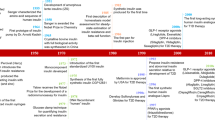
Abstract
Diabetes mellitus (DM) is a chronic metabolic disorder associated with a range of serious complications, including insulin resistance (IR) and cognitive impairments. IR is recognized as a high-risk factor for the development of cognitive decline. However, it remains unclear which endogenous or exogenous factors induce these two pathological features. Recent studies indicate that certain diets, environmental pollutants, and genetic deficiencies or polymorphisms influence the metabolism of endogenous formaldehyde. Formaldehyde has been implicated in the onset of DM. Notably, elevated levels of formaldehyde have been detected in the blood, peripheral organs (such as the spleen and liver), and brains of both DM patients and animal models of diabetes. Formaldehyde-induced hyperglycemia and high glucose levels generate formaldehyde, creating a vicious cycle that speeds up diabetic complications. Further, excessive formaldehyde induces IR through three distinct mechanisms: alteration of the biologically active conformation of insulin, reduction in insulin receptor expression, and modification of insulin receptor structure. In addition, excessive formaldehyde can impair cognitive functions by inhibiting N-methyl-D-aspartate (NMDA) receptors and exacerbating brain IR. In this review, we discuss the pivotal role of diet-derived formaldehyde in diabetes and propose that reduction of formaldehyde by the formaldehyde scavengers may be a new strategy for treating DM patients.

Similar content being viewed by others
Introduction
Foods can increase the risk of diabetes
Numerous studies have demonstrated a positive association between the consumption of ultra-processed foods (such as refined grains, processed meats, artificial and sugary beverages) and the risk of type 2 diabetes [1]. This association is attributed to the high content of sugar, starch, sodium, partially hydrogenated oils, and low content of dietary fiber in food, as well as various food additives and chemicals released during manufacturing and packaging [2].
Certain natural food groups, including red meat, potatoes, eggs, and fructose, have also been associated with an increased risk of type 2 diabetes [3]. Red meat, rich in haem iron, may promote oxidative stress and inflammation, leading to impaired insulin signaling and β-cell dysfunction [4]. Potatoes and related products are characterized by a high glycemic load and may contain trans fats, contributing to glucose dysregulation [5]. Egg consumption may increase the risk of type 2 diabetes through dietary cholesterol–induced β-cell dysfunction and impaired insulin secretion. It may also promote chronic low-grade inflammation, contributing to insulin resistance [6]. High doses of rapidly digested fructose may promote hepatic insulin resistance and contribute to the development of type 2 diabetes, primarily through increased de novo lipogenesis and visceral fat accumulation [3].
Multiple healthy dietary patterns (including the Mediterranean diet, vegetarian diets, Nordic diet, and DASH diet) have been associated with a reduced risk of type 2 diabetes [7]. The Mediterranean diet emphasizes minimally processed whole grains and legumes as dietary staples, along with the daily consumption of abundant and diverse fresh fruits and vegetables. Intake of fish, dairy products, red meat, and processed meats is relatively limited. Although the precise mechanisms remain unclear, the traditional Mediterranean diet may confer protection against metabolic disorders through multiple pathways, including lipid lowering, anti-inflammatory and antioxidant effects, modulation of cancer-related hormones, inhibition of nutrient-sensing pathways through amino acid restriction, and gut microbiota-derived metabolic regulation [8]. The ketogenic diet, characterized by low carbohydrate intake, has shown significant beneficial effects in both the prevention and management of type 2 diabetes, effectively improving HbA1c levels and glycemic control in patients [9]. Although high protein intake can reduce appetite and improve diabetic symptoms, it may concurrently decrease insulin sensitivity and delay the occurrence and development of diabetes [10, 11].
Food and formaldehyde metabolism
Formaldehyde levels in food
Formaldehyde is widely found in air, water, and natural foods. According to the statistics of the World Health Organization, the formaldehyde content is 3.3–60 mg/kg in fruits and vegetables, 5.7–20 mg/kg in meat, 1–3.3 mg/kg in milk and 1–98 mg/kg in fish. It is estimated that an average adult consumes about 1.5–14 mg of formaldehyde per day through food [1].
Accidental contamination of food with formaldehyde can occur through cooking utensils and food additives. Melamine-formaldehyde resins used in food-contact materials with a high formaldehyde-to-melamine (F:M) ratio tend to contain more ether bridges, which are more susceptible to acid- or heat-induced hydrolysis and release twice the amount of formaldehyde compared to methylene bridges [12]. Formaldehyde has been widely used as an additive in food due to its antimicrobial and antioxidant properties. Fish products often contain higher levels of formaldehyde, partly due to the natural demethylation of trimethylamine N-oxide (TMAO) into dimethylamine and formaldehyde, and partly due to the illegal addition of formaldehyde to seafood for infection control and shelf-life extension [1].
Formaldehyde-generating foods increase diabetes risk
Formaldehyde can be generated endogenously from various dietary sources. For instance, red meat contains around 300–500 milligrams of creatine per 100 grams. Creatine transporter-1 (CrT1) and creatine kinase (CK) mainly transfer creatine into the mitochondria and form sarcosine (a formaldehyde precursor) [13]. Creatine supplementation has been shown to transiently elevate hippocampal formaldehyde levels [14, 15]. However, excess formaldehyde (≥450 μM) can act as a cross-linker between cysteine 79 (C79) in the NR1 subunit and lysine 79 (K79) in the NR2B subunit, leading to conformational changes of the NMDAR, suppression of its activity, and subsequent impairment of synaptic transmission and memory formation in mice and rats [14]. Consistently, polymorphisms or knockout of CrT1 and CK lead to cognitive impairments in mice or humans [16,17,18,19].
In addition to its neurotoxic effects, formaldehyde may also contribute to metabolic disorders, including diabetes. Foods rich in carnitine and choline, mainly red meat and seafood [1] (Fig. 1A), can elevate TMAO levels in the body which promotes atherosclerosis and metabolic disorders [20]. TMAO promotes atherosclerosis by upregulating macrophage scavenger receptors such as CD36 and SR-A1, enhancing foam cell formation, and impairing cholesterol metabolism and bile acid synthesis. Its production depends on gut microbiota-mediated conversion of dietary precursors (e.g., choline), followed by hepatic FMO3-driven oxidation, linking microbial metabolism to systemic metabolic disorders [21]. It has been shown that TMAO (demethylated to produce formaldehyde) induces β-cell dysfunction and type 2 diabetes in mice via NLRP3 inflammasome activation and calcium transients [22]. Fructose metabolism is another endogenous source of formaldehyde. In vitro incubation studies have shown that fructose undergoes degradation to form a range of reactive carbonyl compounds, including formaldehyde, with total degradation product yields being 4.6- 271.6-fold higher than those from glucose, potentially contributing to its stronger metabolic reactivity and association with insulin resistance [2].
Figure 1A was reproduced with the permission of ref. [45]. ADH1 alcohol dehydrogenase 1, ADH3 alcohol dehydrogenase 3, ALDH2 aldehyde dehydrogenase 2, CAT catalase, ER endoplasmic reticulum, FA formaldehyde, HCHO formaldehyde, LSD1 lysine special demethylase 1, MeOH methanol, MMA, monomethylamine, MIT mitochondria, SA sarcosine, SARDH sarcosine dehydrogenase, SSAO semicarbazide-sensitive amine oxidase, TET1 TET methylcytosine dioxygenase 1, VOCs volatile organic compounds, TMAOase trimethylamine-N-oxide demethylase.
Exogenous sources such as additives and preservatives in processed and preserved foods may contain formaldehyde or induce formaldehyde production. It can be directly added during food production, such as its use as an antimicrobial agent in cheese manufacturing. In addition, the preservative hexamethylenetetramine may decompose under acidic conditions to release formaldehyde, while microbial fermentation processes can also lead to its natural formation. Furthermore, formaldehyde can react with food proteins to form stable or reversible derivatives, thereby contributing to its residual accumulation in processed foods [23]. Processed meat is one of the major dietary contributors to metabolic diseases among 25–64-year-olds in the US [24].
Formaldehyde-scavenging foods reduce diabetes risk
Pumpkin reduces blood sugar and blood cholesterol levels. Pumpkin seed oil extract can effectively inhibit formaldehyde-induced lipid peroxidation and oxidative stress as well as organ damage [25]. Alkaloid trigonelline extracted from fenugreek seeds can reduce hyperglycemia in diabetic rats and significantly alleviate formaldehyde-induced inflammatory response [26]. Trigonelline significantly reduces TMAO production by inhibiting the gut microbiota–mediated microbial conversion of choline to TMA [27]. Some natural compounds (tea polyphenolic extracts and phytic acid) and sodium citrate can inhibit TMAO demethylase in fish products. The inhibitory mechanism of these compounds is believed to be associated with their iron-chelating ability, as ferrous ions (Fe2+) are essential cofactors for the catalytic activity of TMAO demethylase [28].
Dangers of diabetes
Diabetes is a chronic metabolic disease that causes many serious complications, and the global incidence of the disease is increasing every year. According to the International Diabetes Federation, there were 537 million people with diabetes worldwide in 2021, which will increase to 643 million by 2030 [29]. Type 2 diabetes mellitus (T2DM) is the most common type of diabetes, accounting for more than 90% of all diabetes worldwide. As obesity rates rise globally, the incidence of insulin resistance (IR) is also accelerating and developing into a non-communicable epidemic. Cognitive impairment is one of the major complications of diabetes, including mild cognitive impairment (MCI) and dementia. As of 2022, the global prevalence of MCI is over 15% and is influenced by age, gender, literacy and region [30]. According to the study, the number of dementia patients worldwide will increase from 57.4 million in 2019 to 152.8 million in 2050 [31]. Type 1 diabetes mellitus is characterized by mild to moderate cognitive deficits, mainly in terms of impaired attention and memory. Type 2 diabetes is strongly associated with cognitive deficits, particularly affecting language, learning and memory. A recent Meta-analysis estimated the global prevalence of MCI in T2DM patients to be 45.0%, and T2DM patients in Asia are more likely to have cognitive impairment than those in Europe [32]. Additionally, people with diabetes also have a higher risk of dementia than non-diabetic individuals [33].
IR, defined as a state of reduced responsiveness of insulin-targeted tissues to physiologic levels of insulin, is a key causative factor in type 2 diabetes and many other metabolic diseases. In skeletal muscle, insulin promotes glucose utilization and storage by mediating glucose transport and net glycogen synthesis. In liver and adipose tissue, insulin signaling plays a key role in glucose utilization as insulin promotes glycogen synthesis, increases adipogenic gene expression, decreases gluconeogenesis in the liver, inhibits lipolysis in white adipose tissue and increases glucose transport and adipogenesis [34]. It is generally recognized that IR is associated with both type 2 and I diabetes [35]. In fact, the relationship between IR and diabetes is still not well defined, but the two can contribute to each other’s disease progression [36]. In addition, there are many pathophysiologic factors that can induce IR and worsen the condition, including insulin receptor defects [37,38,39], inflammation [40], adipocytokines [41], and endoplasmic reticulum stress [42]. However, the research conclusions that external and/or internal factor of diabetes are unclear or controversial. Here, the roles of environment-gene-diet derived formaldehyde in diabetes-related insulin resistance and cognitive impairments were discussed in this review.
Exogenous and endogenous formaldehyde affects insulin activity and function
Metabolism of formaldehyde in Life
Primordial gaseous formaldehyde (HCHO) was one of the earliest organically active compounds containing C, H and O elements to appear in the Earth’s early evolution [43]. It is believed to have created the organic compounds that gave rise to life, and to be the primary precursor of organic materials, including amino acids, RNA, DNA and proteins [44]. It is added illegally to food to extend its shelf life due to its antiseptic and preservation properties [45] (Fig. 1A). Gaseous formaldehyde pollution causes a variety of human diseases, such as: Alzheimer’s disease and brain cancer [46] (Fig. 1B).
Under physiological conditions, the level of endogenous formaldehyde in the body is maintained at a low concentration (0.01–0.08 mM in the blood); unexpectedly, it has been considered to be a gaseous neuromodulator to participate in memory formation in the brain [14]. Formaldehyde is produced mainly through demethylation of DNA, RNA and histones. Formaldehyde can also be produced by methanol oxidation. Endogenous methanol is usually produced by intestinal bacterial fermentation or by metabolism of S-adenosylmethionine [47] and then oxidatively deaminated to produce formaldehyde catalyzed by Semicarbazide-sensitive amine oxidase (SSAO, a formaldehyde-generating enzyme). SSAO is ubiquitously present in human body fluids, tissues and cells. SSAO activity is associated with diabetes mellitus. SSAO activity is significantly increased in diabetic patients, which can lead to increased production of toxic metabolites such as formaldehyde [48]. Aldehyde dehydrogenase 2 (ALDH2, a formaldehyde-degrading enzyme) is the main enzyme responsible for oxidizing formaldehyde [49]. ALDH2 deficiency due to mutations in the ALDH2 gene impairs formaldehyde metabolism, leading to formaldehyde accumulation in the body (Fig. 1C).
Acute injection of formaldehyde stimulates insulin secretion
The electrical activity of the cells, especially changes in intracellular calcium ion content, is critical for the release of insulin from them. Insulin is secreted by pancreatic β-cells in the islets of Langerhans in response to an increase in intracellular Ca2+ concentration ([Ca2+]i) [50]. Under normal physiological conditions, the pancreas ingests glucose through the glucose transporter 4 (GLUT4). Then glucose enters the cell through insulin receptor (I-R) and generates ATP. The increased ATP/ADP ratio induces the closure of the ATP-sensitive potassium channel (KATP). It stimulates the opening of the voltage-gated calcium channel (VGCC) and an increase in Ca2+ influx [51], and stimulates cellular cytosolization of insulin-containing vesicles for insulin secretion [52].
High concentrations of formaldehyde activate the endoplasmic reticulum and various ion channels, inducing an increase in intracellular Ca2+ concentration. It has been demonstrated in vitro that a high concentration of formaldehyde (40 mM) induced the release of calcium stored in the endoplasmic reticulum rather than mitochondria in mouse sensory neurons, primary keratinocytes, and non-neuronal cell lines, and inhibited calcium pumps from transporting intracellular Ca2+ to the endoplasmic reticulum [53]. Low concentrations of formaldehyde (<3 mM) also increase intracellular calcium concentration in primary cultured hippocampal neurons by promoting the expression of KATP channel subunits [54], stimulating NMDA receptors and stimulating the opening of T-type calcium channels [55] (Fig. 2).
Briefly, acute FA injection can induce Ca2+ influx. Insulin binding to its receptor allows glucose to enter cells through GLUT4, which is then converted to ATP in the mitochondria, increasing the ATP/ADP ratio. Inhibition of KATP channel activity leads to membrane depolarization, opening of VGCC channels, and an increase in the intracellular Ca2+ levels, ultimately stimulating insulin secretion. AKT protein kinase B, APS adaptor proteins, [Ca2+]i intracellular Ca2+ concentration, CAP c-CBL-associated protein, I-R insulin receptor, KATP ATP-sensitive potassium channel, GLUT4 glucose transporter type 4, IRS insulin receptor substrate, mTORC2 mammalian target of rapamycin Complex 2, PDK1 phosphoinositide-dependent kinase 1, PI3K phosphoinositol 3-kinase, PIP2 phosphatidylinositol-4,5-diphosphate, PIP3 phosphatidylinositol-3,4,5-trisphosphate, VGCC voltage-gated calcium channel.
Vicious cycle: high glucose-induced formaldehyde generation and formaldehyde-elicited hyperglycemia
Injection of formaldehyde induces an elevation in blood glucose
Existing studies have found that injection of formaldehyde at high levels (Formalin) indeed induces a sharp rise in blood glucose (hyperglycemia) in the Carp [56], salmon (Salmo salar) and rainbow trout (Salmo gairdneri) [57], and mice [58]. The increase in blood glucose observed in fish exposed to formalin may be attributed to acute stress responses, including enhanced anaerobic metabolism, impaired oxygen uptake due to gill damage and mucus secretion, and metabolic acidosis, all of which stimulate glycogenolysis and gluconeogenesis [56, 57]. Formaldehyde may also induce hyperglycemia by forming FA-insulin adducts, which impair insulin signaling and reduce glucose uptake, as shown in diabetic mice models with ALDH2 deficiency [58]. Unsurprisingly, high levels of blood formaldehyde were found in the liver and blood in the diabetic model rats [59, 60], and in the blood of T2DM patients [58] (Fig. 3A–C). However, injection of insulin can reduce formalin-induced elevation in blood glucose and pain-like behaviors [61]. These findings indicate that excessive formaldehyde can elicit hyperglycemia.
Figure 3A was reproduced with the permission of ref. [59]. Figure 3B was reproduced with the permission of ref. [60]. Figure 3C, D were reproduced with the permission of ref. [58]. A FA concentration in the liver and spleen of diabetic mice significantly increases and NaHSO3 treatment reduces the FA concentration in the liver and spleen of diabetic mice. B The concentration of FA in the hippocampus of diabetic mice was higher than that of normal mice. C Blood FA levels elevated in T2DM patients than control. D FA aggregates with insulin and leads to insulin dysfunction. E FA binding to specific insulin amino acid residue. F Model of high glucose-induced FA generation via SARDH in the mitochondria and FA-stimulated blood glucose elevation. FA formaldehyde, SARDH sarcosine dehydrogenase, T2DM type-2 diabetic mellitus.
Formaldehyde inactivates Insulin by binding with Arg, Tyr and Lys residues
It is known that TyrA19 of insulin interacts directly with the insulin receptor, and TyrB16 and TyrB26 are amino acid residues important for the binding of insulin to its receptor [62]. Formaldehyde causes a variety of chemical modifications to proteins, such as hydroxymethyl, Schiff base and methylene bridge. Formaldehyde also binds to specific amino acid residues on insulin. Preliminary experiments have concluded that incubating insulin with formaldehyde can cause a decrease in its affinity for the insulin receptor, which in turn triggers an increase in blood glucose levels [58] (Fig. 3D). It is thus conjectured that formaldehyde can react with insulin to change its structure and thus affect its biological activity. When the model peptide was treated with formaldehyde, the amino groups of the N-terminal amino acid residues in the peptide, as well as arginine, cysteine, histidine, and lysine residues reacted [63]. The strong reactivity of formaldehyde with arginine (Arg), tyrosine (Tyr) and lysine (Lys) residues in the insulin protein molecule has been directly supported by experiments [64] (Fig. 3E). Thus, formaldehyde-inactivated insulin contributes to hyperglycemia, because blood glucose cannot be used efficiently.
Application of high glucose induces formaldehyde generation
Surprisingly, the results of in vitro experiment has found that using DM-FA (a newly developed two-photon formaldehyde fluorescent probe), high-glucose stimulates formaldehyde generation in the diabetic cell model [59]. Application of inhibitor of SSAO (a formaldehyde generating enzyme) can alleviate hyperglycemia in diabetic model rats [65]. Previous study has revealed that Ca2+ influx promotes formaldehyde generation in the mitochondria via SARDH [14]. High glucose can elicit extracellular Ca2+ influx via voltage-dependent Ca2+ channel (VDCC) [66,67,68]. Hence, hyperglycemia most likely elicits formaldehyde production and accumulation by activating Ca2+-dependent SARDH; Formaldehyde can stimulate glucogen to form glucose and blood glucose elevation [69,70,71]. This vicious cycle between high glucose-induced formaldehyde generation and formaldehyde-stimulated hyperglycemia contributes to T2DM onset (Fig. 3F). This also gives a possible explanation why only reducing blood sugar alone does not improve diabetes, because the age-associated formaldehyde still exists and impairs memory [60, 72,73,74].
Multiple mechanisms of formaldehyde-induced IR
Formaldehyde has been less well studied in association with IR. Animal studies have shown that pathological concentrations of formaldehyde injection can induce hyperglycemia and cognitive deficits in healthy rats, whereas reduction of formaldehyde by overexpression of human ALDH2 can improve glycemic status and cognitive function in diabetic mice [58]. In population-based studies, a reliable correlation has been observed between increased levels of formaldehyde contained in the air at the workplace and increased risks of dyslipidemia and IR in perimenopausal women workers [75]. Serum formaldehyde concentrations were higher in diabetic patients than in nondiabetic patients [58]. Therefore, formaldehyde is likely involved in the pathogenesis of type 2 diabetes and that endogenous formaldehyde has the potential to be a biomarker for monitoring the early stages of diabetes [59]. Formaldehyde may also be associated with the development of diabetes-related cognitive impairment. Some studies have reported an association between increased expression of the formaldehyde-producing enzymes SSAO as well as sarcosine dehydrogenase (SARDH) and type 2 diabetes mellitus with its complications [76]. One possible mechanism is that increased SSAO activity protects pancreatic islets from the inhibitory effects of methylamine, but produces harmful substances such as formaldehyde and hydrogen peroxide [77].
It has been found that formaldehyde can change insulin structure and reduce its activity when compared with insulin without formaldehyde treatment [58, 63, 64] (Figs. 4A, B). Formaldehyde is a highly reactive compound. It can covalently bind to DNA and proteins [78], inducing adducts formation and cross-linking via Lys, Tyr and Arg residues [64]. Moreover, insulin receptor contains a lot of these amino acid residues [62]. This is a reasonable speculation that formaldehyde can change the structure of I-R (Fig. 4C). Exposure to formaldehyde or to cigarette smoke containing formaldehyde has been linked to the downregulation of genes involved in neural signaling and metabolic regulation [79,80,81,82]. Specifically, formaldehyde has been demonstrated to suppress the expression of synaptic plasticity-related genes in the hippocampus [81], while cigarette smoke exposure, which includes formaldehyde among its toxic constituents, has been reported to impair insulin signaling and reduce insulin receptor (I-R) expression in peripheral tissues [82]. These findings collectively suggest that excessive formaldehyde may contribute to the downregulation of I-R expression (Fig. 4D), potentially through mechanisms involving oxidative stress and epigenetic modifications. Nevertheless, the existence of a direct causal relationship between formaldehyde exposure and reduced I-R expression requires further elucidation. The above studies suggest that endogenous formaldehyde accumulation may be a key factor in the development of IR in diabetes mellitus.
A Under normal conditions, insulin stimulates the movement of GLUT4 from small vesicles to the cell membrane to increase glucose uptake. B Insulin structural changes: chronic FA changes part of the insulin structure, which cannot bind to the insulin receptor, inducing IR. C Chronic FA changes the structure of the insulin receptor, preventing insulin from binding and reducing insulin entry into the cytosol and insulin receptor. D Reduced number of insulin receptors: FA can reduce the number of I-R, leading to a reduction in insulin entering the cell and inducing IR. GLUT4 glucose transporter type 4, I-R insulin receptor, IR insulin resistance.
Molecular mechanisms of formaldehyde-induced T2DM
Formaldehyde and insulin receptor deficiency
Insulin receptor defects may occur due to derangements on three levels: pre-receptor defects, receptor defects and post-receptor defects.
Pre-receptor defects include abnormalities in the insulin molecule itself, the presence of insulin autoantibodies, and abnormal insulin degradation. One example is Insulin Autoimmune Syndrome (IAS), in which spontaneous hypoglycemia occurs due to the presence of insulin autoantibodies triggered by exogenous insulin [83]. Insulin metabolism occurs through the action of the insulin-degrading enzyme (IDE). Clinical studies have shown that elevated levels of glucose and insulin in the body reduce the activity of IDE, which regulates the metabolic rate and sensitivity of insulin [84]. Patients with type 2 diabetes have higher serum IDE levels than healthy individuals [85].
Receptor defects are categorized into structural and functional defects of the receptor. Insulin function is abnormal due to impaired binding of insulin to the receptor is called type A IR syndrome (TAIR) [38]. These factors affects the insulin sensitivity of target tissues [39], thus causing IR.
Post-receptor defects lead to impairment of downstream signal transduction, thereby reducing insulin-mediated cellular glucose uptake [86]. Formaldehyde can bind directly to insulin and reduce its functions [58], it also can bind to the IGF-1 receptor, thereby affecting insulin signaling [87].
Formaldehyde and inflammation
Macrophages infiltrating adipose tissue interfere with the insulin signaling pathway by secreting inflammatory factors. The number of macrophages in adipose tissue is positively correlated with the concentration of several inflammatory cytokines [40]. For example, tumor necrosis factor (TNF-α) mediates IR induced by obesity [88]. It inhibits tyrosine phosphorylation on the insulin receptor and insulin receptor substrate-1β chain [89]. IL-6 plays a decisive role in the induction of IR and type 2 diabetes mellitus by impairing the insulin receptor and inhibiting the phosphorylation of insulin receptor substrate-1 [90]. Formaldehyde can enhance proinflammatory responses in vivo by inducing glycolysis, with increased proinflammatory cytokine release and NO secretion. Specifically, FA activates the PI3K/HIF-1α signaling pathway in a dose-dependent manner, leading to the upregulation of PIK3CA and HIF-1α. This, in turn, increases the expression of downstream glycolytic enzymes such as LDHA and iNOS, which mediate the production of lactate and nitric oxide, respectively [91]. Formaldehyde exposure promotes the secretion of inflammatory cytokines, as well as redox imbalance and oxidative damage [92]. Notably, the protective role of aldehyde dehydrogenase 2 (ALDH2), a key enzyme in FA detoxification, has also been highlighted. In ALDH2-deficient mice, exposure to exogenous formaldehyde triggered exaggerated macrophage glycolysis and proinflammatory responses, suggesting that impaired FA clearance can further amplify inflammation and metabolic dysfunction [93].
Formaldehyde and adipokines
Adipokines, including leptin, adiponectin and lipocalin, are key regulators of insulin sensitivity in major insulin-target organs such as the liver and skeletal muscle. Leptin acts on a functional receptor called OB-Rb which located in the hypothalamus and suppresses appetite [94]. Adiponectin enhances insulin sensitivity. Lipocalin reduces inflammatory cytokines and oxidative stress, thereby improving IR [95]. Reduced adipokine secretion can consequently lead to systemic insulin resistance [41]. Experimental studies have shown that formaldehyde can downregulate the expression of leptin and its receptor in HT22 cells, supporting the notion that impaired leptin signaling contributes to neurodegenerative diseases and highlighting the potential role of formaldehyde in their pathogenesis [96, 97]. Formaldehyde exposure can also disrupt adipokine homeostasis by enhancing the immunoreactivity of two adipokines, asprosin and subfatin, in mouse liver [98].
Formaldehyde and endoplasmic reticulum stress
Endoplasmic reticulum stress is a stress initiated by an imbalance in calcium homeostasis. It can induce IR by interfering insulin signaling pathways [42]. IRE1 phosphorylates JNK, which then results in impaired insulin signaling through inhibition of IRS1 and IkB kinase, leading to NF-κB activation and inflammatory responses [99]. The prolonged activation of NMDA receptors in the body leads to endoplasmic reticulum stress and thus decreases insulin secretion from pancreatic β-cells [100]. Studies have shown that formaldehyde exposure can induce Tau phosphorylation in mice and upregulate the expression of ERS-associated proteins, such as GRP78 and CHOP. These changes thereby induce injury to HT22 cells and cognitive impairment in C57/BL6 mice [101].
Formaldehyde and oxidative stress
Oxidative Stress exerts its effects primarily through reactive oxygen species (ROS) and other reactive substances, including superoxide anion radicals, hydrogen peroxide (H2O2) and hydroxyl radicals. Insulin stimulates adipocytes to produce endogenous H2O2 [102], which is involved in the redox initiation step during insulin receptor activation. Hyperglycemia can lead to an increase in ROS production [103], which can inversely affect insulin, leading to decreased insulin secretion and sensitivity [104]. Hence, it induces IR [105]. In formaldehyde-exposed mice, gene expression of thioredoxin (TRX), an antioxidant protein that regulates redox, was markedly inhibited and thus intracellular ROS levels increased with increasing formaldehyde concentration [106]. Levels of OS biomarkers (15-F2t-IsoP and 8-oxo-dGuo) in occupationally formaldehyde-exposed workers were positively correlated with formaldehyde concentrations [107].
Formaldehyde and mitochondrial dysfunction
Mitochondrial dysfunction plays an important role in lipid metabolism and cellular energy supply. For example, mice knocked out of long-chain Acyl-CoA dehydrogenase exhibit hepatic steatosis and hepatic IR [108]. Knocked out of N-acetyltransferase 1 reduces rates of mitochondrial dysfunction and insulin sensitivity in vivo [109]. The dynamic equilibrium process of mitochondrial fusion and fission is also related to the development of IR [99]. High-fat diets causing IR in skeletal muscle have also been linked to mitochondrial dysfunction, reflected by increased H2O2 release [110] and impaired ADP sensitivity [111]. Formaldehyde induces mitochondrial dysfunction and is highly cytotoxic to a certain extent [112]. This was demonstrated by formaldehyde’s ability to dose-dependently reduce the mitochondrial membrane potential (MMP) of SK-N-SH neuroblastoma cells and inhibit the mitochondrial respiratory enzymes therein, i.e., NADH dehydrogenase (complex I), cytochrome c oxidase (complex IV), and oxidative stress-sensitive aconitase [112, 113]. Elevated intracellular levels of formaldehyde can induce DNA double-strand breaks in the mitochondria [114].
Formaldehyde and obesity
Central obesity is primarily associated with chronic physical inactivity and excessive dietary energy intake, which is often associated with IR [115]. Lipo-toxicity occurs when adipose tissue accumulates in non-fat organs (e.g., liver, pancreas, and muscle tissue), dysfunctionalizing organelles, releasing excess ROS, leading to systemic inflammation and IR [116]. For example, 5 days of a high-fat, high-calorie diet reduces insulin-stimulated glucose disposal in South Asians, but does not reduce blood glucose consumption in Caucasians [117]. Animal experiments revealed a significant increase of SSAO activity in the kidneys and aorta of C57BL/6 mice fed with high cholesterol and fat, as well as a marked increase of methylamine, formic acid and formaldehyde in the urine [118, 119], which may promote IR. Formaldehyde in the air is associated with an increases the risk of overweight, abdominal obesity, and high body fat percentage [120]. Additional studies have shown that selective SSAO inhibitors can lead to reduce weight gain in the KKAy mouse model [121].
Formaldehyde and drug metabolism
Insulin antagonist hormones can cause IR in the body. Hyperglycemia and IR occurs as side effects of various hormonal drugs such as glucocorticoid (GC), growth hormone (GH), thyroid hormone (TH), and adrenaline, epinephrine (A/D). For example, Chronic excess GC affects adipose tissue to secrete lipocalin and leptin, and also leads to impaired fat synthesis and metabolism [122]. GH can promote the catabolism of triglycerides to free fatty acids, increase lipo-toxicity to non-adipose tissues in the body, and lead to IR [123]. Although the role of TH in insulin signaling and glucose metabolism remains unclear, there is a recognized link between thyroid hormone and insulin signaling pathways, whereby dysfunction in one may lead to dysregulation of the other [124]. Formaldehyde may indirectly contribute to the development of insulin resistance induced by insulin antagonist hormones through mechanisms such as adipokine dysregulation and oxidative stress. However, the precise pathways remain to be explored, which may partially explain the adverse metabolic effects of these hormonal drugs.
Formaldehyde and air pollution
Air pollution and its components can induce IR. For example, PM2.5 exposure affects adipose tissue, muscle, liver and brain, inducing IR [125]. Increased levels of formaldehyde in the work environment have been reported abroad to predispose to dyslipidemia and IR [126]. In vitro, it has been found that formaldehyde readily reacts with arginine, tyrosine and lysine residues on bovine insulin [64]. Formaldehyde reacts with human insulin to reduce insulin activity, and insulin bound to formaldehyde performs worse in promoting cellular glucose uptake [58]. Hence, formaldehyde accumulation may be one of the risk factors for IR (Fig. 5A).
A Exogenous and endogenous factors involving FA trigger IR in DM patients. B Metabolic disorder-derived FA impairs memory in individuals with diabetes by suppressing N-methyl-D-aspartate receptors. Figure 5B was reproduced with the permission of ref. [14]. ALDH2 aldehyde dehydrogenase 2 family member, ATD amino-terminal domain C79 cysteine 79, FA formaldehyde, FFA free fatty acids, I-R insulin receptor, LRG1 leucine-rich-alpha-2-glycoprotein-1, K79 lysine 79, NMDAreceptor N-methyl-D-aspartate receptor, NR1 NMDA receptor subunit 1, NR2B NMDA receptor subunit 2, ROS reactive oxygen species, SARDH sarcosine dehydrogenase, SSAO semicarbazide-sensitive amine oxidase.
Formaldehyde impairs cognition by inhibiting NMDA-receptor
It is important to notice that formaldehyde plays a multifaceted role, and at certain concentrations it can enhance memory, promote cell proliferation and even prolong lifespan. According to cellular experiments, neuronal cells incubated with lower concentrations of formaldehyde (0.001–0.05 mM for BV2 cells; 0.01–0.05 mM for SH-SY5Y cells) for three days showed an increase in cell proliferation [44].
High concentrations of formaldehyde directly disrupt memory. Occupational formaldehyde exposure has been found to increase cognitive impairment in young and middle-aged and older adults (≥45 years) [127]. The specific mechanism is that formaldehyde induces cognitive dysfunction by interfering with NO/cGMP signaling pathway and direct oxidative damage to hippocampal cells [128]. Excessive formaldehyde can crosslink NR1 and NR2 subunits to close NMDA-receptor and lead memory decline (Fig. 5B). ALDH2 is an enzyme responsible for oxidizing excess formaldehyde. In ALDH2 knockout mice, formaldehyde accumulation due to ALDH2 hypofunction impairs mice memory, which is consistent with some of the mechanisms occurring in Alzheimer’s disease patients [14].
Abnormal accumulation of formaldehyde in the body exacerbates hyperglycemia and cognitive impairment. The study found that diabetic patients with elevated ALDH2 activity exhibited elevated formaldehyde concentrations in their bodies. Formaldehyde levels demonstrated a positive correlation with blood glucose levels and negative correlation with MMES scores, which is used to evaluate cognitive function. Animal studies demonstrated the injection of formaldehyde to healthy rats or inhibition of ALDH2 expression in mice could elevate blood glucose and formaldehyde concentration. These interventions prolonged the escape latency in the Morris water maze test. ALDH2 overexpression can reverse the cognitive impairment in T2DM mouse models and in mice injected with high glucose [58].
The underlying mechanism of formaldehyde-induced diabetes-related cognitive impairment may be attributed to excess formaldehyde induces to promote insulin aggregation. This will aid in formation of formaldehyde-insulin adducts, and thus decreasing insulin activity [58]. Persistent hyperglycemic stimulation leads to a significant increase in SSAO activity in patients [59], an increase in SSAO substrates (e.g., methylamine or aminopyruvic acetone) [129], and enhanced DNA demethylation. Collectively, these changes contribute to upregulation of formaldehyde production.
Concluding remarks
The number of people with diabetes is increasing worldwide and cognitive impairment is one of the serious complications of diabetes. Imaging studies have shown accelerated atrophy of the cerebral cortex and hippocampus in diabetic patients, and the key mechanism causing neuronal apoptosis and memory loss is the development of IR in the brain. Insulin signaling has a significant effect on cognitive function, and some studies have found that formaldehyde accumulation in diabetic patients is positively correlated with their blood glucose levels, and formaldehyde binds to and reduces the activity of human insulin, so endogenous formaldehyde accumulation may be an important factor in the development of cognitive deficits in diabetic patients.
However, there is still a lack of drugs targeting cognitive impairment in diabetes. Glucose-lowering drugs such as metformin, insulin, DPP-4 inhibitors, and GLP-1R agonists have improved cognitive function in diabetic patients and even in the general population, but the exact mechanism of action is still being explored. Interestingly, some of the hypoglycemic drugs can degrade formaldehyde. More studies have found that formaldehyde scavengers such as OMEGA-3 essential fatty acids, silymarin, and hydrogen sulfide can effectively improve cognition by lowering formaldehyde levels in the body, and metformin in glucose-lowering drugs is also able to bind with formaldehyde and reduce its toxicity, which suggests that formaldehyde scavenger may be a new direction for the treatment of IR and cognitive impairment of diabetic patients’ brains.
However, the clinical application of formaldehyde scavengers in the treatment of insulin resistance (IR) and cognitive impairment still faces several important challenges. First, most currently identified FA scavengers lack specificity for the central nervous system and show limited permeability across the blood-brain barrier, which may restrict their effectiveness in targeting brain-derived formaldehyde. Second, the long-term safety and systemic metabolic effects of FA scavenging agents remain unclear and require further investigation. Third, the precise molecular mechanisms by which formaldehyde contributes to insulin receptor dysfunction—particularly in the context of neuronal signaling and epigenetic regulation—are not yet fully understood. Future research should aim to develop brain-targeted FA scavengers, explore combination strategies with antidiabetic agents such as GLP-1R agonists or metformin, and establish reliable biomarkers to monitor formaldehyde burden and treatment response. Addressing these gaps will be critical for validating FA scavenging as a viable therapeutic approach for diabetic encephalopathy.
References
Mehta NK, Vaishnav A, Priyadarshini MB, Debbarma P, Hoque MS, Mondal P, et al. Formaldehyde contamination in seafood industry: an update on detection methods and legislations. Environ Sci Pollut Res Int. 2024;31:54381–401.
Ohno R, Auditore A, Gensberger-Reigl S, Saller J, Stutzer J, Weigel I, et al. Qualitative and Quantitative Profiling of Fructose Degradation Products Revealed the Formation of Thirteen Reactive Carbonyl Compounds and Higher Reactivity Compared to Glucose. J Agric Food Chem. 2024;72:19131–42.
Mozaffarian D. Dietary and Policy Priorities for Cardiovascular Disease, Diabetes, and Obesity: A Comprehensive Review. Circulation. 2016;133:187–225.
Rohrmann S, Linseisen J. Processed meat: the real villain?. Proc Nutr Soc. 2016;75:233–41.
Halton TL, Willett WC, Liu S, Manson JE, Stampfer MJ, Hu FB. Potato and french fry consumption and risk of type 2 diabetes in women. Am J Clin Nutr. 2006;83:284–90.
Shin JY, Xun P, Nakamura Y, He K. Egg consumption in relation to risk of cardiovascular disease and diabetes: a systematic review and meta-analysis. Am J Clin Nutr. 2013;98:146–59.
Papakonstantinou E, Oikonomou C, Nychas G, Dimitriadis GD. Effects of Diet, Lifestyle, Chrononutrition and Alternative Dietary Interventions on Postprandial Glycemia and Insulin Resistance. Nutrients. 2022;14:823.
Tosti V, Bertozzi B, Fontana L. Health Benefits of the Mediterranean Diet: Metabolic and Molecular Mechanisms. J Gerontol A Biol Sci Med Sci. 2018;73:318–26.
Dynka D, Kowalcze K, Ambrozkiewicz F, Paziewska A. Effect of the Ketogenic Diet on the Prophylaxis and Treatment of Diabetes Mellitus: A Review of the Meta-Analyses and Clinical Trials. Nutrients. 2023;15:500.
Lewgood J, Oliveira B, Korzepa M, Forbes SC, Little JP, Breen L, et al. Efficacy of Dietary and Supplementation Interventions for Individuals with Type 2 Diabetes. Nutrients. 2021;13:2378.
Brandhorst S, Longo VD. Protein Quantity and Source, Fasting-Mimicking Diets, and Longevity. Adv Nutr. 2019;10:S340–S50.
Ebner I, Haberer S, Sander S, Kappenstein O, Luch A, Bruhn T. Release of Melamine and Formaldehyde from Melamine-Formaldehyde Plastic Kitchenware. Molecules. 2020;25:3629.
Scott CR, Clark SH, Teng CC, Swedberg KR. Clinical and cellular studies of sarcosinemia. J Pediatr. 1970;77:805–11.
Ai L, Tan T, Tang Y, Yang J, Cui D, Wang R, et al. Endogenous formaldehyde is a memory-related molecule in mice and humans. Commun Biol. 2019;2:446.
Souza MA, Magni DV, Guerra GP, Oliveira MS, Furian AF, Pereira L, et al. Involvement of hippocampal CAMKII/CREB signaling in the spatial memory retention induced by creatine. Amino Acids. 2012;43:2491–503.
Aksenova MV, Burbaeva G. BB creatine kinase isoenzyme activity in the blood serum of patients with senile dementia, Alzheimer’s disease and schizophrenia. Zh Nevropatol Psikhiatr Im S S Korsakova. 1989;89:113–6.
Kurosawa Y, Degrauw TJ, Lindquist DM, Blanco VM, Pyne-Geithman GJ, Daikoku T, et al. Cyclocreatine treatment improves cognition in mice with creatine transporter deficiency. J Clin Invest. 2012;122:2837–46.
Streijger F, Oerlemans F, Ellenbroek BA, Jost CR, Wieringa B. Van der Zee CE. Structural and behavioural consequences of double deficiency for creatine kinases BCK and UbCKmit. Behav Brain Res. 2005;157:219–34.
Udobi KA-O, Delcimmuto N, Kokenge AN, Abdulla ZI, Perna MK, Skelton MA-O. Deletion of the creatine transporter gene in neonatal, but not adult, mice leads to cognitive deficits. J Inherit Metab Dis. 2019;42:966–74.
Koeth RA, Wang Z, Levison BS, Buffa JA, Org E, Sheehy BT, et al. Intestinal microbiota metabolism of L-carnitine, a nutrient in red meat, promotes atherosclerosis. Nat Med. 2013;19:576–85.
Wang Z, Klipfell E, Bennett BJ, Koeth R, Levison BS, Dugar B, et al. Gut flora metabolism of phosphatidylcholine promotes cardiovascular disease. Nature. 2011;472:57–63.
Kong L, Zhao Q, Jiang X, Hu J, Jiang Q, Sheng L, et al. Trimethylamine N-oxide impairs beta-cell function and glucose tolerance. Nat Commun. 2024;15:2526.
Restani P, Restelli AR, Galli CL. Formaldehyde and hexamethylenetetramine as food additives: chemical interactions and toxicology. Food Addit Contam. 1992;9:597–605.
Micha R, Penalvo JL, Cudhea F, Imamura F, Rehm CD, Mozaffarian D. Association Between Dietary Factors and Mortality From Heart Disease, Stroke, and Type 2 Diabetes in the United States. JAMA. 2017;317:912–24.
Paul M, Sohag MSU, Khan A, Barman RK, Wahed MII, Khan MRI. Pumpkin (Cucurbita maxima) seeds protect against formaldehyde-induced major organ damages. Heliyon. 2020;6:e04587.
Pundarikakshudu K, Shah DH, Panchal AH, Bhavsar GC. Anti-inflammatory activity of fenugreek (Trigonella foenum-graecum Linn) seed petroleum ether extract. Indian J Pharm. 2016;48:441–4.
Anwar S, Bhandari U, Panda BP, Dubey K, Khan W, Ahmad S. Trigonelline inhibits intestinal microbial metabolism of choline and its associated cardiovascular risk. J Pharm Biomed Anal. 2018;159:100–12.
Gomez-Estaca J, Gomez-Guillen MC, Marin-Penalver D, Montero MP. Functional aptitude of hake minces with added TMAO-demethylase inhibitors during frozen storage. Food Chem. 2020;309:125683.
Chou CY, Hsu DY, Chou CH. Predicting the Onset of Diabetes with Machine Learning Methods. J Pers Med. 2023;13:406.
Bai W, Chen P, Cai H, Zhang Q, Su Z, Cheung T, et al. Worldwide prevalence of mild cognitive impairment among community dwellers aged 50 years and older: a meta-analysis and systematic review of epidemiology studies. Age Ageing. 2022;51:afac173.
Collaborators GBDDF. Estimation of the global prevalence of dementia in 2019 and forecasted prevalence in 2050: an analysis for the Global Burden of Disease Study 2019. Lancet Public Health. 2022;7:e105–e25.
You Y, Liu Z, Chen Y, Xu Y, Qin J, Guo S, et al. The prevalence of mild cognitive impairment in type 2 diabetes mellitus patients: a systematic review and meta-analysis. Acta Diabetol. 2021;58:671–85.
Biessels GJ, Despa F. Cognitive decline and dementia in diabetes mellitus: mechanisms and clinical implications. Nat Rev Endocrinol. 2018;14:591–604.
Petersen MC, Shulman GI. Mechanisms of Insulin Action and Insulin Resistance. Physiol Rev. 2018;98:2133–223.
Laakso M, Kuusisto J. Insulin resistance and hyperglycaemia in cardiovascular disease development. Nat Rev Endocrinol. 2014;10:293–302.
Lee SH, Park SY, Choi CS. Insulin Resistance: From Mechanisms to Therapeutic Strategies. Diabetes Metab J. 2022;46:15–37.
Ogawa W, Araki E, Ishigaki Y, Hirota Y, Maegawa H, Yamauchi T, et al. New classification and diagnostic criteria for insulin resistance syndrome. Endocr J. 2022;69:107–13.
Pei J, Wang B, Wang D. Current Studies on Molecular Mechanisms of Insulin Resistance. J Diabetes Res. 2022;2022:1863429.
Singh S, Sharma R, Kumari M, Tiwari S. Insulin receptors in the kidneys in health and disease. World J Nephrol. 2019;8:11–22.
Kunz HE, Hart CR, Gries KJ, Parvizi M, Laurenti M, Dalla Man C, et al. Adipose tissue macrophage populations and inflammation are associated with systemic inflammation and insulin resistance in obesity. Am J Physiol Endocrinol Metab. 2021;321:E105–E21.
Keane KN, Cruzat VF, Carlessi R, de Bittencourt PI Jr, Newsholme P. Molecular Events Linking Oxidative Stress and Inflammation to Insulin Resistance and beta-Cell Dysfunction. Oxid Med Cell Longev. 2015;2015:181643.
Villalobos-Labra R, Subiabre M, Toledo F, Pardo F, Sobrevia L. Endoplasmic reticulum stress and development of insulin resistance in adipose, skeletal, liver, and foetoplacental tissue in diabesity. Mol Asp Med. 2019;66:49–61.
Pinto JP, Gladstone GR, Yung YL. Photochemical Production of Formaldehyde in Earth’s Primitive Atmosphere. Science. 1980;210:183–5.
Li T, Wei Y, Qu M, Mou L, Miao J, Xi M, et al. Formaldehyde and De/Methylation in Age-Related Cognitive Impairment. Genes (Basel). 2021;12:913.
Rahman MB, Hussain M, Kabiraz MP, Nordin N, Siddiqui SA, Bhowmik S, et al. An update on formaldehyde adulteration in food: sources, detection, mechanisms, and risk assessment. Food Chem. 2023;427:136761.
Yuan Y, Wu Y, Zhao H, Ren J, Su W, Kou Y, et al. Tropospheric formaldehyde levels infer ambient formaldehyde-induced brain diseases and global burden in China, 2013-2019. Sci Total Environ. 2023;883:163553.
Dorokhov YL, Shindyapina AV, Sheshukova EV, Komarova TV. Metabolic methanol: molecular pathways and physiological roles. Physiol Rev. 2015;95:603–44.
Yu PH. Deamination of methylamine and angiopathy; toxicity of formaldehyde, oxidative stress and relevance to protein glycoxidation in diabetes. J Neural Transm Suppl. 1998;52:201–16.
Dingler FA, Wang M, Mu A, Millington CL, Oberbeck N, Watcham S, et al. Two Aldehyde Clearance Systems Are Essential to Prevent Lethal Formaldehyde Accumulation in Mice and Humans. Mol Cell. 2020;80:996–1012.e9.
Rorsman P, Ashcroft FM. Pancreatic beta-Cell Electrical Activity and Insulin Secretion: Of Mice and Men. Physiol Rev. 2018;98:117–214.
Kitasato H, Kai R, Ding WG, Omatsu-Kanbe M. The intrinsic rhythmicity of spike-burst generation in pancreatic beta-cells and intercellular interaction within an islet. Jpn J Physiol. 1996;46:363–73.
Gandasi NR, Yin P, Riz M, Chibalina MV, Cortese G, Lund PE, et al. Ca2+ channel clustering with insulin-containing granules is disturbed in type 2 diabetes. J Clin Invest. 2017;127:2353–64.
Fischer MJ, Soller KJ, Sauer SK, Kalucka J, Veglia G, Reeh PW. Formalin evokes calcium transients from the endoplasmatic reticulum. PLoS One. 2015;10:e0123762.
Zhao Y, Ge J, Li X, Guo Q, Zhu Y, Song J, et al. Vasodilatory effect of formaldehyde via the NO/cGMP pathway and the regulation of expression of K(ATP), BK(Ca) and L-type Ca(2+) channels. Toxicol Lett. 2019;312:55–64.
Chi YN, Zhang X, Cai J, Liu FY, Xing GG, Wan Y. Formaldehyde increases intracellular calcium concentration in primary cultured hippocampal neurons partly through NMDA receptors and T-type calcium channels. Neurosci Bull. 2012;28:715–22.
Kakuta I, Namba K, Uematsu K, Murachi S. Physiological response of the fish, Cyprinus carpio, to formalin exposure-I. Effects of formalin on urine flow, heart rate, respiration. Comp Biochem Physiol C Comp Pharm Toxicol. 1991;100:405–11.
Nieminen M, Pasanen P, Laitinen M. Effects of formalin treatment on the blood composition of salmon (Salmo salar) and rainbow trout (Salmo gairdneri). Comp Biochem Physiol C Comp Pharm Toxicol. 1983;76:265–9.
Tan T, Zhang Y, Luo W, Lv J, Han C, Hamlin JNR, et al. Formaldehyde induces diabetes-associated cognitive impairments. FASEB J. 2018;32:3669–79.
Wang J, Li J, Xu L, Tan D, Guo R, Lin W. A robust activatable two-photon fluorescent probe for endogenous formaldehyde biomarker visualization diagnosis and evaluation of diabetes mellitus. Anal Chim Acta. 2023;1266:341371.
Tong Z, Han C, Luo W, Li H, Luo H, Qiang M, et al. Aging-associated excess formaldehyde leads to spatial memory deficits. Sci Rep. 2013;3:1807.
Takeshita N, Yamaguchi I. Insulin attenuates formalin-induced nociceptive response in mice through a mechanism that is deranged by diabetes mellitus. J Pharm Exp Ther. 1997;281:315–21.
Conlon JM. Evolution of the insulin molecule: insights into structure-activity and phylogenetic relationships. Peptides. 2001;22:1183–93.
Metz B, Kersten GF, Hoogerhout P, Brugghe HF, Timmermans HA, de Jong A, et al. Identification of formaldehyde-induced modifications in proteins: reactions with model peptides. J Biol Chem. 2004;279:6235–43.
Metz B, Kersten GF, Baart GJ, de Jong A, Meiring H, ten Hove J, et al. Identification of formaldehyde-induced modifications in proteins: reactions with insulin. Bioconjug Chem. 2006;17:815–22.
Marti L, Abella A, Carpene C, Palacin M, Testar X, Zorzano A. Combined treatment with benzylamine and low dosages of vanadate enhances glucose tolerance and reduces hyperglycemia in streptozotocin-induced diabetic rats. Diabetes. 2001;50:2061–8.
Zou H, Yang W, Schwar G, Zhao R, Alansary D, Yin D, et al. High glucose distinctively regulates Ca(2+) influx in cytotoxic T lymphocytes upon target recognition and thapsigargin stimulation. Eur J Immunol. 2020;50:2095–8.
Shoshan-Barmatz V, De S. Mitochondrial VDAC, the Na(+)/Ca(2+) Exchanger, and the Ca(2+) Uniporter in Ca(2+) Dynamics and Signaling. Adv Exp Med Biol. 2017;981:323–47.
Dingreville F, Panthu B, Thivolet C, Ducreux S, Gouriou Y, Pesenti S, et al. Differential Effect of Glucose on ER-Mitochondria Ca(2+) Exchange Participates in Insulin Secretion and Glucotoxicity-Mediated Dysfunction of beta-Cells. Diabetes. 2019;68:1778–94.
Devor AW, Barichievich RO, Siddiqui B. Continued studies on the formalin method for liver glycogen. Anal Biochem. 1966;14:237–42.
Kobayashi M, Niinou T, Ishida M, Ishida Y. [Influence of adrenalectomy on liver glycogen and blood glucose in rats-with special reference to the effects of formalin, cortisone and ACTH]. Nihon Yakurigaku Zasshi. 1970;66:379–85.
Richardson L. Glycogenic Effect of Formaldehyde as Related to the Adrenal Cortex. Endocrinology. 1962;71:390–4.
Tong Z, Han C, Qiang M, Wang W, Lv J, Zhang S, et al. Age-related formaldehyde interferes with DNA methyltransferase function, causing memory loss in Alzheimer’s disease. Neurobiol Aging. 2015;36:100–10.
Mei Y, Jiang C, Wan Y, Lv J, Jia J, Wang X, et al. Aging-associated formaldehyde-induced norepinephrine deficiency contributes to age-related memory decline. Aging Cell. 2015;14:659–68.
Qiang M, Xiao R, Su T, Wu BB, Tong ZQ, Liu Y, et al. A novel mechanism for endogenous formaldehyde elevation in SAMP8 mouse. J Alzheimers Dis. 2014;40:1039–53.
The list of publication in the journal “Meditsina Truda i Promyshlennaia ekologiia” in 2000. Med Tr Prom Ekol. 2001:1:42–8.
Stolen CM, Madanat R, Marti L, Kari S, Yegutkin GG, Sariola H, et al. Semicarbazide sensitive amine oxidase overexpression has dual consequences: insulin mimicry and diabetes-like complications. Faseb j. 2004;18:702–4.
Boomsma F, van den Meiracker AH, Winkel S, Aanstoot HJ, Batstra MR, Man in ‘t Veld AJ, et al. Circulating semicarbazide-sensitive amine oxidase is raised both in type I (insulin-dependent), in type II (non-insulin-dependent) diabetes mellitus and even in childhood type I diabetes at first clinical diagnosis. Diabetologia. 1999;42:233–7.
Yang M, Ospina M, Tse C, Toth S, Caudill SP, Vesper HW. Ultraperformance Liquid Chromatography Tandem Mass Spectrometry Method To Determine Formaldehyde Hemoglobin Adducts in Humans as Biomarker for Formaldehyde Exposure. Chem Res Toxicol. 2017;30:1592–8.
Ruggiero JL, Voller LM, Shaik JA, Hylwa S. Formaldehyde in Electronic Cigarette Liquid (Aerosolized Liquid). Dermatitis. 2022;33:332–6.
Sodhi SK, Khanna A. More on hidden formaldehyde in e-cigarette aerosols. N Engl J Med. 2015;372:1576.
Ahmed S, Tsukahara S, Tin Tin Win S, Yamamoto S, Kunugita N, Arashidani K, et al. Effects of low-level formaldehyde exposure on synaptic plasticity-related gene expression in the hippocampus of immunized mice. J Neuroimmunol. 2007;186:104–11.
Rehman K, Haider K, Akash MSH. Cigarette smoking and nicotine exposure contributes for aberrant insulin signaling and cardiometabolic disorders. Eur J Pharm. 2021;909:174410.
Ismail AA. The insulin autoimmune syndrome (IAS) as a cause of hypoglycaemia: an update on the pathophysiology, biochemical investigations and diagnosis. Clin Chem Lab Med. 2016;54:1715–24.
Gonzalez-Casimiro CM, Merino B, Casanueva-Alvarez E, Postigo-Casado T, Camara-Torres P, Fernandez-Diaz CM, et al. Modulation of Insulin Sensitivity by Insulin-Degrading Enzyme. Biomedicines. 2021;9:86.
Kullenberg H, Rossen J, Johansson UB, Hagstromer M, Nystrom T, Kumlin M, et al. Increased levels of insulin-degrading enzyme in patients with type 2 diabetes mellitus. Endocrine. 2022;77:561–5.
Abdul-Ghani MA, DeFronzo RA. Pathogenesis of insulin resistance in skeletal muscle. J Biomed Biotechnol. 2010;2010:476279.
King MG, Baskin DG. Effect of paraformaldehyde fixation on localization and characterization of insulin-like growth factor-I (IGF-I) receptors in the rat brain. Anat Rec. 1991;231:467–72.
Xu H, Barnes GT, Yang Q, Tan G, Yang D, Chou CJ, et al. Chronic inflammation in fat plays a crucial role in the development of obesity-related insulin resistance. J Clin Invest. 2003;112:1821–30.
Hotamisligil GS, Spiegelman BM. Tumor necrosis factor alpha: a key component of the obesity-diabetes link. Diabetes. 1994;43:1271–8.
Rehman K, Akash MSH, Liaqat A, Kamal S, Qadir MI, Rasul A. Role of Interleukin-6 in Development of Insulin Resistance and Type 2 Diabetes Mellitus. Crit Rev Eukaryot Gene Expr. 2017;27:229–36.
Ma H, Lin J, Li L, Ding Z, Huang P, Song X, et al. Formaldehyde reinforces pro-inflammatory responses of macrophages through induction of glycolysis. Chemosphere. 2021;282:131149.
Ramos CO, Nardeli CR, Campos KKD, Pena KB, Machado DF, Bandeira ACB, et al. The exposure to formaldehyde causes renal dysfunction, inflammation and redox imbalance in rats. Exp Toxicol Pathol. 2017;69:367–72.
Ma H, Lou K, Shu Q, Song X, Xu H. Aldehyde dehydrogenase 2 deficiency reinforces formaldehyde-potentiated pro-inflammatory responses and glycolysis in macrophages. J Biochem Mol Toxicol. 2024;38:e23518.
Katsiki N, Mikhailidis DP, Banach M. Leptin, cardiovascular diseases and type 2 diabetes mellitus. Acta Pharm Sin. 2018;39:1176–88.
Yanai H, Yoshida H. Beneficial Effects of Adiponectin on Glucose and Lipid Metabolism and Atherosclerotic Progression: Mechanisms and Perspectives. Int J Mol Sci. 2019;20:1190.
Zhu WW, Ning M, Peng YZ, Tang YY, Kang X, Zhan KB, et al. Hydrogen Sulfide Inhibits Formaldehyde-Induced Senescence in HT-22 Cells via Upregulation of Leptin Signaling. Neuromolecular Med. 2019;21:192–203.
Zhan K, Ning M, Wang C, Tang Y, Gu H, Yan C, et al. Formaldehyde accelerates cellular senescence in HT22 cells: possible involvement of the leptin pathway. Acta Biochim Biophys Sin (Shanghai). 2016;48:771–3.
Emre E, Ogeturk M, Aydin S, Kuloglu T, Aksu F, Kavakli A. Carvacrol protects rat liver exposed to formaldehyde by regulating oxidative stress, and asprosin and subfatin hormones. Biotech Histochem. 2023;98:336–45.
Lepretti M, Martucciello S, Burgos Aceves MA, Putti R, Lionetti L. Omega-3 Fatty Acids and Insulin Resistance: Focus on the Regulation of Mitochondria and Endoplasmic Reticulum Stress. Nutrients. 2018;10:350.
Huang XT, Liu W, Zhou Y, Sun M, Sun CC, Zhang CY, et al. Endoplasmic reticulum stress contributes to NMDA-induced pancreatic beta-cell dysfunction in a CHOP-dependent manner. Life Sci. 2019;232:116612.
Chen F, Wang N, Tian X, Su J, Qin Y, He R, et al. The Protective Effect of Mangiferin on Formaldehyde-Induced HT22 Cell Damage and Cognitive Impairment. Pharmaceutics. 2023;15:1568.
May JM, de Haen C. Insulin-stimulated intracellular hydrogen peroxide production in rat epididymal fat cells. J Biol Chem. 1979;254:2214–20.
Luc K, Schramm-Luc A, Guzik TJ, Mikolajczyk TP. Oxidative stress and inflammatory markers in prediabetes and diabetes. J Physiol Pharmacol. 2019;70:809–824.
Moreira PI, Santos MS, Seiça R, Oliveira CR. Brain mitochondrial dysfunction as a link between Alzheimer’s disease and diabetes. J Neurol Sci. 2007;257:206–14.
Heo YJ, Choi SE, Jeon JY, Han SJ, Kim DJ, Kang Y, et al. Visfatin Induces Inflammation and Insulin Resistance via the NF-kappaB and STAT3 Signaling Pathways in Hepatocytes. J Diabetes Res. 2019;2019:4021623.
Jung WW, Kim EM, Lee EH, Yun HJ, Ju HR, Jeong MJ, et al. Formaldehyde exposure induces airway inflammation by increasing eosinophil infiltrations through the regulation of reactive oxygen species production. Environ Toxicol Pharm. 2007;24:174–82.
Ghelli F, Bellisario V, Squillacioti G, Grignani E, Garzaro G, Buglisi M, et al. Oxidative stress induction in woodworkers occupationally exposed to wood dust and formaldehyde. J Occup Med Toxicol. 2021;16:4.
Zhang D, Liu ZX, Choi CS, Tian L, Kibbey R, Dong J, et al. Mitochondrial dysfunction due to long-chain Acyl-CoA dehydrogenase deficiency causes hepatic steatosis and hepatic insulin resistance. Proc Natl Acad Sci USA. 2007;104:17075–80.
Chennamsetty I, Coronado M, Contrepois K, Keller MP, Carcamo-Orive I, Sandin J, et al. Nat1 Deficiency Is Associated with Mitochondrial Dysfunction and Exercise Intolerance in Mice. Cell Rep. 2016;17:527–40.
Anderson EJ, Lustig ME, Boyle KE, Woodlief TL, Kane DA, Lin CT, et al. Mitochondrial H2O2 emission and cellular redox state link excess fat intake to insulin resistance in both rodents and humans. J Clin Invest. 2009;119:573–81.
Miotto PM, LeBlanc PJ, Holloway GP. High-Fat Diet Causes Mitochondrial Dysfunction as a Result of Impaired ADP Sensitivity. Diabetes. 2018;67:2199–205.
Zerin T, Kim JS, Gil HW, Song HY, Hong SY. Effects of formaldehyde on mitochondrial dysfunction and apoptosis in SK-N-SH neuroblastoma cells. Cell Biol Toxicol. 2015;31:261–72.
Tang XQ, Ren YK, Zhou CF, Yang CT, Gu HF, He JQ, et al. Hydrogen sulfide prevents formaldehyde-induced neurotoxicity to PC12 cells by attenuation of mitochondrial dysfunction and pro-apoptotic potential. Neurochem Int. 2012;61:16–24.
Nadalutti CA, Stefanick DF, Zhao ML, Horton JK, Prasad R, Brooks AM, et al. Mitochondrial dysfunction and DNA damage accompany enhanced levels of formaldehyde in cultured primary human fibroblasts. Sci Rep.2020;10:5575.
Rohm TV, Meier DT, Olefsky JM, Donath MY. Inflammation in obesity, diabetes, and related disorders. Immunity. 2022;55:31–55.
Ahmed B, Sultana R, Greene MW. Adipose tissue and insulin resistance in obese. Biomed Pharmacother. 2021;137:111315.
Gemmink A, Bakker LE, Guigas B, Kornips E, Schaart G, Meinders AE, et al. Lipid droplet dynamics and insulin sensitivity upon a 5-day high-fat diet in Caucasians and South Asians.Sci Rep.2017;7:42393.
Yu PH, Deng YL. Endogenous formaldehyde as a potential factor of vulnerability of atherosclerosis: involvement of semicarbazide-sensitive amine oxidase-mediated methylamine turnover. Atherosclerosis. 1998;140:357–63.
Di YL, Yu Y, Zhao SJ, Huang N, Fei XC, Yao DD, et al. Formic acid induces hypertension-related hemorrhage in hSSAO(TG) in mice and human. Exp Neurol. 2022;358:114208.
Chen JK, Wu C, Su TC. Positive Association between Indoor Gaseous Air Pollution and Obesity: An Observational Study in 60 Households. Int J Environ Res Public Health. 2021;18:11447.
Yu PH, Wang M, Fan H, Deng Y, Gubisne-Haberle D. Involvement of SSAO-mediated deamination in adipose glucose transport and weight gain in obese diabetic KKAy mice. Am J Physiol Endocrinol Metab. 2004;286:E634–41.
Beaupere C, Liboz A, Feve B, Blondeau B, Guillemain G. Molecular Mechanisms of Glucocorticoid-Induced Insulin Resistance. Int J Mol Sci. 2021;22:623.
Sharma R, Kopchick JJ, Puri V, Sharma VM. Effect of growth hormone on insulin signaling. Mol Cell Endocrinol. 2020;518:111038.
Martinez B, Ortiz RM. Thyroid Hormone Regulation and Insulin Resistance: Insights From Animals Naturally Adapted to Fasting. Physiology (Bethesda). 2017;32:141–51.
Rao X, Patel P, Puett R, Rajagopalan S. Air pollution as a risk factor for type 2 diabetes. Toxicol Sci. 2015;143:231–41.
Taranenko LA. Features of dyslipidemia development and insulin resistance in female workers engaged in methanol and formaldehyde production. Med Tr Prom Ekol. 2013;11:5–8.
Letellier N, Gutierrez LA, Pilorget C, Artaud F, Descatha A, Ozguler A, et al. Association Between Occupational Exposure to Formaldehyde and Cognitive Impairment. Neurology. 2022;98:e633–e40.
Li F, Yujie Q, Gong S, Zhang H, Ding S. Learning and memory impairment of mice caused by gaseous formaldehyde. Environ Res. 2020;184:109318.
Obata T. Diabetes and semicarbazide-sensitive amine oxidase (SSAO) activity: a review. Life Sci. 2006;79:417–22.
Funding
Funding for this work was provided by the Major Program of the National Natural Science Foundation of China (62394314), the National Natural Science Foundation of China (82071214), Zhejiang Provincial Science and Technology Innovation Program (New Young Talent Program) for College Students (2023R413044).
Author information
Authors and Affiliations
Contributions
H.Y.X., H.S.C., Y.H.L., and T.C.L. were primarily responsible for drafting the review and conducting the related experimental validations. D.R.Z., X.X.C., H.Z.Z., X.Y.H., and H.X. provided valuable feedback on the initial manuscript and offered technical guidance during the experimental procedures. Z.Q.T. made significant contributions by consistently offering insightful and valuable suggestions on both the manuscript and the experimental design at every stage. Y.Q.W. and Y.P.S.T. also participated in supervision and contributed helpful guidance throughout the process.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Xu, H., Chen, H., Li, Y. et al. Dietary formaldehyde: a silent aggravator of diabetes and cognitive impairments. Nutr. Diabetes 15, 35 (2025). https://doi.org/10.1038/s41387-025-00390-x
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41387-025-00390-x