Abstract
Interleukin-17 (IL-17) is a pleiotropic cytokine produced mainly by peripheral T helper 17 cells. Yet, the brain functions of IL-17 derived from central nervous cells remain poorly understood. Here, we find an aberrant IL-17A signaling in the cerebellum of Fmr1-KO mice, a well-established genetic model for autism spectrum disorder (ASD). Cerebellar IL-17A, derived exclusively from microglia, is essential for the regulation of social behaviors by maintaining neuronal excitability and selectively suppressing inhibitory neurotransmission of Purkinje cells (PCs) in the cerebellar Crus I, a brain region critically involved in social cognition. Specific downregulation of IL-17 receptor-mediated signaling in cerebellar PCs recapitulates ASD-like social deficits and repetitive behaviors. Notably, both direct administration of IL-17A and induction of IL-17A release from cerebellar microglia by poly(I:C) effectively restore PC excitability and ameliorate ASD-like symptoms. The findings uncover an indispensable role of microglia-derived IL-17A for cerebellar social processing and suggest potential therapeutic strategies targeting IL-17A signaling for ASD.
Similar content being viewed by others
Introduction
Autism spectrum disorder (ASD) is a highly disabling neurodevelopmental condition characterized by impairments in social communication, along with the presence of restricted interests and repetitive behaviors [1]. Globally, more than 28 million individuals are affected by ASD [2], yet there is still a lack of effective treatment, placing a substantial burden on public health and society. A growing body of studies has highlighted the potential role of the immune/inflammation system in the development and progression of ASD. Notably, maternal immune activation (MIA) has been shown to induce ASD-like social behavioral abnormalities in offspring. Intriguingly, ASD-related behavioral symptoms in some patients appear to transiently improve during infections that spark a fever, a phenomenon referred to as the “fever effect” [3,4,5]. These observations suggest that immune-related factors could both contribute to and alleviate ASD symptoms [6].
Interleukin-17A (IL-17A), a multifaceted cytokine, has garnered significant attention for its involvement in ASD pathogenesis [7,8,9,10]. During pregnancy, maternal inflammation leads to the activation of T helper 17 (Th17) cells, which produce IL-17A, a key mediator in the induction of ASD-like phenotypes in offspring exposed to MIA [7]. In contrast, in adult offspring of the MIA model, IL-17A has been shown to ameliorate sociability deficits during inflammation induced by lipopolysaccharide (LPS) administration [9], a finding that may help explain the “fever effect” in ASD. Additionally, increased baseline serum levels of IL-17A have been observed in ASD patients, even without fever [11]. These results point to the complex and potentially dual roles of IL-17A in both the developing and adult brain. Therefore, understanding the mechanisms by which IL-17A influences brain neurons and ASD-related social behavior is essential for developing novel neuroimmunotherapeutic strategies for ASD.
Emerging evidence has also highlighted the critical role of the cerebellum in ASD [12,13,14,15,16], extending beyond its traditional functions in motor control to encompass contributions to reward processing, emotional regulation, and social communication [17,18,19,20,21,22]. Cerebellar abnormalities, including altered connectivity and Purkinje cell (PC) dysfunction, are consistently implicated in the pathophysiology of ASD [12, 15]. However, the role of the cerebellar neuroimmune system, particularly IL-17 signaling, in ASD remains poorly understood. Therefore, in this study, we examine the function of IL-17A in the cerebellum using a mouse model of Fragile X syndrome (FXS), which represents the most common monogenic cause of ASD, caused by mutations in the fragile X messenger ribonucleoprotein 1 (Fmr1) gene [23, 24]. We find that IL-17A levels are significantly elevated in the cerebellum of Fmr1-KO mice. Contrary to the classical view that IL-17A is produced by peripheral immune cells that infiltrate the central nervous system (CNS), we demonstrate that IL-17A in the cerebellum is exclusively produced by microglia, the resident innate immune cells of the CNS. The microglia-derived IL-17A is essential for maintaining the excitability of PCs, the principal neurons of the cerebellar cortex, and for regulating normal social behaviors. In the Fmr1-KO mice, the elevated cerebellar IL-17A levels may serve a compensatory role, potentially mitigating social behavioral deficits. Furthermore, our findings suggest that IL-17A restores cerebellar PC excitability and ameliorates ASD-like behavioral phenotypes in Fmr1-KO mice, paving a new path for therapeutic intervention targeting IL-17A signaling in ASD.
Methods
Animals
Male (Fmr1−/y) and female (Fmr1−/−) Fmr1-KO mice (aged 8-12 weeks) on an FVB background were provided by Prof. Chen Zhang from Capital Medical University, China. Male FVB (aged 8-12 weeks) and C57BL/6 (aged 8-12 weeks) mice were purchased from Vital River Laboratory Animal Technology Company (Beijing, China). To characterize the developmental trajectory of microglial IL-17A expression, we examined Fmr1-KO and wild-type (WT) littermates at key postnatal stages (P7, P14, and P21), which encompass critical periods of cerebellar synaptogenesis and circuit maturation [25]. Genotypes were confirmed via polymerase chain reaction analysis on extracted DNA. The primers were as follows: mutant forward: 5’-CACGAGACTAGTGAGACGTG-3’; WT forward: 5’-TGTGATAGAATATGCAGCATGTGA-3’; and common primer: 5’-CTTCTGGCACCTCCAGCTT-3’. Mice were housed 2 to 5 per cage under controlled environmental conditions (23 ± 1 °C; 12-h light/dark cycle) with food and water available ad libitum. All experimental procedures were conducted in accordance with the guidelines set forth by the National Institutes of Health Guide for the Care and Use of Laboratory Animals (NIH Publication No. 8023, revised 1978) and were approved by the Animal Ethical and Welfare Committee of Nanjing University. Efforts were made to minimize the number of animals used in the study.
Whole-cell patch-clamp recordings in brain slices
Brain slices containing the Crus I lobule of the cerebellum were prepared for whole-cell patch-clamp recordings as previously described [20, 26, 27]. According to the mouse brain atlas [28], cerebellar sagittal sections (300 μm thick) were obtained using a vibratome (VT 1200S, Leica, Wetzlar, Germany) in ice-cold sucrose-based artificial cerebrospinal fluid (ACSF). The ACSF composition included 212 mM sucrose, 3 mM KCl, 1.25 mM NaH2PO4, 26 mM NaHCO3, 7 mM MgCl2·6H2O, and 10 mM D-glucose, and was equilibrated with a gas mixture of 95% O2 and 5% CO2. Prior to recordings, brain slices were maintained in normal ACSF, consisting of 124 mM NaCl, 2.5 mM KCl, 1.25 mM NaH2PO4, 26 mM NaHCO3, 1.3 mM MgSO4, 2 mM CaCl2, and 20 mM D-glucose, bubbled with 95% O2 and 5% CO2, at 35 ± 0.5 °C for at least 1 h for recovery. An Olympus BX51WI microscope (Olympus, Tokyo, Japan) was used to visualize cerebellar PCs, and images of the patched cells were captured using a charge-coupled device camera (Cohu, Poway, USA) and displayed on a separate monitor. Recording data were amplified (Axopatch 200B; Axon Instruments), digitized (Digidata-1550; Axon Instruments), and analyzed with pClamp 10.4 (Axon Instruments).
Current-clamp recordings were employed to examine spontaneous firing activity, while voltage-clamp recordings at a holding potential of −70 mV were utilized to assess sIPSCs and sEPSCs of PCs. For spontaneous firing rate recordings, glass pipettes (3–5 MΩ) were filled with internal solution comprising 140 mM K-methylsulfate, 7 mM KCl, 2 mM MgCl2, 10 mM HEPES, 0.1 mM EGTA, 4 mM Na2-ATP, and 0.4 mM GTP-Tris, adjusted to pH 7.25 with 1 M KOH. Spontaneous firing activity in PCs was recorded following the bath application of IL17A (6.5 × 10−1 nM, 10 min; R&D Systems, 421-ML-100/CF), anti-IL-17R Ab antibody (4.17 nM, 10 min; R&D Systems, AF448), NF-κB inhibitor pyrrolidine dithiocarbamate (PDTC; 10 nM, 10 min pre-incubation; Sigma-Aldrich, P8765), and knockdown of IL-17RA using current-clamp recordings. Voltage-clamp recordings at a holding potential of −70 mV were used to assess the effects of brief bath application of IL17A (10 min), anti-IL-17R Ab antibody (10 min), PDTC (10 nM, 10 min pre-incubation), and knockdown of IL-17RA on whole-cell and postsynaptic currents of PCs. For sIPSCs recordings, glass pipettes were loaded with high Cl− internal solution containing 135 mM CsCl, 2 mM NaCl, 1 mM MgSO4, 0.2 mM CaCl2, 0.2 mM EGTA, 5 mM QX314, 4 mM Na2-ATP, 0.4 mM Na-GTP, with the pH adjusted to 7.2 using CsOH; D-APV (50 µM; Tocris Bioscience, 0106) and NBQX (20 µM; Tocris Bioscience, 0373) were included to block NMDA and AMPA receptors, respectively. For sEPSCs recordings, glass pipettes were filled with Cs+-methanesulfonate-based internal solution containing 120 mM cesium methanesulfonate, 20 mM CsCl, 10 mM HEPES, 0.2 mM EGTA, 10 mM sodium phosphocreatine, 5 mM QX314, 4 mM Na2-ATP, 0.4 mM Na-GTP, with the pH adjusted to 7.2 using CsOH. SR95531 (20 μM; Tocris Bioscience, 1262) was included to selectively block GABAA receptors (GABAAR).
Flow cytometry and microglia isolation
To analyze cerebellar IL-17A-expressed microglia, single-cell suspensions were prepared from brain tissue as previously described [29]. Briefly, the cerebellum was gently cut into small pieces using a sharp scalpel. The tissue fragments were digested in a dissociation solution containing 1 mg/ml collagenase IV (Sigma, CAS No. 9001121), 1 mg/ml papain (Roche, 10108014001), and 5 U/ml DNase I (Thermo Fisher, EN0521), all dissolved in Hank’s balanced salt solution (HBSS), for 30 min at 37 ◦C. Cell pellets were collected by centrifugation at 350 g for 5 min and washed with HBSS with 0.5% BSA to stop digestion and block. Density gradient (30–70% Percoll layers) centrifugation at 700 g for 30 min without braking was used for mononuclear cell separation. The layer between the interface of 30–70% Percoll solution was collected as mononuclear cells.
The isolated cells were washed and stained with antibodies against the following surface markers: CD45-Percp (Biolegend, clone number: 30-F11), CD11b-BV711 (Biolegend, clone number: M1/70), CD3-APC (Biolegend, clone number: 17A2), and CD4-FITC (Biolegend, clone number: GK1.5). Viability-Aqua reagent (Thermo Fisher, L34957) was used for live/dead cell exclusion. Relative isotypes were included as negative controls. After membrane antigen staining, cells were fixed and permeabilized using the Intracellular Fixation & Permeabilization Buffer Set (Thermo Fisher, 88-8824), followed by staining with PE/Cyanine7 anti-mouse IL-17A antibody (Biolegend, clone number: TC11-18H10.1). Flow cytometric data were acquired using an Attune NxT flow cytometer (Thermo Fisher) and analyzed with FlowJo software (Tree Star).
To purify cerebellar microglia for SMART-sequencing, mononuclear cells were resuspended in 90 μL HBSS-BSA buffer and incubated with CD11b microbeads (Miltenyi Biotec, 130-093-634) for 15 min in the dark at 4 °C. After washing and centrifuging, cell suspension was loaded onto a column placed in a MACS separator (Miltenyi Biotec). The column was washed with MACS buffer and removed from the separator, and purified microglia cells were collected and resuspended in TRIzol reagent (Vazyme Biotech, Nanjing, China). Purified microglia samples were subsequently sent to Majorbio (Majorbio Biopharm Technology, Shanghai, China) for the SMART-RNA sequence assay.
RNA sequencing, SMART-RNA sequencing and pathway analysis
Total RNA from the cerebellum was extracted using TRIzol reagent (Vazyme Biotech, Nanjing, China). All analytical samples were sent to Majorbio (Majorbio Biopharm Technology, Shanghai, China) for RNA sequencing. RNA extraction was performed according to the manufacturer’s instructions using either TRIzol® Reagent (15596018CN, Invitrogen) or the RNeasy Micro Kit (QIAGEN, Hilden, Germany), with genomic DNA removal using DNase I (TaKara). RNA degradation and contamination were monitored on 1% agarose gels. Then RNA quality was determined using the 2100 Bioanalyser (Agilent Technologies) and quantified using the ND-2000 (NanoDrop Technologies). Only high-quality RNA samples (OD260/280 = 1.8 ~ 2.2, OD260/230 ≥ 2.0, RIN ≥ 8.0, 28S:18S ≥ 1.0, > 1 μg) were used to construct the sequencing library. The mRNA was transcribed into full-length cDNA using a Smarter Amplification Kit from OE Biotech Co., Ltd. (Shanghai, China). The library was constructed using a TruSeqTM RNA sample prep Kit (Illumina) following the manufacturer’s protocol. Library quality was verified using the Agilent 2100 Bioanalyzer and the Agilent High Sensitivity DNA kit. The data were analyzed using the Majorbio Cloud Platform (https://cloud.majorbio.com) and R version 4.0. Briefly, transcript abundances from RNA-seq counts were quantified using RSEM. DEGs were selected with a fold change > 1.5 or fold change < 0.67, with statistical significance determined by P < 0.05, utilizing the DESeq2 package. Venn analysis was employed to identify the intersection of DEGs across both comparisons. Volcano plots were generated using tools from the Majorbio Cloud Platform. Additionally, KEGG analysis was conducted to determine if the intersecting genes were significantly enriched.
In addition to sequencing the entire cerebellum, we purified and sequenced cerebellar microglia, analyzing the data as described above. DEGs were selected with fold changes > 1.2 or < 0.83, with statistical significance determined by P < 0.1, using the DESeq2 package. KEGG analysis was similarly performed to assess whether the intersecting genes were significantly enriched.
Real-time quantitative polymerase chain reaction
Animals were deeply anesthetized with isoflurane and subsequently decapitated. The cerebellar cortex or hippocampus was acutely prepared from adult mice. The tissue samples were then rapidly frozen in liquid nitrogen and stored at −80 °C. Total RNA was isolated using TRIzol reagent (Vazyme, R401-01, Nanjing, China) and quantified with a NanoDrop spectrophotometer (Thermo Scientific, Waltham, USA). Three independent groups of RNA pools were used as technical replicates. Equal amounts of RNA for the samples were reverse-transcribed into cDNA using the HiScript III RT SuperMix for quantitative real-time PCR (qPCR) (Vazyme, R323-01). Gene expression levels of IL-17A, IL-17B, IL-17C, IL-17D, IL-17E, IL-17F, IL-17RA, TNF-α, IL-6, IFN-γ, and GABAARα1 were measured by qPCR using Hieff UNICON qPCR SYBR Green Master Mix (Yeasen, 11198ES03) on a CFX RT-qPCR Instrument (BIO-RAD, Hercules, USA). The mouse GAPDH gene was used for normalization. A list of the primers used is provided in Supplementary Table 1.
Western blots
Animals were deeply anesthetized with isoflurane and subsequently decapitated. The hippocampus and cerebellar cortex were acutely prepared from adult mice, which were then freshly frozen in liquid nitrogen. Tissue samples were homogenized in cold RIPA lysis buffer (Beyotime, P0013B) containing protease inhibitors (Beyotime, P1005) and lysed immediately. Protein concentration was determined by a BCA kit (Beyotime, P0009), and equal amounts of protein (30 μg) for each sample were separated by SDS-PAGE gel (10%) before being transferred to a polyvinylidene fluoride (PVDF) membrane. The membrane was blocked with non-fat milk (5%) in Tris-buffered saline containing 0.1% Tween 20, followed by overnight incubation with primary antibodies: goat anti-IL17RA (1:400, R&D Systems, AF448), rabbit anti-phospho-NF-κB p65 (Ser536) (1:1,000, Cell Signaling Technology, Cat# 3033), mouse anti-NF-κB p65 (1:1,000, Cell Signaling Technology, Cat# 6956), rabbit anti-GABAAR α1 (1:1,000, Synaptic Systems, Cat# 224203), and mouse anti-β-ACTIN (1:10,000, Thermo Fisher Scientific, MA5-15739). After a 1-h incubation at room temperature with secondary antibodies, including horseradish peroxidase (HRP)-conjugated rabbit anti-goat IgG (1:5,000, Life Technologies, 31402), HRP-conjugated goat anti-mouse IgG (1:5,000, Life Technologies, 62-6520), and HRP-conjugated anti-rabbit antibody (1:5,000; Life Technologies, Cat# 65-6120), the membrane was then developed using ECL reagents (E422, Vazyme) and visualized with a Tanon imaging system. Protein band intensity was quantified using ImageJ software (National Institutes of Health). The experiment was repeated at least three times independently to account for technical and biological variations. The original gel and blot images are provided in Fig. S10.
Electrochemiluminescence immunoassays
We used a high-sensitivity assay from Meso Scale Discovery (MSD) (S-PLEX; Cat No: K152UTK-1) to assess the levels of IL-17A in the cerebellum, following the manufacturer’s instructions. The cerebellum was weighed and homogenized using an electric homogenizer in ice-cold PBS containing protease inhibitors (MedChemExpress, HY-K0011). The homogenates were then centrifuged at 1500 g for 10 min to remove pellet debris, and the supernatant was collected and stored at −80 °C. IL-17A levels in the cerebellum were quantified using the electrochemiluminescence-based MSD platform.
Immunofluorescent staining
As reported previously [29], mice were perfused with PBS, followed by cold 4% paraformaldehyde. The paraformaldehyde-fixed mouse brains were cryoprotected in 20% and 30% sucrose (48 h each). Frozen coronal cerebellar, hippocampal, prefrontal cortex (PFC), and primary somatosensory cortex dysgranular zone (S1DZ) sections were obtained using a freezing microtome (CM 3050 S, Leica, Wetzlar, Germany) and mounted on gelatin-coated slides. The slices were rinsed in PBST (0.2% Triton X-100) six times for 5 min each and then blocked in 10% FBS (in PBST) for 30 min at room temperature. The slices were stained overnight at 4 °C with primary antibodies, including rabbit anti-Iba1 (1:500, Wako, Cat# 016-20001), mouse anti-IL17A (1:100, eBioscience, Cat# 14-7179-82), goat anti-IL17RA (1:200, R&D, Cat# AF448), rabbit anti-Calbindin (1:500, Cell Signaling Technology, Cat# 13176), mouse anti-Calbindin (1:200, Proteintech, Cat# 66394-1-Ig), rabbit anti-S100β (1:200, Abcam, Cat# ab52642), anti-mouse-CD3-AP3 (1:100, BioLegand, Cat# 100311), and rabbit anti-arc (1:500, Proteintech, Cat# 16290-1-AP), rabbit anti-phospho-NF-κB p65 (Ser536) (1:200, Cell Signaling Technology, Cat# 3033), mouse anti-Gephyrin (1:500, Synaptic Systems, Cat# 147008), and rabbit anti-GABAAR α1 (1:200, Synaptic Systems, Cat# 2224203). Following this, the slices were rinsed in PBST six times for 5 min each, before incubation with secondary antibodies at room temperature for 2 h in the dark. The secondary antibodies used included donkey anti-mouse IgG Alexa Fluor 488 (1:1,000, Thermo, Cat# a21202), donkey anti-rabbit IgG Alexa Fluor 488 (1:1,000, Thermo, Cat# a21206), goat anti-mouse IgG Alexa Fluor 594 (1:1,000, Thermo, Cat# a11020), goat anti-rabbit IgG Alexa Fluor 594 (1:1,000, Thermo, Cat# a11012), donkey anti-goat IgG Alexa Fluor 594 (1:1,000, Thermo, Cat# a11058), and donkey anti-goat IgG Alexa Fluor 647 (1:1,000, Thermo, Cat# a21447). After incubation, the slices were rinsed six times for 5 min each with PBST and dried. The slices were mounted with Antifade Mounting Medium containing DAPI (Beyotime, P0131) and stored at 4 °C. High-resolution fluorescent images were obtained using a laser confocal microscope LSM 880 with Airyscan 2 (Zeiss, Oberkochen, Germany).
Stereotaxic surgery
Mice were anesthetized with sodium pentobarbital (50 mg/kg) and fixed on a stereotaxic frame (Kopf Instruments, Model 962, Tujunga, CA) before making an incision over the scalp. Next, a borehole (0.5 mm diameter) was drilled. According to the mouse brain atlas [28], viral vectors were microinjected bilaterally via a syringe with a 33-gauge metal needle (Hamilton Company, Reno, Nevada) at a volume of 200 nL per site and a rate of 50 nL/min. After microinjection, the needle was left in the brain for 15 min to allow sufficient time for viral absorption. Data from mice with injection sites that were histologically identified to be deviated from the target brain region were excluded from further analysis. To downregulate IL-17RA in cerebellar Crus Ⅰ, pSLenti-U6-shRNA (scrambled)-CMV-mCherry-F2A-Puro-WPRE or pSLenti-U6-shRNA (IL-17RA)-CMV-mCherry-F2A-Puro-WPRE (Obio Technology, Shanghai, China) was used. For selective downregulation of IL-17RA in PCs, a mixture of rAVV-L7-CRE-WPRE-hGHpA (Brainvta, Wuhan, China) and pAAV-hSyn-DIO-shRNA (IL17RA)-eGFP/pAAV-hSyn-DIO-shRNA (scrambled)-eGFP (Shanghai Sunbio Medical Biotechnology, Shanghai, China) was injected into bilateral Crus Ⅰ of the cerebellum (A/P: −6.48 mm, M/L: ±2.5 mm, D/V: −2.0 mm).
For the administration of IL17A or IL17RAb into Crus Ⅰ, two cannulas (RWD) were implanted into bilateral Crus Ⅰ of the cerebellum (A/P: −6.48 mm, M/L: ±2.5 mm, D/V: −1.5 mm, lambda was raised 500 μm above bregma). The cannulas were fitted with dummy cannulas to maintain patency following surgery.
Drug administration
PLX3397 (MedChemExpress, CAS No. 1029044-16-3) was dissolved in DMSO, diluted with corn oil, and mixed into standard chow (290 ppm), which was then fed for 2 weeks to deplete microglia. To induce the immune response, LPS (50 μg/kg) and poly(I:C) (10 mg/kg) were injected intraperitoneally.
Behavioral tests
We used the three-chamber test and marble burying test to evaluate ASD-like behaviors, following previous studies [9, 30, 31]. All tasks were conducted in dim light.
Three-chamber test
As reported previously [7], during the habituation stage, the test mouse was placed in the three-chamber apparatus (Clever TopScan, Reston, USA) for 10 min with the doors opened to determine the preferred side chamber. In the test stage, the mouse was guided to the center chamber, and the doors were closed. A cup containing an object was introduced into the preferred side chamber, while a cup with a stranger mouse was introduced into the other non-preferred side chamber. The doors were then opened to conduct the sociability test, and the movements of the test mouse were tracked for 10 min. For each set of experiments, the orientation of the two cups containing the stranger mouse or the object was counterbalanced. The preference index was calculated to reflect the sociability of the experimental mice, using the formula (Time stranger - Time object)/(Time stranger + Time object) as the preference index.
For the administration of IL-17A or IL-17R antibody, the tests were performed as previously described with minor modifications. Adult male mice were implanted with cannulas into Crus Ⅰ bilaterally and allowed to recover for 1 week prior to the behavioral experiments. During the habituation stage, the test mice were placed in the three-chamber apparatus to exclude location preference. Before the test stage, mice were briefly anesthetized using isoflurane, and either saline or IL-17A (50 ng/side in 0.5 μL at a rate of 0.2 μL/min and 4 h before behavioral tests, R&D, 7956-ML/CF) or IL-17R Ab (10 μg/side in 0.5 μL at a rate of 0.2 μL/min and 0.5 h before behavioral tests, R&D, CAT# MAB4481) was administered bilaterally into Crus Ⅰ before assessing sociability. Additionally, mice received intraperitoneal injections of saline, LPS, or poly(I:C) 6 h before behavioral tests.
Marble burying test
Mice were individually placed in a home-cage-like environment (37.5 × 27 × 16.5 cm) containing 5-cm-thick corncob bedding, with 20 black glass marbles (15 mm in diameter) arranged in a 5 × 4 grid across the whole cage. The mice were left undisturbed for 30 min, and the number of marbles that were at least two-thirds buried at the end of the trial was counted.
Statistical analysis
For all experiments in this study, mice were randomly assigned to groups before surgery and drug treatment. Investigators and data analysts were all blinded to group allocations. Sample sizes were determined based on previous similar studies conducted by our laboratory and others [29, 31]. Data were analyzed using GraphPad 7.0. Two-sided tests were used throughout and results were presented as mean ± SEM. Statistical analyses included unpaired t-tests, paired t-tests, and one-way or two-way analysis of variance (ANOVA) with Tukey’s post hoc tests or Dunnett’s post hoc test. P values of < 0.05 were considered significant. The exact sample size (n) for each group is indicated in the figure legends. Full statistical data are presented in Supplementary Table 2.
Results
IL-17A expression in the cerebellum is elevated in Fmr1-KO mice
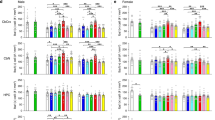
To determine the role of IL-17A in inherited ASD, we employed Fmr1-KO mice, a well-established genetic model for ASD. Fmr1-KO mice displayed typical ASD-related behaviors, including a lack of preference for the novel mouse over the novel object (Fig. S1a, b) and increased marble-burying behavior (Fig. S1c), which are hallmark features of social impairment and repetitive behaviors in ASD. Given the critical role of the cerebellum, particularly PCs of the Crus I lobule, in the pathogenesis of ASD [14, 15, 32], we examined alterations in the neuronal activity of Crus I PCs in Fmr1-KO mice. Whole-cell patch-clamp recordings revealed a significant decrease in the spontaneous firing rate of PCs in Fmr1-KO mice (Fig. 1a, b). In addition, we observed an increase in the amplitude, but not the frequency, of spontaneous inhibitory postsynaptic currents (sIPSCs) in PCs of Fmr1-KO mice (Fig. 1c–e). In contrast, there were no significant differences in the amplitude or frequency of spontaneous excitatory postsynaptic currents (sEPSCs) recorded in PCs between Fmr1-KO and WT mice (Fig. S1d–f). These alternations in the spontaneous firing and synaptic transmission of cerebellar PCs are consistent with previously reported changes in PC electrophysiological properties from other ASD models, such as Shank2-deficient or Tsc1-mutant mice [14, 33].
a Spontaneous firing rates of PCs recorded from cerebellar slices of WT and Fmr1-KO mice. b Group data showing that the spontaneous firing rates of PCs were significantly decreased in Fmr1-KO mice (WT, n = 6 cells from 3 mice; Fmr1-KO mice, n = 6 cells from 3 mice). c Raw current traces showing sIPSCs of cerebellar PCs in WT and Fmr1-KO mice. d, e Group data showing that the amplitude of sIPSCs was increased in PCs of Fmr1-KO mice, while no significant changes were observed in frequency (WT, n = 6 cells from 4 mice; Fmr1-KO mice, n = 6 cells from 2 mice). f Volcano plots of DEGs in the cerebellum of WT and Fmr1-KO mice (n = 4 mice for each group; fold change > 1.5 or fold change < 0.67, P < 0.05). g KEGG enrichment analysis of DEGs showing that upregulated DEGs were primarily associated with immune response-related pathways (including IL-17 signaling pathway), synaptic plasticity, and neuronal activity (P < 0.1). h, i qPCR (WT, n = 5 mice; Fmr1-KO, n = 5 mice) and electrochemiluminescence immunoassay (WT, n = 5 mice; Fmr1-KO, n = 8 mice) showing a significant increase in the relative expression levels of IL-17A mRNA and protein in the cerebellar Crus I of Fmr1-KO mice. j, k Relative IL-17RA mRNA (WT, n = 7 mice; Fmr1-KO, n = 6 mice) and protein expression (WT, n = 8 mice; Fmr1-KO, n = 8 mice) were elevated in the cerebellar Crus I of Fmr1-KO mice. Data are presented as mean ± SEM. *P < 0.05, **P < 0.01, ***P < 0.001, n.s. indicates not significant.
To further explore the pathological and compensatory changes associated with ASD, we conducted transcriptome sequencing to profile gene expression alterations in the Crus I lobule of the cerebellum in Fmr1-KO mice. As shown in Fig. 1f, 380 up-regulated and 300 down-regulated differentially expressed genes (DEGs) were identified between Fmr1-KO and WT mice (fold change > 1.5 or fold change < 0.67, P < 0.05). Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway analysis suggested that these DEGs were mainly enriched in immune response, synaptic plasticity, and neuronal activity, including pathways such as antigen processing and presentation, cell adhesion molecules, cytokine-cytokine receptor interactions, and neuroactive ligand-receptor interactions (Fig. 1g) [34,35,36,37,38]. Notably, the IL-17 signaling pathway was significantly activated in Fmr1-KO mice (Fig. 1g). Therefore, we further analyzed the expression of members of the IL-17 family. The results of qPCR revealed that mRNA expression levels of IL-17A, but not IL-17B-F, were remarkably elevated in the cerebellar Crus I of Fmr1-KO mice (Fig. 1h and Fig. S1g–k). Consistently, Fmr1-KO mice showed an increased expression of IL-17A protein in the cerebellar Crus I lobule (Fig. 1i). Additionally, the mRNA and protein levels of IL-17 receptor A (IL-17RA), which is the main receptor subtype for IL-17A ligand, were also upregulated in the Crus I lobule of Fmr1-KO mice (Fig. 1j, k). These results suggest a pronounced elevation of IL-17A and the activation of its signaling pathway in the cerebellum of Fmr1-KO mice.
Cerebellar IL-17A is solely produced by microglia to enhance sociability by activating PCs
Next, we identified the source of IL-17A in the cerebellum. Given the increased expression of IL-17A mRNA and protein in the cerebellum of Fmr1-KO mice, we hypothesized that IL-17A may be locally produced by resident CNS cells rather than by peripheral immune cells, as is typically the case. In WT mice, we found that IL-17A mRNA was more highly expressed in the cerebellum than in the hippocampus (Fig. 2a), consistent with previous reports of IL-17A and IL-17RA expression in the cerebellum. Immunofluorescent staining revealed that IL-17A immunopositive signals were colocalized with Iba1-positive microglia, but not with calbindin-positive PCs, S100β-positive astrocytes, or CD3-positive T cells, suggesting that IL-17A in the cerebellum is exclusively produced by local resident microglia (Fig. 2b). In Fmr1-KO mice, the observed increase in IL-17A protein levels in the cerebellar Crus I lobule (Fig. 1i) was corroborated at the cellular level by a marked increase in IL-17A immunopositive signals that colocalized with Iba1-positive microglia (Fig. 2c). Quantitative immunofluorescence analysis further revealed that this upregulation was not a late-onset phenomenon but was already evident at the early postnatal stage P7 and persisted throughout key developmental periods (P14 and P21; Fig. 2c) and into adulthood (8 weeks; Fig. 2c).
a Relative IL-17A mRNA expression in the hippocampus (Hippo) and cerebellum of WT mice, showing significantly higher expression in the cerebellum (Hippo, n = 7 mice; Cerebellum, n = 8 mice). b Immunofluorescence images showing IL-17A (green) selectively colocalized with Iba1-positive microglia (red), but not in S100β-positive astrocytes (red), calbindin-positive PCs (red), or CD3-positive T cells (red) in the cerebellum of WT mice. c Immunofluorescent co-labelling of IL-17A (green) and the microglial marker Iba1 (red) in the cerebellar Crus I of WT and Fmr1-KO mice at postnatal days (P) 7, 14, and 21 and adult (8 weeks). Quantification of the mean gray value of IL-17A within Iba1-positive areas in the cerebellar Crus I at P7 (WT, n = 5 mice; Fmr1-KO, n = 4 mice), 14 (WT, n = 5 mice; Fmr1-KO, n = 4 mice), 21 (WT, n = 5 mice; Fmr1-KO, n = 5 mice) and adult (WT, n = 4 mice; Fmr1-KO, n = 5 mice). d Western blot analysis showing higher IL-17RA protein levels in the cerebellum compared with the hippocampus (Hippo, n = 6 mice; Cerebellum, n = 6 mice). e Immunofluorescence signals for IL-17RA (red) are selectively localized in calbindin-positive PCs (green). f Immunofluorescent staining showing IL-17RA (red) were selectively localized in calbindin-positive PCs (green, left), but not in microglia (green, right) of the cerebellar Crus I. g IL-17RA immunofluorescence is selectively enriched in the cerebellar Crus I of Fmr1-KO mice Representative confocal images and quantification show higher IL-17RA signal in Crus I compared with hippocampus, PFC and S1DZ (Cerebellum, n = 5 mice; Hippo, n = 5 mice; PFC, n = 4 mice; S1DZ, n = 6 mice). Data are presented as mean ± SEM. *P < 0.05, **P < 0.01. ***P < 0.001.
Since IL-17A signals through the IL-17RA [39], we further explored the expression and distribution of IL-17RA in the cerebellum in WT and Fmr1-KO mice. In WT mice, IL-17RA protein levels, like those of IL-17A, were significantly elevated in the cerebellum compared to the hippocampus (Fig. 2d). Immunofluorescent staining revealed that IL-17RA was selectively expressed in calbindin-positive PCs, indicating a specific distribution of IL-17RA in cerebellar PCs (Fig. 2e). Importantly, this expression pattern was conserved in Fmr1-KO mice, with IL-17RA remaining confined to PCs and absent from microglia (Fig. 2f). Given that multiple brain regions, including the hippocampus, PFC, and S1DZ, have been implicated in ASD pathology [9, 40, 41], we extended our analysis to assess IL-17RA expression in these areas in Fmr1-KO mice. Notably, among all ASD-relevant regions examined, the cerebellum exhibited the highest level of IL-17RA expression (Fig. 2g), underscoring the particular importance of cerebellar IL-17A signaling in the context of FXS.
To clarify the function of microglia-derived IL-17A in the cerebellum, we assessed the effects of IL-17A on neuronal activity in Crus I PCs and on social behaviors in WT mice. Bath application of IL-17A (10 ng/mL) significantly increased the spontaneous firing frequency of PCs (Fig. 3a, b) and suppressed the amplitude, but not the frequency of sIPSCs (Fig. 3c–e). However, IL-17A had no significant effect on the amplitude or frequency of sEPSCs in PCs (Fig. 3f–h). Moreover, behavioral analysis showed that microinjection of IL-17A (50 ng) into the bilateral cerebellar Crus I lobules significantly increased the preference of mice for an enclosed novel mouse in the three-chamber test (Fig. 3i–k). However, microinjection of IL-17A had no effect on marble-burying behavior, likely due to the already low baseline marble-burying observed in WT mice (Fig. 3l). These results suggest that cerebellar IL-17A enhances sociability, rather than induces ASD-like behaviors, possibly by increasing the firing rates and suppressing inhibitory synaptic strength of PCs.
a Spontaneous firing rates of PCs recorded from cerebellar slices before and during the application of IL-17A (10 ng/mL, 10 min). b Whole-cell patch-clamp recordings showed that bath incubation with IL-17A increased the firing rates of cerebellar PCs (n = 13 cells from 2 mice). c-e IL-17A bath incubation decreased the amplitude, but did not affect the frequency of sIPSCs in cerebellar PCs (n = 6 cells from 3 mice). f-h IL-17A bath incubation had no impact on the amplitude or frequency of sEPSCs in cerebellar PCs (n = 6 cells from 4 mice). i Schematic diagram illustrating the procedures for behavioral tests. IL-17A (50 ng/side) or vehicle was microinjected into the cerebellar Crus I 4 h prior to the behavioral tests. j Heatmaps from the three-chamber test showing social behavior in WT mice after microinjection of vehicle or IL-17A into the cerebellar Crus I. k Microinjection of IL-17A significantly increased both social interaction time and the social preference index with stranger mice in WT mice (Vehicle, n = 9 mice; IL-17A, n = 10 mice). l Microinjection of vehicle or IL-17A into the cerebellar Crus I did not affect the number of buried marbles (Vehicle, n = 8 mice; IL-17A, n = 7 mice). Data are presented as mean ± SEM. *P < 0.05, ***P < 0.001, n.s. indicates not significant.
We negatively manipulated IL-17A signaling in the cerebellar Crus I lobule to further confirm its facilitation effect on PC excitability and sociability in WT mice. Cerebellar slice preparations were exposed to a neutralizing antibody against IL-17RA (0.5 μg/mL), and whole-cell patch-clamp recordings showed that blocking IL-17RA remarkably decreased the spontaneous firing rate of PCs (Fig. S2a–c). In addition, IL-17RA blockade increased the amplitude of sIPSCs without affecting their frequency (Fig. S2d–f). Behavioral results showed that microinjection of IL-17RA antibody into the bilateral Crus I lobules significantly reduced the sociability (Fig. S2g, h) and increased marble-burying behavior (Fig. S2i). Consistent with the pharmacological manipulations, knockdown of IL-17RA in Crus I by delivery of LV-shRNA (IL-17RA)-mCherry also induced ASD-like social disability and repetitive behaviors (Fig. S3). Given that IL-17RA is selectively expressed in PCs, we further assessed the contribution of PCs’ IL-17A signaling to ASD by specifically expressing IL-17RA-targeting shRNA in Crus I PCs. We microinjected AAV expressing Cre under the control of the PC-specific L7 promoter (AAV-L7-Cre) and a Cre-dependent virus carrying either IL-17RA-targeting shRNA (AAV-DIO-shRNA (IL-17RA)-eGFP) or scrambled shRNA (AAV-DIO-shRNA (scrambled)-eGFP) into bilateral Crus I lobules (Fig. 4a). As shown in Fig. 4b, c, eGFP expression was specifically detected in calbindin-positive PCs. In the shRNA group, IL-17RA expression in PCs was significantly reduced (Fig. 4c). Knockdown of IL-17RA in PCs led to a significant decrease in the spontaneous firing rate of PCs (Fig. 4d, e) and an increase in the amplitude, but not the frequency, of sIPSCs (Fig. 4f–h). As a result, mice with IL-17RA knockdown in PCs of cerebellar Crus I exhibited ASD-like behavioral traits, including reduced sociability with strangers in the three-chamber test (Fig. 4i) and increased marble-burying behavior (Fig. 4j). Collectively, these results suggest that cerebellar IL-17A, produced by microglia, plays a crucial role in facilitating the excitability of PCs and promoting sociability. Therefore, the elevated IL-17A levels in the cerebellar Crus I lobule of Fmr1-KO mice may serve as a compensatory, rather than punitive, mechanism to mitigate ASD-related behavioral abnormalities, including social deficits and repetitive behaviors.
a Schematic diagrams illustrating the procedures for electrophysiological recordings and behavioral tests used to assess the effects of selective knockdown of IL-17RA in cerebellar PCs. b, c Confocal image showing immunofluorescence labeling for DIO-eGFP (green), calbindin (red), and IL-17RA (gray). The Cre-dependent virus AAV-DIO-shRNA was locally expressed in cerebellar Crus I PCs. IL-17RA signals in calbindin-positive PCs were significantly reduced in the L7-shIL-17RA group (n = 4 mice) compared to the sham group (n = 4 mice). d, e Whole-cell patch-clamp recordings showing that knockdown of IL-17RA in cerebellar PCs reduced their spontaneous firing rates (Sham, n = 11 cells from 2 mice; shIL-17RA, n = 14 cells from 2 mice). f-h Knockdown of IL-17RA in cerebellar PCs increased the amplitude of sIPSCs (Sham, n = 14 cells from 2 mice; shIL-17RA, n = 12 cells from 2 mice), but did not affect their frequency (Sham, n = 14 cells from 2 mice; shIL-17RA, n = 12 cells from 2 mice). i, j Behavioral tests revealed that knockdown of IL-17RA in cerebellar Crus I PCs decreased social interaction time and social preference index with strangers (Sham, n = 10 mice; shIL-17RA, n = 8 mice), and increased the repetitive stereotyped behaviors (Sham, n = 11 mice; shIL-17RA, n = 10 mice). Data are presented as mean ± SEM. *P < 0.05, **P < 0.01, ***P < 0.001, n.s. indicates not significant.
IL-17A rescues hypoexcitability in PCs and improves ASD-like behaviors in Fmr1-KO mice
We next examined whether the IL-17A-mediated compensatory mechanism in Fmr1-KO mice could serve as a potential therapeutic target for ASD. We tested the effects of intra-Crus I microinjection of IL-17A (10 ng/mL) on aberrant PC activity and ASD-related behavioral phenotypes in Fmr1-KO mice. IL-17A administration significantly restored the reduced spontaneous firing rate of PCs (Fig. 5a, b) and selectively suppressed the elevated amplitude of sIPSCs (Fig. 5c–e). The selective reduction in sIPSC amplitude, without change in frequency, suggests a postsynaptic mechanism likely involving the expression or function of GABAA receptors (GABAARs) [42]. Consistent with this interpretation, qPCR and western blot analyses revealed that IL-17A microinjection downregulated both the mRNA and protein levels of GABAAR in Crus I (Fig. S4a, b). Immunofluorescence staining further confirmed a decrease in GABAAR signal intensity specifically within PCs (Fig. S4c). We also assessed gephyrin, a scaffolding protein essential for postsynaptic clustering and stabilization of GABAARs [43], and found that IL-17A treatment reduced gephyrin levels in PC dendrites (Fig. S4d), supporting a postsynaptic reorganization of inhibitory synapses.
a, b Whole-cell patch-clamp recordings showing that IL-17A bath incubation (10 ng/mL, 10 min) increased the spontaneous firing rates of PCs in Fmr1-KO mice. This effect was blocked by co‑application of the NF‑κB inhibitor PDTC (1.64 ng/mL, 10 min), with no significant difference observed between the Fmr1‑KO + PDTC and Fmr1‑KO + PDTC + IL‑17 A groups (WT, n = 9 cells from 4 mice; Fmr1-KO/Fmr1-KO + IL-17A, n = 5 cells from 3 mice; Fmr1-KO + PDTC /Fmr1‑KO + PDTC + IL‑17 A, n = 5 cells from 3 mice). c-e Bath incubation with IL-17A (10 ng/mL, 10 min) reduced the amplitude of sIPSCs in PCs, but did not affect their frequency in Fmr1-KO mice. The effect of IL‑17 A on sIPSC amplitude was abolished in the presence of PDTC (1.64 ng/mL, 10 min) (WT, n = 7 cells from 2 mice; Fmr1-KO/Fmr1-KO + IL-17A, n = 6 cells from 2 mice; Fmr1-KO + PDTC /Fmr1‑KO + PDTC + IL‑17 A, n = 6 cells from 3 mice). f Schematic diagram illustrating the protocol for behavioral assessment: IL-17A (50 ng/side) or vehicle was microinjected into cerebellar Crus I 4 h prior to testing. g, h Microinjection of IL-17A into Crus I significantly improved social interaction time and the social preference index with stranger mice (WT, n = 8 mice; Fmr1-KO, n = 12 mice; Fmr1-KO + IL-17A, n = 8 mice), and reduced the number of buried marbles in Fmr1-KO mice. (WT, n = 7 mice; Fmr1-KO, n = 5 mice; Fmr1-KO + IL-17A, n = 8 mice). Data are presented as mean ± SEM. *P < 0.05, **P < 0.01, ***P < 0.001, n.s. indicates not significant.
To elucidate the signaling mechanism underlying the IL-17A-induced restoration of PC excitability, we investigated the canonical NF-κB pathway downstream of IL-17R activation, given its established role in modulating neuronal excitability and GABAAR expression [44, 45]. IL-17A treatment robustly activated NF-κB signaling in the Fmr1-KO cerebellum, as indicated by increased phosphorylation and translocation of the p65 subunit in PCs (Fig. S5a, b). Notably, pre-treatment with the NF-κB inhibitor PDTC completely abolished the restorative effects of IL-17A on both spontaneous firing and sIPSC amplitude (Fig. 5a–e), demonstrating that NF-κB activation is required for IL-17A-induced modulation of inhibitory synaptic transmission and the consequent normalization of PC excitability.
Furthermore, intra-Crus I IL-17A microinjection (50 ng) markedly increased sociability, as shown by a heightened preference for social interactions (Fig. 5f, g), and reduced repetitive behavior, as evidenced by a decrease in marble-burying activity (Fig. 5h). These results suggest that the IL-17A signaling pathway in the cerebellum may provide a promising therapeutic target for ASD.
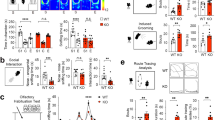
Poly(I:C) treatment enhances microglia-derived IL-17A in the cerebellum and ameliorates ASD-like phenotypes in Fmr1-KO mice
Next, we explored strategies to enhance cerebellar IL-17A signaling. It has been reported that low-dose intraperitoneal injection of bacterial endotoxin LPS induces peripheral cytokine secretion, including IL-17A, to mediate the “fever effect” in MIA offspring, but not in monogenic ASD models [9]. Since fever can result from both bacterial and viral infections, we determined the effects of poly(I:C), a synthetic analogue of viral dsRNA widely used in clinical tumor immunotherapy [46, 47], on IL-17A production and social deficits in Fmr1-KO mice. As shown in Fig. 6a–c, 6 h following intraperitoneal injection, poly(I:C) (10 mg/kg) significantly increased IL-17A mRNA and protein levels in the cerebellum, whereas LPS (50 μg/kg) had no such effect. In addition, unlike LPS, which caused a dramatic increase in pro-inflammatory factors, poly(I:C) induced only a slight elevation of TNF-α, IL-6, IFN-γ in Crus I (Fig. S6a, b). Moreover, poly(I:C) injection did not result in the loss of calbindin-positive PCs (Fig. S6d). Similarly, IL-17A treatment resulted in a mild increase in TNF-α, IL-6, IFN-γ within Crus I, without causing neuronal damage (Fig. S6c, d).
a, b Relative IL-17A mRNA expression 3 or 6 h after i.p. injection of LPS (Fmr1-KO, n = 4 mice; Fmr1-KO + 3 h LPS, n = 4 mice; Fmr1-KO + 6 h LPS, n = 4 mice) or poly(I:C) (Fmr1-KO, n = 7 mice; Fmr1-KO + 3 h poly(I:C), n = 8 mice; Fmr1-KO + 6 h poly(I:C), n = 7 mice). c Immunofluorescent staining for IL-17A (green) and DAPI (blue) showing that poly(I:C) significantly increased IL-17A expression in the cerebellar Crus I (Fmr1-KO, n = 5 mice; Fmr1-KO + poly(I:C), n = 5 mice). d A schematic representation illustrating the purification of cerebellar microglia. Created with Biorender.com. e Volcano plots showing DEGs of purified samples from the cerebellar Crus I of poly(I:C)-treated Fmr1-KO mice compared to untreated Fmr1-KO mice (n = 3 mice for each group; fold change > 1.2 or fold change < 0.84, P < 0.1). f KEGG enrichment analysis of DEGs indicated that poly(I:C) activated immune response-related pathway, including IL-17 signaling pathway (P < 0.05), in cerebellar microglia. g The gating strategy and quantification of IL-17A-positive cerebellar microglia in WT, Fmr1-KO, and poly(I:C)-treated Fmr1-KO mice. Poly(I:C) increased the ratio of IL-17A-producing cerebellar microglia in Fmr1-KO mice (WT, n = 6 mice; Fmr1-KO, n = 6 mice; Fmr1-KO + poly(I:C), n = 6 mice). h Immunofluorescence staining showing that poly(I:C) treatment elevated the number of Arc-positive (green) cerebellar PCs (calbindin-positive cells, green) in Fmr1-KO mice (Fmr1-KO, n = 8 mice; Fmr1-KO + poly(I:C), n = 8 mice). i, j Pharmacological blockade of IL-17A signaling in PCs of the cerebellar Crus I by microinjection of IL-17RA Ab abolished the poly(I:C)-induced improvement of social deficits (WT, n = 7 mice; Fmr1-KO, n = 7 mice; Fmr1-KO + poly(I:C), n = 6 mice; Fmr1-KO + poly(I:C) + IL17RA Ab, n = 7 mice) and reduced repetitive stereotyped behaviors (WT, n = 7 mice; Fmr1-KO, n = 6 mice; Fmr1-KO + poly(I:C), n = 11 mice; Fmr1-KO + poly(I:C) + IL17RA Ab, n = 6 mice). Data are presented as mean ± SEM. *P < 0.05, **P < 0.01, ***P < 0.001, n.s. indicates not significant.
To determine whether the poly(I:C)-induced elevation of IL-17A in the cerebellum was due to microglia or peripheral immune cells, we isolated the cerebellar cortical microglia using magnetic-activated cell sorting (MACS) followed by Switching Mechanism At 5’ end of the RNA Transcript (SMART)-sequencing [48] (Fig. 6d and Fig. S7a). The DEGs were identified using fold change > 1.2 or fold change < 0.83, P < 0.1 (Fig. 6e). KEGG analysis revealed that the IL-17 signaling pathway in cerebellar microglia was significantly enhanced in poly(I:C)-treated Fmr1-KO mice compared to vehicle-treated controls (Fig. 6f), consistent with data from whole cerebellum samples (Fig. S7b, c). These results suggest that microglia are a major source of elevated IL-17A in the cerebellum following poly(I:C) treatment. Flow cytometry further confirmed that poly(I:C) treatment remarkably increased the proportion of IL-17A-expressing microglia in Fmr1-KO mice (Fig. 6g).
Finally, we assessed the impact of poly(I:C) treatment and microglial IL-17A release on PC activity and ASD-like behavioral deficits. As shown in Fig. 6h, poly(I:C)-treated Fmr1-KO mice exhibited increased expression of activity-regulated cytoskeleton-associated (Arc) protein in Crus I PCs, indicating subsequent behavioral effects. Indeed, poly(I:C) treatment significantly improved sociability and reduced repetitive behaviors in Fmr1-KO mice (Fig. 6i, j). Moreover, blockade of Crus I IL-17A signaling by microinjection of antibody against IL-17RA prevented the poly(I:C)-induced improvement in sociability and marble-burying behavior (Fig. 6i, j). To further evaluate the contribution of cerebellar microglia to the therapeutic effect of poly(I:C), we pharmacologically depleted them using PLX3397, a CSF1R inhibitor (Fig. S8a). PLX3397 treatment significantly reduced microglia density in the cerebellum of Fmr1-KO mice (Fig. S8b, c) and lowered cerebellar IL-17A mRNA levels of in poly(I:C)-treated Fmr1-KO mice (Fig. S8d). As a result, the poly(I:C)-induced amelioration of social preference was abolished in microglia-depleted mice (Fig. S8e). These results suggest that poly(I:C) treatment enhances microglia-derived IL-17A in the cerebellum, leading to the amelioration of ASD-like behaviors in Fmr1-KO mice.
Given the well-documented higher prevalence and greater symptom severity of FXS in male patients [49], all primary experiments were conducted in male Fmr1-KO mice to elucidate underlying disease mechanisms. Nonetheless, sex-specific outcomes, such as those observed following lactational poly(I:C) exposure [50], highlight the importance of considering sex as a biological variable. Notably, our extended analyses revealed that poly(I:C)-induced microglial IL-17A release also effectively ameliorated behavioral deficits in female Fmr1-KO mice (Fig. S9). This finding demonstrates that the therapeutic efficacy of this immunomodulatory strategy is preserved across sexes in adult Fmr1-KO mice, thereby broadening its potential translational relevance.
Discussion
IL-17A is a highly versatile cytokine traditionally associated with immune responses, primarily produced by peripheral Th17 cells, and has been implicated in both protective immunity and the pathogenesis of inflammatory diseases [51]. Over the past few years, increasing attention has been directed towards understanding the role of IL-17A in brain function. It is known to mediate peripheral immune activation signals to the brain [52, 53], yet the precise role of IL-17A derived from brain-resident cells remains largely unexplored. In this study, we demonstrate that IL-17A, produced by cerebellar microglia, plays a critical role in regulating the synaptic strength and neuronal firing of PCs through the NF-κB signaling pathway, thereby directly influencing social behaviors. Notably, knocking down IL-17RA in PCs induced ASD-like behaviors in WT mice, whereas activating IL-17A signaling ameliorated ASD-related deficits in Fmr1-KO mice. Furthermore, treatment with poly(I:C), a synthetic viral mimic, significantly enhanced IL-17A production in cerebellar microglia and ameliorated the social and repetitive behavioral deficits of Fmr1-KO mice. These findings suggest that IL-17A signaling in the cerebellum could be a potential therapeutic target for treating ASD.
IL-17A, traditionally recognized as a key mediator of immune responses, has more recently been appreciated for its conserved role in neuroimmune communication, influencing neuronal activity and shaping behaviors [54]. Even in the absence of infection, IL-17A modulates brain function [52]. In Caenorhabditis elegans, IL-17 directly regulates neuronal responsiveness to oxygen and associated aggregation behaviors, underscoring its ancient evolutionary role in neural modulation [55]. In mammals, IL-17A acts on neurons in the PFC and hippocampus to regulate anxiety and memory [41, 56, 57]. While meningeal γδ T cells have been identified as a primary source of IL-17A at homeostasis [52], accumulating evidence indicates that microglia also produce IL-17A within the hippocampus and striatum [58, 59]. Extending this concept to the cerebellum, our study demonstrates that microglia-derived IL-17A critically regulates the activity of PCs, the principal integrators of cerebellar afferent signals [60]. Pharmacological blockade or genetic knockdown of IL-17RA in PCs significantly impaired social behaviors, indicating that cerebellar IL-17A is essential for maintaining social function. Unlike meningeal-derived IL-17A, which diffuses through the cerebrospinal fluid [52], cerebellar microglia-derived IL-17A may provide a more rapid and spatially precise mechanism for modulating local neuronal homeostasis.
The functional consequences of IL-17A signaling are highly context- and region-dependent. Elevated IL-17A can impair hippocampal synaptic plasticity in models of multiple sclerosis [58] and trigger inflammatory cascades in the cerebellum under hyperammonemia conditions [61]. In contrast, we show that in the Fmr1-KO cerebellum, microglia-produced IL-17A restores PC excitability and inhibitory synaptic balance without inducing overt neuroinflammation. Comparative analyses further revealed that IL-17RA expression is highest in the cerebellum among ASD-related regions, including the PFC, hippocampus, and S1DZ, suggesting that the cerebellum is a major target of IL-17A signaling in FXS. While IL-17A likely exerts effects in other ASD-related areas [9, 40, 41], its pronounced receptor enrichment in the cerebellum, together with the robust behavioral rescue observed following local interventions, underscores the particular importance of this region in mediating IL-17A’s beneficial actions.
Social deficits are a core symptom of ASD [1, 62], and the cerebellum, particularly the Crus I region of the cerebellar cortex, has emerged as a key structure in its pathophysiology. Cerebellar abnormalities are among the most consistently reported findings in ASD patients [63, 64]. Here, we identify a significant upregulation of IL-17A mRNA in the cerebellar Crus I lobule of Fmr1-KO mice. Notably, we observed that IL-17A, but not other isoforms like IL-17B-F, was elevated in the cerebellum of ASD mice, and this elevation was primarily attributed to resident microglia. Given that serum IL-17A levels are increased in ASD patients [11], our data suggest that cerebellar microglia may represent an underrecognized source of brain IL-17A in ASD. Importantly, IL-17A delivery ameliorated social and repetitive behavioral deficits, indicating a compensatory and functional role for cerebellar IL-17A signaling. The early onset of IL-17A upregulation, detectable from P7 and sustained through P21, coincides with critical periods of PC electrophysiological maturation [65], suggesting that this represents a developmentally programmed rather than a reactive phenomenon. We therefore propose that loss of Fmr1 intrinsically alters microglial developmental states, leading to sustained activation and autonomous IL-17A production, which in turn initiates a compensatory neuroimmune response during early circuit formation.
Aberrant PC firing and synaptic inhibition are well-established features of Fmr1-KO and other ASD models [66, 67]. Here, exogenous IL-17A restored normal firing rates and reduced excessive inhibitory inputs to PCs, rescuing these core neurophysiological abnormalities. This aligns with prior evidence that modulating cerebellar Crus I PC activity can alleviate social deficits in ASD [14, 15, 68]. At the molecular level, IL-17A treatment decreased both GABAAR and gephyrin expression, consistent with the observed reduction in sIPSC amplitude, together supporting a coherent mechanism of postsynaptic modulation at inhibitory synapses. Mechanistically, we identified NF-κB as the key downstream pathway of IL-17RA activation [51]. IL-17A robustly increased NF-κB p65 phosphorylation, and pharmacological inhibition of NF-κB abolished the IL-17A-mediated rescue of PC firing and inhibitory tone. These results define a signaling axis wherein microglial IL-17A activates canonical NF-κB signaling in PCs, leading to transcriptional modulation of inhibitory synaptic components and normalization of cerebellar output in the Fmr1-KO model.
The phenomenon known as the “fever effect”, temporary behavioral improvement in ASD during febrile episodes, has been reported in subsets of individuals [3,4,5]. Immune activation, rather than hyperthermia itself, has been implicated in this beneficial effect. Both bacterial (LPS) and viral (poly(I:C)) mimetics can induce such immune responses, but IL-17A has been shown to mediate the fever-associated behavioral rescue primarily in maternal immune activation (MIA) models, not in monogenic mutations [9]. Remarkably, our study demonstrates that poly(I:C), but not LPS, triggers cerebellar microglial IL-17A release in Fmr1-KO mice, independent of prior immune priming, leading to a robust improvement in ASD-like behaviors. Microglia depletion abolished these effects, confirming that poly(I:C)-induced behavioral rescue is mediated by microglia-derived IL-17A. These results provide a mechanistic link between immune activation and behavioral improvement, offering new insight into the “fever effect” and identifying a potential therapeutic avenue for heritable ASD forms. Given that both poly(I:C) and IL-17A induce only mild immune activation, do not cause PC loss, and that poly(I:C) has an established safety record in cancer immunotherapy [69,70,71], this approach may represent a viable strategy for clinical translation. Furthermore, although sex-specific differences have been observed following early-life immune challenges [50], poly(I:C) treatment elevated cerebellar IL-17A levels and improved behavioral outcomes in both male and female Fmr1-KO mice, underscoring the robustness and generalizability of this immunomodulatory effect.
While cerebellar neuroimmune interactions likely involve multiple cell types and signaling molecules, our data converge on a central conclusion: microglia-derived IL-17A acting directly on PCs is both necessary and sufficient for the therapeutic effects of poly(I:C) and IL-17A in Fmr1-KO mice. Although contributions from co-released mediators or other immune cell types cannot be fully excluded, three lines of evidence substantiate this pathway: i) poly(I:C)-induced behavioral improvement was abolished by microglial depletion; ii) local blockade of IL-17A signaling in Crus I prevented poly(I:C)-induced rescue of social and repetitive behaviors; and iii) developmental profiling identified microglia as the predominant IL-17A source in the Fmr1-KO cerebellum. Together, these findings establish the microglia-PC IL-17A signaling axis as the key mediator of synaptic and behavioral recovery in this FXS model.
In conclusion, our study uncovers a novel, microglia-dependent role for IL-17A in regulating cerebellar PC function and social behavior. Disruption of IL-17A signaling in the cerebellum produces ASD-like phenotypes, whereas its compensatory enhancement restores synaptic and behavioral function. Poly(I:C)-induced microglial IL-17A release further recapitulates this rescue, providing a potential therapeutic mechanism underlying the “fever effect”. Collectively, these findings expand the physiological understanding of IL-17A in the brain and highlight immune-based modulation of cerebellar circuits as a promising avenue for ASD therapy.
Data availability
The data underlying this article will be shared on reasonable request to the corresponding authors. RNA-sequencing data have been deposited in the National Center for Biotechnology Information (NCBI) Gene Expression Omnibus (GEO) database (GSE287731 and GSE287705). Source data are provided with this paper.
References
Hirota T, King BH. Autism spectrum disorder: a review. JAMA. 2023;329:157–68.
GBD 2021 Dementia Collaborators. Global, regional, and national burden of disorders affecting the nervous system, 1990-2021: a systematic analysis for the Global Burden of Disease Study 2021. Lancet Neurol. 2024;23:344–81.
Curran LK, Newschaffer CJ, Lee LC, Crawford SO, Johnston MV, Zimmerman AW. Behaviors associated with fever in children with autism spectrum disorders. Pediatrics. 2007;120:e1386–92.
Kim E, Huh JR, Choi GB. Prenatal and postnatal neuroimmune interactions in neurodevelopmental disorders. Nat Immunol. 2024;25:598–606.
Cotterill RM. Fever in autistics. Nature. 1985;313:426.
Xu ZP, Zhang XY, Chang H, Kong Y, Ni YY, Liu R, et al. Rescue of maternal immune activation-induced behavioral abnormalities in adult mouse offspring by pathogen-activated maternal Tcells. Nat Neurosci. 2021;24:818–30.
Choi GB, Yim YS, Wong H, Kim S, Kim H, Kim SV, et al. The maternal interleukin-17a pathway in mice promotes autism-like phenotypes in offspring. Science. 2016;351:933–9.
Kim S, Kim H, Yim YS, Ha S, Atarashi K, Tan TG, et al. Maternal gut bacteria promote neurodevelopmental abnormalities in mouse offspring. Nature. 2017;549:528–32.
Reed MD, Yim YS, Wimmer RD, Kim H, Ryu C, Welch GM, et al. IL-17a promotes sociability in mouse models of neurodevelopmental disorders. Nature. 2020;577:249–53.
Andruszewski D, Uhlfelder DC, Desiato G, Regen T, Schelmbauer C, Blanfeld M, et al. Embryo-restricted responses to maternal IL-17A promote neurodevelopmental disorders in mouse offspring. Mol Psychiatr. 2025;30:1585–93.
Al-Ayadhi LY, Mostafa GA. Elevated serum levels of interleukin-17A in children with autism. J Neuroinflammation. 2012;9:158.
Tsai PT, Hull C, Chu Y, Greene-Colozzi E, Sadowski AR, Leech JM, et al. Autistic-like behaviour and cerebellar dysfunction in Purkinje cell Tsc1 mutant mice. Nature. 2012;488:647–51.
Courchesne E, Yeung-Courchesne R, Press GA, Hesselink JR, Jernigan TL. Hypoplasia of cerebellar vermal lobules VI and VII in autism. N Engl J Med. 1988;318:1349–54.
Stoodley CJ, D’Mello AM, Ellegood J, Jakkamsetti V, Liu P, Nebel MB, et al. Altered cerebellar connectivity in autism and cerebellar-mediated rescue of autism-related behaviors in mice. Nat Neurosci. 2017;20:1744–51.
Kelly E, Meng F, Fujita H, Morgado F, Kazemi Y, Rice LC, et al. Regulation of autism-relevant behaviors by cerebellar-prefrontal cortical circuits. Nat Neurosci. 2020;23:1102–10.
Uquillas FD, Sefik E, Li B, Trotter MA, Steele KA, Seidlitz J, et al. Multimodal evidence for cerebellar influence on cortical development in autism: structural growth amidst functional disruption. Mol Psychiatr. 2025;30:1558–72.
Wagner MJ, Kim TH, Savall J, Schnitzer MJ, Luo LQ. Cerebellar granule cells encode the expectation of reward. Nature. 2017;544:96–100.
Gao ZY, Davis C, Thomas AM, Economo MN, Abrego AM, Svoboda K, et al. A cortico-cerebellar loop for motor planning. Nature. 2018;563:113–6.
Carta F, Chen CH, Schott AL, Dorizan S, Khodakhah K. Cerebellar modulation of the reward circuitry and social behavior. Science. 2019;363:eaav0581.
Zhang XY, Wu WX, Shen LP, Ji MJ, Zhao PF, Yu L, et al. A role for the cerebellum in motor-triggered alleviation of anxiety. Neuron. 2024;112:1165–81.
Cai XY, Wang XT, Guo JW, Xu FX, Ma KY, Wang ZX, et al. Aberrant outputs of cerebellar nuclei and targeted rescue of social deficits in an autism mouse model. Protein Cell. 2024;15:872–88.
Jiang SF, Wu HT. The Cerebellum modulates mood with movement. Neurosci Bull. 2024;40:1396–8.
Ramaswami G, Geschwind DH. Genetics of autism spectrum disorder. Handb Clin Neurol. 2018;147:321–9.
Hilal ML, Rosina E, Pedini G, Restivo L, Bagni C. Dysregulation of the mTOR-FMRP pathway and synaptic plasticity in an environmental model of ASD. Mol Psychiatr. 2025;30:1937–51.
Nakayama H, Abe M, Morimoto C, Iida T, Okabe S, Sakimura K, et al. Microglia permit climbing fiber elimination by promoting GABAergic inhibition in the developing cerebellum. Nat Commun. 2018;9:2830.
Shi Y, Zhang YX, Chen JY, Ma S, Xue WX, Li W, et al. Targeting histamine H4 receptor in the rostral ventromedial medulla to relieve hypertension. Adv Sci. 2025;12:e08176.
Zhang BB, Ling XY, Shen QY, Zhang YX, Li QX, Xie ST, et al. Suppression of excitatory synaptic transmission in the centrolateral amygdala via presynaptic histamine H3 heteroreceptors. J Physiol. 2025;603:6015–33.
Paxinos G, Franklin KBJ, Franklin KBJ. The mouse brain in stereotaxic coordinates. Third Edition. New York: Academic Press; 2007.
Xie ST, Fan WC, Zhao XS, Ma XY, Li ZL, Zhao YR, et al. Proinflammatory activation of microglia in the cerebellum hyperexcites Purkinje cells to trigger ataxia. Pharmacol Res. 2023;191:106773.
Tsai PT, Rudolph S, Guo C, Ellegood J, Gibson JM, Schaeffer SM, et al. Sensitive periods for cerebellar-mediated autistic-like behaviors. Cell Rep. 2018;25:357–67.
Shen LP, Li W, Pei LZ, Yin J, Xie ST, Li HZ, et al. Oxytocin receptor in cerebellar purkinje cells does not engage in autism-related behaviors. Cerebellum. 2023;22:888–904.
Gibson JM, Vazquez AH, Yamashiro K, Jakkamsetti V, Ren CY, Lei K, et al. Cerebellar contribution to autism-relevant behaviors in fragile X syndrome models. Cell Rep. 2023;42:113533.
Peter S, Ten Brinke MM, Stedehouder J, Reinelt CM, Wu B, Zhou HB, et al. Dysfunctional cerebellar Purkinje cells contribute to autism-like behaviour in Shank2-deficient mice. Nat Commun. 2016;7:12627.
Pishesha N, Harmand TJ, Ploegh HL. A guide to antigen processing and presentation. Nat Rev Immunol. 2022;22:751–64.
Bourgeron T. From the genetic architecture to synaptic plasticity in autism spectrum disorder. Nat Rev Neurosci. 2015;16:551–63.
Tsai NP, Wilkerson JR, Guo W, Maksimova MA, DeMartino GN, Cowan CW, et al. Multiple autism-linked genes mediate synapse elimination via proteasomal degradation of a synaptic scaffold PSD-95. Cell. 2012;151:1581–94.
Xu LM, Li JR, Huang Y, Zhao M, Tang X, Wei L. AutismKB: an evidence-based knowledgebase of autism genetics. Nucleic Acids Res. 2012;40:D1016–22.
Kong SW, Sahin M, Collins CD, Wertz MH, Campbell MG, Leech JD, et al. Divergent dysregulation of gene expression in murine models of fragile X syndrome and tuberous sclerosis. Mol Autism. 2014;5:16.
Mills KHG. IL-17 and IL-17-producing cells in protection versus pathology. Nat Rev Immunol. 2023;23:38–54.
Lee Y, Ishikawa T, Lee H, Lee B, Ryu C, Davila Mejia I, et al. Brain-wide mapping of immune receptors uncovers a neuromodulatory role of IL-17E and the receptor IL-17RB. Cell. 2025;188:2203–17.
Ribeiro M, Brigas HC, Temido-Ferreira M, Pousinha PA, Regen T, Santa C, et al. Meningeal gammadelta T cell-derived IL-17 controls synaptic plasticity and short-term memory. Sci Immunol. 2019;4:eaay5199.
Gomez-Castro F, Zappettini S, Pressey JC, Silva CG, Russeau M, Gervasi N, et al. Convergence of adenosine and GABA signaling for synapse stabilization during development. Science. 2021;374:eabk2055.
Vithlani M, Terunuma M, Moss SJ. The dynamic modulation of GABA(A) receptor trafficking and its role in regulating the plasticity of inhibitory synapses. Physiol Rev. 2011;91:1009–22.
Zhuang X, Zhan B, Jia Y, Li C, Wu N, Zhao M, et al. IL-33 in the basolateral amygdala integrates neuroinflammation into anxiogenic circuits via modulating BDNF expression. Brain Behav Immun. 2022;102:98–109.
Avloniti M, Evangelidou M, Gomini M, Loupis T, Emmanouil M, Mitropoulou A, et al. IKKbeta deletion from CNS macrophages increases neuronal excitability and accelerates the onset of EAE, while from peripheral macrophages reduces disease severity. J Neuroinflammation. 2024;21:34.
Cunningham C, Campion S, Teeling J, Felton L, Perry VH. The sickness behaviour and CNS inflammatory mediator profile induced by systemic challenge of mice with synthetic double-stranded RNA (poly I:C). Brain Behav Immun. 2007;21:490–502.
Jeong YH, Park JS, Kim DH, Kang JL, Kim HS. Anti-inflammatory mechanism of lonchocarpine in LPS- or poly(I:C)-induced neuroinflammation. Pharmacol Res. 2017;119:431–42.
Hagemann-Jensen M, Ziegenhain C, Chen P, Ramskold D, Hendriks GJ, Larsson AJM, et al. Single-cell RNA counting at allele and isoform resolution using Smart-seq3. Nat Biotechnol. 2020;38:708–14.
Gadban A, Gunapala KM, Taylor V, Benvenisty N. Therapeutic strategies for fragile X syndrome and implications for other gene-silencing disorders. Nat Genet. 2025;57:1812–22.
Haker R, Helft C, Natali Shamir E, Shahar M, Solomon H, Omer N, et al. Characterization of brain abnormalities in lactational neurodevelopmental Poly I:C rat model of schizophrenia and depression using machine-learning and quantitative MRI. J Magn Reson Imaging. 2025;61:2281–91.
Huangfu L, Li R, Huang Y, Wang S. The IL-17 family in diseases: from bench to bedside. Signal Transduct Target Ther. 2023;8:402.
Salvador AF, de Lima KA, Kipnis J. Neuromodulation by the immune system: a focus on cytokines. Nat Rev Immunol. 2021;21:526–41.
McGeachy MJ, Cua DJ, Gaffen SL. The IL-17 family of cytokines in health and disease. Immunity. 2019;50:892–906.
Yang L, Huh JR, Choi GB. One messenger shared by two systems: How cytokines directly modulate neurons. Curr Opin Neurobiol. 2023;80:102708.
Chen CC, Itakura E, Nelson GM, Sheng M, Laurent P, Fenk LA, et al. IL-17 is a neuromodulator of sensory responses. Nature. 2017;542:43–8.
Alves de Lima K, Rustenhoven J, Da Mesquita S, Wall M, Salvador AF, Smirnov I, et al. Meningeal gammadelta T cells regulate anxiety-like behavior via IL-17a signaling in neurons. Nat Immunol. 2020;21:1421–9.
Lv XJ, Lv SS, Wang GH, Chang Y, Cai YQ, Liu HZ, et al. Glia-derived adenosine in the ventral hippocampus drives pain-related anxiodepression in a mouse model resembling trigeminal neuralgia. Brain Behav Immun. 2024;117:224–41.
Di Filippo M, Mancini A, Bellingacci L, Gaetani L, Mazzocchetti P, Zelante T, et al. Interleukin-17 affects synaptic plasticity and cognition in an experimental model of multiple sclerosis. Cell Rep. 2021;37:110094.
Mancini A, Bellingacci L, Canonichesi J, Sciaccaluga M, Megaro A, Zianni E, et al. Immune control of the basal ganglia network: Interleukin-17 as a key modulator of striatal synaptic plasticity. Neurobiol Dis. 2025;213:106994.
Busch SE, Hansel C. Climbing fiber multi-innervation of mouse Purkinje dendrites with arborization common to human. Science. 2023;381:420–7.
Arenas YM, Lopez-Gramaje A, Montoliu C, Llansola M, Felipo V. Increased levels and activation of the IL-17 receptor in microglia contribute to enhanced neuroinflammation in cerebellum of hyperammonemic rats. Biol Res. 2024;57:18.
Zhao WJ, Xu S, Zhang YA, Li DD, Zhu CY, Wang K. The application of extended reality in treating children with autism spectrum disorder. Neurosci Bull. 2024;40:1189–204.
Sathyanesan A, Zhou J, Scafidi J, Heck DH, Sillitoe RV, Gallo V. Emerging connections between cerebellar development, behaviour and complex brain disorders. Nat Rev Neurosci. 2019;20:298–313.
Wang SS, Kloth AD, Badura A. The cerebellum, sensitive periods, and autism. Neuron. 2014;83:518–32.
Kawamura Y, Nakayama H, Hashimoto K, Sakimura K, Kitamura K, Kano M. Spike timing-dependent selective strengthening of single climbing fibre inputs to Purkinje cells during cerebellar development. Nat Commun. 2013;4:2732.
Bagni C, Zukin RS. A synaptic perspective of Fragile X Syndrome and autism spectrum disorders. Neuron. 2019;101:1070–88.
Jiang CC, Lin LS, Long S, Ke XY, Fukunaga K, Lu YM, et al. Signalling pathways in autism spectrum disorder: mechanisms and therapeutic implications. Signal Transduct Target Ther. 2022;7:229.
Cutando L, Puighermanal E, Castell L, Tarot P, Belle M, Bertaso F, et al. Cerebellar dopamine D2 receptors regulate social behaviors. Nat Neurosci. 2022;25:900–11.
Ammi R, De Waele J, Willemen Y, Van Brussel I, Schrijvers DM, Lion E, et al. Poly(I:C) as cancer vaccine adjuvant: knocking on the door of medical breakthroughs. Pharmacol Ther. 2015;146:120–31.
Hu A, Sun L, Lin H, Liao Y, Yang H, Mao Y. Harnessing innate immune pathways for therapeutic advancement in cancer. Signal Transduct Target Ther. 2024;9:68.
Song K, Pun SH. Design and evaluation of synthetic delivery formulations for peptide-based cancer vaccines. Bme Front. 2024;5:0038.
Acknowledgements
This work was supported by STI2030-Major Projects-2021ZD0202805 from the Ministry of Science and Technology of China; grants 82522033, 32530049, 32030044, 32171012, 12227808, 32200948, 32561160133, 32450728, 92468302, 32400811 from the National Natural Science Foundation of China; grants BK20240168, BK20190008 from the Natural Science Foundation of Jiangsu Province, China; grant LNSN-202402 from the State Key Laboratory of Pharmaceutical Biotechnology, the Fundamental Research Funds for the Central Universities (020814380197; 020814380208); Top Talents Program 2024300475 from Nanjing University Integrated Research Platform of the Ministry of Education; F251014 from Beijing Natural Science Foundation, and CX23YZ03 from Chinese Institutes for Medical Research, Beijing.
Author information
Authors and Affiliations
Contributions
JNZ, XYZ, CZ, and STX secured the funding. JNZ, XYZ, CZ, QPZ, and STX organized the project, designed the research, and wrote the paper. JY, WL, LPS, and WLZ analyzed data and wrote the manuscript. JY, WL, LPS, WLZ, JYC, BBZ, YJC, TL, and HZL performed experiments. ZG and CZ reviewed and edited the manuscript. All authors contributed to the preparation of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Yin, J., Li, W., Shen, LP. et al. Cerebellar microglia-derived IL-17A mitigates autism-related behavioral and synaptic deficits. Mol Psychiatry (2026). https://doi.org/10.1038/s41380-026-03454-1
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41380-026-03454-1