Abstract
Background
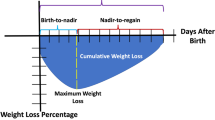
Time to regain birthweight (time-to-regain) includes subcomponents of time from birth to weight loss nadir (birth-to-nadir), time from weight loss nadir to regain birthweight (nadir-to-regain), and cumulative weight loss (CWL; sum of daily percent weight loss from birthweight until regain), yet relationship with in-hospital outcomes remains unknown.
Methods
We performed a secondary analysis of the multicenter Preterm Erythropoietin Neuroprotection (PENUT) Trial including extremely preterm (EP) newborns at 24w0d-27w6d gestation at birth evaluating time-to-regain and its subcomponents with in-hospital adverse outcomes.
Results
Among n = 876 EP newborns, time-to-regain was not significantly associated with in-hospital outcomes. CWL had a U-shaped distribution with >2.5-fold increased odds of NEC for those without weight loss and moderate or severe CWL when compared with mild CWL (>0 to −23 percent-days). Increasing birth-to-nadir was associated with increased odds of NEC (linear trend analysis aOR 1.33, 95% CI 1.05, 1.69). Total fluid intake and time-to-regain are directly related, with total fluid intake strongly associated with markers of severity of illness.
Conclusions
Among EP neonates, time-to-regain is not associated with in-hospital outcomes and is most associated with severity of illness metrics. CWL and birth-to-nadir appear to be more optimal nutritional and fluid status metrics.
Trial registration
This study is a post-hoc secondary analysis of pre-existing data from the PENUT Trial (NCT #01378273). PENUT Trial Registration: NCT01378273. https://clinicaltrials.gov/ct2/show/NCT01378273.
Impact
-
Time to regain birthweight was not associated with in-hospital adverse outcomes, yet cumulative weight loss is significantly associated with necrotizing enterocolitis and is related to long-term neurodevelopmental outcomes.
-
Time to regain birthweight does not appear to be an optimal metric of nutritional or hydration status among extremely preterm newborns.
-
Cumulative weight loss’s relation to both in-hospital and long-term outcomes suggest it is a more promising indicator of optimal nutrition and hydration status.
-
These findings point to the need to revisit current expert recommendations stating that prolonged time to regain birthweight among preterm neonates is a metric of malnutrition; instead, cumulative weight loss may be a more ideal metric.
Similar content being viewed by others
Introduction
The optimal time to regain birthweight (time-to-regain) for extremely preterm (EP) newborns, defined as those born at <28 weeks’ gestation, is currently unknown. Although clinicians caring for EP newborns routinely evaluate daily weights or sodium values to make nutrition- and fluid-based clinical decisions, there is sparse data on whether time-to-regain is associated with short- or long-term harm or benefit.
We previously published that time-to-regain consists of at least three sub-components including: time from birth to the weight loss nadir (birth-to-nadir), time from the weight loss nadir to regain of birthweight (nadir-to-regain), and cumulative weight loss (CWL) defined as the sum of the weight loss percentage for every day below birthweight.1 While published national recommendations suggest longer time-to-regain among preterm newborns is associated with malnutrition,2 longer time-to-regain, increasing CWL, and a more rapid nadir-to-regain have recently been reported to be associated with adverse neurodevelopmental outcomes at two years of age among EP newborns.1 Yet, it remains unclear whether time-to-regain or its sub-components are associated with different in-hospital outcomes among EP newborns.
Via a post-hoc, secondary analysis of the Preterm Erythropoietin (Epo) Neuroprotection (PENUT) Trial,3 we sought to evaluate the association between time-to-regain and its sub-components (birth-to-nadir, nadir-to-regain, CWL) on in-hospital mortality (primary outcome), length of hospital stay, and the following morbidities: necrotizing enterocolitis (NEC), bronchopulmonary dysplasia (BPD), and retinopathy of prematurity (ROP) requiring intervention. Consistent with our previously published findings on 24-month corrected gestational age neurodevelopmental outcomes,1 we hypothesized that a faster time-to-regain, nadir-to-regain and increasingly negative CWL would be associated with increased odds of in-hospital mortality and morbidity as well as a longer length of stay among EP neonates in the United States.
Methods
The PENUT Trial was a double-blinded, placebo-controlled, individually-randomized trial of EP newborns (born between 24 weeks 0 days – 27 weeks 6 days) across 30 neonatal intensive care units in the United States that evaluated whether Epo administration compared to placebo affected 2-year neurodevelopmental outcomes.3 Each study participant’s legal guardian provided informed consent to be enrolled in the PENUT Trial, and institutional review board (IRB) approval was obtained at each site prior to study commencement. The PENUT Trial study protocol and primary outcomes have been previously published.3
We performed a post-hoc secondary analysis of PENUT Trial infants who survived at least 14 days after birth and had daily weight information recorded. A subset of n = 876 of the n = 941 EP infants were therefore included in this analysis.
Statistical analyses
The exposure variables included time-to-regain and its sub-components (birth-to-nadir, nadir-to-regain, and CWL). Outcome analyses were performed by evaluating quartiles of each of these exposure variables with the clinical outcomes of all-cause in-hospital mortality (primary outcome), NEC, BPD, ROP, and length of stay. NEC was defined as Bell’s stage II or higher,4,5 BPD was defined as requiring nasal cannula or higher levels of respiratory support at 36 weeks postmenstrual age,6 and ROP was defined as any ROP requiring intervention such as treatment with laser photocoagulation, cryotherapy, or bevacizumab.
The quartiles for the exposure variables have been previously reported.1 Time-to-regain quartiles were approximated to the nearest day after birth: ≤5 days (including participants who never lost weight), 6–8 days, 9–11 days, and ≥12 days (including those who had not regained birthweight by day 14).1 The quartiles for the time-to-regain subcomponents as previously published and utilized within this study’s analyses are as follows: birth-to-nadir quartiles: ≤1 day, 2 days, 3–4 days, ≥5 days; nadir-to-regain quartiles: ≤1 day, 2–4 days, 5–7 days, and ≥8 days; cumulative weight loss quartiles: <−50 percent-days, −50 to <−23 percent-days, −23 to <0 percent-days, and ≥0 percent-days.1 Also, while maximal weight loss percentage is a sub-component of time-to-regain, the associations between maximal weight loss percentage and in-hospital outcomes among PENUT Trial participants has already been published.7 Because daily weight data were not available as part of the PENUT Trial beyond day 14 after birth, for calculation of time-to-regain, any infant who had not regained their birthweight by day 14 was assumed to have regained their birthweight on day 15 after birth.
For all inferential analyses, generalized estimating equations (GEE) with robust standard errors were used to appropriately account for potential correlation of outcomes for same-birth siblings.3 Baseline and demographic factors were compared across quartiles of time-to-regain using a multivariate Wald test. Fluid administration over the first 14 days was averaged relative to birthweight to provide a value of mL/kg birthweight/day.
Using GEE linear regression models adjusting for gestational age (GA) and Epo treatment group, we examined the association between maternal and infant factors and time-to-regain quartile. Infant factors that were clinically relevant and associated with faster time-to-regain (defined as p ≤ 0.10) were then included as additional potential confounders when assessing the association between time-to-regain and in-hospital clinical outcomes. Maternal factors significantly associated with time-to-regain were not included as confounders in the final outcomes models as they were all associated with birthweight, as detailed in the Results section (Table 1). Final regression models were adjusted for GA, treatment group, mode of delivery, vasopressor use in the first 14 days (including dopamine, norepinephrine, epinephrine, dobutamine, vasopressin, or milrinone), postnatal steroid use in the first 14 days (hydrocortisone or dexamethasone), birthweight z-score, mechanical ventilation at baseline, and quartile of average total fluids in the first 14 days after birth. Only neonates surviving at least 14 days were included in mortality, NEC, and length of stay analyses. Neonates surviving to 36 weeks postmenstrual age were included in BPD and ROP analyses. Multivariable logistic regression models were used for binary outcomes and linear regression model were used for length of stay analyses. Relative to a reference quartile, we estimated the adjusted odds ratio (aOR) associated with a given quartile of time-to-regain, birth-to-nadir, nadir-to-regain, and CWL, all of which were calculated as previously published.1
Finally, to examine the relationships between variables known to be associated with time-to-regain and its sub-components (birth-to-nadir, nadir-to-regain, and CWL), we performed graphical network analyses. Graphical network analyses elucidate significant relationships using a precision matrix of interrelated variables allowing for the identification of relationships after accounting for the interconnectedness of the other variables with one another. This methodology is consistent with the method described by Williams and Rast and our previous publication.8,9 The precision matrix was constructed using a maximum likelihood estimation (MLE) method. Significant relationships were determined using Fisher Z-transformed 95% confidence intervals (CI), resulting in stable networks in which the number of predictors is fewer than the number of observations. Within each graphical network, red lines indicate a significant negative association, and blue lines indicate a significant positive association after all relationships in the network are considered. The width of each line demonstrates the magnitude of the association between variables.
The PENUT trial originally enrolled 941 infants. Five were excluded from the sample: four who died prior to administration of erythropoietin (Epo) or placebo and one who withdrew due to complications with consent, resulting in an effective baseline cohort of 936 infants. For the present study, infants who did not survive the first 14 days after birth (n = 60) were excluded from all analyses, resulting in the total study including n = 876 infants. Cohort size varied by outcome. For analyses requiring survival to 36 weeks corrected gestational age (CGA), such as bronchopulmonary dysplasia (BPD) and retinopathy of prematurity (ROP), 89 infants were excluded (n = 847). For NEC analyses, 18 infants who developed necrotizing enterocolitis (NEC) stage IIA or greater prior to 14 days after birth were excluded from the 876-infant dataset, resulting in n = 858. For length of stay analyses, only infants who survived to discharge home were included (n = 828).
Throughout all analyses, a p < 0.05 was considered statistically significant. All analyses were conducted using the R statistical package (Version 3.6.1, Foundation for Statistical Computing, Vienna, Austria).10
Results
A total of 876 eligible infants had documented weight data to day 14, survived at least 14 days, and were included in this secondary analysis. Of these, 124 (11.5%) infants did not regain birthweight by day 14 after birth. Table 1 shows the maternal and infant characteristics of the overall cohort and by time-to-regain quartiles. Maternal characteristics that were significantly different included: maternal age, pregnancy-induced hypertension, pre-eclampsia, gestational diabetes, antenatal corticosteroid exposure, and cesarean delivery. Infant characteristics that were significantly different based on time-to-regain quartiles were: 5-min Apgar score, birthweight (grams), birthweight z-score, postnatal steroid exposure, postnatal vasopressor exposure, mechanical ventilation, and average total fluid administration over the first 14 days after birth. Of the included infants, n = 97 experienced severe intracranial hemorrhage (Grade III or Grade IV), which was not different across time-to-regain groups (Table 1).
Association between time-to-regain and in-hospital mortality and morbidities
When evaluating the relationship between time-to-regain and in-hospital mortality and morbidities, we first evaluated time-to-regain quartiles with each outcome. Neither unadjusted or adjusted models demonstrated any significant association between time-to-regain quartiles with in-hospital mortality, NEC, or length of stay (Table 2). In unadjusted models, time-to-regain ≤5 days was associated with increased odds of BPD when compared to the reference quartile of ≥12 days (OR 1.74, 95% Confidence Interval [CI] 1.14, 2.66, p = 0.01), but was no longer significantly associated with BPD after adjusting for known confounders (aOR 1.25, 95% CI 0.74, 2.12, p = 0.40). Regarding ROP, in unadjusted models, time-to-regain quartile of 6–8 days was not statistically associated with ROP when compared with time-to-regain of ≥12 days (OR 0.57, 95% CI 0.26, 1.22, p = 0.15). While the association became statistically significant in the multivariate model (aOR 0.38, 95% CI 0.17–0.85, p = 0.02), upon linear trend analyses there was no significant association with time-to-regain and any outcome assessed, including ROP (Table 2).
Association between nadir-to-regain and in-hospital mortality and morbidities
When evaluating quartiles of nadir-to-regain with in-hospital mortality and morbidities, there were no significant associations between nadir-to-regain quartiles and in-hospital mortality, BPD, NEC, or ROP in any multivariate models including adjustment with metrics of severity of illness (Supplemental Table S1). All linear trend analyses similarly demonstrate no significant findings between nadir-to-regain and any assessed outcome.
Association between birth-to-nadir and in-hospital mortality and morbidities
When evaluating quartiles of birth-to-nadir with in-hospital mortality and morbidities, the only significant finding was in relationship with NEC. When compared with a birth-to-nadir of 3–4 days, shorter birth-to-nadir of <2 days and 2 days were both significantly associated with reduced odds of NEC after adjusting for metrics of severity of illness (<2 days aOR 0.32, 95% CI: 0.14, 0.74, p = 0.007; 2 days aOR 0.40, 95% CI 0.17, 0.95, p = 0.04). Significance was retained with linear trend analysis (aOR 1.33, 95% CI 1.05, 1.69, p = 0.02), highlighting increased odds of developing NEC with increasing days of birth-to-nadir (Table 3). There were no other significant associations between birth-to-nadir quartiles or linear trend analysis with any other assessed outcome.
Association between CWL and in-hospital mortality and morbidities
There were no significant associations between CWL quartiles and in-hospital mortality, BPD, or length of stay. In unadjusted analyses, CWL ≥ 0 percent-days (representing no weight loss after birth) was associated with increased odds of NEC (Bell’s stage IIA or higher) when compared to CWL with mild weight loss (−23 to <0 percent days) in adjusted analyses (aOR 2.57, 95% CI 1.07–6.19, p = 0.04). CWL representing moderate weight loss after birth (−50 to <−23 percent-days) was similarly significantly associated with increased odds of NEC when compared with CWL with mild weight loss (−23 to <0 percent days) in adjusted analyses (aOR 2.69, 95% CI 1.02, 7.08, p = 0.045) with similar trend found in more severe CWL (<−50 percent-days; aOR 2.55, 95% CI 0.97, 6.75, p = 0.04), see Table 4.
More severe CWL (<−50 percent-days) was associated with reduced odds of ROP when compared with CWL −23 to <0 percent-days in unadjusted analyses (OR 0.46, 95% CI 0.22, 0.95, p = 0.04), this relationship became nonsignificant within adjusted models (aOR 0.67, 95% CI 0.28, 1.56, p = 0.35). Because of the known non-linear, U-shaped association between CWL with neurodevelopmental outcomes as previously published,1 we did not perform linear trend analyses for CWL with outcomes.
Graphical network analyses
Increasing average total fluid intake (TFI) over the first 14 days, increasing GA, caesarean delivery, and small for gestational age (SGA) status were all associated with a shorter time-to-regain (Supplemental Fig. S1). However, when evaluating relationships with the addition of the subcomponents of time-to-regain such as maximum weight loss percentage, time from birth to weight loss nadir, time from nadir to regain of birthweight, and CWL, only increasing TFI remained associated with a shorter time-to-regain (Fig. 1). Increasing TFI was associated with vasopressor use, postnatal steroids, respiratory distress, and decreasing GA. Increasing GA was associated with decreased TFI, less respiratory distress, and less exposure to vasopressors or antenatal corticosteroids. CWL was positively related to birth-to-nadir.
Red lines indicate a significant negative association, with blue lines indicating a significant positive association after all relationships in the network are taken into account. Only increasing total fluid intake (TFI) remained associated with a shorter time to regain once considering the subcomponents of time-to-regain. Increasing gestational age (GA) was associated with decreased TFI, less respiratory distress, and less exposure to vasopressors or antenatal corticosteroids. Small for gestational age (SGA) status was no longer associated with time-to-regain, but it was associated with increased TFI, caesarean delivery, and increased GA. *Male sex was not associated with any of the other variables in the network. TFI was assessed on a continuous scale.
Discussion
Questioning the utility of time-to-regain as an optimal nutritional metric
Time-to-regain is not significantly associated with any assessed in-hospital adverse health outcomes among EP newborns after adjusting for metrics of severity of illness. Graphical network analyses demonstrate substantial interconnectedness between overall TFI and time-to-regain, and TFI is strongly associated with metrics of illness severity including GA, need for mechanical ventilation, postnatal vasopressor use, and postnatal steroid use. Taken with the lack of association with in-hospital outcomes or neurodevelopmental outcomes,1 these findings suggest time-to-regain is likely moreso a metric of severity of illness than a robust nutritional metric.
Current expert recommendations include time-to-regain of >14 days as an indicator of malnutrition among preterm.2 Yet, the evidence presented to support these recommendations focus on growth metrics with neither clinical correlation to in-hospital outcomes nor long-term outcomes such as neurodevelopment.11,12,13,14,15 The reliance on weight gain or growth velocities as a sole determinant of what is optimal for nutritional metrics among premature newborns is questionable without any evaluation of impact on in-hospital or long-term outcomes. For example, two published multi-center cohorts report that in-hospital linear and weight growth failure among EP neonates is not associated with 2-year neurodevelopmental outcomes.16,17,18 Promoting more rapid time-to-regain may not result in improved long-term outcomes and may be associated with harm, as evidenced by a faster nadir-to-regain being associated with significantly worse neurodevelopmental outcomes.1 Thus, revisiting metrics of optimal nutritional intake, including defining what constitutes malnutrition among premature newborns, is of critical importance.
Our findings are further corroborated by another large study by the National Institute for Child Health and Development Neonatal Research Network that similarly evaluated quartiles of growth velocity on 18–22-month neurodevelopmental outcomes in ELBW neonates.17 The quartile with the highest growth velocity had the longest time-to-regain as well as the lowest occurrence of neurodevelopmental impairment, cerebral palsy, or rehospitalization after discharge from the NICU.17 Remarkably, 49% of the newborns within the quartile with the best neurodevelopmental outcomes had not regained their birthweight by 18 days after birth,17 which is defined as mild malnutrition or worse by current expert recommendations.2
Collectively, our study’s findings along with the increasing body of evidence lacking association of time-to-regain with in-hospital and/or neurodevelopmental outcomes emphasizes the need to revisit optimal metrics for nutritional status among premature newborns and calls into question the current recommendations that a time-to-regain >14 days is an indicator of malnutrition.
Cumulative weight loss may be a superior metric of nutritional and fluid status among EP neonates
The inclusion of both weight loss and duration of weight loss, encompassed by the subcomponent of CWL, may be a superior metric of nutritional and fluid status during the critical fetal-to-neonatal transition period among EP neonates. CWL has a U-shaped association with developing NEC, with >2.5-fold reduced odds of developing NEC with lack of weight loss (CWL ≥ 0 percent-days) or moderate-to-severe weight loss (CWL ≤ −23 percent-days). Of note, while not reaching statistical significance (p = 0.06), severe CWL (defined as <−50 percent-days) similarly had a > 2.5-fold reduced odds of developing NEC, which suggests a potential type 2 error related to the sample size not being able to have adequate power with the multiple covariate regression modeling. Taken together, these findings highlight a consistent finding that a CWL of −23 to <0 percent-days appears most optimal to reduce the odds of developing NEC. While we cannot discern the reasons for the U-shaped associations with NEC, it may be that too little weight loss is associated with generalized edema, including intestinal edema, whereas too much weight loss may be associated with dehydration and splanchnic vasoconstriction. Future studies are needed to explore the potential underlying pathophysiological processes for these significant findings.
Similarly, CWL has a U-shaped association with neurodevelopmental outcomes at 2 years after birth, associated with 4–6 point reduced Bayley Scales of Infant and Toddler Development scores in all assessed domains (cognitive, motor, and language).1,19 Thus, CWL is strongly associated with both in-hospital and long-term neurodevelopmental outcomes of EP neonates, even after accounting for metrics of severity of illness.
While birth-to-nadir was associated with the development of NEC, it is strongly and directly related to CWL as evidenced via graphical network analyses. These findings suggest that EP newborns who have more severe CWL rapidly lose weight soon after birth (a shorter birth-to-nadir). Yet, birth-to-nadir was not found to be associated with neurodevelopmental outcomes.1 Notably, there was nearly a 33% significantly increased odds of developing NEC for each day longer to reach the weight loss nadir. Yet, while birth-to-nadir appears strongly related to odds of developing NEC, CWL may be a more ideal metric of early nutritional and fluid status given its relationship with NEC, long-term outcomes and birth-to-nadir. Further prospective studies and/or clinical trials are needed to confirm these findings.
CWL is defined as the sum of each day’s weight loss until regaining birthweight, which includes the units of percent-days. An example of CWL would be the following: if a newborn is 6% down from birthweight on day 1, 10% down from birthweight on day 2, 5% down from birthweight on day 3, with regain of birthweight on day 4, then the CWL would be −6 + −10 + −5, or −21 percent-days. Such a metric could be integrated within electronic medical records to facilitate daily monitoring by clinicians as is similarly custom for the weight percentages from growth curves as daily weights are recorded. While a likely new term for neonatologists and clinicians, CWL has the strongest association with both in-hospital and long-term outcomes whereas time-to-regain does not. A fundamental reconsideration of whether time-to-regain is an ideal metric given its lack of significant associations with outcomes is imperative.
Strengths and limitations
Our study has notable strengths including the relatively large sample size of 876 EP neonates representing 19 academic centers and 30 NICUs across the United States,3,20 increasing the generalizability of our findings. Additionally, the study includes a relatively contemporary cohort of EP neonates that more accurately reflects current clinical practice.
Our study has several limitations. First, our study is a post-hoc secondary analysis of a randomized trial that did not record weights after 14 days which can only provide insight into association but not causation. Moreover, our findings are likely not generalizable to NICUs in low- and middle-income countries. Additionally, while we included metrics of severity of illness within our multivariate regression models, it is likely that additional metrics or confounding variables were not available to include in the models. For example, there is a lack of detail within the PENUT Trial related to the enteral and parenteral contributions to total fluid intake, which may have differential effects on outcomes. Thus, future studies are needed to explore the impact of enteral compared to parenteral intake in the first 2 weeks after birth on EP neonatal outcomes. Future studies also are needed to explore the relationship between early nutritional practices, such as macro- and micro-nutrient intake, on both short- and long-term outcomes. Furthermore, it is unclear whether anthropometric evaluation used in isolation is similar to, inferior, or superior to other modalities for assessing lean body mass and their relationship with outcomes.
Conclusion
Rapid time-to-regain does not translate to improved in-hospital outcomes among EP neonates. While current expert recommendations suggest time-to-regain within 14 days represents optimal nutrition for premature neonates, our findings indicate that there are no significant short- or long-term benefits with this practice. In fact, there may be harm related to neurodevelopment. Our findings reveal that a rapid time-to-regain is more consistent with metrics of illness severity rather than optimal nutritional intake. Contrary to current recommendations, promoting a rapid time-to-regain may not be ideal for EP neonates. However, birth-to-nadir and cumulative weight loss, a novel metric which includes both weight loss severity and duration of weight loss, is strongly associated with adverse in-hospital (NEC) and neurodevelopmental outcomes highlighting that these time-to-regain subcomponents may be moreso optimal metrics of nutritional and fluid status among EP neonates in the first week after birth rather than time-to-regain itself. Revisiting current recommendations for identifying optimal nutritional metrics of premature newborns is critically needed.
Data availability
De-identified individual participant data is available through the NINDS Data Archive: https://www-ninds-nih-gov.offcampus.lib.washington.edu/Current-Research/Research-Funded-NINDS/Clinical-Research/Archived- Clinical-Research-Datasets. The data is de-identified, and a limited access data set is available through a request form on that page. Data dictionaries, in addition to study protocol, the statistical analysis plan, and the informed consent form will be included. The data will be made available upon publication of all PENUT Trial related manuscripts to researchers who provide a methodologically sound proposal for use in achieving the goals of the approved proposal.
References
Valentine, G. C. et al. Time to regain birthweight and association with neurodevelopmental outcomes among extremely preterm newborns. J. Perinatol. https://doi.org/10.1038/s41372-024-01869-8 (2024).
Goldberg, D. L. et al. Identifying malnutrition in preterm and neonatal populations: recommended indicators. J. Acad. Nutr. Diet. 118, 1571–1582 (2018).
Juul, S. E. et al. A randomized trial of erythropoietin for neuroprotection in preterm infants. N. Engl. J. Med. 382, 233–243 (2020).
Bell, M. J. et al. Neonatal necrotizing enterocolitis. Ther. Decis. Based Clin. Staging Ann. Surg. 187, 1–7 (1978).
Walsh, M. C. & Kliegman, R. M. Necrotizing enterocolitis: treatment based on staging criteria. Pediatr. Clin. North Am. 33, 179–201 (1986).
Jensen, E. A. et al. The diagnosis of bronchopulmonary dysplasia in very preterm infants. an evidence-based approach. Am. J. Respir. Crit. Care Med. 200, 751–759 (2019).
Valentine, G. C. et al. Postnatal maximal weight loss, fluid administration, and outcomes in extremely preterm newborns. J Perinatol. https://doi.org/10.1038/s41372-022-01369-7 (2022).
Juul, S. E. et al. Predicting 2-year neurodevelopmental outcomes in extremely preterm infants using graphical network and machine learning approaches. EClinicalMedicine 56, 101782 (2023).
Williams, D. R. & Rast, P. Back to the basics: rethinking partial correlation network methodology. Br. J. Math. Stat. Psychol. 73, 187–212 (2020).
R Core Team. R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing, Vienna, Austria. https://www.R-project.org/ (2025).
Rochow, N. et al. A nutritional program to improve outcome of very low birth weight infants. Clin. Nutr. 31, 124–131 (2012).
Senterre, T. & Rigo, J. Optimizing early nutritional support based on recent recommendations in VLBW infants and postnatal growth restriction. J. Pediatr. Gastroenterol. Nutr. 53, 536–542 (2011).
Paul, I. M. et al. Weight change nomograms for the first month after birth. Pediatrics 138 https://doi.org/10.1542/peds.2016-2625 (2016).
Moyer-Mileur, L. J. Anthropometric and laboratory assessment of very low birth weight infants: the most helpful measurements and why. Semin Perinatol. 31, 96–103 (2007).
Sakurai, M., Itabashi, K., Sato, Y., Hibino, S. & Mizuno, K. Extrauterine growth restriction in preterm infants of gestational age < or =32 weeks. Pediatr. Int. 50, 70–75 (2008).
Strobel, K. M. et al. Contemporary definitions of infant growth failure and neurodevelopmental and behavioral outcomes in extremely premature infants at two years of age. J Perinatol. https://doi.org/10.1038/s41372-023-01852-9 (2024).
Ehrenkranz, R. A. et al. Growth in the neonatal intensive care unit influences neurodevelopmental and growth outcomes of extremely low birth weight infants. Pediatrics 117, 1253–1261 (2006).
Salas, A. A. et al. Risk assessment of cognitive impairment at 2 years of age in infants born extremely preterm using the INTERGROWTH-21st growth standards. J. Pediatr. 275, 114239 (2024).
Balasundaram, P. & Avulakunta, ID. Bayley Scales Of Infant and Toddler Development. [Updated 2022 Nov 21]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK567715/.
Juul, S. E., Mayock, D. E., Comstock, B. A. & Heagerty, P. J. Neuroprotective potential of erythropoietin in neonates; design of a randomized trial. Matern Health Neonatol. Perinatol. 1, 27 (2015).
Acknowledgements
We would like to acknowledge the tireless dedication and hard work on behalf of the PENUT Trial PIs, Co-Investigators, Research Coordinators, the University of Washington Data Coordinating Center, the PENUT Executive Committee, the PENUT Follow Up Committee, the Independent Medical Monitor, and funding through the NIH and NINDS. We also extend our heartfelt gratitude to each of the families and their children enrolled in the PENUT Trial.
Funding
PENUT was supported by the National Institute of Neurological Disorders and Stroke of the National Institutes of Health under award numbers U01NS077953 and U01NS077955. Dr. Valentine is supported by the National Institutes of Health under award number K23HD105929.
Author information
Authors and Affiliations
Contributions
G.C.V. conceptualized the design of the secondary analysis, composed the initial draft of the manuscript, and revised its subsequent versions. O.B. and T.R.W. performed the statistical analyses. K.M.P., K.M.S., D.E.M., J.B.L., S.N., K.G., S.K., P.J.H., and S.E.J. were involved in revisions to the manuscript, and all agreed to the final draft of the manuscript being submitted.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
Patient informed consent was obtained during the parent PENUT trial.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Valentine, G.C., Brandon, O.C., Perez, K.M. et al. Time to regain birthweight and in-hospital outcomes among United States-born extremely preterm newborns. Pediatr Res 99, 949–957 (2026). https://doi.org/10.1038/s41390-025-04563-3
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41390-025-04563-3
This article is cited by
-
Gregory Valentine: ECI biocommentary
Pediatric Research (2026)