Abstract
This study aimed to explore the clinical characteristics and alteration of orexinergic level in cerebrospinal fluid (CSF) and the volumes of brain grey and white matters, and investigate the roles of orexinergic level on the association between brain atrophy and depression in Alzheimer’s disease (AD) patients. The demographic variables of 156 participants were collected. Orexinergic level in CSF and the volumes of brain grey and white matters were evaluated. The correlations of orexinergic level in CSF with depression and brain volume in AD patients were analyzed. The mediating effect of orexinergic level in CSF on the association between brain atrophy and depression in AD patients was investigated. The joint predictive value of orexinergic level in CSF and brain volume for depression in AD patients was established. AD with depression patients showed significantly elevated levels of orexin A and orexin B in CSF; orexin A level in CSF was positively correlated with HAMD score in AD patients. The elevated orexin A level in CSF mediated 49.6% of total association between the decreased grey matter volume of right dorsal medial thalamic nucleus and depression, and 50.3% of total association between the reduced white matter volume of left amygdala and depression. Combinations of above parameters could predict depression in AD patients with a significantly high area under the curve (AUC = 0.841). Therefore, the elevated orexin A level in CSF mediates its effect on the atrophy of the right dorsal medial thalamic nucleus and the white matter of the left amygdala, eventually alleviating depression in AD.
Similar content being viewed by others
Introduction
Depression is one of the most common neuropsychiatric symptoms in the individuals with Alzheimer’s disease (AD). A recent meta-analysis showed that the prevalence rates of depression in mild, moderate and severe AD were 37% (95% CI 30–44%), 40% (95% CI 31–48%) and 37% (95% CI 16–57%), respectively [1]. A meta-analysis suggested that depression was associated with over a twofold increase in the risk of suffering from AD [2]. Furthermore, depression was found to precipitate cognitive deterioration in AD patients [3]. Therefore, depression is arguably a modifiable target for preventing the progression of AD. However, the clinical characteristics of depression in AD patients are still inconsistent.
The pathological hallmarks of AD include the extracellular insoluble β amyloid (Aβ)- containing plaque and intracellular phosphorylated tau (P-tau)-containing neurofibrillary tangles. It was found that Aβ and P-tau controlled the release of neurotransmitters from presynaptic terminals. Interestingly, a variety of neurotransmitters, including serotonin, dopamine and noradrenaline, were disturbed in AD with depression (AD-D) patients, which might serve as the neurochemical mechanisms underlying AD-D [4]. In recent years, the compromised orexinergic system in depression has attracted increasing attention, but its roles on AD-D have been rarely investigated.
Orexinergic system contains orexin and its receptors. Orexin, comprising orexin A and orexin B, is an excitatory neuropeptide generated from lateral hypothalamus [5]. Orexin receptors, including orexin receptor 1 and orexin receptor 2, bind to orexin A/orexin B, and have significantly different distribution regions in brain. A previous study on orexinergic system mainly focused on its roles on regulating sleep-wake and narcolepsy [6]. However, recent investigations suggested that orexinergic system exerted pivotal role on depression. Orexinergic neurons were increased in the corticosterone-induced depression mouse model [7]. A postmortem study showed that orexin A neurons were increased in the depressed patients compared to controls [8]. However, there are no studies on the orexinergic level in cerebrospinal fluid (CSF) from AD-D patients to date.
It was shown that the orexinergic neurons in lateral hypothalamus extensively projected to other brain regions, such as thalamus, amygdala, hippocampus and medial prefrontal cortex [9]. A meta-analysis indicated that AD patients with late-life depression displayed grey matter atrophy in both thalamus and amygdala [10]. Another meta- analysis presented that white matter atrophy in AD-D patients mainly in the bilateral structures relating to memory formation, such as amygdala, hippocampus and entorhinal cortex [11]. A cohort study recruiting 252 AD patients with late-life depression from Alzheimer’s Disease Neuroimaging Initiative (ADNI) showed that the depressive participants had decreased glucose metabolism in amygdala [12]. However, the correlation of orexinergic level in CSF with the volumes of grey and white matters in above brain regions in AD-D patients is not explored, the role of orexinergic level in CSF on the association between brain atrophy and depression is unknown, and the predictive value of combined orexinergic level in CSF and brain atrophy on depression in AD patients is unclear.
In this study, demographic variables of 44 cases of normal controls (NC), 73 cases of AD with no depression (AD-nD) and 39 cases of AD-D were collected. A variety of rating scales were used to assess cognitive impairment, neuropsychiatric symptoms and activities of daily living (ADL). Orexinergic level in CSF was measured by enzyme-linked immunosorbent assay (ELISA), and the volumes of brain grey and white matter were evaluated by magnetic resonance images (MRI). The correlations of orexinergic level in CSF with depression reflected by the score of Hamilton Depression (HAMD) scale and brain volume in AD patients were analyzed. The mediating effect of orexinergic level in CSF on the association between brain atrophy and depression in AD patients was investigated, and the joint predictive value of orexinergic level in CSF and brain volumes for depression in AD patients was established. The aims of this study were to investigate the clinical characteristics of AD-D and its potential mechanisms involving orexin and brain atrophy.
Methods
Ethics statement
This study was approved by the ethics committee of Beijing Tiantan Hospital, Capital Medical University. All participants recruited and their family members signed the written informed consent forms in accordance with the Declaration of Helsinki.
Inclusion and exclusion criteria for AD patients
Inclusion criteria
AD was diagnosed according to the National Institute of Aging and Alzheimer’s Association criteria [13].
Exclusion criteria
The exclusion criteria of AD included the presence of neurological disorders besides AD that might affect cognitive function, including Lewy body disease, Parkinson’s disease, frontotemporal degeneration, corticobasal degeneration, vascular diseases, etc; a history of traumatic brain injury; major co-morbid psychiatric disorders diagnosed by Diagnostic and Statistical Manual of Mental Disorders, including bipolar disorder and schizophrenia, etc; acute suicidality; a history of substance abuse or dependence over the previous 3 years; or contraindication to MRI.
Diagnosis of depression
Depression was diagnosed with the National Institute of Mental Health Diagnostic Criteria for Depression of AD published in 2001 [14], which was derived from Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition (DSM-IV) criteria for major depression with a few modifications: (1) Three (or more) of the following symptoms must be present during the same 2-week period and represent a change from previous functioning, including clinically significant depressed mood; decreased positive affect or pleasure in response to social contacts and usual activities; social isolation or withdrawal; disruption in appetite; sleep disruption; psychomotor changes; irritability; fatigue or loss of energy; feelings of worthlessness, hopelessness, or excessive or inappropriate guilt; recurrent thoughts of death, suicidal ideation, plan or attempt. At the same time, at least one of the symptoms, either depressed mood or decreased positive affect or pleasure, needs to be met. (2) All criteria met the dementia of AD type. (3) The symptoms cause clinically significant distress or disruption in functioning. (4) The symptoms do not occur exclusively in the course of delirium. (5) The symptoms are not due to the direct physiological effects of a substance. (6) The symptoms are not better accounted for by other conditions, such as bipolar disorder, bereavement, schizophrenia, or schizoaffective disorder.
In this study, depression of subjects was quantitatively evaluated by HAMD scale, which is composed of 7 factors, including anxiety/somatization, weight loss, cognitive impairment, day and night changes, retardation, sleep disturbance and hopelessness. A total score of ≥8 points reflects the existence of depression [15], and 8–19 points, 20–34 points, and ≥35 points were defined as mild, moderate and severe depression, respectively.
Inclusion criteria for NC subjects
NC subjects who matched the age and sex of AD patients and had no complaints of cognitive decline indicated by Mini-Mental State Examination (MMSE) scale score > 26 points, and no depressive symptoms suggested by HAMD scale score < 8 points were included.
In this study, the sample size was calculated using PASS 15.0 software based on a previous study [16]. After assuming a large effect size, adjusting the significance level to α = 0.05 and the statistical power to 0.80 for improved statistical validity, a total of 156 subjects recruited from the Department of Neurology, Beijing Tiantan Hospital, Capital Medical University, were divided into 3 groups: (1) NC group: subjects had no cognitive decline and depression (n = 44); (2) AD-nD group: subjects met the diagnostic criteria of AD but not depression with HAMD score of <8 points (n = 73); (3) AD-D group: subjects met the diagnostic criteria of both AD and depression with HAMD score ≥8 points (n = 39).
Collection of demographic information
Demographic information, including gender, age, age of onset, educational level, and smoking condition, was recorded for each individual.
Assessments of cognitive function, neuropsychiatric symptoms, and ADL
Cognitive function, neuropsychiatric symptoms and ADL of subjects were assessed by a body of following rating scales, which were used in our previous study [17].
Overall cognitive function
Overall cognitive function was assessed by the MMSE and the Montreal Cognitive Assessment (MoCA) scales. MMSE scale evaluates cognitive domains of orientation, recall ability, attention, calculation, memory and language ability. MoCA scale assesses visuospatial and executive function, naming, memory, attention, language, abstract, delayed recall and orientation. The MoCA score ≤26 indicates potential cognitive impairment, and 1 point is added if the educational level of an individual is less than 12 years.
Memory
Memory was evaluated by the Auditory Verbal Learning Test (AVLT). The AVLT rates immediate, delayed and total memory. It consists of 5 learning tests in total. During each test, a list of 12 novel words is read aloud and the subjects are asked to recall 5 times.
Language
Language capability was examined by the 30-item Boston Naming Test (BNT) and the Verbal Fluency Test (VFT). The BNT involves displaying the patients 30 pictures and asking them to name each one. The VFT consists of 3 parts: the subjects are asked to name as many animals and household, and alternated the names of both items as many times as possible in a minute, respectively.
Visuospatial ability
Visuospatial ability was analyzed by the Rey-Osterrieth Complex Figure Test (RCFT), which consists of the RCFT-copying recall test and the RCFT-delayed recall test. The subjects are asked to duplicate a complex figure within 10 min, which is named RCFT-copying recall; and then draw the figure after 25 min, which is named RCFT-delayed recall.
Attention
Attention was evaluated by the Symbol Digit Modalities Test (SDMT) [18], the Trail Making Test-A (TMT-A), the Stroop Color-Word Test-B (SCWT-B) and Stroop Color- Word Test-C (SCWT-C). In the SDMT, the subjects are required to match numbers to the corresponding shape in 90 s as much as possible. In the TMT-A, the subjects are asked to draw a line between 25 consecutive numbers as quickly as possible. In the SCWT-B, the subjects are asked to read 50 circles with 4 colors of red, yellow, blue and green accurately and rapidly. In the SCWT-C, the subjects are asked to read the color of 50 words instead of the words themselves.
Executive function
Executive function was rated by the Stroop Color-Word Test-A (SCWT-A) and the Trail Making Test-B (TMT-B). In the SCWT-A, the subjects are asked to read 50 words accurately and rapidly. In the TMT-B, subjects are asked to connect 25 consecutive numbers in sequence as quickly as possible without lifting the pencil, interconnecting the square and circle shapes while connecting the numbers.
Neuropsychiatric symptoms
Overall neuropsychiatric symptoms
Overall neuropsychiatric symptoms were assessed by the Neuropsychiatric Inventory (NPI), which includes 10 neuropsychiatric symptoms and 2 autonomic symptoms. Each item is evaluated in terms of frequency with 1–4 point (s), severity with 1–3 point (s) and their multiply of 1–12 point (s). The scale is interviewed by both patients and caregivers. The score from caregivers is named NPI-Caregiver Burden.
Anxiety
The 14-item Hamilton Anxiety (HAMA) scale was used to assess anxiety symptoms. HAMA scale is divided into somatic anxiety and psychic anxiety.
Sleep disorder
Pittsburgh Sleep Quality Index (PSQI) was used to assess sleep disorders. It comprises 19 items categorized into 7 components of sleep quality, sleep onset latency, sleep duration, sleep efficiency, sleep disturbances, use of sleeping medication and daytime dysfunction, with the total score ranging from 0 to 21 point (s).
Apathy
The Modified Apathy Estimate Scale (MAES) was used to evaluate apathy. The MAES contains 14 items, and the level for each item consists “of not at all”, “slightly”, “some” and “a lot”. The total score ranged from 0 to 42 points (s).
Agitation
The Cohen-Mansfield Agitation Inventory (CMAI) was used to assess agitation. The CMAI consists of 29 items and rated the frequency of each behavior during the prior 1 month by using a 7-point scale with a range from 1 (never) to 7 (several times an hour).
ADL
The ADL scale, including the basic and the instrumental ADL scales, was used to assess ADL. Basic ADL includes mobility and personal self-care tasks, such as feeding, bathing and toilet use, etc. Instrumental ADL refers to more complex functions, such as telephoning, bookkeeping and managing medications, etc.
Acquisition of the brain volumes of grey and white matters
All participants were scanned on a 3 T MRI scanner (Prisma, Siemens, Germany), and 3-dimensional magnetization prepared rapid-acquisition gradient echo sequence (T1WI-3D-MP RAGE) was acquired with the following parameters: repetition time (TR) = 2300 ms, echo time (TE) = 2.3 ms, inversion time (TI) = 900 ms, scanning field = 240 mm × 240 mm, matrix = 256 × 256, layer thickness = 1 mm and interlayer spacing = 1 mm.
All the structural images in T1 were processed by using FreeSurfer software version 6.0 (http://www.martinos.org/freesurfer), which included motion correction, normalization of signal intensity, skull stripping, Talairach correction and automated segmentation of grey and white matters structures. The brain grey and white matters volumes of a total of 170 regions were calculated by using software template. The technical details of these procedures were described in a prior publication [19].
Detections of APOE genotypes
According to our previous study [20], the venous blood samples of patients with AD were collected from median elbow under fasting condition in the morning following admission and then sent to the clinical laboratory of our hospital. Genotypings for APOE single nucleotide variants (rs429358 C/T and rs7412 C/T), which define APOE ε2, ε3 and ε4, was performed by real‐time fluorescence quantitative polymerase chain reaction using nucleic acid detection reagents (Youzhiyou company, Wuhan, China).
Measurement of orexinergic level in CSF
Anti-AD drugs were withdrawn for 12–14 h if the patient’s condition allowed, and a long time was considered unethical by our ethical committee.
Under fasting condition, 5 ml of CSF was taken in a polypropylene tube (Beijing JingkeHongda Biotechnology Co., Ltd.) through lumbar puncture between 7 and 9 a.m. The samples were centrifuged immediately at 3000 r/min, aliquoted into separate Nunc cryotubes (Beijing JingkeHongda Biotechnology Co., Ltd.), and frozen at −80 °C until assay.
The level of orexinergic system, including orexin A, orexin B, orexin receptor 1 and orexin receptor 2, in CSF was measured. The kits included RK-003-30 RIA kit for orexin A, RK-003-31 RIA kit for orexin B (Phoenix Pharmaceuticals Inc., Germany), EK2497 ELISA Kit for human orexin receptor 1, and EK3718 ELISA Kit for human orexin receptor 2 (Signalway Antibody Company, America). The specific testing procedure for each of above variables followed the corresponding instruction provided by the individual kit.
Data analyses
Statistical analyses were performed by SPSS Statistics software (IBM Corporation, NY, USA). P < 0.05 was considered statistically significant.
Continuous variables, normality and homogeneity of variance were tested. ANOVA (3 groups), or Student T test (2 groups) was used for normally distributed variables with equal population variance, and the non-parametric Kruskal-Wallis test (3 groups) or Mann-Whitney U (2 groups) test was used for the variables with non- homogeneous variance.
When the differences were statistically significant (P < 0.05) among the three groups, post-multiple comparisons were further conducted. Bonferroni method was used when variance was equal; otherwise, Kruskal-Wallis test was used.
When calculating the differences in brain grey and white matters volumes among NC, AD-nD and AD-D groups, grey and white matters volumes were divided into 170 regions, and ANOVA test was performed among NC, AD-nD and AD-D groups. P values were compared to the corrected threshold P’ = 0.05/170 = 0.0003. Bonferroni test was conducted for the further comparison between NC and AD-nD groups, AD-nD and AD-D groups, and NC and AD-D groups.
The chi-squared test was used for the comparisons of categorical variables. Correlation analysis, stepwise multiple linear regression and logistic regression were used to explore the correlation between the related variables.
Receiver operating characteristic (ROC) curves were plotted by calculating the sensitivity and specificity of their diagnostic power.
Mediation analysis was conducted to examine the mediating effect of orexin A level in CSF on the association between brain white and grey matters volumes and depression. The mediating effect is carried out by using r package “lavaan”. Mediation analysis is performed through the establishment of three pathways: (1) exposure to the mediator; (2) mediator to outcome (direct effect); and (3) exposure to outcome (total effect). The total effect reflects the sum of direct and mediating (indirect) effects. The percentage of mediating effect is calculated by using the following formula: (mediating effect/total effect) × 100. Bootstrapping is used for the significance testing for mediation analysis.
Results
The frequency of depression in AD patients
Among 156 subjects recruited in this study, 44 cases were NC subjects and 112 cases were AD patients, in which 39 cases had depression, showing the frequency of depression in AD patients up to 34.82%.
Demographic information of NC, AD-nD and AD-D groups
Demographic information, including age, gender, smoking, education level, BMI and ApoE ε4 carrier status, was compared among NC, AD-nD and AD-D groups. It was found that the above demographic variables were not significantly different among the three groups (all P > 0.05) (Table 1).
Evaluations of cognitive function, neuropsychiatry symptoms and ADL among NC, AD-nD and AD-D groups
The overall cognitive function and multiple cognitive domains, including memory, language, attention, executive function and visuospatial ability, were compared among NC, AD-nD and AD-D groups. Both AD-nD and AD-D groups had significantly decreased scores of MMSE, MoCA, AVLT, BNT, VFT, RCFT and SDMT, and significantly prolonged time of SCWT-A and SCWT-C compared with NC group (all P < 0.05). AD-nD and AD-D groups showed no significant differences in cognitive function (all P > 0.05) (Supplementary Table 1).
The overall and individual neuropsychiatry symptoms were compared among NC, AD-nD and AD-D groups. AD-D group had significantly increased scores of HAMD, HAMA, PSQI, MAES and CMAI and NPI-Caregiver burden compared with AD-nD group (all P < 0.05), suggesting that AD-D group had more severe depression, anxiety, sleep disorder, apathy and agitation than AD-nD group. AD-nD group displayed significantly increased scores of PSQI, MAES and CMAI (all P < 0.05), and showed no significant differences in the scores of HAMA and HAMD scales compared with NC group (all P > 0.05). After adjusting for the effects of apathy and agitation, ANCOVA results revealed significant difference in HAMD scale score among NC, AD-nD and AD-D groups (P < 0.05). Specifically, AD-D group had significantly higher HAMD scale score than AD-nD group (P < 0.05) (Supplementary Table 1).
ADL was compared among NC, AD-nD and AD-D groups. Both AD-D and AD-nD groups had significantly higher ADL scale scores than NC group (P < 0.05). There was no difference in ADL score between AD-D and AD-nD groups (P > 0.05) (Supplementary Table 1).
Association between orexinergic level in CSF and HAMD scale score in AD patients
The levels of orexin and its receptors in CSF were compared among NC, AD-nD and AD-D groups. AD-D group had significantly elevated levels of orexin A and orexin B in CSF compared with AD-nD group (all P < 0.05). There were no significant differences in the levels of orexin receptor 1 and orexin receptor 2 between AD-D and AD-nD groups (all P > 0.05). AD-nD group showed no significantly different levels of orexin and its receptors in CSF compared with NC group (all P > 0.05) (Fig. 1).
The levels of orexin A (A), orexin B (B), orexin receptor 1 (C) and orexin receptor 2 (D) in CSF among NC, AD-nD and AD-D groups were measured and compared. *P < 0.05. NC normal controls, AD-nD Alzheimer’s disease with no depression, AD-D Alzheimer’s disease with depression, CSF cerebrospinal fluid.
The correlation between orexinergic level in CSF and HAMD scale score in AD patients was analyzed. It was found that orexin A level in CSF was significantly and positively correlated with HAMD scale score in AD patients (P < 0.01), and orexin B level in CSF was positively correlated with HAMD scale score in AD patients with P near to statistical significance (P = 0.065). The levels of orexin receptors in CSF were not correlated with HAMD scale score in AD patients (P > 0.05) (Supplementary Table 2).
Correlations between orexinergic level in CSF and brain grey and white matters volumes
In this study, the volumes of grey matter of 170 brain regions by MRI were compared among NC, AD-nD and AD-D groups. The brain regions with statistically significant differences in grey matter volumes between AD-nD and AD-D groups were shown (Supplementary Table 3). Firstly, compared with NC group, the grey matter volumes of most of the brain regions mentioned above were significantly reduced in AD-nD group. Noticeably, compared with AD-nD group, AD-D group had significantly decreased brain grey matter volumes in bilateral precentral gyrus, bilateral superior frontal gyrus, left middle frontal gyrus, right orbitofrontal gyrus, left rolandic operculum, right supplementary motor area, right medial superior frontal gyrus, bilateral rectus, left medial orbitofrontal cortex, right posterior orbitofrontal cortex, bilateral insula, bilateral calcarine cortex, bilateral cuneus, bilateral gyrus lingualis, left middle occipital gyrus, bilateral inferior occipital gyrus, bilateral gyri fusiformis, bilateral postcentral gyrus, right gyri parietalis, right supramarginal gyrus, left paracentral lobule, left transverse temporal gyrus, bilateral inferior temporal gyrus, right anterior ventral thalamic nucleus and dorsal medial thalamic nucleus (all P < 0.05) (Supplementary Table 3). After adjusting for Bonferroni correction, all of the aforementioned regions still showed statistically significant differences (P < 0.0003), except for the right anterior ventral thalamus nucleus. Then, the correlation between orexinergic level in CSF and brain grey matter volume was analyzed in AD patients. It was observed that among the brain grey volumes correlated with HAMD scale score, the grey matter volumes that were negatively correlated with orexin A level in CSF include the right anterior ventral nucleus and the right dorsal medial nucleus thalamus in AD patients (all P < 0.01) (Fig. 2). Further, the correlations between orexin A level in CSF and brain grey matter volumes of the right anterior ventral nucleus and the right dorsal medial nucleus of thalamus in AD-D patients were analyzed. In AD-D group, orexin A level in CSF was negatively correlated with the grey matter volume in the right ventral anterior thalamic nucleus (P < 0.05) and approached statistical significance in the right dorsal medial thalamus nucleus (P = 0.084) (Supplementary Tables 5–7).
Correlations analyses were performed between orexin level in CSF and brain grey matter volumes in the regions with statistically significant differences between AD-nD and AD-D groups. *P < 0.05, **P < 0.01, ***P < 0.001. HAMD Hamilton Depression.
In this study, the volumes of white matter of 170 brain regions by MRI were compared among NC, AD-nD and AD-D groups. The brain regions with statistically significant differences in white matter volumes between AD-nD and AD-D groups were shown (Supplementary Table 4). First of all, compared with NC group, the white matter volumes of left amygdala and left superior parietal gyrus were significantly decreased in AD-nD group (all P < 0.05). Strikingly, compared with AD-nD group, AD-D group had significantly reduced white matter volumes in the right orbitofrontal gyrus, right posterior orbitofrontal cortex, left amygdala, left middle occipital gyrus, right fusiform gyrus, left superior parietal gyrus, left transverse temporal gyrus and left substantia nigra (all P < 0.05). After adjusting for Bonferroni correction, all of the aforementioned regions still showed statistically significant differences (P < 0.0003), except for the right posterior orbitofrontal cortex and left transverse temporal gyrus. Then, the correlation between orexinergic level in CSF and brain white matter volume was analyzed in AD patients. It was found that among the brain white matter volumes correlated with HAMD scale score, the white matter volume in the left amygdala was negatively correlated with orexin A level in CSF (P < 0.01). (Fig. 3). Further, the correlation between orexin A level in CSF and white matter volume in the left amygdala in AD-D group was analyzed. In AD-D group, the white matter volume in the left amygdala was negatively correlated with orexin A level in CSF(P < 0.01) (Supplementary Table 8–10).
Correlations analyses were performed between orexin level in CSF and brain white matter volumes in the regions with statistically significant differences between AD-nD and AD-D groups. *P < 0.05, **P < 0.01, ***P < 0.001. HAMD Hamilton Depression.
Multiple linear regression model including orexin A level in CSF and the grey matter volume of the right anterior ventral thalamus nucleus showed that every 17.24 pg/mL elevation in orexin A level in CSF was associated with a 1-point increase in HAMD scale score, and every 0.01 mm3 decrease in the grey matter volume of the right anterior ventral thalamic nucleus was associated with a 1.4-point increase in HAMD scale score (all P < 0.05) (Supplementary Table 11).
Multiple linear regression model including orexin A level in CSF and the grey matter volume of the right dorsal medial thalamic nucleus displayed that every 0.01 mm3 decrease in the volume of the right dorsal medial thalamic nucleus was associated with a 0.15-point increase in HAMD scale score (P < 0.05) (Supplementary Table 12).
Multiple linear regression model including orexin A level and the white matter volume of the left amygdala revealed that every 0.01 mm3 decrease in the white matter volume of the left amygdala was associated with a 0.94-point increase in HAMD scale score (P < 0.05) (Supplementary Table 13).
Mediating effect of orexin A level in CSF on the association between brain atrophy and depression
In this study, the brain regions presenting significant atrophy in the patients with both AD and depression, and being correlated with orexin A level in CSF were selected. They were right anterior ventral thalamic nucleus, right dorsal medial thalamic nucleus and left amygdala white matter.
To investigate the roles of orexin A in CSF on the association between the atrophy of above three brain regions and depression in AD patients, mediating effect was analyzed. It was found that 49.6% of the total association between the grey matter atrophy of the right dorsal medial thalamic nucleus and depressive symptoms was mediated by the elevated orexin A level in CSF (P < 0.05) (Fig. 4), and 50.3% of the total association between the white matter atrophy of the left amygdala and depressive symptoms was mediated by the elevated orexin A level in CSF in AD patients (P < 0.05) (Fig. 5). Orexin A level in CSF failed to exert mediating effect on the total association between the grey matter volume of the right anterior ventral thalamic nucleus and depressive symptoms in AD patients.
The correlation between orexin A level in CSF and the grey matter volume of right dorsal medial thalamic nucleus in AD patients was analyzed. r = −0.443 [P < 0.001, 95%CI (−0.639, -0.199)] (A). Mediation model assessing whether orexin A level in CSF (M) mediated the relationship between the grey matter volume of right dorsal medial thalamic nucleus (X exposure) and depressive symptoms (Y outcome) (B). Path a: the effect of one-unit change in exposure on mediator; Path b: effect of one-unit change in mediator on outcome adjusted for exposure; Path c′(direct effect): the effect of one-unit change in exposure on outcome independent of mediator value. r = - 15.453 [P < 0.001, 95%CI (−28.84, −4.22)]. a * b (indirect effect): the effect of one-unit change in exposure on the outcome through mediator change. r = −14.617 [P < 0.001, 95%CI (−25.31, −2.18)]. Standardized estimates were provided. The proportion of mediation is the percentage of the mediated variables that explain the correlation between X and Y. Proportion of mediation = 49.2%. CI confidence intervals, CSF cerebrospinal fluid, HAMD Hamilton Depression.
The correlation between orexin A level in CSF and the white matter volume of the left amygdala in AD patients was analyzed. r = −0.451 [P < 0.001, 95%CI (−0.660, -0.201)] (A). Mediation model assessing whether orexin A level in CSF (M) mediated the relationship between the white matter volume of the left amygdala (X exposure) and depressive symptoms (Y outcome) (B). Path a: the effect of one-unit changes in exposure on mediator; Path b: the effect of one-unit changes in mediator on outcome adjusted for exposure; Path c′(direct effect): the effect of one-unit change in exposure on outcome independent of mediator value; r = −91.085 [P < 0.001, 95%CI (−168.77, −33.42)]. a * b (indirect effect): the effect of one-unit change in exposure on the outcome through mediator change. Standardized estimates were provided. r = −92.133 [P = 0.08, 95%CI (−158.741, −4.82)]. The proportion of mediation is the percentage of the mediated variables that explain the correlation between X and Y. Proportion of mediation = 50.3%. CI confidence intervals, CSF cerebrospinal fluid, HAMD Hamilton Depression.
Establishment of predictive model for depression in AD patients
Logistic regression was used to establish the predictive model for AD-D and AD-nD groups by combining orexin A level in CSF, the grey matter volume of the right dorsal medial thalamic nucleus, and the white matter volume of the left amygdala (Supplementary Table 14–16). The results indicated that all tolerances were >0.1, variance inflation factors were <10, eigenvalues were >0, and the condition index was <30, indicating no significant multicollinearity.
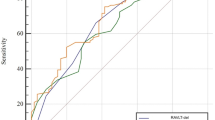
ROC analyses demonstrated that the AUC was 0.751 when only orexin A level in CSF was considered. AUC was increased to 0.807 and 0.824 when orexin A level in CSF was combined with the white matter volume of the left amygdala and the grey matter volume of the right dorsal medial thalamic nucleus, respectively. AUC reached 0.841 when orexin A level in CSF, grey matter volume of right dorsal medial thalamic nucleus and white matter volume of left amygdala were combined simultaneously (Fig. 6).
Receiver operating characteristic (ROC) curves displayed the performance of the models of depression in AD patients. AUC value in the model of combining orexin A level in CSF, the grey matter volume of the right dorsal medial thalamic nucleus and the white matter volume of the left amygdala (red line) was 0.841; AUC value in the model of combining orexin A level in CSF and the grey matter volume of the right dorsal medial thalamic nucleus (green line) was 0.824; AUC value in the model of combining orexin A level in CSF and the white matter volume of the left amygdala (blue line) was 0.807; AUC value in the model of combining orexin A level in CSF and the white matter volume of the left amygdala (yellow line) was 0.751. ROC receiver operating characteristic, CSF cerebrospinal fluid, AUC area under the curve.
Discussion
Recent studies have shown that depression is closely related to AD. A large Meta-analysis reported that the prevalence of depression in AD was 37–40% [1]. In this study, the frequency of depression in AD patients was as high as 34.82%. Therefore, clinicians should pay great attention to the early diagnosis and intervention of depression in AD patients.
In this study, demographic information, including age, gender and education level, were not significantly different among the three groups, excluding the influence of demographic variables on the following results and making the results comparable among the three groups (Table 1).
In this investigation, clinical symptoms of AD, including cognitive function, neuropsychiatry symptoms and ADL, were compared among NC, AD-nD and AD-D groups. It was particularly found that AD-D group suffered from severer neuropsychiatry symptoms. The significantly prominent neuropsychiatry symptoms that AD-D group had in the current investigation included anxiety, sleep disorder, apathy and agitation (Supplementary Table 1), indicating the close relationship of depression with these neuropsychiatric symptoms. Anxiety and sleep disorders are the very common accompanying symptoms in AD patients with depression [21]. Apathy, the most commonly reported neuropsychiatric symptom in AD, shared several overlapping features with depression, including loss of interest and psychomotor retardation due to dysfunction in frontal-subcortical circuits [22]. Agitation, another prevalent symptom, was more severe in AD patients with depression, which was likely due to frontal lobe dysfunction, and alleviating depression might reduce agitation behaviors [23]. In this study, after adjusting for the effects of apathy and agitation, it was still found that AD-D group scored significantly higher in depression than AD-nD group.
Orexinergic system was verified to be involved in the pathological processes of neurological diseases, including AD and depression [24]. In AD, a previous study reported that orexin levels in CSF from patients were 14% lower than those from healthy controls [25]; another investigation showed that orexin level was elevated and correlated with the increased P-tau and total tau in CSF from patients [26]. These contradictory results indicate that the alterations of orexinergic system in AD patients may be complex.
In depression, postmortem hypothalami of depressive patients showed that the immunoreactivity of orexin A was increased [8]; however, other studies failed to find the change of orexin A level in CSF in depressive disease [27, 28]. Currently, there is no consensus on orexin A level in CSF from depressive patients, and no study on the roles of orexinergic level in CSF on depression in AD patients. In the current study, AD-D group showed significantly elevated orexin A and orexin B levels in CSF compared with other groups (Fig. 1), and had a positive correlation between orexin A level in CSF and HAMD scale score (Supplementary Table 2), indicating that orexinergic level in CSF was closely associated with depression in AD patients. Orexinergic neurons and monoaminergic neurons, the typically disturbed neurons in depressed patients, project to each other, and the levels of orexin and monoamine (including serotonin and dopamine) were dynamically balanced by negative feedback [29]. In depression, the inhibitory effect of monoaminergic neurons on orexinergic neurons was decreased [30], which might result in the elevation of orexin A in AD-D patients and its correlation with depressive symptoms. It was known that orexin B had lower stability in CSF than orexin A [31]. Moreover, orexin B did not preferentially bind to orexin receptor 1 because its affinity to this receptor was 1/5–1/100 of orexin A [32]. Thus, the instability in CSF and low affinity to its receptor of orexin B may account for its unobserved correlation with depression in AD patients.
We further investigated the correlation between orexin A level in CSF and the grey matter volume of brain regions in AD-D group. To date, there is no consistent conclusion on the involved brain regions related to depression in AD patients. We found that the brain regions with significantly decreased grey matter volumes included frontal lobe, temporal lobe, parietal lobe, occipital lobe, bilateral calcarine cortex and thalamus (Supplementary Table 3). A previous study reported that patients with late-life depression displayed atrophy in the grey matter of frontal lobe, temporoparietal lobe, occipital lobe and hippocampus [33]. Interestingly, AD patients also exhibited atrophy in the grey matter of bilateral temporoparietal and occipital lobes [34]. A previous study reported that the mechanism underlying the grey matter atrophy might be due to the neurotoxic effects induced by the elevated cortisol, excessive oxidative stress and reduced neurotrophic factors [35]. In this study, the brain regions with the reduced grey matter volumes in AD-D group were partially consistent with the pattern of atrophy in those with AD and late-life depression. Importantly, we for the first time found the significant grey matter atrophy of the right dorsal medial nuclei of thalamus in AD-D group, which might be specifically related to the pathogenic processes in AD with depression, and the roles of orexinergic level in CSF were further explored. It was found that the reduced volumes of dorsal medial and the right anterior ventral nuclei of thalamus were correlated with orexin A level in CSF and depression symptom in AD patients and AD-D group (Fig. 2 and supplementary Table 7). In detail, every 0.01 mm3 decrease in the grey matter volume of the right anterior ventral thalamic nucleus was associated with a 1.4-point increase in HAMD scale score (Supplementary Table 11), and every 0.01 mm3 decrease in the grey matter volume of the right dorsal medial thalamic nucleus was associated with a 0.15-point increase in HAMD scale score (Supplementary Table 12). Moreover, the significantly elevated orexin A level in CSF contributed to 49.2% association between the grey matter atrophy of the right dorsal medial thalamic nucleus and depressive performance in AD patients (Fig. 4), illustrating that orexin A played a pivotal mediating role on depression associated grey matter atrophy of the right dorsal medial thalamic nucleus in AD patients. From the anatomical point of view, orexinergic fibers have a remarkably discrete distribution in thalamus. Dorsal medial nucleus, as part of the dorse of midline thalamus, was found to derive from neurons dispersed throughout the area containing orexinergic neurons [36]. Moreover, orexin A acted through orexin receptor 1 and orexin receptor 2 that were distributed throughout brain, especially in thalamus [37]. From the functional point of view, after injection of orexin into dorsal medial thalamic nucleus, rodents developed depressive behavior [38]. Dorsal medial thalamic nucleus was confirmed as the site with electrophysiological response to limbic area, thus regulating mood disorders, such as depression [39]. Up to now, we for the first time presented anatomical and functional evidence demonstrating that orexin A mediated the association between dorsal medial thalamic nucleus and depression in AD patients.
It was reported that the white matter was less changed than grey matter in AD. In this study, the brain regions with significantly reduced white matter volumes included frontal lobe, temporal lobe, parietal lobe, occipital lobe and left substantia nigra in AD-D group (Supplementary Table 4). In depression, white matter atrophy occurred in frontal and temporal lobes, and midbrain [40], which was associated with cognitive deterioration [41]. In a previous study, the reduced white matter integrity in frontal, parietal, temporal and occipital lobes was associated with late-life depressive symptoms [42]. In this study, we for the first time revealed the white matter atrophy in left amygdala in AD-D group, which might involve a disconnection between cortical-subcortical and interhemispheric tracts. The mechanism underlying the white matter atrophy in AD might be due to the demyelination induced by grey matter degeneration, resulting in cortico-cortical and cortico-subcortical disconnections. Previous study suggested that orexinergic system mediated depression through the action in the amygdala of humans [43]. However, the role of orexinergic level in CSF on the white matter atrophy in AD-D group has not been investigated. In this investigation, it was found that the white matter atrophy of the left amygdala was correlated with the elevated orexin A level in CSF from AD-D group. Detailly, every 17.24 pg/mL elevation in orexin A level in CSF was associated with a 1-point increase in HAMD scale score, and every 0.01 mm3 decrease in the white matter volume of the left amygdala was associated with a 0.94-point increase in HAMD scale score (Supplementary Table 13). Furthermore, the significantly elevated orexin A level in CSF contributed to 50.3% association between the white matter atrophy of the left amygdala and depressive performance in AD patients (Fig. 5), illustrating that orexin A played an essential role on depression associated white matter atrophy of the left amygdala. The orexinergic neurons project prominently to the basal and central nucleus of amygdala, and their fibers were distributed upon amygdala [44]. An animal experiment indicated that orexin A participated in depression-like behaviors by modulating the spontaneous firing activity of amygdala neurons, providing a molecular basis for amygdala atrophy. Intra-injection of orexin A in amygdala resulted in depressive behaviors [45]. These may explain that the enhanced orexin A level in CSF mediates the association between the white matter atrophy of the left amygdala and depression in AD patients.
In this study, we finally established models predicting the occurrence of depression for AD patients by combining orexin A level in CSF, the white matter volume of the left amygdala and the grey matter volume of the right dorsal medial thalamic nucleus using a logistic regression analysis. The AUC was 0.751 when orexin A level in CSF was used, the AUC was increased to 0.807 when orexin A level in CSF was combined with the white matter volume of the left amygdala, the AUC was further elevated to 0.824 when orexin A level in CSF was combined with the grey matter volume of the right dorsal medial thalamic nucleus, and the AUC reached the highest level of 0.841 when above three variables were taken into consideration (Supplementary Table 14–16, Fig. 6). This investigation provides clinical evidence for decreasing the elevated orexin A level in CSF and its mediating effect on the atrophy in the grey matter of the right dorsal medial thalamic nucleus and the white matter of the left amygdala, and eventually alleviating depression in AD patients.
Advantages and limitations
This study has several strengths. This study for the first time demonstrates that orexin A level in CSF is associated with the grey matter atrophy of the right dorsolateral thalamic nucleus/the white matter atrophy of the left amygdala in AD-D patients, orexin A level in CSF mediates the association between the grey matter atrophy of the right dorsal medial thalamic nucleus/the white matter atrophy of the left amygdala and depression in AD patients, and combination of orexin A level in CSF, the white matter volume of left amygdala, and the grey matter volume of the right dorsal medial thalamic nucleus has the most potent predictive role on depression for AD patients.
This study has limitations. To examine the association between the orexinergic system and depression in AD, and focus on the atrophy of depression-related brain regions influenced by the orexinergic system, this study comprises NC group, AD-nD group and AD-D group. Future research will incorporate depression patients with no AD to investigate the alterations in orexinergic system in the patients with depression. Additionally, our study suggests that the elevated orexin A level in CSF partially mediates the relationship between the reduced grey matter volume of the right dorso medial thalamic nucleus and the white matter volume of the left amygdala with depressive symptoms in AD patients. It is important to note that the structural changes in above brain regions jointly contribute to depression in AD patients, yet a bidirectional relationship may exist between depression and brain structural changes. We plan to further explore their complex relationships in the future longitudinal studies.
Conclusion
Depression is highly common in AD patients. Orexin A level in CSF is significantly elevated and correlated with depression in AD patients. AD patients with depression show significant atrophy in the grey matter of the right dorsal medial nuclei of thalamus and the white matter of the left amygdala. The elevated orexin A level in CSF plays an essential role on mediating the association between the atrophy in dorsal medial thalamic nucleus grey matter/amygdala white matter and depression in AD patients. Combining orexin A level in CSF, the white matter volume of amygdala and the grey matter volume of dorsal medial thalamic nucleus presents the highest predictive role on the occurrence of depression for AD patients. This investigation provides clinical evidence for decreasing the elevated orexin A level in CSF and its mediating effect on the atrophy in the right dorsal medial thalamic nucleus and left amygdala, and eventually alleviating depression in AD patients.
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author upon reasonable request.
References
Leung DKY, Chan WC, Spector A, Wong GHY. Prevalence of depression, anxiety, and apathy symptoms across dementia stages: a systematic review and meta-analysis. Int J Geriatr Psychiatry. 2021;36:1330–44.
Dafsari FS, Jessen F. Depression-an underrecognized target for prevention of dementia in Alzheimer’s disease. Transl Psychiatry. 2020;10:160.
Steffens DC. Late-life depression and the prodromes of dementia. JAMA Psychiatry. 2017;74:673–4.
Huang F, Wu X. Brain neurotransmitter modulation by gut microbiota in anxiety and depression. Front Cell Dev Biol. 2021;9:649103.
Broberger C, De Lecea L, Sutcliffe JG, Hökfelt T. Hypocretin/orexin- and melanin-concentrating hormone-expressing cells form distinct populations in the rodent lateral hypothalamus: relationship to the neuropeptide Y and agouti gene-related protein systems. J Comp Neurol. 1998;402:460–74.
Mahoney CE, Cogswell A, Koralnik IJ, Scammell TE. The neurobiological basis of narcolepsy. Nat Rev Neurosci. 2019;20:83–93.
Jalewa J, Wong-Lin K, McGinnity TM, Prasad G, Hölscher C. Increased number of orexin/hypocretin neurons with high and prolonged external stress- induced depression. Behav Brain Res. 2014;272:196–204.
Lu J, Zhao J, Balesar R, Fronczek R, Zhu QB, Wu XY, et al. Sexually dimorphic changes of hypocretin (Orexin) in depression. EBioMedicine. 2017;18:311–9.
Han Y, Yuan K, Zheng Y, Lu L. Orexin receptor antagonists as emerging treatments for psychiatric disorders. Neurosci Bull. 2020;36:432–48.
Boccia M, Acierno M, Piccardi L. Neuroanatomy of Alzheimer’s disease and late-life depression: a coordinate-based meta-analysis of MRI studies. J Alzheimers Dis. 2015;46:963–70.
Li J, Pan P, Huang R, Shang H. A meta-analysis of voxel-based morphometry studies of white matter volume alterations in Alzheimer’s disease. Neurosci Biobehav Rev. 2012;36:757–63.
Touron E, Moulinet I, Kuhn E, Sherif S, Ourry V, Landeau B, et al. Depressive symptoms in cognitively unimpaired older adults are associated with lower structural and functional integrity in a frontolimbic network. Mol Psychiatry. 2022;27:5086–95.
McKhann GM, Knopman DS, Chertkow H, Hyman BT, Jack CR Jr., Kawas CH, et al. The diagnosis of dementia due to Alzheimer’s disease: recommendations from the National institute on aging-Alzheimer’s association workgroups on diagnostic guidelines for Alzheimer’s disease. Alzheimers Dement. 2011;7:263–9.
Olin JT, Schneider LS, Katz IR, Meyers BS, Alexopoulos GS, Breitner JC, et al. Provisional diagnostic criteria for depression of Alzheimer disease. Am J Geriatr Psychiatry. 2002;10:125–8.
Hamilton M. A rating scale for depression. J Neurol Neurosurg Psychiatry. 1960;23:56–62.
Schmidt FM, Arendt E, Steinmetzer A, Bruegel M, Kratzsch J, Strauss M, et al. Csf-hypocretin-1 levels in patients with major depressive disorder compared to healthy controls. Psychiatry Res. 2011;190:240–3.
Yu SY, Lian TH, Guo P, Li LX, Ding DY, Li DN, et al. Correlations of apathy with clinical symptoms of Alzheimer’s disease and olfactory dysfunctions: a cross-sectional study. BMC Neurol. 2020;20:416.
Fellows RP, Schmitter-Edgecombe M. Symbol digit modalities test: regression-based normative data and clinical utility. Arch Clin Neuropsychol. 2019;35:105–15.
Lian TH, Zhu WL, Li SW, Liu YO, Guo P, Zuo LJ, et al. Clinical, structural, and neuropathological features of olfactory dysfunction in patients with Alzheimer’s disease. J Alzheimers Dis. 2019;70:413–23.
He M, Liu Z, Lian T, Guo P, Zhang W, Huang Y, et al. Role of nerve growth factor on cognitive impairment in patients with Alzheimer’s disease carrying apolipoprotein e ε4. CNS Neurosci Ther. 2024;30:e14560.
Amidfar M, Garcez ML, Kim YK. The shared molecular mechanisms underlying aging of the brain, major depressive disorder, and Alzheimer’s disease: The role of circadian rhythm disturbances. Prog Neuropsychopharmacol Biol Psychiatry. 2023;123:110721.
Lanctôt KL, Ismail Z, Bawa KK, Cummings JL, Husain M, Mortby ME, et al. Distinguishing apathy from depression: a review differentiating the behavioral, neuroanatomic, and treatment-related aspects of apathy from depression in neurocognitive disorders. Int J Geriatr Psychiatry. 2023;38:e5882.
Trzepacz PT, Yu P, Bhamidipati PK, Willis B, Forrester T, Tabas L, et al. Frontolimbic atrophy is associated with agitation and aggression in mild cognitive impairment and Alzheimer’s disease. Alzheimers Dement. 2013;9:S95–104.e101.
Wang C, Wang Q, Ji B, Pan Y, Xu C, Cheng B, et al. The orexin/receptor system: molecular mechanism and therapeutic potential for neurological diseases. Front Mol Neurosci. 2018;11:220.
Fronczek R, van Geest S, Frölich M, Overeem S, Roelandse FW, Lammers GJ, et al. Hypocretin (orexin) loss in Alzheimer’s disease. Neurobiol Aging. 2012;33:1642–50.
Osorio RS, Ducca EL, Wohlleber ME, Tanzi EB, Gumb T, Twumasi A, et al. Orexin-A is associated with increases in cerebrospinal fluid phosphorylated-tau in cognitively normal elderly subjects. Sleep. 2016;39:1253–60.
Schmidt FM, Arendt E, Steinmetzer A, Bruegel M, Kratzsch J, Strauss M, et al. CSF-hypocretin-1 levels in patients with major depressive disorder compared to healthy controls. Psychiatry Res. 2011;190:240–3.
Salomon RM, Ripley B, Kennedy JS, Johnson B, Schmidt D, Zeitzer JM, et al. Diurnal variation of cerebrospinal fluid hypocretin-1 (Orexin-A) levels in control and depressed subjects. Biol Psychiatry. 2003;54:96–104.
Baumann CR, Bassetti CL. Hypocretins (orexins): clinical impact of the discovery of a neurotransmitter. Sleep Med Rev. 2005;9:253–68.
Morganstern I, Chang GQ, Barson JR, Ye Z, Karatayev O, Leibowitz SF. Differential effects of acute and chronic ethanol exposure on orexin expression in the perifornical lateral hypothalamus. Alcohol Clin Exp Res. 2010;34:886–96.
Hoang QV, Bajic D, Yanagisawa M, Nakajima S, Nakajima Y. Effects of orexin (hypocretin) on GIRK channels. J Neurophysiol. 2003;90:693–702.
Sakurai T, Amemiya A, Ishii M, Matsuzaki I, Chemelli RM, Tanaka H, et al. Orexins and orexin receptors: a family of hypothalamic neuropeptides and G protein-coupled receptors that regulate feeding behavior. Cell. 1998;92:573–85.
De Winter FL, Emsell L, Bouckaert F, Claes L, Jain S, Farrar G, et al. No association of lower hippocampal volume with Alzheimer’s disease pathology in late-life depression. Am J Psychiatry. 2017;174:237–45.
Jack CR Jr., Holtzman DM. Biomarker modeling of Alzheimer’s disease. Neuron. 2013;80:1347–58.
Ritchie K, Jaussent I, Stewart R, Dupuy AM, Courtet P, Ancelin ML, et al. Association of adverse childhood environment and 5-HTTLPR Genotype with late-life depression. J Clin Psychiatry. 2009;70:1281–8.
Kirouac GJ, Parsons MP, Li S. Orexin (hypocretin) innervation of the paraventricular nucleus of the thalamus. Brain Res. 2005;1059:179–88.
Marcus JN, Aschkenasi CJ, Lee CE, Chemelli RM, Saper CB, Yanagisawa M, et al. Differential expression of orexin receptors 1 and 2 in the rat brain. J Comp Neurol. 2001;435:6–25.
Li Y, Li S, Wei C, Wang H, Sui N, Kirouac GJ. Changes in emotional behavior produced by orexin microinjections in the paraventricular nucleus of the thalamus. Pharmacol Biochem Behav. 2010;95:121–8.
Georgescu IA, Popa D, Zagrean L. The anatomical and functional heterogeneity of the mediodorsal thalamus. Brain Sci. 2020;10:624.
Colloby SJ, Firbank MJ, Vasudev A, Parry SW, Thomas AJ, O’Brien JT. Cortical thickness and VBM-DARTEL in late-life depression. J Affect Disord. 2011;133:158–64.
Marawi T, Zhukovsky P, Rashidi-Ranjbar N, Bowie CR, Brooks H, Fischer CE, et al. Brain-cognition associations in older patients with remitted major depressive disorder or mild cognitive impairment: a multivariate analysis of gray and white matter integrity. Biol Psychiatry. 2023;94:913–23.
Fleischman DA, Arfanakis K, Leurgans SE, Zhang S, Lamar M, Han SD, et al. Late-life depressive symptoms and white matter structural integrity within older Black adults. Front Aging Neurosci. 2023;15:1138568.
Grafe LA, Bhatnagar S. Orexins and stress. Front Neuroendocrinol. 2018;51:132–45.
Schmitt O, Usunoff KG, Lazarov NE, Itzev DE, Eipert P, Rolfs A, et al. Orexinergic innervation of the extended amygdala and basal ganglia in the rat. Brain Struct Funct. 2012;217:233–56.
Gyawali U, James MH. Orexin (Hypocretin) signaling in the basolateral amygdala contributes to individual differences in stress sensitivity. Biol. Psychiatry. 2022;91:775–7.
Acknowledgements
We thank all the participants involved in the present study.
Funding
This study was supported by the Capital’s Funds for Health Improvement and Research (CFH) (2022-2-2048), Science and Technology Innovation 2030 Major Projects (2022ZD0211600), the Research on Mechano-Biomaterial Sciences in Brain Diseases and Neuromodulation, the National Natural Science Foundation of China (T2488101), the Collaborative Research Project of Traditional Chinese and Western Medicine of the Major Difficult Disease-Alzheimer’ s Disease of Beijing (2023BJSZDYNJBXTGG-018), the National Key Research and Development Program of China (2016YFC1306300), the National Natural Science Foundation of China (81970992), the Project of Scientific and Technological Development of Traditional Chinese Medicine in Beijing (JJ2018-48), the Project of Beijing Institute for Brain Disorders (BIBD-PXM2013_014226_07_000084), the Natural Science Foundation Natural Science Foundation of Hebei, China (H2021206416), Medical Science Research Project of Health Commission of Hebei, China(20221377), the National Natural Science Foundation of China (32000792).
Author information
Authors and Affiliations
Contributions
JL contributed to the conception, design and data statistics of the study and paper writing; NW, PG and MH conducted the MRI procedure and acquired the imaging data; YZ, YH and JQ collected collected CSF samples; DL, WZ, and RW collected clinical data; TL, JL, and MW contributed to directing paper writing and data statistics; WZ contributed to the conception and design of the study and the revision of paper writing. The authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval
This study was approved by the Institutional Ethics Board of Tiantan Hospital, Capital Medical University.
Informed consent
Written informed consent was obtained from all participants or their legal guardians.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Li, J., Lian, T., Li, J. et al. Alzheimer’s disease with depression: clinical characteristics and potential mechanisms involving orexin and brain atrophy. Transl Psychiatry 15, 66 (2025). https://doi.org/10.1038/s41398-025-03251-4
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41398-025-03251-4