Abstract
Background
Status epilepticus (SE) is a life-threatening pediatric emergency requiring rapid anticonvulsants. While midazolam and diazepam are standard benzodiazepines, their comparative efficacy across various administration routes remains debated. We synthesized high-quality evidence to guide clinical protocols.
Methods
We searched PubMed, Scopus, Web of Science, and Cochrane for randomized controlled trials comparing buccal, intramuscular, and intravenous formulations. We evaluated treatment outcomes including therapeutic success (seizure cessation), recurrence, and drug-related side effects. We employed meta-analysis, trial sequential analysis (TSA), and GRADE to ensure the robustness of the evidence.
Results
Nine RCT studies (n = 1135 children) were included. Midazolam demonstrated superior therapeutic success (RR = 1.13, 95% CI 1.03–1.25, p = 0.01), with TSA confirming conclusive benefit for the buccal route (RR = 1.30, p = 0.002). Midazolam significantly reduced treatment failure (RR = 0.74, 95% CI 0.57–0.95, p = 0.02) and seizure recurrence (RR = 0.51, p = 0.04). Time-to-cessation was shorter with non-intravenous routes (MD = −2.39 min, p = 0.01). Safety profiles regarding respiratory depression were comparable between groups.
Conclusion
Midazolam is the preferred first-line anticonvulsant for pediatric status epilepticus. It offers superior therapeutic success, lower failure rates, and reduced recurrence, particularly via buccal and intramuscular routes. Given comparable safety, this evidence strongly supports updating emergency medical services guidelines to prioritize non-intravenous midazolam.
Impact
-
Midazolam demonstrates superior efficacy over diazepam, particularly via buccal and intramuscular routes.
-
It addresses critical prehospital delays by bypassing the high failure rates associated with pediatric vascular access.
-
Intramuscular administration matches intravenous efficacy while enabling immediate intervention without specialized equipment.
-
Additionally, buccal and nasal formulations represent the most cost-effective non-intravenous rescue options available.
-
Guidelines must prioritize these practical routes for EMS settings, necessitating policy updates to remove insurance barriers.

Similar content being viewed by others
Introduction
Status epilepticus (SE), a medical emergency that is characterized by a prolonged seizure lasting more than 5 min or the occurrence of multiple seizures without recovery of consciousness in between, As a threatening condition, the primary goal of treatment is to stop the seizure and, in doing so, prevent brain injury. It has a high prevalence rate and potential impact on a child’s neurological development and quality of life, as the yearly occurrence of chronic seizure episodes in children ranges from 10 to 73 cases per 100,000 children, with the highest rates observed in those under 2 years old1 SE is the most common pediatric emergency, affecting about 3–42 per 100,000 children with a mortality of 3–11%.2,3 Death can result either from underlying causes, including febrile SE, the most common cause of pediatric SE, or from immediate complications, including respiratory failure, hemorrhagic shock, or structural brain lesion. Even with the success of treatment to terminate seizures, 15% of survivors can develop long-term neurologic disabilities like epilepsy and cognitive impairment. Also, children with SE have lower developmental scores, psychiatric disorders, and reduced quality of life.4
Benzodiazepines have been considered as the initial treatment of SE due to their rapid onset and high efficacy in terminating seizures. Among them, diazepam has long been considered the gold standard treatment of SE.5 Despite its efficacy, diazepam is suspected to cause more complications compared to other benzodiazepines, including respiratory depression. Also, its prolonged elimination of half-life can result in cumulative effects, including sedation and increased risk of drug interactions. These limitations highlighted the need for alternative, safer treatments6,7
Midazolam is a short-acting, water-soluble benzodiazepine. In addition to intramuscular and intravenous administration, it can be administered via oral, nasal, and rectal routes, making it the preferred option in prehospital or resource-limited settings. Several limitations have been reported, including its short action, necessitating repeated doses, which can increase drug accumulation and side effects. Depending on hepatic metabolism, it can also increase toxicity in children with hepatic dysfunction or who are taking other drugs metabolized by the same pathway.8,9
Most recent meta-analyses have compared the effectiveness of midazolam, diazepam, and lorazepam in the pediatric population. Despite these studies providing valuable evidence, they focus on multiple drug comparisons, leaving behind limited evidence that specifically compares midazolam with diazepam in the pediatric population. Moreover, recent trials have not been included in meta-analyses focusing on both drugs.9,10
In this systematic review and meta-analysis, we aim to summarize and analyze the available evidence for comparing midazolam with diazepam in pediatric SE, regarding specific outcomes, including time to stop seizures, recurrence of seizures, and overall treatment success and adverse events.
Methods
Data sources and searches
We conducted a systematic search of PubMed, Scopus, Web of Science, and the Cochrane Library up to November 28, 2024, for studies evaluating the efficacy and safety of midazolam in the management of pediatric SE. Search terms and their relevant synonyms or MeSH terms included: (“pediatric” OR “neonates”) AND (“midazolam” OR “benzodiazepines”) AND (“status epilepticus” OR “seizures”). Two reviewers screened the study titles and abstracts using Rayyan11 to manage records, ensure blinding, and track conflicts, which were resolved by a third reviewer, and retrieved the full texts for inclusion. The review followed the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines and was prospectively registered in PROSPERO (CRD42024620622)12,13
Study selection and outcomes
We included studies if they met the following criteria: (1) randomized controlled trials comparing midazolam with diazepam (2) pediatric patients with SE, including neonates, infants, children, and adolescents; (3) midazolam administered by any route (intranasal, intravenous, buccal, or intramuscular); (4) reporting at least one of the following outcomes: therapeutic success, time to seizure cessation (minutes), seizure cessation within 10 min, treatment failure, recurrence of seizures, time from arrival to drug administration (minutes), or time from admission to active treatment (minutes) and adverse events.
Exclusion criteria were: (1) studies not published in English; (2) studies on adult patients; (3) use of midazolam for indications other than SE; (4) animal studies, case reports, and editorials without original data; (5) studies without a diazepam comparator arm; and (6) studies with insufficient or unreliable data for extraction. Our study’s primary outcomes were therapeutic success (%), time from admission to active treatment (minutes), time to stop seizing after treatment (minutes), seizure cessation within 10 min, seizure recurrence after initial cessation, and treatment failure (n, %)
Quality assessment and data extraction
We extracted relevant data from the included studies into a predefined online data extraction sheet. The extracted data from each study included: (1) study characteristics; (2) baseline characteristics of the study population; (3) risk of bias domains; and (4) primary and secondary outcome measures. The risk of bias and methodological quality of the included studies were independently assessed by four reviewers, using the Cochrane Risk of Bias 2.0 (RoB 2) tool for randomized controlled trials14 (Fig. 2).
Statistical analysis
We used RevMan 5.4.1 software to do the meta-analysis15. We reported continuous outcomes as mean difference (MD) and dichotomous outcomes as Relative risk (RR); for all outcomes, a confidence interval of 95% was calculated. We used the last time point data in the analysis. We converted study data reported as median (IQR) to mean (SD) when the SD was not directly reported16. We assessed heterogeneity by the Chi-square (I²) test, and a p value less than 0.1 was considered significant. According to Cochrane guidelines, an I2 value of 0–40% indicates low heterogeneity, 30–60% indicates moderate heterogeneity, 50–90% possibly represents substantial heterogeneity, and 75–100% signifies considerable heterogeneity. We used a random effects model when heterogeneity was above 30%; otherwise, we used a fixed effects model. We conducted subgroup analyses based on the route of drug administration, categorizing studies into IV route and non-IV route (buccal, Rectal, intranasal, and intramuscular) subgroups. meta-regression was performed to explore heterogeneity in specific subgroups; detailed model assumptions and justification are provided in the Supplementary Material (Fig. S1 legend). Sensitivity analyses were conducted to explore the robustness of the results and to investigate potential sources of heterogeneity. This involved a leave-one-out method, where studies were sequentially removed to identify single trials contributing disproportionately to the inter-study variance (I2). We also considered the impact of excluding studies with high risk of bias where applicable. When the P value is less than 0.05, the results are statistically significant. We could not perform publication bias analysis using the funnel plot, as the number of studies included was less than ten17. To control the risk of Type I and Type II errors and to ascertain the robustness and conclusiveness of the cumulative evidence, Trial Sequential Analysis (TSA) was performed for our primary outcomes and the key secondary outcomes. These analyses were conducted using the TSA software (version 0.9.5.10 Beta). The TSA parameters were pre-specified as follows: an α of 0.05, a β of 0.2 (power of 80%), a RR for dichotomous outcomes, and a MD for continuous outcomes. A random effects model using the Biggerstaff–Tweedie method was selected. While crossing the sequence monitoring border indicates statistical significance and conclusiveness, we interpreted these results with caution, particularly for outcomes where the accrued sample size remained below the required information size (RIS), acknowledging that further large-scale evidence may still be valuable, especially in the context of small sample size meta-analyses18,19
Certainty of evidence assessment
We assessed the certainty of the evidence for each outcome using the Grading of Recommendations, Assessment, Development and Evaluation (GRADE) approach20. We evaluated the following domains: risk of bias, inconsistency, imprecision, publication bias, and other considerations. For each outcome, we summarized the domains in an evidence profile table that included the number of contributing studies, domain-specific ratings, and the final certainty rating (Table S1).
Results
Literature search
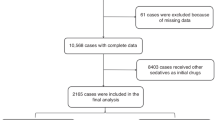
Searching the four major databases PubMed, Scopus, Cochrane Library, and Web of Science resulted in 2337 papers. 524 were duplicates, which have been removed. Title and abstract screening were done for 1813 articles. Full text screening was done for 35 studies after excluding 1769. 9 studies have been included, which is illustrated in PRISMA (Fig. 1).
The flow diagram depicts the systematic review process, detailing the number of records identified through database searching and other sources, the number of duplicates removed, and the number of records screened. It illustrates the exclusion of articles at each stage (screening and eligibility) with specific reasons for exclusion, leading to the final number of studies included in the qualitative and quantitative synthesis (meta-analysis). PRISMA, Preferred Reporting Items for Systematic Reviews and Meta-Analyses.
Study characteristics of included studies
Randomized controlled studies have been conducted in multiple countries enrolling 1115 pediatric patients with SE. Sample size range (32–330) patients. Ages ranged from 1 month to 16 years. The intervention group received midazolam via non-IV routes (buccal, rectal, intranasal (IN), intramuscular (IM), or intravenous (IV) while the control group received diazepam (rectal or IV) or placebo. Primary outcomes were therapeutic success, time for seizure cessation, seizure stopping within 10 min, time to stop seizure after treatment, failure event rate, and adverse events. Follow-up periods ranged from 10 min to 24 h, which are shown in (Table 1).
Baseline characteristics of the included studies
Across 9 RCTs, 563 children were assigned to the midazolam groups and 572 to the diazepam groups. Excluding Ashrafi et al.21, participant ages ranged from 1.53 to 7.17 years. The proportion of males ranged from 0.44 to 0.75 in the midazolam groups compared to 0.46 to 0.65 in the diazepam groups. Multiple etiologies were identified, but Idiopathic and febrile seizures were the most common. Admission temperature ranged 37–38.2 °C. Baseline characteristics are illustrated in (Table 2).
Risk of bias of the included studies
For the 9 included RCTs, we used the Cochrane Risk of Bias 2.0 (RoB 2) tool to assess the risk of bias across five key domains. 3 studies were judged to have low risk of bias overall (Holsti et al.22, Mpimbaza et al.23, Portela et al.24). 3 studies had some concerns (Ashrafi et al.21, Kazmi et al.25, McIntyre et al.26). 3 studies were rated as having a high risk of bias (Tonekaboni et al.27, Thakker et al.28, Momen et al.29), primarily due to issues related to randomization, deviation from intended intervention, or selective outcome reporting (Fig. 2).
"Traffic light" plot presenting the risk of bias summary for each included study across the five domains (D1–D5). Green circles (+) indicate low risk of bias, yellow circles (-) indicate some concerns, and red circles (x) indicate high risk of bias. (b) Summary bar plot showing the proportion of studies with low risk, some concerns, and high risk of bias for each domain and overall. Abbreviations: D1 Bias arising from the randomization process; D2 Bias due to deviations from intended interventions; D3 Bias due to missing outcome data; D4 Bias in measurement of the outcome; D5 Bias in selection of the reported result.
Outcomes
The overall meta-analysis of nine studies, encompassing 563 patients in the midazolam group and 572 in the diazepam group, confirmed a statistically significant therapeutic advantage for midazolam, with a pooled Risk Ratio (RR) of 1.13 (95% CI: 1.03–1.25; p = 0.02). The comprehensive subgroup analysis by administration route (Fig. 3) revealed that the route combination was a strong effect modifier, as indicated by the significant test for subgroup differences (I2 = 70.2%, p = 0.02). Breaking the analysis into specific administration routes demonstrated clear patterns: IV Midazolam was superior to IV Diazepam (RR = 1.20, p = 0.004), and Buccal Midazolam was significantly superior to Rectal Diazepam (RR = 1.30, p = 0.002). Conversely, non-IV midazolam (Buccal/IM/IN) was found to be comparable to IV Diazepam (RR = 1.00, p = 0.98), and IN/IM Midazolam was comparable to Rectal Diazepam (RR = 1.02, p = 0.73). While the subgrouping successfully isolated effects, the Buccal Midazolam vs. Rectal Diazepam comparison retained moderate residual heterogeneity (I2 = 55%). A subsequent random-effects meta-regression was performed using the Midazolam: Diazepam Dose Ratio as a covariate (Fig. S1). The dose ratio was found to be a non-significant predictor of the treatment effect (p = 0.517), confirming that differences in the relative dose did not explain the remaining variability.
The overall pooled analysis demonstrated a statistically significant higher success rate for Midazolam compared to Diazepam (RR = 1.13; 95% CI: [1.03, 1.25], p = 0.01). The overall analysis showed moderate heterogeneity (I2 = 55; t2 = 0.01). Significant therapeutic superiority for Midazolam was observed in the IV vs. IV comparison (RR = 1.20; 95% CI: [1.06, 1.37], p = 0.004) and the Buccal Midazolam vs. Rectal Diazepam comparison (RR = 1.30; 95% CI: [1.10, 1.55], p = 0.002; I2 = 55%, t2 = 0.01). Conversely, no significant difference in success rates was found for IN/IM Midazolam vs. Rectal Diazepam (RR = 1.02; 95% CI: [0.93, 1.11], p = 0.73) or when comparing Non-IV Midazolam (Buccal/IM/IN) vs. IV Diazepam (RR = 1.00; 95% CI: [0.84, 1.19], p = 0.98). The test for subgroup differences was statistically significant (p = 0.02, I2 = 70.2%), confirming that the route of administration significantly modifies the comparative efficacy.
The overall meta-analysis for Time to Seizure Termination (Fig. 4), based on seven studies, showed no statistically significant difference between midazolam and diazepam (MD = −0.62 min, 95% CI: −1.70 to 0.46; p = 0.28). However, substantial heterogeneity was observed across the overall analysis (I2 = 78%), which the significant test for subgroup differences (I2 = 86.8%, p <0.0001) confirmed was highly influenced by the administration route. Subgroup analysis revealed complex patterns: Midazolam was significantly faster than diazepam in the direct IV vs IV Comparison (MD = −1.42 min, p <0.00001) and in the IN/IM vs Rectal Comparison (MD = − 2.39 min, p = 0.010). Conversely, when Non-IV Midazolam (Buccal/IM/IN) was compared against IV Diazepam, Diazepam was significantly faster (MD = 0.88 min, p = 0.04), a clinically relevant finding attributed to the superior systemic bioavailability of the IV route. The Buccal Midazolam vs Rectal Diazepam subgroup showed no significant difference (MD = −2.04 min, p = 0.44), though substantial heterogeneity persisted (I2 = 78%).
The outcome is Mean Difference (MD) in minutes; results to the left of the line favor midazolam (faster termination). The significant test for subgroup differences (I2 = 86.8%, p < 0.0001, t2 = 1.38) indicates that the route of administration is a significant effect modifier. The overall pooled MD was −0.62 min (95% CI: −1.70 to 0.46), showing no statistically significant difference overall. The plot highlights that midazolam is significantly faster in two comparisons (IV vs IV, and IN/IM vs Rectal), while Diazepam is significantly faster in the Non-IV Midazolam vs IV Diazepam comparison.
the overall meta-analysis for Treatment Failure Rate (Fig. 5) showed significantly fewer failures with midazolam (142 events) compared to diazepam (206 events). This resulted in a statistically significant pooled Risk Ratio (RR) of 0.74 (95% CI: 0.57–0.95; p = 0.02), confirming that midazolam significantly reduces the risk of treatment failure compared to diazepam. Subgroup analysis confirmed that the route of administration significantly influenced the failure rate (I2 = 64.6%, p = 0.04). Midazolam showed a significantly lower risk of failure in the direct IV vs IV Comparison (RR = 0.28, p = 0.008) and in the Buccal vs Rectal Comparison (RR = 0.70, p = 0.005). In contrast, comparisons involving IN/IM vs Rectal Diazepam (RR = 1.06, p = 0.81) and Non-IV Midazolam vs IV Diazepam (RR = 1.01, p = 0.98) showed no significant difference in treatment failure rates.
The overall pooled analysis showed a statistically significant benefit for Midazolam, which was associated with a lower failure rate (RR = 0.74; 95% CI: [0.57, 0.95], p = 0.02; I2 = 36%, t2 = 0.04).Subgroup Analysis: significant efficacy favoring Midazolam was observed in the IV vs. IV comparison (RR = 0.28; 95% CI: [0.11, 0.71], p = 0.008) and the Buccal Midazolam vs. Rectal Diazepam comparison (RR = 0.66; 95% CI: [0.49, 0.88], p = 0.005; I2 = 39%, tau2 = 0.02). Conversely, no significant difference in failure rates was observed for IN/IM Midazolam vs. Rectal Diazepam (RR = 1.06; 95% CI: [0.65, 1.72], p = 0.81) or when comparing Non-IV Midazolam vs. IV Diazepam (RR = 1.01; 95% CI: [0.62, 1.62], p = 0.98). The test for subgroup differences was significant (p = 0.04, I2 = 64.6%), confirming that the choice of administration route significantly modifies the comparative efficacy.
Seizure relapse included 152 children in the midazolam group and 137 in the diazepam group. Using the fixed-effect model, a statistically significant benefit for midazolam in reducing seizure relapse has been observed. (RR = 0.51 (95% CI: 0.27, 0.98; p = 0.04) with no heterogeneity (I² = 0%) (Fig. 6).
Meta-analysis of the odds of seizure relapse with Midazolam versus Diazepam. The overall pooled analysis showed a statistically significant benefit for Midazolam, which was associated with a lower odds of seizure relapse (RR = 0.51; 95% (CI): [0.27, 0.98], p = 0.04). The analysis demonstrated no heterogeneity among the included studies (I2 = 0%).
Time from admission to active treatment included 3 studies that showed no statistically significant difference between midazolam and diazepam with substantial heterogeneity (I² = 72%) (Fig. S2). Sensitivity analysis and excluding Holsti et al.22 due to high risk of bias, demonstrated a MD of −2.95 min (95% CI: −4.53, −1.37; p = 0.0003), benefiting midazolam, not diazepam. No heterogeneity among the studies (I² = 0%; p = 0.48) (Fig. S3).
The seizure stopped within 10 min; intubation rate, and respiratory depression showed no statistically significant results with high heterogeneity. However, in Respiratory depression, which showed a trend that favors midazolam in the non-IV route subgroup when compared to diazepam (Figs. S4–S6).
Trial sequential analysis
For the outcome of therapeutic success, TSA showed a RIS of 662 participants. The cumulative Z-curve (blue line) surpassed both the superiority boundary and the RIS, confirming that midazolam achieves a significantly higher therapeutic success rate than diazepam. The evidence is therefore considered sufficient, and no additional studies are required (Fig. 7). Regarding the subgroup of Buccal Midazolam and diazepam it has also been conclusive as shown in (Fig. 8).
Trial sequential analysis of the therapeutic success of Midazolam versus Diazepam was conducted using a Random-effects model (DL). The conventional meta-analysis showed a significant pooled effect (RR = 1.13, 95% CI: 1.02–1.26; P value: 0.0223), with observed heterogeneity (I2 = 0.62, Q = 21.33, P value: 0.0063) and diversity (D2 = 0.73). The TSA was based on a double-sided alpha of 0.05 and a beta of 0.20 (power of 80%). As shown in the TSA plot, the cumulative Z-curve (blue line) crossed both the conventional statistical significance boundary and the superiority boundary. With a Required Information Size (RIS) of 662 patients and an accrued information size of 572 patients, the cumulative evidence is considered conclusive and statistically sufficient to demonstrate a reliable benefit for Midazolam in therapeutic success.
Trial sequential analysis of the therapeutic success of Buccal Midazolam versus Rectal Diazepam was conducted using a Random-effects model (DL). The conventional meta-analysis for this comparison showed a significant pooled effect (RR = 1.32, 95% CI: 1.09 to 1.59; P value: 0.005), with observed heterogeneity (I2 = 0.63, Q = 5.43, P value: 0.0662) and diversity (D2 = 0.72). The TSA was based on a double-sided alpha of 0.05 and a beta of 0.20 (power of 80%). The cumulative Z-curve (blue line) crossed both the conventional statistical significance boundary and superiority boundary. With a Required Information Size (RIS) of 559 patients, the cumulative evidence is considered conclusive and statistically sufficient to demonstrate no need for further trials.
For the outcome of time from admission to active treatment, the RIS was 385 participants. The cumulative Z-curve (blue line) crossed the conventional significance boundary but did not reach the superiority boundary (outer red line), and the accrued sample size remained well below the RIS. Therefore, the evidence is inconclusive, and additional trials are required to establish a definitive conclusion (Fig. S7).
Regarding the outcome of the failure rate, the RIS was 423 participants. The cumulative Z-curve (blue line) crossed the monitoring boundary for superiority (outer red line), indicating conclusive evidence of a significantly lower failure rate in Midazolam group. Further trials are not required to confirm this finding. (Fig. 9).
Trial sequential analysis of the treatment failure rate of Midazolam versus Diazepam was conducted using a Random-effects model (DL). Meta-analysis showed a significant pooled effect (RR = 0.74, 95% CI: 0.57 to 0.96; P value: 0.0215), confirming that Midazolam significantly reduces the risk of failure. The analysis showed low-to-moderate heterogeneity (I2 = 0.37, Q = 12.78, P value: 0.1195) and diversity (D2 = 0.60). The TSA was based on a double-sided alpha of 0.05 and a beta of 0.20 (power of 80%). As shown in the TSA plot the cumulative Z-curve (blue line) crossed both the conventional statistical significance boundary and the superiority boundary (outer red line). With a Required Information Size (RIS) of 423 patients and an accrued information size of 572 patients, the accrued information size exceeded the RIS. Therefore, the cumulative evidence is considered conclusive and no need for further trials.
A TSA was performed for the outcome of seizure relapse. The RIS had 62 participants. The cumulative Z-curve (blue line) crossed the conventional statistical significance boundary but did not cross the trial sequential monitoring boundary for superiority (outer red line). Although the accrued information size of 289 participants exceeded the RIS, the evidence remains inconclusive, and the findings should be interpreted with caution. Further trials may still be needed to confirm the robustness of this result (Fig. S8).
For the outcome of respiratory depression, the RIS was 50 participants. The cumulative Z-curve (blue line) did not cross the conventional boundary or the trial sequential monitoring boundaries and did not reach the RIS. These results indicate that the available evidence is inconclusive, and further studies are required to determine whether there is a significant difference in respiratory depression rates between midazolam and diazepam (Fig. S9).
Discussion
In this systematic review and meta-analysis with TSA, we examined the safety, route of administration and efficacy of midazolam compared with diazepam in treating pediatric SE. Pooled analysis demonstrated a greater therapeutic success with midazolam (RR = 1.13, p = 0.02) and less treatment failure (RR = 0.74, p = 0.02). TSA suggested the existing evidence for both outcomes is sufficient to support the noted advantage. The observed advantage of midazolam likely relates to the route of administration, with IV midazolam being superior to IV diazepam and buccal midazolam performing better than rectal diazepam.
Midazolam was associated with less risk of seizure relapse (RR = 0.51, p = 0.04) yet, TSA raised questions about the certainty of this evidence. Likewise, a sensitivity analysis reported a shorter time from admission to active treatment with midazolam (MD = –2.95 min), but will need further research corroboration. Safety outcomes (including respiratory depression) yielded no substantial difference between drugs, and TSA indicated the existing data may be insufficient to support conclusions.
In summary, midazolam demonstrates overall route-dependent advantages in efficacy, particularly through the IV and buccal routes, while evidence regarding relapse, treatment timing, and safety outcomes remains limited and warrants further investigation.
Our results regarding the superiority of midazolam and its specific routes of administration align with evolving practice patterns. Rather than contradict established approaches, these findings provide quantitative support for recommendations by national and international organizations such as the ILAE, AES/AAN, and NICE1. This recommendation is supported by our findings indicating that midazolam is more effective than diazepam (RR = 1.13, 95% CI 1.03–1.25; p = 0.02), particularly when buccal midazolam is compared to rectal diazepam (RR = 1.30, p = 0.002). TSA provided strong evidence for the buccal subgroup, supporting the ILAE recommendation. Similarly, the AES/AAN recommendations include IM midazolam alongside IV lorazepam and IV diazepam as other first-line treatment options30; the current review supports the recommendation with midazolam reducing treatment failure significantly (RR = 0.74, 95% CI 0.57–0.95; p = 0.02) among the available treatment options. NICE recommends buccal or intranasal midazolam in the community31, which is in accordance with the current subgroup analysis indicating that midazolam was significantly faster at terminating seizures in the IN/IM vs Rectal comparison (MD = −2.39 min, p = 0.01), while it was found that IV diazepam was faster as compared to any of the non-IV midazolam routes (MD = 0.88 min, p = 0.04).
However, this statistical difference in administration speed must be interpreted within the clinical context. Our meta-analysis indicates that non-IV midazolam (IM/IN) demonstrates comparable therapeutic efficacy to IV diazepam (RR = 1.00, p = 0.98). While statistically equivalent in stopping seizures, this finding is clinically transformative. Given that establishing intravenous access in a convulsing child is often delayed or fails in nearly 25% of cases32, The ability of IM midazolam to achieve equivalent seizure control without the rate-limiting step of cannulation supports its prioritization. Thus, while not statistically superior in success rate, IM/IN midazolam offers a superior practical profile for rapid intervention in prehospital and emergency setting.
These findings support earlier systematic reviews. Chhabra et al. suggested that intranasal midazolam is as effective as IV/rectal benzodiazepines with reduced times from admission-to-cessation times33. Correspondence in 2022 (Wheless et al.; Chhabra et al.) added another level of support to the validity of these findings34. In a similar vein, a network meta-analysis in pediatrics (2022) ranked intramuscular midazolam as number one for seizure termination and intranasal midazolam number one when intravenous access was not available—along with subgroup evidence, was more evidence these data are valid.35
For safety, current guidelines emphasize comparable respiratory risk among benzodiazepines1,30,31, which was reflected in our study. The analysis showed no significant differences between midazolam and diazepam regarding respiratory depression or the need for intubation.
However, midazolam demonstrated a relative advantage in reducing seizure relapse risk compared to diazepam. In contrast, a network meta-analysis of benzodiazepine-resistant SE (2022) implicated phenobarbital as potentially most effective but with considerable safety concerns36.
Midazolam’s increased efficacy via non-intravenous administration may be related to its drug-specific pharmacokinetic profile: it transitions from being water-soluble in acidic conditions to lipid-soluble at physiological pH, allowing for rapid absorption and improved bioavailability by buccal, intranasal, or intramuscular routes37.
Its easy passage across the blood–brain barrier facilitates faster seizure control38. Additionally, midazolam’s short half-life and lack of active metabolites may help sustain CNS effects without prolonged sedation39. On the other hand, diazepam’s high lipophilicity and redistribution into fat tissues may reduce its effectiveness via non-IV routes and increase the potential for seizure recurrence37. Distinct from adult management paradigms, our findings emphasize the critical role of non-IV options in the pediatric population. In adults with convulsive SE, intramuscular midazolam and intravenous agents (lorazepam, diazepam) are established as efficacious initial therapy (Level A evidence). Specifically, adult guidelines note that intramuscular midazolam has superior effectiveness compared to intravenous lorazepam when IV access is not yet established. Similarly, our pediatric data support this shift towards non-IV midazolam. Especially, obtaining vascular access in a convulsing child is technically challenging; one-quarter of pediatric peripheral intravenous catheters fail due to device dysfunction32, leading to significant treatment delays. This underscores the absolute necessity of selecting the most effective initial agent. Our results indicate that midazolam, specifically via the buccal route, offers a distinct advantage over rectal diazepam for rapid seizure control when IV access is unavailable30.
This meta-analysis provides more robust evidence, as it only considered RCTs13. This decreases the potential for bias in the results and enhances the validity of the data. Another important advantage is that only children with SE were included, making the conclusions more applicable to this population. The review conducted an extensive search of multiple databases and registries to ensure all recent and pertinent studies were covered40. Additionally, a TSA was conducted to verify our conclusions, which is useful to control random errors and minimize the risks of false-positive or false-negative results, making the results more reliable18,19. Finally, we assessed the certainty of the evidence using the GRADE approach, considering both statistical precision and clinical relevance across all domains.
However, this SRMA has limitations that need to be acknowledged. Many of the included RCTs have fewer than 100 participants, which may limit statistical power (Table 1). Most RCTs offer short-term follow-up, making it difficult to evaluate long-term safety or recurrence of seizures (Table 1). Furthermore, TSA revealed inconclusive results for several outcomes, such as time from admission to active treatment, seizure relapse, and respiratory depression (Figs. S7, S8, and S9). Additionally, the variability of midazolam-to-diazepam dose ratios across included studies may limit generalizability, as the diverse dosing regimens used in the primary studies introduce variability that complicates the precise assessment of relative efficacy. We observed varying heterogeneity across the outcomes: some showed no heterogeneity (I² = 0.0%) and some showed more than substantial heterogeneity (I² > 70%) (Figs. 1 and 4–6). This likely could relate to the routes of administration and/or definitions of outcome (Table 1).
Conclusion
Our findings have significant clinical implications for managing pediatric SE. The evidence from our analysis points to the superiority of midazolam over diazepam, reflected in higher therapeutic success, a significantly lower risk of treatment failure, and reduced seizure relapse. This advantage holds true in key clinical scenarios. For pre-hospital care, buccal midazolam proves to be a more effective agent than rectal diazepam41. In the emergency department, intravenous midazolam is likewise superior to its intravenous diazepam counterpart Accordingly, these findings are critical for prehospital and EMS contexts in which delayed vascular access has a negative prognostic impact42. From a practical implementation stance, data suggest that buccal and nasal midazolam are the most cost-effective non-IV rescue medications available in the United States43 However, insurance-related issues and costs in other regions would need to be accounted for before widespread implementation. These results provide a solid rationale for re-evaluating current practice guidelines, with a view to positioning non-IV midazolam as a preferred first-line benzodiazepine for children with acute seizures44,45.
References
Trinka, E. et al. A definition and classification of status epilepticus–report of the ILAE Task Force on Classification of Status Epilepticus. Epilepsia 56, 1515–1523 (2015).
Sánchez, S. & Rincon, F. Status epilepticus: epidemiology and public health needs. J. Clin. Med. 5, 71 (2016).
Gurcharran, K. & Grinspan, Z. M. The burden of pediatric status epilepticus: epidemiology, morbidity, mortality, and costs. Seizure 68, 3–8 (2019).
Singh, R. K. & Gaillard, W. D. Status epilepticus in children. Curr. Neurol. Neurosci. Rep. 9, 137–144 (2009).
Olkkola, K. T. & Ahonen, J. Midazolam and other benzodiazepines. Handb. Exp. Pharm. 182, 335–360 (2008).
Dhaliwal, J. S., Rosani, A. & Saadabadi, A. Diazepam. xPharm: The Comprehensive Pharmacology Reference 1–7. https://doi.org/10.1016/B978-008055232-3.61585-5 (2023).
Briley, M., Nutt, D., Mcgrath, C., Burrows, G. D. & Norman, T. R. The benzodiazepines: a brief review of pharmacology and therapeutics. Anxiolytics 1–11. https://doi.org/10.1007/978-3-0348-8470-9_1 (2000).
Conway, A., Rolley, J. & Sutherland, J. Midazolam for sedation before procedures. Cochrane Database Syst. Rev. 5, CD009491 (2016).
Hayashi, K. et al. Efficacy of intravenous midazolam for status epilepticus in childhood. Pediatr. Neurol. 36, 366–372 (2007).
Lingamchetty, T. N., Hosseini, S. A., Patel, P. & Saadabadi, A. Midazolam. xPharm: The Comprehensive Pharmacology Reference 1–5. https://doi.org/10.1016/B978-008055232-3.62185-3 (2025).
Ouzzani, M., Hammady, H., Fedorowicz, Z. & Elmagarmid, A. Rayyan—a web and mobile app for systematic reviews. Syst. Rev. 5, 1–10 (2016).
Page, M. et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. PLoS Med. 18, e1003583 (2021).
Cochrane Handbook for Systematic Reviews of Interventions | Cochrane. https://www.cochrane.org/authors/handbooks-and-manuals/handbook.
Sterne, J. et al. RoB 2: a revised tool for assessing risk of bias in randomised trials. BMJ 366, l4898 (2019).
Software | Cochrane [Internet]. [cited 2025 Aug 22]. Available from: https://www.cochrane.org/learn/courses-and-resources/software.
Wan, X., Wang, W., Liu, J. & Tong, T. Estimating the sample mean and standard deviation from the sample size, median, range and/or interquartile range. BMC Med. Res. Methodol. 14, 1–13 (2014).
Lin, L. & Chu, H. Quantifying publication bias in meta-analysis. Biometrics 74, 785–794 (2018).
Maurya, I., Lohiya, A. & Solanki, A. Trial sequential analysis: quality improvement for meta-analysis. Indian J. Anaesth. 68, 1092–1094 (2024).
Kang, H. Trial sequential analysis: novel approach for meta-analysis. Anesth. Pain. Med. 16, 138–150 (2021).
GRADE handbook. https://gdt.gradepro.org/app/handbook/handbook.html.
Ashrafi, M. et al. Efficacy and usability of buccal midazolam in controlling acute prolonged convulsive seizures in children. Eur. J. Paediatr. Neurol. 14, 434–438 (2014).
Holsti, M. et al. Intranasal midazolam vs rectal diazepam for the home treatment of acute seizures in paediatric patients with epilepsy. Arch. Pediatr. Adolesc. Med. 164, 747–753 (2010).
Mpimbaza, A. et al. Comparison of buccal midazolam with rectal diazepam in the treatment of prolonged seizures in Ugandan children: a randomised clinical trial. Pediatrics 121, e58–e64 (2008).
Portela, J. L. et al. Intramuscular midazolam versus intravenous diazepam for treatment of seizures in the paediatric emergency department: a randomised clinical trial. Med. Intensiva 39, 160–166 (2015).
Kazmi, A. et al. A comparison of intravenous midazolam and diazepam in management of status epilepticus in children. J. Pak. Med. Assoc. 71, 640–644 (2021).
McIntyre, J. et al. Safety and efficacy of buccal midazolam versus rectal diazepam for emergency treatment of seizures in children: a randomised controlled trial. Lancet 366, 205–210 (2005).
Tonekaboni, S. H. et al. A comparison of buccal midazolam and intravenous diazepam for the acute treatment of seizures in children. Iran. J. Pediatr. 71, 303–308 (2012).
Thakker, A. & Shanbag, P. A randomised controlled trial of intranasal midazolam versus intravenous diazepam for acute childhood seizures. J. Neurol. 260, 470–474 (2013).
Momen, A. A. et al. Efficacy and safety of intramuscular midazolam versus rectal diazepam in controlling status epilepticus in children. Eur. J. Paediatr. Neurol. 19, 149–154 (2015).
Glauser, T. et al. Evidence-based guideline: treatment of convulsive status epilepticus in children and adults: report of the guideline committee of the American Epilepsy Society. Epilepsy Curr. 16, 48–61 (2016).
National Institute for Health and Care Excellence (NICE). Epilepsies: diagnosis and management (NICE Guideline NG217, 2022).
Malyon, L. et al. Peripheral intravenous catheter duration and failure in paediatric acute care: a prospective cohort study. Emerg. Med. Australas. 26, 602–608 (2014).
Chhabra, R., Gupta, R. & Gupta, L. K. Intranasal midazolam versus intravenous/rectal benzodiazepines for acute seizure control in children: a systematic review and meta-analysis. Epilepsy Behav. 125, 108398 (2021).
Chhabra, R., Gupta, R. & Gupta, L. K. Reply to comment on “Intranasal midazolam versus intravenous/rectal benzodiazepines for acute seizure control in children: a systematic review and meta-analysis. Epilepsy Behav. 128, 108573 (2022).
Hasan, S. U. et al. Termination of seizures in the paediatric age group, best benzodiazepine and route of administration: a network meta-analysis. Eur. J. Neurosci. 56, 4234–4245 (2022).
Jain, P., Aneja, S., Cunningham, J., Arya, R. & Sharma, S. Treatment of benzodiazepine-resistant status epilepticus: systematic review and network meta-analyses. Seizure 102, 74–82 (2022).
Dundee, J. W., Halliday, N. J., Harper, K. W. & Brogden, R. N. Midazolam: a review of its pharmacological properties and therapeutic use. Drugs 28, 519–543 (1984).
Mandema, J. W. et al. Pharmacokinetic-pharmacodynamic modeling of the central nervous system effects of midazolam and its main metabolite α-hydroxymidazolam in healthy volunteers. Clin. Pharmacol. Ther. 51, 715–728 (1992).
Greenblatt, D. J., Gan, L., Harmatz, J. S. & Shader, R. I. Pharmacokinetics and pharmacodynamics of single-dose triazolam: electroencephalography compared with the Digit-Symbol Substitution Test. Br. J. Clin. Pharmacol. 60, 244–248 (2005).
Liberati, A. et al. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: explanation and elaboration. J. Clin. Epidemiol. 62, e1–e34 (2009).
Chin, R. F. et al. Treatment of community-onset, childhood convulsive status epilepticus: a prospective, population-based study. Lancet Neurol. 7, 696–703 (2008).
Gaínza-Lein, M. et al. Association of time to treatment with short-term outcomes for pediatric patients with refractory convulsive status epilepticus. JAMA Neurol. 75, 410–418 (2018).
Sánchez Fernández, I., Gaínza-Lein, M. & Loddenkemper, T. Nonintravenous rescue medications for pediatric status epilepticus: a cost-effectiveness analysis. Epilepsia 58, 1349–1359 (2017).
Caraballo, R. Randomized trial of three anticonvulsant medications for status epilepticus. Arch. Argent. Pediatr. 118, E211–E216 (2020).
Abend, N. S. & Loddenkemper, T. Management of pediatric status epilepticus. Curr. Treat. Options Neurol. 16, 301 (2014).
Funding
Open access funding provided by The Science, Technology & Innovation Funding Authority (STDF) in cooperation with The Egyptian Knowledge Bank (EKB).
Author information
Authors and Affiliations
Contributions
A.K. and N.H. contributed equally to this work and are considered first authors. A.K. conducted statistical analysis, performed all data synthesis including Forest plots and Trial Sequential Analysis (TSA), and wrote the “Results and Methods” sections. N.H. conceived the idea, designed the study, performed initial data collection, screened studies, and wrote the “Discussion” section. M.M., M.M., and I.A. contributed significantly to data screening and extraction, and assisted in drafting the manuscript. AH performed GRADE assessment. L.H.T., M.S.H. and S.A. assisted in data extraction and critically reviewed sections of the manuscript. Y.N. provided overall supervision and critical manuscript review. O.M.A. oversaw the study. All authors contributed to the writing of the manuscript and approved the final version.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Kertam, A., Hatem, N., AL-AZZAWI, O.M. et al. Efficacy, safety, route of administration of midazolam and diazepam for pediatric status epilepticus: systematic review, meta-analysis, and trial sequential analysis. Pediatr Res (2026). https://doi.org/10.1038/s41390-025-04722-6
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41390-025-04722-6