TAS2Rs or other pattern-recognition receptors remains to be clarified. Additionally, they found that the clearance of parasite infection mediated by tuft cell-mediated type II innate immune response is dependent on TAS2Rs, as demonstrated using the TAS2R inhibitor allyl isothiocyanate. Heterologous expression assays also revealed that extracts from T. spiralis could activate the specific mouse bitter taste receptor Tsa2r143. Consistently, activation of Tas2r143 by salicin induced tuft cell expansion and type II innate immune response. These findings suggest that intestinal tuft cells recognize parasite infection through TAS2Rs and initiate the type II immune defense to eliminate parasites. In terms of downstream signaling mechanisms, their study proposed that TAS2R expressed by intestinal tuft cells function via G proteins Gα-gustducin/Gβ1γ13 and Gαo/Gβ1γ13. However, some studies have indicated that intestinal tuft cells do not express TAS2Rs. Miller et al., for instance, reported in experiments conducted on mouse small intestine tuft cells no expression of any known members from the TAS2R family.129 Therefore, further animal studies are required to confirm whether intestinal tuft cells express TAS2Rs and their functionalities, and verification using human tissue samples is particularly important.
Although the study by Luo et al. did not directly identify sweet taste receptors in intestinal tuft cells, their findings suggest that the use of sweeteners enhances the tuft cell response to parasite infection imply a potential role for sweet taste receptors. Howitt et al. observed a significant tuft cell expansion in both BALB/c mice and C57BL/6 J mice when infected with the parasites Heligomomoides polygyrus (proximal small intestinal infection). However, when infected with the protozoa Tritrichomonas muris (distal small intestinal infection), the tuft cell response was merely observed in C57BL/6 J mice, not in BALB/c mice.130 They hypothesized that this difference may be attributed to loss-of-function mutations in BALB/c TAS1R3 protein. Subsequently, they confirmed high expression levels of TAS1R3 specifically in the distal small intestine (ileum) and identified it as the most highly expressed taste GPCR in ileal tuft cells.130 Mechanistic studies revealed that TAS1R3 knockout resulted in decreased gut Tuft cell numbers at homeostasis and impaired tuft cell response upon stimulation from parasites or succinate.130 The specific mechanism underlying these observations remains unknown. Further exploration is warranted to determine if it involves binding between TAS1R3 and glucose or other intestinal contents to activate downstream pathways. Of note, Howitt et al. found weak expression levels of TAS1R1 and TAS1R2 in isolated ileal tuft cells.130 The expression of TAS1R2 in the intestine is also suggested to be either absent or significantly low by some researchers.131,132,133 Therefore, it is worth exploring whether TAS1R3 plays a role in gut tuft cells independently from TAS1R2, such as through the formation of homodimers or heterodimers with another Class C GPCR.
Urinary tract infections
Urinary tract infection is one of the most prevalent diseases affecting the urinary system and a significant risk factor for severe kidney disease and male infertility.134 In their study, Deckmann et al. identified urethral tuft cells in close proximity to the urethral opening and at the end of glandular ducts within the urethra.18 They found that isolated mouse urethral tuft cells expressed Tas1r1, Tas1r3, and Tas2r108, while Tas1r2 was not detected. The majority (86%) of these tuft cells responded to exogenous glutamate, denatonium, and heat-killed uropathogenic E. coli. However, saccharin did not elicit any response in mice urethral tuft cells. Furthermore, under denatonium stimulation, acetylcholine released by rat urethral tuft cells acts on adjacent sensory nerve fibers to enhance bladder detrusor activity, aiding in the removal of harmful substances from the urethra and preventing bacterial colonization. Recently, through transcriptome analysis conducted by Deckmann’s group, it was discovered that both TAS1R2 and TAS1R3 are expressed in urethral tuft cells.19 By examining changes in intracellular Ca2+ concentrations in isolated mouse urethral tuft cells upon glucose stimulation, researchers observed that 78% of these cells responded to sucrose while 65% responded to fructose. To investigate the role of TAS1R3 in urine glucose recognition further, they compared wild-type mice with TAS1R3 knockout mice and found a significantly reduced proportion of responsive urethral tuft cells as well as diminished intracellular Ca2+ changes after sucrose stimulation in the latter group. Additionally, when treating mice with gurmarin, an inhibitor targeting mouse TAS1R3, there was a notable decrease in sucrose-induced responses among their urethral tuft cells. However, neither the knockout of the TAS1R3 gene nor the application of the inhibitor gurmarin could completely abolish the response of urethral tuft cells to sucrose, indicating that there exists an alternative “sweet” detection pathway in urethral tuft cells that is independent of TAS1R3. Similarly, in animal experiments, they discovered that transurethral instillation of sucrose and glucose could stimulate acetylcholine secretion by urethral tuft cells and significantly enhance bladder detrusor contractile activity in rats compared to saline. However, when combined with the TAS1R3 inhibitor gurmarin, sucrose and glucose did not enhance bladder detrusor contractile activity. This suggests that after sugar stimulation, urethral tuft cells enhance bladder detrusor activity in a manner dependent on TAS1R3. The increase in urine glucose concentration caused by diabetes is also a risk factor for urinary tract infections, and 50% of diabetic patients experience bladder dysfunction, cystopathy, and overactive bladder.135 These conditions may be associated with the glucose recognition mechanism of urethral tuft cells. Currently, it remains unclear how taste receptor activation promotes bladder contraction, and there are limited studies on the function of urethral tuft cells and taste GPCRs.
Metabolic diseases
Metabolic diseases refer to a broad category of disorders resulting from systemic metabolic dysfunctions rather than a single condition, including but not limited to type 2 diabetes (T2D) and obesity. Deregulated glucose homeostasis is one of the key pathogenic mechanisms underlying these diseases.136
Enteroendocrine cells (EECs) can sense glucose and other substances through taste GPCRs, thereby regulating the secretion of related hormones, maintaining glucose homeostasis, and contributing to the development of metabolic diseases. EECs can be classified into eight subtypes, commonly designated as S, I, L, K, D, N, A, and enterochromaffin cells.137 Notably, the A, K, and L cell subtypes can express receptors for sweet, bitter, and umami tastes.138 Glucagon-like peptide-1 (GLP-1) and GLP-2 are intestinal hormones secreted by L cells. GLP-1 enhances insulin secretion from the pancreas following oral glucose intake, whereas GLP-2 exerts its effects by facilitating intestinal glucose absorption.22,139 Therefore, GLP-1 receptor agonists and GLP-1 analogues have emerged as viable therapeutic options for diabetes and obesity management.140 In addition, ghrelin and cholecystokinin (CCK) are two important gastrointestinal hormones. Ghrelin accelerates gastric emptying and inhibits insulin secretion, while CCK regulates blood glucose levels by suppressing appetite and delaying gastric emptying.141 Glucose induces the secretion of GLP-1 by L cells, involving several glucose sensors, including sweet taste GPCRs, sodium-glucose cotransporter 1 (SGLT-1), and glucose transporter 2 (GLUT2). Huang et al. discovered that the effects of different glucose concentrations on STC-1 cell GLP-1 secretion and the protein levels of SGLT1, GLUT2, and sweet taste GPCRs were different, and they found that SGLT1 mainly participates in the induction of GLP-1 secretion by low glucose concentrations, while sweet taste GPCRs and GLUT2 mainly participate in the induction of GLP-1 secretion by high glucose concentrations.142 Margolskee et al. proved that TAS1R3 and gustducin are expressed in EECs and are the basis for sensing sugar and regulating SGLT1.23 They found that compared to low-sugar diet, wild-type mice had increased expression of SGLT1 mRNA in high-sugar diet, while the expression of SGLT1 mRNA in TAS1R3 and gustducin knockout mice was consistent with that of wild-type mice on low-sugar diet, suggesting that there is an independent pathway that maintains the basal expression of SGLT1 mRNA. The expression of GLP-1 in the human intestinal endocrine L cell line NCI-H716 was confirmed by immunofluorescence staining and RT-PCR. Additionally, relevant taste signaling components were detected within this cell line. Stimulation of NCI-H716 cells with sucrose, glucose, or sucralose resulted in a concentration-dependent release of GLP-1 into the culture medium. The application of Lactisole, a sweet taste inhibitor targeting TAS1R3, inhibited the reactive release of GLP-1 in response to sucralose and also reduced the expression of gustducin in NCI-H716 cells. It was observed that glucose-mediated secretion of GLP-1 was diminished. Thus, both TAS1R3 and gustducin are involved in mediating sweetener-induced secretion of GLP-1 from NCI-H716 cells.22 Smith et al. proposed that besides GLP-1, GLP-2 is also implicated in taste GPCRs-mediated glucose metabolism. The intestinal TAS1R2/TAS1R3 can enhance glucose absorption by activating GLUT2 transporters mediated by GLP-2, facilitating GLUT2 translocation to the apical membrane of enterocytes.139 Furthermore, they found reduced glucose absorption both in vivo in TAS1R2 knockout mice and ex vivo in intestinal organoids. However, the administration of teduglutide (a GLP-2 analog) completely restored intestinal glucose absorption. This indicates that GLP-2 acts as a downstream effector of TAS1R2 signaling. In conjunction with previous studies on GLP-1, they suggest that TAS1R2-mediated secretion of both GLP-1 and GLP-2 jointly regulates postprandial glucose levels. In addition, some preliminary studies suggest that the intestinal TAS2Rs may be involved in regulating glucose homeostasis. Researchers have proposed a relationship between TAS2Rs and the release of ghrelin, CCK, and GLP-1.143 For example, oral administration of bitter gourd extract to high-fat-fed mice was shown to stimulate GLP-1 and insulin secretion following either oral or intraperitoneal glucose injection, resulting in reduced postprandial blood glucose levels. This effect could be inhibited by the specific GLP-1 receptor antagonist exendin-9.144 The in vitro induction of GLP-1 secretion by STC-1 induced by bitter gourd extract was partially abolished by TAS2R inhibitor (probenecid) and PLCβ2 inhibitor (U-73122).144
The pancreas also expresses sweet taste GPCRs as sweetness sensors, which may regulate the release of insulin as an independent signaling pathway from nutrient metabolism.145,146 The expression and function of sweet taste GPCRs in the β cells play an important role in regulating basal insulin release, especially in regulating basal insulin release in the short-term fasting glucose concentration, sweet taste GPCRs sense the glucose and inhibit insulin secretion over time. On the contrary, in diabetic and diet-induced obese mouse models, the expression and function of sweet taste GPCRs are reduced, leading to an increase in basal insulin, which can offset the peripheral insulin resistance during the development of diabetes.145 There is a difference among researchers on whether the sweet taste GPCRs in the pancreas function as typical TAS1R2/TAS1R3 heterodimers or TAS1R3 homodimers. The Kojima research group discovered that the mRNA and protein levels of TAS1R3 in mouse pancreatic tissue and MIN6 cells were significantly higher than those of TAS1R2, with the mRNA level of TAS1R2 being less than 1% of that of TAS1R3. Immunofluorescence analysis was unable to detect any presence of TAS1R2 protein. Following the knockout of TAS1R3, artificial sweeteners failed to further enhance glucose-induced insulin secretion in MIN6 cells. However, the knockout of TAS1R2 did not have a significant impact on this process.147,148,149 Consequently, they proposed that the homologous dimerization of TAS1R3 may function as a glucose sensor on the surface of pancreatic β cells. Conversely, other researchers hold differing views. Kyriazis et al. identified expression for both TAS1R2 and TAS1R3 in human pancreatic islets and their role in mediating fructose’s enhancement effect on insulin secretion. In β cells from mice lacking TAS1R2, fructose-induced calcium influx and insulin release were diminished. Therefore, they posited that the heterodimeric structure formed by TAS1R2/TAS1R3 plays a critical role within β cells.145,146 The expression of TAS2Rs in β cells and their potential role in regulating insulin release remains an underexplored area of research. Some studies have indicated that denatonium, can stimulate insulin secretion from rat pancreatic islets. However, the effects of denatonium on β cells do not appear to be mediated by the activation of TAS2R receptors or gustducin.150 Current investigations into the expression and function of taste GPCRs in the pancreas predominantly focus on animal models and cellular experiments. The implications of these findings for human physiology remain uncertain. Therefore, further research is necessary to elucidate the role of taste GPCRs-mediated regulation of insulin release within human physiological contexts and its interactions with other established mechanisms governing insulin regulation.
Diabetes mellitus is a complicated endocrine disease characterized by hyperglycemia.151 A study has indicated that the expression of intestinal sweet taste GPCRs in T2D patients is negatively correlated with fasting blood glucose levels.139 However, it should be noted that under conditions of rapid fluctuations in luminal glucose levels, sweet taste GPCRs expression may be influenced. Yang et al. employed a randomized crossover design involving healthy subjects and T2D patients and found that the absolute transcription levels of sweet taste GPCRs were not significantly affected by acute changes in fasting blood glucose but were responsive to luminal glucose exposure.152 Specifically, normal glycemic conditions exposed to luminal glucose resulted in increased TAS1R2 expression. Conversely, hyperglycemic states led to decreased TAS1R2 expression. In contrast, exposure to luminal glucose in T2D patients resulted in elevated TAS1R2 expression. This suggests that dysregulation of sweet taste GPCRs expression may contribute to persistent postprandial hyperglycemia observed in individuals with T2D. Quinoa has been reported to possess notable glucose-lowering properties. To explore its mechanism of action, researchers administered quinoa to T2D mouse models induced by a high-fat diet (HFD) combined with streptozotocin (STZ). They observed that compared to normal control mice, T2D mice exhibited increased expression of TAS1R3 and TRPM5 in the colon, while GLP-1 expression was reduced. Both quinoa and metformin were able to reverse these changes. Thus, they hypothesized that one mechanism through which quinoa ameliorates hyperglycemia is via nutrient sensing through taste GPCRs.153 Furthermore, there is evidence indicating potential alterations in bitter taste receptor expression among diabetic patients as well. Bartakova et al. compared the genotypes related to bitter taste receptors between pregnant women with gestational diabetes and healthy pregnant controls and found a lower frequency of the TAS2R9 rs3741845 allele among those with gestational diabetes.154 Also, a study has suggested that the rs3741845 SNP of TAS2R9 results in an alanine-to-valine substitution at position 187 (Ala187Val) in the predicted ligand-binding pocket, leading to a defective response to certain bitter ligands and subsequently affecting glucose homeostasis.155
It has been reported that there is a notable correlation between obesity and taste GPCRs. A recent review elucidates how individuals can consume sweet substances, triggering feelings of satisfaction through central reward pathways.156 The authors emphasize that sugar may induce addiction similarly to nicotine, leading to dulled taste perception among obese populations. Furthermore, they found that inhibiting sweet or umami perception could facilitate weight loss. Interestingly, the authors note that while non-nutritive sweeteners may result in short-term weight reduction, the prevalence of obesity and metabolic dysfunction continues to rise. This trend may be associated with an increased risk of disease linked to the use of non-nutritive sweeteners. Additionally, previous research by Liszt et al. indicates that obesity alters TAS2R expression in intestinal cells in a region-dependent manner and reduces EECs sensitivity to TAS2R agonist denatonium benzoate concerning ghrelin expression.157 Obesity may also influence TAS2R expression and sensitivity in other cell types, such as Paneth cells and goblet cells.
In conclusion, there exists a significant relationship between digestive system taste GPCRs and metabolic diseases. An increasing body of evidence suggests that taste GPCRs are substantially involved in nutrient sensing, monitoring changes in energy storage, and initiating metabolic and behavioral responses essential for maintaining energy balance. By targeting taste GPCRs, novel drugs with specificity and efficacy can be developed for improved management of body weight and glucose homeostasis.
Asthma
Airway smooth muscle cells (ASM) express a variety of TAS2Rs. Interestingly, under the stimulation of bitter compounds, ASM cells exhibit severe relaxation instead of contraction. This implies that bitter taste receptor agonists act as bronchodilators and are three times more effective than β-adrenergic receptor agonists.50 The underlying mechanisms include: 1) TAS2R-mediated increase in intracellular Ca2+, accompanied by BKCa channel-mediated increase in Ca2+-dependent K+ conductance, leading to membrane hyperpolarization and relaxation.50 2) Separation of the Gβγ subunit, thus inhibiting the voltage-dependent Ca2+ channels (VDCC).51 3) Dissociation of TAS2R-coupled G protein Gαt from the Tas2R-Gαt/Gβγ complex, with the released Gαt binding to acetylcholine receptors (AChR), competitively inhibiting the function of coupled G protein Gq and preventing AChR-induced Ca2+ signaling.52 4) Inhibition of RhoA activity and myosin light chain phosphatase (MYPT)1 phosphorylation at the T853 residue modulating histamine-induced myosin light chain (MLC)20 phosphorylation, leading to relaxation of contracted ASM.53 In addition to these mechanisms, TAS2R agonists have concentration-dependent anti-mitogenic effects on ASM and can treat asthma by alleviating ASM remodeling.158 Furthermore, TAS2R agonists also regulate immune cells during asthma. For example, they inhibit pro-inflammatory factor release by macrophages159 and polarize them towards M2 type.160 Additionally, TAS2R agonists like denatonium and chloroquine can inhibit IgE-dependent mast cell activation, thereby alleviating asthma symptoms.161 Therefore, TAS2Rs are considered potential targets for anti-asthma therapy. However, it is important to note that in an animal study, researchers observed no significant difference in bronchodilation induced by bitter tastants between wild-type mice and their corresponding TAS2R knockout counterparts (Tas2r143/Tas2r135/Tas2r126 triple knockout).162 This finding suggests that the mechanism of bronchodilation mediated by bitter tastants in mice may operate independently of TAS2Rs.
However, the lack of tested safe and effective agonists that act at low effective concentrations has hindered the clinical approval of TAS2R-based agents for asthma treatment. Nevertheless, certain drugs approved for asthma treatment, such as the macrolide antibiotic azithromycin,112 the anti-inflammatory analgesic flufenamic acid, and the antihistamine diphenhyramine,163 have shown potential as TAS2R ligands. A recent study utilizing BitterDB database identified flufenamic acid as a highly effective compound capable of rapidly activating TAS2R14 and leading to ASM relaxation at a low concentration (1 μM).164 Of note, as aforementioned, the protein structures of two complexes formed by TAS2R14 with gustducin and Gi1 were resolved, providing insights into ligand recognition of TAS2Rs and laying the foundation for future development of TAS2R-based anti-asthma drugs.85,86
Reproductive dysfunction
In the male reproductive system, taste GPCRs may play a crucial role in sperm chemotaxis, fertility, and sterility.165 As early as 2001, Max et al. identified TAS1R3 expressed in mouse testes.5 Subsequently, researchers discovered gustducin, a key component of taste transduction, in mammalian spermatozoa.166 Mosinger et al. discovered that both genetic deletion and pharmacological blockade of the TAS1R3 receptor result in reproductive disorders in male mice. These alterations include decreased sperm count, abnormal sperm morphology, and impaired sperm motility within the testes, highlighting the crucial involvement of TAS1R3 in spermatogenesis and sperm function.167 Moreover, bitter tastants such as chemokines or progesterone secreted by cumulus–oocyte complex have the potential to activate TAS2Rs in spermatozoa and promote sperm chemotaxis, capacitation, and acrosomal reactions.168 Although these studies indicate the presence of taste GPCRs in the reproductive system and suggest their potential role in reproductive function, there is currently no clear evidence regarding endogenous ligands under physiological conditions, and a lack of comprehensive functional studies to determine their role under such conditions. Li and Zhou found that the absence of TAS2R5+ cells resulted in testicular shrinkage and male infertility in mice.169 Additionally, a study demonstrated that non-synonymous SNPs of TAS2R14 and TAS2R3 were associated with decreased sperm motility and structural abnormalities.170 Polycystic ovary syndrome (PCOS) is one of the most prevalent reproductive disorders among women. Wu et al. discovered that using agonists for Tas2r108 effectively improved endocrine and metabolic disorders associated with PCOS while promoting recovery of reproductive function.171 Currently, further exploration is needed to understand the mechanism behind taste GPCRs’ involvement in reproductive dysfunction.
Neurodegenerative diseases
Taste receptors, including TAS1Rs, TAS2Rs, and their downstream effector proteins, have been detected in various regions of the central nervous system (CNS), suggesting a neuromodulatory role beyond gustation.172,173,174 Many neurodegenerative disorders, such as Alzheimer’s disease (AD) and Parkinson’s disease (PD), share overlapping mechanisms, including oxidative stress, mitochondrial dysfunction, neuroinflammation, and protein misfolding.175 Under normal conditions, TAS2Rs-mediated sensing of pathogenic, toxicogenic, and microbial molecules in the brain is crucial for preventing neuroinflammatory responses and supporting immune system function.26 Accordingly, dysfunctional signaling or downregulation of key components of the taste signaling pathway has been associated with the cellular damage characteristic of neurodegenerative diseases. Although sweet receptors were once believed to function primarily in learning and memory, recent evidence indicates that disruptions in sweet taste receptor signaling are also linked to inflammatory responses.176,177,178 Thus, sweet taste receptor signaling may contribute to neuroinflammation as well.
Alzheimer’s disease
AD is one of the most common neurodegenerative disorders, characterized by progressive and irreversible cognitive decline, ultimately compromising patient independence.179 In AD, pathophysiological changes involving protein alterations lead to oxidative stress, dysfunction across multiple cellular pathways and cell death. Although many studies describe the neuroprotective effects of bitter compounds in AD, the role of TAS2Rs in mediating these effects remains unclear. For example, bitter compounds have been reported to reduce apoptosis, inflammation, and oxidative stress by regulating inflammatory markers (TNF-α, IL-1β, iNOS) and antioxidant enzymes (superoxide dismutase, glutathione, catalase).180,181,182,183 Alves et al. reported a correlation between TAS2Rs expression and AD progression: TAS2Rs genes, along with other AD-associated genes, were progressively downregulated from the early stages of AD, with continued decline as the disease advanced.184 These findings suggest that TAS2Rs may serve as both potential biomarkers and therapeutic targets, warranting further investigation.
Parkinson’s disease
PD, the second most prevalent neurodegenerative disorder after AD, results from the progressive loss of dopaminergic neurons in the substantia nigra and striatum of the midbrain.185 This neuronal degeneration is driven by α-synuclein accumulation, which triggers neuroinflammation and neurodegeneration in PD by inducing degrading enzymes, inflammatory mediators and NLRP3 inflammasome activation.186 NF-κB and the NLRP3 inflammasome are central regulators of inflammatory networks and contribute to both neuroinflammatory processes and TAS2Rs signaling dysregulation.187,188 Defects in TAS2Rs signaling can lead to the overproduction of pro-inflammatory cytokines such as IL-1β, which further activate NF-κB via Toll-like receptor pathways.189 Certain TAS2Rs agonists have been shown to mitigate lipopolysaccharide (LPS)-induced neuroinflammation, oxidative stress, and apoptosis by suppressing NLRP3 and NF-κB signaling.190,191,192,193,194 These findings suggest a potential role for TAS2Rs in mediating the protective effects of their ligands for treating CNS disorders. Furthermore, TAS2Rs expression has been reported to change in PD patients, with TAS2R5 and TAS2R50 downregulated, while TAS2R10 and TAS2R13 are upregulated.195 Despite these researches, further research is needed to clarify the specific roles of TAS2Rs in PD pathophysiology.
Cancers
The expression of taste GPCRs has been detected in various types of tumor cells and is differentially expressed in tumor tissues. Ryan M et al. conducted an analysis on the expression of TAS1Rs and TAS2Rs in 45 types of solid tumors using data from TCGA, revealing that compared to normal tissues, TAS2R4, TAS2R5, TAS2R14, TAS2R19, TAS2R20, and TAS2R31 are generally downregulated in tumors while TAS1R3 and TAS2R38 are usually upregulated.196 Furthermore, the correlation between taste GPCR expression levels and patient prognosis varies among different tumors. For instance, increased expression of TAS2R14 is associated with improved survival in non-papillary bladder cancer but decreased survival in adrenal cortex carcinoma and esophageal adenocarcinoma.196 Therefore, taste GPCRs hold promise for tumor prognosis analysis and therapy. A study utilizing data from TCGA and GEO databases identified 7 TAS2Rs that are linked to the survival of colon cancer patients, leading to the definition of two molecular subtypes of colon cancer.197 These subtypes exhibit low immune cell infiltration and poor prognosis when high levels of TAS2Rs are expressed, suggesting a potential association between TAS2Rs and tumor immune invasion to some extent. Moreover, the activation of pancreatic cancer cells TAS1R2/TAS1R3 by natural or artificial sweeteners can induce transactivation of glycosylated receptors, leading to the manifestation of tumor metastasis phenotypes such as epithelial mesenchymal transformation and enhanced cell migration, suggesting that the activation of sweet receptors in pancreatic cancer cells may be associated with cancer metastasis.198 In contrast, Seo et al. demonstrated that TAS2R8 and TAS2R10, endogenously expressed in neuroblastoma (NB) cancer cells, play significant roles in suppressing cancer stemness, migration, and invasion by reducing matrix metalloproteinase-2 (MMP-2) activity, dampening hypoxia-related programs, and downregulating cancer stem cell (CSC) markers.199 These findings are consistent with research by Wölfle’s team, which showed that salicin, a TAS2R16 agonist, promotes neurite outgrowth through bitter taste receptor activation.200 Additionally, a study has also demonstrated that gustducin can play a role in inhibiting pancreatic cancer.201 Knockout of gustducin increases the expression of CXCL1 and CXCL2 in epithelial cells, thereby increasing the number of CXCR2+ myeloid suppressor cells.201
Moreover, taste GPCRs offer novel targets for cancer treatment. For instance, the TAS2R14 agonist noskapine and diphenhydramine have demonstrated their ability to induce the pro-apoptotic effect in ovarian cancer cell line SKOV3.202 Kaempferol is also recognized as a ligand of TAS2R14 and TAS2R39, which promotes apoptosis in human cervical cancer cells through the PI3K/AKT and telomerase pathways or in ovarian cancer cells via the death receptor/Fas associated death domain (FADD)/caspase-8 pathways.203,204 In head and neck squamous cell carcinoma (HNSCC), particularly among patients with HPV infection, elevated expression of TAS2R14 has been observed. Lidocaine has been found to activate TAS2R14 and induce apoptosis of HNSCC cancer cells, thereby exhibiting therapeutic potential.205 Furthermore, it can enhance the apoptotic effect when combined with a GLUT1 inhibitor.206 Salvestrini et al. extended the relevance of taste GPCRs from solid tumors to hematologic malignancies. In their study, TAS2Rs expressed in AML cells were activated by denatonium (DEN), leading to suppressed proliferation and clonogenicity, induction of apoptosis, reduced migration, and altered metabolism. DEN exerted these effects by activating 9 out of 25 TAS2Rs, five of which (TAS2R4, TAS2R8, TAS2R10, TAS2R13, and TAS2R30/47) were also expressed at protein level in both AML patient samples and AML cell lines.207 Therefore, taste receptor agonists may serve as promising avenues for cancer therapy. Additionally, overexpressed taste receptors offer new targets for drug design. Jessica et al. designed a TAS2R9-targeted liposome specifically aimed at delivering CXCR2 inhibitors to cancer-related fibroblasts within pancreatic ductal adenocarcinoma tissue to achieve therapeutic effects.208 However, these studies still lack specific genetic evidence that definitively elucidates the role of taste receptors in cancer research such as utilizing cell-specific knockout mice, thus they remain at an early stage of investigation with considerable distance from clinical drug development.
Conclusion
Taste GPCRs, particularly TAS1Rs and TAS2Rs, play multifaceted roles beyond taste perception, influencing metabolism, immunity, and cellular signaling across various tissues. Their dysfunctions and polymorphisms are closely linked to the pathogenesis of diseases such as metabolic disorders, respiratory infections, dental caries, and certain cancers. Advances in understanding their signaling mechanisms and structural characteristics have revealed critical insights into their physiological and pathological roles, highlighting their potential as therapeutic targets. This review underscores the importance of taste GPCRs as regulators of systemic processes and advocates for further exploration of their functions in non-taste tissues. Despite challenges in resolving their structural complexity and understanding their interactions in diverse physiological contexts, ongoing research and technological advancements offer new opportunities for therapeutic innovation. By targeting these receptors, it may be possible to develop novel treatments for a range of diseases, from metabolic and immune disorders to cancer. Continued investigation into the molecular mechanisms of taste GPCRs will undoubtedly deepen our understanding of their contributions to health and disease.
References
Yarmolinsky, D. A., Zuker, C. S. & Ryba, N. J. Common sense about taste: from mammals to insects. Cell 139, 234–244 (2009).
Breslin, P. A. An evolutionary perspective on food and human taste. Curr. Biol. 23, R409–R418 (2013).
Hoon, M. A. et al. Putative mammalian taste receptors: a class of taste-specific GPCRs with distinct topographic selectivity. Cell 96, 541–551 (1999).
Nelson, G. et al. Mammalian sweet taste receptors. Cell 106, 381–390 (2001).
Max, M. et al. Tas1r3, encoding a new candidate taste receptor, is allelic to the sweet responsiveness locus Sac. Nat. Genet. 28, 58–63 (2001).
Kitagawa, M., Kusakabe, Y., Miura, H., Ninomiya, Y. & Hino, A. Molecular genetic identification of a candidate receptor gene for sweet taste. Biochem. Biophys. Res. Commun. 283, 236–242 (2001).
Sainz, E., Korley, J. N., Battey, J. F. & Sullivan, S. L. Identification of a novel member of the T1R family of putative taste receptors. J. Neurochem. 77, 896–903 (2001).
Montmayeur, J. P., Liberles, S. D., Matsunami, H. & Buck, L. B. A candidate taste receptor gene near a sweet taste locus. Nat. Neurosci. 4, 492–498 (2001).
Bachmanov, A. A. et al. Positional cloning of the mouse saccharin preference (Sac) locus. Chem. Senses 26, 925–933 (2001).
Munk, C. et al. GPCRdb: the G protein-coupled receptor database - an introduction. Br. J. Pharm. 173, 2195–2207 (2016).
Schiöth, H. B. & Fredriksson, R. The GRAFS classification system of G-protein coupled receptors in comparative perspective. Gen. Comp. Endocrinol. 142, 94–101 (2005).
Liu, D. & Liman, E. R. Intracellular Ca2+ and the phospholipid PIP2 regulate the taste transduction ion channel TRPM5. Proc. Natl. Acad. Sci. USA 100, 15160–15165 (2003).
Dutta Banik, D., Martin, L. E., Freichel, M., Torregrossa, A. M. & Medler, K. F. TRPM4 and TRPM5 are both required for normal signaling in taste receptor cells. Proc. Natl. Acad. Sci. USA 115, E772–e781 (2018).
Tizzano, M. et al. Nasal chemosensory cells use bitter taste signaling to detect irritants and bacterial signals. Proc. Natl. Acad. Sci. USA 107, 3210–3215 (2010).
Lee, R. J. et al. Bitter and sweet taste receptors regulate human upper respiratory innate immunity. J. Clin. Invest 124, 1393–1405 (2014).
Shah, A. S., Ben-Shahar, Y., Moninger, T. O., Kline, J. N. & Welsh, M. J. Motile cilia of human airway epithelia are chemosensory. Science 325, 1131–1134 (2009).
Schneider, C., O’Leary, C. E. & Locksley, R. M. Regulation of immune responses by tuft cells. Nat. Rev. Immunol. 19, 584–593 (2019).
Deckmann, K. et al. Bitter triggers acetylcholine release from polymodal urethral chemosensory cells and bladder reflexes. Proc. Natl. Acad. Sci. USA 111, 8287–8292 (2014).
Schmidt, P. et al. Tas1R3 dependent and independent recognition of sugars in the urethra and the role of tuft cells in this process. Adv. Biol. 8, e2400117 (2024).
Xi, R., Zheng, X. & Tizzano, M. Role of taste receptors in innate immunity and oral health. J. Dent. Res 101, 759–768 (2022).
Dong, H. et al. Oral microbiota-host interaction mediated by taste receptors. Front Cell Infect. Microbiol 12, 802504 (2022).
Jang, H. J. et al. Gut-expressed gustducin and taste receptors regulate secretion of glucagon-like peptide-1. Proc. Natl. Acad. Sci. USA 104, 15069–15074 (2007).
Margolskee, R. F. et al. T1R3 and gustducin in gut sense sugars to regulate expression of Na+-glucose cotransporter 1. Proc. Natl. Acad. Sci. USA 104, 15075–15080 (2007).
Roura, E. et al. Taste and hypertension in humans: targeting cardiovascular disease. Curr. Pharm. Des. 22, 2290–2305 (2016).
Yu, Q. et al. Bitter taste cells in the ventricular walls of the murine brain regulate glucose homeostasis. Nat. Commun. 14, 1588 (2023).
Singh, N., Vrontakis, M., Parkinson, F. & Chelikani, P. Functional bitter taste receptors are expressed in brain cells. Biochem. Biophys. Res Commun. 406, 146–151 (2011).
Lee, S. J., Depoortere, I. & Hatt, H. Therapeutic potential of ectopic olfactory and taste receptors. Nat. Rev. Drug Discov. 18, 116–138 (2019).
Lu, P., Zhang, C. H., Lifshitz, L. M. & ZhuGe, R. Extraoral bitter taste receptors in health and disease. J. Gen. Physiol. 149, 181–197 (2017).
Kinnamon, S. C. Taste receptor signalling—from tongues to lungs. Acta Physiol. 204, 158–168 (2012).
Sanematsu, K., Yoshida, R., Shigemura, N. & Ninomiya, Y. Structure, function, and signaling of taste G-protein-coupled receptors. Curr. Pharm. Biotechnol. 15, 951–961 (2014).
Huang, L. et al. Ggamma13 colocalizes with gustducin in taste receptor cells and mediates IP3 responses to bitter denatonium. Nat. Neurosci. 2, 1055–1062 (1999).
Rössler, P. et al. G protein betagamma complexes in circumvallate taste cells involved in bitter transduction. Chem. Senses 25, 413–421 (2000).
Ahmad, R. & Dalziel, J. E. G protein-coupled receptors in taste physiology and pharmacology. Front. Pharmacol. 11, 587664 (2020).
Hisatsune, C. et al. Abnormal taste perception in mice lacking the type 3 inositol 1,4,5-trisphosphate receptor. J. Biol. Chem. 282, 37225–37231 (2007).
Romanov, R. A. et al. Chemical synapses without synaptic vesicles: Purinergic neurotransmission through a CALHM1 channel-mitochondrial signaling complex. Sci. Signal 11, https://doi.org/10.1126/scisignal.aao1815 (2018).
Ma, Z. et al. CALHM3 Is Essential for Rapid Ion Channel-Mediated Purinergic Neurotransmission of GPCR-Mediated Tastes. Neuron 98, 547–561 e510 (2018).
Vandenbeuch, A. et al. Postsynaptic P2X3-containing receptors in gustatory nerve fibres mediate responses to all taste qualities in mice. J. Physiol. 593, 1113–1125 (2015).
Wang, P. et al. Recent advancements in the taste transduction mechanism, identification, and characterization of taste components. Food Chem. 433, 137282 (2024).
Avenet, P., Hofmann, F. & Lindemann, B. Transduction in taste receptor cells requires cAMP-dependent protein kinase. Nature 331, 351–354 (1988).
Béhé, P., DeSimone, J. A., Avenet, P. & Lindemann, B. Membrane currents in taste cells of the rat fungiform papilla. Evidence for two types of Ca currents and inhibition of K currents by saccharin. J. Gen. Physiol. 96, 1061–1084 (1990).
Varkevisser, B. & Kinnamon, S. C. Sweet taste transduction in hamster: role of protein kinases. J. Neurophysiol. 83, 2526–2532 (2000).
Kinnamon, S. C. Umami taste transduction mechanisms. Am. J. Clin. Nutr. 90, 753s–755s (2009).
He, W. et al. Umami taste responses are mediated by alpha-transducin and alpha-gustducin. J. Neurosci. Off. J. Soc. Neurosci. 24, 7674–7680 (2004).
Ruiz-Avila, L. et al. Coupling of bitter receptor to phosphodiesterase through transducin in taste receptor cells. Nature 376, 80–85 (1995).
Yan, W. et al. Bitter taste transduced by PLC-β2-dependent rise in IP3 and α-gustducin-dependent fall in cyclic nucleotides. 280, C742−C751, (2001).
Yan, W. et al. Bitter taste transduced by PLC-beta(2)-dependent rise in IP(3) and alpha-gustducin-dependent fall in cyclic nucleotides. Am. J. Physiol. Cell Physiol. 280, C742–C751 (2001).
Clapp, T. R. et al. Tonic activity of Galpha-gustducin regulates taste cell responsivity. FEBS Lett. 582, 3783–3787 (2008).
Luo, X. C. et al. Infection by the parasitic helminth Trichinella spiralis activates a Tas2r-mediated signaling pathway in intestinal tuft cells. Proc. Natl. Acad. Sci. USA 116, 5564–5569 (2019).
Singh, N. et al. Bitter taste receptor T2R14-Gαi coupling mediates innate immune responses to microbial quorum sensing molecules in cystic fibrosis. iScience 27, 111286 (2024).
Deshpande, D. A. et al. Bitter taste receptors on airway smooth muscle bronchodilate by localized calcium signaling and reverse obstruction. Nat. Med. 16, 1299–1304 (2010).
Zhang, C. H. et al. The cellular and molecular basis of bitter tastant-induced bronchodilation. PLoS Biol. 11, e1001501 (2013).
Zhou, Y. W. et al. Tas2R activation relaxes airway smooth muscle by release of Galpha(t) targeting on AChR signaling. Proc. Natl. Acad. Sci. USA 119, e2121513119 (2022).
Conaway, S. Jr et al. Molecular mechanism of bitter taste receptor agonist-mediated relaxation of airway smooth muscle. Faseb J. 38, e23842 (2024).
Kuhn, C. & Meyerhof, W. Oligomerization of sweet and bitter taste receptors. Methods Cell Biol. 117, 229–242 (2013).
Li, X. et al. Human receptors for sweet and umami taste. Proc. Natl. Acad. Sci. USA 99, 4692–4696 (2002).
Jiang, P. et al. Molecular mechanisms of sweet receptor function. Chem. Senses 30, i17–i18 (2005).
Xu, H. et al. Different functional roles of T1R subunits in the heteromeric taste receptors. Proc. Natl. Acad. Sci. USA 101, 14258–14263 (2004).
Masuda, K. et al. Characterization of the modes of binding between human sweet taste receptor and low-molecular-weight sweet compounds. PLoS One 7, e35380 (2012).
Jiang, P. et al. Identification of the cyclamate interaction site within the transmembrane domain of the human sweet taste receptor subunit T1R3. J. Biol. Chem. 280, 34296–34305 (2005).
Sun, Y. et al. Comprehensive new insights into sweet taste transmission mechanisms and detection methods. Foods 14, https://doi.org/10.3390/foods14132397 (2025).
Cai, C. et al. Characterization of the sweet taste receptor Tas1r2 from an Old World monkey species rhesus monkey and species-dependent activation of the monomeric receptor by an intense sweetener perillartine. PLoS One 11, e0160079 (2016).
Zhao, M., Xu, X. Q., Meng, X. Y. & Liu, B. The heptahelical domain of the sweet taste receptor T1R2 is a new allosteric binding site for the sweet taste modulator amiloride that modulates sweet taste in a species-dependent manner. J. Mol. Neurosci. 66, 207–213 (2018).
Zhao, G. Q. et al. The receptors for mammalian sweet and umami taste. Cell 115, 255–266 (2003).
Jiang, P. et al. The cysteine-rich region of T1R3 determines responses to intensely sweet proteins. J. Biol. Chem. 279, 45068–45075 (2004).
Zhang, F. et al. Molecular mechanism of the sweet taste enhancers. Proc. Natl. Acad. Sci. USA 107, 4752–4757 (2010).
Nie, Y. et al. Expression and purification of functional ligand-binding domains of T1R3 taste receptors. Chem. senses 31, 505–513 (2006).
Cheng, L. et al. Structure, function and drug discovery of GPCR signaling. Mol. Biomed. 4, 46 (2023).
Li, X. et al. Heterologous expression and purification of GPCRs. Methods Mol. Biol. 2507, 295–312 (2022).
Norskov-Lauritsen, L. & Brauner-Osborne, H. Role of post-translational modifications on structure, function and pharmacology of class C G protein-coupled receptors. Eur. J. Pharm. 763, 233–240 (2015).
Lavington, S. & Watts, A. Lipid nanoparticle technologies for the study of G protein-coupled receptors in lipid environments. Biophys. Rev. 12, 1287–1302 (2020).
Juen, Z. et al. The structure of human sweetness. Cell 188, 4141–4153.e4118 (2025).
Shi, Z. et al. Structural and functional characterization of human sweet taste receptor. Nature https://doi.org/10.1038/s41586-025-09302-6 (2025).
Wang, H. et al. Structure and activation mechanism of human sweet taste receptor. Cell Res. https://doi.org/10.1038/s41422-025-01156-x (2025).
Lang, T., Di Pizio, A., Risso, D., Drayna, D. & Behrens, M. Activation Profile of TAS2R2, the 26th human bitter taste receptor. 67, 2200775, (2023).
Di Pizio, A. A bitter taste receptor activated in a surprising way. Nature 628, 506−507, (2024).
Ziaikin, E., David, M., Uspenskaya, S. & Niv, M. Y. BitterDB: 2024 update on bitter ligands and taste receptors. Nucleic Acids Res. 53, D1645–d1650 (2025).
Behrens, M. & Ziegler, F. Structure-function analyses of human bitter taste receptors-where do we stand? Molecules 25, https://doi.org/10.3390/molecules25194423 (2020).
Brockhoff, A., Behrens, M., Niv, M. Y. & Meyerhof, W. Structural requirements of bitter taste receptor activation. Proc. Natl. Acad. Sci. USA 107, 11110–11115 (2010).
Nowak, S. et al. Reengineering the ligand sensitivity of the broadly tuned human bitter taste receptor TAS2R14. Biochim. Biophys. Acta Gen. Subj. 1862, 2162–2173 (2018).
Singh, N., Pydi, S. P., Upadhyaya, J. & Chelikani, P. Structural basis of activation of bitter taste receptor T2R1 and comparison with Class A G-protein-coupled receptors (GPCRs). J. Biol. Chem. 286, 36032–36041 (2011).
Pydi, S. P., Bhullar, R. P. & Chelikani, P. Constitutively active mutant gives novel insights into the mechanism of bitter taste receptor activation. 122, 537−544, (2012).
Pydi, S. P., Singh, N., Upadhyaya, J., Bhullar, R. P. & Chelikani, P. The third intracellular loop plays a critical role in bitter taste receptor activation. Biochim. Biophys. Acta 1838, 231–236 (2014).
Pydi, S. P. et al. Cholesterol modulates bitter taste receptor function. Biochim. Biophys. Acta 1858, 2081–2087 (2016).
Mueller, K. L. et al. The receptors and coding logic for bitter taste. Nature 434, 225–229 (2005).
Kim, Y. et al. Bitter taste receptor activation by cholesterol and an intracellular tastant. Nature 628, 664–671 (2024).
Hu, X. et al. Bitter taste TAS2R14 activation by intracellular tastants and cholesterol. Nature 631, 459–466 (2024).
Tao, L. et al. Bitter taste receptor TAS2R14 activation and G protein assembly by an intracellular agonist. Cell Res. 34, 735–738 (2024).
Peri, L. et al. A bitter anti-inflammatory drug binds at two distinct sites of a human bitter taste GPCR. Nat. Commun. 15, 9991 (2024).
Xu, W. et al. Structural basis for strychnine activation of human bitter taste receptor TAS2R46. Science 377, 1298–1304 (2022).
Srivastava, A. et al. High-resolution structure of the human GPR40 receptor bound to allosteric agonist TAK-875. Nature 513, 124–127 (2014).
Claff, T. et al. Structural basis for lipid-mediated activation of G protein-coupled receptor GPR55. Nat. Commun. 16, 1973 (2025).
Priestap, H. A., de los Santos, C. & Quirke, J. M. Identification of a reduction product of aristolochic acid: implications for the metabolic activation of carcinogenic aristolochic acid. J. Nat. Products 73, 1979–1986 (2010).
Waterloo, L. et al. Discovery of 2-Aminopyrimidines as Potent Agonists for the Bitter Taste Receptor TAS2R14. J. Med. Chem. 66, 3499–3521 (2023).
Vitaku, E., Smith, D. T. & Njardarson, J. T. Analysis of the structural diversity, substitution patterns, and frequency of nitrogen heterocycles among U.S. FDA approved pharmaceuticals. J. Med. Chem. 57, 10257–10274 (2014).
Brockhoff, A., Behrens, M., Massarotti, A., Appendino, G. & Meyerhof, W. Broad tuning of the human bitter taste receptor hTAS2R46 to various sesquiterpene lactones, clerodane and labdane diterpenoids, strychnine, and denatonium. J. Agric. Food Chem. 55, 6236–6243 (2007).
Jayasinghe, S. N. et al. Is sweet taste perception associated with sweet food liking and intake? Nutrients 9, https://doi.org/10.3390/nu9070750 (2017).
Haznedaroglu, E. et al. Association of sweet taste receptor gene polymorphisms with dental caries experience in school children. Caries Res. 49, 275–281 (2015).
Kulkarni, G. V. et al. Association of GLUT2 and TAS1R2 genotypes with risk for dental caries. Caries Res. 47, 219–225 (2013).
Eny, K. M., Wolever, T. M., Corey, P. N. & El-Sohemy, A. Genetic variation in TAS1R2 (Ile191Val) is associated with consumption of sugars in overweight and obese individuals in 2 distinct populations. Am. J. Clin. Nutr. 92, 1501–1510 (2010).
Ramos-Lopez, O., Panduro, A., Martinez-Lopez, E. & Roman, S. Sweet taste receptor TAS1R2 polymorphism (Val191Val) is associated with a higher carbohydrate intake and hypertriglyceridemia among the population of West Mexico. Nutrients 8, 101 (2016).
Izakovicova Holla, L. et al. GLUT2 and TAS1R2 polymorphisms and susceptibility to dental caries. Caries Res. 49, 417–424 (2015).
Fushan, A. A., Simons, C. T., Slack, J. P., Manichaikul, A. & Drayna, D. Allelic polymorphism within the TAS1R3 promoter is associated with human taste sensitivity to sucrose. Curr. Biol. 19, 1288–1293 (2009).
Bufe, B. et al. The molecular basis of individual differences in phenylthiocarbamide and propylthiouracil bitterness perception. Curr. Biol. 15, 322–327 (2005).
Kim, U. K. et al. Positional cloning of the human quantitative trait locus underlying taste sensitivity to phenylthiocarbamide. Science 299, 1221–1225 (2003).
Gil, S. et al. Genotype-specific regulation of oral innate immunity by T2R38 taste receptor. Mol. Immunol. 68, 663–670 (2015).
Medapati, M. R. et al. Bitter taste receptor T2R14 modulates gram-positive bacterial internalization and survival in gingival epithelial cells. Int. J. Mol. Sci. 22, https://doi.org/10.3390/ijms22189920 (2021).
Zheng, X. et al. Gingival solitary chemosensory cells are immune sentinels for periodontitis. Nat. Commun. 10, 4496 (2019).
Williams, D. W. et al. Human oral mucosa cell atlas reveals a stromal-neutrophil axis regulating tissue immunity. Cell 184, 4090–4104 e4015 (2021).
Huang, N. et al. SARS-CoV-2 infection of the oral cavity and saliva. Nat. Med. 27, 892–903 (2021).
Zhou, Z. et al. TAS2R16 activation suppresses LPS-induced cytokine expression in human gingival fibroblasts. Front. Immunol. 12, 726546 (2021).
Zhang, Z. et al. Salicin alleviates periodontitis via Tas2r143/gustducin signaling in fibroblasts. Front. Immunol. 15, 1374900 (2024).
Jaggupilli, A. et al. Chemosensory bitter taste receptors (T2Rs) are activated by multiple antibiotics. FASEB J. 33, 501–517 (2019).
Finger, T. E. et al. Solitary chemoreceptor cells in the nasal cavity serve as sentinels of respiration. Proc. Natl. Acad. Sci. USA 100, 8981–8986 (2003).
Saunders, C. J., Christensen, M., Finger, T. E. & Tizzano, M. Cholinergic neurotransmission links solitary chemosensory cells to nasal inflammation. Proc. Natl. Acad. Sci. USA 111, 6075–6080 (2014).
Barham, H. P. et al. Solitary chemosensory cells and bitter taste receptor signaling in human sinonasal mucosa. Int. Forum Allergy Rhinol. 3, 450–457 (2013).
Lee, R. J. et al. Bacterial d-amino acids suppress sinonasal innate immunity through sweet taste receptors in solitary chemosensory cells. Sci. Signal 10, https://doi.org/10.1126/scisignal.aam7703 (2017).
Montoro, D. T. et al. A revised airway epithelial hierarchy includes CFTR-expressing ionocytes. Nature 560, 319–324 (2018).
Krasteva, G. et al. Cholinergic chemosensory cells in the trachea regulate breathing. Proc. Natl. Acad. Sci. USA 108, 9478–9483 (2011).
Li, Y. H. et al. G protein subunit Ggamma13-mediated signaling pathway is critical to the inflammation resolution and functional recovery of severely injured lungs. Elife 12, https://doi.org/10.7554/eLife.92956 (2024).
Lee, R. J. et al. T2R38 taste receptor polymorphisms underlie susceptibility to upper respiratory infection. J. Clin. Investig. 122, 4145–4159 (2012).
Douglas, J. E., Saunders, C. J., Reed, D. R. & Cohen, N. A. A role for airway taste receptor modulation in the treatment of upper respiratory infections. Expert Rev. Respir. Med. 10, 157–170 (2016).
Takemoto, K. et al. Functional alteration and differential expression of the bitter taste receptor T2R38 in human paranasal sinus in patients with chronic rhinosinusitis. Int. J. Mol. Sci. 24, https://doi.org/10.3390/ijms24054499 (2023).
Adappa, N. D. et al. Correlation of T2R38 taste phenotype and in vitro biofilm formation from nonpolypoid chronic rhinosinusitis patients. Int. Forum Allergy Rhinol. 6, 783–791 (2016).
Gerbe, F. et al. Intestinal epithelial tuft cells initiate type 2 mucosal immunity to helminth parasites. Nature 529, 226–230 (2016).
von Moltke, J., Ji, M., Liang, H. E. & Locksley, R. M. Tuft-cell-derived IL-25 regulates an intestinal ILC2-epithelial response circuit. Nature 529, 221–225 (2016).
Nadjsombati, M. S. et al. Detection of succinate by intestinal tuft cells triggers a type 2 innate immune circuit. Immunity 49, 33–41 e37 (2018).
Howitt, M. R. et al. Tuft cells, taste-chemosensory cells, orchestrate parasite type 2 immunity in the gut. Science 351, 1329–1333 (2016).
Hoover, B. et al. The intestinal tuft cell nanostructure in 3D. Sci. Rep. 7, 1652 (2017).
Miller, C. N. et al. Thymic tuft cells promote an IL-4-enriched medulla and shape thymocyte development. Nature 559, 627–631 (2018).
Howitt, M. R. et al. The taste receptor TAS1R3 regulates small intestinal tuft cell homeostasis. Immunohorizons 4, 23–32 (2020).
Ki, S. Y. & Jeong, Y. T. Taste receptors beyond taste buds. Int. J. Mol. Sci. 23, https://doi.org/10.3390/ijms23179677 (2022).
Dyer, J., Salmon, K. S., Zibrik, L. & Shirazi-Beechey, S. P. Expression of sweet taste receptors of the T1R family in the intestinal tract and enteroendocrine cells. Biochem. Soc. Trans. 33, 302–305 (2005).
Bezencon, C., le Coutre, J. & Damak, S. Taste-signaling proteins are coexpressed in solitary intestinal epithelial cells. Chem. Senses 32, 41–49 (2007).
Bhushan, S. et al. Testicular innate immune defense against bacteria. Mol. Cell Endocrinol. 306, 37–44 (2009).
Arrellano-Valdez, F., Urrutia-Osorio, M., Arroyo, C. & Soto-Vega, E. A comprehensive review of urologic complications in patients with diabetes. SpringerPlus 3, 549 (2014).
Efeyan, A., Comb, W. C. & Sabatini, D. M. Nutrient-sensing mechanisms and pathways. Nature 517, 302–310 (2015).
Haber, A. L. et al. A single-cell survey of the small intestinal epithelium. Nature 551, 333–339 (2017).
Furness, J. B., Rivera, L. R., Cho, H. J., Bravo, D. M. & Callaghan, B. The gut as a sensory organ. Nat. Rev. Gastroenterol. Hepatol. 10, 729–740 (2013).
Smith, K. et al. T1R2 receptor-mediated glucose sensing in the upper intestine potentiates glucose absorption through activation of local regulatory pathways. Mol. Metab. 17, 98–111 (2018).
Doyle, M. E. & Egan, J. M. Mechanisms of action of glucagon-like peptide 1 in the pancreas. Pharm. Ther. 113, 546–593 (2007).
Steinert, R. E. et al. Ghrelin, CCK, GLP-1, and PYY(3-36): secretory controls and physiological roles in eating and glycemia in health, obesity, and after RYGB. Physiol. Rev. 97, 411–463 (2017).
Huang, F. et al. The effects of different concentrations of glucose on glucose sensors and GLP-1 secretion in the enteroendocrine cell line STC-1. Gen. Physiol. Biophys. 39, 79–87 (2020).
Reimann, F., Tolhurst, G. & Gribble, F. M. G-protein-coupled receptors in intestinal chemosensation. Cell Metab. 15, 421–431 (2012).
Huang, T. N., Lu, K. N., Pai, Y. P., Chin, H. & Huang, C. J. Role of GLP-1 in the hypoglycemic effects of wild bitter gourd. Evid. Based Complement. Altern. Med. 2013, 625892 (2013).
Kyriazis, G. A., Smith, K. R., Tyrberg, B., Hussain, T. & Pratley, R. E. Sweet taste receptors regulate basal insulin secretion and contribute to compensatory insulin hypersecretion during the development of diabetes in male mice. Endocrinology 155, 2112–2121 (2014).
Kyriazis, G. A., Soundarapandian, M. M. & Tyrberg, B. Sweet taste receptor signaling in beta cells mediates fructose-induced potentiation of glucose-stimulated insulin secretion. Proc. Natl. Acad. Sci. USA 109, E524–E532 (2012).
Nakagawa, Y. et al. Sweet taste receptor expressed in pancreatic beta-cells activates the calcium and cyclic AMP signaling systems and stimulates insulin secretion. PLoS One 4, e5106 (2009).
Medina, A. et al. Expression of the glucose-sensing receptor T1R3 in pancreatic islet: changes in the expression levels in various nutritional and metabolic states. Endocr. J. 61, 797–805 (2014).
Kojima, I. et al. Return of the glucoreceptor: Glucose activates the glucose-sensing receptor T1R3 and facilitates metabolism in pancreatic beta-cells. J. Diab. Investig. 6, 256–263 (2015).
Straub, S. G., Mulvaney-Musa, J., Yajima, H., Weiland, G. A. & Sharp, G. W. Stimulation of insulin secretion by denatonium, one of the most bitter-tasting substances known. Diabetes 52, 356–364 (2003).
Stumvoll, M., Goldstein, B. J. & van Haeften, T. W. Type 2 diabetes: principles of pathogenesis and therapy. Lancet 365, 1333–1346 (2005).
Young, R. L. et al. Disordered control of intestinal sweet taste receptor expression and glucose absorption in type 2 diabetes. Diabetes 62, 3532–3541 (2013).
Zheng, C. Y. et al. Revealing the mechanism of quinoa on type 2 diabetes based on intestinal flora and taste pathways. Food Sci. Nutr. 11, 7930–7945 (2023).
Bartakova, V., Kuricova, K., Zlamal, F., Belobradkova, J. & Kankova, K. Differences in food intake and genetic variability in taste receptors between Czech pregnant women with and without gestational diabetes mellitus. Eur. J. Nutr. 57, 513–521 (2018).
Dotson, C. D. et al. Bitter taste receptors influence glucose homeostasis. PLoS One 3, e3974 (2008).
Egan, J. M. Physiological integration of taste and metabolism. N. Engl. J. Med. 390, 1699–1710 (2024).
Liszt, K. I. et al. Human intestinal bitter taste receptors regulate innate immune responses and metabolic regulators in obesity. J. Clin. Investig. 132, https://doi.org/10.1172/JCI144828 (2022).
Sharma, P. et al. Antimitogenic effect of bitter taste receptor agonists on airway smooth muscle cells. Am. J. Physiol. Lung Cell Mol. Physiol. 310, L365–L376 (2016).
Schierbeck, H., Wahamaa, H., Andersson, U. & Harris, H. E. Immunomodulatory drugs regulate HMGB1 release from activated human monocytes. Mol. Med. 16, 343–351 (2010).
Vrancic, M. et al. Azithromycin distinctively modulates classical activation of human monocytes in vitro. Br. J. Pharm. 165, 1348–1360 (2012).
Ekoff, M. et al. Bitter taste receptor (TAS2R) agonists inhibit IgE-dependent mast cell activation. J. Allergy Clin. Immunol. 134, 475–478 (2014).
Lu, P. et al. Genetic deletion of the Tas2r143/Tas2r135/Tas2r126 cluster reveals that TAS2Rs may not mediate bitter tastant-induced bronchodilation. J. Cell Physiol. 236, 6407–6423 (2021).
Camoretti-Mercado, B. et al. Pleiotropic effects of bitter taste receptors on [Ca2+]i mobilization, hyperpolarization, and relaxation of human airway smooth muscle cells. PLoS One 10, e0131582 (2015).
Ni, K. et al. BitterDB database analysis plus cell stiffness screening identify flufenamic acid as the most potent TAS2R14-based relaxant of airway smooth muscle cells for therapeutic bronchodilation. Theranostics 14, 1744–1763 (2024).
Lu, P., Moore Simas, T. A., Delpapa, E. & ZhuGe, R. Bitter taste receptors in the reproductive system: Function and therapeutic implications. J. Cell Physiol. 239, e31179 (2024).
Fehr, J. et al. Expression of the G-protein alpha-subunit gustducin in mammalian spermatozoa. J. Comp. Physiol. A Neuroethol. Sens. Neural Behav. Physiol. 193, 21–34 (2007).
Mosinger, B. et al. Genetic loss or pharmacological blockade of testes-expressed taste genes causes male sterility. Proc. Natl. Acad. Sci. USA 110, 12319–12324 (2013).
Correia, J., Michelangeli, F. & Publicover, S. Regulation and roles of Ca2+ stores in human sperm. Reproduction 150, R65–R76 (2015).
Li, F. & Zhou, M. Depletion of bitter taste transduction leads to massive spermatid loss in transgenic mice. Mol. Hum. Reprod. 18, 289–297 (2012).
Gentiluomo, M. et al. Taste receptor polymorphisms and male infertility. Hum. Reprod. 32, 2324–2331 (2017).
Wu, S., Xue, P., Grayson, N., Bland, J. S. & Wolfe, A. Bitter taste receptor ligand improves metabolic and reproductive functions in a murine model of PCOS. Endocrinology 160, 143–155 (2019).
Duarte, A. C. et al. Bitter taste receptors profiling in the human blood-cerebrospinal fluid-barrier. Biochem. Pharmacol. 177, 113954 (2020).
Herrera Moro Chao, D. et al. Impact of obesity on taste receptor expression in extra-oral tissues: emphasis on hypothalamus and brainstem. Sci. Rep. 6, 29094 (2016).
Dehkordi, O. et al. Neuronal expression of bitter taste receptors and downstream signaling molecules in the rat brainstem. Brain Res. 1475, 1–10 (2012).
Dash, U. C. et al. Oxidative stress and inflammation in the pathogenesis of neurological disorders: mechanisms and implications. Acta Pharmaceutica Sin. B 15, 15–34 (2025).
Welcome, M. O. & Mastorakis, N. E. The taste of neuroinflammation: Molecular mechanisms linking taste sensing to neuroinflammatory responses. Pharmacol. Res. 167, 105557 (2021).
Zhou, L. et al. Sweet taste receptors mediated ros-nlrp3 inflammasome signaling activation: implications for diabetic nephropathy. 2018, 7078214, (2018).
Shin, Y.-J. et al. Enhanced expression of the sweet taste receptors and alpha-gustducin in reactive astrocytes of the rat hippocampus following ischemic injury. Neurochem. Res. 35, 1628–1634 (2010).
Paarmann, K. et al. French maritime pine bark treatment decelerates plaque development and improves spatial memory in Alzheimer’s disease mice. Phytomedicine 57, 39–48 (2019).
Wang, K. et al. Naringenin reduces oxidative stress and improves mitochondrial dysfunction via activation of the Nrf2/ARE signaling pathway in neurons. Int. J. Mol. Med. 40, 1582–1590 (2017).
Chiang, M.-C., Nicol, C. J. & Cheng, Y.-C. Resveratrol activation of AMPK-dependent pathways is neuroprotective in human neural stem cells against amyloid-beta-induced inflammation and oxidative stress. Neurochem. Int. 115, 1–10 (2018).
Cheng, X., Yang, Y.-L., Yang, H., Wang, Y.-H. & Du, G.-H. Kaempferol alleviates LPS-induced neuroinflammation and BBB dysfunction in mice via inhibiting HMGB1 release and down-regulating TLR4/MyD88 pathway. Int. Immunopharmacol. 56, 29–35 (2018).
El-kott, A. F. et al. Kaempferol protects against cadmium chloride-induced hippocampal damage and memory deficits by activation of silent information regulator 1 and inhibition of poly (ADP-Ribose) polymerase-1. Sci. Total Environ. 728, 138832 (2020).
Alves, V. C., Figueiro-Silva, J., Ferrer, I. & Carro, E. Epigenetic silencing of OR and TAS2R genes expression in human orbitofrontal cortex at early stages of sporadic Alzheimer’s disease. Cell. Mol. Life Sci. 80, 196 (2023).
Giráldez-Pérez, R. M., Antolín-Vallespín, M., Muñoz, M. D. & Sánchez-Capelo, A. Models of α-synuclein aggregation in Parkinson’s disease. Acta Neuropathol. Commun. 2, 176 (2014).
Kong, F. et al. Curcumin represses NLRP3 inflammasome activation via TLR4/MyD88/NF-κB and P2X7R signaling in PMA-induced macrophages. 7, 2016, (2016).
Welcome, M. O. & Mastorakis, N. E. Stress-induced blood brain barrier disruption: molecular mechanisms and signaling pathways. Pharmacol. Res. 157, 104769 (2020).
Welcome, M. O. Cellular mechanisms and molecular signaling pathways in stress-induced anxiety, depression, and blood–brain barrier inflammation and leakage. Inflammopharmacology 28, 643–665 (2020).
Jian, B., Yang, S., Chaudry, I. H. & Raju, R. Resveratrol restores sirtuin 1 (SIRT1) activity and pyruvate dehydrogenase kinase 1 (PDK1) expression after hemorrhagic injury in a rat model. Mol. Med. 20, 10–16 (2014).
Qi, Y. et al. Intracerebroventricular injection of resveratrol ameliorated Aβ-induced learning and cognitive decline in mice. Metab. Brain Dis. 34, 257–266 (2019).
Lee, G.-S. et al. The calcium-sensing receptor regulates the NLRP3 inflammasome through Ca2+ and cAMP. Nature 492, 123–127 (2012).
Ogunrinade, F. A. et al. Zanthoxylum zanthoxyloides inhibits lipopolysaccharide- and synthetic hemozoin-induced neuroinflammation in BV-2 microglia: roles of NF-κB transcription factor and NLRP3 inflammasome activation. J. Pharm. Pharmacol. 73, 118–134 (2021).
Wei, Y. et al. Rosmarinic acid mitigates lipopolysaccharide-induced neuroinflammatory responses through the inhibition of TLR4 and CD14 expression and NF-κB and NLRP3 inflammasome activation. Inflammation 41, 732–740 (2018).
Thawkar, B. S. & Kaur, G. Inhibitors of NF-κB and P2X7/NLRP3/Caspase 1 pathway in microglia: Novel therapeutic opportunities in neuroinflammation induced early-stage Alzheimer’s disease. J. Neuroimmunol. 326, 62–74 (2019).
Garcia-Esparcia, P. et al. Functional genomics reveals dysregulation of cortical olfactory receptors in Parkinson disease: novel putative chemoreceptors in the human brain. J. Neuropathol. Exp. Neurol. 72, 524–539 (2013).
Carey, R. M., Kim, T., Cohen, N. A., Lee, R. J. & Nead, K. T. Impact of sweet, umami, and bitter taste receptor (TAS1R and TAS2R) genomic and expression alterations in solid tumors on survival. Sci. Rep. 12, 8937 (2022).
Bi, S. et al. Comprehensive analysis of the function and prognostic value of TAS2Rs family-related genes in colon cancer. Int. J. Mol. Sci. 25, https://doi.org/10.3390/ijms25136849 (2024).
Skapinker, E. et al. Artificial and natural sweeteners biased T1R2/T1R3 taste receptors transactivate glycosylated receptors on cancer cells to induce epithelial-mesenchymal transition of metastatic phenotype. Nutrients 16, https://doi.org/10.3390/nu16121840 (2024).
Seo, Y., Kim, Y.-S., Lee, K. E., Park, T. H. & Kim, Y. Anti-cancer stemness and anti-invasive activity of bitter taste receptors, TAS2R8 and TAS2R10, in human neuroblastoma cells. PLOS ONE 12, e0176851 (2017).
Wölfle, U. et al. Salicin from Willow Bark can modulate neurite outgrowth in human neuroblastoma SH-SY5Y cells. 29, 1494-1500, (2015).
Hoffman, M. T. et al. The gustatory sensory G-protein GNAT3 suppresses pancreatic cancer progression in mice. Cell Mol. Gastroenterol. Hepatol. 11, 349–369 (2021).
Martin, L. T. P. et al. Bitter taste receptors are expressed in human epithelial ovarian and prostate cancers cells and noscapine stimulation impacts cell survival. Mol. Cell Biochem. 454, 203–214 (2019).
Gao, Y., Yin, J., Rankin, G. O. & Chen, Y. C. Kaempferol induces G2/M cell cycle arrest via checkpoint kinase 2 and promotes apoptosis via death receptors in human ovarian carcinoma A2780/CP70 cells. Molecules 23, https://doi.org/10.3390/molecules23051095 (2018).
Kashafi, E., Moradzadeh, M., Mohamadkhani, A. & Erfanian, S. Kaempferol increases apoptosis in human cervical cancer HeLa cells via PI3K/AKT and telomerase pathways. Biomed. Pharmacother. 89, 573–577 (2017).
Miller, Z. A. et al. Lidocaine induces apoptosis in head and neck squamous cell carcinoma through activation of bitter taste receptor T2R14. Cell Rep. 42, 113437 (2023).
Miller, Z. A. et al. GLUT1 inhibitor BAY-876 induces apoptosis and enhances anti-cancer effects of bitter receptor agonists in head and neck squamous carcinoma cells. Cell Death Discov. 10, 339 (2024).
Salvestrini, V. et al. Denatonium as a bitter taste receptor agonist modifies transcriptomic profile and functions of acute myeloid leukemia cells. 10, 2020, (2020).
Hung, J. et al. A Bitter taste receptor as a novel molecular target on cancer-associated fibroblasts in pancreatic ductal adenocarcinoma. Pharmaceuticals 16, https://doi.org/10.3390/ph16030389 (2023).
Funding
Open Access funding enabled and organized by Projekt DEAL.
Author information
Authors and Affiliations
Contributions
Conceptualization and supervision: W.Y. and X.Z.. Original draft: R.Z., X.Y., W.Y. and X.Z.. Review and editing: P.J., M.T., Z.S., W.Y. and X.Z..
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Zhai, R., Yong, X., Jiang, P. et al. The structure and function of taste G protein-coupled receptors and their implications in diseases. Int J Oral Sci 18, 34 (2026). https://doi.org/10.1038/s41368-026-00436-5
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41368-026-00436-5