Abstract
Background
Few studies evaluated the effect of different proportions of dietary plant- and animal-based protein on metabolic syndrome (MetS) and its related biomarkers.
Objective
Considering the effect of various types of dietary sources of protein on metabolic health and inconsistent results, this study sought to examine the impact of partially substituting animal protein with plant protein in the diet on the components of MetS, atherogenic index of plasma, and serum adropin values.
Methods
In this parallel, randomized clinical trial with two arms, 73 participants with MetS were randomly allocated to one of two slightly calorie-restricted intervention diets with different proportions of protein sources, including a plant-based protein diet (70% plant-based protein and 30% animal-based protein) and an animal-based protein diet (30% plant-based protein and 70% animal-based protein) for 10 weeks. All analyses were performed based on both intention-to-treat and per-protocol principles.
Results
Mean age in the plant and animal-based protein groups was 44.0 ± 9.8 and 43.9 ± 9.8 years, respectively. Within each group, weight, body mass index, systolic and diastolic blood pressure, and atherogenic index of plasma significantly decreased, and adropin levels significantly increased after the intervention (P < 0.05). However, waist circumference (WC) and triglyceride (TG) levels showed a significant decrease only in the plant protein group, and high-density lipoprotein cholesterol (HDL-c) levels illustrated a significant increase only in the animal protein group (P < 0.05). Between-group differences in both crude and adjusted models did not show any significant changes between the two intervention arms (P > 0.05). However, findings of per-protocol analyses illustrated a significant difference only in mean adropin values in two study arms.
Conclusion
Our findings revealed that both diets based on plant and animal protein were associated with improved in anthropometric indices, MetS components, atherogenic index of plasma, and elevated serum adropin levels. Nevertheless, no statistically significant difference was identified between the two groups.
Similar content being viewed by others
Introduction
Metabolic syndrome (MetS), as a worldwide public health concern, is related to a cluster of disorders, including abdominal obesity, impaired fasting blood glucose (FBG), dyslipidemia (low high-density lipoprotein cholesterol (HDL-c) and high triglycerides (TG) levels), and high blood pressure [1]. This syndrome is closely linked to an increased risk of non-communicable diseases, including cardiovascular diseases, type 2 diabetes mellitus (T2DM), renal failure, cancers, as well as mortality [2]. The global prevalence of MetS in adults is in the range of 12.5 to 31.4% based on different applied diagnostic criteria, and it has gradually increased in many countries [3].
Recently, the discovery of novel biomarkers, including adropin and the atherogenic index of plasma (AIP), has opened a new era in the diagnosis and management of MetS [4, 5]. Serum adropin levels serve as a significant indicator for predicting metabolic state and represent a recently identified peptide that is crucial for maintaining energy homeostasis, obesity, insulin resistance, and nutrient intake [6, 7]. It has been involved in a variety of biological processes and is regulated by carbohydrates and lipids intake [8]. Previous studies suggested that low circulating levels of adropin can elevate the risk of obesity, insulin resistance, MetS, and T2DM [9, 10]. Another index that is related to MetS and obesity is AIP, which is a logarithmically transformed ratio of concentrations of TG to HDL-c [11]. The AIP exhibits a heightened sensitivity in capturing the interaction between atherogenic lipoproteins and protective lipoproteins compared to other atherogenic indices. [5]. Previous investigations revealed that AIP was significantly connected to waist circumference (WC) and body mass index (BMI) [12]. Moreover, elevated AIP levels lead to a higher risk of MetS, diabetes, and high blood pressure [13, 14].
Several modifiable lifestyle factors, especially dietary nutrient content, may have a critical role in the prevalence of MetS, its components, and related biomarkers [15, 16]. In addition to multiple dietary approaches to prevent MetS, the percentage and type of macronutrients could affect the management of MetS and its biomarkers [17, 18]. Among macronutrients, the association of dietary protein with MetS and its related components and biomarkers has received little consideration. Therefore, it is important to examine the effect of partial substitution of dietary animal protein with plant protein on cardiometabolic health, MetS components, atherogenic index of plasma, and adropin.
There are multiple possible reasons for the varying association between animal- and plant-based protein and cardiometabolic risk factors. One potential explanation is that the distinct amino acid compositions found in animal- and plant-based protein sources may account for different associations [19]. Previous evidence has shown that methionine, an essential amino acid, and its metabolic byproduct, homocysteine, may play a role in the progression of chronic diseases, MetS, and its components [20, 21]. On the other hand, glycine, which is abundant in plant-based proteins, is effective in treating metabolic disorders, inflammatory diseases, obesity, diabetes, and cancers [22]. Another explanation is that the various associations of animal-based and plant-based protein foods with cardiometabolic risk factors may be due to additional nutrients in these foods. Plant-based protein sources like nuts, legumes, and whole grains have lower levels of saturated fat but higher levels of monounsaturated and polyunsaturated fats, fiber, polyphenols, antioxidants, and other biologically active compounds. These compounds could have a beneficial effect on metabolic syndrome and its components [23, 24]. In 2021, we conducted a cross-sectional study on a sample of Iranian adults to evaluate the associations between a plant-based diet and MetS, high AIP level, and adropin values [25]. The usual diet of these adult participants consisted of 30% plant protein and 70% animal-based. Our findings showed that more adherence to an unhealthy plant-based diet could increase the odds of MetS, while a healthy plant-based diet could decrease high AIP levels. As cross-sectional investigations could not establish causality, a randomized clinical trial was required to affirm such findings. Due to few number of interventions that assessed the effect of animal and plant protein on MetS and its components, as well as the lack of studies that have investigated this effect on novel biomarkers of MetS, including adropin and AIP in a clinical trial manner, we aimed to conduct a randomized clinical trial in adults with MetS to examine the effect of partial replacement of animal protein with plant protein in the diet on components of MetS, adropin, and atherogenic index of plasma. We hypothesized that substitution of animal protein with plant protein would provide a better metabolic status, higher levels of adropin, and lower levels of AIP.
Materials and Methods
Participants
The current intervention study was a parallel randomized clinical trial. This study was carried out as a part of a representative population-based cross-sectional study, which was carried out on adults in Isfahan, Iran, in 2021 [25]. A total of 151 participants with MetS, as determined in the previous cross-sectional study, were asked to take part in this intervention study. According to the Joint Interim Statement (JIS) criteria [26], at least three of the following factors were necessary for a diagnosis of MetS: abdominal obesity (WC ≥ 94 cm in men and ≥80 cm in women), high TG levels (TG ≥ 150 mg/dl (1.7 mmol/L) or on drug treatment for high triglycerides), high blood pressure (systolic blood pressure (SBP) ≥ 130 mmHg and/or diastolic blood pressure (DBP) ≥ 85 mmHg or on antihypertensive drug treatment in participants with hypertension), impaired FBG (FBG ≥ 100 mg/dl or on drug treatment for high FBG), and low HDL-c levels (HDL-c < 40 mg/dl (1.03 mmol/L) in men and <50 mg/dl (1.3 mmol/L) in women). Participants were teachers aged 20 to 55 years working in Isfahan with a diagnosis of metabolic syndrome based on JIS criteria. Inclusion criteria included a body mass index (BMI) between 25 and 35 kg/m² and no adherence to any special diet within the past 6 months. Participants were excluded if they used dietary supplements or had a history of kidney, liver, cardiovascular, thyroid, or cancer diseases. Additional exclusion criteria were use of hormonal drugs (including birth control pills and glucocorticoids), use of medications that could affect blood sugar, blood pressure, or lipid control, being pregnant or lactating, drinking alcohol, or smoking cigarettes or tobacco products. All participants provided informed consent and expressed a desire to participate in the study. Finally, 73 participants with MetS were included at the baseline of the study. A participant’s flow diagram is illustrated in Fig. 1. The study received approval from the ethics committee at Isfahan University of Medical Sciences. (No: IR.MUI.RESEARCH.REC.1400.370; 2021/12/04) and was registered on the Iranian Registry of Clinical Trials website (www.irct.ir) (No: IRCT20150909023957N10, 2022/05/16). Informed consent was acquired in written form from all participants.
The study participant flow chart.
Study design
This was a parallel study with two arms following two types of diets with different proportions of protein sources, including a plant-based protein diet (70% plant-based protein and 30% animal-based protein) and an animal-based protein diet (30% plant-based protein and 70% animal-based protein). The primary outcomes were adropin levels, MetS components, and probability of having high AIP levels. Participants were randomly allocated to 1 of 2 intervention diets for 10 weeks. This time frame of 10 weeks was chosen based on previous evidence indicating that 8–12 weeks of lifestyle and dietary interventions could lead to significant improvements in MetS components [27, 28]. This duration could provide a balance between allowing enough time to observe measurable changes or improvements and ensuring participants’ adherence and feasibility of the study. Random allocation was performed based on a permuted block design with block sizes of 4, using a computer random number generator. On account of the nature of dietary intervention, it was not possible to implement blinding for either the participants or the researchers; however, statisticians were blinded to the treatment assignment. The intervention was only in the form of nutritional and dietary recommendations, and food items were not given to the subjects. These recommendations were personalized for each participant and were designed for each individual by an expert dietician. Participants were visited at the research site every 2 weeks during 10 weeks of the intervention.
Participants were instructed to maintain their regular physical activity levels and to refrain from using any medications or supplements that could potentially influence the study outcomes during the 10-week intervention period. Physical activity was recorded for two days during the study. Dietary intake was evaluated using a 3-day diet record. In addition, diet compliance was evaluated using a 3-day diet record every two weeks. To decrease recall bias and reliance on memory, participants recorded their food intake at the time of food consumption. The food intake (g/day) was analyzed using Nutritionist IV software (Version 7; N-squared computing, OR, USA) to calculate total energy and nutrient intake.
Sample size
Due to a lack of studies assessing the effect of partial replacement of animal-based protein with plant-based protein on adropin levels (as the main outcome), the sample size calculation method for a pilot study was applied to calculate sample size (by considering a power of 90%, two-sided 5% significance, and having a standardized small effect size). According to this method [29, 30], 25 participants were at least considered for each treatment arm based on the minimum standardized effect size.
Dietary interventions
Two types of dietary plans with different proportions of plant- and animal-based protein were designed. The first was a plant-based protein diet consisting of 70% plant-based protein and 30% animal-based protein. The plant sources of protein were mainly from whole grains, soy protein, legumes, nuts, and seeds. The second diet was an animal-based protein diet containing 30% plant-based protein and 70% animal-based protein. This diet was based on animal sources of protein, including low-fat meat, poultry, fish, eggs, and low-fat dairy products. Details of intervention diets are explained in Supplementary Table 1. Daily calorie requirement was estimated individually using the 2002 Institute of Medicine, Food and Nutrition Board dietary reference intakes for energy in overweight and obese participants [31]. Both groups were instructed to slightly restrict energy intake by 500 Kcal/day. Both diets provided 20% of the total daily energy requirement from protein. In addition, carbohydrate and fat intakes were respectively designed to be 50% and 30% of total daily energy requirements in both diets [32].
Anthropometric, blood pressure, and biochemical assessment
Anthropometric and biochemical measurements were made at baseline and after intervention. Weight measurements were conducted using the body composition analyzer (Tanita MC-780MA, Tokyo, Japan) with an accuracy of 0.01 kg, while the participants were in a fasting condition and light clothing without shoes. Height was assessed using a tape measure affixed to the wall, recorded to the nearest 0.1 cm, with participants positioned in a natural shoulder alignment and standing without shoes. Waist circumference was assessed with an unstretched tape measure at the narrowest point between the top of the iliac crest and the lower margin of the last palpable rib. BMI was calculated as weight (kg)/[height (m)]2.
Arterial blood pressure (BP) was evaluated twice using a digital sphygmomanometer (OMRON, M3, HEM-7154-E, Japan), with a 0.5 mmHg accuracy, on the left arm, after at least 5-min rest in a sitting position. The mean of the first and second measurements was used for the blood pressure report. After an overnight fast (>12 h), a 10-ml peripheral blood sample was taken using venipuncture from each participant before and after the intervention. Serum was separated by a 10-minute centrifuge at 1500 g. The levels of FBG and lipid profiles, including serum concentrations of total cholesterol, HDL-c, low-density lipoprotein cholesterol (LDL-c), and TG, were measured using an enzymatic colorimetric method with the BioSystem Kit and the Biosystem A15 auto-analyzer. AIP was computed as a logarithmic transformation of the ratio of TG to HDL-c [33]. Serum adropin levels were quantified utilizing the ELISA kit developed by ZellBio GmbH (Germany), employing the biotin double antibody sandwich methodology. The assay demonstrated a detection range for adropin from 30 to 960 pg/mL, with a sensitivity of 4 pg/mL. The coefficients of variation for intra-assay and inter-assay reproducibility were found to be less than 10% and 12%, respectively.
Statistical analysis
The normal distribution of continuous variables was evaluated using the Kolmogorov–Smirnov test. Data were illustrated as mean (SD) for quantitative variables and frequency (percentage) for qualitative variables. Independent samples t-tests were employed to compare baseline characteristics and nutrient intakes for continuous variables between the two groups, while Pearson’s chi-square tests were applied for categorical variables. A paired samples t-test was applied to assess within-group changes of variables during 10 weeks of intervention. An independent samples t-test was applied to assess differences between two intervention groups for continuous variables. Between-group differences for any variables at the end of the intervention were evaluated using analysis of covariance (ANCOVA). In ANCOVA models, the baseline value of each variable was used as a covariate. In addition, age and gender were considered as other covariates. All analyses were performed based on both per-protocol and intention-to-treat (ITT) principles. For per-protocol analysis, participants who completed the study protocol were included in the analyses. For ITT, the last-observation-carried-forward method was used to treat missing values. SPSS software version 20 (IBM, Chicago, IL) was used to conduct all analyses, and a p-value of less than 0.05 (two-tailed) was deemed statistically significant.
Results
Among individuals with MetS in the cross-sectional phase, 73 participants agreed to take part in this randomized clinical trial. At the end of the trial, 52 participants were analyzed in the per-protocol method. By replacing the missing data with the last observed value, all 73 participants were analyzed in the ITT method. Among 73 patients with MetS, 21 participants (n = 10 in the plant-based protein diet, and n = 11 in the animal-based protein diet) were excluded due to pregnancy (n = 1), Coronavirus infection (n = 14), low compliance rate (n = 2), and decline to continue (n = 4). The details of the follow-up process are summarized in Fig. 1.
Demographic characteristics and baseline cardio-metabolic factors of 73 study participants are displayed in Table 1. Mean age in the plant and animal-based protein groups was respectively 44 ± 9.8 and 43.9 ± 9.8 years, with 45.9% and 50% of men. A significantly higher HDL-c (52.65 vs. 47.49, P = 0.01) and lower AIP (0.49 vs. 0.57, P = 0.02) were found in the plant-based protein group. There were no significant differences in other cardio-metabolic factors and anthropometric indices. Moreover, after per-protocol analysis of 52 participants, only HDL-c levels were significantly higher in the plant protein compared to the animal protein group (53.44 vs. 47.58, P = 0.02), as shown in Supplementary Table 2.
Daily dietary intakes of participants at baseline and end of intervention are presented in Table 2. There were no significant differences between the two groups in terms of energy or macronutrient intakes at baseline. During 10 weeks of intervention, the amounts of fiber intake (20.96 vs. 12.74 g/day, P < 0.001), polyunsaturated fatty acid (22.67 vs. 19.36 g/day), magnesium (375.55 vs. 324.87 mg/day), and thiamin (1.58 mg vs.1.27 mg/day) in the plant protein group were significantly higher than the animal protein group. Also in this group, elevation of plant protein intake (79.55% vs. 31.85%, P < 0.001) was significant that showing great adherence of patients to the prescribed diets. However, intakes of cholesterol, saturated fatty acids, monounsaturated fatty acids, calcium, riboflavin, and niacin were higher in the animal-based protein group (P < 0.05).
For physical activity levels, no significant difference was found between the plant-based protein (35.99 ± 5.26 MET-h/day) and animal-based protein groups (38.51 ± 6.04 MET-h/day) throughout the intervention period (P = 0.12).
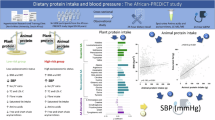
Table 3 and Fig. 2 illustrate the effect of partial animal-based protein replacement in the diet with plant-based protein on anthropometric indices, MetS components, adropin, and AIP (by intention-to-treat analyses, n = 73). Within-group analyses revealed that weight, BMI, systolic and diastolic blood pressure, and AIP were significantly decreased, and adropin levels were significantly increased after the intervention in both groups compared to the baseline values (P < 0.05). While WC and TG in the plant protein group showed a significant decrease compared to the baseline values, there was no significant difference in the other group. However, higher HDL-c levels just in the animal protein group after 10 weeks. Results of between-group analyses and analysis of covariance (with adjustments for baseline values, age, and sex) showed no statistically significant difference between the two intervention groups in terms of the investigated indicators (P < 0.05). Per-protocol analysis on 52 participants illustrated a significant difference only in terms of mean adropin levels in both study groups, as depicted in Supplementary Table 3.
Schematic summary of the effect of partial animal-based protein replacement in the diet with plant-based protein on anthropometric indices, MetS components, adropin, and AIP (obtained from intention-to-treat analyses, n = 73).
Discussion
The present study was the first randomized clinical trial that investigated the effect of partial replacement of animal-based with plant-based protein in diets with a slight energy restriction on AIP and adropin levels among adults with MetS. Our findings revealed about 2.5% weight loss in both study groups. Moreover, BMI, AIP, and systolic and diastolic blood pressure after intervention decreased in both groups, showing that neither of the two regimes was superior to the other. A reduction in TG levels and waist circumference in the plant-based protein group and an elevation of HDL-c levels in the animal-based protein group after 10 weeks of intervention were significantly found. A significant increase in adropin levels was observed in both groups. However, no statistically significant difference was seen between the two study groups.
In the current intervention, the percentage of protein was considered as 20%, meanwhile participants’ diet at baseline indicated that about 13% of their energy intake came from protein; this enhancement in protein intake might have increased satiety and preserved lean body mass during weight loss [34]. Notably, both intervention groups in the present trial experienced an average weight loss of approximately 2.5% over the 10 weeks. The observed weight loss was most likely attributable to the prescribed 500 kcal/day energy restriction in both arms of the intervention. Previous studies showed that a high-protein diet (HPD) could result in weight loss. A meta-analysis of clinical trials revealed that HPD (25–30% of energy) could cause weight loss and improved MetS components, regardless of the source of protein intake, in comparison to a standard diet (12–18%) [35]. Moreover, Ma et al. have indicated that the impact of HPD on weight reduction, body fat, and blood lipid profiles surpasses that of calorie-restricted diets or intermittent fasting [36]. The underlying mechanism might be an increase in satiety and energy expenditure. Increased satiety might be through elevations in anorexigenic hormones, diet-induced thermogenesis (DIT), blood amino acid levels, hepatic gluconeogenesis, ketogenesis, and decreased levels of orexigenic hormones. Protein is known to increase energy expenditure by a higher increment in DIT than carbohydrates and fat and to prevent the reduction of fat-free mass [37].
We found that both types of studied diets improved anthropometric indices and MetS components, without any significant difference between the two groups. Hill et al. examined the effect of two types of diet (based on plant and animal protein) and indicated a 5% reduction in weight and improvement in MetS components in two groups, without any significant difference between them [38]. The results of the mentioned study were consistent with ours. These findings showed that weight loss, as a result of a slight energy restriction, could play the main role in MetS improvement, regardless of the source of protein intake. In other words, heart-healthy weight-loss dietary patterns based on animal or plant-based protein could improve components of MetS similarly.
The European Prospective Investigation into Cancer and Nutrition (EPIC) study reported that consuming protein from either plants or animals did not show significant associations with the development of CVDs, ischemic heart disease, or stroke, after considering confounders in analyses [39]. In addition, a study by Sawyer et al. reported that consuming plant-based protein was linked to a reduced risk of MetS, whereas animal-based protein did not have a significant effect on MetS; so that, this research did not suggest reducing animal-based protein intake [40]. A review of the effects of various types of protein on components of MetS and risk factors of CVDs revealed that multiple studies, both observational and intervention-based, have attempted to determine whether plant-based and animal-based proteins have different effects on reducing risk factors for CVDs. However, the current evidence is inconclusive and is likely to remain so, due to challenges in isolating the specific effects of individual proteins. It is important to consider the influence of other components of plant vs. animal foods and the overall dietary pattern [41]. The underlying factors contributing to the specific impacts of plant and animal proteins are multifaceted and diverse; different amino acid composition of these foods, presence of other non-protein compounds in the whole diet, and interactions with gut microbiome may be involved [41,42,43,44,45]. To lower the MetS and CVDs risk factors, previous research suggested that diets emphasizing protein-rich plant foods and incorporating some animal-based protein sources that are low in saturated fat and unprocessed foods, including seafood, poultry, lean meats, low-fat dairy, and eggs, instead of refined carbohydrates and processed meats, might be beneficial [41, 46].
In the current trial, in both intervention arms, participants were advised to consume low-fat dairy products, to reduce their total energy intake by approximately 500 kcal, and to increase their dietary protein to about 20% of total energy. Due to having different sources of protein in groups, notable differences emerged in the consumption of saturated fats and other macronutrients, which may have influenced the components of MetS, adropin, and AIP. Those in the plant-based protein group had a significantly lower intake of saturated fatty acids (5.28% of energy or 11.30 g/day) compared to the animal-based protein group (7.80% of energy or 15.21 g/day; P < 0.001). Additionally, the plant-based group (vs. animal-based protein group) had a higher dietary fiber intake (20.96 vs. 12.74 g/day; P < 0.001) and a lower cholesterol intake (110.79 vs. 290.98 g/day; P < 0.001). However, during 10 weeks of intervention, the intake of total fat, carbohydrate, and protein was similar in both groups. Recent dietary guidelines recommend reducing saturated fat intake to ≤10% of total daily energy, as more than 10% saturated fat intake is linked to increased LDL cholesterol and a higher risk of atherosclerotic cardiovascular disease (ASCVD) [47]. However, another study revealed that the unhealthy effects of saturated fat might rely on the total dietary context, including carbohydrate intake and food sources of saturated fat [48]. For example, even though whole-fat dairy products and unprocessed meats are rich in saturated fat, their consumption as part of an overall balanced diet does not consistently raise the risk of cardiovascular disease as part of a balanced diet [48].
Epidemiological evidence on the relationship between animal protein intake, particularly red meat intake, and risk of cardiovascular diseases and components of MetS is inconsistent. The study by Chung et al. assessed the association of animal and plant protein intake with MetS and its components in men and women, separately. Their report illustrated that a higher intake of animal-based protein increased the MetS risk factors in men. However, the intake of both animal and plant proteins did not show a significant association with any MetS risk factors in women [49]. One of the reasons for the various effects of animal protein consumption on metabolic parameters in men and women could be related to the different ratios of dietary intake of animal and plant protein. In other words, women consumed a greater proportion of plant-based protein compared to animal-based protein, while men had a higher proportion of animal-based protein that was similar to Western diets. Also, the association between the ratio of animal to plant dietary protein and MetS and its components was investigated in the cross-sectional study [50]. The findings showed that a higher ratio of animal to plant protein intake was linked to components of MetS, such as increased waist circumference and fasting blood glucose levels. Nevertheless, the consumption of plant protein did not appear to have any impact on glucose regulation or body composition [50]. In addition, a study by Vasei et al. evaluated the effects of substituting plant-based- with animal-based proteins in the DASH diet on components of MetS in participants with obesity. The results revealed that the plant-based DASH diet has greater effects on reducing blood sugar and systolic blood pressure, with no significant effect on lipid profiles in both plant- and animal-based DASH diets [51].
While some studies illustrated an adverse effect of animal protein, others report no significant association, particularly when processed and unprocessed meats are examined separately [52,53,54,55]. A number of studies have shown a relation between consumption of red meat and MetS due to its saturated fat content, although this relation was not always substantial [56,57,58]. Red meat is usually limited in heart-healthy diets because of its saturated fat content. However, a study by Damião et al. demonstrated that adjusting the intake of saturated fats disappeared the unfavorable association between red meat and MetS [59]. In our study, the diet in both groups was designed according to a healthy diet, and the type of meat and dairy was considered to be low-fat. Also, patients were advised not to consume processed meat products; these factors reduced the intake of saturated fats. Therefore, the evidence showed that the type of protein that was consumed was not as important as the reduction in energy and saturated fatty acid intake to manage MetS or related risk factors.
Previous studies have reported that eating lean meat or a lower intake of saturated fatty acids can reduce TC and LDL-c levels in hypercholesterolemia participants [60, 61], but we found no significant difference in TC or LDL-c levels in the two study arms. It is worth noting that cholesterol values in both study groups were close to normal ranges; this issue might reduce the effectiveness of the diets [62]. In addition, a study by Meinertz et al. evaluated the effects of different sources of dietary protein (soy as a plant-based vs. casein as an animal-based protein) on plasma lipoproteins in healthy individuals with normal lipid levels. The plasma level of cholesterol and LDL-c reduced in both intervention arms, and the results could not show a substantial difference between the two arms [63]. Therefore, it seems that the type of dietary protein has minimal to no impact on the levels of plasma lipoproteins in healthy individuals. However, a significantly higher HDL-c in the animal protein group was observed after 10 weeks. In this case, the role of saturated fatty acids intake in increasing HDL-c should be highlighted [64, 65]. In addition, the current study showed a decrease in LDL-c/HDL-c ratio in the animal-based protein group. In contrast, Li et al. suggested that the lacto-ovo vegetarian and vegan groups showed notably lower levels of plasma TC, LDL-c, and LDL-c/HDL-c ratio compared to both high- and moderate-meat-eater groups. However, moderate-meat-eaters had significantly higher HDL-c levels than vegans. This study did not recommend that meat eaters switch to a vegetarian diet. However, it might be advisable for meat eaters to reduce their intake of saturated fatty acids to improve their lipid profile [66].
Values of TG in the plant protein group were significantly decreased after the intervention. It is necessary to know that the amount of polyunsaturated fats might be higher in the plant-based protein diet because of higher nut consumption. A network meta-analysis of clinical trials reported that pistachio and walnut-enriched diets could lower TG, TC, and LDL-c [67]. Another meta-analysis suggested that incorporating nuts into the diet can positively affect serum TG and LDL-c in healthy adults with overweight or obesity [68]. Nuts are rich in unsaturated fats and many bioactive compounds such as antioxidants, dietary fiber, and plant sterols [69]. In the animal-based protein group, our findings revealed that the levels of TG decreased after 10 weeks of intervention, but the result was not significant. A previous cross-sectional study showed that an increase in overall protein consumption was linked to a lower likelihood of elevated TG levels in both men and women. When the types of protein were categorized, it was found that consuming animal protein was only associated with a reduced risk of elevated TG levels in men, but not in women [70]. Weight loss, as a result of increased dietary protein intake (especially with low saturated fatty acids intake), could be considered as a potential mechanism for improving TG levels [71]. In addition, polyunsaturated fats, especially omega-3 fats, could lower TG. Omega-3 could also reduce the de novo synthesis of fatty acids and TG, increase TG catabolism in adipose and muscle tissue, and increase the clearance of TG-rich lipoproteins through activation of the peroxisome proliferator-activated receptors (PPARs) gene [72].
In the current study, the AIP levels decreased in both groups after 10 weeks of intervention. A case-control study evaluated the AIP values in vegetarian and non-vegetarians groups. The mentioned study did not find any variation in AIP levels between different groups. Therefore, the mentioned research did not suggest that a vegetarian diet would be superior in the management and prevention of heart disease risk factors [73]. In addition, a cross-sectional study evaluated the association between different types of plant-based dietary indices and AIP levels. The results showed that greater compliance with the overall plant-based diet index and moderate adherence to the healthful plant-based diet index were linked to reduced likelihood of elevated AIP levels [25]. It is worth noting that the effect of different types of dietary protein on triglycerides and HDL-c, as components of AIP, could predict AIP levels. In the current study, plant-based protein intake decreased TG, and animal-based protein intake increased HDL-c levels that resulting in decreased AIP levels in both intervention groups.
In both groups of the present trial, there was no difference in fasting blood sugar (FBS) after 10 weeks of intervention. Although previous studies suggested that decreased energy intake and subsequent weight loss could be associated with lower FBS, our study could not obtain such a result. Some studies illustrated that blood glucose reduction might be greater in people with higher initial blood sugar than normal or near-normal blood sugar [38, 74]. On the other hand, an elevation in fiber intake might be related to lower blood sugar [75, 76]. In the present study, neither group could meet the recommended amount of fiber intake, which is equivalent to 14 grams per 1000 kcal per day, and this could be a reason for no positive changes in blood sugar.
The intake of macronutrients plays a significant role in the prevention and treatment of MetS and its components. A community-based, large genomic study by Park et al. illustrated that individuals with MetS exhibited reduced consumption of animal-source proteins, along with elevated consumption of plant-source carbohydrates and proteins [77]. This finding might be related to the correlation of dietary plant-based protein and carbohydrates that could alter the association between plant-based protein and MetS [78].
There was a significant increase in adropin levels in both intervention groups, and after adjustment for baseline adropin concentrations, age, and sex, the levels of adropin were higher in the plant protein group than in the animal protein. Onge et al., in a crossover trial, investigated the effect of regular and limited sleep combined with a controlled meal, 30% of energy from fat, 55% from carbohydrate, and 15% from protein, during 4 days. Their results showed that adropin could not be affected by sleep status. However, adropin was correlated with food choices in women. Such that plasma adropin was directly correlated with total and saturated fat and inversely with carbohydrate intake [79]. Furthermore, a study on mice illustrated that a diet consisting of low carbohydrate and high fat was associated with higher levels of adropin, whereas a high-carbohydrate low-fat diet was associated with lower adropin values [80].
Strengths and limitations
The present study contains some strengths. It is the first study that examined the effect of replacing animal-based with plant-based protein on the aforesaid parameters. Also, participants had high adherence to the prescribed diet due to being motivated and somewhat educated. However, we acknowledge some limitations. A great percentage of participants dropped out due to conducting the study during the COVID-19 epidemic, as well as the long duration of the study. Since we live in a developing country, it was not possible to feed the participants and only diets were recommended to them. Due to financial constraints, both the duration and the sample size of the study were relatively limited. While the 10-week intervention period allowed us to observe meaningful improvements in metabolic syndrome parameters, a longer duration might have resulted in more pronounced or sustained effects. It is worth noting that a longer dietary intervention, on the other hand, might result in lower participants’ adherence to dietary recommendations. The relatively modest sample size of 60 participants could also limit the statistical power to detect small differences between groups and might restrict the generalizability of our findings. In addition, diet compliance was evaluated using a 3-day diet record every two weeks, and self-reported dietary intake might increase measurement error and information bias. Also, this study could not establish a difference between various types of dietary protein intake and components of MetS; further studies with optimized designs are needed to evaluate the effect of different types of dietary protein on components of MetS and its novel biomarkers.
In conclusion, our findings revealed that both diets based on plant and animal protein were associated with improvement in anthropometric indices, MetS components, plasma atherogenic index, and elevated serum adropin levels. Apart from adropin values, no significant difference was observed between the two intervention groups. Further prospective studies, as well as intervention studies with a larger sample size and a longer period of time,e are needed.
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Nilsson PM, Tuomilehto J, Rydén L. The metabolic syndrome–What is it and how should it be managed?. Eur J Prev Cardiol. 2019;26:33–46.
Potenza MV, Mechanick JI. The metabolic syndrome: definition, global impact, and pathophysiology. Nutr Clin Pract. 2009;24:560–77.
Noubiap JJ, Nansseu JR, Lontchi-Yimagou E, Nkeck JR, Nyaga UF, Ngouo AT, et al. Geographic distribution of metabolic syndrome and its components in the general adult population: A meta-analysis of global data from 28 million individuals. Diab Res Clin Pr. 2022;188:109924.
Yosaee S, Khodadost M, Esteghamati A, Speakman JR, Shidfar F, Nazari MN, et al. Metabolic syndrome patients have lower levels of adropin when compared with healthy overweight/obese and lean subjects. Am J Men’s health. 2017;11:426–34.
Zhang X, Li X, Feng J, Chen X. Association of metabolic syndrome with atherogenic index of plasma in an urban Chinese population: a 15-year prospective study. Nutr Metab Cardiovasc Dis. 2019;29:1214–9.
Kumar KG, Trevaskis JL, Lam DD, Sutton GM, Koza RA, Chouljenko VN, Kousoulas KG, Rogers PM, Kesterson RA, Thearle M, Ferrante AW. Identification of adropin as a secreted factor linking dietary macronutrient intake with energy homeostasis and lipid metabolism. Cell Metab. 2008;8:468–81.
Yosaee S, Khodadost M, Esteghamati A, Speakman JR, Shidfar F, Nazari MN, Bitarafan V, Djafarian K. Metabolic syndrome patients have lower levels of adropin when compared with healthy overweight/obese and lean subjects. Am J Men's Health. 2017;11:426–34.
Ali II, D’Souza C, Singh J, Adeghate E. Adropin’s role in energy homeostasis and metabolic disorders. Int J Mol Sci. 2022;23:8318.
Zang H, Jiang F, Cheng X, Xu H, Hu X. Serum adropin levels are decreased in Chinese type 2 diabetic patients and negatively correlated with body mass index. Endocr J. 2018;65:685–91.
Zhang H, Chen N. Adropin as an indicator of T2DM and its complications. Food Sci Hum Wellness. 2022;11:1455–63.
Zhu X, Yu L, Zhou H, Ma Q, Zhou X, Lei T, et al. Atherogenic index of plasma is a novel and better biomarker associated with obesity: a population-based cross-sectional study in China. Lipids Health Dis. 2018;17:37.
Niroumand S, Khajedaluee M, Khadem-Rezaiyan M, Abrishami M, Juya M, Khodaee G, Dadgarmoghaddam M. Atherogenic Index of Plasma (AIP): A marker of cardiovascular disease. Med J Islam Repub Iran. 2015;29:240.
Onat A, Can G, Kaya H, Hergenç G. “Atherogenic index of plasma”(log10 triglyceride/high-density lipoprotein− cholesterol) predicts high blood pressure, diabetes, and vascular events. J Clin Lipidol. 2010;4:89–98.
Li Y-W, Kao T-W, Chang P-K, Chen W-L, Wu L-W. Atherogenic index of plasma as predictors for metabolic syndrome, hypertension and diabetes mellitus in Taiwan citizens: a 9-year longitudinal study. Sci Rep. 2021;11:9900.
Vrablík M, Dobiasova M, Zlatohlávek L, Urbanová Z, Ceska R. Biomarkers of cardiometabolic risk in obese/overweight children: effect of lifestyle intervention. Physiol Res. 2014;63:743.
Fernández-Verdejo R, Moya-Osorio JL, Fuentes-Lopez E, Galgani JE. Metabolic health and its association with lifestyle habits according to nutritional status in Chile: A cross-sectional study from the National Health Survey 2016-2017. PLoS One. 2020;15:e0236451.
Moghaddam E, Vogt JA, Wolever TM. The effects of fat and protein on glycemic responses in nondiabetic humans vary with waist circumference, fasting plasma insulin, and dietary fiber intake. J Nutr. 2006;136:2506–11.
Chiavaroli L, Lee D, Ahmed A, Cheung A, Khan T, Mejia SB, et al. Low Glycemic Index/load dietary patterns and glycemia and cardiometabolic risk factors in diabetes: a systematic review and meta-analysis of randomized controlled trials. Curr Dev Nutr. 2021;5:1018.
Mariotti F. Animal and plant protein sources and cardiometabolic health. Adv Nutr. 2019;10:S351–S66.
Shih YL, Shih CC, Huang TC, Chen JY. The relationship between elevated homocysteine and metabolic syndrome in a community-dwelling middle-aged and elderly population in Taiwan. Biomedicines. 2023;11:378.
Liu C, Liu L, Wang Y, Chen X, Liu J, Peng S, et al. Hyperhomocysteinemia increases risk of metabolic syndrome and cardiovascular death in an elderly Chinese community population of a 7-year follow-up study. Front Cardiovasc Med. 2022;8:811670.
Razak MA, Begum PS, Viswanath B, Rajagopal S. Multifarious Beneficial Effect of Nonessential Amino Acid, Glycine: A Review. Oxid Med Cell Longev. 2017;2017:1716701.
Zhu R, Fogelholm M, Poppitt SD, Silvestre MP, Møller G, Huttunen-Lenz M, et al. Adherence to a plant-based diet and consumption of specific plant foods—associations with 3-year weight-loss maintenance and cardiometabolic risk factors: a secondary analysis of the PREVIEW intervention study. Nutrients. 2021;13:3916.
Castro-Barquero S, Ruiz-León AM, Sierra-Pérez M, Estruch R, Casas R. Dietary strategies for metabolic syndrome: a comprehensive review. Nutrients. 2020;12:2983.
Shahdadian F, Saneei P, Lotfi K, Feizi A, Askari G, Safavi SM. Association of plant-based diets with adropin, atherogenic index of plasma, and metabolic syndrome and its components: A cross-sectional study on adults. Front Nutr. 2023;10:1077709.
Alberti KG, Eckel RH, Grundy SM, Zimmet PZ, Cleeman JI, Donato KA, et al. Harmonizing the metabolic syndrome: a joint interim statement of the international diabetes federation task force on epidemiology and prevention; National Heart, lung, and Blood Institute; American Heart Association; World Heart Federation; International Atherosclerosis Society; and International Association for the Study of Obesity. Circulation. 2009;120:1640–5.
Wang H, Dai Y, Huang S, Rong S, Qi Y, Li B. A new perspective on special effective interventions for metabolic syndrome risk factors: a systematic review and meta-analysis. Front Public Health. 2023;11:1133614.
Joseph MS, Konerman MA, Zhang M, Wei B, Brinza E, Walden P, et al. Long-term outcomes following completion of a structured nutrition and exercise lifestyle intervention program for patients with metabolic syndrome. Diab Metab Syndr Obes. 2018;11:753–9.
Whitehead AL, Julious SA, Cooper CL, Campbell MJ. Estimating the sample size for a pilot randomised trial to minimise the overall trial sample size for the external pilot and main trial for a continuous outcome variable. Stat methods Med Res. 2016;25:1057–73.
Viechtbauer W, Smits L, Kotz D, Budé L, Spigt M, Serroyen J, et al. A simple formula for the calculation of sample size in pilot studies. J Clin Epidemiol. 2015;68:1375–9.
Institute of Medicine. Dietary Reference Intakes for Energy, C, Fiber, fat, fatty acids, cholesterol, protein and amino acids. Food and Nutrition Board. Washington, DC: National Academy Press; 2002.
Phillips JA. Dietary Guidelines for Americans, 2020–2025. Workplace Health Saf. 2021;69:395.
Dobiás̆ová M, Frohlich J. The plasma parameter log (TG/HDL-C) as an atherogenic index: correlation with lipoprotein particle size and esterification rate inapob-lipoprotein-depleted plasma (FERHDL). Clin Biochem. 2001;34:583–8.
Leidy HJ, Carnell NS, Mattes RD, Campbell WW. Higher protein intake preserves lean mass and satiety with weight loss in pre-obese and obese women. Obesity. 2007;15:421–9.
Wycherley TP, Moran LJ, Clifton PM, Noakes M, Brinkworth GD. Effects of energy-restricted high-protein, low-fat compared with standard-protein, low-fat diets: a meta-analysis of randomized controlled trials. Am J Clin Nutr. 2012;96:1281–98.
Ma Y, Sun L, Mu Z. Effects of different weight loss dietary interventions on body mass index and glucose and lipid metabolism in obese patients. Medicine. 2023;102:e33254.
Moon J, Koh G. Clinical evidence and mechanisms of high-protein diet-induced weight loss. J Obes Metab Syndr. 2020;29:166.
Hill AM, Harris Jackson KA, Roussell MA, West SG, Kris-Etherton PM. Type and amount of dietary protein in the treatment of metabolic syndrome: a randomized controlled trial. Am J Clin Nutr. 2015;102:757–70.
Zheng J-S, Steur M, Imamura F, Freisling H, Johnson L, van der Schouw YT, et al. Dietary intake of plant-and animal-derived protein and incident cardiovascular diseases: the pan-European EPIC-CVD case–cohort study. Am J Clin Nutr. 2024;119:1164–74.
Sawyer B, Stone KA, Kotarsky CJ, Johnson N, Bradley A, Scheffert RA, et al. Animal-based dietary protein intake is not a risk factor for metabolic syndrome among young or middle-aged females. Nutr Metab Insights. 2022;15:11786388221107800.
Richter CK, Skulas-Ray AC, Champagne CM, Kris-Etherton PM. Plant protein and animal proteins: do they differentially affect cardiovascular disease risk?. Adv Nutr. 2015;6:712–28.
Appel LJ. The effects of protein intake on blood pressure and cardiovascular disease. Curr Opin Lipido. 2003;14:55–9.
Larsen R, Eilertsen K-E, Elvevoll EO. Health benefits of marine foods and ingredients. Biotechnol Adv. 2011;29:508–18.
El Khoury D, Anderson GH. Recent advances in dietary proteins and lipid metabolism. Curr Opin Lipido. 2013;24:207–13.
Tang WW, Wang Z, Levison BS, Koeth RA, Britt EB, Fu X, et al. Intestinal microbial metabolism of phosphatidylcholine and cardiovascular risk. N Engl J Med. 2013;368:1575–84.
Johnson N, Stone K, Stastny S, McGrath R, Hackney K. Beef consumption and functional performance in middle-aged and older adults: a narrative review. J Food Nutr Sci. 2021;3:18–31.
Maki KC, Dicklin MR, Kirkpatrick CF. Saturated fats and cardiovascular health: Current evidence and controversies. J Clin Lipido. 2021;15:765–72.
Astrup A, Magkos F, Bier DM, Brenna JT, de Oliveira Otto MC, Hill JO, et al. Saturated fats and health: a reassessment and proposal for food-based recommendations: JACC State-of-the-Art Review. J Am Coll Cardiol. 2020;76:844–57.
Chung S, Chung M-Y, Choi H-K, Park JH, Hwang J-T, Joung H. Animal protein intake is positively associated with metabolic syndrome risk factors in middle-aged Korean Men. Nutrients. 2020;12:3415.
Azemati B, Rajaram S, Jaceldo-Siegl K, Haddad EH, Shavlik D, Fraser GE. Dietary animal to plant protein ratio is associated with risk factors of metabolic syndrome in participants of the AHS-2 Calibration Study. Nutrients. 2021;13:4296.
Vasei MH, Hosseinpour-Niazi S, Ainy E, Mirmiran P. Effect of dietary approaches to stop hypertension (DASH) diet, high in animal or plant protein on cardiometabolic risk factors in obese metabolic syndrome patients: A randomized clinical trial. Prim Care Diab. 2022;16:634–9.
Micha R, Wallace SK, Mozaffarian D. Red and processed meat consumption and risk of incident coronary heart disease, stroke, and diabetes mellitus: a systematic review and meta-analysis. Circulation. 2010;121:2271–83.
Pan A, Sun Q, Bernstein AM, Schulze MB, Manson JE, Stampfer MJ, et al. Red meat consumption and mortality: results from 2 prospective cohort studies. Arch Intern Med. 2012;172:555–63.
Rohrmann S, Overvad K, Bueno-de-Mesquita HB, Jakobsen MU, Egeberg R, Tjønneland A, et al. Meat consumption and mortality-results from the European Prospective Investigation into Cancer and Nutrition. BMC Med. 2013;11:1–12.
Sinha R, Cross AJ, Graubard BI, Leitzmann MF, Schatzkin A. Meat intake and mortality: a prospective study of over half a million people. Arch Intern Med. 2009;169:562–71.
Cocate PG, Natali AJ, Oliveira AD, Alfenas RDCG, Peluzio Mdo C, Longo GZ, et al. Red but not white meat consumption is associated with metabolic syndrome, insulin resistance and lipid peroxidation in Brazilian middle-aged men. Eur J Prev Cardiol. 2015;22:223–30.
León EA, Henriquez P, Serra-Majem L. Mediterranean diet and metabolic syndrome: a cross-sectional study in the Canary Islands. Public Health Nutr. 2006;9:1089–98.
Babio N, Sorlí M, Bulló M, Basora J, Ibarrola-Jurado N, Fernández-Ballart J, et al. Association between red meat consumption and metabolic syndrome in a Mediterranean population at high cardiovascular risk: cross-sectional and 1-year follow-up assessment. Nutr, Metab Cardiovasc Dis. 2012;22:200–7.
Damiao R, Castro TG, Cardoso MA, Gimeno SG, Ferreira SR. Group JBDS. Dietary intakes associated with metabolic syndrome in a cohort of Japanese ancestry. Br J Nutr. 2006;96:532–8.
Roussell M, Hill A, Gaugler T, West S, Ulbrecht J, Vanden Heuvel J, et al. Effects of a DASH-like diet containing lean beef on vascular health. J Hum Hypertens. 2014;28:600–5.
Roussell MA, Hill AM, Gaugler TL, West SG, Vanden Heuvel JP, Alaupovic P, et al. Beef in an Optimal Lean Diet study: effects on lipids, lipoproteins, and apolipoproteins. Am J Clin Nutr. 2012;95:9–16.
Flock MR, Green MH, Kris-Etherton PM. Effects of adiposity on plasma lipid response to reductions in dietary saturated fatty acids and cholesterol. Adv Nutr. 2011;2:261–74.
Meinertz H, Faergeman O, Nilausen K, Chapman MJ, Goldstein S, Laplaud PM. Effects of soy protein and casein in low cholesterol diets on plasma lipoproteins in normolipidemic subjects. Atherosclerosis. 1988;72:63–70.
Hayek T, Ito Y, Azrolan N, Verdery R, Aalto-Setälä K, Walsh A, et al. Dietary fat increases high density lipoprotein (HDL) levels both by increasing the transport rates and decreasing the fractional catabolic rates of HDL cholesterol ester and apolipoprotein (Apo) AI. Presentation of a new animal model and mechanistic studies in human Apo AI transgenic and control mice. J Clin Investig. 1993;91:1665–71.
Siri-Tarino PW, Sun Q, Hu FB, Krauss RM. Saturated fatty acids and risk of coronary heart disease: modulation by replacement nutrients. Curr Atheroscler Rep. 2010;12:384–90.
Li D, Sinclair A, Mann N, Turner A, Ball M, Kelly F, et al. The association of diet and thrombotic risk factors in healthy male vegetarians and meat-eaters. Eur J Clin Nutr. 1999;53:612–9.
Liu K, Hui S, Wang B, Kaliannan K, Guo X, Liang L. Comparative effects of different types of tree nut consumption on blood lipids: a network meta-analysis of clinical trials. Am J Clin Nutr. 2020;111:219–27.
Eslami O, Khorramrouz F, Sohouli M, Bagheri N, Shidfar F, Fernandez ML. Effect of nuts on components of metabolic syndrome in healthy adults with overweight/obesity: A systematic review and meta-analysis. Nutr Metab Cardiovasc Dis. 2022;32:2459–69.
Kim Y, Keogh JB, Clifton PM. Benefits of nut consumption on insulin resistance and cardiovascular risk factors: multiple potential mechanisms of actions. Nutrients. 2017;9:1271.
Jamshidi A, Farjam M, Ekramzadeh M, Homayounfar R. Evaluating type and amount of dietary protein in relation to metabolic syndrome among Iranian adults: cross-sectional analysis of Fasa Persian cohort study. Diabetol Metab Syndr. 2022;14:42.
Halton TL, Hu FB. The effects of high protein diets on thermogenesis, satiety and weight loss: a critical review. J Am Coll Nutr. 2004;23:373–85.
Liu QK. Triglyceride-lowering and anti-inflammatory mechanisms of omega-3 polyunsaturated fatty acids for atherosclerotic cardiovascular risk reduction. J Clin Lipido. 2021;15:556–68.
Sultana AW, Begum N, Abdullah AS, Khan MAH, Rahman MR, Kamal AHM, et al. Lipid Profile and Atherogenic Index of Plasma (AIP) in Vegetarians. J Enam Med Col. 2015;5:30–33.
Group U. UK Prospective Diabetes Study 7: response of fasting plasma glucose to diet therapy in newly presenting type II diabetic patients. Metabolism. 1990;39:905–12.
Kobayakawa A, Suzuki T, Ikami T, Saito M, Yabe D, Seino Y. Improvement of fasting plasma glucose level after ingesting moderate amount of dietary fiber in Japanese men with mild hyperglycemia and visceral fat obesity. J Diet Suppl. 2013;10:129–41.
Mao T, Huang F, Zhu X, Wei D, Chen L. Effects of dietary fiber on glycemic control and insulin sensitivity in patients with type 2 diabetes: A systematic review and meta-analysis. J Funct Foods. 2021;82:104500.
Park H, Kityo A, Kim Y, Lee S-A. Macronutrient intake in adults diagnosed with metabolic syndrome: Using the Health Examinee (HEXA) Cohort. Nutrients. 2021;13:4457.
Park HA. Adequacy of protein intake among Korean elderly: an analysis of the 2013–2014 Korea National Health and Nutrition Examination Survey data. Korean J Fam Med. 2018;39:130.
St-Onge MP, Shechter A, Shlisky J, Tam CS, Gao S, Ravussin E, et al. Fasting plasma adropin concentrations correlate with fat consumption in human females. Obesity. 2014;22:1056–63.
Ganesh-Kumar K, Zhang J, Gao S, Rossi J, McGuinness OP, Halem HH, et al. Adropin deficiency is associated with increased adiposity and insulin resistance. Obesity. 2012;20:1394–402.
Acknowledgements
This study was extracted from Ph.D. dissertation, which was approved by Isfahan University of Medical Sciences (no: 3400603).
Funding
The funding for conception, design, data analysis, and manuscript preparation was provided by Isfahan University of Medical Sciences in Isfahan, Iran.
Author information
Authors and Affiliations
Contributions
FS, MR, PR, AF, GA, ZM, PS, and SMS were involved in the conception and design of the study, data collection and interpretation, drafting of the manuscript, approval of the final version, and consented to all aspects of the research.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical Approval and Consent to Participate
The research methodology was conducted in accordance with the Declaration of Helsinki and complied with the CONSORT guidelines. All participants provided informed written consent prior to their inclusion in the study. The protocol was also reviewed and approved by the Ethics Committee at Isfahan University of Medical Sciences.
Consent to participate
Informed consent was obtained from all participants involved in the study.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Shahdadian, F., Rezazadegan, M., Rouhani, P. et al. Does partial replacement of animal protein with plant protein in the diet affect components of metabolic syndrome, adropin levels, and the atherogenic index of plasma? Results from a parallel randomized clinical trial in adults with metabolic syndrome. Nutr. Diabetes 15, 43 (2025). https://doi.org/10.1038/s41387-025-00401-x
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41387-025-00401-x