Abstract
Background
Prediabetes is a public health concern, and its prevalence is growing around the world. Providing an effective strategy to prevent prediabetes progression and, consequently, type-2 diabetes mellitus (T2DM) could be useful for global health. The present study aimed to investigate the impact of vitamin D supplementation on metabolic factors, serum omentin-1 levels, and anthropometric indices in women with prediabetes.
Methods
This randomized controlled trial (RCT) study was carried out on women with prediabetes aged 18–65 years. The women were allocated to the intervention (n = 48) or placebo (n = 48) groups to receive vitamin D (50000 IU) or a placebo every two weeks for 12 weeks, respectively. Fasting blood samples, anthropometric measurements, and information about the dietary intake and physical activity of the participants were collected at the beginning and after the intervention. Data analyses were done using IBM SPSS Statistics software.
Results
Vitamin D intervention significantly enhanced 25-hydroxyvitamin D (MD (CI 95%): 11.610 ng/ml (8.264, 14.956)), insulin (MD (CI 95%): 0.413 µIU/mL (0.321, 0.505)), Homeostatic Model Assessment of Insulin Resistance (MD (CI 95%): 0.411 (0.319, 0.502)), and Homeostatic Model Assessment of Insulin Resistance-β cell function (MD (CI 95%): 29.505% (22.114, 36.986)) levels compared to the placebo at the end of trial (all, p < 0.05). Moreover, a significant reduction in QUICKI was observed after the intervention (MD (CI 95%): −0.062 (−0.076, −0.048)). Changes in fasting blood sugar (FBS), lipid profiles, omentin-1 levels, and anthropometric indices were not significant between the groups after the intervention.
Conclusion
The findings revealed that vitamin D administration improved insulin secretion but failed to affect FBS, lipid profiles, anthropometric indices, and body composition in women with prediabetes. Due to inconsistent results, more well-designed RCTs are required to clarify the effects of vitamin D supplementation on these patients.
Similar content being viewed by others
Introduction
Prediabetes is a metabolic condition characterized by elevated fasting blood glucose (FBG) levels that are higher than normal but not yet the diabetes criteria [1]. According to the American Diabetes Association (ADA), prediabetes is defined as fasting blood sugar (FBS) between 100 and 125 mg/dL (impaired fasting glucose (IFG)), a 2-h glucose level between 140 and 199 mg/dL (impaired glucose tolerance (IGT)), or a glycated hemoglobin (HbA1c) between 5.7 and 6.4% [1]. The worldwide prevalence of IFG and IGT was 5.8% (298 million) and 9.1% (464 million) in 2021, respectively. It was reported that 18.2% and 15.4% of males and females in Iran had prediabetes, respectively [2]. Prediabetes prevalence is increasing around the world, and it is expected that 6.5% (414 million) and 10.0% (638 million) of the population will have IFG and IGT by 2045, respectively [3]. Individuals with prediabetes have a significantly high risk of developing type 2 diabetes mellitus (T2DM) and associated complications, including cardiovascular disease and metabolic disorders [4].
Prediabetes and obesity are closely connected. Adipose tissue secretes adipocytokines, which contribute to the pathogenesis of insulin resistance and diabetes [5,6,7]. Omentin-1 is a visceral adipose tissue secretory adipokine with anti-inflammatory effects [8]. It was reported that the levels of serum omentin-1 were lower in subjects with impaired glucose regulation and T2DM than in the normal glucose tolerance group [9]. Previous studies have also shown that plasma omentin-1 levels are inversely correlated with insulin resistance, obesity, and body fat mass [9,10,11].
Lifestyle modifications, such as diet, weight management, and exercise, are the primary strategies for preventing prediabetes progression [12, 13]. Some evidence suggests that vitamin D supplementation may influence glucose metabolism, insulin sensitivity, and weight loss [14,15,16]. Vitamin D is a fat-soluble vitamin with well-established functions in bone health, but its role in glucose homeostasis has gained increasing attention [17]. Studies indicate that vitamin D receptors (VDRs) are present in pancreatic β-cells, which influence insulin secretion, and insulin-responsive tissues such as skeletal muscle and adipose tissue [18, 19]. Moreover, VDRs play a role in glucose metabolism by suppressing gluconeogenesis via inhibiting key enzymes such as phosphoenolpyruvate carboxykinase and glucose-6-phosphatase [20]. The findings of the previous studies about the effects of vitamin D supplementation on glycemic factors were inconsistent in patients with prediabetes [21,22,23]. In the study by Niroomand et al. on adults with prediabetes and vitamin D deficiency, vitamin D intervention did not change FBG or 2-h oral glucose tolerance test plasma glucose (2hPG) concentration, but it reduced the Homeostatic Model Assessment of Insulin Resistance (HOMA-IR) score [21]. Another study by Zarrin et al. on males and females with prediabetes indicated that FBS levels were reduced, and insulin, HbA1c, and HOMA-IR levels did not alter after vitamin D supplementation [22]. The association of vitamin D levels with serum omentin-1 concentration was also controversial in patients with metabolic syndrome and healthy women [24, 25]. There is no research on the effect of vitamin D intervention on omentin-1 levels in patients with prediabetes. In the study on women with T2DM, vitamin D treatment did not change serum omentin-1 levels [26]. Moreover, Cheshmazar et al. indicated that an 8-week vitamin D intervention together with a low-calorie diet did not affect omentin-1 levels in overweight or obese adults with vitamin D deficiency [27]. However, they did not assess the effects of vitamin D on the glycemic factors among their participants [27].
Some evidence indicated that vitamin D metabolism and its impact on insulin sensitivity may be modulated by sex hormones, with potential differences in VDR expression and signaling between men and women [28, 29]. By focusing on women with prediabetes, the study targets a high-risk group with a distinct physiological context, enhancing the ability to detect meaningful effects of vitamin D supplementation on glucose regulation and diabetes prevention. As vitamin D deficiency potentially influences the progression of prediabetes to T2DM and the findings of a few studies that have evaluated the effects of vitamin D administration on patients with prediabetes were inconsistent [21,22,23]; and no study evaluated the vitamin D effects on omentin-1 levels in these patients, this randomized controlled trial (RCT) aimed to explore the effects of vitamin D supplementation on metabolic factors, serum omentin-1, and anthropometric indices in women with prediabetes.
Methods
Participants
This study is a randomized, double-blind, placebo-controlled trial conducted on 96 women with prediabetes aged 18–65 years. The participants were recruited from the healthcare center in Tabriz, Iran, using the convenience sampling method and according to the eligibility criteria. The inclusion criteria were women aged 18–65 years diagnosed with prediabetes who had signed an informed consent to participate. Prediabetes was recognized based on the ADA criteria (FBS: 100–125 mg/dL or HbA1c: 5.7–6.4%) [1]. Serum vitamin D levels >100 μg/L; morbid obesity (body mass index (BMI) ≥ 40 kg/m2) or wasting (BMI < 18.5 kg/m2); pregnancy or lactation; smoking or alcohol addiction; renal failure (in dialysis); cardiovascular, thyroid, liver, or kidney diseases; cancer; chronic neurological diseases; Cushing’s syndrome; rheumatoid arthritis; surgery in the last three months; use of vitamin/nutritional supplements current or last three months; following weight loss diets; use of various drugs such as glycemic controller or lipid-lowering drugs; concomitant participation in another experimental study; and other conditions in which the researchers believes the subject were the exclusion criteria for this study. All procedures were conducted following the Declaration of Helsinki and by considering the Good Clinical Practice (GCP) guidelines [30]. The protocol of the current project was approved by the Ethics Committee of Tabriz University of Medical Sciences (Ethical code: IR.TBZMED.REC.1402.618) and is registered in the Iranian Registry of Clinical Trials (Registration number: IRCT20100408003664N26).
Study design
Ninety-six women with prediabetes were randomly assigned to two intervention or placebo groups with a 1:1 allocation ratio, receiving either treatment (intervention group (n = 48)) or placebo (control group (n = 48)) for 12 weeks. Stata 18 (StataCorp, College Station, Texas 77845 USA) with the ralloc methods was used for generating the randomization sequence by randomly permuted block procedure with a size of 2. Then, organized in sealed opaque envelopes by a person who is not one of the investigators. The assignment sequence was concealed from the researcher before the randomization. Participants and researchers were blinded to the groups’ allocation. Individuals in the intervention groups were taking a vitamin D pearl with a dose of 50,000 units every two weeks for 12 weeks. The vitamin D supplementation dosage was considered based on the prior research [26, 31]. Since the normal upper limit of vitamin D intake is 10,000 IU/d, the recommended dosage (50000 IU every 2 weeks) was regarded as safe [32]. The placebo was sunflower oil, which was similar to the intervention of taking one pearl every two weeks. The shape, size, and color of the vitamin D pearls and placebo were the same. Vitamin D and placebo pearls were purchased from Zahravi Pharmaceutical Company (Zahravi Pharmaceutical Co, Tabriz, Iran, web address: https://www.zahravi.com/en/). Supplements were delivered to the participants at the beginning of the study and the end of each four weeks, and the follow-up visits were also carried out every four weeks. Moreover, the participants’ supplement consumption was followed up every two weeks by a telephone call. The participants’ compliance with the study procedure was evaluated by recursive pearls, together with delivering the supplements every four weeks, and serum vitamin D levels changed after the intervention period. Additionally, the participants were asked not to change their usual diet, physical activity, and lifestyle.
Outcome measures
The primary outcome measures were the changes in serum levels of 25-hydroxyvitamin D (25(OH)D), glycemic indices (FBS, insulin, and HOMA-IR, quantitative insulin-sensitivity check index (QUICKI), and Homeostatic Model Assessment of β cell function (HOMA-β) levels), lipid profiles (total cholesterol (TC), Low-density lipoprotein cholesterol (LDL-C), triglycerides (TG), and high-density lipoprotein cholesterol (HDL-C)), and omentin-1 from the baseline to 12 weeks. Secondary outcome measures were the changes in anthropometric indices, including weight, BMI, waist circumference (WC), hip circumference (HC), waist-to-hip ratio (WHR), waist-to-height ratio (WHtR), and body composition alteration from baseline to 12 weeks.
Data collection
A structured, content-based, and face-validated questionnaire checklist (by the research team) was administered to collect information regarding general characteristics and medical and treatment history. Moreover, the duration of sunlight exposure during a day was obtained by a questionnaire designed for this purpose. The participants answered questions about their average sun exposure hours on a usual day, duration of exposure, time of day when exposure occurred, how often sunscreen or sun hats were used, and described the clothing degree worn when outside. The participants’ physical activity levels were assessed using a short version of the International Physical Activity Questionnaire (IPAQ) [33], which has been translated and validated for use in Persian. This survey requires participants to indicate the number of days, hours, and minutes they engaged in strenuous activities, such as aerobics, or moderate activities, like carrying lightweight objects, during the previous week. Additionally, it asks about the number of days, hours, and minutes participants walked for a minimum of 10 min, as well as the duration of time they spent seated for any activity over the last 7 days.
The participants’ dietary intake was recorded using a 24-h dietary recall for three days (two regular days and one weekend) at the beginning and after the intervention. Then, these three-day recalls were analyzed by Nutritionist-4 software, and the three-day mean caloric and nutrient intakes of participants were estimated.
Anthropometric measurements were recorded at the start and the end of the supplementation period. A digital platform scale was used to measure weight, with participants standing barefoot in minimal clothing at the center. The same scale was utilized for height measurement, with subjects standing barefoot, arms hanging freely at their sides, and palms facing their thighs. BMI was determined by dividing weight (kg) by height (m) squared. To measure WC, a flexible plastic tape was placed at the midpoint between the lower costal margin and iliac crest, perpendicular to the body. During this measurement, the participant was standing with feet approximately 20 cm apart and arms hanging freely. HC was assessed at the widest point over the buttocks, with the researcher kneeling beside the participant to accurately observe the maximum extension level. WHR and WHtR were calculated by dividing WC (cm) by HC (cm) and WC (cm) by height (m), respectively. Additionally, the body composition (fat-free mass (FFM) and fat mass (FM)) of the participants was measured by the bioelectrical impedance analysis method using Tanita’s body composition analyzer.
A professional individual collected blood samples after 8–12 h of fasting at the baseline and after 12 weeks. The obtained blood sample was centrifuged at 22 °C, 5000 rpm, for 6 min, and the serum was stored in microtubes at −70 °C for later analysis. 25(OH)D3 concentration was measured using an enzyme-linked immunosorbent assay (ELISA) kit (Monobind kit, Monobind Inc., Lake Forest, CA, USA). FBS was evaluated by the enzymatic colorimetric method (kit: Pars Azmoon, Tehran, Iran), and serum insulin was assessed by the ELISA technique (Monobind Inc, Lake Forest, CA, USA). The HOMA-IR, HOMA-β, and QUICKI were calculated using the formulas (HOMA-IR = [Fasting Insulin (μIU/ml) * Fasting Glucose (mg/dl)]/405; HOMA-β = (Fasting Insulin (μIU/ml) * 360)/(Fasting Glucose (mg/dl) - 63); QUICKI = 1/[log(Fasting Insulin (μIU/ml)) + log(Fasting Glucose (mg/dl))] [34, 35].
The enzymatic colorimetric method was used for assessing lipid profiles, including TC, HDL-C, and serum TG (kit: Pars Azmoon, Tehran, Iran). LDL-C was calculated applying the Friedewald formula: LDL-C = TC (mg/dl) _ ([HDL-C (mg/dl) + TG (mg/dl)]/5). Serum omentin-1 levels were evaluated using the ELIZA method (Monobind kit, Monobind Inc., Lake Forest, CA, USA). The intra- and inter-assay coefficients of variation for omentin-1 were <10% and <12%, respectively, and the sensitivity was 2 ng/L.
Statistical analyses
Following the completion of data gathering, analysis was conducted using IBM SPSS Statistics software (version 20, IBM Corp, Armonk, USA). To evaluate the normality of data distribution, the Kolmogorov-Smirnov test and distribution measures (skewness and kurtosis) were employed. Numerical variables with normal distribution were presented as mean ± standard deviation (SD), while those with non-normal distribution were expressed as median and interquartile range (IQR). Absolute and relative frequencies were used to represent categorical variables. Comparisons of baseline numerical variables between intervention and placebo groups were performed using the independent sample t-test or the Mann–Whitney U test. Fisher’s exact test was utilized to compare qualitative variables. To assess within-group changes of variables post-intervention, the paired sample t-test or Wilcoxon rank-sum test was applied. For normally distributed variables, mean difference (MD) and confidence interval (CI), and for variables with non-normal distribution, differences in medians (DIM) and change percent were reported. The change percent was calculated using the formula: ((post-intervention mean − pre-intervention mean)/pre-intervention mean) × 100.
The impact of vitamin D supplementation was evaluated using analysis of covariance (ANCOVA) through three distinct models. The first model, referred to as the baseline model, adjusted for baseline values and age. The second model incorporated adjustments for baseline values, age, and changes in energy intake and physical activity. The third model, known as the full model, adjusted for baseline values, age, changes in energy intake and physical activity, dietary vitamin D consumption, sun exposure, and baseline serum 25(OH)D levels. For variables that were not normally distributed, ANCOVA was performed after logarithmic transformation. All statistical analyses adhered to the intention-to-treat principle. For variables with missing data, mean imputation was used, whereby missing values were replaced with the mean of the observed values for that variable. This approach assumes that data were missing completely at random [36]. All statistical analyses were statistically significant at p < 0.05 using a two-tailed approach.
Results
General characteristics
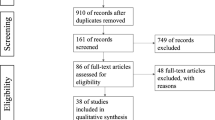
The process of the study is outlined in Fig. 1. Ninety-six women with prediabetes were enrolled in this study, and 89 women (45 in the vitamin D group and 44 in the placebo group) completed the 12-week study duration. The percentage of lost to follow-up was 7.29%, and the participant compliance with the study procedure was more than 95% based on the number of recursive pearls. Finally, 48 patients were included in the ITT analysis in each group.
Study flow diagram, ITT Intention-to-treat.
The general characteristics of the women are presented in Table 1. The mean age and BMI were 50.91 ± 9.04 years and 32.52 ± 4.45 kg/m2, respectively. Except for one person, all of them were married, and there was no significant difference in age and educational level between the groups. At the beginning of the study, 83.3% and 64.6% of women had vitamin D deficiency in the intervention and placebo groups, respectively. Moreover, 66.7% and 75.0% of participants were obese in the vitamin D and placebo groups, respectively. However, the percentage of vitamin D deficiency and overweight/obesity was not significantly different between the two groups. In addition, there were no significant differences in the sunlight exposure duration, levels of physical activity, and family history of diabetes (first-degree relative) between the groups.
Dietary intake
Table 2 indicates the dietary intake data of the women. Energy, carbohydrate, protein, fat, and vitamin D intakes of participants were not significantly different between the two groups at the beginning of the study. Moreover, changes in these variables were not significant within the groups during the intervention (p > 0.05).
Based on the ANCOVA adjusted in three models for baseline values, age, energy intake change, physical activity change, sunlight exposure time, and dietary vitamin D intake, there was no significant difference in the mean of dietary energy, carbohydrate, protein, fat, and vitamin D intakes between the intervention and placebo groups at the end of the trial (p > 0.05).
Biochemical parameters
The biochemical parameters of the participants are shown in Table 3. There were no significant differences in the biochemical values between the two groups at baseline (p > 0.05). Serum vitamin D and insulin concentrations and HOMA-β percent were significantly increased within the vitamin D group during the trial (all, p < 0.001). HOMA-IR was also significantly enhanced within both groups after the intervention compared to the baseline (p < 0.001). However, QUICKI was significantly decreased within both groups after the intervention (p < 0.001 and p = 0.038 for the vitamin D and placebo groups, respectively). Moreover, serum HDL-C levels were significantly increased within the placebo group during the study (p < 0.001).
According to the ANCOVA adjusted in three models for baseline values, age, energy intake change, physical activity change, sunlight exposure time, and dietary vitamin D intake, significant differences were observed in serum levels of 25(OH)D3 (MD (CI 95%): 11.610 ng/ml (8.264, 14.956), p < 0.001), insulin (MD (CI 95%): 0.422 µIU/mL (0.317, 0.526), p < 0.001), HOMA-IR (MD (CI 95%): 0.420 ng/dl (0.316, 0.524), p < 0.001), HOMA-β (MD (CI 95%): 28.459 (20.084, 36.834), p < 0.001), and QUICKI (MD (CI 95%): −0.063 (−0.079, −0.048), p < 0.001) between the groups after the intervention. Changes in serum levels of omentin-1, FBS, TC, TG, HDL-C, and LDL-C were not significant between the two groups after vitamin D administration at the end of the study (p > 0.05).
The results of the subgroup analysis based on the BMI categories (25.0–29.9 and 30.0–39.9) showed that vitamin D supplementation led to a significant increase in levels of insulin, HOMA-IR, and HOMA-β and a decrease in QUICKI in both categories of BMI (Supplementary Table 1). However, vitamin D intervention decreased serum levels of TC and LDL-C in participants with overweight but not in patients with obesity (Supplementary Table 1).
Anthropometric measurements
As shown in Table 4, there was no significant difference between the intervention and placebo groups in terms of anthropometric measures at the beginning of the study (p > 0.05). Within both groups, WC and WHthR were significantly enhanced at the end of the intervention period compared to the baseline. Moreover, a significant decrease in WHR and FM% and an increase in FFM% were observed in the placebo group. Based on the ANCOVA adjusted for age and baseline values, the anthropometric indices were not significantly different between the vitamin D and placebo groups after the intervention (p > 0.05); these results did not change after further adjustment for energy intake change, physical activity change, sunlight exposure time, and dietary vitamin D intake.
Discussion
Patients with prediabetes have a 50% higher risk of developing T2DM than healthy people [37], and 70% of them progress to T2DM if left untreated [38]. The strategies for preventing the progression of prediabetes to T2DM are critical. More than 80% of our participants had a vitamin D deficiency, and the mean of 25(OH)D levels was lower than the normal limits (≥30 ng/ml) [39] in both the vitamin D and placebo groups at the beginning of the study. While the vitamin D deficiency prevalence was not statistically significant between the groups, clinically, its prevalence was higher in the vitamin D group than in the placebo group. Moreover, vitamin D intervention significantly increased 25(OH)D levels but did not reach sufficiency levels.
The findings of this RCT revealed that in patients with prediabetes, vitamin D administration resulted in enhanced levels of 25(OH)D, insulin, HOMA-IR, and HOMA-β and decreased QUICKI. However, vitamin D effects on serum FBS were not significant. The results of the current study were similar in part to previous trials evaluating the effects of vitamin D supplementation on glycemic control. Niroomand et al. reported no significant changes in fasting plasma glucose (FPG) [21]. Another study by Bhat et al. on overweight/obese Asian Indian adult women with prediabetes and vitamin D deficiency indicated that after supplementation with 60000 IU vitamin D once per week for eight weeks, serum FBS levels were decreased, but insulin concentration and HOMA-IR score did not change [40]. Moreover, the results of Tuomainen et al. showed that 40 μg/day or 80 μg/day vitamin D supplementation for 5 months in overweight subjects with prediabetes aged ≥60 years did not alter glucose metabolism [41]. Davidson et al. did not find any significant change in FPG, HOMA-IR, and HOMA-β levels after 6 months of vitamin D administration in patients with prediabetes and hypovitaminosis D [19]. Nevertheless, a study by Lu et al. reported that vitamin D treatment improved insulin sensitivity in individuals with impaired glucose regulation after one year of intervention [42].
There are some explanations for why vitamin D supplementation in this study did not affect the FBS levels but enhanced the HOMA-β and insulin levels. Vitamin D is known to enhance pancreatic β-cell function, which can lead to increased insulin secretion [43, 44]; however, in the presence of peripheral insulin resistance, elevated insulin levels may not lead to improved glucose uptake or lower fasting glucose [45]. Similarly, vitamin D increased β-cell function as well as insulin resistance and did not change FBS levels in this study. However, an increase in insulin may reflect a compensatory response to underlying insulin resistance, particularly in the context of obesity [20, 46]. About 80% of participants in this study were overweight or obese. Visceral adiposity is strongly linked with impaired insulin signaling and chronic inflammation [47]. As the majority of our participants were obese, having high HOMA-IR and low QUICKI was expected in these patients, and the observed increase in central obesity among participants likely contributed to sustained insulin resistance. Therefore, increasing HOMA-IR, decreasing QUICKI, and consequently enhancing insulin secretion may be because of increased central obesity in these women. Vitamin D could not decrease general and central obesity and consequently increase insulin sensitivity; higher intervention duration may enhance insulin sensitivity by reducing obesity. Nevertheless, individual variability in response, potentially influenced by vitamin D receptor (VDR) gene polymorphisms, may also play a role in the observed vitamin D effects on insulin secretion and fasting glucose levels [48, 49]. However, FBS levels are regulated by a complex network of hormonal and metabolic factors, including glucagon and liver glucose output. Furthermore, recent trials suggest that vitamin D may enhance insulin action in muscle and adipose tissues, leading to improved postprandial glucose metabolism rather than significant reductions in FBS [50]. A higher duration or dose of vitamin D may be required to normalize vitamin D levels in patients with prediabetes who are overweight or obese.
Based on the findings, omentin-1, as an adipocytokine inversely related to insulin resistance and impaired glucose tolerance [15, 19, 21], did not increase by the end of the trial. Our findings were similar to some studies in patients with T2DM that have reported no significant changes in serum concentration of omentin-1 following vitamin D supplementation. For example, Fazelian et al. revealed that vitamin D intervention did not increase serum levels of omentin-1 in subjects with T2DM; however, according to subgroup analysis, omentin-1 levels were increased significantly in patients with lower baseline serum 25(OH)D levels [26]. Moreover, Cheshmazar et al. did not detect a significant difference in serum omentin-1 concentration after intervention [27]. However, Jafari et al. showed that fortified yogurt with vitamin D for 12 weeks enhanced the serum levels of omentin-1 in postmenopausal women with T2DM [51]. Amirkhizi et al. also indicated a significant positive relationship between the serum levels of 25(OH)D and omentin-1 [24]. Therefore, the non-significant effects of vitamin D intervention on insulin resistance could be partially related to the lack of vitamin D influence on omentin-1 levels. Moreover, the metabolic status of participants may interfere with the effects of vitamin D in these patients.
The present study found that vitamin D supplementation did not significantly affect lipid profile levels, which aligns with several previous clinical trials and meta-analyses. Bhatt et al. reported that vitamin D supplementation did not change serum TC, TG, LDL-C, and HDL-C levels in patients with prediabetes [40]. A recent meta-analysis indicated that vitamin D intervention significantly reduced TG levels, but had no significant effects on TC, HDL-C, and LDL-C concentrations in individuals with prediabetes [52]. However, some studies demonstrated that low serum 25(OH)D levels were associated with adverse lipid status [52]. The lack of vitamin D supplementation’s significant effect on improving lipid profiles in this study may be because the baseline levels of lipid profiles in the participants were approximately within the normal range. However, subgroup analysis revealed that vitamin D intervention decreased TC and LDL-C levels in participants with overweight compared to the obese. Vitamin D, possibly via modulating hepatic cholesterol metabolism by downregulating HMG-CoA reductase and upregulating LDL receptors, and facilitating greater clearance of LDL-C from the blood, could affect serum TC and LDL-C levels [53].
In terms of anthropometric measurements and body composition, we did not obtain any significant alteration in women with prediabetes following vitamin D administration. Similarly, the studies reported that anthropometric measures did not alter significantly after vitamin D supplementation in patients with prediabetes [22, 26]. Results from the meta-analysis showed that vitamin D supplementation without calorie restriction failed to decrease the weight or the FM percentage [16]. However, cross-sectional surveys reported that serum 25(OH)D level is connected with body FM regulation and increasing lean body mass [54,55,56]. Although the increased central obesity was significant only within each group and was not significant between the groups, it was likely due to hormonal changes, such as the decline in estrogen, elevated cortisol levels resulting from chronic psychological stress, and insulin resistance, which is associated with increased abdominal adiposity.
Possible explanations for the controversy of the findings regarding the vitamin D effects on patients with prediabetes are the duration of supplementation and dosage used, and variations in individual response to supplementation based on genetics, lifestyle, and coexisting metabolic conditions, which may have influenced the outcomes. Some studies suggest that short-term supplementation (e.g., <6 months) may not be sufficient to induce measurable metabolic changes, as the role of vitamin D in glucose metabolism is indirect and long-term [57]. Higher doses of vitamin D (>4000 IU/day) have been hypothesized to produce stronger effects, but results remain inconsistent across studies. Additionally, genetic variability in VDR polymorphisms may influence individual responses to supplementation, affecting its impact on glucose metabolism [49]. Moreover, the majority of participants in this RCT were overweight or obese. There is a hypothesis that in obese individuals, 1,25-dihydroxyvitamin D (1,25(OH)D) concentration is high, which can act to limit the production of its precursor and reduce the levels of 25(OH)D. In obese individuals, the fat tissue located beneath the skin exhibits reduced levels of CYP2R1, an enzyme crucial for vitamin D 25-hydroxylation. Additionally, there is a potential decrease in the expression of 1-α hydroxylase in obese subjects. These findings indicate that obesity may impair both the 25-hydroxylation and 1-α hydroxylation processes of vitamin D metabolism [58]. Moreover, it is demonstrated that the percentage of body FM has an inverse association with 25(OH)D levels regardless of age, latitude, and longitude [59]. The FM percentage in more than 97% of women in this study was higher than the normal range. Vitamin D, as a fat-soluble substance, preferentially deposits in body fat, decreasing vitamin D supplement bioavailability and consequently the potential effects [60]. A meta-analysis also indicated that vitamin D supplementation in T2DM patients showed more improvement in glycemic markers among non-obese individuals than obese individuals [61]. The researchers propose that obese individuals, even those with insufficient vitamin D levels, may not gain additional benefits from supplementation due to the potential storage of some supplemented doses in their adipose tissue [61]. It is proposed that obese people’s response to vitamin D supplementation is approximately 30% lower compared to non-obese individuals, and vitamin D replacement therapy should be tailored based on body size to reach the desired serum 25(OH)D levels [62]. In this study, serum 25(OH)D levels increased significantly after vitamin D supplementation, but not to the extent that therapeutic effects were seen. According to the older Endocrine Society Guidelines and other investigations, maintaining 25(OH)D levels preferably at 40.0–60.0 ng/mL (100–150 nmol/L) is recommended to achieve the optimal overall health benefits of vitamin D [39, 63, 64]. Intakes at least 4000–6000 IU/day from vitamin D from all sources would be required to maintain 25(OH)D levels in the 40 to 60 ng/mL range [39]. Therefore, to achieve the therapeutic effects of vitamin D in individuals with overweight and obesity, it is suggested to supplement with a higher dose of vitamin D [39].
Sun exposure time of participants was adjusted in the analysis; however, misreporting by the participants was unavoidable and was a limitation of the study. Moreover, because of the relatively shorter intervention duration of the study, studies with longer follow-up periods or combined interventions such as vitamin D aligned with lifestyle modifications may be useful in patients with vitamin D deficiency.
Conclusion
The findings indicated that vitamin D supplementation increased the levels of 25(OH)D, insulin, HOMA-IR, and HOMA-β and decreased QUICKI without significant effects on serum FBS concentrations, omentin-1 levels, and anthropometric measures in patients with prediabetes. However, vitamin D intervention decreased TC and LDL-C levels in overweight participants. Future research should focus on long-term clinical trials or higher doses of vitamin D to clarify these relationships and determine the effects of vitamin D supplements in both genders of populations with prediabetes.
Data availability
The data supporting the current study are available from the corresponding author on reasonable request.
References
Association AD. Diagnosis and classification of diabetes mellitus. Diabetes care. 2014;37:S81–S90.
Shahraz S, Moghaddam SS, Azmin M, Peykari N, Yoosefi M, Mohebi F, et al. Prevalence of diabetes and prediabetes, and achievements in diabetes control in Iran; the results of the STEPS of 2016. Archives Iran Med. 2022;25:591.
Rooney MR, Fang M, Ogurtsova K, Ozkan B, Echouffo-Tcheugui JB, Boyko EJ, et al. Global prevalence of prediabetes. Diabetes Care. 2023;46:1388–94.
Mancini GJ, Cheng AY, Connelly K, Fitchett D, Goldenberg R, Goodman SG, et al. Diabetes for cardiologists: practical issues in diagnosis and management. Canadian J Cardiol. 2017;33:366–77.
Martinez KE, Tucker LA, Bailey BW, LeCheminant JD. Expanded normal weight obesity and insulin resistance in US adults of the National Health and Nutrition Examination Survey. J Diabetes Res. 2017;2017:9502643.
Cheng Y-H, Tsao Y-C, Tzeng I-S, Chuang H-H, Li W-C, Tung T-H, et al. Body mass index and waist circumference are better predictors of insulin resistance than total body fat percentage in middle-aged and elderly Taiwanese. Medicine. 2017;96:e8126.
Meah FA, DiMeglio LA, Greenbaum CJ, Blum JS, Sosenko JM, Pugliese A, et al. The relationship between BMI and insulin resistance and progression from single to multiple autoantibody positivity and type 1 diabetes among TrialNet Pathway to Prevention participants. Diabetologia. 2016;59:1186–95.
Schäffler A, Neumeier M, Herfarth H, Fürst A, Schölmerich J, Büchler C. Genomic structure of human omentin, a new adipocytokine expressed in omental adipose tissue. Biochim Biophys Acta BBA Gene Struct Expr. 2005;1732:96–102.
Pan H-Y, Guo L, Li Q. Changes of serum omentin-1 levels in normal subjects and in patients with impaired glucose regulation and with newly diagnosed and untreated type 2 diabetes. Diabetes Res Clin Pract. 2010;88:29–33.
Tan BK, Adya R, Farhatullah S, Lewandowski KC, O’Hare P, Lehnert H, et al. Omentin-1, a novel adipokine, is decreased in overweight insulin-resistant women with polycystic ovary syndrome: ex vivo and in vivo regulation of omentin-1 by insulin and glucose. Diabetes. 2008;57:801–8.
Abd-Elbaky AE, Abo-ElMatty DM, Mesbah NM, Ibrahim SM. Omentin and apelin concentrations in relation to obesity, diabetes mellitus type two, and cardiovascular diseases in Egyptian population. Int J Diabetes Develop Ctries. 2016;36:52–8.
Galaviz KI, Suvada K, Merchant R, Dharanendra S, Haw JS, Narayan KV, et al. Interventions for reversing prediabetes: a systematic review and meta-analysis. Am J Prevent Med. 2022;62:614–25.
Denova-Gutiérrez E, Vargas-Chanes D, Hernández S, Muñoz-Aguirre P, Napier D, Barquera S. Asociación entre las inequidades socioeconómicas y la diabetes tipo 2 mediada por la obesidad y factores del estilo de vida en adultos mexicanos: un enfoque de modelos de ecuaciones estructurales. Salud Pública México. 2020;62:192–202.
Yu L, Zhai Y, Shen S. Association between vitamin D and prediabetes: a PRISMA-compliant meta-analysis. Medicine. 2020;99:e19034.
Zhang Y, Tan H, Tang J, Li J, Chong W, Hai Y, et al. Effects of vitamin D supplementation on prevention of type 2 diabetes in patients with prediabetes: a systematic review and meta-analysis. Diabetes Care. 2020;43:1650–8.
Pathak K, Soares M, Calton E, Zhao Y, Hallett J. Vitamin D supplementation and body weight status: a systematic review and meta-analysis of randomized controlled trials. Obes Rev. 2014;15:528–37.
Heaney, RP. Vitamin D: Role in The Calcium and Phosphorus Economies. Vitamin D 607–624 (Elsevier, 2011).
Szymczak-Pajor I, Śliwińska A. Analysis of association between vitamin D deficiency and insulin resistance. Nutrients. 2019;11:794.
Davidson MB, Duran P, Lee ML, Friedman TC. High-dose vitamin D supplementation in people with prediabetes and hypovitaminosis D. Diabetes Care. 2013;36:260–6.
Palomer X, González-Clemente JM, Blanco-Vaca F, Mauricio D. Role of vitamin D in the pathogenesis of type 2 diabetes mellitus. Diabetes Obes Metab. 2008;10:185–97.
Niroomand M, Fotouhi A, Irannejad N, Hosseinpanah F. Does high-dose vitamin D supplementation impact insulin resistance and risk of development of diabetes in patients with pre-diabetes? A double-blind randomized clinical trial. Diabetes Res Clin Pract. 2019;148:1–9.
Zarrin R, Ayremlou P, Ghassemi F. The effect of vitamin D supplementation on the glycemic status and the percentage of body fat mass in adults with prediabetes: a randomized clinical trial. Iran Red Crescent Med J. 2016;3:1–8.
Hoseini SA, Aminorroaya A, Iraj B, Amini M. The effects of oral vitamin D on insulin resistance in pre-diabetic patients. J Res Med Sci Off J Isfahan Univ Med Sci. 2013;18:47.
Amirkhizi F, Khademi Z, Hamedi− Shahraki S, Rahimlou M. Vitamin D insufficiency and its association with adipokines and atherogenic indices in patients with metabolic syndrome: a case-control study. Front Endocrinol. 2023;14:1080138.
Zorlu M, Kiskac M, Cakirca M, Karatoprak C, Güler E, Çelik K, et al. Evaluation of the relation between vitamin d and serum omentin and vaspin levels in women. Exp Clin Endocrinol Diabetes. 2016;124:440–3.
Fazelian S, Paknahad Z, Khajehali L, Kheiri S, Amani R. The effects of supplementation with vitamin D on inflammatory biomarkers, omentin, and vaspin in women with type 2 diabetes: a randomized double-blind placebo-controlled clinical trial. J Food Biochem. 2018;42:e12631.
Cheshmazar E, Hosseini AF, Yazdani B, Razmpoosh E, Zarrati M. Effects of vitamin D supplementation on omentin-1 and spexin levels, inflammatory parameters, lipid profile, and anthropometric indices in obese and overweight adults with vitamin D deficiency under low-calorie diet: a randomized placebo controlled trial. Evid Based Complement Altern Med. 2020;2020:3826237.
Muscogiuri G, Altieri B, de Angelis C, Palomba S, Pivonello R, Colao A, Orio F. Shedding new light on female fertility: the role of vitamin D. Rev Endocr Metab Disord. 2017;18:273–83.
Jensen MB. Vitamin D metabolism, sex hormones, and male reproductive function. Reproduction. 2012;144:135–52.
World Health Organization. Handbook for Good Clinical Research Practice (GCP): Guidance for Implementation (World Health Organization, 2005).
Rajabi-Naeeni M, Dolatian M, Qorbani M, Vaezi AA. The effect of omega-3 and vitamin D co-supplementation on glycemic control and lipid profiles in reproductive-aged women with pre-diabetes and hypovitaminosis D: a randomized controlled trial. Diabetol Metab Syndr. 2020;12:1–1.
Holick MF, Binkley NC, Bischoff-Ferrari HA, Gordon CM, Hanley DA, Heaney RP, Murad MH, Weaver CM. Evaluation, treatment, and prevention of vitamin D deficiency: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2011;96:1911–30.
Craig CL, Marshall AL, Sjöström M, Bauman AE, Booth ML, Ainsworth BE, et al. International physical activity questionnaire: 12-country reliability and validity. Med Sci Sports Exerc. 2003;35:1381–95.
Pisprasert V, Ingram KH, Lopez-Davila MF, Munoz AJ, Garvey WT. Limitations in the use of indices using glucose and insulin levels to predict insulin sensitivity: impact of race and gender and superiority of the indices derived from oral glucose tolerance test in African Americans. Diabetes Care. 2013;36:845–53.
Katz A, Nambi SS, Mather K, Baron AD, Follmann DA, Sullivan G, et al. Quantitative insulin sensitivity check index: a simple, accurate method for assessing insulin sensitivity in humans. J Clin Endocrinol Metab. 2000;85:2402–10.
Little RJ, Rubin DB. Statistical Analysis With Missing Data (John Wiley & Sons, 2019).
Association AD. Standards of medical care in diabetes—2014. Diabetes Care. 2014;37:S14–S80.
Tabák AG, Herder C, Rathmann W, Brunner EJ, Kivimäki M. Prediabetes: a high-risk state for diabetes development. Lancet. 2012;379:2279–90.
Holick MF, Binkley NC, Bischoff-Ferrari HA, Gordon CM, Hanley DA, Heaney RP, et al. Guidelines for preventing and treating vitamin D deficiency and insufficiency revisited. J Clin Endocrinol Metab. 2012;97:1153–8.
Bhatt SP, Misra A, Pandey RM, Upadhyay AD, Gulati S, Singh N. Vitamin D supplementation in overweight/obese Asian Indian women with prediabetes reduces glycemic measures and truncal subcutaneous fat: a 78 weeks randomized placebo-controlled trial (PREVENT-WIN Trial). Sci Rep. 2020;10:220.
Tuomainen T-P, Virtanen JK, Voutilainen S, Nurmi T, Mursu J, de Mello VD, et al. Glucose metabolism effects of vitamin D in prediabetes: the vitdmet randomized placebo-controlled supplementation study. J Diabetes Res. 2015;2015:672653.
Lu J, Liu Y, Zhang J, Lou P, Liu Y, Zhou Y. Effect of vitamin D intervention on the outcome of glucose metabolism in patients with impaired glucose regulation. Chin J Clin Nutrition. 2019;6:349–53.
Chiu KC, Chu A, Go VL, Saad MF. Hypovitaminosis D is associated with insulin resistance and β cell dysfunction. Am J Clin Nutr. 2004;79:820–5.
MAESTRO B, CAMPIÓN J, DÁVILA N, CALLE C. Stimulation by 1, 25-dihydroxyvitamin D3 of insulin receptor expression and insulin responsiveness for glucose transport in U-937 human promonocytic cells. Endocrine J. 2000;47:383–91.
Pittas AG, Lau J, Hu FB, Dawson-Hughes B. The role of vitamin D and calcium in type 2 diabetes. A systematic review and meta-analysis. J Clin Endocrinol Metab. 2007;92:2017–29.
Contreras-Bolívar V, García-Fontana B, García-Fontana C, Muñoz-Torres M. Mechanisms involved in the relationship between vitamin D and insulin resistance: impact on clinical practice. Nutrients. 2021;13:3491.
Zatterale F, Longo M, Naderi J, Raciti GA, Desiderio A, Miele C, Beguinot F. Chronic adipose tissue inflammation linking obesity to insulin resistance and type 2 diabetes. Front Physiol. 2020;10:1607.
Wang TJ, Zhang F, Richards JB, Kestenbaum B, Van Meurs JB, Berry D, Kiel DP, Streeten EA, Ohlsson C, Koller DL, Peltonen L. Common genetic determinants of vitamin D insufficiency: a genome-wide association study. Lancet. 2010;376:180–8.
Usategui-Martín R, De Luis-Román DA, Fernández-Gómez JM, Ruiz-Mambrilla M, Pérez-Castrillón JL. Vitamin D receptor (VDR) gene polymorphisms modify the response to vitamin D supplementation: a systematic review and meta-analysis. Nutrients. 2022;14:360.
Mitri J, Muraru M, Pittas A. Vitamin D and type 2 diabetes: a systematic review. Eur J Clin Nutr. 2011;65:1005–15.
Jafari T, Faghihimani E, Feizi A, Iraj B, Javanmard SH, Esmaillzadeh A, Fallah AA, Askari G. Effects of vitamin D-fortified low fat yogurt on glycemic status, anthropometric indexes, inflammation, and bone turnover in diabetic postmenopausal women: a randomised controlled clinical trial. Clin Nutr. 2016;35:67–76.
Liu E, Meigs JB, Pittas AG, McKeown NM, Economos CD, Booth SL, et al. Plasma 25-hydroxyvitamin D is associated with markers of the insulin resistant phenotype in nondiabetic adults. Journal Nutr. 2009;139:329–34.
Yang Y, Yan S, Yao N, Guo Y, Wang H, Sun M, et al. Effects of vitamin D supplementation on the regulation of blood lipid levels in prediabetic subjects: a meta-analysis. Front Nutr. 2023;10:983515.
Salehpour A, Shidfar F, Hosseinpanah F, Vafa M, Razaghi M, Amiri F. Does vitamin D3 supplementation improve glucose homeostasis in overweight or obese women? A double-blind, randomized, placebo-controlled clinical trial. Diabetic Med. 2013;30:1477–81.
Zhou J, Zhao L-J, Watson P, Zhang Q, Lappe JM. The effect of calcium and vitamin D supplementation on obesity in postmenopausal women: secondary analysis for a large-scale, placebo controlled, double-blind, 4-year longitudinal clinical trial. Nutr Metab. 2010;7:1–9.
Rosenblum JL, Castro VM, Moore CE, Kaplan LM. Calcium and vitamin D supplementation is associated with decreased abdominal visceral adipose tissue in overweight and obese adults. Am J Clin Nutr. 2012;95:101–8.
George P, Pearson E, Witham M. Effect of vitamin D supplementation on glycaemic control and insulin resistance: a systematic review and meta-analysis. Diabetic Med. 2012;29:e142–e50.
Vanlint S. Vitamin D and obesity. Nutrients. 2013;5:949–56.
Griz LHM, Bandeira F, Gabbay MAL, Dib SA, Carvalho EFD. Vitamin D and diabetes mellitus: an update 2013. Arq Bras Endocrinol Metab. 2014;58:1–8.
Didriksen A, Grimnes G, Hutchinson MS, Kjærgaard M, Svartberg J, Joakimsen RM, et al. The serum 25-hydroxyvitamin D response to vitamin D supplementation is related to genetic factors, BMI, and baseline levels. Eur J Endocrinol. 2013;169:559–67.
Li X, Liu Y, Zheng Y, Wang P, Zhang Y. The effect of vitamin D supplementation on glycemic control in type 2 diabetes patients: a systematic review and meta-analysis. Nutrients. 2018;10:375.
Drincic A, Fuller E, Heaney RP, Armas LA. 25-Hydroxyvitamin D response to graded vitamin D3 supplementation among obese adults. J Clin Endocrinol Metab. 2013;98:4845–51.
Bischoff-Ferrari HA, Giovannucci E, Willett WC, Dietrich T, Dawson-Hughes B. Estimation of optimal serum concentrations of 25-hydroxyvitamin D for multiple health outcomes. Am J Clin Nutr. 2006;84:18–28.
Charoenngam N, Holick MF. Immunologic effects of vitamin D on human health and disease. Nutrients. 2020;12:2097.
Acknowledgements
We thank the Nutrition Research Center of Tabriz University of Medical Sciences, Tabriz, Iran, and the Iran National Science Foundation for their financial support. This article has been written based on a data set of Ph.D. thesis registered at Tabriz University of Medical Sciences, Tabriz, Iran (ID: 72938). We also thank the participants of this study.
Funding
This work was supported by the Nutrition Research Center of Tabriz University of Medical Sciences, Tabriz, Iran (grant number: 72938) and the Iran National Science Foundation (grant number: 4030940). The funders had no role in study design, data collection and analysis, or manuscript writing.
Author information
Authors and Affiliations
Contributions
MR, RMG, and MAJ designed the study. RMG and SAY contributed to data collection. MAJ conducted the statistical analysis, and RMG wrote the manuscript draft. MR and RMG contributed to the discussion and revision of the manuscript. MR, RMG, and MAJ critically reviewed the manuscript. All authors read the manuscript and approved the integrity of the study procedure.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Molani-Gol, R., Rafraf, M., Asghari Jafarabadi, M. et al. Effects of vitamin D supplementation on metabolic factors, serum omentin-1, and anthropometric indices in middle-aged women with prediabetes: a double-blind randomized controlled trial. Nutr. Diabetes 15, 44 (2025). https://doi.org/10.1038/s41387-025-00402-w
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41387-025-00402-w