Abstract
Surface acoustic wave (SAW) sensors demonstrate significant potential in environmental monitoring due to their high sensitivity and fast response capabilities. However, conventional single-component gas-sensitive materials struggle to achieve both wide detection ranges and rapid response simultaneously. This study developed a high-performance composite film through heterostructure engineering to enhance carbon dioxide (CO₂) sensing performance. A bilayer composite gas-sensing functional layer was fabricated by sequentially depositing tin oxide (SnO₂) and copper oxide (CuO) films on a lithium niobate (LiNbO₃) substrate via magnetron sputtering. Experimental results demonstrated that the SnO₂-CuO composite sensor exhibited a CO₂ sensitivity of 11.35 mV/%, representing 4.3-fold and 10.3-fold improvements over pure CuO (2.65 mV/%) and SnO₂ (1.10 mV/%), respectively. The detection range was extended to 0.1-4vol%, with response and recovery times reduced to 9.3 s and 28.9 s at room temperature (25 °C). In addition, the SAW sensor demonstrated excellent repeatability, humidity interference resistance, high selectivity and long-term stability (5.7% signal attenuation over 30 days). Density functional theory (DFT) calculations revealed that the enhanced performance was attributed to heterointerface charge modulation, which increased the adsorption capacity for CO₂ molecules.

Similar content being viewed by others
Introduction
The relentless rise in atmospheric CO₂ levels, exacerbated by industrial emissions and urbanization, has intensified demands for real-time gas monitoring in both environmental and indoor settings1,2,3. Prolonged exposure to CO₂ concentrations exceeding 1000 ppm impairs human cognitive function and respiratory health, while levels above 5000 ppm pose acute toxicity risks4. In carbon capture, utilization, and storage (CCUS) systems, undetected CO₂ leaks may lead to catastrophic asphyxiation hazards and groundwater acidification5,6. These challenges underscore the critical need for CO₂ sensors capable of rapid response (<30 s), wide detection ranges (0.1–4 vol%), and reliable operation under ambient conditions.
Conventional CO₂ sensors can be categorized into three types based on their sensing principles: optical sensors7,8 (e.g., non-dispersive infrared, NDIR), electrochemical sensors9,10, and metal-oxide-semiconductor (MOS) sensors11,12. These CO₂ sensors struggle to balance rapid response, low power consumption, and selectivity, especially under room-temperature conditions. In contrast, surface acoustic wave (SAW) sensors leverage piezoelectric substrate mediated mechanical wave modulation, enabling room-temperature operation, miniaturization, and direct gas-phase interaction without labeling13. SAW gas sensors function by depositing a sensitive thin film on the propagation path of the surface acoustic wave14,15. The frequency variation is caused by the mass loading effect of gas molecules adsorbed on the sensitive film, which modulates the propagation characteristics of the surface acoustic wave through changes in substrate surface mass16,17. From the perspective of sensing mechanisms, the gas-sensitive film, as the critical component of surface acoustic wave devices, predominantly governs the performance of SAW sensors17,18. The sensing film’s material composition, interfacial characteristics, and microstructural homogeneity directly dictate key operational metrics, including gas adsorption kinetics, charge transfer efficiency, and long-term stability.
The development of SAW-based CO₂ sensing has focused on single-phase metal oxide films. For example, Muhammad et al.19 demonstrated SnO₂-coated SAW sensors achieving a 0-2000 ppm detection range, and Abhishek et al.20 reported calcium doped ZnO thin film on langasite substrates achieving 400 °C operation with the 87 s response time. Despite these efforts, CO2 SAW sensors exhibit narrow dynamic ranges and slow response time, due to insufficient gas adsorption capacity and sluggish charge transfer kinetics in sensing films21. The performance bottleneck highlights the urgent need for innovative gas-sensitive material designs to optimize interfacial interactions and enhance CO₂ binding energetics, which remains a pivotal yet underexplored avenue for advancing SAW sensor technology. Recent density functional theory (DFT) simulations have demonstrated that heterostructured metal oxide interfaces can enhance adsorption energies for target gas molecules compared to single-phase materials, thereby theoretically improving sensing performance through optimized charge transfer and catalytic activity22,23. This enhancement is attributed to interfacial electron redistribution at heterojunctions, which strengthens gas-surface interactions and lowers reaction energy barriers24. However, experimental validation of these theoretical advantages in SAW sensor designs is rarely reported.
In this work, we develop a room-temperature SAW-CO₂ sensor using a magnetron-sputtered SnO₂-CuO bilayer film on a LiNbO₃ substrate. Specifically, a 200 nm SnO₂ underlayer is deposited to ensure strong adhesion and lattice compatibility with the LiNbO₃ surface, followed by a 40 nm CuO overlayer to enhance CO₂ adsorption. The SnO₂-CuO interface is engineered to form a type-II heterojunction, promoting charge separation and modulating surface conductivity upon gas exposure. The proposed sensor achieves a response time of 9.3 s and a detection limit of 0.1 vol%. Experimental results reveal that the hierarchical structure not only amplifies SAW velocity shifts induced by CO₂ adsorption but also minimizes cross-response to humidity and other interfering gases (C₂H5OH, H₂, CO, H₂S, NH₃, CH₄, C₂H₂) at 25 °C. This work establishes a scalable platform for energy-efficient gas sensors in smart environmental monitoring systems.
Material and methods
Fabrication of the SAW sensor
The SAW device utilized a 128° Y-X cut lithium niobate (LiNbO₃) piezoelectric substrate, chosen for its strong Rayleigh wave excitation and high temperature stability. The delay-line-patterned SAW structure was fabricated via photolithography process where photoresist was spin-coated onto the substrate surface, followed by UV exposure and development to form the interdigital transducer (IDT) pattern. The device was designed with an operating wavelength (λ) and frequency of 15.7 μm and 200 MHz, respectively. Two aluminum-metallized IDTs (120 nm thick) were positioned with a 4 mm acoustic path between them for sensing interface deposition. To reduce insertion loss, a single-phase unidirectional transducer (SPUDT) configuration was adopted, with electrode widths set to λ/4 and λ/8. The SPUDT arrays comprised 100 and 40 electrode pairs, respectively, to optimize acoustic energy directionality and signal efficiency.
Synthesis of SnO₂-CuO composite film
The SnO₂-CuO composite film was deposited on a LiNbO₃ substrate through a sequential magnetron sputtering process. The substrate underwent ultrasonic cleaning in acetone, ethanol, and deionized water prior to deposition. The SnO₂ layer (200 nm) was sputtered using a metallic oxide SnO2 target under Ar flow, DC power (35 W), and working pressure (3.0 mTorr) for 40 min. Subsequently, the CuO layer (40 nm) was deposited using a CuO target with Ar, RF power (100 W), and working pressure (2.5 mTorr) for 8 min. A 10-minute interval between depositions ensured interfacial stability. Optimized sputtering parameters (total thickness: 240 nm, Sn:Cu molar ratio = 3:1) yielded uniform elemental distribution and low surface roughness, critical for SAW signal stability.
Carbon dioxide sensor testing platform
As shown in Fig. S1, the testing platform comprised three components: a gas mixing system, a SAW sensor, and a signal output unit. The gas generator operated at a flow rate of 450 mL/min, dynamically blending a high-concentration CO₂ source gas (20 vol% CO₂ balanced with N₂, purity ≥99.999%) with dry synthetic air (79 vol% N₂ and 21 vol% O₂, purity ≥99.999%) to generate CO₂/air mixtures in the range of 0.1–4 vol%. To extract sensing signals, the fabricated sensor device was encapsulated in a sealed gas chamber (volume: ~500 mL) and integrated into a phase detector circuit, forming the CO₂ sensing system. The phase information of the SAW CO₂ sensor shifted with gas concentration variations, and the SAW sensing system converted these phase changes into a voltage output.
Characterization
The morphologies of gas sensitive materials were characterized by scanning electron microscopy (SEM, ZEISS Sigma 360) and atomic force microscope (AFM, Bruker Dimension Icon). The energy dispersive spectroscopy (EDS) and the elemental mapping images were explored by SEM attachment. To analyze the surface chemistry of SnO2, CuO and SnO2-CuO composite, the X-ray photoelectron spectrometer (XPS, Sigma Probe, Thermo Scientific K-Alpha) with Al Kα radiation (1486.6 eV) was used. We calibrated the binding energies by referencing the C 1 s peak at 284.8 eV.
Results and discussion
Characterization of the gas sensors
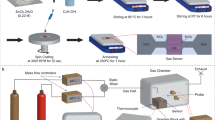
The SnO₂-CuO composite gas-sensitive thin film was fabricated by sequentially depositing SnO₂ and CuO thin films on a lithium niobate (LiNbO₃) substrate using magnetron sputtering technology. Figure 1a was the schematic of the SAW sensor, highlighting the sensing film area (3.1 mm × 1.6 mm) on the acoustic path. The surface acoustic wave (SAW) sensor consisted of three parts: interdigital transducers (IDTs), the SnO₂-CuO composite gas-sensitive layer, and the LiNbO₃ piezoelectric substrate. The physical image of the device (Fig. 1b) revealed its dimensions of 6 mm (length) × 3.4 mm (width), with the gas-sensitive region appearing light yellow, indicating the composite nature of SnO₂ and CuO. The surface morphology of the film was characterized by scanning electron microscopy (SEM) (Fig. 1c), which showed a dense and defect-free surface, confirming the uniformity of the sputtering process. Figure S2 displayed the SEM images of pure CuO and pure SnO₂ films, both exhibiting good surface density. The elemental distribution mapping (Fig. 1d) demonstrated the uniform distribution of Sn (cyan), Cu (green), and O (red) on the film surface, indicating excellent interfacial bonding between SnO₂ and CuO on the substrate. Cross-sectional SEM analysis (Fig. 1e) further revealed the bilayer structure of the composite film: a 200 nm SnO₂ layer at the bottom and a 40 nm CuO layer on top, with a total thickness of 240 nm. Atomic force microscopy (AFM) characterization (Fig. 1f) measured the film thickness to be approximately 240 nm, consistent with the SEM results. The surface exhibited an ultra-smooth morphology with an arithmetic average roughness (Ra) of 0.886 nm, confirming the uniformity of the sputtering process. These results confirmed the successful preparation of the SnO₂-CuO heterojunction film on the LiNbO₃ substrate.
a Schematic diagram for the synthesis of the CO2 SAW sensor with SnO2-CuO film. b The digital photo, c SEM image, d EDX mapping energy dispersive spectroscopy, e cross-section SEM image and f AFM image of SnO2-CuO film
XPS chemical state analysis was performed on CuO-SnO₂ composite samples, pure SnO₂, and pure CuO. Deconvolution spectra of individual elements were further analyzed to gain insights into their respective chemical states (Fig. 2a–c). As shown in the high-resolution Sn 3 d spectra (Fig. 2a), the CuO-SnO₂ composite exhibited two peaks centered at 486.6 eV (Sn 3 d₅/₂) and 495.1 eV (Sn 3 d₃/₂). Compared to pure SnO₂, these peaks showed a positive shift of 0.4 eV in the composite, indicating electron transfer from SnO₂ to CuO and an elevated oxidation state of Sn. The Cu 2p spectrum (Fig. 2b) displayed two broad peaks corresponding to Cu 2p₁/₂ and Cu 2p₃/₂, along with satellite peaks. In the composite, the Cu 2p₃/₂ and Cu 2p₁/₂ peaks appeared at 932.4 eV and 952.6 eV, respectively, which were ~1.7 eV lower than those of pure CuO (~934.1 eV and 954.3 eV), suggesting electron acceptance by Cu in the composite. The O 1 s spectra of all samples (Fig. 2c) were fitted with two components at 529.7 eV (lattice oxygen from Sn-O-Sn or Cu-O-Cu bonds) and 531.5 eV (vacancy-associated oxygen from O⁻ and O²⁻ species in oxygen-deficient regions)25,26. The composite demonstrated a higher oxygen species content than pure SnO₂, implying enhanced donor content. A slight red shift in the O 1 s peak of the composite was attributed to the intimate mixing between SnO₂ and CuO. EDS analysis (Fig. 2d) revealed a SnO₂-to-CuO molar ratio of approximately 3:1. These XPS and EDS results collectively confirmed the formation of a stable p-n heterojunction interface through electron interactions between SnO₂ and CuO, where charge redistribution and oxygen vacancy enrichment were highlighted as critical factors for enhanced CO₂ sensing performance.
a Sn 3d core level XPS spectra of SnO2-CuO and pure SnO2. b Cu 2p core level XPS spectra of SnO2-CuO and pure CuO. c O 1s core level XPS spectra of SnO2-CuO, pure SnO2 and CuO. d Energy dispersive spectroscopy spectrum of SnO2-CuO film
Gas sensing performance
To evaluate the gas-sensing performance of the SnO₂-CuO composite film, SAW devices based on SnO₂, CuO, and SnO₂-CuO were tested for CO₂ sensing at room temperature (25 °C). Figure 3a presents the real-time response curves of the three samples to different CO₂ concentrations (0.1–4 vol%). The SnO₂-CuO composite device exhibited significant advantages: its response to 4 vol% CO₂ reached 43 mV, far exceeding those of CuO (13 mV) and SnO₂ (6 mV). Even at a low concentration of 0.1 vol%, its response remained at 1.3 mV, while the pure SnO₂ and CuO devices showed almost no response (Fig. S3). A sensitivity comparison (Fig. 3b) revealed that the SnO₂-CuO composite device had a sensitivity of 11.35 mV/%, which was 4.3 times and 10.3 times higher than those of pure CuO (2.65 mV/%) and SnO₂ (1.10 mV/%), respectively. Figure 3c analyzed the response and recovery times of the SnO₂-CuO composite film to 4 vol% CO₂. The composite device exhibited a response time (T₉₀, time to reach 90% of the saturation value) of 9.3 s, significantly shorter than those of CuO (12.2 s) and SnO₂ (14.7 s) (Fig. S4), with a recovery time of 28.9 s. Additionally, repeatability tests (Fig. 3d) showed highly overlapping response curves for three cycles of 0.5 vol% CO₂, confirming the stability of SAW devices. The experimental results demonstrated that the SAW device based on the SnO₂-CuO composite film had a wide detection range of 0.1–4 vol% at room temperature, with overall performance (sensitivity, response speed, and repeatability) superior to single-component devices and most reported room-temperature metal oxide-based CO₂ sensors (Table 1). This performance enhancement could be attributed to the synergistic charge transfer effect at the SnO₂-CuO heterojunction interface and the promotion of CO₂ adsorption-reaction processes by oxygen vacancies.
a Sensing response, b sensitivity, c the response time and recovery time and d repeatability towards 0.5 vol% carbon dioxide
Humidity and cross-gas interference resistance
To assess the practical application potential of the SnO₂-CuO composite film SAW device, its humidity interference resistance, long-term stability, and gas selectivity were systematically investigated (Fig. 4). As shown in Fig. 4a, humidity interference tests were conducted under 10%–90% relative humidity (RH) at 25 °C. the response signal of SAW sensor slightly increased (from 0.8 mV to 1.3 mV) with rising humidity, but its maximum value (1.3 mV at 90% RH) was only 3.0% of the response to 4 vol% CO₂ (43 mV), indicating negligible humidity interference on CO₂ detection. This characteristic was attributed to the high preferential adsorption capacity of oxygen vacancies in the SnO₂-CuO composite film for CO₂, effectively suppressing competitive adsorption by water molecules. Long-term stability tests (Fig. 4b) showed that the response value of the sensor to 0.5 vol% CO₂ decreased by only 5.7% (from 7.9 mV to 7.45 mV) over 30 days, confirming its stable interfacial structure and negligible performance degradation. Gas selectivity tests (Fig. 4c) further compared the responses to 200 ppm interfering gases (C₂H₅OH, H₂, CO, H₂S, NH₃, CH₄, C₂H₂) and 0.5 vol% CO₂. The results showed that all interfering gases produced responses below 1.2 mV (with ethanol showing the highest response of 1.2 mV), demonstrating the excellent selectivity of the SAW sensor for CO₂. These results indicated that the sensor could maintain high-precision CO₂ detection in complex environments (high humidity, multiple interfering gases), providing critical performance assurance for practical deployment.
a Relationship between the SAW sensor response and humidity change. b Long-term stability of SnO2-CuO to 0.5 vol% CO2 at room temperature (25 °C). c Sensor selectivity for 0.5 vol% CO2 and 200 ppm of other gases at room temperature (25 °C)
Gas sensing mechanism
As shown in Fig. 5a, b, the high sensitivity and rapid response of the SnO₂-CuO nanocomposite toward CO₂ can be explained by dynamic band structure modulation. SnO₂ (n-type) and CuO (p-type) formed a p-n heterojunction at their interface, where electrons transferred from SnO₂ to CuO27,28. XPS results confirmed reduced electron density in SnO₂ and electron-enriched states in CuO due to interfacial charge redistribution. This charge transfer strengthened the interfacial electric field, creating a pronounced electron depletion layer (green region) at the SnO₂-CuO interface. The conduction band (Ec), Fermi level (Ef), and valence band (Ev) positions are defined, with bandgap energies (Eg) of 1.8 eV for CuO and 3.6 eV for SnO₂29,30. Beyond the material-level charge modulation, the SAW sensing mechanism is attributed to the acousto-electric effect as the dominant factor. When CO₂ molecules adsorb onto the SnO₂-CuO film, the heterojunction’s conductivity increases. This conductivity change attenuates the SAW propagation energy through acousto-electric effect, thereby shifting the phase velocity and resonant frequency of the acoustic wave31,32.
When the composite was exposed to air at elevated temperatures (Fig. 5a), oxygen molecules adsorb on the surface, forming chemisorbed oxygen species (O₂⁻, O⁻, or O²⁻) via reactions:
Schematic of band diagrams for the SnO2-CuO film constructed (a) in air and (b) in CO2. DFT-optimized structures of CO2 molecules adsorption on (c) SnO2, d CuO and (e) SnO2-CuO
This electron capture intensified the depletion layer barrier, resulting in a high-resistance state. Upon CO₂ exposure (Fig. 5b), CO₂ reacted with adsorbed oxygen species (O⁻) according to:
The reaction reduced hole concentration in CuO via electron-hole recombination and lowered the carrier concentration gradient across the heterojunction. Consequently, carrier diffusion weakened, the interfacial depletion layer narrowed, and conductivity increased significantly. The metastable carbonate intermediated rapidly decompose during recovery, restoring resistance.
Additionally, DFT calculations further validated the enhanced CO₂ sensing sensitivity of the SnO₂-CuO nanocomposite. Three models SnO₂, CuO, and the SnO₂-CuO composite were constructed, with their optimized structures shown in Fig. S5 (modeling details provided in the Supplementary Information). The adsorption energies (Eads) of CO₂ molecules on these systems were investigated. CO₂ exhibited an adsorption energy of +0.62 eV on the SnO₂ surface (Fig. 5c), where the positive value indicated weak physisorption and difficulty in capturing CO₂. In contrast, the CuO and SnO₂-CuO systems showed adsorption energies of −1.01 eV (Fig. 5d) and −1.84 eV (Fig. 5e), respectively, demonstrating effective chemisorption sites for CO₂. DFT calculations revealed that the SnO₂-CuO heterojunction exhibited the strongest CO₂ adsorption energy at interfacial oxygen vacancy sites (Fig. 5e), compared to CuO and SnO₂. This enhanced adsorption directly accelerated the CO₂ capture kinetics, reducing the response time to 9.3 s. The DFT results align with experimental gas-sensing data, confirming that the heterojunction interface significantly enhanced gas-sensing performance by optimizing charge transfer and surface reactivity.
Conclusion
This work demonstrated a high-performance SnO₂-CuO heterostructure-based SAW sensor for CO₂ detection, achieving a sensitivity of 11.35 mV/%, rapid response (t₉₀ = 9.3 s), and high selectivity. The enhanced performance was attributed to interfacial charge redistribution at the SnO₂-CuO p-n junction, where electron transfer created a built-in electric field and widened the depletion layer. XPS analysis and DFT simulations confirmed interfacial electron coupling (Sn3d/Cu3p binding energy shifts) and stronger CO₂ adsorption energy (−1.84 eV vs. −1.01 eV for CuO), aligning with experimental sensitivity trends. The sensor exhibited 0.1-4vol% detection range, humidity resistance and long-term stability (5.7% signal loss over 30 days). This heterostructure strategy, integrating band engineering and DFT-guided active site design, offers a universal approach for advanced gas sensors.
Change history
03 March 2026
A Correction to this paper has been published: https://doi.org/10.1038/s41378-026-01223-2
References
Haldar, T. et al. Exploring MOF-derived CuO/rGO heterostructures for highly efficient room temperature CO2 sensors. ACS Sens. 9, 5856–5865 (2024).
Zhou, Z., Xu, Y., Qiao, C., Liu, L. & Jia, Y. A novel low-cost gas sensor for CO2 detection using polymer-coated fiber Bragg grating. Sens. Actuators B Chem. 332, 129482 (2021).
Qu, R., Junge, K. & Beller, M. Hydrogenation of carboxylic acids, esters, and related compounds over heterogeneous catalysts: a step toward sustainable and carbon-neutral processes. Chem. Rev. 123, 1103–1165 (2023).
Fanget, S. et al. Gas sensors based on gravimetric detection—a review. Sens. Actuators B Chem. 160, 804–821 (2011).
Liang, B. et al. Carbon capture, utilization and storage (CCUS) in oil and gas reservoirs in China: status, opportunities and challenges. Fuel 375, 132353 (2024).
Romanak, K., Sherk, G. W., Hovorka, S. & Yang, C. Assessment of alleged CO2 leakage at the Kerr farm using a simple process-based soil gas technique: implications for carbon capture, utilization, and storage (CCUS) monitoring. Energy Procedia 37, 4242–4248 (2013).
Vafaei, M. & Amini, A. Chamberless NDIR CO2 sensor robust against environmental fluctuations. ACS Sens. 6, 1536–1542 (2021).
Ng, D. K. T. et al. NDIR CO2 gas sensing using CMOS compatible MEMS ScAlN-based pyroelectric detector. Sens. Actuators B Chem. 346, 130437 (2021).
Jeamjumnunja, K., Cheycharoen, O., Phongzitthiganna, N., Hannongbua, S. & Prasittichai, C. Surface-modified halloysite nanotubes as electrochemical CO2 sensors. ACS Appl. Nano Mater. 4, 3686–3695 (2021).
Struzik, M., Garbayo, I., Pfenninger, R. & Rupp, J. L. M. A simple and fast electrochemical CO2 sensor based on Li7La3Zr2O12 for environmental monitoring. Adv. Mater. 30, 1804098 (2018).
Bolli, E. et al. Engineered SnO2-based thin films for efficient CO2 gas sensing at room temperature. Appl. Surf. Sci. 683, 161795 (2025).
Masuda, Y. Recent advances in SnO2 nanostructure based gas sensors. Sens. Actuators B Chem. 364, 131876 (2022).
Ren, Z. et al. Temperature and humidity effects on SAW hydrogen sensor and compensation method. IEEE Sens. J. 24, 22317–22325 (2024).
Jin, J. et al. Dimensionality regulation of ZIF anti-interference layer for high-selectivity and fast-response hydrogen sensing. Int. J. Hydrog. Energy 50, 903–911 (2024).
Pan, Y. et al. A passive wireless surface acoustic wave (SAW) sensor system for detecting warfare agents based on fluoroalcohol polysiloxane film. Microsyst. Nanoeng. 10, 4 (2024).
Li, X. et al. Advances in sensing mechanisms and micro/nanostructured sensing layers for surface acoustic wave-based gas sensors. J. Mater. Chem. A 11, 9216–9238 (2023).
Kumar, A. & Prajesh, R. The potential of acoustic wave devices for gas sensing applications. Sens. Actuators A Phys. 339, 113498 (2022).
Pasupuleti, K. S. et al. High performance langasite based SAW NO2 gas sensor using 2D g-C3N4@TiO2 hybrid nanocomposite. J. Hazard. Mater. 427, 128174 (2022).
Aslam, M. Z., Zhang, H., Good, M., Guo, Y. & Larche, M. A multimodal lithium niobate-based SAW gas sensor for accurate detection of carbon dioxide at elevated temperature. Ultrasonics 138, 107198 (2024).
Ghosh, A., Zhang, C., Shi, S. & Zhang, H. High temperature CO2 sensing and its cross-sensitivity towards H2 and CO gas using calcium doped ZnO thin film coated langasite SAW sensor. Sens. Actuators B Chem. 301, 126958 (2019).
Sun, X. et al. Enhanced sensitivity of SAW based ammonia sensor employing GO-SnO2 nanocomposites. Sens. Actuators B Chem. 375, 132884 (2023).
Ruksana, S. et al. Highly efficient CuO-anchored SnO2 nanofiber for low-concentration H2S gas. Sens. ACS Appl. Eng. Mater. 2, 431–442 (2024).
Wu, R. et al. A lateral built-in field of the 2D/2D SnS2/SnSe2 in-plane heterostructure with boosted interfacial charge transfer. J. Mater. Chem. A 10, 14810–14819 (2022).
Zhao, Q. et al. Edge-enriched Mo2TiC2Tx/MoS2 heterostructure with coupling interface for selective NO2 monitoring. Adv. Funct. Mater. 32, 2203528 (2022).
Bai, S. et al. Gas sensing properties of Cd-doped ZnO nanofibers synthesized by the electrospinning method. J. Mater. Chem. A 2, 16697–16706 (2014).
Bai, S. et al. Synthesis of SnO2–CuO heterojunction using electrospinning and application in detecting of CO. Sens. Actuators B Chem. 226, 96–103 (2016).
Bhowmick, T., Ghosh, A., Nag, S. & Majumder, S. B. Sensitive and selective CO2 gas sensor based on CuO/ZnO bilayer thin-film architecture. J. Alloy. Compd. 903, 163871 (2022).
Ullah, E. et al. Fabrication of 1.6 V asymmetric supercapacitor in an aqueous electrolyte using a CuO-SnO2 composite and activated carbon electrodes. Mater. Today Commun. 37, 106889 (2023).
Zhang, J., Ma, S., Wang, B. & Pei, S. Hydrothermal synthesis of SnO2-CuO composite nanoparticles as a fast-response ethanol gas sensor. J. Alloy. Compd. 886, 161299 (2021).
Park, K.-R. et al. Design of highly porous SnO2-CuO nanotubes for enhancing H2S gas sensor performance. Sens. Actuators B Chem. 302, 127179 (2020).
Yuan, Y. et al. Analysis of the acoustoelectric response of SAW gas sensors using a COM model. Microsyst. Nanoeng. 10, 69 (2024).
Lu, X. et al. Harnessing exceptional points for ultrahigh sensitive acoustic wave sensing. Microsyst. Nanoeng. 11, 44 (2025).
Ghica, C. et al. Influence of relative humidity on CO2 interaction mechanism for Gd-doped SnO2 with respect to pure SnO2 and Gd2O3. Sens. Actuators B Chem. 368, 132130 (2022).
Xiong, Y. et al. Effective CO2 detection based on LaOCl-doped SnO2 nanofibers: insight into the role of oxygen in carrier gas. Sens. Actuators B Chem. 241, 725–734 (2017).
Xiong, Y., Zhang, G., Zhang, S., Zeng, D. & Xie, C. Tin oxide thick film by doping rare earth for detecting traces of CO2: operating in oxygen-free atmosphere. Mater. Res. Bull. 52, 56–64 (2014).
Wang, D. et al. CO2-sensing properties and mechanism of nano-SnO2 thick-film sensor. Sens. Actuators B Chem. 227, 73–84 (2016).
Lee, Z. Y., Hawari, H. F., Djaswadi, G. W. & Kamarudin, K. A highly sensitive room temperature CO2 gas sensor based on SnO2-rGO hybrid composite. Materials 14, 522 (2021).
Kumar, V. et al. Environment-sensitive and fast room temperature CO2 gas sensor based on ZnO, NiO and Ni-ZnO nanocomposite materials. Environ. Funct. Mater. 2, 167–177 (2023).
Abdelkarem, K. et al. Design of high-sensitivity La-doped ZnO sensors for CO2 gas detection at room temperature. Sci. Rep. 13, 18398 (2023).
Acknowledgements
This work was financially supported by the National Key Research and Development Program (2024YFE0199800), the National Natural Science Foundation of China (No. 12404543), China Postdoctoral Science Foundation National Key Research (2023M733687), and Key Research Program of Frontier Sciences, Chinese Academy of Sciences, Grant No. ZDBS-LY-7023.
Author information
Authors and Affiliations
Contributions
J.J.: writing—original draft, visualization, validation, methodology, investigation, formal analysis, conceptualization. Q.Y.: methodology; A.H.: conceptualization, resources; B.C.: validation, methodology; X.X.: supervision; Y.L.: methodology; F.S.: resources; K.L.: supervision, resources; W.W.: conceptualization, funding acquisition, project administration, writing—review & editing.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Jin, J., Yang, Q., Hu, A. et al. Heterojunction interface-engineered SnO₂-CuO SAW sensor for room-temperature CO₂ detection with fast response and high selectivity. Microsyst Nanoeng 11, 226 (2025). https://doi.org/10.1038/s41378-025-01062-7
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41378-025-01062-7