Abstract
Objective
This study aims to systematically investigate activation in brain regions during early-stage motor sequence learning (MSL) and to identify convergent neural activation patterns, thereby advancing our understanding of the neural mechanisms underlying MSL.
Methods
This meta-analysis integrated functional magnetic resonance imaging (fMRI) data from 12 studies (219 participants) using the serial reaction time task (SRTT) paradigm, applying anisotropic effect-size seed-based d mapping (AES-SDM) to identify common brain activation regions underlying MSL. After the primary meta-analysis, subgroup analyses were conducted on the young adult group, implicit learning group, and bimanual/right-hand task group to investigate the specific effects of age and task type on brain region activation in MSL. Additionally, an independent fMRI dataset from 64 healthy participants performing SRTT was used to validate the meta-analysis results.
Results
We identified convergent activations during SRTT in cortico-basal ganglia-cerebellar circuit, encompassing the supplementary motor area (SMA), precentral gyrus (PreCG), rolandic operculum (ROL), and other cortical regions, alongside basal ganglia and cerebellum, collectively underscoring their critical roles in early-stage MSL. Subgroup analyses revealed age- and task-related modulation of neural activation patterns. Results were largely replicated in the independent validation dataset.
Conclusions
Our findings provide reliable evidence for the involvement of a cortico-basal ganglia-cerebellar circuit in MSL.
Significance
Our meta-analysis contributes to unraveling the brain activation patterns underlying early-stage MSL, offering insights for clinical interventions in motor dysfunction rehabilitation.
Similar content being viewed by others
Introduction
Motor sequence learning (MSL) refers to the brain’s ability to learn and internalize ordered motor patterns, such as the finger movements in playing a piano melody, leg coordination in riding a bicycle, or hand-eye sequences in typing [1, 2]. Through practice, this process yields increasingly efficient and automatic skill execution [1, 2]. As a fundamental cognitive process underlying the acquisition, refinement, and automation of sequential motor skills, MSL is critical for a wide range of human behaviors and daily activities [3, 4]. Over decades, diverse experimental paradigms, such as serial reaction time task (SRTT), variants of SRTT (e.g., incompatible SRTT), explicit sequence learning task (e.g., finger tapping task) and other paradigms such as pursuit rotor task have been developed to explore deep insights into the mechanisms of MSL [2]. Among these, SRTT is one of the most extensively utilized paradigms to investigate how the brain engages in sequence learning, originally developed by Nissen and Bullemer [5, 6]. In contrast to other paradigms (e.g., finger tapping task), the SRTT allows for stringent control over perceptual, motor, and other confounding factors through the rigorously standardized design, thereby providing a clear and unparalleled advantage in probing sequence-specific learning within MSL research [5]. Specifically, during the SRTT, participants are instructed to rapidly respond to visual stimuli in two conditions: a repeating sequence condition (in which the visual cues follow a recurring sequence of positions unbeknownst to the participants) and a random condition (in which the visual cue no longer follow a repeating pattern). These conditions are typically presented in separate experimental blocks. By contrasting neural activation between these two conditions, the SRTT isolates sequence-specific learning processes from general motor execution, even when participants exhibit no conscious awareness of embedded patterns [5, 7], thereby enabling researchers to pinpoint the neural mechanisms underlying sequence acquisition with sensitivity guaranteed [8, 9]. Thus, the robust experimental design of the SRTT could offer a powerful framework for elucidating the mechanisms of MSL. Investigation into the neural basis of MSL contributes to the advancement of neuroscience research and simultaneously informs therapeutic interventions for neurological disorders characterized by motor dysfunction, such as Parkinson’s disease and motor deficits following stroke [10, 11].
In recent decades, neuroimaging studies have greatly improved our understanding of brain activation patterns during MSL. The learning process involves a transition from novel sequence encoding to fluent execution, mediated by coordinated activity in cortico-striatal and cortico-cerebellar networks [12,13,14]. Functional magnetic resonance imaging (fMRI) studies demonstrate that the supplementary motor area (SMA) plays a key role in motor planning and sequencing, with increased activity observed during the early stages of learning [15,16,17]. The basal ganglia, particularly the caudate and putamen, are essential for recognizing patterns and reinforcing learning [4, 5, 18]. This is evidenced by studies comparing patients with Parkinson’s disease to healthy controls on motor sequence task performance, which show that impairments in the striatum interfere with sequence learning and highlight its importance in implicit learning [10]. Concurrently, the cerebellum contributes to movement accuracy and timing precision, with cerebellar activation linked to reduced reaction time variability during skill consolidation [19,20,21].
Despite these significant advances in identifying neural substrates of the MSL, inconsistencies persist across studies. Some studies, for instance, have indicated that the striatum has been identified as critical for MSL [4, 19, 22], yet patients with Parkinson’s disease, who have impaired striatum due to the disease, still exhibit detectable motor performance on MSL tasks [23, 24]. Moreover, the role of the cerebellum in early motor sequence acquisition also remains debated. Some studies suggested cerebellar involvement during the early learning phase, particularly in error correction and motor coordination [13, 25], while others argued that cerebellar activation is not specifically linked to that learning phase [26]. To reconcile these discrepancies, meta-analyses including SRTT-related neuroimaging studies have been conducted, yet consensus remains elusive. For instance, one meta-analysis identified the basal ganglia and cerebellum as critical neural substrates underlying MSL [5], whereas another reported predominant activation in the motor cortex and thalamus, alongside weaker subcortical contributions [27].
Importantly, the two meta-analyses may have been limited by heterogeneity in included studies’ task paradigms (e.g., varying degrees of sequence complexity, implicit/explicit learning designs) and conflation of distinct MSL stages (e.g., early encoding vs. late automated performance). Such methodological heterogeneity and conflation could inherently contribute to the observed contradictory findings. These results, in turn, reflect the multifaceted nature of MSL and emphasize the necessity for systematic investigations into how task-specific factors (e.g., learning modality), age-related neural adaptations, and analytical variations (e.g., ROI definitions, statistical thresholds) collectively shape brain activation patterns. Furthermore, limited sample sizes in individual neuroimaging studies further exacerbate variability and contribute to mixed results. To address these challenges, we applied anisotropic effect-size seed-based d mapping (AES-SDM) [28, 29], a meta-analytic tool designed to quantify whole-brain activation consistency through cross-study data integration, to studies employing the SRTT paradigm to evaluate MSL performance. Briefly, this method synthesizes peak coordinate data from multiple studies and employs anisotropic effect-size modeling to reconstruct maps quantifying the spatial consistency of brain activation across studies. Through this approach, AES-SDM minimizes spatial bias and enhances sensitivity in detecting consistent activation patterns, even amid methodological heterogeneity [30], such as variations in fMRI acquisition parameters (e.g., TR: 2.0–3.0 s). Additionally, subgroup analyses were conducted to examine age- and task-dependent effects on MSL. To enhance reliability, findings from the meta-analysis were externally validated using an independent fMRI dataset. Ultimately, this study aims to map consistently engaged brain networks that drive motor skill acquisition, with a focus on the motor cortex, basal ganglia, and cerebellum, thereby translating mechanistic insights into clinical strategies for motor dysfunction rehabilitation.
Materials and methods
Study selection
We conducted a comprehensive search from the inception of each database (PubMed, Web of Science, and Embase) up to August 26, 2024. The search strategy employed the following keywords: ((motor sequence learning OR serial reaction time task OR SRT OR SRTT) AND (fMRI OR functional magnetic resonance imaging)). The literature search was restricted to peer-reviewed studies published in English. Additionally, a manual search was performed by screening references from included studies and relevant reviews pertaining to the topic of the meta-analysis. We conducted this systematic review fully compliant with the PRISMA 2020 guidelines, with the completed checklist provided as Supplementary Table S1. Two authors (WC, TL) independently carried out the search process, and any discrepancies in search results were resolved through discussion to ensure consistency prior to the screening phase. For the initial screening of titles and abstracts (where potential discrepancies may be more frequent), inter-rater agreement was assessed using Cohen’s kappa coefficient (K = 0.85, 95% CI: 0.80–0.90) [31]. The detailed calculation process is provided in the Supplementary Materials. This meta-analysis was registered in PROSPERO (registered number: CRD42024584359, titled ‘From Bits to Beats: Unraveling the Human Brain’s Adventure in Motor Sequence Learning’, https://www.crd.york.ac.uk/PROSPERO).
We included studies that met the following criteria: (1) studies used fMRI to examine neural activation during MSL tasks, using whole-brain analysis, and reported peak coordinates in standard reference space (Talairach/Tournoux or MNI); (2) studies provided data from healthy adults aged ≥ 18 years; (3) sample size was larger than 5 in single group; (4) the MSL task involved a classic form of the SRTT. In these tasks, participants were instructed to press corresponding buttons with their fingers, using either both hands or one hand when a visual stimulus was sequentially presented to them. Each response button spatially corresponded to different stimulus locations (e.g., four response buttons corresponding to four stimulus locations); (5) participants were not informed of the existence of a sequential condition, or participants were informed of the existence of a sequence condition but were not informed of the specific sequence; (6) for studies containing multiple independent healthy cohorts or mixed patient-healthy control (HC) cohorts, only data from each HC cohort were extracted and treated as an independent experiment in the meta-analysis. For studies investigating brain activation across multiple stages of MSL, only the data from early/fast learning stage (encoding) were included. Exclusion criteria were as follows: (1) animal studies or studies included only patients; (2) studies using other modalities such as PET, EEG, MEG, structural MRI or DTI; (3) studies only performed analysis within regions of interest (ROIs) and did not report whole-brain activation results; (4) studies involve individuals with exceptional manual dexterity as participants (e.g., pianists); (5) studies involving the sequence learning task did not employ the classic form of SRTT (e.g., finger tapping task); (6) studies involve participants who are asked to respond only to certain trials, and/or the response buttons are not spatially aligned with the stimulus cues; (7) studies involve participants who are asked to respond to visual stimuli in other ways, such as pressing with feet or gazing with eyes. In addition, the corresponding authors would be contacted in cases additional data is needed for the present meta-analysis.
Global analysis
The meta-analysis was conducted using anisotropic effect-size seed-based d mapping software (AES-SDM version 5.15, http://www.sdmproject.com) following established protocols [28, 29]. Specifically, Peak coordinates and t-values of brain regions contrasting sequential versus random conditions were extracted from the included studies. Non-MNI coordinates (i.e., TAL space) were converted to MNI space using the icbm2tal nonlinear transform [32], minimizing template bias and enhancing spatial accuracy [33]. If studies reported z-values or p-values, these values were converted to t-values using SDM’s conversion utility (www.sdmproject.com/utilities/?show=Statistics). These t-values were transformed into Hedges’g effect sizes, and an anisotropic Gaussian kernel (full-width half-maximum = 20 mm) was applied to account for spatial uncertainty, assigning higher effect sizes to voxels with stronger spatial correlation to reported peaks. Signed effect size and variance maps were reconstructed and combined via a random-effects model that adjusted for sample size, intra-study variability, and between-study heterogeneity. The meta-analytic maps were reported using the recommended threshold (uncorrected voxel p < 0.005, cluster extent ≥ 10 voxels, peak SDM-Z > 1), which corresponds approximately to a corrected p < 0.05 and offers an optimal balance between sensitivity and specificity [28]. All meta-maps with statistical significance were visualized using the BrainNet Viewer (version 1.7, https://www.nitrc.org/projects/bnv/) [34].
Subgroup analysis
We further conducted subgroup analyses on the young adult group (age = 18–40), the implicit learning group, and the bimanual/right-hand task group extracted from the overall sample, aiming to investigate whether age and task type exerted specific effects on the activation of brain regions associated with MSL.
Jackknife analyses and publication bias
The meta-analytic results may have been biased by individual studies, potentially leading to false-positive findings or reduced statistical power [35,36,37]. To eliminate this possibility, we conducted leave-one-out analyses (also called jackknife analyses), where in each iteration one experiment was excluded and the meta-analysis was recomputed (voxel p < 0.005, peak height Z ≥ 1, cluster extent ≥ 10 voxels). This approach allowed us to assess whether the observed meta-analytic clusters were critically dependent on any single influential study or robust against potential false-positive effects. Subsequently, we performed heterogeneity analyses using a random-effects model and Q statistics [28] to generate a statistical map (voxel p < 0.005, peak height Z ≥ 1, cluster extent ≥ 10 voxels), thereby exploring whether those observed brain activation patterns had significant variations across studies. To evaluate potential publication bias, funnel plots were generated to visually inspect the symmetry of effect size distributions for each cluster. Additionally, Egger’s test was employed to objectively quantify funnel plot asymmetry using a linear regression model [38]. A statistical significance threshold of p < 0.05 in Egger’s test was used to indicate the presence of significant publication bias.
Validation analysis
To validate the reproducibility and generalizability of the meta-analysis results, an independent dataset comprising 64 healthy participants (age = 22.09 ± 2.50, 32 females) was employed to generate whole-brain activation maps, while the participants performed both left-hand and right-hand SRTT (contrast maps: sequential vs. random conditions). Statistical maps were threshold ed at p < 0.001, corrected for the Family-Wise Error (FWE), with nonparametric permutation test (5000 permutations) using threshold-free cluster enhancement (TFCE) [39,40,41]. Details of validation analyses were shown in Supplementary Materials.
Results
Included studies and sample characteristics
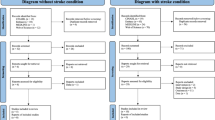
After removing 777 duplicates from an initial pool of 2141 articles, we carefully screened and ultimately included 12 papers that met the eligibility criteria for meta-analysis [9, 42,43,44,45,46,47,48,49,50,51,52]. The study identification and exclusion process are illustrated in the PRISMA flowchart (Fig. 1). The primary meta-analysis included data from 219 participants. Detailed demographic information, task types, and other relevant characteristics of the included studies are summarized in Table 1.
The flowchart describing search results and study selection.
Primary analysis in the full sample
The primary meta-analysis identified significant clusters of brain activation (voxel p < 0.005, peak height Z ≥ 1, cluster extent ≥ 10 voxels) associated with MSL (Table 2, Fig. 2). The most prominent activation was localized to the right rolandic operculum (ROL), with significant bilateral SMA engagement and the left basal ganglia (encompassing the putamen, caudate, and globus pallidus). Distributed activation patterns emerged in the left middle frontal gyrus (MFG), left calcarine cortex (CAL), bilateral precentral gyrus (PreCG), right fusiform gyrus (FFG), and right cerebellum. Conversely, deactivations were noted in the right inferior temporal gyrus (ITG), inferior frontal gyrus, orbital part (ORBinf), superior frontal gyrus, orbital part (ORBsup). Sensitivity analyses demonstrated high consistency across studies, with most clusters showing robustness in 14 out of 14 experiments. While heterogeneity was minimal for most clusters, the SMA, basal ganglia, and CAL clusters showed significant heterogeneity. For all clusters, Egger’s test indicated no significant publication bias (all p > 0.05), further validating the reliability of the findings.
L left, R right, CAL calcarine fissure / surrounding cortex, FFG fusiform gyrus, ITG inferior temporal gyrus, LING lingual gyrus, MFG medial frontal gyrus, ORBinf inferior frontal gyrus, orbital part, ORBsup superior frontal gyrus, orbital part, PCL paracentral lobule, PreCG precentral gyrus, ROL rolandic operculum, SMA supplementary motor area.
Subgroup analysis in young participants
The subgroup analysis focusing on young participants (age = 18–40, n = 140), comprising 10 experiments, revealed significant clusters of brain activation linked to sequence learning (voxel p < 0.005, peak height Z ≥ 1, cluster extent ≥ 10 voxels). The primary activations focus on bilateral SMA. Additional reliable activations were identified in the left MFG, basal ganglia and ORBinf. Activations in the right insula, PreCG and ROL were also observed. In contrast, deactivations were detected in the right middle temporal gyrus (MTG) and ORBinf (Table 3, Fig. 3A). Results of jackknife sensitivity and heterogeneity analyses for all subgroups were shown in Table 3. No publication bias (all p > 0.05) was observed in the results of the subgroup analysis of young participants.
A All included participants were young adults; B All tasks performed by participants were implicit; C All included participants performed tasks with both hands. L left, R right, CAL calcarine fissure / surrounding cortex, FFG fusiform gyrus, INS insula, LING lingual gyrus, MFG medial frontal gyrus, MTG middle temporal gyrus, ORBinf inferior frontal gyrus, orbital part, PCL paracentral lobule, PreCG precentral gyrus, ROL rolandic operculum, SMA supplementary motor area, STG superior temporal gyrus.
Subgroup analysis based on implicit learning task
The subgroup analysis examining implicit MSL-related brain activation pattern, based on 11 experiments with 166 participants in total who were not informed of the existence of a sequential condition, identified significant clusters of brain activation (voxel p < 0.005, peak height Z ≥ 1, cluster extent ≥ 10 voxels). Significant activations were localized in the bilateral superior temporal gyrus (STG) and SMA, in the left basal ganglia, lingual gyrus (LING), and MFG, as well as in right-hemispheric regions encompassing the paracentral lobule (PCL), PreCG, FFG, and cerebellum. Conversely, deactivations were observed in the right MTG and bilateral ORBsup (Table 3, Fig. 3B). Notably, publication bias was detected in the STG (p = 0.029) and ORBsup (p = 0.033) regions.
Subgroup analysis based on bimanual task
Subgroup analysis of bimanual task (9 experiments, n = 145) showed significant activation clusters (voxel p < 0.005, peak height Z ≥ 1, cluster extent ≥ 10 voxels). The strongest activation localized in the right ROL, with significant engagement of the bilateral SMA. Left-hemispheric activations emerged in the basal ganglia, CAL, MTG and STG, while right-hemispheric activations involved the PCL, PreCG and FFG. Deactivation patterns were observed in the right MTG and ORBsup (Table 3, Fig. 3C). Egger’s test revealed publication bias in the ROL (p = 0.030), FFG (p = 0.028), and cerebellum (p = 0.028) regions. As part of ancillary analyses, results from the right-hand task subgroup are provided in Supplementary Table 3.
Validation analysis
To validate the meta-analytic findings, we administered separate left-hand and right-hand SRTT runs in an independent cohort of 64 healthy participants and found significant activation contrasts between sequential and random conditions during the SRTT, thresholded at p < 0.001 (FWE corrected) using TFCE with 5000 permutations. Critically, all activation patterns identified in the meta-analysis were replicated in this independent dataset (Fig. 4). This included prominent activations in the right ROL, bilateral SMA, and left basal ganglia—specifically the putamen, caudate and globus pallidus. Additional activation clusters emerged in the bilateral preCG, left MFG, ORBinf, CAL, LING, right FFG and PCL. Furthermore, consistent deactivation was observed in the right ITG, ORBinf, and left ORBsup regions.
Statistical maps were thresholded at p < 0.001 (FWE corrected) using TFCE with 5000 permutations. L left, R right, CAL calcarine fissure / surrounding cortex, FFG fusiform gyrus, ITG inferior temporal gyrus, LING lingual gyrus, MFG medial frontal gyrus, ORBinf inferior frontal gyrus, orbital part, ORBsup superior frontal gyrus, orbital part, PCL paracentral lobule, PreCG precentral gyrus, ROL rolandic operculum, SMA supplementary motor area.
Discussion
The present meta-analysis employed AES-SDM to synthesize fMRI studies investigating brain regions associated with MSL, specifically focusing on those that employed the SRTT paradigm. Our primary analysis identified convergent activations in the motor cortex, basal ganglia, and cerebellum, thereby supporting the involvement of a cortico-basal ganglia-cerebellar circuit in the early-stage MSL [11, 14, 27, 53]. Additionally, subgroup analyses further indicated that age, learning type, and task modality may influence the activation patterns observed in brain regions. Finally, validation with an independent dataset demonstrates the rigor of our findings.
Specifically, as a critical region within the motor cortex, the SMA plays an essential role in motor execution and the acquisition of sequential rules, with its activation consistently observed across analyses and validated through independent datasets, highlighting its centrality in sequence learning [54,55,56]. This aligns with prior evidence, specifically findings on the role of the SMA in linking action phases during the encoding stage of sequential movement learning, action execution, and imagined rhythmic motor planning [57,58,59,60]. It is noteworthy that the SMA exhibited significant heterogeneity (“Yes”) in both the primary analysis and the young participants subgroup analysis, which may be related to the inclusion of different types of learning tasks in these two analyses (e.g., some included experiments in these two analyses involved both explicit and implicit sequence learning task) [61]; in contrast, no heterogeneity was found to be significant (“No”) in the implicit task subgroup analysis. Moreover, the primary analysis revealed additional activation in other motor cortical regions, such as the PreCG and ROL, and other associated areas, with our validation dataset further demonstrating the widespread engagement of these cortical regions in supporting MSL. The PreCG was linked to motor planning and execution, particularly in hand movements and action-related processing [62], while the ROL likely supports sensorimotor integration by interacting with brain regions involved in movement planning and sensory processing [63]. Although there has been some prior research on the contributions of these regions to MSL, their specific role in motor learning has not often been the central focus of empirical studies. Consequently, additional research is needed to fully characterize their involvement.
Notably, previous studies have pointed out the primary motor cortex (M1) was a critical neural substrate for motor execution, playing an essential role in both skill acquisition and maintenance [64,65,66,67]. Nevertheless, recent studies have provided a refined perspective, suggesting that M1 activation may not be consistently detected during MSL and appears less directly involved in sequence encoding compared to its established role in elementary motor execution [68, 69]. Our meta-analysis aligns with this distinction: no significant activation in M1 was observed, an outcome likely attributable to our paradigm’s focus on implicit sequential rule acquisition during motor learning (i.e., SRTT) rather than elementary motor execution tasks (e.g., finger-tapping protocols). This dissociation suggests that M1 may not be involved in the encoding phase of MSL, during which tasks prioritize acquiring sequence rules over motor performance alone. It should be noted that our findings are explicitly constrained to early learning dynamics and do not rule out the potential involvement of M1 in subsequent consolidation stages. For instance, long-term motor training induces expanded neural representations of movement sequences in M1, as evidenced by prior fMRI studies in healthy adults [70]. Thus, how M1 functions across learning stages in MSL merits further investigation.
The basal ganglia, subcortical nuclei including the striatum (e.g., caudate nucleus, putamen) and globus pallidus, play a central role in motor control, sequence learning, and habit formation [71]. Previous studies highlight their involvement in chunking and automating motor sequences via cortico-striatal circuits, particularly during rule extraction and action selection in early learning [4, 72]. Our meta-analysis and validation analysis of significant basal ganglia activation in early MSL further confirm their critical role in encoding sequential rules. Basal ganglia lesions (e.g., stroke) impair sequence chunking, leading to fragmented movements, error-prone execution, and reduced learning efficiency [10]. Similarly, striatal degeneration in patients with Parkinson’s disease disrupts implicit sequence learning [73]. Therefore, The prominent activation of the basal ganglia is consistent with their well-established role in initiating sequences and integrating rules [74]. Of note, the basal ganglia did not exhibit significant between-study heterogeneity in the young participants subgroup, but marked heterogeneity was present in the primary analysis and other subgroups, which may suggest that age-related neural differences could impact the activation pattern of the basal ganglia [75].
Regarding the cerebellum, extensive research has highlighted its pivotal role in motor learning networks, principally engaged in temporal anticipation and error correction during early skill acquisition stages [13, 76]. Our results corroborate this framework by consistently demonstrating cerebellar activation across analyses (except for the subgroup analysis of young participants), providing support for its involvement in initial learning phases. Further empirical data from short-term MSL paradigms reveals that the cerebellum primarily facilitates initial motor coordination and internal model formation rather than long-term skill automatization [25]. This collective evidence directly contradicts previous claims that cerebellar activity is not linked to specific learning stages but is merely essential to performance adaptation [26]. However, the absence of significant cerebellar activation observed in young adults remains unclear due to limited empirical investigation. These unresolved issues underscore the need for longitudinal studies to clarify age-dependent neural trajectories in motor skill acquisition.
Taken together, this study elucidated the neural mechanisms underlying the early stage of MSL, advanced the understanding of prior inconsistencies in reported brain activation patterns, and provided critical insights at the neural circuit level into strategies for guiding clinical interventions. Numerous studies have shown that patients with movement disorders, such as stroke and Parkinson’s disease, are associated with deficits in critical regions within the motor network [77, 78]. However, there is a growing consensus that such impairments also involve functional alterations at the circuit or network level [79, 80], and our results, which indicate that early-stage MSL engages the motor cortico-basal ganglia-cerebellar circuit, suggesting synchronous activity in those areas, consistently align with this perspective. Recent researches have demonstrated that noninvasive brain stimulations (NIBS), such as repetitive transcranial magnetic stimulation (rTMS), hold the ability to change the brain activity at the targeted site as well as remote regions that are functionally connected to it [81, 82]. Accumulating evidence indicates that early-stage MSL performance can be modulated by applying these NIBS to the key nodes within motor cortico-basal ganglia-cerebellar circuit in healthy participants [83,84,85]. These findings suggest that the application of NIBS to the motor cortico-basal ganglia-cerebellar circuit may serve as an alternative treatment for patients with early-stage MSL deficits which could be explored in future studies. Furthermore, the identification of critical nodes within this circuit suggests that targeting such areas with NIBS may yield optimal outcomes [86]. It has been revealed that rTMS targeting the SMA is more effective than stimulating the M1 in improving early MSL performance [57, 87], a finding that aligns with our result of SMA activation in the absence of M1 engagement. Moreover, since critical nodes often subserve specific functions, they provide a foundation for individualized interventions and rehabilitation monitoring tailored to specific disorders, such as SMA modulation might enhance implicit sequence encoding [87], whereas basal ganglia activation could facilitate sequence consolidation [80]. Although rTMS exhibits limited penetration depth [88], restricting its ability to effectively stimulate deep-seated key nodes (e.g., the basal ganglia) within the neural circuits involved in early MSL, recent studies have shown that cortical targets can be determined based on functional connectivity strength with subcortical regions. This approach allows indirect modulation of the activity in deep nuclei, ultimately enhancing behavioral performance [89, 90]. Thus, for patients with movement disorders exhibiting MSL deficits, future therapeutic strategies could identify the region which exhibit abnormal activation within the relevant circuits during MSL task. These neural features, along with clinical symptoms, can inform the selection of NIBS parameters and stimulation targets, particularly for basal ganglia-related dysfunction. Based on these insights, we further recommend that future research focus on stratifying early-stage MSL patients according to circuit-specific dysfunction patterns to better guide individualized intervention and rehabilitation monitoring.
Despite these findings, the present meta-analysis has several limitations. First, while our study focuses specifically on the SRTT paradigm for early-stage MSL to ensure internal validity, this limits the generalizability of our findings to other MSL domains. Moreover, restriction to early learning stages precludes mapping the full neural trajectory from rules acquisition through consolidation to final automation. Future studies are encouraged to incorporate diverse paradigms and integrate longitudinal designs across different learning stages to systematically elucidate the specificity and continuity of neural mechanisms underlying MSL. Secondly, the limited number of included studies necessitates cautious interpretation of both the overall regional brain activation patterns and findings regarding heterogeneity or bias, particularly in subgroup analyses, where small sample sizes compromise the statistical reliability of such results [35]. For example, methodological heterogeneity (e.g., variations in brain coverage, such as the exclusion of cerebellar activation data in Zedkova et al. (2006), which was included in the current meta-analysis) may have been amplified in these smaller subsets, potentially generating spurious bias signals in Egger’s tests [38]. Additionally, the predominance of younger adults in the included studies limits the generalizability of the observed neural patterns to older populations, where movement disorders are more prevalent. Thus, we anticipate that future research will conduct targeted investigations into how MSL is neurally represented in aging cohorts. Similarly, the predominant use of bimanual tasks across studies precludes deeper analysis of hemispheric lateralization and lateralized hand-specific activation patterns, which deserves in-depth investigation in future studies. Where appropriate, we also recommend incorporating detailed handedness metrics to better assess the influence of hand preference strength on lateralized activation. Finally, while stable static activation patterns during early-stage MSL were identified, dynamic interactions among regions remain unclear. Further research is needed to build spatiotemporal connectomes of these networks, delineating information flow within motor learning circuits. Addressing these in future work would provide a more comprehensive and clearer foundation for informing clinical interventions.
Conclusion
In summary, our study systematically synthesizes studies employing the SRTT and validates these findings with an independent dataset, which identifies the cortico-basal ganglia-cerebellar circuit as a stable neural substrate supporting early-stage MSL. These findings resolve prior inconsistencies in reported brain activation patterns and delineate a functional network essential for MSL. By advancing our understanding of neural mechanisms underlying MSL, especially in early learning phases, this study provides a theoretical foundation for clinical interventions in motor dysfunction-related disorders.
Data availability
The coordinate data used are available in the GitHub repository: https://github.com/VivieChen/MotorSequenceLearning-MetaAnalysis. The data that support the findings in validation analysis of this study are available from the corresponding author upon request.
References
Schmidt RA, Lee TD Motor learning and performance: from principles to application. Sixth edition. Champaign, IL: Human Kinetics; 2020.
Willingham DB. A neuropsychological theory of motor skill learning. Psychol Rev. 1998;105:558–84.
Dayan E, Cohen LG. Neuroplasticity Subserving Motor Skill Learning. Neuron. 2011;72:443–54.
Doyon J, Bellec P, Amsel R, Penhune V, Monchi O, Carrier J, et al. Contributions of the basal ganglia and functionally related brain structures to motor learning. Behav Brain Res. 2009;199:61–75.
Janacsek K, Shattuck KF, Tagarelli KM, Lum JAG, Turkeltaub PE, Ullman MT. Sequence learning in the human brain: A functional neuroanatomical meta-analysis of serial reaction time studies. NeuroImage. 2020;207:116387.
Nissen MJ, Bullemer P. Attentional requirements of learning: Evidence from performance measures. Cognit Psychol. 1987;19:1–32.
Robertson EM. The serial reaction time task: Implicit motor skill learning?. J Neurosci. 2007;27:10073–5.
Grafton ST, Hazeltine E, Ivry R. Functional mapping of sequence learning in normal humans. J Cogn Neurosci. 1995;7:497–510.
Rauch SL, Whalen PJ, Savage CR, Curran T, Kendrick A, Brown HD, et al. Striatal recruitment during an implicit sequence learning task as measured by functional magnetic resonance imaging. Hum Brain Mapp. 1997;5:124–32.
Boyd LA, Edwards JD, Siengsukon CS, Vidoni ED, Wessel BD, Linsdell MA. Motor sequence chunking is impaired by basal ganglia stroke. Neurobiol Learn Mem. 2009;92:35–44.
Krakauer JW, Mazzoni P. Human sensorimotor learning: adaptation, skill, and beyond. Curr Opin Neurobiol. 2011;21:636–44.
Ashe J, Lungu OV, Basford AT, Lu X. Cortical control of motor sequences. Curr Opin Neurobiol. 2006;16:213–21.
Doyon J, Penhune V, Ungerleider LG. Distinct contribution of the cortico-striatal and cortico-cerebellar systems to motor skill learning. Neuropsychologia. 2003;41:252–62.
Penhune VB, Steele CJ. Parallel contributions of cerebellar, striatal and M1 mechanisms to motor sequence learning. Behav Brain Res. 2012;226:579–91.
Steele CJ, Penhune VB. Specific increases within global decreases: a functional magnetic resonance imaging investigation of five days of motor sequence learning. J Neurosci Off J Soc Neurosci. 2010;30:8332–41.
Tanji J. Sequential Organization of Multiple Movements: Involvement of Cortical Motor Areas. Annu Rev Neurosci. 2001;24:631–51.
Wiestler T, Diedrichsen J. Skill learning strengthens cortical representations of motor sequences. Elife. 2013;2:e00801.
Poldrack RA, Sabb FW, Foerde K, Tom SM, Asarnow RF, Bookheimer SY, et al. The neural correlates of motor skill automaticity. J Neurosci Off J Soc Neurosci. 2005;25:5356–64.
Albouy G, Sterpenich V, Balteau E, Vandewalle G, Desseilles M, Dang-Vu T, et al. Both the hippocampus and striatum are involved in consolidation of motor sequence memory. Neuron. 2008;58:261–72.
Hikosaka O. A new approach to the functional systems of the brain. Epilepsia. 2002;43:9–15.
Ma L, Wang B, Narayana S, Hazeltine E, Chen X, Robin DA, et al. Changes in regional activity are accompanied with changes in inter-regional connectivity during 4 weeks motor learning. Brain Res. 2010;1318:64–76.
Packard MG, Knowlton BJ. Learning and Memory Functions of the Basal Ganglia. Annu Rev Neurosci. 2002;25:563–93.
Gamble KR, Cummings Jr. TJ, Lo SE, Ghosh PT, Howard Jr. JH, Howard DV. Implicit sequence learning in people with Parkinson’s disease. Front Hum Neurosci. 2014;8.
Seidler RD, Tuite P, Ashe J. Selective impairments in implicit learning in Parkinson’s disease. Brain Res. 2007;1137:104–10.
Floyer-Lea A, Matthews PM. Distinguishable brain activation networks for short- and long-term motor skill learning. J Neurophysiol. 2005;94:512–8.
Seidler RD, Purushotham A, Kim SG, Ugurbil K, Willingham D, Ashe J. Cerebellum activation associated with performance change but not motor learning. Science. 2002;296:2043–6.
Hardwick RM, Rottschy C, Miall RC, Eickhoff SB. A quantitative meta-analysis and review of motor learning in the human brain. NeuroImage. 2013;67:283–97.
Radua J, Mataix-Cols D, Phillips ML, El-Hage W, Kronhaus DM, Cardoner N, et al. A new meta-analytic method for neuroimaging studies that combines reported peak coordinates and statistical parametric maps. Eur Psychiatry. 2012;27:605–11.
Radua J, Rubia K, Canales-Rodríguez EJ, Pomarol-Clotet E, Fusar-Poli P, Mataix-Cols D. Anisotropic kernels for coordinate-based meta-analyses of neuroimaging studies. Front Psychiatry. 2014;5:13.
Müller VI, Cieslik EC, Laird AR, Fox PT, Radua J, Mataix-Cols D, et al. Ten simple rules for neuroimaging meta-analysis. Neurosci Biobehav Rev. 2018;84:151–61.
Landis JR, Koch GG. The measurement of observer agreement for categorical data. Biometrics. 1977;33:159.
Lancaster JL, Tordesillas-Gutiérrez D, Martinez M, Salinas F, Evans A, Zilles K, et al. Bias between MNI and Talairach coordinates analyzed using the ICBM-152 brain template. Hum Brain Mapp. 2007;28:1194–205.
Laird AR, Robinson JL, McMillan KM, Tordesillas-Gutiérrez D, Moran ST, Gonzales SM, et al. Comparison of the disparity between Talairach and MNI coordinates in functional neuroimaging data: Validation of the Lancaster transform. NeuroImage. 2010;51:677–83.
Xia M, Wang J, He Y. BrainNet viewer: A network visualization tool for human brain connectomics. PLoS ONE. 2013;8:e68910.
Button KS, Ioannidis JPA, Mokrysz C, Nosek BA, Flint J, Robinson ESJ, et al. Power failure: why small sample size undermines the reliability of neuroscience. Nat Rev Neurosci. 2013;14:365–76.
Ioannidis JPA. Why most published research findings are false. PLoS Med. 2005;2:e124.
Thirion B, Pinel P, Mériaux S, Roche A, Dehaene S, Poline J-B. Analysis of a large fMRI cohort: Statistical and methodological issues for group analyses. NeuroImage. 2007;35:105–20.
Egger M, Smith GD, Schneider M, Minder C. Bias in meta-analysis detected by a simple, graphical test. BMJ. 1997;315:629–34.
Smith S, Nichols T. Threshold-free cluster enhancement: Addressing problems of smoothing, threshold dependence and localisation in cluster inference. NeuroImage. 2009;44:83–98.
Winkler AM, Ridgway GR, Douaud G, Nichols TE, Smith SM. Faster permutation inference in brain imaging. NeuroImage. 2016;141:502–16.
Yan CG, Wang XD, Zuo XN, Zang YF. DPABI: Data processing & analysis for (Resting-State) brain imaging. Neuroinformatics. 2016;14:339–51.
Daselaar SM, Rombouts SARB, Veltman DJ, Raaijmakers JGW, Jonker C. Similar network activated by young and old adults during the acquisition of a motor sequence. Neurobiol Aging. 2003;24:1013–9.
Fitzroy AB, Kainec KA, Seo J, Spencer RMC. Encoding and consolidation of motor sequence learning in young and older adults. Neurobiol Learn Mem. 2021;185:107508.
Karim HT, Huppert TJ, Erickson KI, Wollam ME, Sparto PJ, Sejdic E, et al. Motor sequence learning-induced neural efficiency in functional brain connectivity. Behav Brain Res. 2017;319:87–95.
Landau SM, D’Esposito M. Sequence learning in pianists and nonpianists: an fMRI study of motor expertise. Cogn Affect Behav Neurosci. 2006;6:246–59.
Martis B, Wright CI, McMullin KG, Shin LM, Rauch SL. Functional magnetic resonance imaging evidence for a lack of striatal dysfunction during implicit sequence learning in individuals with animal phobia. Am J Psychiatry. 2004;161:67–71.
Muller R-A, Cauich C, Rubio MA, Mizuno A, Courchesne E. Abnormal activity patterns in premotor cortex during sequence learning in autistic patients. Biol Psychiatry. 2004;56:323–32.
Naismith SL, Lagopoulos J, Ward PB, Davey CG, Little C, Hickie IB. Fronto-striatal correlates of impaired implicit sequence learning in major depression: an fMRI study. J Affect Disord. 2010;125:256–61.
Oishi K, Toma K, Bagarinao ET, Matsuo K, Nakai T, Chihara K, et al. Activation of the precuneus is related to reduced reaction time in serial reaction time tasks. Neurosci Res. 2005;52:37–45.
Werheid K, Zysset S, Muller A, Reuter M, von Cramon DY. Rule learning in a serial reaction time task: an fMRI study on patients with early Parkinson’s disease. Brain Res Cogn Brain Res. 2003;16:273–84.
Woodward ND, Tibbo P, Purdon SE. An fMRI investigation of procedural learning in unaffected siblings of individuals with schizophrenia. Schizophr Res. 2007;94:306–16.
Zedkova L, Woodward ND, Harding I, Tibbo PG, Purdon SE. Procedural learning in schizophrenia investigated with functional magnetic resonance imaging. Schizophr Res. 2006;88:198–207.
Jouen AL, Verwey WB, van der Helden J, Scheiber C, Neveu R, Dominey PF, et al. Discrete sequence production with and without a pause: the role of cortex, basal ganglia, and cerebellum. Front Hum Neurosci. 2013;7:492.
Lacourse MG, Orr EL, Cramer SC, Cohen MJ. Brain activation during execution and motor imagery of novel and skilled sequential hand movements. NeuroImage. 2005;27:505–19.
Lin CH, Chiang MC, Wu AD, Iacoboni M, Udompholkul P, Yazdanshenas O, et al. Age related differences in the neural substrates of motor sequence learning after interleaved and repetitive practice. NeuroImage. 2012;62:2007–20.
Wymbs NF, Grafton ST. The human motor system supports sequence-specific representations over multiple training-dependent timescales. Cereb Cortex. 2015;25:4213–25.
Chen J, Fan Y, Jia X, Fan F, Wang J, Zou Q, et al. The supplementary motor area as a flexible hub mediating behavioral and neuroplastic changes in motor sequence learning: A TMS and TMS-EEG study. Neurosci Bull. 2025;41:837–52.
Narayana S, Laird AR, Tandon N, Franklin C, Lancaster JL, Fox PT. Electrophysiological and functional connectivity of the human supplementary motor area. NeuroImage. 2012;62:250–65.
Oullier O, Jantzen KJ, Steinberg FL, Kelso JAS. Neural substrates of real and imagined sensorimotor coordination. Cereb Cortex. 2005;15:975–85.
Säfström D, Domellöf E. Brain activations supporting linking of action phases in a sequential manual task. NeuroImage. 2018;172:608–19.
Honda M. Dynamic cortical involvement in implicit and explicit motor sequence learning. A PET study. Brain. 1998;121:2159–73.
Harpaintner M, Sim E-J, Trumpp NM, Ulrich M, Kiefer M. The grounding of abstract concepts in the motor and visual system: An fMRI study. Cortex. 2020;124:1–22.
Triarhou LC. Cytoarchitectonics of the Rolandic operculum: morphofunctional ponderings. Brain Struct Funct. 2021;226:941–50.
Steel A, Song S, Bageac D, Knutson KM, Keisler A, Saad ZS, et al. Shifts in connectivity during procedural learning after motor cortex stimulation: A combined transcranial magnetic stimulation/functional magnetic resonance imaging study. Cortex. 2016;74:134–48.
van Mier HI, Perlmutter JS, Petersen SE. Functional changes in brain activity during acquisition and practice of movement sequences. Motor Control. 2004;8:500–20.
Wilkinson L, Steel A, Mooshagian E, Zimmermann T, Keisler A, Lewis JD, et al. Online feedback enhances early consolidation of motor sequence learning and reverses recall deficit from transcranial stimulation of motor cortex. Cortex. 2015;71:134–47.
Zhang X, De Beukelaar TT, Possel J, Olaerts M, Swinnen SP, Woolley DG, et al. Movement observation improves early consolidation of motor memory. J Neurosci. 2011;31:11515–20.
Berlot E, Popp NJ, Diedrichsen J. A critical re-evaluation of fMRI signatures of motor sequence learning. Elife 2020;9.
Berlot E, Popp NJ, Grafton ST, Diedrichsen J. Combining repetition suppression and pattern analysis provides new insights into the role of M1 and parietal areas in skilled sequential actions. J Neurosci Off J Soc Neurosci. 2021;41:7649–61.
Karni A, Meyer G, Jezzard P, Adams MM, Turner R, Ungerleider LG. Functional MRI evidence for adult motor cortex plasticity during motor skill learning. Nature. 1995;377:155–8.
Halsband U, Lange RK. Motor learning in man: A review of functional and clinical studies. J Physiol-Paris. 2006;99:414–24.
Rieckmann A, Fischer H, Backman L. Activation in striatum and medial temporal lobe during sequence learning in younger and older adults: Relations to performance. NeuroImage. 2010;50:1303–12.
Schendan HE, Tinaz S, Maher SM, Stern CE. Frontostriatal and mediotemporal lobe contributions to implicit higher-order spatial sequence learning declines in aging and Parkinson’s disease. Behav Neurosci. 2013;127:204–21.
Bostan AC, Strick PL. The basal ganglia and the cerebellum: nodes in an integrated network. Nat Rev Neurosci. 2018;19:338–50.
Mattay VS, Fera F, Tessitore A, Hariri AR, Das S, Callicott JH, et al. Neurophysiological correlates of age-related changes in human motor function. Neurology. 2002;58:630–5.
Bernard JA, Nguyen AD, Hausman HK, Maldonado T, Ballard HK, Jackson TB, et al. Shaky scaffolding: Age differences in cerebellar activation revealed through activation likelihood estimation meta-analysis. Hum Brain Mapp. 2020;41:5255–81.
Dahms C, Brodoehl S, Witte O, Klingner C. The importance of different learning stages for motor sequence learning after stroke. Hum BRAIN Mapp. 2020;41:270–86.
Turner RS, Grafton ST, McIntosh AR, DeLong MR, Hoffman JM. The functional anatomy of parkinsonian bradykinesia. NeuroImage. 2003;19:163–79.
Dahms C, Noll A, Wagner F, Schmidt A, Brodoehl S, Klingner CM. Connecting the dots: Motor and default mode network crossroads in post-stroke motor learning deficits. Neuroimage Clin. 2024;42:103601.
Dovern A, Fink GR, Timpert DC, Saliger J, Karbe H, Weiss PH, et al. Timing matters? learning of complex spatiotemporal sequences in left-hemisphere stroke patients. J Cogn Neurosci. 2016;28:223–36.
Polanía R, Nitsche MA, Ruff CC. Studying and modifying brain function with non-invasive brain stimulation. Nat Neurosci. 2018;21:174–87.
Rossini PM, Burke D, Chen R, Cohen LG, Daskalakis Z, Di Iorio R, et al. Non-invasive electrical and magnetic stimulation of the brain, spinal cord, roots and peripheral nerves: Basic principles and procedures for routine clinical and research application. An updated report from an I.F.C.N. Committee. Clin Neurophysiol. 2015;126:1071–107.
Koch G, Esposito R, Motta C, Casula EP, Di Lorenzo F, Bonnì S, et al. Improving visuo-motor learning with cerebellar theta burst stimulation: Behavioral and neurophysiological evidence. NeuroImage. 2020;208:116424.
Shimizu T, Hanajima R, Shirota Y, Tsutsumi R, Tanaka N, Terao Y, et al. Plasticity induction in the pre-supplementary motor area (pre-SMA) and SMA-proper differentially affects visuomotor sequence learning. Brain Stimulat. 2020;13:229–38.
Thong S, Doery E, Biabani M, Rogasch N, Chong T, Hendrikse J, et al. Disinhibition across secondary motor cortical regions during motor sequence learning: A TMS-EEG STUDY. J Neurosci. 2025;45.
Hartwigsen G, Volz LJ. Probing rapid network reorganization of motor and language functions via neuromodulation and neuroimaging. NeuroImage. 2021;224:117449.
Kim YK, Shin SH. Comparison of effects of transcranial magnetic stimulation on primary motor cortex and supplementary motor area in motor skill learning (randomized, cross over study). Front Hum Neurosci 2014;8.
Deng ZD, Lisanby SH, Peterchev AV. Electric field depth–focality tradeoff in transcranial magnetic stimulation: Simulation comparison of 50 coil designs. Brain Stimulat. 2013;6:1–13.
Cash RFH, Weigand A, Zalesky A, Siddiqi SH, Downar J, Fitzgerald PB, et al. Using brain imaging to improve spatial targeting of transcranial magnetic stimulation for depression. Biol Psychiatry. 2021;90:689–700.
Wang JX, Rogers LM, Gross EZ, Ryals AJ, Dokucu ME, Brandstatt KL, et al. Targeted enhancement of cortical-hippocampal brain networks and associative memory. Science. 2014;345:1054–7.
World Medical Association. World Medical Association Declaration of Helsinki: Ethical principles for medical research involving human subjects. JAMA. 2013;310:2191.
Acknowledgements
This work was supported by grants from Medical and Health Science Program of Zhejiang Province (No. 2025HY0687). This work was supported by grants from Science and Technology Innovation 2030- “Brain Science and Brain-like Research” Major Project (2022ZD0210800), the Construction Fund of Key Medical Disciplines of HangZhou (2025HZGF02), the Zhejiang Provincial Natural Science Foundation of China (No. LGJ22H180001), and the Postgraduate Research Innovation Promotion Project of Hangzhou Normal University (No. 2025HSDYJSKY053).
Author information
Authors and Affiliations
Contributions
Weiwei Chen: Conceptualization, data curation, formal analysis, investigation, methodology, resources, validation, visualization, writing - original draft preparation. Tongyue Li: Data curation, investigation, methodology. Ji Luo: Data curation. Jing Chen: Conceptualization, methodology, project administration, supervision, writing - review & editing. Yating Lv: Conceptualization, funding acquisition, methodology, project administration, resources, supervision, validation, writing - review & editing.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Compliance with ethical standards
The validation study was approved by the Ethics Committee of the Affiliated Hospital of Hangzhou Normal University (No. 2023 (E2) - HS - 089) and was conducted in accordance with the Declaration of Helsinki [91]. All participants provided written informed consent prior to participation.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Chen, W., Li, T., Luo, J. et al. Consistent cortico-basal ganglia-cerebellar activation in early motor sequence learning: a systematic review and meta-analysis. Transl Psychiatry 16, 175 (2026). https://doi.org/10.1038/s41398-026-03851-8
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41398-026-03851-8