Abstract
Vascular Cognitive Impairment and Dementia (VCID), the second most common form of dementia, is becoming increasingly prevalent worldwide. However, currently there are no FDA-approved therapies for VCID. In this review, we explore treatments with extant supporting evidence as well as treatments in development and outline the path forward for future research. Donepezil, rivastigmine, and memantine all have some evidence as cognitive enhancers in patients with Vascular Dementia (VaD) and are approved in several non-US jurisdictions. Nutraceuticals, such as Ginkgo biloba and butylphthalide, have some supportive data and may be similarly helpful. Prevention of VCID is an important objective, and there is support for the intensive control of hypertension and treatment of hyperglycemia to prevent cognitive decline. Asymptomatic cerebral small vessel disease is commonly detected in neuropsychiatric cohorts and healthy individuals, and protocols and trials are being developed to help guide management to minimize the risk of cognitive impairment. A range of novel treatment strategies, including noninvasive neurostimulation, stimulant treatment, and neurotrophic factors, are being explored. Future research into VCID treatment needs to incorporate a range of novel technologies, including blood-based biomarkers, automation of brain image segmentation and quantification using artificial intelligence, automated remotely administered neuropsychological testing, and rational drug discovery.
Similar content being viewed by others
Introduction
Vascular Dementia (VaD) comprises 15 – 20% of all dementia cases and is the second most common form of dementia after Alzheimer’s Disease [1]. While a minority of cases are “pure” VaD, vascular pathology is present in most patients with dementia in late life (i.e. older than 65). A longitudinal post-mortem study found high rates of atherosclerosis (86%), arteriolosclerosis (74%), and gross infarcts (44%) [2] in those with a pre-mortem diagnosis of dementia. The population prevalence of VaD is estimated to be 116 per 10,000 persons over the age of 50 [3], and this increases exponentially with age, rising from 44 per 10,000 in those aged 50 – 59 years to 732 per 10,000 in those over 90 years of age [3]. Vascular Cognitive Impairment and Dementia (VCID), which comprises the full spectrum of cognitive disorders, from vascular mild cognitive impairment (VaMCI) to VaD, is estimated to be three times as common as VaD itself [4].
Despite the substantial burden of disease attributable to VCID, there has been little progress in the development of novel therapeutics, and there are no existing FDA-approved treatments [5]. There are currently four Food and Drug Administration (FDA)-approved therapies for Alzheimer’s disease (AD) that act as cognitive enhancers (three cholinesterase inhibitors and memantine). More recently, two promising anti-amyloid monoclonal antibodies for AD have received FDA approval (lecanemab and donanemab) [6]. While there are a range of modifiable mid-life risk factors for VaD, by the time patients reach late life, when VCID is most prevalent, treating these risk factors may not substantially mitigate dementia risk [7]. The lack of research progress and absence of FDA-approved therapies can lead to clinician demoralization, with a feeling of hopelessness that very little can be done for this patient population [8].
This review outlines the available clinical trial evidence for treatments in VCID, explores the putative mechanisms of these treatments, and outlines a path forward to advance the field. This review will not cover non-pharmacological interventions for VCID, which have been reviewed in detail elsewhere [9]. Understanding the specific management of VCID is becoming increasingly important for physicians as dementia subtypes become better demarcated by fluid-based biomarkers [10] and the prevalence of VCID increases with the aging population.
Defining VCID
Before exploring current and future treatments for VCID, we will briefly describe some clinical definitions of the disorder, as classifications have changed considerably over the years, which may affect the interpretation of clinical trials [11,12,13]. VCID in some recent studies refers to the transdiagnostic concept of “Vascular Contributions to Cognitive Impairment and Dementia” [14]. While this dimensional understanding of dementia may be important for the future development of the science, this review examines studies which focus on cognitive impairment and dementia with etiology in cerebrovascular disease, and in the literature have been referred to under the categories of VCID, VaMCI and VaD.
Most clinical trials assessing the safety and efficacy of cognitive enhancers in VaD have used the 1993 NINDS-AIREN criteria (Supplementary Table 1) [12]. According to these criteria, a diagnosis of dementia requires memory deficits and at least two other cognitive domains. To diagnose probable VaD, a clear temporal relationship between stroke and neuroimaging evidence is required. Neuroimaging evidence included multiple large vessel strokes or a single strategically located large vessel stroke in the angular gyrus, thalamus, basal forebrain, or posterior or anterior cerebral artery territories. Importantly, the criteria state that the absence of focal neurologic signs other than cognitive disturbance makes the diagnosis of VaD unlikely or uncertain.
The VASCOG-2 WSO criteria (Table 1) recently updated the 2014 VASCOG-1 criteria [15] for VCID using the Delphi Survey Method. These differ from the NINDS-AIREN criteria in several ways. First, they covered the spectrum of VCID from VaMCI to VaD. Second, they recognize the clinical reality that memory is not impaired in all cases of VCID and that significant deficits in any one cognitive domain are sufficient to diagnose cognitive impairment. The distinction between VaMCI and VaD is based not only on the severity of cognitive deficits, but also on the impact on daily function, with the latter being associated with an inability to function independently. Third, the VASCOG-2 WSO criteria also detail the significant neuroimaging findings required for defining mild and major vascular cognitive disorders, with the explicit recognition that markers of small vessel disease in the absence of large infarcts may be sufficient. The DSM-5 criteria (Supplementary Table 2) have also been commonly used for diagnosis, and are similar to the VASCOG-1 criteria, albeit less detailed. It is also noteworthy that many individuals are identified on brain imaging as having a significant burden of small vessel disease (SVD) or cerebrovascular disease in general (CVD), but are either asymptomatic or present with non-specific symptoms, such as depression, apathy, fatigue, or other neuropsychiatric symptoms. They have been referred to as having covert cerebral SVD (ccSVD) [16] or as ‘pre-clinical’ cases, and may be candidates for the prevention of VCID.
Vascular brain lesions: Frequency and reversibility
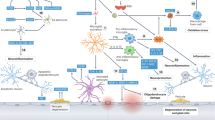
There are a panoply of underlying radiological and pathological vascular brain changes associated with VCID. To understand the current and future treatment targets of VCID, these lesions are briefly reviewed below (Figs. 1 and 2). In the 1970s and 80 s, the diagnosis of VaD was generally restricted to those who had experienced a stroke, referred to as post-stroke dementia. In addition to post-stroke dementia not being exclusively vascular in origin, we now recognize that VCID may be caused by various diseases of the large and small cerebral vessels, leading to thromboembolic disease, cerebral ischemia or hemorrhage, acute or chronic cerebral hypoperfusion, impaired angiogenesis and diminished oxygen and nutrient blood-brain barrier transport (see Table 2 for list of underlying pathologies) [17]. The developing understanding of these diverse pathophysiological mechanisms has allowed for a greater number of novel treatment approaches, that we will explore in later sections.
Schematic diagram summarizing the macroscopic lesions associated with VCID.
a Acute lacunar infarct in right thalamus on DWI. b Old cavitated lacune in posterior limb of left internal capsule on FLAIR. c Confluent white matter hyperintensities on FLAIR. d Deep CMB on gradient echo scan. e Enlarged PVS intracerebral. f Basal ganglia hemorrhage on CT. Image and description taken directly from “Markus Hugh S, de Leeuw Frank Erik. Cerebral small vessel disease: Recent advances and future directions. Int J Stroke. 2022;18:4–14 - Fig. 1” [167] under Creative Commons Attribution 4.0 License (https://creativecommons.org/licenses/by/4.0/) which permits any use, reproduction and distribution of the work without further permission.
Large vessel infarcts have the strongest association with VCID, and in the 2–6 months following a stroke, about 10% of patients without prior dementia will develop dementia, with an overall 44% experiencing VCID [18]. Infarcts due to large vessel disease are present in 11% of people in their 60 s and around 35% of those older than 80 [19]. It is now recognized that the majority of VCID cases are attributable to cerebral SVD, which can be well-delineated using MRI. Several SVD lesions have been described and the terminology for these lesions has been standardized using the STRIVE criteria [20]. The lesions are briefly described as follows.
Lacunes are caused by vascular insufficiency due to the deep penetration of blood vessels. They affect 8–10% of individuals in their 60 s and increase with age [21]. They are seen as hypodense circular or ovoid lesions, 3–15 mm in diameter on MRI and, like larger strokes, once present, they are irreversible. White Matter Hyperintensities (WMH) (aka leukoaraiosis) are seen as hyperintense lesions on T2-weighted or fluid-attenuated inversion recovery MRI and are generally divided into periventricular and deep WMH. The pathology underlying this radiological change in its early form may be a reversible increase in interstitial fluid; however, in its later form, it represents demyelination and axonal damage [22]. In addition to vascular insufficiency, WMH may be caused by demyelination, inflammation, trauma, neoplasms, and degeneration; however, there is good evidence that most of these lesions in older people are ischemic in origin [23]. They are the most common form of vascular lesions, present in approximately 90% of older adults [24, 25], 72% of those in mid-life [25], and almost 50% of those in their late 40 s [16]. Because it is commonly observed in cognitively normal individuals, its association with VCID is variable. Automated quantification of WMH volume has increasingly been used to better indicate the severity of the disease and likely prognosis of cognitive decline [26, 27]. WMH are at least partially reversible, with one study of 190 patients post minor stroke (mean age = 65.3) found that over 1 year, 37% of participants showed a decrease in WMH volume, particularly in those with effective blood pressure control [28]. In studies of other conditions, such as migraine and Reversible Cerebral Vasconstriction Syndrome (RCVS), WMH may be entirely transient and reversible [29]. Cerebral microbleeds (CMBs) are a very common form of vascular pathology, present in 15–40% of older persons [30, 31]. CMBs within the cerebral lobes are associated with underlying cerebral amyloid angiopathy (CAA), whereas CMBs within deeper cerebral structures are much more closely associated with hypertension. CMBs have a weak or inconsistent relationship with cognitive decline, with one systematic review finding no such association [32]. However, several studies have found that the risk of cognitive decline increases only once more than 3–5 CMBs are present [33, 34]. Although CMBs, like other hemorrhages, may diminish in size over time, there is little available evidence that they are entirely reversible. Enlarged perivascular spaces (ePVS) are extremely common in the general population, with some estimates that they are present in nearly 100% of older persons [35]. ePVS increases with age, hypertension, and TBI, among other risk factors [36, 37]. Studies have variably found present or absent associations between cognitive decline and ePVS [38]. Studies on several different diseases indicate that ePVS may be partially reversible [36].
There is evidence from animal models that most pathologies underlying these lesions, particularly atherosclerosis and arteriolosclerosis, are partially reversible, particularly in their early forms [39, 40]. This approach is to find effective treatments to improve cerebral blood flow, thereby improving cognitive function and/or mitigating further decline. Moreover, the risk factors for CVD have been extensively examined, and many are modifiable, thereby permitting the potential prevention of CVD and VCID.
Medications and their mechanisms for VCID management
While the USA-based Federal Drugs Administration (FDA) has not approved any medications for cognitive symptoms of VCID in other nations, regulatory approval for use in VaD has been granted for donepezil (e.g., New Zealand, India, South Korea, Romania, and Thailand) and memantine (e.g., Argentina, Brazil, and Mexico) [41]. Chinese jurisdictions advocate the use of herbal extracts, including butylphthalide and Ginkgo biloba, and the evidence for their use is discussed below.
A challenge in interpreting clinical trials for VaD is that the outcomes used are often specifically designed for Alzheimer’s disease (e.g., using the Alzheimer’s Disease Assessment Scale-Cognitive subscale – ADAS-Cog) and may not accurately reflect beneficial effects in cognitive domains more frequently affected by VaD, such as processing speed and executive function [42]. Additionally, clinical trials up to this point have not used fluid biomarkers and have not been able to reliably disambiguate VCID from AD [43], only being able to diagnose “probable” or “possible” VaD. Other problems, include short trial duration, population heterogeneity, small sample size and the high frequency of mixed dementia cases. With these challenges in mind, the evidence for the treatment of VCID is reviewed below.
Cognitive enhancers
Acetylcholine concentration and acetyltransferase activity are reduced in patients with both AD and VCID [44]. Cholinesterase inhibitors aim to correct this deficit by increasing the levels of the neurotransmitter acetylcholine in synaptic clefts by reducing its enzymatic degradation [45]. However, VCID presents in heterogeneous subtypes, and trials suggest that they may be less efficacious in some forms (e.g., multi-infarct dementia) than others (e.g., subcortical vascular dementia) [46].
Donepezil
Donepezil is a non-competitive reversible cholinesterase antagonist. It shows limited binding to butyryl-cholinesterase and is highly selective for acetylcholinesterase [47]. Donepezil has a half-life between 60 and 90 h and is administered once per day at doses of either 5 or 10 mg. In a 2021 Cochrane review and meta-analysis [48], the authors assessed three RCTs of donepezil hydrochloride 5 mg or 10 mg in VaD. The meta-analysis found that donepezil 5 mg and 10 mg both significantly reduced the rate of cognitive decline, as measured by the ADAS-Cog (range of 0 – 70) (5 mg: Mean Difference (MD) = -0.92, 95%CI[-1.44, −0.40], and 10 mg; MD = −2.21, 95%CI[ − 3.07, −1.35]). The minimal change in ADAS-Cog deemed to be clinically relevant is between 2 and 3 [48], and the authors suggest that the 5 mg dose is unlikely to have a clinically significant effect, despite its statistical significance. The most commonly reported adverse effects were headache, high blood pressure, nausea/vomiting, diarrhea, and dizziness. Compared to placebo, the 10 mg dose caused significantly more adverse effects, whereas the 5 mg dose did not (5 mg: OR = 1.22, 95%CI[0.94, 1.58] AND 10 mg: OR = 1.95, 95%C [1.20, 3.15]). These findings are supported by other meta-analyses [49], including one 2022 network meta-analysis [50], which found that donepezil 10 mg was the most effective intervention and only one that improved executive function. Indeed, a randomized controlled trial of donepezil 10 mg in participants with Cerebral Autosomal Dominant Arteriopathy with Subcortical Infarcts and Leukoencephalopathy (CADASIL) (n = 168 patients, mean age = 54.8 years) [51] found cognitive function improvements only in tests measuring executive or attention function but not general cognitive function.
Galantamine
Galantamine is a competitive, reversible cholinesterase antagonist that binds almost exclusively to acetylcholinesterase with little butyryl-cholinesterase activity [52]. It has a half-life of approximately 7 h and is administered at doses between 4 mg twice daily and 12 mg twice daily (between 8 and 24 mg daily). Two trials of galantamine in VaD [53, 54] (n = 1380) have been included in several systematic reviews and meta-analyses [48, 55]. Battle et al. [48, 55] found a statistically significant reduction in the ADAS-Cog score (MD = -2.01, 95%CI[ − 3.18, −0.85]) and improvement in Clinical Global Impression-Severity (OR = 1.32, 95%CI[1.03, 1.70]) compared to placebo. Shi et al. [55] found that galantamine had the largest positive benefit on the ADAS-Cog score (MD = -1.99, 95%CI[-3.03, -0.95]) of the compared medications but also had the second highest risk of adverse events (OR = 1.68, 95%CI[1.18, 2.37]). As mentioned above, although galantamine seems to have a significant effect, it rests on the borderline of what is considered clinically relevant.
Rivastigmine
Rivastigmine acts on both acetylcholinesterase and butyryl-cholinesterase as a “pseudo-irreversible” antagonist. Its antagonistic duration of action is approximately 7 h; however, it has a plasma half-life of approximately 1.5 h. It can be delivered as a patch or as a twice-daily oral dose of 3–12 mg/day. There have been three reported clinical trials (n = 800) of rivastigmine in VCID, including two for VaD [56, 57] and one for VaMCI [58]. Individually, the trials showed no significant effect on ADAS-Cog scores or functional outcomes, and a meta-analysis showed no positive effect [48]. Shi et al. [55] reported a small, clinically insignificant positive effect of rivastigmine (MD = 0.60, 95%CI[0.10, 1.10]).
Memantine
Memantine is a non-competitive antagonist of extra-synaptic N-methyl-D-aspartate (NMDA) receptors that disrupts regular synaptic transmission and limits excitotoxic neuronal damage. Its half-life is approximately 60 h, and it is administered once daily at doses between 5 mg and 20 mg (immediate release). Two trials comparing memantine (20 mg) to placebo (n = 836) [59, 60] found that memantine significantly reduced cognitive decline, with one of the largest effect sizes in one network meta-analysis (MD = -1.89, 95%CI[-2.93, -0.86]). Compared to placebo, there were no significantly greater rates of adverse events (OR = 1.25, 95%CI[0.82, 1.92]), and it was found to be more acceptable than cholinesterase inhibitors [55].
Butylphthalide
Butylphthalide is one of the key chemical constituents of celery oil and contributes to its distinct flavor. It is commonly used in Chinese medicine and has been attributed to several pharmacological effects, including reduced inflammation, improved mitochondrial function, diminished platelet aggregation, reduced oxidative stress, and prevention of neuronal apoptosis [61, 62]. In 2002, it was approved in China for the treatment of cerebral ischemia [60], and since then, there have been four clinical trials (n total = 665) [61, 63,64,65] testing butylphthalide 200 mg three times daily against placebo, donepezil, or as a combination therapy with another herbal treatment. Butylphthalide was generally well tolerated, and trials have had significant positive outcomes, resulting in significant improvements in ADAS-Cog, Clinician Interview Based Impression of Change (CIBIC), and MMSE scores. However, in the most robust trial [61], the change in ADAS-Cog was small (MD = -1.07 points; 95%CI [-0.12, -2.02], p = 0.03) and may not be clinically relevant. A range of mechanisms, including anti-oxidant and anti-inflammatory effects, have been proposed; however, it is not clear which mechanism(s) induce the therapeutic effect. Clinicians may consider its use in current populations, but further delineation of its mechanisms and confirmation of its effects in non-Chinese populations are required.
Ginkgo biloba
Ginkgo biloba, a herbal supplement taken from the ginkgo tree, is commonly used in Chinese medicine and is thought to have anti-oxidant, anti-inflammatory, and anti-thrombotic effects. It is generally administered in tablet form once daily at doses between 60 mg and 240 mg. A 2010 meta-analysis [66], including nine trials (n = 2372) that incorporated patients with all-cause dementia, VaD, and AD, found that Ginkgo had a significant positive effect on cognition (SMD = -0.58, 95%CI[-1.14, -0.01], p = 0.04) but not on Activities of Daily Living (ADLs), although the heterogeneity was high (I2 = 97%). Like Butylphthalide, Ginkgo was well tolerated, with no significant difference in rates of adverse effects compared to placebo. Since then, there has been one further, non-Chinese, 6 month clinical trial [67] (n = 90, mean age (SD) = 67.1 ± 8.0 years) that found ginkgo produced a significantly greater improvement in CGI-S, but none of the other three main cognitive and functional outcome measures. In summary, there is weak evidence that Ginkgo biloba, a well-tolerated herbal extract, would be helpful in patients with VCID, although more trials with larger, more diverse populations are needed.
Cerebrolysin
Cerebrolysin is a patented combination of pig-derived, enzymatically treated peptides including brain-derived neurotrophic factor (BDNF), glial cell line-derived neurotrophic factor (GDNF), nerve growth factor (NGF), and ciliary neurotrophic factor (CNTF). It is administered as an intravenous infusion, normally once daily, for 28–30 days. It has regulatory approval for use in post-stroke patients in Russia, China, South Korea, Austria and Germany [68].
To date, two randomized trials of cerebrolysin for VaD have been conducted. Xiao et al. [69] (n = 148, age range = 55–85) compared 4 weeks of cerebrolysin to placebo infusion, assessing outcomes at 2 and 4 weeks. At 4 weeks, those in the cerebrolysin group showed a significantly larger improvement in MMSE (Change from baseline = 2.68 vs. 1.72, p = 0.028), although there were no differences in functional outcomes (ADLs) or CGI-S. Guekht et al. [70] (n = 242, age range = 50 to 85) compared 2 four-week treatment cycles of cerebrolysin with placebo infusion and assessed outcomes at 24 weeks after treatment initiation. They found that in the cerebrolysin group, there were significantly greater improvements in ADAS-Cog (-10.6 points vs -4.4 in placebo, MD = 6.2, p < 0.001) and a clinician-rated measure of change (75.3% showed improvement vs. 37.4% in placebo group). Interestingly, in this trial, there was no significant difference between the groups for change in MMSE, but for functional outcomes, the cerebrolysin group showed significant improvement (MD = 6.33 points; 95%CI[4.19, 8.46], p < 0.001). While promising, this trial should be interpreted with caution given the inconsistency in the large, significant ADAS-cog result and the small, non-significant MMSE result as well as the fact that the placebo group had such a large improvement in cognitive and functional outcomes. Taken together, cerebrolysin has some preliminary evidence supporting its use in VCID, but it remains a burdensome treatment that requires a strong evidence base prior to its use.
Other medications
Nimodipine, a calcium channel blocker with central vasoactive properties, was studied in combination with galantamine in mixed dementia (Alzheimer’s disease with cerebrovascular disease) [69]; however, the combination was not found to be better than galantamine alone. Similarly, a trial of nimodipine alone (n = 251) found that compared to placebo, it had no significant benefit for cognitive or social outcomes.
Isolated or small clinical trials have been conducted for other medications, including pentoxyifyline, piracetam, aspirin, choline alfoscerate, propentofylline, naftirdofuryl, idebenone, buflomedil, Yangxue Qingnao, and several other Chinese Herbal preparations [71]. Further studies are needed before these can be considered as treatments for VCID.
In summary, there are few cognitive benefits in VCID with the use of donepezil, galantamine, and memantine, generally at higher doses of these medications. Butylphthalide and Ginkgo Biloba are well-tolerated herbal supplements that have some clinical trial evidence indicating small cognitive benefits, but data remain limited, especially in non-Chinese populations. Nimodipine, cerebrolysin, and several other medications have been tested in various trials; however, larger studies and more consistent results are needed before any recommendations can be made.
Symptomatic treatment of neuropsychiatric symptoms of VCID
The vast majority of those with VCID experience neuropsychiatric symptoms [72], with common presentations including depression, apathy, irritability, appetite change, delusions, and hallucinations. Non-pharmacological strategies are the first line of management [73]. These strategies include behavioral management, care staff communication training, sensory interventions (e.g., light therapy, sensory garden, and aromatherapy), music therapy, dance therapy, reminiscence therapy, and exercise therapy. These interventions have been reviewed in detail elsewhere [9, 74].
Treating depression in VCID
Depression and anxiety are common in VCID, affecting 53% and 36% of those with VaD and 46% and 31% of those with VaMCI, respectively. The relationship may be bidirectional, with depression both predicting and being predicted by VCID. A 2013 meta-analysis [75] of prospective studies found that depression was associated with significantly greater risk of VaD (HR = 2.52, 95%CI [1.77, 3.59], P < 0.001, n = 5 studies) than AD (HR = 1.65, 95%CI [1.42, 1.92], P < 0.001, n = 15 studies).
As mentioned above, there is evidence of several non-pharmacological strategies [76, 77] in people with depression and dementia, but the outcomes in this population are generally poor [78]. Depression in dementia increases cognitive decline, mortality risk, carer stress and the rate of admission to residential aged care facilities [79,80,81].
Despite the high prevalence of depression in VCID, very few clinical trials have been conducted to guide its management. Dudas et al. [82], in a systematic review of antidepressants in dementia, found that, overall there were ten studies (n = 1592), but the vast majority examined AD exclusively, with only one small study (n = 31) including a mixture of vascular dementia and AD [83]. While there is no evidence to specifically guide the use of antidepressants in VaD, this systematic review provided insights into their application in AD and all-cause dementia. Antidepressants had no significant effect on depression rating scores between 6 and 13 weeks or between 6 and 9 months after treatment. They also had no impact on functional outcomes (activities of daily living) or cognitive scores (MMSE) 13 weeks after treatment. Those taking antidepressants were 51% more likely to drop out of the trials (OR = 1.51, 95%CI[1.07, 2.14]) and 55% more likely to experience an adverse event (OR = 1.55, 95%CI[1.21, 1.98]), which could include dizziness, falls, dry mouth, or confusion. While evidence is lacking for use in VaD, there is frequent overlap of vascular and Alzheimer’s pathologies in dementia, and the extant research shows that higher burdens of vascular risk factors and worse cognitive function predict reduced response to antidepressants [84]. These data indicate that antidepressants are unlikely to be effective in patients with VaD or depression.
However, in patients with vascular disease and only MCI, there is reasonable evidence that anti-depressants continue to have efficacy. Sheline et al. [84] (n = 217, mean age (SD) = 68.4 (7.2), age range = 60 + ), in a randomized trial of sertraline for late-life depression, found that while lower cognitive scores predicted poorer responses to antidepressants, 62.3% of those who remitted had a Fazekas score of greater than 2 (i.e., substantial white matter change on MRI), and this was not significantly different (p = 0.18) from 72.4% of non-remitters who had elevated Fazekas scores. Taragano et al. [85] (n = 101, mean age (SD) = 69.9 (7.8)) found in their clinical trial examining patients with “vascular depression” that 40.5% of patients remitted with fluoxetine, although there was a substantially greater proportion of patients who remitted with fluoxetine-nimodipine treatment compared to fluoxetine alone (54% vs. 27%, p = 0.006). There is strong evidence that antidepressants improve physical and psychological outcomes in patients with depression following stroke. A 2020 network meta-analysis incorporating 51 randomized trials [86] identified that citalopram, escitalopram, sertraline, mirtazapine, venlafaxine, and paroxetine were significantly more effective than placebo, whereas amitriptyline was less effective and more poorly tolerated.
Based on the extant literature, there is good evidence that antidepressants are effective and therapeutic in post-stroke patients and have a role in the treatment of depression in those with vascular brain disease, although cSVD may be a relative mediator of treatment resistance. In those with the most severe forms of VCID (i.e. dementia), there is limited evidence suggesting that antidepressants are not likely to be effective.
Treating psychotic symptoms and agitation in VCID
Individuals with VCID frequently present with agitation (33%), delusions (20%), and hallucinations (11%) [87]. These can be some of the most challenging symptoms to manage, particularly when caregivers and loved ones experience aggression. While non-pharmacological interventions are the first line of treatment, pharmacological strategies can be used in refractory cases where there are substantial risks to the patient and others.
In a Cochrane systematic review and meta-analysis, Mühlbauer et al. [88] identified 24 clinical trials that examined the effects of antipsychotics on psychosis and agitation in patients with AD and VaD. Of these, nine included mixed samples of AD, VaD, and mixed dementias, whereas the other 15 focused exclusively on AD. Within trials that included a mix of dementia subtypes, the proportion of the sample with vascular or mixed dementia varied from 14.8% [89] to 27% [90]. Because of the small number of participants with VaD in each trial, subgroup analyses were generally not performed.
Considering the 9 trials that assessed both AD and VaD populations, there were significant positive results in treating agitation and psychosis for quetiapine (200 mg daily) [89, 91], tiapride (100–300 mg daily), haloperidol (2–6 mg daily) [92] and risperidone (0.5–2 mg daily) [90, 93]. One trial [94] comparing both olanzapine and risperidone to placebo found that both active drugs produced no better outcomes over 10 weeks, as measured by the Neuropsychiatric Inventory and Clinical Global Impression-Severity of Psychosis scale. Overall, the results are encouraging, indicating that drug use, especially of quetiapine and risperidone, may be efficacious, although all nine studies had at least some commercial funding and the results have not been confirmed in independent studies.
The positive results of these studies are reflective of the wider meta-analysis performed by Mühlbauer et al. [88]. They found that in patients with dementia (AD and/or VaD), atypical antipsychotics, including risperidone, olanzapine, aripiprazole, and quetiapine, likely reduced agitation (SMD = -0.21, 95% CI [-0.30, -0.12]), but had a minimal effect on psychosis (SMD = -0.11, 95%CI [-0.18, -0.03]). They were associated with an increased risk of side effects, including somnolence (RR = 1.93, 95% CI [1.57, 2.39]), extrapyramidal symptoms (RR = 1.39, 95%CI[1.14, 1.68]), serious adverse events (RR = 1.32, 95%CI [1.09, 1.61]), and death (RR = 1.36, 95%CI[0.90, 2.05]). Many medical regulatory bodies, including the FDA, require black box warnings noting the increased risk of mortality in patients with dementia. Risperidone was the best studied drug, with seven trials, and its safety and efficacy was similar to those of atypical antipsychotics considered as a whole. First-generation antipsychotics, including haloperidol and thiothixene, also appeared to reduce agitation (SMD = -0.36, 95%CI [-0.57, -0.15]) and psychosis (SMD = -0.29, 95%CI[-0.55, -0.03]), although the study numbers were smaller and the certainty of the estimate was comparably lower.
Taken together, antipsychotics seem to have some efficacy reducing agitation in all-cause dementia; however, there is no clear evidence for their specific effects on VCID. The application of antipsychotics in VCID should be approached with extreme caution given the adverse effect profile, including extrapyramidal side effects, increased risk of falls, stroke, pneumonia, thrombosis, and all-cause mortality [73].
Covert Cerebral Small Vessel Disease (ccSVD) and VCID prevention
The prevalence of dementia worldwide is predicted to steadily increase in the coming decades as the world’s population ages [95]. However, the bright line in this often pessimistically cited fact is that the age-specific risk of all-cause dementia in developed countries has decreased by 20% every 10 years since the late 20th century [96,97,98], and this has been mirrored in the age-specific decrease in cerebral small vessel disease (cSVD) [99]. Much of this relates to the better detection and treatment of vascular risk factors [96], strongly indicating that populations have previously reduced dementia risk by prevention, suggesting that such reductions may continue into the future. The management of vascular risk factors for the prevention of stroke and dementia has been reviewed in detail elsewhere [100]. The following section explores the available evidence related to the prevention of covert cerebral small vessel disease (ccSVD) and VCID.
ccSVD is a common incidental finding on imaging and is present in 90% of older persons [23, 24], and nearly half of those in their late 40 s [16]. The most common form of ccSVD is WMH, but it may include any of the aforementioned lesions of cSVD (e.g., lacunes, cerebral microbleeds, ePVS). Very few trials have examined how ccSVD should be managed, and prior clinical practice has often dismissed these changes as merely age-related. However, there is increasing appreciation that ccSVD increases the risk of VCID, all-cause dementia, stroke, intracerebral hemorrhage, and mortality. It is associated with vascular risk factors including age, hypertension, high cholesterol, smoking, and diabetes. The 2024 Lancet Commission on the Prevention of Dementia estimated that 45% of the burden of dementia is potentially modifiable, largely through treatable vascular risk factors [7, 101]. The majority of this preventable burden relates to AD, which shares many vascular risk factors with VCID but is substantially more common. This estimate provides a useful estimate for ccSVD but should be considered in balance with the fact that ccSVD is highly heritable (e.g., the heritability of WMH was reported to be as high as 0.76) [102] and occurs in many cases with no history of vascular risk factors [103].
A number of guidelines have been published in the past few years for the management of ccSVD, including two from the European Stroke Organisation [104, 105] and one from an Australian group [106]. Both made similar recommendations, with a particular focus on controlling hypertension, treating diabetes, and hyperlipidemia, while recognizing that the quantity and quality of evidence to support these recommendations is very weak. We examine the evidence for the prevention of ccSVD and VCID in the following section.
Antihypertensives
An individual participant data (IPD) meta-analysis of longitudinal studies (n = 19,209) performed by our group found that untreated hypertension in late-life was associated with a 71% higher risk of VaD compared to those with treated hypertension (mean age, 72 years; HR, 1.71; 95%CI: 1.03–2.84) [107, 108]. Mid-life hypertension is a well-established, highly prevalent and often undertreated risk factor for both VaD and other forms of dementia [109,110,111].
Peters et al. [112], in an IPD meta-analysis of five randomized trials of antihypertensive drugs (n = 28,008), found that hypertension treatment significantly reduced the risk of incident dementia (OR = 0.87, 95%CI[0.75, 0.99]) and that dementia risk continued to decline with lower blood pressure down to an SBP of approximately 100 mmHg. The PRESERVE trial [113] randomized 111 patients with MRI-confirmed lacunar infarcts and confluent WMH to either standard (SBP = 130–140 mmHg) or intensive (systolic<125 mmHg) blood pressure management. They found that over two years, there were no significant differences between the groups in diffusion tensor imaging measures of white matter structural integrity, WMH volume, or cognitive performance. There is good evidence that treating hypertension helps prevent VCID, although it is not clear if intensive blood pressure targets are associated with improved clinical outcomes in the setting of ccSVD.
Lipid lowering therapy
There is little to no direct clinical trial evidence that the use of statins or other antilipid therapies reduces the risk of cognitive impairment. There have been no randomized trials of statins examining VaD as a primary outcome, although in the few that have included all-cause dementia as a secondary outcome, no association has been found [114,115,116,117]. In two trials examining the effect of antihypertensive and lipid-lowering agents, a lower volume of WMH in the rosuvastatin arm was observed over five years compared to placebo [118, 119], although no differences between the treatment and control groups were found in the PROSPER study [120]. Although randomized clinical trials have not provided strong evidence for the use of lipid-lowering agents for VCID prevention, a meta-analysis of 31 observational studies found that statin use substantially reduced the risk of non-AD dementia (RR = 0.81, 95%CI[0.73, 0.89], p < 0.01), with greater reductions in dementia risk at higher doses of statins [121].
Antihyperglycemics
Type 2 diabetes mellitus is a common risk factor for VCID, which increases the relative risk of VaD by 137% [122]. Although there is a clear positive relationship between VaD risk and HbA1c [123, 124], the specific effects of different classes of antihyperglycemics remain unclear. Metformin, in a 2018 meta-analysis, was associated with a reduced risk of cognitive impairment (OR = 0.55, 95%CI[0.38, 0.78]) [121], but in another study, there was no association [125]; in fact, insulin therapy was associated with an increased risk of dementia. In a meta-analysis of randomized trials, one study found that dementia was substantially lower in those randomized to GLP-1 receptor agonists (HR = 0.47, 95%CI[0.25–0.86]) compared to placebo, and in a concurrently run national registry study, there was also evidence that GLP-1 RA use reduced dementia risk (HR = 0.89, 95% CI[0.86–0.93]) [126]. In summary, there is good evidence that glycemic control is protective against VCID, and that some treatments may have greater cognitive protective effects than others.
Antiplatelet agents and anticoagulation
Antiplatelet therapy and anticoagulation are known to be important interventions for reducing stroke risk, especially in the context of atrial fibrillation. However, in large meta-analyses of 11 clinical trials (n = 109,860 participants; mean age (SD) = 66.2 (7.9); mean follow up = 5.8 years) antiplatelet therapy was found to have no effect on incident cognitive decline [127]. In a meta-analysis of 5 longitudinal studies and one randomized controlled trial of patients with atrial fibrillation anticoagulants were found to significantly reduce dementia risk (RR = 0.79; 95%CI[0.67, 0.93]; p = 0.005), a finding that has been supported by a more recent network meta-analysis [128].
Carotid artery stenosis endarterectomy and stenting
Carotid stenosis secondary to atherosclerotic disease is a known cause of cerebral hypoperfusion and VCID. Most commonly carotid artery disease is managed conservatively with medications and lifestyle interventions however in severe cases direct intervention in the form of carotid artery endarterectomy (CEA) or stenting (CAS) is used. In a systematic review of 13 longitudinal pre- post-procedure studies of carotid endarterectomy, 7 studies found that there was improvement in cognitive function after the intervention while 6 studies showed no change or decline [129]. Importantly, the only randomized clinical trial included in that review (n = 58) found that there were no cognitive benefits from CAE [130]. Studies comparing CEA and CAS found that both significantly improved cognitive function with no difference in their effects [131].
Multidomain interventions
Trials of multidomain interventions targeting vascular and cognitive reserve-related risk factors have yielded highly variable results. The FINGER trial (n = 1260) was a multidomain intervention aimed at improving diet, exercise, vascular risk monitoring, and cognitive training [132], and found a small but significant effect in the intervention arm. However, comparable multidomain interventions showed no significant effects on the main outcomes in Dutch pre-DIVA [133] and French MAPT trials [134]. More recently, an Australian trial (n = 6104, age range = 55–77) of an online multidomain intervention in a non-demented population found that at three years, those in the intervention group had a significantly greater improvement in global cognitive composite scores compared to controls [135]. Specific data on vascular risk reduction and VCID are not unavailable. There is strong evidence from large meta-analyses of over 60 RCTs that, in those older than 65, exercise interventions improve global cognition (SMD = 0.42; 95%CI[0.37–0.48]), memory (SMD = 0.27; 95%CI[0.20–0.35]) and executive function (0.23 (0.20–0.26)) [136].
Current clinical trials and future VCID treatments
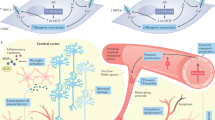
Investigations into potential therapeutics for VCID, compared to other diseases, have been limited; however, there are several drugs and therapies currently under investigation. These treatments have a range of modalities and mechanisms, including neurotransmitter systems, cellular metabolism, cerebral blood flow, and non-invasive neurostimulation (see Fig. 3 and Supplementary Tables 3 and 4).
The top half of the diagram outlines treatments with current evidence and the bottom half outlines treatments still under development. The medications and medication classes listed are not exhaustive and are intended to give a sense of the breadth of approaches being taken. AChE Acetylcholinesterase, Ach Acetylcholine, ACEI Angiotensin Converting Enzyme Inhibitor, ARB Angiotensin Receptor Blocker, BB Beta-Blockers, Ca2+ Calcium, CCB Calcium Channel Blockers, DAT dopamine reuptake transport, D receptor Dopamine receptor, GLP-RA1 glucagon-like peptide-1 receptor agonists, ISMN Isosorbide mononitrate, Na+ Sodium, NA RT Noradrenaline reuptake transporter, TMS Transcranial Magnetic Stimulation, tPBM Transcranial photobiomodulation.
Neurotransmitter systems
Methylphenidate is a noradrenaline and dopamine reuptake inhibitor that is primarily used as a stimulant treatment for attention deficit disorders. A single-dose pilot trial (n = 30, mean age (SD) = 67 (8)) in patients with VCID showed improved executive function (MD = 1.40%; 95%CI [0.56, 2.25]) and verbal memory (MD = 0.59 words; 95%CI [0.03, 1.15]) compared with placebo [137]. Oxiracetam is a nootropic and mild stimulant derived from the neurotransmitter GABA [138]. It has been tested in combination with the α1-adrenergic receptor antagonist nicergoline, a vasodilator used in vascular migraine and Raynaud disease. Participants (n = 120, age range = 40–80 years) with cognitive decline within three months of ischemic or hemorrhagic stroke received either nicergoline or nicergoline with oxiracetam. After 4 weeks, the combination group had significantly better outcomes [139]. This was a small, limited, and short-term study; however, oxiracetam was also tested in a 36-week trial of 500 post-stroke patients (age > 50 years) with subjective cognitive decline. The final results of this trial are yet to be published (KCT0005137) [140].
Cellular metabolism
Metformin, a commonly prescribed antihyperglycemic agent, inhibits hepatic gluconeogenesis and opposes the action of glucagon. Several trials have examined cognition as a secondary outcome in the treatment of diabetes mellitus, and several new trials are now assessing its efficacy in normoglycemic patients with cognitive impairment. The MetMemory trial (NCT-00592735) will examine the effect of metformin in insulin-resistant normoglycemic participants with MCI over three years. MET-FINGER (NCT-05109169) and Metformin in Alzheimer’s Prevention (NCT-04098666) are multimodal trials combining metformin with lifestyle intervention [141] in participants with AD.
Actovegin was developed from calf blood that has been ultra-filtered, purified, and deproteinized, and comprises electrolytes, amino acids, oligopeptides, nucleosides, and glycosphingolipids. It has been found to improve plasma membrane transport of glucose and tissue oxygenation and has been used for performance enhancement in sports, most famously by the competitive cyclist Lance Armstrong [142]. In the ARTEMIDA trial [143], post-stroke patients (n = 503, mean age (SD) = 69.9 (6.9), age range > 60 years) were administered Actovegin (initially an IV infusion and then a daily oral dose) or a placebo. At six months the treatment arm showed significantly better improvement in ADAS-cog (MD = -2.3, 95%CI [-3.9, -0.7]; p = 0.005).
Apabetalone inhibits the Bromodomain and extra-terminal (BET) protein, acting epigenetically to reduce harmful gene expression in chronic diseases [144]. It was tested in patients with type 2 diabetes and acute coronary syndrome in a 2 year clinical trial (n = 464, mean age (range) = 73 (71–76)), and it was found to improve MoCA scores in those with a low cognitive baseline (MoCA < 22) (MD = 2.1; p = 0.02), but not in those with a higher baseline (MoCA = 22 or greater) [145].
Cerebral blood flow
The Lacunar Intervention Trial-2 (LACI-2) is a randomized phase 2 clinical trial assessing the use of cilostazol and isosorbide mononitrate (ISMN) in patients following lacunar stroke (n = 363). They found that the combination of cilostazol and ISMN significantly reduced the risk of cognitive impairment (aOR = 0.44 [95% CI, 0.23 to 0.85]), dependence (aOR = 0.14 [95% CI, 0.03 to 0.59]), and improved quality of life (adjusted mean difference = 0.10 [95% CI, 0.03 to 0.17]) [146].
Tadalafil and Sildenafil are phosphodiesterase inhibitors that are used to treat pulmonary hypertension and male erectile dysfunction. Tadalafil was trialed in a small group of patients (N = 55, mean age (SD) = 66.8 (8.6) years) with SVD. There was no significant effect on cerebral blood flow, as measured using MRI [147]. In contrast, sildenafil was found in early trials to improve cerebral blood flow over three weeks [148, 149], although further studies are needed to assess the effect of this change on cognition over time.
Adrenomedullin is a peptide thought to improve cerebral circulation in patients with vascular diseases by improving angiogenesis, vasodilation, and reducing inflammation. A phase 2 trial (NCT06072118) [150] has been completed, although not yet published, to assess whether it improves cerebral blood flow in 60 patients with CADASIL, a genetic form of VCID.
Coumarin, the precursor to warfarin, has a derivative named daphnetin that provides protection to blood vessels and ischemic tissues through anticoagulation, modulating vasoactive substances, improving circulation, and reducing inflammation [151]. At the time of writing, a clinical trial (NCT06507488) was conducted to test its effectiveness over six months in 206 individuals with vascular cognitive impairment.
Non-invasive neurostimulation
Transcranial Magnetic Stimulation (TMS) [152] is a well-established therapy for mood disorders, and several small studies have investigated its potential for post-stroke cognitive impairment. Researchers have suggested that it functions by modulating synaptic plasticity, activating the default mode network, and improving CNS angiogenesis. The few small trials that have been conducted have shown improvements in cognition, but the treatment settings and measured outcomes have been highly variable [152].
Transcranial photobiomodulation (tPBM) uses noninvasive laser therapy to upregulate mitochondrial activity and increase cerebral oxygenation and cerebral blood flow. It has improved cognitive function in some animal populations and is currently being tested in a trial (NCT06497413) of 100 adults (18–80 years of age) to assess whether it can induce improvements in cerebral blood flow and neurocognitive function over four weeks [153].
Future research directions
The fields of dementia research and clinical practice are now in an exciting state of flux from developments in blood-based biomarkers, advanced artificial intelligence (AI) imaging analysis, and the rollout of disease-modifying immunotherapies for AD. Given this rapid rate of change, the direction of VCID research needs to be future-proof, judicious, and well calibrated. Academic research is littered with examples of positive publication bias, misdirected funds, falsified research, setting the field back years, and wasting vast amounts of money [154]. We outline some key strategic foci for future research on VCID treatment in the following section.
While new clinical trials and longitudinal studies are crucial, future research should exploit the large extant trial data and national registries as a more expedient way of understanding treatments. For example, as discussed in previous sections, there have been nine studies examining the effect of antipsychotics in mixed samples of patients with AD and VaD, but in each of them, the sample of VaD patients was too small to analyze independently. Collecting individual participant data (IPD) from these studies to develop a database of VaD patients for meaningful analysis would help develop more targeted treatment protocols. Similarly, trials of antihypertensive and antihyperglycemic agents are often too small to provide clear evidence of the effect of one subclass of treatment compared to the other. Promoting transparency in equally publishing both negative and positive studies would aid in data collection. Thus, concatenating extant trial data into IPD meta-analyses and using national registry data may provide greater insight into the utility of various medication subclasses.
In future trials, blood-based and imaging biomarkers should be applied to delineate the overlapping and distinct contributions of Alzheimer’s and vascular pathology to dementia to better target treatment. P-tau 181, p-tau 217, GFAP, NFL, and A-Beta 40/42 are blood tests that are well validated to quantify Alzheimer’s pathology [155]. While there are no currently validated biomarker for VCID, the NIH funded MarkVCID1 study assessed four fluid-based biomarkers (NfL, EGF, ExE, and CSF-PlGF) and MarkVCID2 is currently assessing utility of NfL in a cohort of approximately 1800 participants [156]. Simultaneously, AI segmentation and quantification of grey matter volume, white matter volume, WMH, ePVS, lacunes, strokes, and CMBs are becoming more widely available and can provide a more reliable and precise assessment of vascular brain burden relative to age [157]. In addition to traditional MRI markers of VCID, novel diffusion tensor imaging (DTI) measures of white matter integrity, such as the peak width of the skeletonized mean diffusivity (PSMD) and difference in diffusion function (DDF) [158], have been shown to predict cognition and cognitive decline better than previous structural MRI measures [159]. These are current research tools, but their clinical application for diagnosis, prognosis, and as a biomarker should continue to be investigated. Applying these tools in randomized trials and longitudinal studies will allow for much greater precision in prescribing treatments for underlying pathologies, as well as providing more accurate prognostic information for VCID, AD, and mixed cases. Similar to the revolution in precision medicine for oncology [160], the long-term ambition for VCID and AD treatment is that treatment mechanisms and doses should be targeted to the subtype and severity of the underlying pathology.
Most studies indicate that the more advanced forms of VCID show little treatment responsiveness [161], but to detect earlier and milder forms of VCID affordable, scalable, targeted neuropsychological testing systems need to be validated and made available [156]. A key challenge in detecting VCID in its early stages is that mild deficits can be difficult to detect and thus often require time-consuming and expensive neuropsychological testing. Furthermore, traditional neuropsychological batteries used in dementia clinical practice have been oriented towards AD, at times underestimating or failing to measure cognitive domains more commonly affected in VCID (e.g., processing speed and executive function). To improve the widespread detection of mild, early clinical trials should utilize validated online, patient-driven, neuropsychological testing batteries designed for VCID assessment to assess their applicability in clinical practice and treatment algorithm development. As part of the effort to detect and treat VCID in its early or prodromal phase, studies of asymptomatic individuals should include markers of ccSVD (e.g., WMH, ePVS, lacunes) to assess the best practice for their treatment and the extent of their reversibility.
Finally, in the future, more rational, data-driven drug discovery is needed to select the most likely candidates for successful treatment. There is a poor understanding of the mechanism for many current treatments for VCID (e.g., non-specific anti-inflammatory or anti-oxidant effects), and without mechanistic understanding, selecting compounds for initial trials can be highly speculative and unreliable. In other fields of medicine, both monogenic forms of diseases and large-scale genome wide association studies (GWAS) have been used to identify druggable targets and develop treatments [162]. For example, identification of CCR5 gene variants in an HIV GWAS was used to develop the commonly used medication Maraviroc [163]. Fongang et al. (2024) [164], in the largest GWAS of VaD patients to date, identified several possible candidate genes including TOMM40, MARCHF10, APOE, SPRY2, ERBB4, RASGRP1 and FOXA2. Using the drug-gene interaction database [165] and more advanced druggable Mendelian randomization approaches, novel drug targets and existing drugs can be identified for targeted drug development and repurposing, respectively. For example, byrostatin 1, a macrolide that acts as a protein kinase C (PKC) agonist, is known to interact with RASGRP1, a candidate gene. It has shown some early promise in AD mouse models [166], and thus may be a candidate for further testing in VCID models and trials. Taken together, the field needs to use large amounts of clinical and genetic data to better select candidate targets and drugs for future testing.
In conclusion, our review shows that, despite our increasing knowledge of the mechanisms of CVD, VCID remains a major health challenge in the 21st century. Even though it is a potentially highly preventable form of dementia, its risk factors continue to increase on a global scale, and no effective treatments exist. Research into VCID lags significantly behind that on both cardiovascular diseases and AD. This must change if we are to meet the dementia challenge faced by most aging societies.
Funding
We acknowledge the Dementia Australia Research Foundation for providing funding support for this work under the Royce Simmons Foundation Project Grant.
References
Nichols E, Steinmetz JD, Vollset SE, Fukutaki K, Chalek J, Abd-Allah F, et al. Estimation of the global prevalence of dementia in 2019 and forecasted prevalence in 2050: an analysis for the Global Burden of Disease Study 2019. Lancet Public Health. 2022;7:105–e125.
Power MC, Mormino E, Soldan A, James BD, Yu L, Armstrong NM, et al. Combined neuropathological pathways account for age-related risk of dementia. Ann Neurol. 2018;84:10–22.
Cao Q, Tan CC, Xu W, Hu H, Cao XP, Dong Q, et al. The Prevalence of Dementia: A Systematic Review and Meta-Analysis. J Alzheimers Dis. 2020;73:1157–66.
Rockwood K, Wentzel C, Hachinski V, Hogan DB, MacKnight C, McDowell I, et al. Prevalence and outcomes of vascular cognitive impairment. Neurology. 2000;54:447.
Cleveland Clinic. Vascular Dementia. Dis Cond. 2024:1–3.
Peng Y, Jin H, Xue Y-H, Chen Q, Yao S-Y, Du M-Q, et al. Current and future therapeutic strategies for Alzheimer’s disease: an overview of drug development bottlenecks. Front Aging Neurosci. 2023;15:1206572.
Livingston G, Huntley J, Liu KY, Costafreda SG, Selbæk G, Alladi S, et al. Dementia prevention, intervention, and care: 2024 report of the Lancet standing Commission. Lancet. 2024;404:572–628.
National Health Service (NHS UK). Vascular Dementia. Cond AZ. 2023:1–2.
Wang G, Albayrak A, van der Cammen TJM. A systematic review of non-pharmacological interventions for BPSD in nursing home residents with dementia: from a perspective of ergonomics. Int Psychogeriatrics. 2019;31:1137–49.
Dubois B, von Arnim CAF, Burnie N, Bozeat S, Cummings J. Biomarkers in Alzheimer’s disease: role in early and differential diagnosis and recognition of atypical variants. Alzheimers Res Ther. 2023;15:175.
Chui HC, Victoroff JI, Margolin D, Jagust W, Shankle R, Katzman R. Criteria for the diagnosis of ischemic vascular dementia proposed by the State of California Alzheimer’s Disease Diagnostic and Treatment Centers. Neurology. 1992;42:473–80.
Román GC, Tatemichi TK, Erkinjuntti T, Cummings JL, Masdeu JC, Garcia JH, et al. Vascular dementia: diagnostic criteria for research studies. Report of the NINDS-AIREN International Workshop. Neurology. 1993;43:250–60.
Erkinjuntti T, Inzitari D, Pantoni L, Wallin A, Scheltens P, Rockwood K, et al. Research criteria for subcortical vascular dementia in clinical trials. J Neural Transm Suppl. 2000;59:23–30.
Corriveau RA, Bosetti F, Emr M, Gladman JT, Koenig JI, Moy CS, et al. The Science of Vascular Contributions to Cognitive Impairment and Dementia (VCID): A Framework for Advancing Research Priorities in the Cerebrovascular Biology of Cognitive Decline., 2016.
Sachdev P, Kalaria R, O’Brien J, Skoog I, Alladi S, Black SE, et al. Diagnostic criteria for vascular cognitive disorders: a VASCOG statement. Alzheimer Dis Assoc Disord. 2014;28:206–18.
Wen W, Sachdev PS, Li JJ, Chen X, Anstey KJ. White matter hyperintensities in the forties: their prevalence and topography in an epidemiological sample aged 44-48. Hum Brain Mapp. 2009;30:1155–67.
Iadecola C, Duering M, Hachinski V, Joutel A, Pendlebury ST, Schneider JA, et al. Vascular Cognitive Impairment and Dementia: JACC Scientific Expert Panel. J Am Coll Cardiol. 2019;73:3326–44.
Lo JW, Crawford JD, Desmond DW, Godefroy O, Jokinen H, Mahinrad S, et al. Profile of and risk factors for poststroke cognitive impairment in diverse ethnoregional groups. Neurology. 2019;93:e2257–71.
Chauhan G, Adams HHH, Satizabal CL, Bis JC, Teumer A, Sargurupremraj M, et al. Genetic and lifestyle risk factors for MRI-defined brain infarcts in a population-based setting. Neurology. 2019;92:e486–503.
Duering M, Biessels GJ, Brodtmann A, Chen C, Cordonnier C, de Leeuw F-E, et al. Neuroimaging standards for research into small vessel disease-advances since 2013. Lancet Neurol. 2023;22:602–18.
Chen X, Wen W, Anstey KJ, Sachdev PS. Prevalence, incidence, and risk factors of lacunar infarcts in a community sample. Neurology. 2009;73:266–72.
Wardlaw JM, Valdés Hernández MC, Muñoz-Maniega S. What are white matter hyperintensities made of? Relevance to vascular cognitive impairment. J Am Heart Assoc. 2015;4:1140.
Lin J, Wang D, Lan L, Fan Y. Multiple Factors Involved in the Pathogenesis of White Matter Lesions. Biomed Res Int. 2017;2017:9372050.
de Leeuw FE, de Groot JC, Achten E, Oudkerk M, Ramos LM, Heijboer R, et al. Prevalence of cerebral white matter lesions in elderly people: a population based magnetic resonance imaging study. The Rotterdam Scan Study. J Neurol Neurosurg Psychiatry. 2001;70:9–14.
Wen W, Sachdev P. The topography of white matter hyperintensities on brain MRI in healthy 60- to 64-year-old individuals. Neuroimage. 2004;22:144–54.
Han F, Zhai F-F, Wang Q, Zhou L-X, Ni J, Yao M, et al. Prevalence and Risk Factors of Cerebral Small Vessel Disease in a Chinese Population-Based Sample. J Stroke. 2018;20:239–46.
Igwe KC, Lao PJ, Vorburger RS, Banerjee A, Rivera A, Chesebro A, et al. Automatic quantification of white matter hyperintensities on T2-weighted fluid attenuated inversion recovery magnetic resonance imaging. Magn Reson Imaging. 2022;85:71–79.
Wardlaw JM, Chappell FM, Valdés Hernández MDC, Makin SDJ, Staals J, Shuler K, et al. White matter hyperintensity reduction and outcomes after minor stroke. Neurology. 2017;89:1003–10.
Chen S-P, Chou K-H, Fuh J-L, Huang Y-H, Huang C-C, Lirng J-F, et al. Dynamic Changes in White Matter Hyperintensities in Reversible Cerebral Vasoconstriction Syndrome. JAMA Neurol. 2018;75:1106–13.
Vernooij MW, van der Lugt A, Ikram MA, Wielopolski PA, Niessen WJ, Hofman A, et al. Prevalence and risk factors of cerebral microbleeds: the Rotterdam Scan Study. Neurology. 2008;70:1208–14.
Graff-Radford J, Botha H, Rabinstein AA, Gunter JL, Przybelski SA, Lesnick T, et al. Cerebral microbleeds: Prevalence and relationship to amyloid burden. Neurology. 2019;92:e253–262.
Debette S, Schilling S, Duperron M-G, Larsson SC, Markus HS. Clinical Significance of Magnetic Resonance Imaging Markers of Vascular Brain Injury: A Systematic Review and Meta-analysis. JAMA Neurol. 2019;76:81–94.
Ding J, Sigurðsson S, Jónsson PV, Eiriksdottir G, Meirelles O, Kjartansson O, et al. Space and location of cerebral microbleeds, cognitive decline, and dementia in the community. Neurology. 2017;88:2089–97.
Poels MMF, Ikram MA, van der Lugt A, Hofman A, Niessen WJ, Krestin GP, et al. Cerebral microbleeds are associated with worse cognitive function: the Rotterdam Scan Study. Neurology. 2012;78:326–33.
Zhu Y-C, Dufouil C, Mazoyer B, Soumaré A, Ricolfi F, Tzourio C, et al. Frequency and location of dilated Virchow-Robin spaces in elderly people: a population-based 3D MR imaging study. AJNR Am J Neuroradiol. 2011;32:709–13.
Walter AE, Savalia K, Yoon J, Morrison J, Schneider ALC, Diaz-Arrastia R, et al. Change in Enlarged Perivascular Spaces over Time and Associations with Outcomes After Traumatic Brain Injury. Neurotrauma Rep. 2024;5:738–48.
Smeijer D, Ikram MK, Hilal S. Enlarged Perivascular Spaces and Dementia: A Systematic Review. J Alzheimers Dis. 2019;72:247–56.
Hilal S, Tan CS, Adams HHH, Habes M, Mok V, Venketasubramanian N, et al. Enlarged perivascular spaces and cognition: A meta-analysis of 5 population-based studies. Neurology. 2018;91:e832–842.
Chistiakov DA, Myasoedova VA, Revin VV, Orekhov AN, Bobryshev YV. The phenomenon of atherosclerosis reversal and regression: Lessons from animal models. Exp Mol Pathol. 2017;102:138–45.
Björkegren JLM, Hägg S, Talukdar HA, Foroughi Asl H, Jain RK, Cedergren C, et al. Plasma Cholesterol–Induced Lesion Networks Activated before Regression of Early, Mature, and Advanced Atherosclerosis. PLOS Genet. 2014;10:e1004201.
Burns A, O’Brien J, Auriacombe S, Ballard C, Broich K, Bullock R, et al. Clinical practice with anti-dementia drugs: a consensus statement from British Association for Psychopharmacology. J Psychopharmacol. 2006;20:732–55.
Erkinjuntti T, Kurz A, Gauthier S, Bullock R, Lilienfeld S, Damaraju CV. The assessment of treatment effects in vascular dementia: A critical review of scales used in clinical trials. Int J Geriatr Psychiatry. 2002;17:805–15.
Knopman DS, DeKosky ST, Cummings JL, Chui H, Corey-Bloom J, Relkin N, et al. Practice parameter: diagnosis of dementia (an evidence-based review). Report of the Quality Standards Subcommittee of the American Academy of Neurology. Neurology. 2001;56:1143–53.
Roman GC, Kalaria RN. Vascular determinants of cholinergic deficits in Alzheimer disease and vascular dementia. Neurobiol Aging. 2006;27:1769–85.
Wang J, Zhang H-Y, Tang X-C. Cholinergic deficiency involved in vascular dementia: possible mechanism and strategy of treatment. Acta Pharmacol Sin. 2009;30:879–88.
Luchsinger JA, Reitz C, Honig LS, Tang MX, Shea S, Mayeux R. Aggregation of vascular risk factors and risk of incident Alzheimer disease. Neurology. 2005;65:545–51.
Dawbarn D, Allen SJ Neurobiology of Alzheimer’s Disease. 3rd edition. Oxford: Oxford University Press; 2007.
Battle CE, Abdul-Rahim AH, Shenkin SD, Hewitt J, Quinn TJ Cholinesterase inhibitors for vascular dementia and other vascular cognitive impairments: a network meta-analysis. Cochrane Database Syst Rev. 2021. 2021. https://doi.org/10.1002/14651858.CD013306.pub2.
Muir RT, Hill MD, Black SE, Smith EE. Minimal clinically important difference in Alzheimer’s disease: Rapid review. Alzheimer’s Dement. 2024;20:3352–63.
Kavirajan H, Schneider LS. Efficacy and adverse effects of cholinesterase inhibitors and memantine in vascular dementia: a meta-analysis of randomised controlled trials. Lancet Neurol. 2007;6:782–92.
Dichgans M, Markus HS, Salloway S, Verkkoniemi A, Moline M, Wang Q, et al. Donepezil in patients with subcortical vascular cognitive impairment: a randomised double-blind trial in CADASIL. Lancet Neurol. 2008;7:310–8.
Lilienfeld S. Galantamine-a novel cholinergic drug with a unique dual mode of action for the treatment of patients with Alzheimer’s disease. CNS Drug Rev. 2002;8:159–76.
Auchus AP, Brashear HR, Salloway S, Korczyn AD, De Deyn PP, Gassmann-Mayer C. Galantamine treatment of vascular dementia: a randomized trial. Neurology. 2007;69:448–58.
Erkinjuntti T, Kurz A, Gauthier S, Bullock R, Lilienfeld S, Damaraju CV. Efficacy of galantamine in probable vascular dementia and Alzheimer’s disease combined with cerebrovascular disease: a randomised trial. Lancet. 2002;359:1283–90.
Shi X, Ren G, Cui Y, Xu Z. Comparative Efficacy and Acceptability of Cholinesterase Inhibitors and Memantine Based on Dosage in Patients with Vascular Cognitive Impairment: A Network Meta-analysis. Curr Alzheimer Res. 2022;19:133–45.
Ballard C, Sauter M, Scheltens P, He Y, Barkhof F, van Straaten ECW, et al. Efficacy, safety and tolerability of rivastigmine capsules in patients with probable vascular dementia: the VantagE study. Curr Med Res Opin. 2008;24:2561–74.
Mok V, Wong A, Ho S, Leung T, Lam WWM, Wong KS. Rivastigmine in Chinese patients with subcortical vascular dementia. Neuropsychiatr Dis Treat. 2007;3:943–8.
Narasimhalu K, Effendy S, Sim CH, Lee JM, Chen I, Hia SB, et al. A randomized controlled trial of rivastigmine in patients with cognitive impairment no dementia because of cerebrovascular disease. Acta Neurol Scand. 2010;121:217–24.
Wilcock G, Möbius HJ, Stöffler A. A double-blind, placebo-controlled multicentre study of memantine in mild to moderate vascular dementia (MMM500). Int Clin Psychopharmacol. 2002;17:297–305.
Orgogozo J-M, Rigaud A-S, Stöffler A, Möbius H-J, Forette F. Efficacy and safety of memantine in patients with mild to moderate vascular dementia: a randomized, placebo-controlled trial (MMM 300). Stroke. 2002;33:1834–9.
Jia J, Wei C, Liang J, Zhou A, Zuo X, Song H, et al. The effects of DL-3-n-butylphthalide in patients with vascular cognitive impairment without dementia caused by subcortical ischemic small vessel disease: A multicentre, randomized, double-blind, placebo-controlled trial. Alzheimers Dement. 2016;12:89–99.
Abdoulaye IA, Guo YJ. A Review of Recent Advances in Neuroprotective Potential of 3-N-Butylphthalide and Its Derivatives. Biomed Res Int. 2016;2016:5012341.
Wang Y, Kou S, Yang S, Zhang C, Wang S, Wang Y. Effect of Butylphthalide soft capsules on cognitive function and dementia-related factors in elderly patients with Parkinson’s disease dementia during the COVID-19 pandemic. Am J Transl Res. 2024;16:496–505.
Yang DM, Chang WL, Tang P. Effects of Butylphthalide and Danhong injection on cognitive function and serum related factors in patients with vascular dementia. Chinese J Clin Res. 2019;32:1041–4.
Zhang H, Wu H, Qi X, Wu F, Zhang D. Effect of butylphthalide combined with idebenone on vascular dementia: A retrospective observational analysis. Medicine. 2024;103:e37495.
Weinmann S, Roll S, Schwarzbach C, Vauth C, Willich SN. Effects of Ginkgo biloba in dementia: systematic review and meta-analysis. BMC Geriatr. 2010;10:14.
Demarin V, Bašić Kes V, Trkanjec Z, Budišić M, Bošnjak Pašić M, Črnac P, et al. Efficacy and safety of Ginkgo biloba standardized extract in the treatment of vascular cognitive impairment: a randomized, double-blind, placebo-controlled clinical trial. Neuropsychiatr Dis Treat. 2017;13:483–90.
Fiani B, Covarrubias C, Wong A, Doan T, Reardon T, Nikolaidis D, et al. Cerebrolysin for stroke, neurodegeneration, and traumatic brain injury: review of the literature and outcomes. Neurol Sci. 2021;42:1345–53.
Xiao S, Yan H, Yao P. The Efficacy of Cerebrolysinin Patients With Vascular Dementia: Results of A Chinese Multicentre, Randomised, Double-Blind, Placebo-Controlled Trial. Hong Kong J Psychiatry. 1999;9:13–19.
Guekht AB, Moessler H, Novak PH, Gusev EI. Cerebrolysin in vascular dementia: Improvement of clinical outcome in a randomized, double-blind, placebo-controlled multicenter trial. J Stroke Cerebrovasc Dis. 2011;20:310–8.
Masserini F, Baso G, Gendarini C, Pantoni L. Therapeutic strategies in vascular cognitive impairment: A systematic review of population, intervention, comparators, and outcomes. Alzheimer’s Dement. 2023;19:5795–804.
Caramelli P, Laks J, Palmini ALF, Nitrini R, Chaves MLF, Forlenza OV, et al. Effects of galantamine and galantamine combined with nimodipine on cognitive speed and quality of life in mixed dementia: a 24-week, randomized, placebo-controlled exploratory trial (the REMIX study). Arq Neuropsiquiatr. 2014;72:411–7.
Tampi RR, Jeste DV. Dementia Is More Than Memory Loss: Neuropsychiatric Symptoms of Dementia and Their Nonpharmacological and Pharmacological Management. Am J Psychiatry. 2022;179:528–43.
Abraha I, Rimland JM, Trotta FM, Dell’Aquila G, Cruz-Jentoft A, Petrovic M, et al. Systematic review of systematic reviews of non-pharmacological interventions to treat behavioural disturbances in older patients with dementia. The SENATOR-OnTop series. BMJ Open. 2017;7:e012759.
Diniz BS, Butters MA, Albert SM, Dew MA, Reynolds CF. 3rd. Late-life depression and risk of vascular dementia and Alzheimer’s disease: systematic review and meta-analysis of community-based cohort studies. Br J Psychiatry. 2013;202:329–35.
Orgeta V, Qazi A, Spector A, Orrell M. Psychological treatments for depression and anxiety in dementia and mild cognitive impairment: systematic review and meta-analysis. Br J Psychiatry. 2015;207:293–8.
Watt JA, Goodarzi Z, Veroniki AA, Nincic V, Khan PA, Ghassemi M, et al. Comparative efficacy of interventions for reducing symptoms of depression in people with dementia: systematic review and network meta-analysis. BMJ. 2021;372:n532.
Fritze F, Ehrt U, Hortobagyi T, Ballard C, Aarsland D. Depressive symptoms in Alzheimer’s disease and lewy body dementia: a one-year follow-up study. Dement Geriatr Cogn Disord. 2011;32:143–9.
Asmer MS, Kirkham J, Newton H, Ismail Z, Elbayoumi H, Leung RH, et al. Meta-Analysis of the Prevalence of Major Depressive Disorder Among Older Adults With Dementia. J Clin Psychiatry. 2018;79:17r11772.
Lavretsky H, Zheng L, Weiner MW, Mungas D, Reed B, Kramer JH, et al. Association of depressed mood and mortality in older adults with and without cognitive impairment in a prospective naturalistic study. Am J Psychiatry. 2010;167:589–97.
Feng L, Scherer SC, Tan BY, Chan G, Fong NP, Ng TP. Comorbid cognitive impairment and depression is a significant predictor of poor outcomes in hip fracture rehabilitation. Int Psychogeriatrics. 2010;22:246–53.
Dudas R, Malouf R, McCleery J, Dening T. Antidepressants for treating depression in dementia. Cochrane Database Syst Rev. 2018;8:CD003944.
de Vasconcelos Cunha UG, Lopes Rocha F, Avila de Melo R, Alves Valle E, de Souza Neto JJ, Mendes Brega R, et al. A placebo-controlled double-blind randomized study of venlafaxine in the treatment of depression in dementia. Dement Geriatr Cogn Disord. 2007;24:36–41.
Sheline YI, Pieper CF, Barch DM, Welsh-Bohmer K, McKinstry RC, MacFall JR, et al. Support for the vascular depression hypothesis in late-life depression: results of a 2-site, prospective, antidepressant treatment trial. Arch Gen Psychiatry. 2010;67:277–85.
Taragano FE, Bagnatti P, Allegri RF. A double-blind, randomized clinical trial to assess the augmentation with nimodipine of antidepressant therapy in the treatment of ‘vascular depression. Int Psychogeriatrics. 2005;17:487–98.
Li X, Zhang C. Comparative efficacy of nine antidepressants in treating Chinese patients with post-stroke depression: A network meta-analysis. J Affect Disord. 2020;266:540–8.
Tiel C, Sudo FK, Alves GS, Ericeira-Valente L, Moreira DM, Laks J, et al. Neuropsychiatric symptoms in Vascular Cognitive Impairment: a systematic review. Dement Neuropsychol. 2015;9:230–6.
Mühlbauer V, Möhler R, Dichter MN, Zuidema SU, Köpke S, Luijendijk HJ Antipsychotics for agitation and psychosis in people with Alzheimer’s disease and vascular dementia. Cochrane Database Syst Rev. 2021;2021.
Tariot PN, Schneider L, Katz IR, Mintzer JE, Street J, Copenhaver M, et al. Quetiapine treatment of psychosis associated with dementia: a double-blind, randomized, placebo-controlled clinical trial. Am J Geriatr Psychiatry Off J Am Assoc Geriatr Psychiatry. 2006;14:767–76.
Katz IR, Jeste DV, Mintzer JE, Clyde C, Napolitano J, Brecher M. Comparison of risperidone and placebo for psychosis and behavioral disturbances associated with dementia: a randomized, double-blind trial. J Clin Psychiatry. 1999;60:107–15.
Zhong KX, Tariot PN, Mintzer J, Minkwitz MC, Devine NA. Quetiapine to treat agitation in dementia: a randomized, double-blind, placebo-controlled study. Curr Alzheimer Res. 2007;4:81–93.
Allain H, Dautzenberg PH, Maurer K, Schuck S, Bonhomme D, Gérard D. Double blind study of tiapride versus haloperidol and placebo in agitation and aggressiveness in elderly patients with cognitive impairment. Psychopharmacology. 2000;148:361–6.
Brodaty H, Ames D, Snowdon J, Woodward M, Kirwan J, Clarnette R, et al. A randomized placebo-controlled trial of risperidone for the treatment of aggression, agitation, and psychosis of dementia. J Clin Psychiatry. 2003;64:134–43.
Deberdt WG, Dysken MW, Rappaport SA, Feldman PD, Young CA, Hay DP, et al. Comparison of olanzapine and risperidone in the treatment of psychosis and associated behavioral disturbances in patients with dementia. Am J Geriatr Psychiatry. 2005;13:722–30.
Nichols E, Szoeke CEI, Vollset SE, et al. Global, regional, and national burden of Alzheimer’s disease and other dementias, 1990–2016: a systematic analysis for the Global Burden of Disease Study 2016. Lancet Neurol. 2019;18:88–106.
Satizabal CL, Beiser AS, Chouraki V, Chêne G, Dufouil C, Seshadri S. Incidence of Dementia over Three Decades in the Framingham Heart Study. N Engl J Med. 2016;374:523–32.
Grasset L, Brayne C, Joly P, Jacqmin-Gadda H, Peres K, Foubert-Samier A, et al. Trends in dementia incidence: Evolution over a 10-year period in France. Alzheimers Dement. 2016;12:272–80.
Matthews FE, Stephan BCM, Robinson L, Jagger C, Barnes LE, Arthur A, et al. A two decade dementia incidence comparison from the Cognitive Function and Ageing Studies I and II. Nat Commun. 2016;7:11398.
Schrijvers EMC, Verhaaren BFJ, Koudstaal PJ, Hofman A, Ikram MA, Breteler MMB. Is dementia incidence declining?: Trends in dementia incidence since 1990 in the Rotterdam Study. Neurology. 2012;78:1456–63.
Bushnell C, Kernan WN, Sharrief AZ, Chaturvedi S, Cole JW, Cornwell WK, et al. 2024 Guideline for the Primary Prevention of Stroke: A Guideline From the American Heart Association/American Stroke Association. Stroke. 2024;55:e344–e424.
Smith JR, Pike JR, Gottesman RF, Knopman DS, Lutsey PL, Palta P, et al. Contribution of Modifiable Midlife and Late-Life Vascular Risk Factors to Incident Dementia. JAMA Neurol. 2025. 2 June 2025. https://doi.org/10.1001/jamaneurol.2025.1495.
Sachdev PS, Thalamuthu A, Mather KA, Ames D, Wright MJ, Wen W. White Matter Hyperintensities Are Under Strong Genetic Influence. Stroke. 2016;47:1422–8.
Kerber KA, Whitman GT, Brown DL, Baloh RW. Increased risk of death in community-dwelling older people with white matter hyperintensities on MRI. J Neurol Sci. 2006;250:33–38.
Wardlaw JM, Debette S, Jokinen H, De Leeuw F-E, Pantoni L, Chabriat H, et al. ESO Guideline on covert cerebral small vessel disease. Eur Stroke J. 2021;6:CXI–CLXII.
Wardlaw JM, Chabriat H, de Leeuw F-E, Debette S, Dichgans M, Doubal F, et al. European stroke organisation (ESO) guideline on cerebral small vessel disease, part 2, lacunar ischaemic stroke. Eur Stroke J. 2024;9:5–68.
Pepper E, Bateman G, Fiorentino M, Brodtmann A. Consensus statement for the management of incidentally found brain white matter hyperintensities in general medical practice. Med J Aust. 2023;219:278–84.
Lennon MJ, Lam BCP, Lipnicki DM, Crawford JD, Peters R, Schutte AE, et al. Use of Antihypertensives, Blood Pressure, and Estimated Risk of Dementia in Late Life: An Individual Participant Data Meta-Analysis. JAMA Netw Open. 2023;6:e2333353.
Lennon MJ, Lipnicki DM, Lam BCP, Crawford JD, Schutte AE, Peters R, et al. Blood Pressure, Antihypertensive Use, and Late-Life Alzheimer and Non-Alzheimer Dementia Risk: An Individual Participant Data Meta-Analysis. Neurology. 2024;103:e209715.
Lennon MJ, Makkar SR, Crawford JD, Sachdev PS. Midlife Hypertension and Alzheimer’s Disease: A Systematic Review and Meta-Analysis. J Alzheimers Dis. 2019;71:307–16.
Lennon MJ, Koncz R, Sachdev PS Hypertension and Alzheimer’s disease: is the picture any clearer? Curr Opin Psychiatry. 2021;34.
Lennon MJ, Lam BCP, Crawford J, Brodaty H, Kochan NA, Trollor JN, et al. Does Antihypertensive Use Moderate the Effect of Blood Pressure on Cognitive Decline in Older People? J. Gerontol A Biol Sci Med Sci. 2021;76:859–66.
Peters R, Xu Y, Fitzgerald O, Aung HL, Beckett N, Bulpitt C, et al. Blood pressure lowering and prevention of dementia: an individual patient data meta-analysis. Eur Heart J. 2022;43:4980–90.
Markus HS, Egle M, Croall ID, Sari H, Khan U, Hassan A, et al. PRESERVE: Randomized Trial of Intensive Versus Standard Blood Pressure Control in Small Vessel Disease. Stroke. 2021;52:2484–93.
Bath PM, Scutt P, Blackburn DJ, Ankolekar S, Krishnan K, Ballard C, et al. Intensive versus Guideline Blood Pressure and Lipid Lowering in Patients with Previous Stroke: Main Results from the Pilot ‘Prevention of Decline in Cognition after Stroke Trial’ (PODCAST) Randomised Controlled Trial. PLoS ONE. 2017;12:e0164608.
Shepherd J, Blauw GJ, Murphy MB, Bollen ELEM, Buckley BM, Cobbe SM, et al. Pravastatin in elderly individuals at risk of vascular disease (PROSPER): a randomised controlled trial. Lancet. 2002;360:1623–30.
Giugliano RP, Wiviott SD, Blazing MA, De Ferrari GM, Park J-G, Murphy SA, et al. Long-term Safety and Efficacy of Achieving Very Low Levels of Low-Density Lipoprotein Cholesterol: A Prespecified Analysis of the IMPROVE-IT Trial. JAMA Cardiol. 2017;2:547–55.
Bosch J, O’Donnell M, Swaminathan B, Lonn EM, Sharma M, Dagenais G, et al. Effects of blood pressure and lipid lowering on cognition: Results from the HOPE-3 study. Neurology. 2019;92:e1435–e1446.
Zhang H, Cui Y, Zhao Y, Dong Y, Duan D, Wang J, et al. Effects of sartans and low-dose statins on cerebral white matter hyperintensities and cognitive function in older patients with hypertension: a randomized, double-blind and placebo-controlled clinical trial. Hypertens Res. 2019;42:717–29.
Ji T, Zhao Y, Wang J, Cui Y, Duan D, Chai Q, et al. Effect of Low-Dose Statins and Apolipoprotein E Genotype on Cerebral Small Vessel Disease in Older Hypertensive Patients: A Subgroup Analysis of a Randomized Clinical Trial. J Am Med Dir Assoc. 2018;19:995–1002.e4.
ten Dam VH, van den Heuvel DMJ, van Buchem MA, Westendorp RGJ, Bollen ELEM, Ford I, et al. Effect of pravastatin on cerebral infarcts and white matter lesions. Neurology. 2005;64:1807–9.
Zhang X, Wen J, Zhang Z. Statins use and risk of dementia: A dose-response meta analysis. Medicine. 2018;97:e11304.
Biessels GJ, Despa F. Cognitive decline and dementia in diabetes mellitus: mechanisms and clinical implications. Nat Rev Endocrinol. 2018;14:591–604.
Ramirez A, Wolfsgruber S, Lange C, Kaduszkiewicz H, Weyerer S, Werle J, et al. Elevated HbA1c is associated with increased risk of incident dementia in primary care patients. J Alzheimers Dis. 2015;44:1203–12.
Campbell JM, Stephenson MD, de Courten B, Chapman I, Bellman SM, Aromataris E. Metformin Use Associated with Reduced Risk of Dementia in Patients with Diabetes: A Systematic Review and Meta-Analysis. J Alzheimers Dis. 2018;65:1225–36.
Weinstein G, Davis-Plourde KL, Conner S, Himali JJ, Beiser AS, Lee A, et al. Association of metformin, sulfonylurea and insulin use with brain structure and function and risk of dementia and Alzheimer’s disease: Pooled analysis from 5 cohorts. PLoS ONE. 2019;14:e0212293.
Nørgaard CH, Friedrich S, Hansen CT, Gerds T, Ballard C, Møller DV, et al. Treatment with glucagon-like peptide-1 receptor agonists and incidence of dementia: Data from pooled double-blind randomized controlled trials and nationwide disease and prescription registers. Alzheimer’s Dement. 2022;8:e12268.
Kitt K, Murphy R, Clarke A, Reddin C, Ferguson J, Bosch J, et al. Antiplatelet therapy and incident cognitive impairment or dementia-a systematic review and meta-analysis of randomised clinical trials. Age Ageing. 2023;52:afad197.
Ning W, Wang S, Tang H, Wu S, Huang X, Liu B, et al. Effect of different oral anticoagulants on cognitive function in patients with atrial fibrillation: A Bayesian network meta-analysis. Medicine. 2024;103:e37750.
Sridharan ND, Asaadi S, Thirumala PD, Avgerinos ED. A systematic review of cognitive function after carotid endarterectomy in asymptomatic patients. J Vasc Surg. 2022;75:2074–85.
Aleksic M, Huff W, Hoppmann B, Heckenkamp J, Pukrop R, Brunkwall J. Cognitive Function Remains Unchanged After Endarterectomy of Unilateral Internal Carotid Artery Stenosis Under Local Anaesthesia. Eur J Vasc Endovasc Surg. 2006;31:616–21.
Lal BK, Younes M, Cruz G, Kapadia I, Jamil Z, Pappas PJ. Cognitive changes after surgery vs stenting for carotid artery stenosis. J Vasc Surg. 2011;54:691–8.
Ngandu T, Lehtisalo J, Solomon A, Levälahti E, Ahtiluoto S, Antikainen R, et al. A 2 year multidomain intervention of diet, exercise, cognitive training, and vascular risk monitoring versus control to prevent cognitive decline in at-risk elderly people (FINGER): a randomised controlled trial. Lancet. 2015;385:2255–63.
Moll van Charante EP, Richard E, Eurelings LS, van Dalen J-W, Ligthart SA, van Bussel EF, et al. Effectiveness of a 6-year multidomain vascular care intervention to prevent dementia (preDIVA): a cluster-randomised controlled trial. Lancet. 2016;388:797–805.
Andrieu S, Guyonnet S, Coley N, Cantet C, Bonnefoy M, Bordes S, et al. Effect of long-term omega 3 polyunsaturated fatty acid supplementation with or without multidomain intervention on cognitive function in elderly adults with memory complaints (MAPT): a randomised, placebo-controlled trial. Lancet Neurol. 2017;16:377–89.
Brodaty H, Chau T, Heffernan M, Ginige JA, Andrews G, Millard M, et al. An online multidomain lifestyle intervention to prevent cognitive decline in at-risk older adults: a randomized controlled trial. Nat Med. 2025;31:565–73.
Singh B, Bennett H, Miatke A, Dumuid D, Curtis R, Ferguson T, et al. Effectiveness of exercise for improving cognition, memory and executive function: a systematic umbrella review and meta-meta-analysis. Br J Sports Med. 2025;59:866 LP–876.
Leijenaar JF, Groeneveld GJ, Klaassen ES, Leeuwis AE, Scheltens P, Weinstein HC, et al. Methylphenidate and galantamine in patients with vascular cognitive impairment-the proof-of-principle study STREAM-VCI. Alzheimers Res Ther. 2020;12:10.
Xu J, Qi Q, Lv P, Dong Y, Jiang X, Liu Z. Oxiracetam ameliorates cognitive deficits in vascular dementia rats by regulating the expression of neuronal apoptosis/autophagy-related genes associated with the activation of the Akt/mTOR signaling pathway. Brazilian J Med Biol Res Rev Bras Pesqui Medicas e Biol. 2019;52:e8371.
Wang B, Zhong L, Qiao P, Ma Z. Clinical efficacy and safety of nicergoline combined with oxiracetam in the treatment of vascular cognitive impairment. Pak J Pharm Sci. 2020;33:417–22.
Lim J-S, Lee J, Kang Y, Park H-T, Kim D-E, Cha J-K, et al. Efficacy and safety of oxiracetam in patients with vascular cognitive impairment: A multicenter, randomized, double-blinded, placebo-controlled, phase IV clinical trial. Contemp Clin Trials. 2023;126:107108.
Barbera M, Lehtisalo J, Perera D, Aspö M, Cross M, De Jager Loots CA, et al. A multimodal precision-prevention approach combining lifestyle intervention with metformin repurposing to prevent cognitive impairment and disability: the MET-FINGER randomised controlled trial protocol. Alzheimers Res Ther. 2024;16:23.
Reichl F-X, Högg C, Liu F, Schwarz M, Teupser D, Hickel R, et al. Actovegin® reduces PMA-induced inflammation on human cells. Eur J Appl Physiol. 2020;120:1671–80.
Guekht A, Skoog I, Edmundson S, Zakharov V, Korczyn AD. ARTEMIDA Trial (A Randomized Trial of Efficacy, 12 Months International Double-Blind Actovegin): A Randomized Controlled Trial to Assess the Efficacy of Actovegin in Poststroke Cognitive Impairment. Stroke. 2017;48:1262–70.
Dhulkifle H, Diab MI, Algonaiah M, Korashy HM, Maayah ZH. Apabetalone (RVX-208): A Potential Epigenetic Therapy for the Treatment of Cardiovascular, Renal, Neurological, Viral, and Cancer Disorders. ACS Pharmacol Transl Sci. 2024;7:546–59.
Kalantar-Zadeh K, Schwartz GG, Nicholls SJ, Buhr KA, Ginsberg HN, Johansson JO, et al. Effect of Apabetalone on Cardiovascular Events in Diabetes, CKD, and Recent Acute Coronary Syndrome: Results from the BETonMACE Randomized Controlled Trial. Clin J Am Soc Nephrol. 2021;16:705–16.
Bath PM, Mhlanga I, Woodhouse LJ, Doubal F, Oatey K, Montgomery AA, et al. Cilostazol and isosorbide mononitrate for the prevention of progression of cerebral small vessel disease: baseline data and statistical analysis plan for the Lacunar Intervention Trial-2 (LACI-2) (ISRCTN14911850). Stroke Vasc Neurol. 2023;8:134–43.
Pauls MMH, Binnie LR, Benjamin P, Betteridge S, Clarke B, Dhillon M-PK, et al. The PASTIS trial: Testing tadalafil for possible use in vascular cognitive impairment. Alzheimers Dement. 2022;18:2393–402.
Webb AJS, Birks JS, Feakins KA, Lawson A, Dawson J, Rothman AMK, et al. Cerebrovascular Effects of Sildenafil in Small Vessel Disease: The OxHARP Trial. Circ Res. 2024;135:320–31.
Sheng M, Lu H, Liu P, Li Y, Ravi H, Peng S-L, et al. Sildenafil Improves Vascular and Metabolic Function in Patients with Alzheimer’s Disease. J Alzheimers Dis. 2017;60:1351–64.
Washida K, Saito S, Tanaka T, Nakaoku Y, Ishiyama H, Abe S, et al. A multicenter, single-arm, phase II clinical trial of adrenomedullin in patients with cerebral autosomal dominant arteriopathy with subcortical infarcts and leukoencephalopathy. Cereb Circ Cogn Behav. 2024;6:100211.
Javed M, Saleem A, Xaveria A, Akhtar MF. Daphnetin: A bioactive natural coumarin with diverse therapeutic potentials. Front Pharmacol. 2022;13:993562.
Li S, Xiao Z. Recent Research Progress on the Use of Transcranial Magnetic Stimulation in the Treatment of Vascular Cognitive Impairment. Neuropsychiatr Dis Treat. 2024;20:1235–46.
Lin H, Li D, Zhu J, Liu S, Li J, Yu T, et al. Transcranial photobiomodulation for brain diseases: review of animal and human studies including mechanisms and emerging trends. Neurophotonics. 2024;11:10601.
Spires-Jones TL. How do we prevent scientific fraud? Brain Commun. 2022;4:fcac217.
Blanco K, Salcidua S, Orellana P, Sauma-Pérez T, León T, Steinmetz LCL, et al. Systematic review: fluid biomarkers and machine learning methods to improve the diagnosis from mild cognitive impairment to Alzheimer’s disease. Alzheimers Res Ther. 2023;15:176.
Wilcock D, Jicha G, Blacker D, Albert MS, D’Orazio LM, Elahi FM, et al. MarkVCID cerebral small vessel consortium: I. Enrollment, clinical, fluid protocols. Alzheimers Dement. 2021;17:704–15.
Xue C, Kowshik SS, Lteif D, Puducheri S, Jasodanand VH, Zhou OT, et al. AI-based differential diagnosis of dementia etiologies on multimodal data. Nat Med. 2024;30:2977–89.
Du J, Koch FC, Xia A, Jiang J, Crawford JD, Lam BCP, et al. Difference in distribution functions: A new diffusion weighted imaging metric for estimating white matter integrity. Neuroimage. 2021;240:118381.
Luckey AM, Ghosh S, Wang C-P, Beiser A, Bernal R, Li Z, et al. Biological validation of peak-width of skeletonized mean diffusivity as a VCID biomarker: The MarkVCID Consortium. Alzheimer’s Dement. 2024;20:8814–24.
Edsjö A, Holmquist L, Geoerger B, Nowak F, Gomon G, Alix-Panabières C, et al. Precision cancer medicine: Concepts, current practice, and future developments. J Intern Med. 2023;294:455–81.
Baskys A, Hou AC. Vascular dementia: pharmacological treatment approaches and perspectives. Clin Interv Aging. 2007;2:327–35.
Su WM, Gu XJ, Dou M, Duan QQ, Jiang Z, Yin KF, et al. Systematic druggable genome-wide Mendelian randomisation identifies therapeutic targets for Alzheimer’s disease. J Neurol Neurosurg Psychiatry. 2023;94:954–61.
Lopalco L. CCR5: From Natural Resistance to a New Anti-HIV Strategy. Viruses. 2010;2:574–600.
Fongang B, Weinstein G, Guðjónsson A, Mishra A, Bis JC, Yang Q, et al. A meta-analysis of genome-wide association studies identifies new genetic loci associated with all-cause and vascular dementia. Alzheimer’s Dement. 2021;17:e056081.
Freshour SL, Kiwala S, Cotto KC, Coffman AC, McMichael JF, Song JJ, et al. Integration of the Drug-Gene Interaction Database (DGIdb 4.0) with open crowdsource efforts. Nucleic Acids Res. 2021;49:D1144–D1151.
Nelson TJ, Sun M-K, Lim C, Sen A, Khan T, Chirila FV, et al. Bryostatin Effects on Cognitive Function and PKCɛ in Alzheimer’s Disease Phase IIa and Expanded Access Trials. J Alzheimers Dis. 2017;58:521–35.
Markus HS, de Leeuw FE. Cerebral small vessel disease: Recent advances and future directions. Int J Stroke. 2022;18:4–14.
Funding
Open Access funding enabled and organized by CAUL and its Member Institutions.
Author information
Authors and Affiliations
Contributions
MJL was responsible for the initial conceptualization of the paper, development of the figures and tables, initial drafting of the paper, and reviewing and editing the drafts of the paper. The PSS was responsible for the initial conceptualization of the paper, providing direction for the work, and reviewing and editing multiple rounds of drafts.
Corresponding authors
Ethics declarations
Competing interests
PSS served on the Advisory Committees for Biogen (2020 and 2021), Roche Australia (2022), Eli Lilly (2025), and Novo Nordisk (2025). MJL has nothing to disclose.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Lennon, M.J., Sachdev, P.S. Vascular cognitive impairment and dementia: Prevention, treatments, mechanisms and management options for the future. Neuropsychopharmacol. (2026). https://doi.org/10.1038/s41386-026-02331-3
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41386-026-02331-3
This article is cited by
-
Neuropsychiatric illness in the later years of life: summary and synthesis
Neuropsychopharmacology (2026)