Abstract
Multiple myeloma (MM) cells secrete high levels of immunoglobulin and are therefore addicted to mechanisms that maintain proteome homeostasis (proteostasis). While proteasome inhibitors that target the degradative aspect of proteostasis have proven effective, only limited attempts have been made to target protein secretion. Here we show that the receptor tyrosine kinase LTK is a regulatory node in the proteostasis network that responds to secretory load and helps cells maintain a high secretory output. LTK is a highly similar paralog to ALK and by repurposing existing ALK inhibitors, we demonstrate that targeting LTK causes immunoglobulin retention, ER stress and subsequent apoptosis of primary MM cells, even in patients refractory to proteasome inhibitors. Thus, LTK is a novel therapeutic target in the biosynthetic pathway of proteostasis, with significant potential for MM treatment.
Similar content being viewed by others
Introduction
Multiple myeloma (MM) is a plasma cell malignancy that is characterized in most cases by the excessive secretion of immunoglobulins (M-protein). Plasma cells produce thousands of immunoglobulins per second [1, 2] and the in vivo secretion rates of MM cells were estimated to be up to 85,000 molecules of IgG per cell per minute [3]. MM cells are therefore potentially addicted to mechanisms that maintain protein homeostasis (proteostasis) [4, 5]. Proteostasis involves both the synthesis and trafficking of proteins, as well as the degradation of misfolded proteins. Misfolded proteins in the endoplasmic reticulum (ER) are targeted to the proteasome to ER-associated degradation. The proteasome inhibitor (PI) bortezomib has been successfully used in myeloma treatment, leading to the subsequent approval of second generation proteasomal inhibitors, which are part of the many lines of standard of care for most myeloma patients [6, 7]. The biosynthetic and degradative parts of proteostasis are highly interconnected. However, no clinically or preclinically successful attempts have been made to target the biosynthetic part of the proteostasis network. This is partly because secretion has been considered notoriously undruggable. Identifying druggable targets within the secretion machinery holds promise not only for new therapies against MM, but also for other malignancies and conditions [8,9,10].
The ER is a major site for proteostasis and is equipped with various control systems that sense and respond to imbalances in the proteome, with the unfolded protein response (UPR) being the most prominent. The export of secretory proteins from the ER occurs in a COPII-dependent manner. Components of the COPII coat are not druggable, making it challenging to target ER export pharmacologically. In our previous work, we identified the receptor tyrosine kinase LTK as a regulator of COPII-dependent ER-export [11,12,13]. Blocking LTK function led to the inhibition of secretory protein export from the ER [14]. LTK is a paralog of anaplastic lymphoma kinase ALK [15], and is only expressed in myeloma cells following the rare event of an ALK-translocation; hence, ALK-inhibitors have not generally been used in myeloma therapies [16]. However, we previously demonstrated that ALK inhibitors blocked secretory trafficking from the ER [14]. We therefore hypothesized that targeting LTK in ALK-negative MM cells might inhibit immunoglobulin export from the ER, induce ER stress and ultimately trigger cell death. In the current study, we show that inhibition of LTK triggers cell death in a manner dependent on the secretory load of the cells. We demonstrate that LTK is expressed across different stages of MM and in various MM cell lines. Our work establishes LTK as a promising novel target for myeloma therapy.
Materials and methods
Patient samples and primary MM cell sample processing
MM patients were recruited from the Oslo Myeloma Center at Oslo University Hospital following signed informed consent in compliance with the Declaration of Helsinki. The study was approved by the Regional Committee for Medical and Health Research Ethics of South-East Norway (REC#2016/947 and 2012/174). Bone marrow mononuclear cells (BMMCs) were prepared from patient bone marrow aspirates using LymphoprepTM density gradient centrifugation. For MM cells, we followed a previously published prototocol [17, 18]. Briefly, CD8+ cytotoxic T lymphocytes were depleted by using CD8 Dynabeads (Life Technologies) and BMMCs were subsequently stimulated by expanding T helper cells in the presence of Human T-Activator CD3/CD28 Dynabeads (Life Technologies), and 100 U/ml human interleukin-2 (hIL-2, Roche, Germany). After 48 h, BMMCs were subjected to CD138+ enrichment to isolate MM plasma cells using MACS CD138+ microbeads (Miltenyi Biotec, Germany) These cells were used for drug treatment assay. For other assays CD138+ enrichment was performed on fresh cells that were assayed immediately.
Drug treatment and cell viability assay
The drugs used included crizotinib, ceritinib, entrectinib, ensartinib, alectinib, brigatinib and lorlatinib (Selleck Chemicals LLC). CD138+ MM cells (5000–10,000 cells/well) from activation assays were tested in 384-well plates against ALK-inhibitors concentrations ranging from 0.01 to 10 µM, as previously described [18]. Drug sensitivity score (DSS) was calculated as previously described [19]. See also Supplementary Methods for further details.
Cell lines
MM cell lines L363 and L363-BTZ were maintained in the RPMI-1640 culture medium (Sigma Aldrich, Buchs, Switzerland) supplemented with 10% heat-inactivated fetal bovine serum (FBS), 100 µg/ml streptomycin and 100U/ml penicillin/streptomycin (Sigma Aldrich, Buchs, Switzerland). The bortezomib-resistant cell lines were established and maintained from their parental cell line by continuous exposure to the drugs [20, 21]. The URVIN cell line was established from a patient by us [22], supplemented with 1 ng/ml IL-6 (R&D systems). The human myeloma cell lines (HMCLs) used for qPCR were OH-2 [23, 24], IH-1 [25], VOLIN, KJON [26], URVIN, JJN3, INA-6 [27] and U266. Cell lines has been verified using binominal fingerprint score as shown in [22], and regularly tested for mycoplasma (Lonza MycoAlert PLUS Mycoplasma Detection, BioNordika). RPMI-8226 was used for immunofluorescence. HeLa cells with inducible IgM expression has been described previously [28]. HeLa cells expressing LTK with LTK∆exon7 DNA constructs are detailed in the Supplementary Methods.
Immunofluorescence, immunoblotting and mass spectroscopy
HeLa cells grown on glass coverslips were transfected with a construct encoding GFP-LTK∆exon7 and stained with rabbit anti-CLIMP63 (CKAP4) pAb (1:500, gift from Hans-Peter Hauri) [29]. Details for immunoblotting, immunoprecipitation and mass spectroscopy are found in the Supplementary Methods.
Gene expression data and DNA constructs
Omics data were downloaded from the CoMMpass℠ study, IA-13 and IA-17 builds and included 767 and 921 MM samples respectively (www.themmrf.org). These data were generated as part of the Multiple Myeloma Research Foundation Personalized Medicine Initiatives (https://research.themmrf.org). RNA seq data were represented as Fragments Per Kilobase of transcript per Million mapped reads, FPKM. Data were visualized using Graphpad Prizm 10 with violin plots and FKPM < 1 was considered negative. Details on RT-PCR, site-directed PCR mutagenesis and pcDNA-LTK∆exon7 [30] are provided in the Supplementary Methods.
ELISA
IgG positive CD138+ separated cells (1×106/ml) were seeded in serum free human plasma like medium (HLPM, Thermo Fisher Scientific) with 5 µM ALK inhibitors or DMSO control for 3 h. The medium was collected, diluted two-fold, and analyzed using a ELISA total IgG kit (Invitrogen) according to the manufacturer’s instructions.
Intracellular immunoglobulin staining with fluorescence flow cytometry
URVIN or IgG positive CD138+ separated cells (1 × 106/ml) from patients were seeded in HLPM with 5 µM ALK inhibitors for 18 h (URVIN) or 3 h for primary cells. Cells were harvested, stained with Dead Cell Stain FarRed IR (Thermo Fisher Scientific), fixed with the TF fix kit (eBioscience), barcoded with Pacific Blue/Orange as previously described [31], and stained with anti-IgL PE-Cy-7 clone MHL-38 (316622, BioLegend) and/or anti IgK-AlexaFluor700 clone-G20-193 (561319, BD Pharmingen). Cells were acquired using an Attune Flow cytometer (Thermo Fischer Scientific) and data were analyzed using FlowJo 10.
Results
LTK is expressed in MM patients
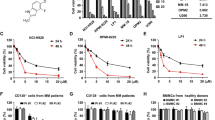
Expression of LTK and ALK was analyzed using RNA sequencing data from 767 MM patients from the CoMMpass study [32]. We found that the majority of patients (83%) had myeloma cells expressing LTK, but not ALK (0.1%) (Fig. 1A). Further analysis of LTK expression by qPCR was conducted in smouldering myeloma (SMM), newly diagnosed MM, relapsed/refractory MM, human myeloma cell lines (HMCLs), and healthy donor plasmablasts expanded from CD19+ peripheral blood cells. We found no significant difference in LTK expression between SMM, primary MM and relapsed MM (Fig.1B). LTK expression was also not significantly different between lines of treatment and in longitudinal samples from MM patients (Supplementary Fig. S1A, B). Moreover, MM cells showed, on average, over 10-fold higher LTK expression than healthy donor plasmablasts (Fig. 1B). Two relapsed/refractory MM patients had intermediate/low LTK levels. The HMCLs expressed low LTK, as could be expected since many HMCLs secrete relatively low levels of immunoglobulins. To confirm the expression at the protein level, cell lysates were tested for the presence of LTK by mass spectroscopy. In five out of all five samples tested, LTK peptide sequences were detected, including the MM cells from both intermediate/low LTK expressing relapsed/refractory MM patients (Fig. 1B, red symbols).
A LTK expression versus ALK expression from MM patients enrolled in the CoMMpass study (build IV13). FPKM: Fragments Per Kilobase Million. B qPCR analysis of LTK in smouldering multiple myeloma (SMM), newly diagnosed treatment naive MM, relapsed/refractory MM, human myeloma cell line (HMCL), and healthy donor plasmablasts. Red filled circles indicate samples tested and validated by mass spectrometry for LTK protein. One-way ANOVA, SMM vs. Diagnosis p = 0.8235; SMM vs. Relapse p = 0.4813; Diagnosis vs. Relapse p = 0.2111. C LTK transcript isoforms, LTK-201 (ENST00000263800), LTK-202 (ENST00000355166), and LTK-203 (ENST00000453182) in MM patients enrolled in the CoMMpass study (build IV17) and the sum of the three transcripts are shown. Medians, means, and fractions of transcript positive MM patients are indicated (Transcripts per million, TPM > 1). D Confocal micrograph of HeLa cells expressing LTK∆exon7, the dominant MM isoform of LTK (LTK-202). Left: LTK∆exon7-GFP signal (green), right: ER localization visualized through anti-Cytoskeleton-Associated Protein 4 (CKAP4) (red). Images are maximum intensity projections of several confocal sections. Scale bars: 10 μm. E Western blots of cell lysates from LTK∆exon7 HeLa cells or mock transfectants exposed to crizotinib or ceritinib, as indicated. Immunoblots of anti-phospho-LTK and anti-LTK with anti-vinculin as loading control are shown.
We next examined LTK expression in the CoMMpass study in more detail. The LTK gene is located on 15q15.1, but although chromosome 15 is frequently triploid in hyperdiploid myeloma patients, we did not find increased LTK expression due to chromosomal amplification (p = 0.4874, one-way ANOVA, Supplementary Fig. S1C), nor did we find any correlation between LTK mRNA levels and secreted M-protein (data not shown). However, we identified MM cells that had no or very low IgH expression (IgG1, IgG2, IgG3, IgG4, IgA1, IgA2), these had significantly less LTK expression than MM with above average IgH expression (p = 0.0009, Mann-Whitney, Supplementary Fig. S1D). A similar significant difference was seen in IgG1-positive MM: LTK expression in IgG1HI patients with higher than average IgG1 was increased and significantly different from that found in MM with lower than average IgG1 expression (p = 0.027, Mann-Whitney, Supplementary Fig. S1E). A similar LTK expression was found in non-malignant bone marrow plasma cells (Supplementary Fig S1F) [33]. There were no correlations between LTK expression level and the presence of common mutations in RAS, TP53 or DIS3 (Supplementary Fig. S1G). Analysis of data from the CoMMpass study also provided evidence that the main isotype of LTK transcript in MM cells was the LTK-202 isoform, which lacks exon 7 (LTK-202) (Fig. 1C). To validate ER localization of this isoform, we generated GFP-tagged LTK∆exon7 transfected HeLa cells and found co-localization of the LTK∆exon7 protein with CKAP4, a known ER marker (Fig. 1D). We also confirmed LTK localization in an HMCL, RPMI-8226, and in a primary myeloma patient sample, finding it localized to intracellular ER-like membranes (Supplementary Fig. S1H, I).
As the LTK kinase domain has very high homology to ALK, we tested whether the ALK inhibitors ceritinib and crizotinib could dephosphorylate LTK∆exon7 in HeLa cells. When cells were exposed to either of these drugs, we observed a dramatic downregulation in the ratio of phosphorylated LTK (pLTK) to dephosphorylated LTK in Western blot analyses (Fig. 1E).
Taken together we confirm that myeloma cells express LTK, but not ALK, and that the main isoform of LTK in myeloma cells localizes to the ER and can be inhibited by ALK inhibitors.
LTK plays a role for MM cells to cope with elevated secretory load
Since MM cells produce high amounts of immunoglobulins, we wanted to test whether LTK could mediate an adaptation to high secretory cargo load. We therefore used HeLa cells with inducible expression of secretory heavy chain of IgM [28]. Expression of the IgM heavy chain resulted in ER overload and was shown to trigger the unfolded protein response, UPR [28, 34]. Induction of IgM expression in HeLa cells for 24 h resulted in a mild activation of the UPR, as indicated by an increase in spliced X-box binding protein-1 (XBP1s) levels (Fig. 2A). When LTK expression was silenced, cells responded with higher levels of ER stress upon induction of IgM expression (Fig. 2A).
A HeLa cells expressing inducible IgM (heavy and light chain) were transfected with control or LTK siRNA. After 72 h, cells were treated with mifepristone to induce IgM expression followed by lysis and immunoblotting against the indicated proteins. B LTK mRNA is fourfold higher in HeLa cells with 2 weeks IgM expression compared to control. C Immunoblot of HeLa cells with induce IgM expression over a period of 2 weeks, subsequently treated with 1 μM crizotinib for 24 h. D L363 and bortezomib resistant L363-BTZ cells were treated with 5 µM crizotinib and the expression of spliced vs non-spliced XBP1 was not statistically different regardless of bortezomib sensitivity (p =0.533) for L363 vs L363-BTZ, two-way ANOVA (see also Supplementary Fig. S2A, B for analysis of crizotinib vs no drug for each cell line). E ATF4 was determined 4, 8 and 12 h post-treatment. Spliced vs non-spliced XBP1 p = 0.533 for L363 vs L363-BTZ, as well as ATF4 p = 0.267 between L363 and L363-BTZ and p = 0.013 for ATF4 expression over time (Supplementary Fig. S2).
In the same model system, IgM was induced for 14 days to allow cells to mount long-term adaptive responses to this increased secretory load. In the hypersecretory cells, we found a 4-fold increase of LTK mRNA (Fig. 2B), consistent with LTK playing an important role in helping cells cope with elevated secretory load. This result aligned with the correlation between IgG1 and LTK expression in primary MM cells as described above (Supplementary Fig. S1C). The IgM-expressing cells showed only moderately increased ER stress levels (Fig. 2C). Furthermore, when LTK was inhibited by crizotinib, hypersecretory cells exhibited a 2.5-fold increase in ER stress. In comparison, the non-IgM secreting control cells showed no LTK adaptation and no ER stress regardless of crizotinib treatment (Fig. 2C).
To determine whether LTK inhibition induces ER stress in HMCL, wild-type (L363) and bortezomib-resistant (L363-BTZ) cells were treated with crizotinib. Consistent the ER stress induction shown above, crizotinib treatment induced ER stress as demonstrated by increased splicing of XBP1 and induction of ATF4 (p = 0.013 over time) in both wildtype and bortezomib-resistant HMCL cells (Fig. 2D, E, Supplementary Fig. S2A, B). There was no significant difference between L363 and L363-BTZ response to crizotinib (XBP1 p = 0.533 and ATF4 p = 0.267, Fig. 2D, E)). These data suggest that LTK plays an important role in helping cells cope with a high secretory load, such as immunoglobulin secretion, with a significant increase in ER stress markers upon treatment with the ALK-inhibitor crizotinib.
Targeting LTK reduces the viability of multiple myeloma cells
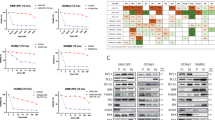
Induction of ER stress in hypersecretory cells prompted us to test whether LTK inhibition would trigger cell death. In the same experiment as in Fig. 2C, we found that the ER-stressed cells with hypersecretion of IgM had cleaved caspase 3 when LTK was inhibited with crizotinib. No apoptosis was detected in non-treated cells, nor in the non-IgM secreting controls (with or without crizotinib) (Fig. 3A). Thus, the secretory load of cells determines the sensitivity to ALK inhibitors.
A Immunoblot of caspase-3 in HeLa cells with induced IgM expression over a period of 2 weeks, subsequently treated with 1 μM crizotinib for 24 h. B Dose-response curve for alectinib, ceritinib and crizotinib, after 24 h of treatment in L363 and L363-BTZ cells. The results represent the means and ±SD of three independent experiments using the CCK-8 assay. C Viability (CellTiterGlo) dose-response curve in for ceritinib, entrectinib and bortezomib after 48 h of treatment in CD138+ bortezomib resistant myeloma patient samples (n = 3). Further drug combinations are shown in Supplementary Fig. S2E. D Combination of ceritinib with bortezomib in CD138+ MM cells from bortezomib sensitive patient samples (n = 3). The shown data are the concentration of closest to IC50 as possible for each single drugs for each patient, and the combination of these two concentrations. A full viability (CellTiterGlo) dataset is shown in Supplementary Fig. S2F for all combinations in one representative patient. E Combination of entrectinib with bortezomib in CD138+ bortezomib sensitive patient samples (n = 4), as in (D). Patient information for (C–E) is shown in Supplementary Table S1. One-way ANOVA p < 0.05 (*). F CD138+ cells from 22 myeloma patients were treated with ceritinib in increasing concentrations for 72 h before viability was measured with the CellTiterGlo assay. G Drug sensitivity score (DSS) of crizotinb was measured in the 22 patient samples, and the samples are sorted by their response to their last treatment: progressive disease (PD), stable disease (SD), partial response (PR), very good partial response (VGPR), and complete remission (CR) (n = 22, 72 h p = 0.8146 by ANOVA summary). H LTK RNA expression from MM cells from the CoMMpass study (build IV17) sorted similarly as in G (n = 139, P = 0.43, ANOVA multiple comparison). I, J In the same type of drug assay as in (F), six additional patients were treated with ceritinib, crizotinib entrectinib, ensartinib and lorlatinib. I Relative viability (%) is shown as average of n = 6 patient samples for each drug, with three technical replicates per patient sample. J DSS values are shown for all six patient samples, statistical comparisons with low-activity lorlatinib are shown. One-way ANOVA p < 0.0001 (****). See also Supplementary Fig. S3A for individual patient drug response curves and Supplementary Tables S2, 3 for patient information related to (F, G, I, J).
We next tested whether HMCL cells were sensitive to LTK inhibition. The L363 and L363-BTZ cells were treated with increasing concentrations of crizotinib, ceritinib and alectinib. We observed the same concentration-dependent cytotoxicity in both bortezomib-sensitive and bortezomib-resistant cells (Fig. 3B). This was repeated with patient samples previously treated with bortezomib, we observed a concentration-dependent cytotoxicity for ceritinib and entrectinib in all three patient samples tested (Fig. 3C). For bortezomib responsive patient samples we found a significant additive effect when comparing the closest value to IC50 in our assay for bortezomib and ceritinib (Fig. 3D) or entrectinib (Fig. 3E). Similar data from HMCLs are shown in Supplementary Fig. S2C, D.
To extend these results to primary MM cells, we tested the efficacy of crizotinib in MM cells from a cohort of patients, where 17 out of 22 patients that had become refractory to at least one proteasome inhibitor (Supplementary Table S2). In the presence of crizotinib, we found a reduced cell viability with a median IC50 of 1.93 µM crizotinib (Fig. 3F). Using a measure that provides a metric for the area under the curve (AUC), we calculated the drug sensitivity score (DSS) [18, 19] for the 22 MM patients, and results were sorted according to patient response to their previous treatment; progressive disease (PD), stable disease (SD), partial response (PR), very good partial response (VGPR), and complete remission (CR) derived from the International Myeloma Working group (IMWG) response criteria from 2016 [35]. There was no difference in the response to LTK inhibition between the groups (n = 22, p = 0.87) indicating that LTK inhibition had similar effect regardless of the preceding response status (Fig. 3G). In line with these results, there were no significant differences in LTK expression in the corresponding groups after analysis of data from the CoMMpass study (n = 139, p = 0.43), Fig. 3H.
To extend the drug effect analysis to additional ALK-inhibitors beyond crizotinib, we also tested ceritinib, entrectinib, ensartinib and lorlatinib on MM cells from six additional patients (Fig. 3I, J). With some variation between the drugs, we found that all drugs, except lorlatinib, showed a significant effect on MM cell viability (Fig. 3; see Supplementary Table 1 and 2 for patient information, and Supplementary Fig. S3A for individual response curves). Lorlatinib, which has a different chemical structure and binding site than the other ALK inhibitors and more than 38x lower cell free (IC50) activity for LTK than for ALK [36], resulted in only minor inhibition at 10 µM. DMSO served as negative control, and lorlatinib, as the only ALK-inhibitor with a low LTK activity, was also used for statistical comparisons. All 6 patients had received proteasome inhibitors (bortezomib or carfilzomib) in at least one line of previous treatment. ALK-inhibitors, except lorlatinib, were also effective in a range of HMCLs representing different genetic, mutational backgrounds and gene expression (Supplementary Figs. S3B, S4A, B). Additional assays with LTK inhibition of primary myeloma cells in bone marrow mononuclear cell (BMMC) cultures for 48 h are shown in Supplementary Fig. S5. In summary, we observed a decrease in viability in MM cells regardless of previous treatments when treated with the various ALK inhibitors.
Targeting LTK reduces antibody secretion
To further define specific functional correlates of LTK inhibition, we tested whether LTK inhibition caused intracellular retention of M-protein and correspondingly reduced secretion ability. We first tested the various ALK-inhibitors on the IgGκ-secreting HMCL URVIN, which was established by us [22]. After 18 h incubation, intracellular flow cytometry showed M-protein retention, detected by intracellular anti-κ light chain staining (Fig. 4A, B) in response to ceritinib, crizotinib, entrectinib, ensartinib, brigatinib and lorlatinib. All of these ALK-inhibitors except lorlatinib (that has a 38x reduced IC50 for LTK [36]) caused an increased mean fluorescence intensity (MFI) of κ light chain staining, (Fig. 4A). Lorlatinib had no effect, with κ light chain staining overlapping with untreated URVIN cells. The intracellular retention of κ was quantified in Fig. 4B, and showed a significant increase for all drugs with activity against LTK compared to lorlatinib (n = 4).
We tested the effect of LTK inhibition on HMCL (A, B) and primary MM cells (C–G) in short term culture, in terms of retained intracellular Ig (A–C) and reduced Ig secretion (E) and investigated the covariance of Ig secretion with entrectinib and ceritinib treatment. A The URVIN cell line was treated with 5 µM of ceritinib (black), crizotinib (pink), entrectinib (green), ensartinib (purple), brigatinib (brown) and the control lorlatinib (beige) for 18 h or left untreated (DMSO, gray filled histogram). Intracellular staining intensity for κ light chain is shown. B Relative expression is correlated to untreated cells. Statistical comparisons to lorlatinib, a low-activity control, is shown (n = 4 biological replicates of URVIN, one-way ANOVA). C Primary MM cells were treated for 3 h with 5 µM ceritinib (black), entrectinib (green), alectinib (orange) or lorlatinib (beige) or left untreated (gray histogram) for 3 h. D The change in intracellular κ or λ light chain (appropriate κ or λ per patient) is shown for primary myeloma samples (ceritinib, n = 7, entrectinib n = 10, alectinib n = 5, lorlatinib n = 7). E Secreted M-protein from MM cell cultures in assays without drug, after treatment for 3 h (ceritinib, n = 11, entrectinib n = 14, alectinib n = 11). F Linear regression analysis of increased light chain stain in MM samples (as in C, D) after treatment with ceritinib vs entrectinib (n = 13). G Linear regression of inhibited secretion of M-protein in primary MM cultures (as in E) with ceritinib vs entrectinib (n = 19). Kruskal–Wallis comparisons with Dunn’s multiple comparisons test in (D and E) are indicated, p < 0.05 (denoted by *), p < 0.01 (**) and p < 0.001 (***). See also Supplementary Tables S4 and S5 for patient information.
The intracellular retention experiments were repeated with freshly isolated CD138+ primary MM cells in a short-term 3 h assay to ensure MM cell viability. Even in 3 h assays, we observed increased staining intensity of the appropriate intracellular light chain in cells treated with ceritinib, entrectinib, and alectinib, but not with lorlatinib. An example from one primary patient sample where a shift to the right demonstrates a significant retention of intracellular light chains is shown in Fig. 4C. Quantification of the patient samples (n = 5–10) revealed significant M-protein (appropriate light chain) retention when comparing the ALK-inhibitors with lorlatinib (Fig. 4D). With intracellular retention of M-protein, it is expected that there would be a reduced secretion of M-protein from the cells. We therefore measured M-protein in supernatants in further patient samples after 3 h of inhibition, and found a significantly reduced secretion (Fig. 4E, n = 11–20).
As the various ALK-inhibitors may differ in off-target effects, we investigated whether the M protein retention in primary MM cells was a common feature that correlated across different drugs. Entrectinib is an ALK, NTREK1-3 and ROS1 inhibitor [37], while ceritinib is an ALK, IGF1R, INSR, TSSK1B and FLT3 inhibitor [38]. We found that both the intracellular retention of M protein (n = 12, p = 0.0029, Fig. 4F) and the reduced secretion (n = 18, p = 0.0029, Fig. 4G) showed a linear correlation between these two drugs, strongly suggesting that the observed retention was a direct result of LTK inhibition and not a consequence of different potential off-target effects. MM cells are generally negative for NTREK1-3 (present in 12–30% of patients), ROS-1 (0%), and TSSK1B (0%), but can be positive for IGF1R (43%), INSR (100%), and FLT3 (44%), data from CoMMpass, not shown. These data show that inhibition of LTK results in intracellular retention of M-protein and a significant reduction of antibody secretion from MM cells.
Discussion
In the current work, we identified LTK as a novel target for the treatment of MM. We demonstrated that inhibiting the biosynthetic branch of proteostasis, regulated by LTK, induces ER stress and ultimately apoptosis in vitro. RNA sequencing data from 767 patients in the CoMMpass study revealed that 83% of patients expressed LTK, while ALK expression was absent. Subsequent qPCR analysis confirmed LTK expression across various stages of MM, including smouldering myeloma, newly diagnosed MM, and relapsed/refractory MM. MM cells expressed over ten times higher LTK levels than healthy donor plasmablasts. Mass spectrometry verified LTK presence at the protein level in all tested samples, including those from relapsed/refractory patients with lower LTK levels. Further analysis revealed the primary isotype of LTK in MM cells as the LTK-202 isoform, which lacks exon 7 and localizes to the ER, as confirmed by co-localization studies in HeLa cells and primary myeloma samples. Functional assays revealed that LTK acts as a regulatory node in the proteostasis network, particularly influencing the unfolded protein response (UPR). In IgM expressing HeLa cells, LTK inhibition increased ER stress markers, particularly under conditions of high secretory load, supporting these predictions. Additionally, MM cells treated with ALK inhibitors, including crizotinib, ceritinib, and alectinib, demonstrated a concentration-dependent reduction in cell viability in concentrations that are in the range (3.1–11.29 µM) measured in serum from patients [39, 40], affecting both proteasome-sensitive and resistant cells. This was also demonstrated in three patients that were sensitive to ceritinib and entrectinib, despite being resistant towards bortezomib. Extending these findings to primary MM cells from patients refractory to proteasome inhibitors (PIs) showed similar results, with crizotinib reducing cell viability across patients with different response statuses to prior treatments. In addition, we demonstrated that in bortezomib sensitive patient cells, there was a significant additive effect of combining bortezomib and ALK-inhibitors. LTK inhibition caused intracellular retention of the M-protein and decreased its secretion, as shown in both HMCLs and primary MM cells. This retention and reduced secretion were observed consistently across treatment with various ALK inhibitors, suggesting that these effects were specific to LTK inhibition rather than off-target activities. Moreover, the correlation between M-protein retention and LTK inhibition across different drugs highlighted the potential of LTK as a therapeutic target in MM. Overall, the data demonstrated that targeting LTK disrupts cell viability by inducing ER-stress and impairing protein secretion, underscoring its therapeutic potential in managing MM.
MM therapy has significantly advanced in the last 20 years, including the introduction of PIs, such as bortezomib, ixazomib, and carfilzomib; immunomodulatory agents (IMIDs), like lenalidomide and pomalidomide; monoclonal antibodies targeting myeloma cell surface antigens (CD38: daratumumab and isatuximab; SLAMF7: elotuzumab); and autologous hematopoietic stem cell transplantation [41, 42]. Overall survival for newly diagnosed MM has dramatically improved from about 30 months before the year 2000 [43] to more than 120 months with standard first-line therapy comprising lenalidomide, bortezomib, and dexamethasone induction therapy [44]. Another common triplet therapy is daratumumab, lenalidomide, and dexamethasone (DRd) [41, 42]. Almost all patients with MM eventually relapse, and median progression free survival (PFS) and overall survival (OS) in patients with relapsed MM refractory to lenalidomide and bortezomib is poor, with median times of 5 months and 9 months, respectively [45].
Relapsed patients are mostly treated with triple therapies, but triple-refractory MM patients (refractory to a PI, an immunomodulatory drug (IMID) and an anti-CD38 mAb) and penta-refractory MM patients (refractory to two PIs, two IMIDs and an anti-CD38 mAb) are not uncommon. Thus, new agents are required for such multi-refractory patients. Selinexor, a selective inhibitor of nuclear export (SINE), has shown promise for these patients [46]. This drug covalently binds to the cargo-binding groove of exportin-1 (XPO1), interfering with protein trafficking from the nucleus to the cytosol. XPO1 inhibition causes the accumulation of tumor suppressor proteins such as p53, p21, and p27 in the nucleus, reactivating tumor suppressive pathways, inducing cell cycle arrest, and promoting pro-apoptotic pathways leading to apoptosis in cancer cells [47].
Our findings that LTK is expressed in MM cells, combined with the observation that this kinase was activated by secretory flux, make LTK a promising drug target for myeloma therapy. Moreover, the current demonstration that LTK inhibition remains effective in PI-refractory patients suggests that LTK inhibition could play a role in the treatment of MM patients refractory to one or more PIs. Furthermore, as LTK inhibition targets the biosynthetic part of the proteostasis network and the secretory pathway, it could add to PI inhibition of the degradation part of the proteostasis network and be part of combinational therapies. SINE and PI are already a potent combinational therapy [48], and a third potential synergy could involve SINE with the two other parts of the proteostasis network, targeting the biosynthetic part and secretory pathway (LTK) and protein degradation (proteasome).
LTK inhibition is of special interest in MM as even in end-stage MM a hypersecretory phenotype of MM cells is maintained in most patients, as indicated by persistently increased M-protein levels. Early estimates suggested that individual MM cells secrete up to 85,000 molecules of IgG per minute [3]. Current results demonstrate that MM cells likely depend on proper LTK function, the first discovered ER-resident receptor tyrosine kinase that regulates export from the ER [14, 49]. The ER is a major site for proteostasis, but little was known about the adaptation to higher secretory load of appropriately folded protein and the regulation at ER entry sites [11,12,13]. We showed an upregulation of LTK expression in HeLa cells as an adaptation to IgM secretion, and that such cells, but not non-transfected cells, became sensitive to LTK inhibition. Further, LTK correlated with IgG1 expression in IgG1+ MM and that short-term inhibition resulted in M-protein retention and apoptosis of MM cells regardless of prior therapy.
Besides having relevance for plasma cell malignancies, our results may also be of interest for other diseases featuring hypersecretion. Non-malignant plasma cells that also secrete high levels of Ig are relevant targets, especially in autoantibody-driven autoimmune disorders. Interestingly, a gain-of-function mutation in LTK was identified in the LTK kinase domain in lupus-prone mice and in patients with systemic lupus erythematosus [50]. In light of the current results, increased LTK function could potentially play a role in B cell differentiation and the generation of plasma blasts as well as long-lived plasma cells.
Besides LTK inhibition, the ALK-inhibitors, tested here, may have potential beneficial synergistic effects in MM due to the inhibition of various kinases by each particular drug. For example, ceritinib has activity on IGF1R and INSR, both of which may impact metabolically active MM cells. Inhibition of MET, the most MM-relevant non-ALK/LTK target for crizotinib, would not induce ER-stress [51] nor cause M protein retention, but may play a role in inhibiting MM responses to HGF including reduced proliferation, and microenvironment effects [52]. However, inhibition of other target kinases could not explain the findings of M-protein retention within the 3-hour scope of our assays. In this regard, the finding of a linear correlation with the effect of ceritinib versus entrectinib (ALK, NTRK1-3) strongly argues against other targets than LTK.
The results suggest that MM cells are particularly vulnerable targets of LTK inhibition and suggest that potential synergies may be favorable both in combination with existing therapies (PIs and SINE) and may provide additional MM-relevant inhibition for repurposed ALK inhibitors.
Data availability
All material described in the manuscript, including all relevant raw data will be freely available to any researcher wishing to use them for non-commercial purposes, without breaching participant confidentiality. Access to patient sensitive data is restricted (REC#2016/947 and 2012/174).
References
Helmreich E, Kern M, Eisen HN. The secretion of antibody by isolated lymph node cells. J Biol Chem. 1961;236:464–73.
Hibi T, Dosch HM. Limiting dilution analysis of the B cell compartment in human bone marrow. Eur J Immunol. 1986;16:139–45.
Salmon SE, Smith BA. Immunoglobulin synthesis and total body tumor cell number in IgG multiple myeloma. J Clin Investig. 1970;49:1114–21.
Auner HW, Cenci S. Recent advances and future directions in targeting the secretory apparatus in multiple myeloma. Br J Haematol. 2015;168:14–25.
Palumbo A, Anderson K. Multiple myeloma. N Engl J Med. 2011;364:1046–60.
Moreau P, Richardson PG, Cavo M, Orlowski RZ, San Miguel JF, Palumbo A, et al. Proteasome inhibitors in multiple myeloma: 10 years later. Blood. 2012;120:947–59.
PDQ Adult Treatment Editorial Board. Plasma Cell Neoplasms (Including Multiple Myeloma) Treatment (PDQ®): Health Professional Version. 2025 Apr 25. In: PDQ Cancer Information Summaries [Internet]. Bethesda (MD): National Cancer Institute (US); 2002–.
Deshaies RJ. Proteotoxic crisis, the ubiquitin-proteasome system, and cancer therapy. BMC Biol. 2014;12:94.
Balch WE, Morimoto RI, Dillin A, Kelly JW. Adapting proteostasis for disease intervention. Science. 2008;319:916–9.
Dejeans N, Manie S, Hetz C, Bard F, Hupp T, Agostinis P, et al. Addicted to secrete - novel concepts and targets in cancer therapy. Trends Mol Med. 2014;20:242–50.
Liu L, Cai J, Wang H, Liang X, Zhou Q, Ding C, et al. Coupling of COPII vesicle trafficking to nutrient availability by the IRE1alpha-XBP1s axis. Proc Natl Acad Sci USA. 2019;116:11776–85.
Acosta-Alvear D, Zhou Y, Blais A, Tsikitis M, Lents NH, Arias C, et al. XBP1 controls diverse cell type- and condition-specific transcriptional regulatory networks. Mol cell. 2007;27:53–66.
Farhan H, Weiss M, Tani K, Kaufman RJ, Hauri HP. Adaptation of endoplasmic reticulum exit sites to acute and chronic increases in cargo load. EMBO J. 2008;27:2043–54.
Centonze FG, Reiterer V, Nalbach K, Saito K, Pawlowski K, Behrends C, et al. LTK is an ER-resident receptor tyrosine kinase that regulates secretion. J Cell Biol. 2019;218:2470–80.
De Munck S, Provost M, Kurikawa M, Omori I, Mukohyama J, Felix J, et al. Structural basis of cytokine-mediated activation of ALK family receptors. Nature. 2021;600:143–7.
Masood A, Christ T, Asif S, Rajakumar P, Gustafson BA, Shune LO, et al. Non-secretory multiple myeloma with unusual TFG-ALK fusion showed dramatic response to ALK inhibition. NPJ Genom Med. 2021;6:23.
Wang D, Floisand Y, Myklebust CV, Burgler S, Parente-Ribes A, Hofgaard PO, et al. Autologous bone marrow Th cells can support multiple myeloma cell proliferation in vitro and in xenografted mice. Leukemia. 2017;31:2114–21.
Giliberto M, Thimiri Govinda Raj DB, Cremaschi A, Skanland SS, Gade A, Tjonnfjord GE, et al. Ex vivo drug sensitivity screening in multiple myeloma identifies drug combinations that act synergistically. Mol Oncol. 2022;16:1241–58.
Giliberto M, Santana LM, Holien T, Misund K, Nakken S, Vodak D, et al. Mutational analysis and protein profiling predict drug sensitivity in multiple myeloma cell lines. Front Oncol. 2022;12:1040730.
Soriano GP, Besse L, Li N, Kraus M, Besse A, Meeuwenoord N, et al. Proteasome inhibitor-adapted myeloma cells are largely independent from proteasome activity and show complex proteomic changes, in particular in redox and energy metabolism. Leukemia. 2016;30:2198–207.
Besse A, Besse L, Kraus M, Mendez-Lopez M, Bader J, Xin BT, et al. Proteasome inhibition in multiple myeloma: head-to-head comparison of currently available proteasome inhibitors. Cell Chem Biol. 2019;26:340–51 e343.
Behsen AD, Holien T, Micci F, Rye M, Rasmussen JM, Andersen K, et al. Cell surface marker heterogeneity in human myeloma cell lines for modeling of disease and therapy. Sci Rep. 2024;14:28805.
Borset M, Waage A, Brekke OL, Helseth E. TNF and IL-6 are potent growth factors for OH-2, a novel human myeloma cell line. Eur J Haematol. 1994;53:31–37.
Vatsveen TK, Tian E, Kresse SH, Meza-Zepeda LA, Gabrea A, Glebov O, et al. OH-2, a hyperdiploid myeloma cell line without an IGH translocation, has a complex translocation juxtaposing MYC near MAFB and the IGK locus. Leuk Res. 2009;33:1670–7.
Hjertner O, Hjorth-Hansen H, Borset M, Seidel C, Waage A, Sundan A. Bone morphogenetic protein-4 inhibits proliferation and induces apoptosis of multiple myeloma cells. Blood. 2001;97:516–22.
Vatsveen TK, Borset M, Dikic A, Tian E, Micci F, Lid AH, et al. VOLIN and KJON-Two novel hyperdiploid myeloma cell lines. Genes Chromosomes Cancer. 2016;55:890–901.
Burger R, Guenther A, Bakker F, Schmalzing M, Bernand S, Baum W, et al. Gp130 and ras mediated signaling in human plasma cell line INA-6: a cytokine-regulated tumor model for plasmacytoma. Hematol J. 2001;2:42–53.
Bakunts A, Orsi A, Vitale M, Cattaneo A, Lari F, Tade L, et al. Ratiometric sensing of BiP-client versus BiP levels by the unfolded protein response determines its signaling amplitude. Elife. 2017;6:e27518.
Vedrenne C, Klopfenstein DR, Hauri HP. Phosphorylation controls CLIMP-63-mediated anchoring of the endoplasmic reticulum to microtubules. Mol Biol Cell. 2005;16:1928–37.
Ben-Tekaya H, Miura K, Pepperkok R, Hauri HP. Live imaging of bidirectional traffic from the ERGIC. J Cell Sci. 2005;118:357–67.
Hermansen JU, Yin Y, Rein ID, Skanland SS. Immunophenotyping with (phospho)protein profiling and fluorescent cell barcoding for single-cell signaling analysis and biomarker discovery. NPJ Precis Oncol. 2024;8:107.
Skerget S, Penaherrera D, Chari A, Jagannath S, Siegel DS, Vij R, et al. Comprehensive molecular profiling of multiple myeloma identifies refined copy number and expression subtypes. Nat Genet. 2024;56:1878–89.
Alzamareh DF, Meednu N, Nandedkar-Kulkarni N, Krenitsky D, Barnard J, Yasaka K, et al. Interferon activation in bone marrow long-lived plasma cells in systemic lupus erythematosus. Front Immunol. 2024;15:1499551.
Reimold AM, Iwakoshi NN, Manis J, Vallabhajosyula P, Szomolanyi-Tsuda E, Gravallese EM, et al. Plasma cell differentiation requires the transcription factor XBP-1. Nature. 2001;412:300–7.
Kumar S, Paiva B, Anderson KC, Durie B, Landgren O, Moreau P, et al. International Myeloma Working Group consensus criteria for response and minimal residual disease assessment in multiple myeloma. Lancet Oncol. 2016;17:e328–e346.
Johnson TW, Richardson PF, Bailey S, Brooun A, Burke BJ, Collins MR, et al. Discovery of (10R)-7-amino-12-fluoro-2,10,16-trimethyl-15-oxo-10,15,16,17-tetrahydro-2H-8,4-(metheno)pyrazolo[4,3-h][2,5,11]-benzoxadiazacyclotetradecine-3-carbonitrile (PF-06463922), a macrocyclic inhibitor of anaplastic lymphoma kinase (ALK) and c-ros oncogene 1 (ROS1) with preclinical brain exposure and broad-spectrum potency against ALK-resistant mutations. J Med Chem. 2014;57:4720–44.
Rolfo C, Ruiz R, Giovannetti E, Gil-Bazo I, Russo A, Passiglia F, et al. Entrectinib: a potent new TRK, ROS1, and ALK inhibitor. Expert Opin Investig Drugs. 2015;24:1493–1500.
Marsilje TH, Pei W, Chen B, Lu W, Uno T, Jin Y, et al. Synthesis, structure-activity relationships, and in vivo efficacy of the novel potent and selective anaplastic lymphoma kinase (ALK) inhibitor 5-chloro-N2-(2-isopropoxy-5-methyl-4-(piperidin-4-yl)phenyl)-N4-(2-(isopropylsulfonyl)phenyl)pyrimidine-2,4-diamine (LDK378) currently in phase 1 and phase 2 clinical trials. J Med Chem. 2013;56:5675–90.
Meneses-Lorente G, Bentley D, Guerini E, Kowalski K, Chow-Maneval E, Yu L, et al. Characterization of the pharmacokinetics of entrectinib and its active M5 metabolite in healthy volunteers and patients with solid tumors. Invest N Drugs. 2021;39:803–11.
Kurata Y, Miyauchi N, Suno M, Ito T, Sendo T, Kiura K. Correlation of plasma crizotinib trough concentration with adverse events in patients with anaplastic lymphoma kinase positive non-small-cell lung cancer. J Pharm Health Care Sci. 2015;1:8.
Cowan AJ, Green DJ, Kwok M, Lee S, Coffey DG, Holmberg LA, et al. Diagnosis and management of multiple myeloma: a review. JAMA. 2022;327:464–77.
Rajkumar SV. Multiple myeloma: 2022 update on diagnosis, risk stratification, and management. Am J Hematol. 2022;97:1086–107.
Bergsagel DE. Is aggressive chemotherapy more effective in the treatment of plasma cell myeloma? Eur J Cancer Clin Oncol. 1989;25:159–61.
Joseph NS, Kaufman JL, Dhodapkar MV, Hofmeister CC, Almaula DK, Heffner LT, et al. Long-term follow-up results of lenalidomide, bortezomib, and dexamethasone induction therapy and risk-adapted maintenance approach in newly diagnosed multiple myeloma. J Clin Oncol. 2020;38:1928–37.
Kumar SK, Lee JH, Lahuerta JJ, Morgan G, Richardson PG, Crowley J, et al. Risk of progression and survival in multiple myeloma relapsing after therapy with IMiDs and bortezomib: a multicenter international myeloma working group study. Leukemia. 2012;26:149–57.
Martino EA, Vigna E, Bruzzese A, Labanca C, Mendicino F, Lucia E, et al. Selinexor in multiple myeloma. Expert Opin Pharmacother. 2024;25:421–34.
Parikh K, Cang S, Sekhri A, Liu D. Selective inhibitors of nuclear export (SINE)–a novel class of anti-cancer agents. J Hematol Oncol. 2014;7:78.
Delforge M, Raddoux J, Antonis C, Clement C, Kint N, Vanhellemont A, et al. Selinexor, bortezomib and dexamethasone: an effective salvage regimen for heavily pretreated myeloma patients. Onco Targets Ther. 2022;15:243–50.
Farhan H. Tyrosine kinase signaling in and on the endoplasmic reticulum. Biochem Soc Trans. 2020;48:199–205.
Li N, Nakamura K, Jiang Y, Tsurui H, Matsuoka S, Abe M, et al. Gain-of-function polymorphism in mouse and human Ltk: implications for the pathogenesis of systemic lupus erythematosus. Hum Mol Genet. 2004;13:171–9.
Slordahl TS, Denayer T, Moen SH, Standal T, Borset M, Ververken C, et al. Anti-c-MET nanobody - a new potential drug in multiple myeloma treatment. Eur J Haematol. 2013;91:399–410.
Baljevic M, Zaman S, Baladandayuthapani V, Lin YH, de Partovi CM, Berkova Z, et al. Phase II study of the c-MET inhibitor tivantinib (ARQ 197) in patients with relapsed or relapsed/refractory multiple myeloma. Ann Hematol. 2017;96:977–85.
Acknowledgements
HF, LAM, KT acknowledge support from the Research Council of Norway (project numbers 310054, 310045). LAM is supported by Research Centre’s of Excellence scheme, (PRIMA project number 332727), the Norwegian Cancer society (grant 190369), and the South Eastern Regional Health Authority (project number 2019606). KT and SSS: Centre for Digital Life PINpOINT project 294916 and the Norwegian Centre for Clinical Cancer Research, MATRIX: Research Council of Norway and the Norwegian Cancer society joint grant 328827, and RCN ERA PerMed, project number 322898), the South Eastern Regional Health Authority (KT and LAM, project 2015031), and the Anders Jahre Foundation. LAM, KT and SSS: the KG Jebsen Foundation grant 19. HF is supported by grants from the Norwegian Cancer Society (project 208015), by the Research Council of Norway (project 302452), by H2020 MSCA-innovative training networks SECRET and SAND (H2020‐MSCA‐ITN‐860035; H2020‐MSCA‐ITN‐859962), by grants from the Austrian Science Foundation (FWF, projects: 10.55776/P35832, 10.55776/P36600, 10.55776/FG20). VR is supported by a grant from the Austrian Science Foundation (FWF, project: 10.55776/P36925). CD is supported by a Swiss National Foundation grant (SNF 310030_182492), LB and AB are supported by the project National Institute for Cancer Research (Program EXCELES, ID Project No. LX22NPO5102) - Funded by the European Union – Next Generation EU. RNAseq data from the CoMMpass study were generated as part of the Multiple Myeloma Research Foundation Personalized Medicine Initiatives (https://research.themmrf.org and www.themmrf.org).
Author information
Authors and Affiliations
Contributions
HF, LAM, FHS and KT conceived the study. TKV, LAM, HF, LB and FC wrote the paper. All authors contributed to the manuscript and approved the final version. TKV, LAM, HF, MG, VB, FC, AB, AT, AA, YF, SSS, JFI, KM, VR, MZ, CD and LB performed experiments and analyzed data. FHS provided clinical data and patient samples.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Våtsveen, T.K., Giliberto, M., Bjornsdottir, V. et al. Targeting proteostasis in multiple myeloma through inhibition of LTK. Leukemia 39, 2237–2245 (2025). https://doi.org/10.1038/s41375-025-02682-8
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41375-025-02682-8
This article is cited by
-
Towards a unified framework for the function of endoplasmic reticulum exit sites
Nature Reviews Molecular Cell Biology (2025)