Abstract
GLP-1 and its synthetic analogs have emerged as significant therapeutic agents for the management of metabolic disorders, merging glycemic control with weight loss through innovative structural and delivery breakthroughs. This review provides a meticulous exploration of GLP-1, elucidating its origin, secretion, and the challenges associated with its clinical application due to its fragility in the presence of DPP-IV, resulting in a short half-life. To overcome this limitation, various modifications and delivery strategies to enhance the pharmacokinetic properties and therapeutic efficacy of GLP-1 analogs have been studied. The review delves into the intricacies of different modification approaches, including N and C-terminal modifications, Fatty acid Side chain Modifications, and Large Molecule Conjugation Modifications, highlighting their rationale and resulting improvements in half-life, stability, receptor binding, and bioactivity. Additionally, the importance of optimized delivery strategies to ensure sustained and controlled release of GLP-1 analogs is discussed. The culmination of these scientific advancements provides valuable insights for the development of more effective treatments for metabolic disorders, ultimately paving the way for improved patient outcomes in the realm of metabolic health.

Similar content being viewed by others
Introduction
Diabetes is a chronic metabolic disorder involving hyperglycemia and is characterized by defects in insulin secretion, insulin action, or both. If undiagnosed, diabetes can cause cardiovascular diseases, neuropathy (nerve damage), nephropathy (kidney damage), retinopathy (eye disorders), and even lower-limb amputation [1]. Diabetes has reached epidemic proportions globally, with an estimated 537 million adults affected in 2021. In the United States, the Centers for Disease Control and Prevention (CDC) reports that over 38.4 million individuals, approximately 11.6% of the population, are currently living with diabetes [2].
There are two types of diabetes: type 1 and type 2. Type 1 is an autoimmune disease in which insulin-producing β-cells are attacked by the host’s immune system, and is thought to be caused by a combination of viral infection, an environmental trigger, or gene malfunction [3, 4]. Type 1 diabetes mellitus (T1DM) patients typically need insulin injections for survival. Moreover, individuals with diabetes require continuous monitoring to prevent acute complications such as hypoglycemia and diabetic ketoacidosis. Type 2 diabetes mellitus (T2DM) is the predominant form, accounting for over 90% of all diabetes cases globally. It is characterized by insulin resistance, wherein peripheral tissues exhibit a diminished response to insulin, eventually resulting in impaired insulin utilization and relative insulin deficiency [3].
The current therapeutic paradigm for diabetes management entails a comprehensive, multi-modal strategy encompassing both lifestyle and pharmacological interventions. Foundational components include structured dietary modifications, regular physical activity, and weight management. Pharmacological treatment typically begins with non-insulin antihyperglycemic agents, either as monotherapy or in combination. These agents include thiazolidinediones (PPAR-γ agonists), dipeptidyl peptidase-4 (DPP-4) inhibitors, sulfonylureas, sodium-glucose co-transporter 2 (SGLT2) inhibitors, α-glucosidase inhibitors, and glucagon-like peptide-1 (GLP-1) receptor agonists. Insulin therapy, delivered via subcutaneous (SC) injections or continuous subcutaneous insulin infusion (CSII) systems, remains indispensable, particularly in individuals with T1DM and those with progressive or inadequately controlled T2DM.
GLP-1 and its analogs have emerged as significant therapeutic agents for managing metabolic disorders. GLP-1 is an endogenous incretin hormone produced by pancreatic L-cells through the proteolytic breakdown of the preproglucagon molecule. This breakdown generates two forms of GLP-1; a biologically active form GLP-1-(7–36) amide and an inactive form GLP-1-(1-37). The inactive form is considered a “precursor” as it gets converted into an active peptide GLP-1 (7-37) by cleaving a single arginine residue (Fig. 1) [5, 6]. Due to its insulinotropic action (stimulates insulin secretion), GLP-1-based treatments effectively prevent postprandial hyperglycemia (post-meal glucose spikes) in patients with T2DM. Additionally, its ability to suppress appetite contributes to weight loss and therefore offers synergistic benefits for managing T2DM.
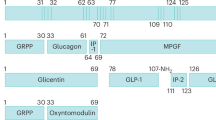
A figure illustrating the intricate relationship between the synthesis, processing, and tissue-specific physiological handling of glucagon precursors. The preproglucagon gene encodes proglucagon, which undergoes tissue-dependent post-translational processing. In the intestinal and neural tissues, prohormone convertase PCSK1/3 mediates the cleavage of proglucagon, producing glicentin-related pancreatic polypeptide (GRPP), oxyntomodulin (OXM), glucagon-like peptide-1 (GLP-1), intervening peptide-2 (IP-2), and glucagon-like peptide-2 (GLP-2). Conversely, in pancreatic islet α-cells, PCSK2 serves as the primary processing enzyme, generating glucagon, GRPP, intervening peptide-1 (IP-1), and a distinct proglucagon fragment.
However, despite these advantages, the clinical use of native GLP-1 is significantly limited by its short plasma half-life (1–2 min), owing to rapid enzymatic degradation by DPP-4 and renal clearance. These challenges necessitate frequent dosing or the use of modified analogs and innovative delivery systems to sustain therapeutic concentrations. The therapeutic potential of GLP-1 and its analogs extends beyond glycemic control and obesity management. Emerging evidence suggests their neuroprotective effects in neurodegenerative disorders, particularly Alzheimer’s disease, as well as potential anti-tumor properties in various malignancies [7].
Scope
This review explores GLP-1 receptor agonists as an emerging therapeutic trend in the management of diabetes. Despite their significant clinical potential, they are associated with certain limitations, including suboptimal pharmacokinetics and patient adherence challenges, necessitating the development of advanced drug delivery systems to optimize efficacy and improve compliance. Various chemical modifications, formulation techniques, and innovative approaches are being explored in an attempt to alleviate these limitations. This review seeks to provide a comprehensive analysis of these developments in GLP-1 formulation and delivery systems, along with insights into how these modifications enhance the efficacy and utility of GLP-1-based therapies.
GLP-1
Origin
Initially, it was hypothesized that GLP-1 was derived from proglucagon, a 158-amino-acid precursor, specifically from the N-terminal region of the major proglucagon fragment (MPGF). However, subsequent studies identified endogenous GLP-1 as corresponding to the proglucagon fragment spanning amino acids 78 to 107. Proglucagon, a precursor polypeptide, undergoes tissue-specific post-translational processing to yield various biologically active peptides in the pancreas and the gut/brain. In pancreatic cells, proglucagon is cleaved to produce glucagon, glicentin-related pancreatic polypeptide (GRPP), intervening peptide-1 (IP-1), and the major proglucagon fragment (MPGF), constituting GLP-1, IP-2, and GLP-2. In contrast, in intestinal cells and certain brain regions, prohormone convertase 1/3 (PC1/3) processes proglucagon to generate GLP-1, intervening peptide-2 (IP-2), and GLP-2. The hormones produced by the enzymatic processing of proglucagon then play essential roles in glucose metabolism and homeostasis [8, 9]. Figure 1 shows a schematic representation of the origin of GLP-1 via proglucagon.
Receptor binding and mechanism of action
The main signaling mechanism of GLP-1 is the activation of adenylate cyclase via the Gαs subunit, leading to increased production of cyclic AMP [6, 10]. This elevated cyclic AMP activates protein kinase A (PKA) and Epac2 pathways to potentiate glucose-dependent insulin secretion and insulin exocytosis, respectively [6]. Moreover, GLP-1 triggers a change in conformation of its receptor, leading to the activation of a Gs protein and the production of secondary messengers. Alanine scanning studies have identified specific amino acids, including His7, Gly10, Phe12, Thr13, Asp15, Phe28, and Ile29, that play crucial roles in receptor binding and activation. Substituting these amino acids with alanine resulted in a significant decrease in binding affinity and cAMP production. The N-terminal region of GLP-1 is considered critical for receptor activation, whereas the C-terminal region predominantly facilitates receptor binding. Figure 2 presents a schematic flowchart illustrating the sequential steps involved in the mechanism of GLP-1 action.
This figure delineates the molecular signaling mechanism activated by glucagon-like peptide-1 (GLP-1) in pancreatic β cells. Binding of GLP-1 to its receptor initiates G-protein activation, leading to stimulation of adenylyl cyclase and subsequent elevation of intracellular cyclic AMP (cAMP) levels. Increased cAMP activates protein kinase A (PKA), which, alongside CaMKIV, phosphorylates CREB to promote insulin gene transcription. Simultaneously, PKA triggers membrane depolarization and opens voltage-gated Ca²⁺ channels, elevating cytosolic Ca²⁺ concentrations. The rise in Ca²⁺ is a critical signal that facilitates exocytosis of insulin granules, coupling GLP-1 receptor activation to enhanced insulin secretion and synthesis.
Metabolism
GLP-1 gets rapidly metabolized in the circulation by dipeptidyl peptidase-4 (DPP-IV), resulting in a short plasma half-life of approximately 2 min [11]. DPP-IV cleaves GLP-1 at the peptide bond between Ala8-Glu9 (Fig. 3), generating the resultant metabolite, GLP-1(9-36)-NH2. This metabolite exhibits markedly reduced binding affinity and agonistic activity, with over a 100-fold lower than that of the intact peptide (Fig. 3). Consequently, only about 10–20% of total plasma GLP-1 remains in its biologically active form, with the majority rapidly inactivated by DPP-IV [12].
This image shows how DPP-4 cleaves active GLP-1 (left), rapidly converting it into an inactive form (right), thereby reducing its ability to stimulate insulin secretion.
GLP-1 receptor agonists (GLP-1 RAs)
GLP-1 receptor agonists, known as GLP-1 analogs, are structurally modified incretin mimetics that are specifically engineered to address and overcome the pharmacokinetic limitations of native GLP-1. Current formulations of GLP-1 analogs rely primarily on subcutaneous injections, which may pose challenges to patient convenience and long-term adherence. Figure 4 presents a flow chart outlining the currently available GLP-1 receptor agonists.
This table shows how modifying GLP-1 receptor agonists, by modulating terminal amino acids, adding fatty acid chains, or attaching large molecules, often extend their activity. These alterations aid the drug to last longer in the body and resist breakdown, making treatment more convenient and effective. Examples include drugs such as exenatide, liraglutide, and dulaglutide, each using a different modification strategy.
N and C terminal Modifications
Exenatide
Exendin-4 is a 39-amino acid peptide sourced from the venom of the Heloderma lizard, boasting a 53% homology with native human GLP-1 (Fig. 5A). It effectively evades DPP-IV inactivation by substituting alanine with glycine in the 8th position. Exenatide, a synthetic variant of exendin-4, incorporates strategic modifications that substitute asparagine (Asn) at the C-terminal with aspartic acid (Asp) and swap alanine (Ala) at the 8th position with glycine (Gly). These enhancements significantly bolster resistance to enzymatic degradation, leading to a half-life of 2–3 h (Fig. 5B). The difference between Exendin-4 and Exenatide lies primarily in the replacement of Ala with Valine (Val) at the 19th position and Asn with aspartic acid at the 28th position in Exendin-4. These critical modifications confer a slightly higher affinity to the GLP-1 receptor for both peptides, enabling lower concentrations to achieve equivalent receptor occupancy compared to native GLP-1 [13].
A GLP-1, B Exenatide, C Lixisenatide, D Taspoglutide | E Liraglutide, F Semaglutide, G Tirzepatide | H Dulaglutide, I Albiglutide, J Efpeglenatide.
Lixisenatide
Lixisenatide (Fig. 5C) is a GLP-1 receptor agonist that is based on the structure of exendin-4. It was developed by Sanofi Aventis under a license from Zealand Pharma. This peptide consists of 44 amino acids, featuring a modified C-terminus that includes the addition of six Lysine (Lys) residues and the removal of one Proline (Pro) residue. This structural modification enhances lixisenatide’s resistance to physiological degradation by DPP-IV, resulting in a half-life of approximately 2–4 h. Additionally, its binding affinity is four times greater than that of native human GLP-1, which supports the formulation of a convenient once-a-day dosage regimen [14, 15].
Taspoglutide
GLP-1 analogs can be effectively modified by incorporating alpha-aminoisobutyric acid (Aib) in place of Ala at the 8th position and Gly at the 35th position (Fig. 5D). Aib mimics the structure of d-amino acids, which considerably increases resistance against protease enzymes such as DPP-IV, leading to an extended half-life of up to 5 days. Moreover, Aib significantly promotes the formation of alpha helices within the peptide structure, which enhances the molecule’s enzymatic stability and potency without significantly altering the structural integrity of the native peptide [16].
Fatty acid side chain modifications
Liraglutide
The conjugation of a fatty acid chain to GLP-1 analogs significantly enhances their binding affinity to human serum albumin, thereby generating long-acting peptide formulations with improved pharmacokinetic profiles. This structural modification strategy has been pivotal in the development of GLP-1 receptor agonists with extended plasma half-lives [17].
The prolonged half-life of GLP-1 analogs is achieved through several mechanisms, such as delayed absorption from the subcutaneous injection site, albumin binding, which shields the molecule from enzymatic degradation by DPP-4, and reduced renal clearance. Liraglutide exemplifies this approach through specific molecular modifications. It features a C16 palmitic acid moiety conjugated to the Lys residue at the 26th position through a Glu residue. Additionally, the Ala at the 8th position is substituted with Gly, and the Arg at the 34th position is replaced by Lys (Fig. 5E). These alterations preserve approximately 97% sequence homology with native human GLP-1 [18].
In systemic circulation, only about 1–2% of liraglutide exists in the unbound (free) form, while the remainder is reversibly bound to albumin, effectively serving as a depot that enables sustained pharmacological action. The resulting elimination half-life of approximately 13 h supports a once-daily dosing regimen [19].
Semaglutide
Building upon the pharmacological success of liraglutide, Novo Nordisk developed semaglutide with strategic structural modifications to enhance its pharmacokinetic profile. Like liraglutide, semaglutide incorporates an Aib substitution at the 8th position in place of Ala, which confers resistance to enzymatic degradation by DPP-4. Additionally, Arg at the 34th position is replaced with Lys to accommodate site-specific acylation. A key differentiating feature is the attachment of a C18 fatty acid via a spacer to Lys at the 26th position, facilitating strong binding to albumin and thereby significantly prolonging plasma half-life. These structural refinements result in a half-life of approximately 160 h, enabling once-weekly subcutaneous administration while maintaining effective glycemic control (Fig. 5F) [19].
Tirzepatide
Tirzepatide is a novel, synthetic peptide engineered to achieve dual agonism of both the glucose-dependent insulinotropic polypeptide (GIP) receptor and GLP-1. This 39-amino-acid molecule incorporates structural elements derived from both native GIP and GLP-1, with targeted amino acid substitutions that optimize receptor binding and functional activity. Tirzepatide exhibits C-terminal homology to exenatide and is conjugated to a C20 fatty diacid moiety via a Lys-linked hydrophilic spacer, a modification strategy similar to that employed in semaglutide [20]. These modifications confer an extended elimination half-life of approximately 116.7 h, facilitating once-weekly dosing. Furthermore, tirzepatide demonstrates enhanced affinity for GLP-1R relative to native GLP-1, while simultaneously engaging the GIP receptor, thereby amplifying its therapeutic efficacy in glycemic control and weight reduction (Fig. 5G).
Tirzepatide’s mechanism of action combines the effects of two incretin hormones: GIP and GLP-1. Chronic hyperglycemia in diabetes downregulates GIP receptor (GIPR) expression and impairs GIP-mediated insulin secretion [21], rendering high-doses of GIP infusions ineffective [22]. Tirzepatide addresses the limitations of standalone GIP therapy in diabetes by combining GIPR and GLP-1 receptor (GLP-1R) agonism, leveraging their complementary mechanisms [23]. Its GLP-1 activity reduces blood glucose levels, creating an environment that restores GIPR sensitivity [24].
Tirzepatide exerts its therapeutic effects through dual agonism of two key incretin receptors: the GIPR and the GLP-1R. In individuals with T2DM, chronic hyperglycemia is known to downregulate GIPR expression and impair GIP-stimulated insulinotropic activity, thereby limiting the efficacy of GIP monotherapy [21]. Consequently, even high-dose GIP infusions have demonstrated minimal glucose-lowering effects in this population [22]. Tirzepatide overcomes these limitations by simultaneously activating both GIPR and GLP-1R, thereby harnessing their complementary mechanisms of action [25]. In parallel, the reduction in glycemic burden induced by GLP-1 activity creates a metabolic milieu that may restore GIPR sensitivity and functionality. This synergistic interaction enhances overall insulinotropic and glucoregulatory efficacy, positioning tirzepatide as a first-in-class dual incretin receptor agonist with superior metabolic outcomes.
Large molecule conjugation modification
Dulaglutide
Dulaglutide is a long-acting GLP-1 receptor agonist that employs Fc-fusion protein technology to achieve prolonged systemic exposure. The molecule consists of a modified GLP-1 analog, containing three key amino acid substitutions: Ala8→Gly, Gly22→Glu, and Arg36→Gly, covalently fused to the Fc (constant) region of human immunoglobulin G subclass 4 (IgG4) (Fig. 5H) [26]. This Fc domain serves a dual function: it sterically hinders access to DPP-4, thereby protecting the GLP-1 moiety from enzymatic degradation, and it engages the neonatal Fc receptor (FcRn), facilitating receptor-mediated recycling and preventing lysosomal degradation following cellular uptake.
Eli Lilly’s application of Fc-fusion technology in dulaglutide has significantly enhanced both the pharmacokinetic and pharmacodynamic properties of the peptide. This innovation results in an elimination half-life of approximately 90 h (~4 days), supporting a once-weekly SC dosing regimen. Clinical trials have demonstrated that weekly dulaglutide administration provides glycemic control comparable to once-daily liraglutide, while offering improved adherence and patient convenience.
Albiglutide
Albiglutide achieves extended systemic activity through genetic fusion with human serum albumin (HSA), a strategy distinct from the fatty acid-based albumin binding used in agents like liraglutide and semaglutide. Unlike these analogs, which bind reversibly to albumin via lipid side chains, albiglutide consists of two tandem GLP-1 (7–36) analog sequences covalently fused to a single HSA molecule in a defined molecular configuration (Fig. 5I). This design ensures spatial separation of the GLP-1 N-terminus from the albumin moiety, preserving receptor-binding capacity and maintaining full biological activity.
In addition to its HSA-mediated pharmacokinetic extension, albiglutide exhibits enhanced stability against enzymatic degradation by DPP-4. This is conferred by a targeted amino acid substitution at the 8th position, replacing alanine with glutamic acid, a modification also found in exenatide-based GLP-1 therapies. The combination of structural resistance to DPP-4 and prolonged half-life via HSA fusion allows for once-weekly administration, positioning albiglutide as a viable and efficacious GLP-1 receptor agonist for long-term glycemic control [14].
Efpeglenatide
Efpeglenatide is a novel, long-acting GLP-1 receptor agonist developed by Hanmi Pharmaceuticals (South Korea) utilizing the proprietary LAPSCOVERY™ (Long-Acting Protein/Peptide Discovery) technology platform. This technology enables extended pharmacological activity through the site-specific conjugation of a cysteine-albumin–conjugated Exendin-4 analog (CA-Exendin-4) to the Fc fragment of human immunoglobulin G subclass 4 (IgG4), linked via a flexible peptide spacer (Fig. 5J) [27]. The flexible linker ensures spatial separation between the Fc region and the GLP-1 receptor–binding domain, preserving receptor accessibility and bioactivity.
The Fc domain not only provides steric protection against proteolytic enzymes but may also reduce immunogenicity by minimizing the generation of anti-drug antibodies. Furthermore, the increased molecular weight of the Fc-conjugated construct decreases renal filtration and clearance. These combined mechanisms contribute to an extended elimination half-life of approximately 5.6 to 7.5 days [28]. This prolonged half-life permits reduced injection frequency compared to conventional GLP-1 analogs, offering improved patient adherence and sustained glycemic control [29].
Drug delivery systems: transforming GLP-1 analogues (as illustrated in Fig. 6)
Nano-formulations
Nano-formulations are advanced drug delivery systems in which active pharmaceutical ingredients (APIs) are formulated at the nanoscale, typically with particle sizes less than 1 µm [30, 31]. These nanosystems offer a transformative platform for enhancing the pharmacological and biopharmaceutical performance of various drugs, including GLP-1 receptor agonists, which are otherwise limited by poor oral bioavailability, enzymatic degradation, and rapid renal clearance.
This figure highlights various drug delivery technologies, from microneedles to oral tablets, each engineered to tailor the drug delivery and release profiles.
Common types of nanoformulations include liposomes, polymeric nanoparticles, solid lipid nanoparticles (SLNs), nanostructured lipid carriers (NLCs), nano-emulsions, micelles, dendrimers, nanocrystals, and nano-complexes [32]. Each system can be tailored to achieve specific objectives such as prolonged circulation, enhanced tissue targeting, protection from proteolytic enzymes, and controlled or sustained drug release. These technologies are summarized in Table 1A.
In the context of GLP-1 analogs, nanoformulations have shown significant promise in addressing their inherent pharmacokinetic limitations. For instance, liposomal carriers have been developed to encapsulate exenatide and liraglutide, demonstrating improved stability and sustained release upon subcutaneous or intranasal administration. Polymeric nanoparticles, especially those derived from PLGA (poly(lactic-co-glycolic acid)), have been used to prepare long-acting formulations of GLP-1 that can extend the release period to weeks with a single dose, reducing injection frequency and improving patient adherence [33].
Recent advances have explored PEGylated lipid nanoparticles, which combine the benefits of stealth properties (to evade immune clearance) and controlled delivery. For example, PEG-lipid modified exenatide-loaded nanoparticles have been studied for their ability to cross mucosal barriers and improve oral bioavailability [34]. Another promising direction involves mucoadhesive nanocarriers that anchor to intestinal epithelium and release GLP-1 analogs in a site-specific manner, minimizing systemic degradation [35].
Nanocrystal suspensions of poorly water-soluble GLP-1 analogs are also under investigation to increase dissolution rates and promote lymphatic absorption. Additionally, thermosensitive nanogels and stimuli-responsive nanocarriers that respond to pH or glucose levels have been proposed for precision-controlled GLP-1 delivery, enabling intelligent drug release based on glycemic fluctuations.
These nano-based strategies not only optimize pharmacokinetics and therapeutic efficacy, but also hold potential for non-invasive delivery routes, including oral, buccal, transdermal, and pulmonary administration. As such, nanoformulations represent a crucial frontier in advancing the next generation of GLP-1-based therapeutics for the effective management of type 2 diabetes and related metabolic disorders [36, 37].
Microparticles
Microparticles are discrete, spherical entities typically ranging in diameter from 10 μm to 1 mm and are widely employed as drug delivery systems. Structurally, they can be categorized into microcapsules, which consist of an API enclosed within a polymeric or lipid shell, and microspheres, in which the API is homogeneously distributed throughout the polymeric matrix [33, 34]. These systems are fabricated using a variety of materials, including biodegradable polymers, lipids, and inorganic compounds, depending on the desired release profile and route of administration. The principal advantages of microparticulate delivery systems include controlled and sustained drug release, enhanced bioavailability, improved physicochemical stability, and site-specific targeting, all of which contribute to optimized therapeutic outcomes and increased patient adherence [35]. Microparticles can be formulated into diverse dosage forms such as dry powders, injectable suspensions (e.g., microspheres and liposomes), and are suitable for multiple routes of administration, including oral, pulmonary, and parenteral delivery, as summarized in Table 1B.
Microneedles
Microneedles (MNs) are minimally invasive devices engineered as micron-scale projections, typically ranging from 25 μm to 2 mm in length. They are designed to facilitate the precise and painless delivery of therapeutic agents through the skin and other biological barriers. [36, 37] Unlike conventional hypodermic needles, microneedle systems create transient microchannels that bypass the stratum corneum, enabling efficient transdermal transport of a wide range of molecules, including peptides, proteins, and small drugs, without reaching nerve-rich deeper layers and thus minimizing pain and risk of infection [38, 39].
Recent advances have expanded the use of MNs for GLP-1 receptor agonists, which are traditionally limited by poor oral bioavailability and require frequent subcutaneous injections. Innovative studies have demonstrated the feasibility of biodegradable microneedle patches incorporating GLP-1 analogs such as exenatide and liraglutide, achieving sustained glucose-lowering effects in preclinical models. These systems often use biocompatible polymers like polyvinylpyrrolidone (PVP), polyvinyl alcohol (PVA), sodium alginate, or gelatin, and may include functional components such as egg-derived proteins or silk fibroin to mimic biological properties and enhance mechanical strength and drug stability [40].
Furthermore, smart microneedle systems have emerged, incorporating glucose-responsive materials such as phenylboronic acid or glucose oxidase-modified hydrogels. These platforms enable on-demand insulin or GLP-1 release triggered by elevated blood glucose levels, providing a closed-loop feedback mechanism that enhances therapeutic precision and reduces the risk of hypoglycemia [41].
Another promising direction involves dissolving microneedles, which completely degrade upon administration, eliminating the need for disposal and improving patient compliance. In vivo studies have shown that microneedle-mediated delivery of semaglutide analogs leads to comparable or improved pharmacokinetics versus subcutaneous injections, with potential for once-weekly or biweekly dosing schedules [42].
In addition, 3D printing and microfabrication technologies now allow for precise customization of microneedle geometry, drug loading capacity, and release kinetics, tailored to individual patient needs and molecular characteristics of the peptide drug. These advancements position microneedle technology as a transformative approach for delivering GLP-1 receptor agonists, offering the potential to improve therapeutic adherence, minimize systemic side effects, and support decentralized care models for diabetes management [43]. Recent developments and formulations are summarized in Table 1C.
Injectable hydrogels
Injectable hydrogels are stimuli-responsive polymeric networks designed to undergo sol-to-gel transitions under physiological conditions, thereby enabling localized and sustained drug delivery via minimally invasive SC administration [44]. These systems typically employ temperature- or pH-sensitive biodegradable polymers that form viscoelastic depots in situ, facilitating controlled release of therapeutic agents such as GLP-1 receptor agonists while circumventing systemic toxicity [45].
By modulating electrostatic interactions between charged peptide drugs and the polymer matrix, these hydrogels enable sustained and steady drug elution over extended periods, ranging from days to weeks, significantly reducing dosing frequency from daily to weekly or even monthly intervals [46]. Their shear-thinning properties permit administration through standard syringe needles, and their biodegradability ensures gradual clearance from the body without local inflammation [46]. Various hydrogel formulations, differentiated by their physicochemical properties and release kinetics, are summarized in Table 1D.
Implants
Implantable SC implants and osmotic pumps represent advanced technologies for sustained drug delivery, leveraging distinct mechanisms to achieve long-term therapeutic efficacy. For instance, Vivani’s NanoPortal™ technology utilizes nanostructured titanium oxide membranes with vertically aligned nanotubes to enable sustained, tunable drug delivery through pore-size-dependent diffusion kinetics [47]. This subdermal implant platform eliminates pumps and electronics, instead relying on customizable nanotube dimensions to achieve steady release profiles for peptides and proteins over a long time. By maintaining pore sizes slightly larger than the drug molecule, the system ensures near-constant elution rates [47]. Osmotic pumps have proven to be another implantable solution to achieve sustained therapeutic efficacy. They utilize osmotic pressure gradients generated by salt-based cores to drive drug release through semipermeable membranes, enabling precise kinetics independent of physiological conditions [48]. Both technologies eliminate daily dosing facilitated by their subcutaneous placement, which avoids first-pass metabolism and gastrointestinal degradation. Table 1E summarizes the implantable technologies involved in the development of a formulation for sustained delivery.
Current marketed formulations
Semaglutide (Rybelsus®, Ozempic®, and Wegovy®)
Oral drug delivery offers significant advantages in terms of patient adherence, particularly in the chronic management of T2DM. A major advancement in this area is Emisphere’s Eligen® technology, which employs sodium N-(8-[2-hydroxybenzoyl] amino) caprylate (SNAC) as a permeation enhancer to facilitate the oral bioavailability of peptide-based drugs. This technology underpins the formulation of oral semaglutide (Rybelsus®) and enables its effective absorption in the stomach, as illustrated in Fig. 7 [49, 50]. SNAC functions through a multifaceted mechanism: it buffers the local gastric pH, thereby protecting semaglutide from acidic degradation and proteolytic enzymes, and enhances drug solubility and monomerization. Importantly, SNAC transiently fluidizes the gastric epithelial membrane, promoting transcellular absorption of semaglutide while preserving tight junction integrity.
This illustration demonstrates how SNAC in the tablet helps oral semaglutide survive stomach acid. SNAC raises local pH, protects the drug from gastric enzymes, and increases its absorption through the stomach lining.
Preclinical pharmacoscintigraphic imaging and studies in canine models have demonstrated that semaglutide is primarily absorbed in the stomach, with higher drug concentrations detected in the splenic vein relative to the portal vein, indicating site-specific uptake [51]. This innovative approach effectively bypasses the traditional barriers associated with oral peptide delivery, offering a clinically viable and patient-preferred alternative to injectable formulations.
Semaglutide is also available as injectable formulations, including Ozempic® and Wegovy®, which are widely recognized for their efficacy in the management of T2DM and obesity. Both formulations are administered as once-weekly SC injections due to the long half-life of Semaglutide (approximately one week). Furthermore, Ozempic® is primarily used for glycemic control in diabetes patients, while Wegovy® targets weight management by reducing hunger and enhancing satiety through activation of GLP-1 receptors [52]. The pharmacokinetic profiles of these formulations indicate that maximum plasma concentrations are typically attained within 1 to 3 days following SC administration, while steady-state plasma levels are generally achieved after 4 to 5 weeks of once-weekly dosing. Dosing regimens for both products involve gradual titration to optimize therapeutic outcomes; Ozempic® progresses from 0.25 mg to a maintenance dose of up to 1 mg, whereas Wegovy® reaches a higher maintenance dose of 2.4 mg to support weight loss goals [52, 53].
Dulaglutide (Trulicity®)
Dulaglutide, marketed as Trulicity® formulated as a fusion protein. Its extended half-life of approximately 4.7 days is attributed to its structural design, discussed in the large molecule conjugation section of this paper. This pharmacokinetic profile supports once-weekly subcutaneous administration, delivered via prefilled autoinjector pens available in 0.75 mg, 1.5 mg, 3.0 mg, and 4.5 mg per 0.5 mL dose strengths [54]. The formulation is stabilized using excipients such as citric acid, mannitol, and polysorbate 80, while an automated needle mechanism facilitates self-injection by extending and retracting the needle, thereby improving patient adherence and ease of use.
Clinically, dulaglutide exhibits dose-dependent efficacy. In the FREEDOM-1 trial, daily doses of 40–60 µg resulted in HbA1c reductions of 1.1–1.2% over 39 weeks (p < 0.001 vs. placebo), while higher weekly doses (3.0–4.5 mg) achieved reductions of up to 1.51% at 36 weeks. Concurrently, patients also experienced sustained weight loss of 2.3–3.0 kg (p ≤ 0.015 vs. placebo), highlighting its dual glycemic and weight-lowering benefits [55]. Furthermore, the REWIND trial demonstrated a 12% reduction in major adverse cardiovascular events (MACE) (p = 0.026), including among patients without established cardiovascular disease, alongside renoprotective effects evidenced by reductions in albuminuria [56]. Dulaglutide’s safety profile is consistent with other GLP-1 receptor agonists, with transient nausea being the most frequently reported adverse event (14.6% incidence).⁵⁵ Dose escalation from 0.75 to 1.5 mg is recommended after at least four weeks, based on individual glycemic control needs [57].
Exenatide (Byetta® and Bydureon®)
Exenatide is currently marketed in two primary formulations: Byetta® and Bydureon®, representing the immediate-release and extended-release forms, respectively. Byetta® is administered twice daily via SC injection and delivers rapid, short-term GLP-1 receptor activation. In contrast, Bydureon® is a long-acting, once-weekly formulation, developed using a microsphere-based delivery system. The extended-release (ER) formulation encapsulates exenatide within biodegradable poly(D, L-lactide-co-glycolide) (PLGA) microspheres, which form a polymeric matrix that allows sustained drug release following SC administration [13, 14, 58]. Upon injection, the PLGA matrix undergoes gradual hydrolysis, releasing the peptide over time. Pharmacokinetic data indicate that fasting plasma glucose (FPG) levels begin to decrease within two weeks, and steady-state plasma concentrations are typically achieved after six weeks of weekly dosing [59]. Interestingly, the plasma concentration achieved with once-weekly exenatide ER is comparable to that of a single injection of immediate-release exenatide (Byetta®) [60, 61]. This extended-release delivery not only improves glycemic control but also enhances patient adherence by reducing the injection frequency from twice daily to once weekly.
Discontinued formulations
Efpeglenatide and taspoglutide
Efpeglenatide and taspoglutide were once-promising, long-acting GLP-1 receptor agonists developed for the treatment of T2DM, both designed for once-weekly administration. Despite demonstrating robust glycemic efficacy, both agents were eventually discontinued due to distinct tolerability and safety concerns, diverging in severity and clinical implications.
Efpeglenatide, formulated using advanced Fc-PEG conjugation technology, exhibited dose-dependent gastrointestinal adverse events consistent with the GLP-1 class profile. Reported incidences included nausea (5.9–22.2%), diarrhea (8.8–25.3%), vomiting (2.9–9.1%), and constipation (8.8–16.2%), with discontinuation rates reaching 19% in high-dose monthly regimens (8–16 mg) [62,63,64]. Importantly, efpeglenatide’s immunogenicity remained relatively low, with few patients developing clinically significant anti-drug antibodies, allowing its safety profile to be viewed as manageable within class expectations. However, these tolerability issues ultimately impeded its regulatory and clinical progression.
In contrast, taspoglutide’s development was halted due to severe immunogenic and hypersensitivity complications. Up to 55% of patients developed anti-drug antibodies [65], which were associated with systemic allergic reactions in approximately 5% of cases and injection-site hypersensitivity, including anaphylaxis, which necessitated early trial termination [66, 67]. These immune-mediated events were compounded by intolerable gastrointestinal side effects, including nausea in 59% and vomiting in 37% of participants, along with reports of acute pancreatitis, culminating in a dropout rate of 26–34% in Phase 3 trials [65].
While both agents struggled with the inherent GI burden typical of GLP-1RAs, efpeglenatide’s limitations were largely dose-related and within manageable expectations, whereas taspoglutide’s combination of severe immunogenicity and hypersensitivity reactions rendered it clinically unviable.
5.7.2. Albiglutide (Tanzeum)
Albiglutide, marketed as Tanzeum, is a fusion protein similar to Dulaglutide. The drug is delivered via a dual-chamber prefilled injection pen containing lyophilized albiglutide powder and a diluent. The powder is reconstituted with the diluent before administration, yielding a yellow solution stabilized by excipients such as mannitol, polysorbate 80, sodium phosphate, and trehalose dihydrate. [68] Clinical trials have demonstrated the efficacy of albiglutide in lowering HbA1c levels and improving glycemic control. In Phase III studies under the Harmony program, patients treated with albiglutide showed statistically significant reductions in HbA1c compared to placebo. At Week 52, HbA1c reductions were: −0.7% for the 30 mg dose and −0.9% for the 50 mg dose versus +0.2% with placebo (p < 0.001). Additionally, 49% of patients on the 30 mg dose achieved HbA1c levels below 7%, compared to only 21% in the placebo group. Fasting plasma glucose levels also improved significantly, with reductions of −16 mg/dL (30 mg dose) and −25 mg/dL (50 mg dose) compared to an increase of +18 mg/dL with placebo (p < 0.001) [69]. The Harmony Outcomes trial further revealed that albiglutide reduced MACE by 22% over a median follow-up period of 1.6 years in patients with type 2 diabetes and cardiovascular disease (p = 0.0006) [70]. While albiglutide demonstrated good glycemic efficacy and tolerability, adverse effects included upper respiratory tract infections, diarrhea, nausea, and injection site reactions. Weight changes were minimal and not significantly different from placebo [71]. These findings underscore albiglutide’s potential as an effective adjunct therapy for glycemic control and cardiovascular risk reduction in T2DM management.
Future direction
The evolution of GLP-1RAs is increasingly shifting toward oral formulations, aiming to enhance patient adherence by eliminating the need for injections and addressing the logistical complexities associated with traditional peptide therapies [72, 73]. The preclinical studies on GLP-1 receptor agonists have been comprehensively summarized in Table 2. Despite significant progress observed in clinical trial development, there remains a scarcity of published clinical data, particularly regarding long-term safety, efficacy across diverse populations, and real-world effectiveness. Notably, only one registered clinical study exists to date, which evaluates an implant-based delivery system of Exenatide and is currently in Phase 1 with an ‘Active, not recruiting’ status (NCT05670379), initiated on December 20, 2024, and expected to conclude by August 2025. Unlike injectable formulations that typically require cold-chain storage, oral GLP-1RAs bypass refrigeration constraints, thereby reducing manufacturing and distribution costs and improving accessibility in resource-limited settings [73]. Moreover, small-molecule GLP-1RAs offer a solution to the bioavailability limitations faced by oral peptide-based agents, which often depend on absorption enhancers (e.g., SNAC) and strict fasting protocols [74]. Their compact and chemically stable structures facilitate efficient gastrointestinal absorption, independent of dietary restrictions, which simplifies both patient administration and large-scale manufacturing.
Eli Lilly’s orforglipron represents a breakthrough in this transition as the first oral small-molecule GLP-1RA to complete Phase 3 trials. In the ACHIEVE-1 study, a 36 mg dose resulted in a reduction in HbA1c of 1.3–1.6% from a baseline of 8.0%, and an average weight loss of 7.9% (approximately 16.0 lbs) over 40 weeks. Regulatory submissions are anticipated in late 2025 for obesity and in 2026 for type 2 diabetes [75]. Orforglipron’s oral delivery route and scalable production capacity position it as a frontrunner in democratizing access to incretin-based therapies, particularly for patients who are hesitant or unable to adhere to injectable regimens. Its development marks a strategic pivot toward oral-first therapeutic models, potentially redefining the landscape of metabolic disease management while alleviating global supply chain limitations.
Conclusion
This comprehensive review highlights the therapeutic potential of GLP-1 and its analogs in the management of metabolic disorders, particularly T2DM. The inherent pharmacokinetic limitations of native GLP-1, such as its short biological half-life, have catalyzed significant advancements through in-depth research into its physiology, secretion dynamics, and molecular characteristics. To overcome these limitations, this review discusses a range of structural modifications, including N-terminal and C-terminal substitutions, fatty acid conjugation, and large molecule fusion technologies, which have collectively contributed to enhanced half-life, increased stability, improved receptor affinity, and retained or augmented bioactivity of GLP-1 analogs. Furthermore, the review explores novel drug delivery strategies, such as microsphere encapsulation, injectable hydrogels, Fc-fusion, and oral small-molecule formulations, aimed at achieving sustained and controlled release to reduce dosing frequency and improve patient compliance.
Beyond glycemic control, the pleiotropic actions of GLP-1 on various organs and metabolic pathways are emphasized, underscoring its potential utility in addressing cardiovascular, renal, and obesity-related complications. Collectively, these insights position GLP-1 and its analogs as versatile and promising agents in the broader context of metabolic disease management.
References
Gregg EW, Li Y, Wang J, Rios Burrows N, Ali MK, Rolka D, et al. Changes in diabetes-related complications in the United States, 1990-2010. N Engl J Med. 2014;370:1514–23. https://doi.org/10.1056/NEJMoa1310799.
CDC [Internet]. 2024. National Diabetes Statistics Report. Available from: https://www.cdc.gov/diabetes/php/data-research/index.html.
Atkinson MA, Eisenbarth GS, Michels AW. Type 1 diabetes. Lancet. 2014;383:69–82. https://doi.org/10.1016/S0140-6736(13)60591-7.
Craig ME, Hattersley A, Donaghue KC. Definition, epidemiology and classification of diabetes in children and adolescents. Pediatr Diab. 2009;10:3–12. https://doi.org/10.1111/j.1399-5448.2009.00568.x.
Smith NK, Hackett TA, Galli A, Flynn CR. GLP-1: molecular mechanisms and outcomes of a complex signaling system. Neurochem Int. 2019;128:94–105. https://doi.org/10.1016/j.neuint.2019.04.010.
Ahrén B. Glucagon-like peptide-1 (GLP-1): a gut hormone of potential interest in the treatment of diabetes. Bioessays. 1998;20:642–51.
Drucker DJ. Mechanisms of action and therapeutic application of glucagon-like peptide-1. Cell Metab. 2018;27:740–56. https://doi.org/10.1016/j.cmet.2018.03.001.
Sandoval DA, D’Alessio DA. Physiology of proglucagon peptides: role of glucagon and GLP-1 in health and disease. Physiol Rev. 2015;95:513–48. https://doi.org/10.1152/physrev.00013.2014.
Vrang N, Larsen PJ. Preproglucagon derived peptides GLP-1, GLP-2 and oxyntomodulin in the CNS: role of peripherally secreted and centrally produced peptides. Prog Neurobiol. 2010;92:442–62. https://doi.org/10.1016/j.pneurobio.2010.07.003.
Marzook A, Tomas A, Jones B. The interplay of glucagon-like peptide-1 receptor trafficking and signalling in pancreatic beta cells. Front Endocrinol. 2021;12:678055. https://doi.org/10.3389/fendo.2021.678055.
Ismail R, Csóka I. Novel strategies in the oral delivery of antidiabetic peptide drugs-Insulin, GLP 1 and its analogs. Eur J Pharm Biopharm. 2017;115:257–67. https://doi.org/10.1016/j.ejpb.2017.03.015.
Manandhar B, Ahn JM. Glucagon-like peptide-1 (GLP-1) analogs: recent advances, new possibilities, and therapeutic implications. J Med Chem. 2015;58:1020–37. https://doi.org/10.1021/jm500810s.
Mayo KE, Miller LJ, Bataille D, Dalle S, Göke B, Thorens B, et al. International Union of Pharmacology. XXXV. The glucagon receptor family. Pharm Rev. 2003;55:167–94. https://doi.org/10.1124/pr.55.1.6.
Seewoodhary J, Davies J. Novel GLP-1 mimetics in diabetes: lixisenatide and albiglutide. Future Prescr. 2011;12:20–2. https://doi.org/10.1002/fps.79.
Barnett AH. Lixisenatide: evidence for its potential use in the treatment of type 2 diabetes. Core Evid. 2011;67–79. https://doi.org/10.2147/CE.S15525.
Sharma D, Verma S, Vaidya S, Kalia K, Tiwari V. Recent updates on GLP-1 agonists: current advancements & challenges. Biomed Pharmacother. 2018;108:952–62. https://doi.org/10.1016/j.biopha.2018.08.088.
Heinemann L, Sinha K, Weyer C, Loftager M, Hirschberger S, Heise T. Time-action profile of the soluble, fatty acid acylated, long-acting insulin analogue NN304. Diabet Med. 1999;16:332–8. https://doi.org/10.1046/j.1464-5491.1999.00081.x.
Juhl CB, Hollingdal M, Sturis J, Jakobsen G, Agersø H, Veldhuis J, et al. Bedtime administration of NN2211, a long-acting GLP-1 derivative, substantially reduces fasting and postprandial glycemia in type 2 diabetes. Diabetes. 2002;51:424–9. https://doi.org/10.2337/diabetes.51.2.424.
Knudsen LB. Inventing liraglutide, a glucagon-like peptide-1 analogue, for the treatment of diabetes and obesity. ACS Pharm Transl Sci. 2019;2:468–84. https://doi.org/10.1021/acsptsci.9b00048.
Gault VA, Flatt PR, Harriott P, Mooney MH, Bailey CJ, O’Harte FPM. Improved biological activity of Gly^ 2-and Ser^ 2-substituted analogues of glucose-dependent insulinotrophic polypeptide. J Endocrinol. 2003;176:133–42. https://doi.org/10.1677/joe.0.1760133.
Min T, Bain SC. The role of tirzepatide, dual GIP and GLP-1 receptor agonist, in the management of type 2 diabetes: the SURPASS clinical trials. Diab Ther. 2021;12:143–57. https://doi.org/10.1007/s13300-020-00981-0.
Mentis N, Vardarli I, Köthe LD, Holst JJ, Deacon CF, Theodorakis M, et al. GIP does not potentiate the antidiabetic effects of GLP-1 in hyperglycemic patients with type 2 diabetes. Diabetes. 2011;60:1270–6. https://doi.org/10.2337/db10-1332.
Pratley RE. GIP: an inconsequential incretin or not?. Diab Care Am Diab Assoc. 2010;33:1691–2. https://doi.org/10.2337/dc10-0704.
Coskun T, Sloop KW, Loghin C, Alsina-Fernandez J, Urva S, Bokvist KB, et al. LY3298176, a novel dual GIP and GLP-1 receptor agonist for the treatment of type 2 diabetes mellitus: from discovery to clinical proof of concept. Mol Metab. 2018;18:3–14. https://doi.org/10.1016/j.molmet.2018.09.009.
WHO [Internet]. 2024 [cited 2024 Dec 30]. Disease Outbreak News (DONs). Available from: https://www.who.int/emergencies/disease-outbreak-news
Naver SV, Jimenez-Solem E, Christensen M, Andersen JT, Knop FK. Dulaglutide: a novel once-weekly glucagon-like peptide-1 receptor agonist. 2014; https://doi.org/10.4155/cli.14.65.
Zheng Z, Zong Y, Ma Y, Tian Y, Pang Y, Zhang C, et al. Glucagon-like peptide-1 receptor: mechanisms and advances in therapy. Signal Transduct Target Ther. 2024;9:234 https://doi.org/10.1038/s41392-024-01931-z.
Rosenstock J, Sorli CH, Trautmann ME, Morales C, Wendisch U, Dailey G, et al. Once-weekly efpeglenatide dose-range effects on glycemic control and body weight in patients with type 2 diabetes on metformin or drug naive, referenced to liraglutide. Diab Care. 2019;42:1733–41. https://doi.org/10.2337/dc18-2648.
Reid J, Rana K, Segel SA, Sheikh-Ali M, Choksi RR, Goldfaden RF. Efpeglenatide. Glucagon-like peptide 1 (GLP-1) receptor agonist, treatment for type 2 diabetes. Drugs Future. 2019;44. https://doi.org/10.1358/dof.2019.44.6.2927591.
International Pharmaceutical Industry [Internet]. Nanoformulation for enhanced drug delivery and better patient compliance. Available from: https://international-pharma.com/wp-content/uploads/2023/09/Nanoformulation-for-Enhanced-Drug-Delivery-and-Better-Patient-Compliance.pdf.
Jeevanandam J, San Chan Y, Danquah MK. Nano-formulations of drugs: recent developments, impact and challenges. Biochimie. 2016;128:99–112. https://doi.org/10.1016/j.biochi.2016.07.008.
Tiwary P, Oswal K, Varghese R, Anchan H, Gupta P. Solid lipid nanoparticles in the diagnosis and treatment of prostate cancer: a comprehensive review of two decades of advancements. Chem Phys Lipids. 2025;105510. https://doi.org/10.1016/j.chemphyslip.2025.105510.
Freitas S, Merkle HP, Gander B. Microencapsulation by solvent extraction/evaporation: reviewing the state of the art of microsphere preparation process technology. J Controlled Release. 2005;102:313–32. https://doi.org/10.1016/j.jconrel.2004.10.015.
Bao Z, Kim J, Kwok C, Le Devedec F, Allen C. A dataset on formulation parameters and characteristics of drug-loaded PLGA microparticles. Sci Data. 2025;12:364. https://doi.org/10.1038/s41597-025-04621-9.
da Silva RYP, de Menezes DLB, Oliveira V, da S, Converti A, de Lima ÁAN. Microparticles in the development and improvement of pharmaceutical formulations: an analysis of in vitro and in vivo studies. Int J Mol Sci. 2023;24:5441 https://doi.org/10.3390/ijms24065441.
Hulimane Shivaswamy R, Binulal P, Benoy A, Lakshmiramanan K, Bhaskar N, Pandya HJ. Microneedles as a promising technology for disease monitoring and drug delivery: a review. ACS Mater Au. 2024. https://doi.org/10.1021/acsmaterialsau.4c00125.
Kim YC, Park JH, Prausnitz MR. Microneedles for drug and vaccine delivery. Adv Drug Delivery Rev. 2012;64:1547-68. https://doi.org/10.1016/j.addr.2012.04.005.
Avcil M, Çelik A. Microneedles in drug delivery: progress and challenges. Micromachines. 2021;12:1321 https://doi.org/10.3390/mi12111321.
Chen J, Ren H, Zhou P, Zheng S, Du B, Liu X, et al. Microneedle-mediated drug delivery for cutaneous diseases. Front Bioeng Biotechnol. 2022;10:1032041. https://doi.org/10.3389/fbioe.2022.1032041.
Chen G, Yu J, Gu Z. Glucose-responsive microneedle patches for diabetes treatment. J Diab Sci Technol. 2019;13:41–8. https://doi.org/10.1177/1932296818778607.
Chen MC, Ling MH, Lai KY, Pramudityo E. Chitosan microneedle patches for sustained transdermal delivery of macromolecules. Biomacromolecules. 2012;13:4022–31. https://doi.org/10.1021/bm301293d.
Prausnitz MR, Langer R. Transdermal drug delivery. Nat Biotechnol. 2008;26:1261–8. https://doi.org/10.1038/nbt.1504.
Bodhale DW, Nisar A, Afzulpurkar N. Design, fabrication, and analysis of silicon microneedles for transdermal drug delivery applications. In: The Third International Conference on the Development of Biomedical Engineering in Vietnam: BME2010, 11-14 January, 2010, Ho Chi Minh City, Vietnam. Springer; 2010:84–9. https://doi.org/10.1007/978-3-642-12020-6_21.
Salehi S, Naghib SM, Garshasbi HR, Ghorbanzadeh S, Zhang W. Smart stimuli-responsive injectable gels and hydrogels for drug delivery and tissue engineering applications: a review. Front Bioeng Biotechnol. 2023;11:1104126. https://doi.org/10.3389/fbioe.2023.1104126.
Trinh TA, Le TMD, Nguyen HTT, Nguyen TL, Kim J, Huynh DP, et al. pH-temperature responsive hydrogel-mediated delivery of exendin-4 encapsulated chitosan nanospheres for sustained therapeutic efficacy in type 2 diabetes mellitus. Macromol Biosci. 2023;23:2300221. https://doi.org/10.1002/mabi.202300221.
Li J, Mooney DJ. Designing hydrogels for controlled drug delivery. Nat Rev Mater. 2016;1:1–17. https://doi.org/10.1038/natrevmats.2016.71.
NanoPortalTM Platform Technology - Vivani [Internet]. [cited 2025 Apr 15]. Available from: https://vivani.com/nanoportal/.
Almoshari Y. Osmotic pump drug delivery systems-a comprehensive review. Pharmaceuticals. 2022;15:1430 https://doi.org/10.3390/ph15111430.
Lewis AL, McEntee N, Holland J, Patel A. Development and approval of rybelsus (oral semaglutide): ushering in a new era in peptide delivery. Drug Deliv Transl Res. 2022;12:1–6. https://doi.org/10.1007/s13346-021-01000-w.
Jensen SB, Sauerberg P, Nielsen FS, Pedersen BL, Skibsted E. Tablet formulation comprising semaglutide and a delivery agent. Novo Nordisk AS. 2018. https://lens.org/097-884-899-351-39X.
Twarog C, Fattah S, Heade J, Maher S, Fattal E, Brayden DJ. Intestinal permeation enhancers for oral delivery of macromolecules: a comparison between salcaprozate sodium (SNAC) and sodium caprate (C10). Pharmaceutics. 2019;11:78 https://doi.org/10.3390/pharmaceutics11020078.
Singh G, Krauthamer M, Bjalme-Evans M. Wegovy (semaglutide): a new weight loss drug for chronic weight management. J Investig Med. 2022;70:5–13. https://doi.org/10.1136/jim-2021-001952.
Highlights of Prescribing Information. Ozempic (Semaglutide) Injection, for subcutaneous use. Novo Nordisk. 2017. Available from: https://www.accessdata.fda.gov/drugsatfda_docs/label/2024/209637s032lbl.pdf.
Idrees T, Castro-Revoredo IA, Migdal AL, Moreno EM, Umpierrez GE. Update on the management of diabetes in long-term care facilities. BMJ Open Diab Res Care. 2022;10. https://doi.org/10.1136/bmjdrc-2021-002705.
Frias JP, Bonora E, Nevarez Ruiz L, Li YG, Yu Z, Milicevic Z, et al. Efficacy and safety of dulaglutide 3.0 mg and 4.5 mg versus dulaglutide 1.5 mg in metformin-treated patients with type 2 diabetes in a randomized controlled trial (AWARD-11). Diab Care. 2021;44:765–73. https://doi.org/10.2337/dc20-1473.
Gerstein HC, Colhoun HM, Dagenais GR, Diaz R, Lakshmanan M, Pais P, et al. Dulaglutide and renal outcomes in type 2 diabetes: an exploratory analysis of the REWIND randomised, placebo-controlled trial. Lancet. 2019;394:131–8. https://doi.org/10.1016/S0140-6736(19)31150-X.
Amblee A. Mode of administration of dulaglutide: implications for treatment adherence. Patient Prefer Adherence. 2016;975–82. https://doi.org/10.2147/PPA.S82866.
DeYoung MB, MacConell L, Sarin V, Trautmann M, Herbert P. Encapsulation of exenatide in poly-(D, L-lactide-co-glycolide) microspheres produced an investigational long-acting once-weekly formulation for type 2 diabetes. Diab Technol Ther. 2011;13:1145–54. https://doi.org/10.1089/dia.2011.0050.
Kim D, MacConell L, Zhuang D, Kothare PA, Trautmann M, Fineman M, et al. Effects of once-weekly dosing of a long-acting release formulation of exenatide on glucose control and body weight in subjects with type 2 diabetes. Diab Care. 2007;30:1487–93. https://doi.org/10.2337/dc06-2375.
Kyriacou A, Ahmed AB. Exenatide use in the management of type 2 diabetes mellitus. Pharmaceuticals. 2010;3:2554–67. https://doi.org/10.3390/ph3082554.
Lund A, Knop FK, Vilsbøll T. Glucagon-like peptide-1 receptor agonists for the treatment of type 2 diabetes: differences and similarities. Eur J Intern Med. 2014;25:407–14. https://doi.org/10.1016/j.ejim.2014.03.005.
Del Prato S, Kang J, Trautmann ME, Stewart J, Sorli CH, Derwahl M, et al. Efficacy and safety of once-monthly efpeglenatide in patients with type 2 diabetes: results of a phase 2 placebo-controlled, 16-week randomized dose-finding study. Diab Obes Metab. 2020;22:1176–86. https://doi.org/10.1111/dom.14020.
Frias JP, Choi J, Rosenstock J, Popescu L, Niemoeller E, Muehlen-Bartmer I, et al. Efficacy and safety of once-weekly efpeglenatide monotherapy versus placebo in type 2 diabetes: the AMPLITUDE-M randomized controlled trial. Diab Care. 2022;45:1592–600. https://doi.org/10.2337/dc21-2656.
Pratley RE, Kang J, Trautmann ME, Hompesch M, Han O, Stewart J, et al. Body weight management and safety with efpeglenatide in adults without diabetes: a phase II randomized study. Diab Obes Metab. 2019;21:2429–39. https://doi.org/10.1111/dom.13824.
Rosenstock J, Balas B, Charbonnel B, Bolli GB, Boldrin M, Ratner R, et al. The fate of taspoglutide, a weekly GLP-1 receptor agonist, versus twice-daily exenatide for type 2 diabetes: the T-emerge 2 trial. Diab Care. 2013;36:498–504. https://doi.org/10.2337/dc12-0709.
Raz I, Fonseca V, Kipnes M, Durrwell L, Hoekstra J, Boldrin M, et al. Efficacy and safety of taspoglutide monotherapy in drug-naive type 2 diabetic patients after 24 weeks of treatment: results of a randomized, double-blind, placebo-controlled phase 3 study (T-emerge 1). Diab Care. 2012;35:485–7. https://doi.org/10.2337/dc11-1942.
Bergenstal RM, Forti A, Chiasson JL, Woloschak M, Boldrin M, Balena R. Efficacy and safety of taspoglutide versus sitagliptin for type 2 diabetes mellitus (T-emerge 4 trial). Diab Ther. 2012;3:1–19. https://doi.org/10.1007/s13300-012-0013-8.
fda, cder. HIGHLIGHTS OF PRESCRIBING INFORMATION [Internet]. Available from: www.TANZEUM.com.
American Health Drug Benefits [Internet]. 2015. Albiglutide: A New GLP-1 Receptor Agonist Option. Available from: https://www.ahdbonline.com/drug-updates/tanzeum-albiglutide-a-once-weekly-glp-1-receptor-agonist-subcutaneous-injection-approved-for-the-treatment-of-patients-with-type-2-diabetes.
Mafham M, Preiss D. HARMONY or discord in cardiovascular outcome trials of GLP-1 receptor agonists?. Lancet. 2018;392:1489–90. https://doi.org/10.1016/S0140-6736(18)32348-1.
Kamrul-Hasan ABM, Dutta D, Nagendra L, Bhattacharya S, Singla R, Kalra S. Efficacy and safety of albiglutide, a once-weekly glucagon-like peptide-1 receptor agonist, in patients with type 2 diabetes: a systematic review and meta-analysis. Medicine. 2024;103:e38568 https://doi.org/10.1097/MD.0000000000038568.
Andersen A, Knop FK, Vilsbøll T. A pharmacological and clinical overview of oral semaglutide for the treatment of type 2 diabetes. Drugs. 2021;81:1003–30. https://doi.org/10.1007/s40265-021-01499-w.
Yu YB, Briggs KT, Taraban MB, Brinson RG, Marino JP. Grand challenges in pharmaceutical research series: ridding the cold chain for biologics. Pharm Res. 2021;38:3–7. https://doi.org/10.1007/s11095-021-03008-w.
Aroda VR, Blonde L, Pratley RE. A new era for oral peptides: SNAC and the development of oral semaglutide for the treatment of type 2 diabetes. Rev Endocr Metab Disord. 2022;23:979–94. https://doi.org/10.1007/s11154-022-09735-8.
Lilly investors [Internet]. 2025. Lilly’s oral GLP-1, orforglipron, demonstrated statistically significant efficacy results and a safety profile consistent with injectable GLP-1 medicines in successful Phase 3 trial. Available from: https://investor.lilly.com/news-releases/news-release-details/lillys-oral-glp-1-orforglipron-demonstrated-statistically.
Bao X, Qian K, Yao P. Oral delivery of exenatide-loaded hybrid zein nanoparticles for stable blood glucose control and β-cell repair of type 2 diabetes mice. J Nanobiotechnol. 2020;18:67 https://doi.org/10.1186/s12951-020-00619-0. Available from.
He Y, Huang Y, Xu H, Yang X, Liu N, Xu Y, et al. Aptamer-modified M cell targeting liposomes for oral delivery of macromolecules. Colloids Surf B Biointerfaces. 2023;222:113109. https://doi.org/10.1016/j.colsurfb.2022.113109. Available from.
Shi Y, Sun X, Zhang L, Sun K, Li K, Li Y, et al. Fc-modified exenatide-loaded nanoparticles for oral delivery to improve hypoglycemic effects in mice. Sci Rep. 2018;8:726 https://doi.org/10.1038/s41598-018-19170-y.
Çelik Tekeli M, Yalçın Y, Verdi H, Aktaş Y, Çelebi N. In vitro cellular uptake and insulin secretion studies on INS-1E cells of exendin-4-loaded self-nanoemulsifying drug delivery systems. Pharm Dev Technol. 2024;29. Available from: https://doi.org/10.1080/10837450.2024.2423823.
Zhang L. Tf ligand-receptor-mediated exenatide-Zn(2+) complex oral-delivery system for penetration enhancement of exenatide. J Drug Target. 2018;26:931 https://doi.org/10.1080/1061186X.2018.1455839.
Li Y, Tian H, Zeng H, Zhang Y, Yin T, He H, et al. Chitosan based surface modulation of core-shell nanoparticles for oral delivery of exenatide via balancing mucus penetration and cellular uptake. Int J Pharm. 2025;672:125319. https://doi.org/10.1016/j.ijpharm.2025.125319.
Kim JY, Lee H, Oh KS, Kweon S, Jeon Ocheol, Byun Y, et al. Multilayer nanoparticles for sustained delivery of exenatide to treat type 2 diabetes mellitus. Biomater. 2013;34:8444–9. https://doi.org/10.1016/j.biomaterials.2013.07.040.
Huang Y, Wang X, Li W, Yue F, Wang M, Zhou F. Exenatide-modified deferoxamine-based nanoparticles ameliorates neurological deficits in Parkinson’s disease mice. Int J Nanomed. 2024;19:10401–14. https://doi.org/10.2147/IJN.S479670.
Uhl P, Bajraktari-Sylejmani G, Witzigmann D, Bay C, Zimmermann S, Burhenne J, et al. A nanocarrier approach for oral peptide delivery: evaluation of cell-penetrating-peptide-modified liposomal formulations in dogs. Adv Ther [Internet]. 2023;6:2300021. https://doi.org/10.1002/adtp.202300021.
Herbster L, Pander G, Böhmann M, Eisenzapf T, Wohlfart S, Mühlberg E, et al. Liposomes containing tetraether lipids and cell-penetrating peptides as platform technology for oral delivery of peptides: a comparative study on GLP-1 Analogs. Nano Sel. 2025. https://doi.org/10.1002/nano.70010.
Ismail R, Sovány T, Gácsi A, Ambrus R, Katona G, Imre N, et al. Synthesis and statistical optimization of poly (Lactic-Co-Glycolic Acid) nanoparticles encapsulating glp1 analog designed for oral delivery. Pharm Res. 2019;36. https://doi.org/10.1007/s11095-019-2620-9.
Senduran N, Yadav HN, Vishwakarma VK, Bhatnagar P, Gupta P, Bhatia J, et al. Orally deliverable nanoformulation of liraglutide against type 2 diabetic rat model. J Drug Deliv Sci Technol. 2020;56:101513. https://doi.org/10.1016/j.jddst.2020.101513.
Presas E, Tovar S, Cuñarro J, O’Shea JP, O’Driscoll CM. Pre-clinical evaluation of a modified cyclodextrin-based nanoparticle for intestinal delivery of liraglutide. J Pharm Sci. 2021;110:292–300. https://doi.org/10.1016/j.xphs.2020.10.058.
Subedi L, Bamjan AD, Phuyal S, Shim JH, Cho SS, Seo JB, et al. An oral liraglutide nanomicelle formulation conferring reduced insulin-resistance and long-term hypoglycemic and lipid metabolic benefits. J Controlled Release. 2025;378:637–55. https://doi.org/10.1016/j.jconrel.2024.12.039.
Kim GL, Song JG, Han HK. Enhanced oral efficacy of semaglutide via an ionic nanocomplex with organometallic phyllosilicate in type 2 diabetic rats. Pharmaceutics. 2024;16:886. https://doi.org/10.3390/pharmaceutics16070886
Domingues I, Yagoubi H, Zhang W, Marotti V, Kambale EK, Vints K, et al. Effects of semaglutide-loaded lipid nanocapsules on metabolic dysfunction-associated steatotic liver disease. Drug Deliv Transl Res. 2024;14:2917–29. https://doi.org/10.1007/s13346-024-01576-z.
Wang B, Su Z, Kuang M, Luo Y, Xu M, Sun M, et al. Chitosan hydrochloride coated and nonionic surfactant modified niosomes: a better way for oral administration of semaglutide. Biomed Mater. 2025;20:035001 https://doi.org/10.1088/1748-605X/adb2cf.
Pinto S, Viegas J, Cristelo C, Pacheco C, Barros S, Buckley ST, et al. Bioengineered nanomedicines targeting the intestinal fc receptor achieve the improved glucoregulatory effect of semaglutide in a type 2 diabetic mice model. ACS Nano. 2024;18:28406–24. https://doi.org/10.1021/acsnano.4c11172.
Izadi R, Arkan E, Jalalvand AR, Akbari V. Multivariate analyses to develop a novel drug delivery system: trying to expanding the system to bio-sensing of the human serum albumin. Sens Biosensing Res. 2022;36. 100489. https://doi.org/10.1016/j.sbsr.2022.100489.
Sandmeier M, Ricci F, To D, Lindner S, Stengel D, Schifferle M, et al. Design of self-emulsifying oral delivery systems for semaglutide: reverse micelles versus hydrophobic ion pairs. Drug Deliv Transl Res. 2024; Available from: https://doi.org/10.1007/s13346-024-01729-0.
Xiao P, Wang H, Liu H, Yuan H, Guo C, Feng Y, et al. Milk exosome-liposome hybrid vesicles with self-adapting surface properties overcome the sequential absorption barriers for oral delivery of peptides. ACS Nano. 2024;18:21091–111. https://doi.org/10.1021/acsnano.4c02560.
Zhang Y, Han J, Wu W, Dang B. Oral delivery of semaglutide and tirzepatide using milk-derived small extracellular vesicles. 2025; Available from: https://doi.org/10.1101/2024.12.28.630566.
Dong N, Zhu C, Jiang J, Huang D, Li X, Quan G, et al. Development of composite PLGA microspheres containing exenatide-encapsulated lecithin nanoparticles for sustained drug release. Asian J Pharm Sci. 2020;15:347–55. https://doi.org/10.1016/j.ajps.2019.01.002.
Chen C, Zheng H, Xu J, Shi X, Li F, Wang X. Sustained-release study on Exenatide loaded into mesoporous silica nanoparticles: in vitro characterization and in vivo evaluation. DARU J Pharm Sci. 2017;25:20 https://doi.org/10.1186/s40199-017-0186-9.
Lu Y, Wu L, Lin M, Bao X, Zhong H, Ke P, et al. Double layer spherical nanoparticles with hyaluronic acid coating to enhance oral delivery of exenatide in T2DM rats. Eur J Pharm Biopharm. 2023;191:205–18. https://doi.org/10.1016/j.ejpb.2023.09.003.
Abeer MM, Meka AK, Pujara N, Kumeria T, Strounina E, Nunes R, et al. Rationally designed dendritic silica nanoparticles for oral delivery of exenatide. Pharmaceutics. 2019;11. Available from: https://doi.org/10.3390/pharmaceutics11080418.
Wang Y, Song X, Zhuang L, Lang H, Yu L, Yan X, et al. Kinetic control of Phytic acid/Lixisenatide/Fe (III) ternary nanoparticles assembly process for sustained peptide release. Int J Pharm. 2022;611:121317. https://doi.org/10.1016/j.ijpharm.2021.121317.
Qian F, Mathias N, Moench P, Chi C, Desikan S, Hussain M, et al. Pulmonary delivery of a GLP-1 receptor agonist, BMS-686117. Int J Pharm. 2009;366:218–20. https://doi.org/10.1016/j.ijpharm.2008.10.020.
Shi S, Shuang S, Xiangdong L, Guoqiang Z, Feng D, Wenshuang Z, et al. Construction and performance of exendin-4-loaded chitosan-PLGA microspheres for enhancing implant osseointegration in type 2 diabetic rats. Drug Deliv. 2022;29:548–60. https://doi.org/10.1080/10717544.2022.2036873.
Tian H, Chang M, Lyu Y, Dong N, Yu N, Yin T, et al. Intramuscular injection of palmitic acid-conjugated Exendin-4 loaded multivesicular liposomes for long-acting and improving in-situ stability. Expert Opin Drug Deliv. 2024;21:169–85. https://doi.org/10.1080/17425247.2024.2305110.
Tang Y, Arbaugh B, Park H, Scher HB, Bai L, Mao L, et al. Targeting enteric release of therapeutic peptides by encapsulation in complex coacervated matrix microparticles by spray drying. J Drug Deliv Sci Technol. 2023;79:104063. https://doi.org/10.1016/j.jddst.2022.104063.
Jiang W, Wang Q, Zhang X, Yang H, Yang X. Semaglutide sustained-release microspheres with single-phase zero-order release behavior: Effects of polymer blending and surfactants. J Drug Deliv Sci Technol. 2025;107:106793. https://doi.org/10.1016/j.jddst.2025.106793.
Maharjan R, Lim DS, Baik HJ, Park HE, Kim MS, Kim KH, et al. Preparation of semaglutide long-acting injectable microcapsules with physicochemical properties, long-term stability, and pharmacokinetics and pharmacodynamics. J Pharm Investig. 2024;54:667–81. https://doi.org/10.1007/s40005-024-00681-y.
Zeng H, Song J, Li Y, Guo C, Zhang Y, Yin T, et al. Effect of hydroxyethyl starch on drug stability and release of semaglutide in PLGA microspheres. Int J Pharm. 2024;654:123991. https://doi.org/10.1016/j.ijpharm.2024.123991.
Xu B, Liu H, Yang G, Zhang S, Zhou Z, Gao Y. Novel double-layered PLGA microparticles-dissolving microneedle (MPs-DMN) system for peptide drugs sustained release by transdermal delivery. Int J Pharm. 2025;670:125128. https://doi.org/10.1016/j.ijpharm.2024.125128.
Liu H, Zhang S, Zhou Z, Xing M, Gao Y. Two-layer sustained-release microneedles encapsulating exenatide for type 2 diabetes treatment. Pharmaceutics. 2022;14. https://doi.org/10.3390/pharmaceutics14061255.
You J, Juhng S, Song J, Park J, Jang M, Kang G, et al. Egg microneedle for transdermal delivery of active liraglutide. Adv Health Mater. 2023. https://doi.org/10.1002/adhm.202202473.
Rabiei M, Kashanian S, Bahrami G, Derakhshankhah H, Barzegari E, Samavati SS, et al. Dissolving microneedle-assisted long-acting Liraglutide delivery to control type 2 diabetes and obesity. Eur J Pharm Sci. 2021;167:106040. https://doi.org/10.1016/j.ejps.2021.106040.
Chen W, Wainer J, Ryoo SW, Qi X, Chang R, Li J, et al. Dynamic omnidirectional adhesive microneedle system for oral macromolecular drug delivery. Sci Adv. 2025;8:eabk1792. https://doi.org/10.1126/sciadv.abk1792.
Chen Y, Li Y, Shen W, Li K, Yu L, Chen Q, et al. Controlled release of liraglutide using thermogelling polymers in treatment of diabetes. Sci Rep. 2016;6. https://doi.org/10.1038/srep31593.
Zhuang Y, Yang X, Li Y, Chen Y, Peng X, Yu L, et al. Sustained release strategy designed for lixisenatide delivery to synchronously treat diabetes and associated complications. ACS Appl Mater Interfaces. 2019;11:29604–18. https://doi.org/10.1021/acsami.9b10346.
Harris E. September 2024 therapeutic delivery: industry update. Ther Deliv. 2025;16:1–8. https://doi.org/10.1080/20415990.2024.2442298.
Klotsman M, Anderson WH, Gilor C. Drug release profile of a novel exenatide long-term drug delivery system (OKV-119) administered to cats. BMC Vet Res. 2024;20:211 https://doi.org/10.1186/s12917-024-04051-6.
Klotsman M, Adin CA, Anderson WH, Gilor C. Safety, tolerability, and proof-of-concept study of OKV-119, a novel exenatide long-term drug delivery system, in healthy Cats. Front Vet Sci. 2021;8. https://doi.org/10.3389/fvets.2021.661546.
Rosenstock J, Buse JB, Azeem R, Prabhakar P, Kjems L, Huang H, et al. Efficacy and safety of ITCA 650, a novel drug-device GLP-1 receptor agonist, in type 2 diabetes uncontrolled with oral antidiabetes drugs: the FREEDOM-1 Trial. Diab Care. 2017;41:333–40. https://doi.org/10.2337/dc17-1306.
Zangabad PS, Vasani RB, Tong Z, Anderson D, Rudd D, Hick CA, et al. Porous silicon microparticles enable sustained release of GLP-1R agonist peptides for the treatment of Type 2 diabetes. Adv Ther. 2024;7:2400136. https://doi.org/10.1002/adtp.202400136.
Wu J, Williams GR, Branford-White C, Li H, Li Y, Zhu LM. Liraglutide-loaded poly (lactic-co-glycolic acid) microspheres: preparation and in vivo evaluation. Eur J Pharm Sci. 2016;92:28–38. https://doi.org/10.1016/j.ejps.2016.06.018.
Schneider EL, Hangasky JA, Fernández RdelV, Ashley GW, Santi DV. The limitation of lipidation: conversion of semaglutide from once-weekly to once-monthly dosing. Proc Natl Acad Sci USA. 2024;121:e2415815121. https://doi.org/10.1073/pnas.2415815121.
Zhu S, Zhang B, Wang Y, He Y, Qian G, Deng L, et al. A bilayer microneedle for therapeutic peptide delivery towards the treatment of diabetes in db/db mice. J Drug Deliv Sci Technol. 2021;62:102336. https://doi.org/10.1016/j.jddst.2021.102336.
Lin H, Liu J, Hou Y, Yu Z, Hong J, Yu J, et al. Microneedle patch with pure drug tips for delivery of liraglutide: pharmacokinetics in rats and minipigs. Drug Deliv Transl Res. 2025;15:216–30. https://doi.org/10.1007/s13346-024-01582-1.
Li QX, Gao H, Guo YX, Wang BY, Hua R, Gao L, et al. GLP-1 and underlying beneficial actions in Alzheimer’s disease, hypertension, and NASH. Front Endocrinol. 2021;12:721198 https://doi.org/10.3389/fendo.2021.721198.
Author information
Authors and Affiliations
Contributions
RVP and HA were responsible for conceptualization, methodology, writing the original draft, and contributing to critical revision. RV and KJ contributed to writing, reviewing and editing the manuscript, as well as validation and visualization. PG and RVP were responsible for review administration, supervision, and critical revision. RV contributed to image curation and visualization. All authors reviewed subsequent revisions and provided references.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Peri, R.V., Anchan, H., Jonnalagadda, K. et al. Designing GLP-1 delivery: structural perspectives and formulation approaches for optimized therapy. Nutr. Diabetes 15, 53 (2025). https://doi.org/10.1038/s41387-025-00397-4
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41387-025-00397-4



![Fig. 1: Origin of GLP-1, obtained from Li et al. 2021 [126], under the Creative Commons Attribution License (CC BY) License.](http://media.springernature.com/lw685/springer-static/image/art%3A10.1038%2Fs41387-025-00397-4/MediaObjects/41387_2025_397_Fig1_HTML.png)





