Abstract
NeuroHIV has emerged as a significant unmet challenge, manifesting as impairments in learning, memory, sensorimotor, and language in people living with HIV (PLWH). Despite the widespread use of antiretroviral therapy, the incidence of NeuroHIV remains high among PLWH. Microglial cells, as the main targets and reservoirs for HIV-1 infection, play crucial roles in sustaining HIV-1 persistence and contributing to neuroinflammation and neuroHIV pathogenesis. This review summarizes recent molecular studies that elucidate the role of HIV-1-related factors—such as the viral proteins trans-activator of transcription (Tat), gp120, and Vpr, HIV-1 RNAs, and associated microRNA and long non-coding RNAs and extracellular vesicles—in microglial activation through mechanisms including cellular senescence, ferroptosis, defective mitophagy, NLR family pyrin domain containing 3 activation, and NF-κB pathway modulation. These processes collectively lead to neuroinflammation and the development of NeuroHIV. This review presents the current understanding of non-replicative mechanisms in NeuroHIV, outlining challenges, shortcomings, and the current treatment status. Further, it identifies potential therapeutic targets linked to microglial activation. The overall goal is to stimulate further research into novel strategies for mitigating the neuroinflammatory processes driving cognitive decline.

Similar content being viewed by others
Introduction
According to current international consensus criteria, NeuroHIV encompasses a spectrum of disorders, including asymptomatic neurocognitive impairment (ANI), mild neurocognitive disorder (MND), and HIV-associated dementia (HAD), each characterized by a wide range of clinical symptoms such as cognitive, behavioral, and motor deficits [1]. Before antiretroviral therapy (ART) became widely available, severe cognitive impairments were observed in as many as 50% of individuals living with HIV (PLWH) [2]. With the introduction of ART, the estimated prevalence of NeuroHIV is approximately 42.6%. Within this population, the reported rates of ANI, MND, and HAD are 23.5, 13.3, and 5.0%, respectively [1]. Additionally, PLWH have a 58% increased likelihood of developing dementia compared to those without HIV-1 [3]. Notably, the use of ART has not been shown to reduce this risk, highlighting that HIV-1 is closely associated with the development of NeuroHIV even in those undergoing ART. This ongoing challenge underscores the need for effective interventions to address HAND in both ART-treated and ART-naïve individuals [4].
Several factors predispose individuals to NeuroHIV, including the direct neurotoxic effects of HIV-1, the inflammation it triggers in the brain (neuroinflammation), the potential neurotoxicity of ART drugs, and sociodemographic characteristics like age and the presence of co-occurring health conditions [5]. While the specific roles and interactions of direct viral mechanisms, indirect effects mediated by the host immune response, and these other contributing factors in NeuroHIV pathogenesis remain complex and not fully elucidated, growing evidence increasingly highlights the central role of microglial activation and subsequent neuroinflammation. These studies employ a diverse range of experimental models to investigate these processes, including human induced pluripotent stem cell (hiPSC)-derived tri-cultures of microglia, astrocytes, and neurons; single-cell RNA sequencing of human cerebrospinal fluid (CSF) microglia and myeloid cells; primary microglia isolated from mice; murine models of HIV infection; brain tissues from adult humans and nonhuman primates infected with HIV-1; and human brain organoids [6,7,8,9,10].
Microglia, the resident immune cells of the brain, a primary target and reservoir for HIV-1 in the central nervous system (CNS) due to several unique features supporting viral persistence [11]. The natural expression of HIV entry co-receptors, including C-C chemokine receptor type (CCR) 3, CCR5, and C-X-C chemokine receptor type 4 (CXCR4), by these cells promotes viral latency. This is achieved through mechanisms such as PR- and Nef-dependent induction of regulated upon activation, normal T cell expressed and secreted/C-C chemokine ligand 5 [12, 13]. Furthermore, compared to other CNS cells like macrophages and astrocytes, microglia maintain lower drug concentrations, exhibit a longer lifespan and a slower division rate, and are capable of extensive brain colonization [11]. These characteristics contribute to their role as both a significant viral reservoir and active mediator of neuroinflammation. Compelling evidence also demonstrates that HIV-1 related proteins (e.g., HIV-1 envelope glycoprotein 120) and RNAs from infected cells act as potent pro-inflammatory stimuli, triggering neurotoxic microglial activation [14, 15]. This activation leads to the release of cytokines, proteases, and reactive oxygen species, regulated by inflammasome activation, cell death pathways, and altered mitochondrial function [14, 15]. Collectively, microglia likely play a key role in maintaining HIV reservoirs and ongoing viral protein expression within the brain, ultimately contributing to neuroinflammation, neuronal damage, and the development of NeuroHIV.
In this review, we explore recent advances in understanding how HIV-1 related factors contribute to microglial activation, the induction of neuroinflammation, and the underlying mechanisms driving neurological impairments in NeuroHIV. Considering the substantial global prevalence of neurological disorders among PLWH, it is imperative to gain a deeper understanding of the pathobiology and identify potential therapeutic targets to develop new strategies for the prevention and treatment of NeuroHIV. To guide interpretation, we adopt an operational hierarchy of microglial signaling in PLWH. MicroRNA (miRNA)-sirtuin 3 (SIRT3), Acyl-CoA synthetase long-chain family member 4 (ACSL4), nuclear factor kappa B (NF-κB) and NLR family pyrin domain containing (NLRP) 3 act as upstream hubs; and ferroptosis, impaired mitophagy, and cellular senescence represent downstream effectors that sustain neuroinflammation. This hierarchy reflects necessity/sufficiency and temporal precedence reported across studies and clarifies which pathways are more upstream or dominant.
Overview of microglia and HIV-1 facrors
Microglia
Microglia, first published in the 1930s, are derived from the yolk sac and are characterized by the expression of the fractalkine receptor and the alpha chain of β(2) integrin Mac-1 [16, 17]. Their development, survival, and function are regulated by the colony-stimulating factor 1 receptor, the transcription factor PU.1, and interferon regulatory factor 8 [18]. However, the specific expression of these markers may vary across species and developmental stages. Microglia are unique from most tissue macrophages due to their distinct developmental origin and self-renewal capacity. Unlike macrophages that originate from hematopoietic stem cells in the bone marrow, microglia arise from precursors within the CNS during embryonic development. Under normal physiological conditions, they are largely self-sustaining, maintaining their population through local proliferation within the CNS, minimizing reliance on circulating monocytes [19, 20]. Studies estimate the lifespan of human cortical microglia to be approximately 4.2 years, suggesting a relatively slow turnover rate under homeostatic conditions. Nonetheless, their self-renewal and proliferation rates are modulated by various factors, including the brain region, disease state, and age, reflecting the dynamic and adaptable nature of microglial populations [18]. Microglia are widely distributed throughout the CNS and account for approximately 5–15% of all brain cells [21]. Notably, microglia exhibit considerable heterogeneous and dynamic changes in terms of genetic profiles, cell colonization, densities, morphology, as well as mobility and motility in the context of health or disease [22].
Microglia exhibit diverse functional states, enabling crosstalk with other CNS cell types and participation in peripheral immunity [22]. Acting as the brain’s first line of defense, microglia recognize pathogens through pattern recognition receptors, enabling them to present antigens, activate immune cells, and initiate protective immune responses within the CNS [23]. However, microglial activation can become detrimental under pathological conditions. For example, in vitro activation induced by Toll-like receptor 4 ligands like lipopolysaccharide leads to the production of pro-inflammatory cytokines and chemokines, resulting in reduced neuronal survival [24]. In HIV-1 infection, elevated levels of pro-inflammatory cytokines, including interleukin (IL)-8, IL-6, monocyte chemotactic protein 1 (MCP-1), and tumor necrosis factor (TNF)-α, have been observed in microglia within the human brain, microglial models, and studies using rhesus macaques [25]. The adoption of a pro-inflammatory phenotype in microglia can be driven by various factors, including the presence of pro-inflammatory mediators, infiltration of peripheral immune cells, pathological deposits (such as tau aggregates), and viral proteins [e.g., proteins trans-activator of transcription (Tat)] [26, 27].
Beyond their immune functions, microglia also perform critical non-immune roles, including synapse formation, pruning, and elimination, thereby regulating synaptic function to support neurogenesis [21]. They achieve these functions through phagocytosis of dying neurons and cellular debris and the secretion of neurotrophic factors and cytokines that modulate synaptic plasticity during both development and adulthood [28]. Moreover, microglia constantly monitor the microenvironment for potential signals in real-time through interactions with astrocytes, neurons, oligodendrocytes, and other immune cells; this is referred to as microglial surveillance [29]. However, as highlighted in other review articles, microglial dysfunction, often involving impaired communication with other cells, can contribute to neurodegeneration [14, 30]. For example, dysregulated microglial pruning can disrupt brain circuitry and serve as an indicator of cognitive decline [31, 32]. Furthermore, interaction with activated microglia can induce astrocytes to adopt a neurotoxic phenotype, impairing their essential support functions for neurons and exacerbating neuroinflammation [33]. Overall, these findings underscore the central role of microglial activation in the pathogenesis of various neurodegenerative diseases, including Alzheimer’s disease, Parkinson’s disease, multiple system atrophy, and viral infections of the brain [14, 27].
HIV-1 viral factors
HIV-1 proviruses may retain the ability to transcribe viral mRNAs and translate viral proteins [33]. The HIV-1 genome encodes 15 distinct proteins, including two structural proteins: Gag (matrix, capsid, nucleocapsid, and p6) for the virion core and Env (gp120 and gp41) for the outer membrane; one protease, Pol (reverse transcriptase, integrase, and protease); four accessory proteins: Vif, HIV-1 viral protein R (Vpr), viral protein U (Vpu) and Nef; and two regulatory proteins: Tat and Rev. Gp120, Tat, Vpr, and Nef are actively secreted by infected cells during HIV-1 infection, even in the presence of effective ART [34]. In addition to viral replication, these proteins can trigger responses and induce cytotoxic and pro-inflammatory effects in neurons, dendritic cells (DCs), microglia, and macrophages, and astrocyte across the CNS, thereby contributing to the development of NeuroHIV [35,36,37].
Additionally, the integrated HIV-1 provirus is approximately 9700 bp in length, and HIV-1 Rev binds directly to the Rev response element, leading to the accumulation of unspliced, partially spliced, and fully spliced RNA species, as well as rare ‘cryptic’ splice donors (5′ splice sites) and acceptors (3′ splice sites) [38]. Moreover, incompletely spliced viral transcripts facilitate their export from the nucleus by interacting with chromosomal region maintenance protein 1 (CRM1). This interaction leads to the formation of the Rev-CRM1-Ras-related nuclear protein (Ran)-guanosine triphosphate (GTP) complex, established through coupling with the GTP-bound form of Ran, a transport cofactor. Subsequently, CRM1 binds to nucleoporins, specifically Nucleoporin 214/Can protein, directing the complex toward the nuclear pore for transport, while fully spliced HIV-1 mRNAs can exit the nucleus without Rev [38]. Substantial evidence indicates that HIV RNAs persist in CSF and CNS cells despite effective ART [39, 40]. Among these species are unspliced transcripts whose nuclear export requires Rev binding to the RRE and CRM1. In this context, HIV-1 intron-containing RNA (icRNA)—the unspliced, intron-retaining transcript that is Rev/RRE-dependent and exported via CRM1—is detectable in previously infected CD4⁺ T cells, macrophages, and microglia. Export requires cis-acting RNA elements; when Rev is provided in trans, cytoplasmic accumulation and post-transcriptional handling of HIV-1 RNA are sensed as a danger signal, activating innate immunity [15, 41, 42]. Additionally, single-stranded RNA oligonucleotides derived from viruses, such as HIV-1, the influenza virus, and hepatitis C virus have been shown to activate immune response via toll-like receptor 7 and 8 [43, 44]. Uridine-rich single-stranded RNA oligonucleotides (ssRNA40) derived from HIV-1 can prompt natural killer cells, and microglia to secrete pro-inflammatory cytokines [45, 46]. These results demonstrate that the continued presence of HIV RNAs in microglia contributes to the ongoing inflammatory state and the development of neurological disorders during ART. HIV-associated inflammation can induce indoleamine 2,3-dioxygenase/Tryptophan 2,3-dioxygenase-mediated tryptophan catabolism to kynurenine (KYN), branching toward kynurenic acid (astrocyte-biased, anti-excitatory) and quinolinic acid (QUIN, microglia-biased, N-methyl-D-aspartate agonist) [47]. Elevated CSF QUIN has been linked to neurocognitive impairment, suggesting a bridge between immune activation and neurotoxic effects [48]. KYN metabolites also signal via NLRP2 inflammasome, potentially interfacing with NF-κB programs [49]. While direct evidence that specific HIV-1 proteins or transcripts drive microglial kynurenine pathway activation remains limited, acknowledging this axis improves completeness and indicates testable crosstalk with the hierarchy above.
The miRNAs and long non-coding RNAs (lncRNAs) and extracellular vesicles (EVs) induced by viral factors
MiRNAs and lncRNAs are two pivotal classes of regulatory RNAs. MiRNAs primarily fine-tune gene expression by post-transcriptionally repressing mRNA translation, while lncRNAs act more broadly through interactions with DNA, RNA, and proteins to regulate processes from chromatin remodeling to protein synthesis [50, 51]. Both classes mediate a complex interplay between host gene regulation and HIV-1, contributing to viral pathogenesis and associated neurodegenerative disorders [52, 53]. EVs are lipid bilayer–enclosed structures released by cells that carry nucleic acids, proteins, and metabolites reflective of their cell of origin. In the central nervous system, glia-derived EVs support neuronal homeostasis under physiological conditions but can become pathogenic during HIV-1 infection and neuroinflammation by delivering inflammatory signals, stress-response proteins, viral small RNAs, HIV proteins, and other neurotoxic mediators that impair neuronal function [54,55,56]. Mechanistically, glutaminase-mediated glutaminolysis and downstream α-ketoglutarate production are essential for pro-inflammatory EV release and microglial activation during HIV-1 infection and immune activation [54, 57]. In addition, HIV-1 Tat inhibits autophagy and proteasomal degradation in microglia and astrocytes, which in turn modulates EV biogenesis pathways and may amplify intercellular propagation of inflammatory signals [58, 59].
In recent years, the role of HIV transcripts in mediating microglial activation has garnered significant attention as a research hotspot in the progression of NeuroHIV, primarily due to their strong induction and sustainment of the inflammatory response.
The molecular mechanisms of HIV-1-related factors in microglial activation and neuroinflammation
HIV-1 Tat -mediated microglial activation
The persistence of HIV-1 reservoirs in PLWH with virological suppression results in the accumulation of HIV-1 Tat [36]. Early work established that Tat induces TNFα, IL-1β, nitric oxide, and increases nitric oxide synthase (NOS) activity and expression in microglia [60, 61]. Building on this foundation, we outline Tat’s effects primarily through miRNA-mediated pathways involving SIRT3, ACSL4, NF-κB, and NLRP3, as well as by impairing mitophagy/autophagy (Table 1). Additional chemokine programs are summarized below.
HIV-1 Tat-mediated microglial senescence by miRNA-505-SIRT3 pathway
Senescence was initially defined as the cessation of cell division after multiple replication cycles [62] and has expanded to include premature senescence triggered by telomere shortening, oxidative stress, mitochondrial dysfunction, and lysosomal alterations [63]. It features diminished proliferative capacity and increased secretion of pro-inflammatory cytokines and adhesion molecules [64, 65], and has been linked to AD and NeuroHIV [64, 65]. HIV-1 Tat exposure induces a senescence-like phenotype in microglia with elevated senescence markers and pro-inflammatory cytokines [66, 67]. Mitochondrial dysfunction can drive senescence via reactive oxygen species (ROS) accumulation [68, 69], and Tat (± morphine) perturbs mitochondrial function, increasing ROS, protein oxidation, and cytokine release in microglia [60, 70]. SIRT3, a class III HDAC associated with longevity, deacetylates Forkhead box protein O3a (FOXO3a) and promotes transcription of antioxidant enzymes (e.g., manganese superoxide dismutase and catalase), thereby constraining mitochondrial ROS and preserving redox homeostasis [71, 72]. Recent work shows that activating the SIRT3-FOXO3A axis promotes mitochondrial biogenesis and antioxidant enzyme expression [73]. Consistent with this mechanism, Tat upregulates miR-505, which represses SIRT3, leading to ROS accumulation, mitochondrial stress, and a senescence-like microglial phenotype; conversely, SIRT3 overexpression is protective [66]. These observations place the Tat/miR-505/SIRT3 axis upstream of microglial senescence in NeuroHIV (Fig. 1a). Exploring the relationship between senescence markers observed in vitro and their direct impact on in vivo NeuroHIV pathology, is necessary to fully understand how microglial senescence specifically affects neurocognitive disorders in PLWH. These findings may represent promising therapeutic targets, such as miR-505 and SIRT3, for addressing HIV-1 Tat-associated microglial senescence and NeuroHIV.
a HIV Tat-mediated microglial senescence. HIV-1 Tat triggers the miR-505, which negatively regulated the expression of sirtuin(SIRT3). This process may inhibit the expression of antioxidant genes such as manganese superoxide dismutase (MnSOD) and catalase by deacetylating forkhead box O3a (Foxo3a), contributing to mitochondrial oxidative stress and microglial senescence. b HIV Tat-mediated microglial ferroptosis via miR-204-Acyl-CoA synthetase long-chain family member 4 (ACSL4) signaling axis. The upregulation of ACSL4, resulting from the inhibition of miR-204, enhances the synthesis of phospholipid hydroperoxides (PLOOH). Concurrently, the downregulation of glutathione peroxidase-4 (GPX4) exacerbates the accumulation of phospholipid hydroperoxides (PLOOH), supplying fatty acids that promote lipid peroxidation and drive ferroptosis. c HIV Tat-mediated microglial activation by impairing mitophagy. HIV-1 Tat up-regulates mitophagy signaling proteins such as PTEN induced kinase 1 (PINK1), parkin RBR E3 ubiquitin protein ligase (PRKN) and dynamin-1-like protein (DNM1L) by decreasing the mitochondrial membrane potential. These processes impair PRKN-dependent mitophagy and the clearance of damaged mitochondria. d HIV Tat-mediated microglial activation by impairing mitophagy. Methamphetamine and HIV-1 Tat induces microglial autophagy via activation of the nuclear factor erythroid-2-related factor 2 (NRF2)/NAD(P)H:quinone oxidoreductase 1 (NQO1)/heme oxygenase-1 (HO-1) signal pathway.
HIV-1 Tat-mediated microglial ferroptosis via miR-204-ACSL4 signaling axis
Ferroptosis, a regulated form of cell death, has emerged as a relevant process in neurodegenerative disease pathogenesis [74]. During ferroptosis, Acyl-CoA synthetase long-chain family member 4 (ACSL4)-catalyzed arachidonic acid-CoA drives the production of phospholipid hydroperoxides (PLOOH) [75]. Ferritin heavy chain-1 (FTH1) plays an important role in storing iron, while glutathione peroxidase-4 (GPX4) helps to suppress ferroptosis by converting lipid hydroperoxides to lipid alcohols [76]. Iron accumulation, lipid peroxidation, and decreased GPX4 expression, which are characteristic hallmarks of ferroptosis, contribute to inflammation through the induction of oxidative stress and the activation of the high mobility group box-1 protein1-advanced glycosylation end-product-specific receptor signaling pathway and extracellular signal-regulated kinase, respectively [77,78,79]. Microglia are the most iron-rich cells in the CNS [80]. HIV-1 Tat resulted in microglial activation and pro-inflammatory responses by inducing ferroptosis with dramatically increased ACSL4 and FTH1, and decreased GPX4 [27]. The induction of Tat-mediated ferroptosis was observed following the downregulation of miR-204 expression, underscoring the significance of the miR-204-ACSL4 signaling axis (Fig. 1b) [27]. The nuclear factor erythroid-2-related factor 2 (NRF2) can block HIV-1 Tat- and methamphetamine-induced microglia ferroptosis through upregulating ATF4-induced solute carrier family 7a member 11 (SLC7A11) and GPX4 expression [81].
HIV-1 Tat-mediated microglial activation by impairing mitophagy/autophagy
Mitophagy selectively removes damaged mitochondria through a progression from phagophore to autophagosome and then to lysosome, thereby preserving organelle integrity and cellular homeostasis [82]. When this process is impaired, dysfunctional mitochondria accumulate, ROS rises, and inflammatory signaling is engaged—features linked to neurodegeneration [83, 84]. In microglia, HIV-1 Tat upregulates PTEN-induced kinase 1 (PINK1), Parkin RBR E3 ubiquitin protein ligase (PRKN), and dynamin-1-like protein (DNM1L), marking damaged mitochondria for turnover and promoting fission, yet sequestosome 1 (SQSTM1)-dependent flux is blocked, lowering the mitochondrial membrane potential and fostering microglial activation and neuroinflammation (Fig. 1c) [70, 85]. Enhancing the clearance of damaged mitochondria may therefore represent a therapeutic avenue in NeuroHIV.
Beyond mitophagy, macroautophagy also shapes microglial reactivity [86]. Tat, with or without methamphetamine, increases microtubule associated protein 1 light chain 3-II (LC3-II), Beclin-1, and the autophagy-related proteins ATG5 and ATG7. Activation of the NRF2 pathway, including induction of NAD(P)H:quinone oxidoreductase 1 (NQO1) and heme oxygenase-1 (HO-1), promotes autophagic clearance and restrains over-activation [87] (Fig. 1d). Under exposure to Tat, cocaine, or ART, lysosomal dysfunction is observed: lysosomal pH rises, lysosome-associated membrane protein 2 (LAMP2) decreases, and cathepsin D decreases. These alterations coincide with higher levels of Beclin-1, LC3B-II, and SQSTM1 and contribute to neuroinflammation and neurodegeneration [88, 89]. Microglia-specific autophagy also maintains lipid homeostasis, thereby modulating the inflammatory tone relevant to neurodegenerative disease [26, 86, 90].
HIV-1 Tat-induced microglial activation via ionic efflux and miR-223/ miR-124-NLRP3 -dependent pyroptosis
The NLRP3 inflammasome comprises three core components: NLRP3, a danger-sensing receptor; ASC, an adaptor that bridges NLRP3 to caspase-1; and caspase-1, the effector protease that processes IL-1β/IL-18 and activates gasdermin D (GSDMD) to induce pyroptosis [91]. NLRP3 activation proceeds in two stages. Priming is initiated when pattern-recognition receptors detect pathogen- or damage-associated molecular patterns and is reinforced by inflammatory cytokines; activation is then triggered by diverse microbial or sterile stimuli and is typically accompanied by potassium (K+) efflux and other ionic changes [91]. Ca2+ and cAMP can facilitate NLRP3 activation via the calcium-sensing receptor (CaSR) [92]. In HIV-specific contexts, Tat elevates intracellular Ca2+ by engaging N-methyl-d-aspartate receptors (NMDARs) and activating the NOS/sGC/PKG pathway in mixed glial–neuronal cultures, thereby potentiating neurotoxicity [93,94,95,96]. Tat also mobilizes Ca2+ from IP3-sensitive stores and activates novel PKC isoforms, enhancing TNF-α production across macrophages, neurons, and astrocytes [97, 98]. In BV2 microglia, ATP/P2X7-dependent Ca2+ influx gates the SUR1–TRPM4 channel, promoting Na+ entry, membrane depolarization, and secondary K+ efflux—a canonical trigger for NLRP3 inflammasome assembly [99]. Collectively, these HIV-related mechanisms link Ca2+ signals (for activation via CaSR and Ca2+ mobilization) with K+ efflux (as the proximal activation signal), integrating ion fluxes with microglial sensing and inflammasome priming/activation. Recent studies further connect sustained Ca2+ entry (via NMDAR or P2X7) to loss of mitochondrial membrane potential, increased mitochondrial ROS, and suppression of mitophagic clearance, collectively potentiating NLRP3 activation in microglia under HIV-related conditions [100].
Consistent with the ion-centric model above, HIV-1 Tat can upregulate NLRP3, promote ASC oligomerization and caspase-1 activation, and augment IL-1β release by inhibiting miR-223, thereby contributing to microglial activation [101] (Fig. 2a). In parallel, Tat-mediated downregulation of miR-124 increases SREBP2 expression and lipid-droplet accumulation in microglia, a metabolic context that further activates the NLRP3 inflammasome [102]. Once assembled, the NLRP3 complex triggers GSDMD-mediated pyroptosis, resulting in the release of pro-inflammatory cytokines and amplification of neuroinflammation [103,104,105,106] (Fig. 2a).
a HIV-1 Tat mediated the PYD domains-containing protein 3 (NLRP3) inflammasome activation by inhibiting miR-223. b HIV-1 Tat mediated the nuclear factor-kappaB (NF-κB) pathway through the upregulation of miR-34a, which subsequently downregulates the family member NLR caspase recruitment domain (CARD) containing 5 (NLRC5). This cascade ultimately leads to an increased expression of NF-κB p65, thereby activating NF-κB signaling.
HIV-1 Tat-induced microglial activation via miRNA-34a-NF-κB pathway
The NF-κB system, encompassing canonical and non-canonical branches, mediates inflammatory gene induction. In the canonical branch, IKKβ phosphorylates IκBα bound to p65/p50 heterodimers, targeting IκBα for proteasomal degradation; the liberated p65/p50 complex translocates to the nucleus and drives transcription [107,108,109]. Consistent with this framework, Tat activates NF-κB and promotes pro-inflammatory cytokine release in microglia [110]. At the molecular level, Tat upregulates miR-34a, which suppresses CARD-containing adaptors and increases NF-κB p65, thereby augmenting NF-κB signaling and triggering a pro-inflammatory microglial response that contributes to neuroinflammation [111] (Fig. 2b).
Other mechanisms involved in Tat-induced microglial activation
Tat also engages epigenetic and chemokine programs that reinforce microglial activation. It promotes DNA methylation of pro-inflammatory genes via the MECP2/STAT3 axis or the promoter DNA hypermethylation of miR-124 [112, 113]. In a COX-2–dependent manner, Tat induces CCL2/MCP-1, IL-1β, TNF-α, and iNOS; activation of NF-κB and the FABP4/NF-κB feedback loop facilitates recruitment of immune cells to the CNS, amplifying inflammation [114, 115]. Through MAPK and PI3K signaling, Tat further elevates chemokines: ERK1/2 and PI3K increase CCL2 and CCL4; p38 MAPK drives CXCL8 and CCL3; and combined ERK1/2, PI3K, and p38 MAPK signaling upregulates CXCL10 in microglia [116].
HIV-1 gp120-mediated microglial activation
Gp120, a glycoprotein on the surface of HIV-1, contributes significantly to the pathogenesis of HAND and leads to cognitive impairment and other neurological complications in PLWH (Table 1). Understanding the mechanism of gp120 in microglial activation is crucial for developing targeted therapeutic interventions to mitigate its effects on the CNS.
HIV-1 gp120-mediated microglial activation via CXCR4-NLRP3-dependent pyroptosis
During HIV-1 infection, the released gp120 activates microglia, resulting in rapid inflammasome activation. This process leads to increased expression of COX-2 and iNOS, as well as the release of various cytokines [117, 118]. Another study also showed that gp120 alone or together with methamphetamine activate NLRP3 and promote inflammatory response in microglia [37, 119]. Moreover, HIV-1 gp120 upregulated Kv1.3 expression via CXCR4 and p38-MAPK signaling (Fig. 3①) [120,121,122]. The binding of gp120 to CXCR4 causes both NF-κB activation and K+ efflux. CXCR4 signaling activates NF-κB, which upregulates the transcription of NLRP3 and pro-IL-1β, preparing the stage for inflammasome assembly (Fig. 3②). Additionally, K+ efflux triggers NLRP3 inflammasome assembly, caspase-1 activation, and IL-1β release and pyroptosis[123, 124]. These data demonstrate that HIV-1 gp120 triggers a pro-inflammatory response and neuropathology via CXCR4-NLRP3-dependent pyroptosis (Fig. 3①, ②). However, further elucidation of the connection between Kv1.3 and the NLRP3 inflammasome in gp120-mediated microglial activation could deepen our understanding of NeuroHIV [125].
a NLRP3 Inflammasome Activation: ① HIV-1 gp120 enhances Kv1.3 expression via C-X-C chemokine receptor type 4 (CXCR4) and p38-mitogen-activated protein kinase (p38-MAPK) signaling, leading to the activation of the NLRP3 inflammasome. ⑤ The viral protein Vpr directly activates the NLRP3 inflammasome. ⑥ HIV-1 ssRNA40 triggers NLRP3 inflammasome activation through toll-like receptor (TLR) 7/8-mediated mitochondrial damage. b NF-κB Pathway Activation: ② Binding of gp120 to CXCR4 activates the nuclear factor-kappa B (NF-κB) pathway, which then induces the transcription and expression of NLRP3 and pro-IL-1β. ③ HIV-1 gp120 also facilitates microglial activation through the purinergic 2×7 receptor (P2X7R)-mediated NF-κB signaling pathway. ④ Gp120 activate microglia by TLR 2-NF-κB signaling and by the direct binding of p62 and protein kinase C (PKC) through the IKK/NF-κB signaling pathway.
HIV-1 gp120-mediated microglial activation by NF-κB pathway
Another study has shown that gp120 induces over-activation of microglia by up-regulating P2X7R and promoting the formation of a p65 homodimer (Fig. 3③) [126, 127]. As reported, ATP can activate P2X7R by pannexin channels, subsequently activate NF-κB pathway through inducing the translocation of NF-κB and facilitating phosphorylation of IκB [128, 129]. These processes may result in NF-κB activation and maybe promote the expression of IL-1β, IL-6 and TNF-α in microglia (Fig. 3③) [127, 129]. However, the roles of P2X7R and NF-κB in microglia-associated neurotoxicity require more comprehensive understanding in HAND in future studies. Two other studies showed that gp120 activates microglia by TLR2-NF-κB signaling, and by the direct binding of p62 and PKC through the IKK/NF-κB signaling pathway (Fig. 3④) [130, 131].
Other HIV-1-related proteins on the microglial activation
While Tat and gp120 have received considerable attention, other HIV-1 proteins like Vpr and Nef are also known to play a role in microglial activation and inflammation. However, research exploring their specific mechanisms is still evolving [132,133,134]. This review provides a summary of the current research (Table 1). After Vpr stimulation, microglia exhibited increased translocation of inflammasome scaffold proteins from the nucleus to the cytoplasm, along with elevated levels of NLRP3, caspase-1, and IL-1 in both in vivo and in vitro settings [133] (Fig. 3⑤). Soluble Vpr released from infected microglia activates HIF-1α and perturbs neuronal–glial homeostasis, driving oxidative stress, elevation of intracellular Ca²⁺, excess ROS, and disruption of neuronal glutamatergic signaling [135, 136]. Meanwhile, Nef promotes neuropathology by upregulating CCL2 expression in microglia and augmenting IL-6 and IL-8 production in astrocytes via the PI3K/Akt/PKC, p38 MAPK, NF-κB, C/EBP, and AP-1 signaling pathways [137,138,139]. Nef also activates NADPH oxidase through the Vav/Rac/PAK pathway in phagocytes, amplifying neuroinflammation and promoting myelin injury and neuronal damage [134, 140]. Other studies have indicated that Vpr and Nef, or Nef and Tat in conjunction with gp41 exposure in microglia, stimulate the production of cytokines and chemokines, thereby contributing to the neuropathogenesis of HIV-1 [81, 141].
The roles of HIV-1 RNAs on microglia
HIV-1 icRNA-mediated microglial activation via Rev-CRM1-dependent nuclear export
HIV-1 icRNA has been shown to promote the release of type I interferon (IFN-I) and inflammatory cytokines in DCs, macrophages, and CD4+ T cells [142]. Further studies have implied that HIV-1 icRNA is sensed and signals via the mitochondrial antiviral signaling protein (MAVS) pathway, inducing IFN-I-dependent pro-inflammatory responses in macrophages [41]. Inhibition of transforming growth factor beta-activated kinase 1, NF-κB, and non-receptor tyrosine kinases, which are involved in MAVS activation, resulted in reduced Siglec1 expression and interferon-inducible protein-10 secretion in macrophages [41, 143]. These findings reveal the causative mechanisms of IFN-I-dependent pro-inflammatory response even in the absence of HIV-1 replication in PLWH [41, 142]. Recent research has also reported that HIV-1 icRNA could induce pro-inflammatory responses in microglia via Rev-CRM1-dependent nuclear export [15]. It also induces NLRP1 inflammasome activation and IL-1β secretion in macrophages and microglia independently of RLR and endosomal TLR pathways [144]. Rev-CRM1-dependent nuclear export and cytosolic sensing of HIV-1 icRNA trigger pro-inflammatory responses in productively infected microglia [15].
HIV-1 ssRNA40-mediated microglial activation via NLRP3 inflammasome
HIV-1 ssRNA40, as a potent trigger, is sufficient to prime NLRP3 inflammasome activation and the secretion of IL-1β, IL-18, TNF-α and IL-1α. Exposure to HIV-1 ssRNA40 induces the upregulation of pro-IL-1β, pro-IL-18, and NLRP3 through TLR7/TLR8. Activation of the NLRP3 inflammasome increased ROS generation with a loss of mitochondrial membrane potential and mitochondrial integrity in human microglial cells, independent of virus replication. By inhibiting autophagy/mitophagy with reduced optineurin, p62, TANK-binding kinase 1 phosphorylation, and PRKN expression, prolonged exposure of microglial cells to ssRNA40 amplified NLRP3 inflammasome activity, leading to increased inflammatory cytokine and ROS production and pyroptosis [46] (Fig. 3⑥).
The role of miRNAs, lncRNAs, and EVs induced by viral factors in microglial activation and inflammatory signaling
The role of miRNAs in microglial activation
The roles and mechanisms of specific microRNAs—including miR-204, miR-124, miR-505, and miR-34a—have been previously integrated into discussions of HIV-1 Tat-mediated microglial activation. Within this context, several Tat-responsive miRNAs critically influence innate immune signaling and redox homeostasis. For instance, miR-32 disrupts antiviral control by directly inhibiting TRAF3 and the downstream IRF3/IRF7 pathway [145]. Furthermore, Tat-induced suppression of miR-17 elevates NOX2/NOX4 levels, thereby increasing ROS production and augmenting microglial activation and cytokine release [146]. These findings collectively highlight a microRNA-driven mechanism underlying Tat-induced neuroinflammation.
The role of IncRNAs in microglial activation
HIV-1 Tat also engages lncRNA circuits that converge on chemokine signaling and neuronal homeostasis. Tat upregulates lncRNA Xist, which sponges miR-124, leading to CCL2 overexpression and microglial activation [147]. In parallel, Tat increases lncRNA-U1, which modulates the neuropeptide B/W receptor 1 locus and disrupts neuronal homeostatic signaling [52].
The role of EVs in microglial activation
EVs have a dual and consequential role in NeuroHIV. As potential biomarkers, CSF-derived EVs from neurons and glia exhibit distinct protein signatures that show promise for identifying cognitive impairment in PLWH [56]. Mechanistically, EVs act as delivery vehicles for neurotoxic cargo. Microglia- and astrocyte-derived EVs can carry HIV Nef and Tat; EV-associated Nef impairs the blood–brain barrier, induces oxidative stress, promotes Alzheimer’s-like pathology, and contributes to axonal degeneration, whereas EV-associated Tat directly causes neurite shortening and neuronal death [59, 139, 148,149,150,151]. EVs also mediate indirect toxicity by transferring miRNAs (e.g., miR-29b and miR-7) from astrocytes to neurons, thereby suppressing neuroprotective factors and disrupting synaptic integrity—effects that are potentiated by morphine [152, 153]. Additionally, Tat-conditioned microglial EVs enriched for NLRP3 convey pro-inflammatory signals and contribute to synaptodendritic injury, further impairing neuronal structure and function [58]. Taken together, EV trafficking furnishes a conduit for cell crosstalk, amplifies microglial activation and neuroinflammation, and links viral proteins, non-coding RNAs, and inflammatory mediators to progressive CNS pathology in NeuroHIV.
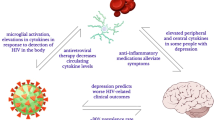
HIV-1 factors—including Tat, gp120, Vpr, Nef, viral RNAs, and related miRNA, IncRNA and EVs—collaboratively trigger pathogenic microglial responses, driving neuroinflammation and neurotoxicity in HAND through several converging pathways (Table 1). These include: 1) oxidative stress and mitochondrial damage, where viral factors induce ROS accumulation and mitochondrial dysfunction, leading to cellular damage, impaired autophagy/mitophagy, and primed inflammatory pathways; 2) NLRP3 inflammasome activation, triggered by Tat, gp120, and viral RNAs, which is exacerbated by ROS, ion flux changes, and impaired autophagy, with NF-κB acting as a crucial priming signal for pyroptosis and pro-inflammatory cytokine release; 3) NF-κB signaling, consistently activated by Tat, gp120, and Nef, which orchestrates the expression of inflammatory genes and inflammasome components, amplifying the inflammatory cascade; and 4) senescence and ferroptosis, both induced by Tat. Common upstream events, such as impaired autophagy/mitophagy and ROS accumulation, predispose microglia to these forms of dysfunction and death, thus contributing to a neurotoxic phenotype. Therefore, overlap is expected under sustained stimulation. Notably, senescence-associated changes in iron handling and lipid metabolism may feedback over time to lower the threshold for ferroptosis. However, their precise temporal sequence requires further investigation. While distinct mechanisms such as miRNA regulation and EVs transfer add complexity, these converging pathways collectively sustain a hazardous cycle of microglial activation, oxidative stress, programmed cell death, and chronic inflammation, ultimately driving neurodegeneration in HAND. Understanding these intricate interactions is essential for developing effective therapies.
Overview of the roles of microglial activation and HIV-1 factors in NeuroHIV
Microglial cells are primary target cells of HIV-1 infection and serve as reservoirs, contributing to the persistence of the virus in CNS. The connection between HIV-1 proteins, microglial activation, and neuronal cell death has been demonstrated, highlighting the crucial role of dysfunctional microglia in the development of NeuroHIV through various mechanisms.
First, microglia are considered one of the primary sources of viral proteins and RNAs within the CNS. These viral factors contribute to the progression of NeuroHIV by causing both “direct injury” and “bystander injury”. “Direct injury” occurs when gp120 and Tat directly damage neurons, for example, through impaired mitophagy [143]. Tat leads to neuronal death by activating calcium release from IP3-sensitive intracellular pools, causing endolysosomal dysfunction and dendritic impairments, which ultimately contribute to spatial memory impairment and premature brain aging [154,155,156,157,158].
In terms of “bystander injury”, viral factors induce neuronal toxicity through inflammatory processes. Tat and gp120 disrupt the BBB by downregulating tight junction proteins and promoting endothelial cell apoptosis [159,160,161]. This disruption promotes chemokine release in HIV-infected glial cells, which in turn recruits peripheral immune cells and induces a pro-inflammatory response within glial and peripheral immune cells [162,163,164]. For instance, Tat mediates astrocyte senescence and the production of ROS and pro-inflammatory cytokines, while gp120 and methamphetamine induce apoptosis [165, 166]. As for microglia, HIV-1 Tat and gp120 proteins promote cell loss and pro-inflammatory response via NLRP3 or NF-κB, which is strongly associated with NeuroHIV [27, 125]. In oligodendrocytes, Tat induces apoptosis, myelin damage, and white matter damage [94, 167, 168]. The dysfunction of neurons, astrocyte, and other cells exacerbates microglial activation, neuroinflammation, and NeuroHIV [169, 170].
Beyond the established neurotoxic roles of Tat and gp120, other HIV-1 components contribute to neurotoxicity through distinct mechanisms. Soluble Vpr released from infected microglia induces oxidative stress, disrupts cellular signaling, and impairs neuronal glutamate transmission [135, 137]. Nef promotes neuropathology by upregulating CCL2 in microglia and enhancing IL-6 and IL-8 production in astrocytes [137,138,139], while also activating NADPH oxidase in phagocytes, thereby exacerbating neuroinflammation, myelin damage, and neuronal injury [134, 140]. Concurrently, HIV-1 icRNA activates macrophages and microglia [144, 171] and triggers pro-inflammatory responses in productively infected microglia [15]. Similarly, HIV-1 ssRNA40 activates microglia, natural killer cells, dendritic cells, and macrophages [46, 172,173,174,175]. Furthermore, HIV-1 Tat, upon astrocytic exposure, induces the release of EV-encapsulated miR-7, which neurons subsequently internalize. This uptake leads to the downregulation of neuronal neuroligin 2 and ultimately results in synaptic alterations [153].
These viral factors and transcripts amplify cell–cell crosstalk across glia and neurons. Microglial cytokines and chemokines potentiate astrocyte activation, while astrocyte-derived mediators feed back to sustain microglial reactivity; together they intensify synaptic dysfunction and neuronal vulnerability. This reciprocity fosters intermittent cycles of viral reactivation and silencing in microglia and augments inflammatory signaling in macrophages, microglia, and astrocytes, creating a self-sustaining pro-inflammatory milieu in the CNS. The resulting positive-feedback loop perpetuates and intensifies neuroinflammation, thereby driving NeuroHIV pathogenesis [133, 136, 137]. In summary, these findings highlight the triad of microglia–astrocytes–neurons as an integrated network through which HIV-1 factors propagate CNS pathology (Fig. 4).
This figure illustrates the multifaceted roles of various brain cells in the pathogenesis of NeuroHIV, driven by HIV-1 factors and microglial activation: a Microglia activation: HIV-1 replication and transcription, the production of HIV-1 factors and associated miRNA, IncRNA and EVs, and proinflammatory cytokines, and interaction with other cells in the brain. b Neuron: neuronal death, endolysosomal dysfunction, and dendritic impairments, contributing to neurodegeneration; c The blood-brain barrier (BBB): reducing the expression of tight junction proteins, and inducing endothelial cell apoptosis. d Astrocytes: apoptosis, cellular senescence, and the release of proinflammatory cytokines; e Oligodendrocytes: apoptosis, myelin sheath damage, and white matter injury; Immune Cells: a proinflammatory environment in the brain; (f) Immune cells: the production of proinflammatory cytokines. Collectively, these cellular dysfunctions create a proinflammatory environment in the brain, leading to the spectrum of clinical manifestations of NeuroHIV, including dementia, mild neurocognitive disorder, and asymptomatic neurocognitive impairment.
Challenges and future aspects
Challenges in the experimental tools to study the exact mechanism of HIV-1-related factors on microglial activation
Experimental model limitations: many current models fail to replicate the complex microenvironment of the CNS, which often results in incomplete or potentially misleading interpretations of activation mechanisms. First, the representation of the CNS microenvironment in existing models is insufficient. While immortalized cell lines and conventional animal models are valuable, they do not accurately replicate the complex physiological and biochemical features of the human CNS in vivo. For instance, the interactions between neurons, glial cells (including microglia), and the extracellular matrix are intricately regulated and differ substantially from those observed in simplified laboratory settings. Moreover, species differences between animal models and humans complicate the translation of findings. Immune responses and neurobiological pathways in rodents may not fully mirror those in humans, leading to discrepancies in data interpretation and the effectiveness of potential therapeutic strategies. Second, accessing human brain tissue presents both ethical and logistical barriers that further complicate research. Restricted access limits researchers’ ability to study these cells in conditions that closely resemble in vivo environments, hindering the exploration of HIV-1-related factors in a relevant biological context. Viral factor representation: the representation and complexity of viral factors in experimental models are often inadequate. The viral factors used in in vitro experiments and animal studies frequently do not accurately mirror the structure, concentration, and dynamics present in actual HIV-1-infected individuals. Maintaining the correct conformation, concentrations, and post-translational modifications of HIV-1 proteins in experimental settings is particularly challenging. Furthermore, the complexity of HIV-1 is compounded by the existence of numerous viral subtypes that vary across different geographical regions. These variations can significantly influence the virus’s behavior, pathogenicity, and response to therapeutic interventions. Models that rely on specific viral factors may lack the generalizability needed to inform clinical outcomes effectively, potentially overlooking critical differences in viral dynamics and host interactions. This discrepancy can skew research findings. Given these challenges, there is an urgent need for the development of new experimental models that more accurately reflect the human CNS environment, as follows: 1) Innovative in vitro models: Developing advanced 3D culture systems or organoid models that replicate the cellular architecture and microenvironment of the CNS could offer more relevant insights into HIV-1 interactions with microglia and other brain cells. Recent approaches have adopted pluripotent stem cell cultures and 3D brain organoid systems that incorporate microglia derived from hematopoietic progenitor cells into these organoids [10, 176, 177]. Three-dimensional brain organoids derived from human iPSCs, augmented with HIV-1–susceptible microglia, recapitulate key features of human cellular diversity and permit direct visualization of neuroinvasion, including directed migration of infected microglia into neural tissue. In parallel, humanized mouse models with peripheral immune reconstitution and microglial engraftment enable evaluation of neuroinvasion, reservoir dynamics, and microglia-targeted interventions under systemic antiretroviral exposure. 2) Humanized animal models: Creating humanized mouse models that incorporate human CNS components and immune systems may help bridge the gap between animal studies and human biology. 3) Characterization of viral factors: Investigating the precise structure and concentration of HIV-1-related factors in HIV-1-infected patients is essential.
While considerable research has focused on Tat and gp120 in microglial cells, more detailed and in-depth studies are still required. Additionally, although Vpr, Nef, and HIV-1 icRNA, ssRNA40, miRNA, IncRNA and EVs play significant roles in microglial activation, current research on these factors remains limited. Therefore, it is essential to further investigate the roles of these factors. Addressing these gaps is crucial for enhancing our understanding and developing effective treatments for NeuroHIV.
Challenges in the therapy of NeuroHIV
As ART alone is insufficient to fully suppress HIV-1 replication, including the transcription and translation of HIV-1 provirus in both T-cell reservoirs and non-CD4 T-cell reservoirs, there is a critical need to develop new brain-penetrating agents that can effectively target both CNS and peripheral immune cells to simultaneously silence HIV-1 and reduce the risk of neuroinflammation. Currently, eliminating the latent HIV-1 reservoir to achieve a functional cure for HIV/AIDS remains a top priority. Immunotherapies such as broadly neutralizing antibodies, HIV-specific CAR-T cells, and immune-checkpoint blockers are particularly attractive for inducing a sustained HIV-1-specific immune response [178,179,180]. However, the safety and efficacy of these treatments in the context of NeuroHIV require thorough investigation, particularly due to CNS‑related toxicities. Understanding the biology of the latent HIV-1 reservoir is critical for developing effective curative strategies. Research on latency mechanisms and the interplay between HIV-1 and host immune responses will be instrumental in designing targeted therapies. Additionally, advancements in gene-editing technologies, such as CRISPR/Cas9, show promise for excising proviral DNA from the host genome [181]. Although developing effective curative interventions is challenging, continued progress in elucidating the biology of the latent HIV-1 reservoir will enhance our ability to achieve a functional cure.
As ART alone is insufficient to fully suppress HIV-1 replication, including the transcription and translation of the HIV-1 provirus, there is a critical need to develop new brain-penetrat agents that can effectively target both the CNS and the peripheral immune system to silence HIV-1 and reduce the risk of neuroinflammation. Recent research has shown that the nerve growth factor IB-like receptor (Nurr1/NR4A2) and the glucocorticoid receptor (GR, NR3C1) not only effectively silence HIV-1 in reactive microglial cells by repressing HIV-1 gene expression, but also induce a quiescent state in these cells [182, 183]. Additionally, epigenetic-related molecules, including histone lysine methyltransferases and histone deacetylases are involved in HIV-1 post-integration latency. For instance, lysine 4 of histone H3 and histone H3 lysine 27 trimethylation were found to be enriched in latent HIV-1 chromatin in both microglial cells and CD4+ T cells, suggesting that targeting these epigenetic pathways may suppress HIV-1 latency and microglial activation [184, 185]. Furthermore, several approaches aim to target transcription and/or RNA export to counteract the effects of proinflammatory cytokines and inhibit viral protein synthesis: 1) Inhibition of Tat protein expression through didehydro-cortistatin A, 2) Inhibition of HIV integration and relocalization of the residual virus by Lens epithelium-derived growth factor p75, 3) Small interfering RNA (siRNA) silencing HIV gene transcription by recruiting Argonaute1, histone deacetylase 1, and histone methyltransferase, 4) Heat shock protein 90 inhibitors, Janus kinase (Jak)-STAT inhibitors, kinase inhibitors, and bromodomain protein 4 regulators, 5) CRISPR/Cas9-guided RNA [100, 186,187,188]. However, it remains to be investigated whether these agents or RNA molecules can effectively cross the blood-brain barrier and safely inhibit HIV-1 expression in microglia.
Recent research into microglia and HIV-1 factors has underscored the central mechanisms involved in this process include NLRP3 inflammasome activation (e.g., GSDMD, GSDME, NEK7, ASC, caspase-1 and IL-1β) and NF-κB signaling pathways (e.g., NF-κB, P2X7R, IKK, nuclear translocation inhibitors, DNA binding inhibitors), along with cellular processes such as autophagy (e.g., PINK1, Parkin and DNM1L), ferroptosis (e.g., FTH1, ACSL4, miR-204, and GPX4), and senescence (e.g., miR-505 and SIRT3), highlighting critical targets for further investigation and therapeutic intervention in NeuroHIV. In preclinical CNS models, multiple strategies targeting the NLRP3 inflammasome axis have shown promise. These include direct NLRP3 inhibitors (e.g., CY-09, MCC950, OLT1177/dapansutrile, oridonin, INF39, tranilast), blockade of pyroptosis or downstream effectors (e.g., disulfiram, dimethyl fumarate, maresin-1), and interference with adaptor or cytokine pathways such as ASC, NEK7, caspase-1, and IL-1β (e.g., lycorine, entrectinib, VX-765/X-740, canakinumab). Across diverse CNS disease contexts, these agents have reduced inflammatory readouts and improved functional outcomes in vivo [189,190,191]. Furthermore, some CNS-penetrant NLRP3 inhibitors have entered phase human trials for neuroinflammatory diseases and neurodegenerative diseases, such as VTX3232, Emlenoflast, NT-0796. Emlenoflast and NT-0796 were announced safe and well-tolerated, suggesting promising clinical efficacy in adult patients [153,154,155, 192,193,194]. Although minocycline showed anti-inflammatory and neuroprotective effects preclinically and crosses the blood–brain barrier [195, 196], randomized clinical trials in HAND did not improve cognitive benefit [197]. We therefore present minocycline as negative clinical evidence that nonetheless informed microglial-targeting hypotheses rather than as an effective therapy. These findings indicate the transition from preclinical studies to clinical applications remains a challenge. Several constraints limit brain-penetrant anti-inflammatory agents in NeuroHIV/HAND. First, pathway complexity raises on- and off-target risks when modulating ASC, caspase-1, GSDMD, IL-1β, NF-κB, P2X7R, IKK, FTH1, ACSL4, miR-204, or GPX4, and microglia-specific target engagement is difficult to confirm. Second, restricts distribution and is subject to active efflux, yielding heterogeneous brain exposure across regions and patients. Third, most inhibitors of NLRP3, NF-κB, autophagy, ferroptosis, or senescence remain preclinical and lack regulatory approval, and there is a paucity of validated pharmacodynamic biomarkers in humans (e.g., CSF IL-1β/IL-18, GSDMD-N, ex vivo NLRP3 assays) to confirm target engagement. Additional translational hurdles include potential drug–drug interactions with antiretroviral therapy, uncertainty in therapeutic timing relative to disease stage, and variability of cognitive endpoints in HAND trials. Opportunities for progress include rational brain-exposure design with efflux avoidance, development of microglia-directed delivery systems, and incorporation of human target-engagement biomarkers into early-phase studies. Notably, some nanomaterials can themselves activate NLRP3 in the CNS [198], underscoring the need for careful nanoengineering and mechanistic evaluation. Drug repurposing remains attractive when compounds already possess favorable pharmacokinetic properties and demonstrable BBB penetrance. For instance, microRNAs—particularly microRNA-7—and nanobodies may exhibit remarkable therapeutic potential [199]. This is especially important for biologics that can cross the blood-brain barrier to effectively control inflammatory injury and mitigate the corresponding deleterious effects on the brain and peripheral tissues.
Outlook
This review focused on how HIV-1 proteins and RNAs activate microglia. However, further research is needed to fully understand these mechanisms and their effects on other CNS and blood cells. Understanding HIV-1 factors could improve our knowledge of the complex disease process and lead to new treatments targeting harmful bystander effects, independent of viral replication.
Conclusion
In conclusion, this review provides a comprehensive summary of recent molecular research shedding light on the complex mechanisms through which HIV-1-related factors—such as viral proteins and RNAs—mediate microglial activation, leading to neuroinflammation and the development of NeuroHIV. By highlighting the various pathways involved, including NLRP3 inflammasome activation, NF-κB signaling, and autophagy/mitophagy dysfunction, this review aims to inspire further investigation into these intricate molecular processes in the absence of active viral replication in the brain.
References
Wang Y, Liu M, Lu Q, Farrell M, Lappin JM, Shi J, et al. Global prevalence and burden of HIV-associated neurocognitive disorder: a meta-analysis. Neurology. 2020;95:e26 10–e2621.
Grant I, Atkinson JH, Hesselink JR, Kennedy CJ, Richman DD, Spector SA, et al. Evidence for early central nervous system involvement in the acquired immunodeficiency syndrome (AIDS) and other human immunodeficiency virus (HIV) infections. studies with neuropsychologic testing and magnetic resonance imaging. Ann Intern Med. 1987;107:82 8–36.
Lam JO, Hou CE, Hojilla JC, Anderson AN, Gilsanz P, Alexeeff SE, et al. Comparison of dementia risk after age 50 between individuals with and without HIV infection. AIDS. 2021;35:821–8.
Borrajo A, Pérez-Rodríguez D, Fernández-Pereira C, Prieto-González JM, Agís-Balboa RC. Genomic factors and therapeutic approaches in HIV-Associated neurocognitive disorders: a comprehensive review. Int J Mol Sci. 2023;24:14364.
Mastrorosa I, Pinnetti C, Brita AC, Mondi A, Lorenzini P, Del Duca G, et al. Declining prevalence of human immunodeficiency virus (HIV)-Associated neurocognitive disorders in recent years and associated factors in a large cohort of antiretroviral therapy-treated individuals with HIV. Clin Infect Dis. 2023;76:e629–e637.
Ryan SK, Gonzalez MV, Garifallou JP, Bennett FC, Williams KS, Sotuyo NP, et al. Neuroinflammation and EIF2 Signaling persist despite antiretroviral treatment in an hiPSC Tri-culture Model of HIV Infection. Stem Cell Rep. 2020;14:703–16.
Esaulova E, Cantoni C, Shchukina I, Zaitsev K, Bucelli RC, Wu GF, et al. Single-cell RNA-seq analysis of human CSF microglia and myeloid cells in neuroinflammation. Neurol Neuroimmunol Neuroinflamm. 2020;7:e732.
Jin M, Shiwaku H, Tanaka H, Obita T, Ohuchi S, Yoshioka Y, et al. Tau activates microglia via the PQBP1-cGAS-STING pathway to promote brain inflammation. Nat Commun. 2021;12:6565.
Branton WG, Fernandes JP, Mohammadzadeh N, Doan MAL, Laman JD, Gelman BB, et al. Microbial molecule ingress promotes neuroinflammation and brain CCR5 expression in persons with HIV-associated neurocognitive disorders. Brain Behav Immun. 2023;107:110–23.
Kong W, Frouard J, Xie G, Corley MJ, Helmy E, Zhang G, et al. Neuroinflammation generated by HIV-infected microglia promotes dysfunction and death of neurons in human brain organoids. PNAS Nexus. 2024;3:pgae179.
Wallet C, De Rovere M, Van Assche J, Daouad F, De Wit S, Gautier V, et al. Microglial Cells: the Main HIV-1 Reservoir in the Brain. Front Cell Infect Microbiol. 2019;9:362.
He J, Chen Y, Farzan M, Choe H, Ohagen A, Gartner S, et al. CCR3 and CCR5 are co-receptors for HIV-1 infection of microglia. Nature. 1997;385:645–9.
Silva MJA, Marinho RL, Rodrigues YC, Brasil TP, Dos Santos PAS, Silva CS, et al. Molecular Role of HIV-1 Human Receptors (CCL5-CCR5 Axis) in neuroAIDS: a systematic review. Microorganisms. 2024;12:782.
Gao C, Jiang J, Tan Y, Chen S. Microglia in neurodegenerative diseases: mechanism and potential therapeutic targets. Signal Transduct Target Ther. 2023;8:359.
Akiyama H, Jalloh S, Park S, Lei M, Mostoslavsky G, Gummuluru S. Expression of HIV-1 Intron-Containing RNA in microglia induces inflammatory responses. J Virol. 2021;95:e01386-20.
Kierdorf K, Erny D, Goldmann T, Sander V, Schulz C, Perdiguero EG, et al. Microglia emerge from erythromyeloid precursors via Pu.1- and Irf8-dependent pathways. Nat Neurosci. 2013;16:273–80.
Tay TL, Savage JC, Hui CW, Bisht K, Tremblay MÈ. Microglia across the lifespan: from origin to function in brain development, plasticity and cognition. J Physio. 2017;595:1929–45.
Tay, Mai TL, Dautzenberg D, Fernández-Klett J, Lin F, Sagar G, et al. A new fate mapping system reveals context-dependent random or clonal expansion of microglia. Nat Neurosci. 2017;20:793–803.
Prinz M, Jung S, Priller J. Microglia biology: one century of evolving concepts. Cell. 2019;179:292–311.
Gomez Perdiguero E, Schulz C, Geissmann F. Development and homeostasis of “resident” myeloid cells: the case of the microglia. Glia. 2013;61:112–20.
Thion MS, Ginhoux F, Garel S. Microglia and early brain development: an intimate journey. Science. 2018;362:185–9.
Tan YL, Yuan Y, Tian L. Microglial regional heterogeneity and its role in the brain. Mol Psychiatry. 2020;25:351–67.
Rodríguez-Gómez JA, Kavanagh E, Engskog-Vlachos P, Engskog MKR, Herrera AJ, Espinosa-Oliva AM, et al. Microglia: agents of the CNS Pro-Inflammatory Response. Cells. 2020;9:1717.
Durafourt BA, Moore CS, Zammit DA, Johnson TA, Zaguia F, Guiot MC, et al. Comparison of polarization properties of human adult microglia and blood-derived macrophages. Glia. 2012;60:717–27.
Chen NC, Partridge AT, Sell C, Torres C, Martín-García J. Fate of microglia during HIV-1 infection: From activation to senescence?. Glia. 2017;65:431–44 6.
Xu Y, Propson NE, Du S, Xiong W, Zheng H. Autophagy deficiency modulates microglial lipid homeostasis and aggravates tau pathology and spreading. Proc Natl Acad Sci USA. 2021;118:e2023418118.
Kannan M, Sil S, Oladapo A, Thangaraj A, Periyasamy P, Buch S. HIV-1 Tat-mediated microglial ferroptosis involves the miR-204-ACSL4 signaling axis. Redox Biol. 2023;62:102689.
Cornell J, Salinas S, Huang HY, Zhou M. Microglia regulation of synaptic plasticity and learning and memory. Neural Regen Res. 2022;17:705–16.
Prinz M, Masuda T, Wheeler MA, Quintana FJ. Microglia and central nervous system-associated macrophages-from origin to disease modulation. Annu Rev Immunol. 2021;39:251–77.
Salter MW, Stevens B. Microglia emerge as central players in brain disease. Nat Med. 2017;23:1018–27.
Koffie RM, Hyman BT, Spires-Jones TL. Alzheimer’s disease: synapses gone cold. Mol Neurodegener. 2011;6:63.
Wilton DK, Dissing-Olesen L, Stevens B. Neuron-Glia signaling in synapse elimination. Annu Rev Neurosci. 2019;42:107–27.
Dubé M, Tastet O, Dufour C, Sannier G, Brassard N, Delgado GG, et al. Spontaneous HIV expression during suppressive ART is associated with the magnitude and function of HIV-specific CD4+ and CD8+ T cells. Cell Host Microbe. 2023;31:1507–1522.e5.
Maubert ME, Pirrone V, Rivera NT, Wigdahl B, Nonnemacher MR. Interaction between Tat and Drugs of Abuse during HIV-1 infection and central nervous system disease. Front Microbiol. 2016;6:1512.
Santerre M, Arjona SP, Allen CN, Callen S, Buch S, Sawaya BE. HIV-1 Vpr protein impairs lysosome clearance causing SNCA/alpha-synuclein accumulation in neurons. Autophagy. 2021;17:1768–82.
Marino J, Maubert ME, Mele AR, Spector C, Wigdahl B, Nonnemacher MR. Functional impact of HIV-1 Tat on cells of the CNS and its role in HAND. Cell Mol Life Sci. 2020;77:5 079–5099.
He X, Yang W, Zeng Z, Wei Y, Gao J, Zhang B, et al. NLRP3-dependent pyroptosis is required for HIV-1 gp120-induced neuropathology. Cell Mol Immunol. 2020;17:283–99.
Ocwieja KE, Sherrill-Mix S, Mukherjee R, Custers-Allen R, David P, Brown M, et al. Dynamic regulation of HIV-1 mRNA populations analyzed by single-molecule enrichment and long-read sequencing. Nucleic Acids Res. 2012;40:10345–55.
Peluso MJ, Ferretti F, Peterson J, Lee E, Fuchs D, Boschini A, et al. Cerebrospinal fluid HIV escape associated with progressive neurologic dysfunction in patients on antiretroviral therapy with well controlled plasma viral load. AIDS. 2012;26:1765–74.
Dahl V, Peterson J, Fuchs D, Gisslen M, Palmer S, Price RW. Low levels of HIV-1 RNA detected in the cerebrospinal fluid after up to 10 years of suppressive therapy are associated with local immune activation. AIDS. 2014;28:2251–8.
Akiyama H, Miller CM, Ettinger CR, Belkina AC, Snyder-Cappione JE, Gummuluru S. HIV-1 intron-containing RNA expression induces innate immune activation and T cell dysfunction. Nat Commun. 2018;9:3450.
McCauley SM, Kim K, Nowosielska A, Dauphin A, Yurkovetskiy L, Diehl WE, et al. Intron-containing RNA from the HIV-1 provirus activates type I interferon and inflammatory cytokines. Nat Commun. 2018;9:5305.
Zhang YL, Guo YJ, Bin Li, Sun SH. Hepatitis C virus single-stranded RNA induces innate immunity via Toll-like receptor 7. J Hepatol. 2009;51:29–38.
Heil F, Hemmi H, Hochrein H, Ampenberger F, Kirschning C, Akira S, et al. Species-specific recognition of single-stranded RNA via toll-like receptor 7 and 8. Science. 2004;303:1526–9.
Ao X, Gan Q, Huang X, Bao D, Wu X, Lin Q, et al. TLR8 agonist partially improves IFN-γ deficiency of NK cells in chronic hepatitis B through the synergy of monocytes. Aliment Pharmacol Ther. 2023;57:387–98.
Rawat P, Teodorof-Diedrich C, Spector SA. Human immunodeficiency virus Type-1 single-stranded RNA activates the NLRP3 inflammasome and impairs autophagic clearance of damaged mitochondria in human microglia. Glia. 2019;67:802–24.
Kandanearatchi A, Brew BJ. The kynurenine pathway and quinolinic acid: pivotal roles in HIV associated neurocognitive disorders. FEBS J. 2012;279:1366–74.
Li X, Edén A, Malwade S, Cunningham JL, Bergquist J, Weidenfors JA, et al. Central and peripheral kynurenine pathway metabolites in COVID-19: Implications for neurological and immunological responses. Brain Behav Immun. 2025;124:163–76.
Zhang Q, Sun Y, He Z, Xu Y, Li X, Ding J, et al. Kynurenine regulates NLRP2 inflammasome in astrocytes and its implications in depression. Brain Behav Immun. 2020;88:471–81.
Fang JH, Zhou HC, Zeng C, Yang J, Liu Y, Huang X, et al. MicroRNA-29b suppresses tumor angiogenesis, invasion, and metastasis by regulating matrix metalloproteinase 2 expression. Hepatology. 2011;54:1729–40.
Herman AB, Tsitsipatis D, Gorospe M. Integrated lncRNA function upon genomic and epigenomic regulation. Mol Cell. 2022;82:2252–66.
Torkzaban B, Natarajaseenivasan K, Mohseni Ahooyi T, Shekarabi M, Amini S, Langford TD, et al. The lncRNA LOC102549805 (U1) modulates neurotoxicity of HIV-1 Tat protein. Cell Death Dis. 2020;11:835.
Eacker SM, Dawson TM, Dawson VL. Understanding microRNAs in neurodegeneration. Nat Rev Neurosci. 2009;10:837–41. https://doi.org/10.1038/nrn2726.
Wu B, Liu J, Zhao R, Li Y, Peer J, Braun AL, et al. Glutaminase 1 regulates the release of extracellular vesicles during neuroinflammation through key metabolic intermediate alpha-ketoglutarate. J Neuroinflammation. 2018;15:79.
Pérez PS, Romaniuk MA, Duette GA, Zhao Z, Huang Y, Martin-Jaular L, et al. Extracellular vesicles and chronic inflammation during HIV infection. J Extracell Vesicles. 2019;8:1687275.
Guha D, Lorenz DR, Misra V, Chettimada S, Morgello S, Gabuzda D. Proteomic analysis of cerebrospinal fluid extracellular vesicles reveals synaptic injury, inflammation, and stress response markers in HIV patients with cognitive impairment. J Neuroinflammation. 2019;16:254.
Gao G, Li C, Zhu J, Wang Y, Huang Y, Zhao S, et al. Glutaminase 1 regulates neuroinflammation after cerebral ischemia through enhancing microglial activation and pro-inflammatory exosome release. Front Immunol. 2020;11:161.
Kannan M, Singh S, Chemparathy DT, Oladapo AA, Gawande DY, Dravid SM, et al. HIV-1 Tat induced microglial EVs leads to neuronal synaptodendritic injury: microglia-neuron cross-talk in NeuroHIV. Extracell Vesicles Circ Nucl Acids. 2022;3:133–49.
Chemparathy DT, Ray S, Ochs C, Ferguson N, Gawande DY, Dravid SM, et al. Neuropathogenic role of astrocyte-derived extracellular vesicles in HIV-associated neurocognitive disorders. J Extracell Vesicles. 2024;13:e12439.
Turchan-Cholewo J, Dimayuga FO, Gupta S, Keller JN, Knapp PE, Hauser KF, et al. Morphine and HIV-Tat increase microglial-free radical production and oxidative stress: possible role in cytokine regulation. J Neurochem. 2009;108:202–15.
Turchan-Cholewo J, Dimayuga VM, Gupta S, Gorospe RM, Keller JN, Bruce-Keller AJ. NADPH oxidase drives cytokine and neurotoxin release from microglia and macrophages in response to HIV-Tat. Antioxid Redox Signal. 2009;11:193–204.
Hayflick L. The limited in vitro lifetime of human diploid cell strains. Exp Cell Res. 1965;37:614–36.
Liu RM. Aging, cellular senescence, and alzheimer’s disease. Int J Mol Sci. 2022;23:1989.
Baker DJ, Petersen RC. Cellular senescence in brain aging and neurodegenerative diseases: evidence and perspectives. J Clin Invest. 2018;128:1208–16.
Marino J, Wigdahl B, Nonnemacher MR. Extracellular HIV-1 tat mediates increased glutamate in the CNS Leading to Onset of Senescence and Progression of HAND. Front Aging Neurosci. 2020;12:168.
Thangaraj A, Chivero ET, Tripathi A, Singh S, Niu F, Guo ML, et al. HIV TAT-mediated microglial senescence: Role of SIRT3-dependent mitochondrial oxidative stress. Redox Biol. 2021;40:101843.
Chen NC, Partridge AT, Tuzer F, Cohen J, Nacarelli T, Navas-Martín S, et al. Induction of a senescence-like phenotype in cultured human fetal microglia during HIV-1 infection. J Gerontol A Biol Sci Med Sci. 2018;73:1187–96.
Miwa S, Kashyap S, Chini E, von Zglinicki T. Mitochondrial dysfunction in cell senescence and aging. J Clin Invest. 2022;132:e158447. https://doi.org/10.1172/JCI158447.
Shi Y, Li H, Chu D, Lin W, Wang X, Wu Y, et al. Rescuing nucleus pulposus cells from senescence via dual-functional greigite nanozyme to alleviate intervertebral disc degeneration. Adv Sci. 2023;10:e2300988. https://doi.org/10.1002/advs.202300988.
Thangaraj A, Periyasamy P, Liao K, Bendi VS, Callen S, Pendyala G, et al. HIV-1 TAT-mediated microglial activation: role of mitochondrial dysfunction and defective mitophagy. Autophagy. 2018;14:1596–619.
Liu M, Xuan A, Zheng L, Li D, Chen C, Liu H, et al. Novel coumarin derivative SZC-6 as an allosteric activator of SIRT3 alleviates diabetic kidney disease via the SIRT3-Foxo3a signaling axis. Free Radic Biol Med. 2025;240:29–45. https://doi.org/10.1016/j.freeradbiomed.2025.08.019.
Qiu J, Liu D, Li P, Zhou L, Zhou L, Liu X, et al. NADPH oxidase mediates oxidative stress and ventricular remodeling through SIRT3/FOXO3a pathway in diabetic mice. Antioxidants. 2022;11:1745 https://doi.org/10.3390/antiox11091745.
Yoon H, Park SG, Kim HJ, Shin HR, Kim KT, Cho YD, et al. Nicotinamide enhances osteoblast differentiation through activation of the mitochondrial antioxidant defense system. Exp Mol Med. 2023;55:1531–43.
Wang Y, Wu S, Li Q, Sun H, Wang H. Pharmacological inhibition of ferroptosis as a therapeutic target for neurodegenerative diseases and strokes. Adv Sci. 2023;10:e2300325.
Chen X, Li J, Kang R, Klionsky DJ, Tang D. Ferroptosis: machinery and regulation. Autophagy. 2021;17:2054–81.
Fang Y, Chen X, Tan Q, Zhou H, Xu J, Gu Q. Inhibiting ferroptosis through disrupting the NCOA4-FTH1 interaction: a new mechanism of action. ACS Cent Sci. 2021;7:980–9.
Xu Y, Jia B, Li J, Li Q, Luo C. The interplay between ferroptosis and neuroinflammation in central neurological disorders. Antioxidants. 2024;13:395.
Wen Q, Liu J, Kang R, Zhou B, Tang D. The release and activity of HMGB1 in ferroptosis. Biochem Biophys Res Commun. 2019;510:278–83.
Chen L, Hambright WS, Na R, Ran Q. Ablation of the ferroptosis inhibitor glutathione peroxidase 4 in neurons results in rapid motor neuron degeneration and paralysis. J Biol Chem. 2015;290:28097–106.
Reinert A, Morawski M, Seeger J, Arendt T, Reinert T. Iron concentrations in neurons and glial cells with estimates on ferritin concentrations. BMC Neurosci. 2019;20:25.
Sheng WS, Hu S, Hegg CC, Thayer SA, Peterson PK. Activation of human microglial cells by HIV-1 gp41 and Tat proteins. Clin Immunol. 2000;96:243–51.
Lin S, Cheng H, Yang G, Wang C, Leung CK, Zhang S, et al. NRF2 Antagonizes HIV-1 Tat and Methamphetamine-Induced BV2 Cell Ferroptosis by Regulating SLC7A11. Neurotox Res. 2023;41:398–407.
Li J, Yang D, Li Z, Zhao M, Wang D, Sun Z, et al. PINK1/Parkin-mediated mitophagy in neurodegenerative diseases. Ageing Res Rev. 2023;84:101817.
Fang EF, Hou Y, Palikaras K, Adriaanse BA, Kerr JS, Yang B, et al. Mitophagy inhibits amyloid-β and tau pathology and reverses cognitive deficits in models of Alzheimer’s disease. Nat Neurosci. 2019;22:401–12.
Sun K, Jing X, Guo J, Yao X, Guo F. Mitophagy in degenerative joint diseases. Autophagy. 2021;7:2082–92.
Cheng J, Liao Y, Dong Y, Hu H, Yang N, Kong X, et al. Microglial autophagy defect causes parkinson disease-like symptoms by accelerating inflammasome activation in mice. Autophagy. 2020;16:2193–205.
Yang G, Li J, Leung CK, Shen B, Wang C, Xu Y, et al. Methamphetamine and HIV-1 Tat proteins synergistically induce microglial autophagy via activation of the Nrf2/NQO1/HO-1 signal pathway. Neuropharmacology. 2022;220:109256.
Singh S, Thangaraj A, Chivero ET, Guo ML, Periyasamy P, Buch S. Role of Dysregulated Autophagy in HIV Tat, cocaine, and cART mediated NLRP3 Activation in Microglia. J Neuroimmune Pharmacol. 2023;18:327–47.
Quick JD, Silva C, Wong JH, Lim KL, Reynolds R, Barron AM, et al. Lysosomal acidification dysfunction in microglia: an emerging pathogenic mechanism of neuroinflammation and neurodegeneration. J Neuroinflammation. 2023;20:185.
Wu AG, Zhou XG, Qiao G, Yu L, Tang Y, Yan L, et al. Targeting microglial autophagic degradation in NLRP3 inflammasome-mediated neurodegenerative diseases. Ageing Res Rev. 2021;65:101202.
Fu J, Wu H. Structural mechanisms of NLRP3 inflammasome assembly and activation. Annu Rev Immunol. 2023;41:301–16.
Lee GS, Subramanian N, Kim AI, Aksentijevich I, Goldbach-Mansky R, Sacks DB, et al. The calcium-sensing receptor regulates the NLRP3 inflammasome through Ca2+ and cAMP. Nature. 2012;492:123–7.
Mahdi F, Shariat-Madar Z, Paris JJ. HIV-1 tat impairment of mitochondrial respiration via complexes I and II can be ameliorated by allopregnanolone in opioid-exposed or opioid-naïve cells and mice. Antioxidants. 2025;14:420.
Zou S, Fuss B, Fitting S, Hahn YK, Hauser KF, Knapp PE. Oligodendrocytes Are Targets of HIV-1 Tat: NMDA and AMPA receptor-mediated effects on survival and development. J Neurosci. 2015;35:11384–98.
Fitting S, Knapp PE, Zou S, Marks WD, Bowers MS, Akbarali HI, et al. Interactive HIV-1 Tat and morphine-induced synaptodendritic injury is triggered through focal disruptions in Na+ influx, mitochondrial instability, and Ca²⁺ overload. J Neurosci. 2014;34:12850–64.
Krogh KA, Wydeven N, Wickman K, Thayer SA. HIV-1 protein Tat produces biphasic changes in NMDA-evoked increases in intracellular Ca2+ concentration via activation of Src kinase and nitric oxide signaling pathways. J Neurochem. 2014;130:642–56.
Mayne M, Holden CP, Nath A, Geiger JD. Release of calcium from inositol 1,4,5-trisphosphate receptor-regulated stores by HIV-1 Tat regulates TNF-alpha production in human macrophages. J Immunol. 2000;164:6538–42.
Haughey NJ, Holden CP, Nath A, Geiger JD. Involvement of inositol 1,4,5-trisphosphate-regulated stores of intracellular calcium in calcium dysregulation and neuron cell death caused by HIV-1 protein tat. J Neurochem. 1999;73:1363–74.
He Y, Chang Y, Peng Y, Zhu J, Liu K, Chen J, et al. Glibenclamide directly prevents neuroinflammation by targeting SUR1-TRPM4-Mediated NLRP3 inflammasome activation in microglia. Mol Neurobiol. 2022;59:6590–607.
Borrajo A, Spuch C, Penedo MA, Olivares JM, Agís-Balboa RC. Important role of microglia in HIV-1 associated neurocognitive disorders and the molecular pathways implicated in its pathogenesis. Ann Med. 2021;53:43–69.
Chivero ET, Guo ML, Periyasamy P, Liao K, Callen SE, Buch S. HIV-1 Tat primes and activates microglial NLRP3 inflammasome-mediated neuroinflammation. J Neurosci. 2017;37:3599–609.
Cheng Y, Jung J, Guo L, Shuboni-Mulligan DD, Chen JF, Hu W, et al. HIV-TAT dysregulates microglial lipid metabolism through SREBP2/miR-124 axis: Implication of lipid droplet accumulation microglia in NeuroHIV. Brain Behav Immun. 2025;123:108–22.
Kim SK, Park KY, Choe JY. Toll-Like Receptor 9 Is Involved in NLRP3 inflammasome activation and IL-1β production through monosodium urate-induced mitochondrial DNA. Inflammation. 2020;43:2301–11.
Coll RC, Schroder K, Pelegrín P. NLRP3 and pyroptosis blockers for treating inflammatory diseases. Trends Pharmacol Sci. 2022;43:653–68.
Wei X, Xie F, Zhou X, Wu Y, Yan H, Liu T, et al. Role of pyroptosis in inflammation and cancer. Cell Mol Immunol. 2022;19:971–92.
Benelli R, Barbero A, Ferrini S, Scapini P, Cassatella M, Bussolino F, et al. Human immunodeficiency virus transactivator protein (Tat) stimulates chemotaxis, calcium mobilization, and activation of human polymorphonuclear leukocytes: implications for Tat-mediated pathogenesis. J Infect Dis. 2000;182:1643.
Yu H, Lin L, Zhang Z, Zhang H, Hu H. Targeting NF-κB pathway for the therapy of diseases: mechanism and clinical study. Signal Transduct Target Ther. 2020;5:209.
Park JH, Seo YH, Jang JH, Jeong CH, Lee S, Park B. Asiatic acid attenuates methamphetamine-induced neuroinflammation and neurotoxicity through blocking of NF-kB/STAT3/ERK and mitochondria-mediated apoptosis pathway. J Neuroinflammation. 2017;14:240.
Berry CT, May MJ, Freedman BD. STIM- and Orai-mediated calcium entry controls NF-κB activity and function in lymphocytes. Cell Calcium. 2018;74:131–43.
Nicolini A, Ajmone-Cat MA, Bernardo A, Levi G, Minghetti L. Human immunodeficiency virus type-1 Tat protein induces nuclear factor (NF)-kappaB activation and oxidative stress in microglial cultures by independent mechanisms. J Neurochem. 2001;79:713–6.
Periyasamy P, Thangaraj A, Bendi VS, Buch S. HIV-1 Tat-mediated microglial inflammation involves a novel miRNA-34a-NLRC5-NFκB signaling axis. Brain Behav Immun. 2019;80:227–37.
Periyasamy P, Thangaraj A, Guo ML, Hu G, Callen S, Buch S. Epigenetic Promoter DNA Methylation of miR-124 Promotes HIV-1 Tat-Mediated microglial activation via MECP2-STAT3 Axis. J Neurosci. 2018;38:5367–83.
Periyasamy P, Thangaraj A, Kannan M, Oladapo A, Buch S. The Epigenetic Role of miR-124 in HIV-1 tat- and cocaine-mediated microglial activation. Int J Mol Sci. 2022;23:15017.
Flora G, Pu H, Hennig B, Toborek M. Cyclooxygenase-2 is involved in HIV-1 Tat-induced inflammatory responses in the brain. Neuromolecular Med. 2006;8:337–52.
Zhou X, Zhou S, Tao J, Gao Y, Meng G, Cao D, et al. HIV-1 Tat drives the Fabp4/NF-κB feedback loop in microglia to mediate inflammatory response and neuronal apoptosis. J Neurovirol. 2022;28:483–96.
D’Aversa TG, Yu KO, Berman JW. Expression of chemokines by human fetal microglia after treatment with the human immunodeficiency virus type 1 protein Tat. J Neurovirol. 2004;10:86–97.
Lee YJ, Yeo IJ, Choi DY, Yun J, Son DJ, Han SB, et al. Amyloidogenic, neuroinflammatory and memory dysfunction effects of HIV-1 gp120. Arch Pharm Res. 2021;44:689–701.
Louboutin JP, Reyes BA, Agrawal L, Van Bockstaele EJ, Strayer DS. HIV-1 gp120-induced neuroinflammation: relationship to neuron loss and protection by rSV40-delivered antioxidant enzymes. Exp Neurol. 2010;221:231–45.
Dutta D, Liu J, Xu E, Xiong H. Methamphetamine Enhancement of HIV-1 gp120-Mediated NLRP3 inflammasome activation and resultant proinflammatory responses in rat microglial cultures. Int J Mol Sci. 2024;25:3588.
Xu C, Liu J, Chen L, Liang S, Fujii N, Tamamura H, et al. HIV-1 gp120 enhances outward potassium current via CXCR4 and cAMP-dependent protein kinase a signaling in cultured rat microglia. Glia. 2017;59:997–1007.
Liu J, Xu E, Tu G, Liu H, Luo J, Xiong H. Methamphetamine potentiates HIV-1gp120- induced microglial neurotoxic activity by enhancing microglial outward K+ current. Mol Cell Neurosci. 2017;82:167–75.
Liu J, Xu C, Chen L, Xu P, Xiong H. Involvement of Kv1.3 and p38 MAPK signaling in HIV-1 glycoprotein 120-induced microglia neurotoxicity. Cell Death Dis. 2012;3:e254.
Leu WJ, Chu JC, Hsu JL, Du CM, Jiang YH, Hsu LC, et al. Chalcones display Anti-NLRP3 inflammasome activity in macrophages through inhibition of both priming and activation steps-structure-activity- relationship and mechanism studies. Molecules. 2020;25:5960.
Mangan MSJ, Olhava EJ, Roush WR, Seidel HM, Glick GD, Latz. E. Targeting the NLRP3 inflammasome in inflammatory diseases. Nat Rev Drug Discov. 2018;17:688.
Walsh JG, Reinke SN, Mamik MK, McKenzie BA, Maingat F, Branton WG, et al. Rapid inflammasome activation in microglia contributes to brain disease in HIV/AIDS. Retrovirology. 2014;11:35.
Chen Q, Wu H, Qin S, Liu C, Chen Y, Yang Y, et al. The P2X7 Receptor Involved in gp120-Induced Cell Injury in BV2 Microglia. Inflammation. 2016;39:1814–26.
Ferrari D, Wesselborg S, Bauer MK, Schulze-Osthoff K. Extracellular ATP activates transcription factor NF-kappaB through the P2Z purinoreceptor by selectively targeting NF-kappaB p65. J Cell Biol. 1997;139:1635–43.
Ibrahim SSA, Huttunen KM. Orchestrated modulation of rheumatoid arthritis via crosstalking intracellular signaling pathways. Inflammopharmacology. 2021;29:965–74.
Zeng D, Yao P, Zhao H. P2X7, a critical regulator and potential target for bone and joint diseases. J Cell Physiol. 2019;234:2095–103.
Sun Y, Zhang C, Lei T, Lin F, Huang J, Hu Y, et al. HIV1 gp120 activates microglia via TLR2-nf-κb signaling to up-regulate inflammatory cytokine expression and induce neuropathic pain. Neuropharmacology. 2024;260:110136.
Wang H, Zuo Q, Li X, Liu Y, Gan L, Wang L, et al. p62 Binding to Protein Kinase C Regulates HIV-1 gp120 V3 Loop Induced Microglial Inflammation. Inflammation. 2025;48:2772–82.
James T, Nonnemacher MR, Wigdahl B, Krebs FC. Defining the roles for Vpr in HIV-1-associated neuropathogenesis. J Neurovirol. 2016;22:403–15.
Mamik MK, Hui E, Branton WG, McKenzie BA, Chisholm J, Cohen EA, et al. HIV-1 Viral Protein R Activates NLRP3 Inflammasome in Microglia: implications for HIV-1 associated neuroinflammation. J Neuroimmune Pharmacol. 2017;12:233–48.
Acharjee S, Branton WG, Vivithanaporn P, Maingat F, Paul AM, Dickie P, et al. HIV-1 Nef expression in microglia disrupts dopaminergic and immune functions with associated mania-like behaviors. Brain Behav Immun. 2014;40:74–84.
Rom I, Deshmane SL, Mukerjee R, Khalili K, Amini S, Sawaya BE. HIV-1 Vpr deregulates calcium secretion in neural cells. Brain Res. 2009;1275:81–6.
Deshmane SL, Mukerjee R, Fan S, Del Valle L, Michiels C, Sweet T, et al. Activation of the oxidative stress pathway by HIV-1 Vpr leads to induction of hypoxia-inducible factor 1alpha expression. J Biol Chem. 2009;284:11364–73.
Vilhardt F, Plastre O, Sawada M, Suzuki K, Wiznerowicz M, Kiyokawa E, et al. The HIV-1 Nef protein and phagocyte NADPH oxidase activation. J Biol Chem. 2002;277:42136–43.
Schenck JK, Clarkson-Paredes C, Pushkarsky T, Wang Y, Miller RH, Bukrinsky MI. Nef mediates neuroimmune response, myelin impairment, and neuronal injury in EcoHIV-infected mice. Life Sci Alliance. 2024;8:e202402879.
Raymond AD, Diaz P, Chevelon S, Agudelo M, Yndart-Arias A, Ding H, et al. Microglia-derived HIV Nef+ exosome impairment of the blood-brain barrier is treatable by nanomedicine-based delivery of Nef peptides. J Neurovirol. 2016;22:129–39.
Liu X, Kumar A. Differential signaling mechanism for HIV-1 Nef-mediated production of IL-6 and IL-8 in human astrocytes. Sci Rep. 2015;5:9867.
Si Q, Kim MO, Zhao ML, Landau NR, Goldstein H, Lee S. Vpr- and Nef-dependent induction of RANTES/CCL5 in microglial cells. Virology. 2002;301:342–53.
Song T, Wei C, Zheng Z, Xu Y, Cheng X, Yuan Y, et al. c-Abl tyrosine kinase interacts with MAVS and regulates innate immune response. FEBS Lett. 2010;584:33–8.
Teodorof-Diedrich C, Spector SA. Human Immunodeficiency Virus Type 1 gp120 and tat induce mitochondrial fragmentation and incomplete mitophagy in human neurons. J Virol. 2018;92:e00993-18.
Jalloh S, Hughes IK, Akiyama H, Gojanovich AD, Quiñones-Molina AA, Yang M, et al. Expression of intron-containing HIV-1 RNA induces NLRP1 inflammasome activation in myeloid cells. PLoS Biol. 2025;23:e3003320.
Mishra R, Chhatbar C, Singh SK. HIV-1 Tat C-mediated regulation of tumor necrosis factor receptor-associated factor-3 by microRNA 32 in human microglia. J Neuroinflammation. 2012;9:131.
Jadhav VS, Krause KH, Singh SK. HIV-1 Tat C modulates NOX2 and NOX4 expressions through miR-17 in a human microglial cell line. J Neurochem. 2014;131:803–15.
Periyasamy P, Singh S, Oladapo A, Kannan M, Buch S. Role of lncRNA Xist-miR-124-CCL2 axis in HIV Tat-mediated microglial activation and neuroinflammation. Front Immunol. 2025;16:1558842.
Schenck JK, Karl MT, Clarkson-Paredes C, Bastin A, Pushkarsky T, Brichacek B, et al. Extracellular vesicles produced by HIV-1 Nef-expressing cells induce myelin impairment and oligodendrocyte damage in the mouse central nervous system. J Neuroinflammation. 2024;21:127.
Pužar Dominkuš P, Ferdin J, Plemenitaš A, Peterlin BM, Lenassi M. Nef is secreted in exosomes from Nef.GFP-expressing and HIV-1-infected human astrocytes. J Neurovirol. 2017;23:713–24.
Rahimian P, He JJ. Exosome-associated release, uptake, and neurotoxicity of HIV-1 Tat protein. J Neurovirol. 2016;22:774–88.
Sami Saribas A, Cicalese S, Ahooyi TM, Khalili K, Amini S, Sariyer IK. HIV-1 Nef is released in extracellular vesicles derived from astrocytes: evidence for Nef-mediated neurotoxicity. Cell Death Dis. 2017;8:e2542.
Hu G, Yao H, Chaudhuri AD, Duan M, Yelamanchili SV, Wen H, et al. Exosome-mediated shuttling of microRNA-29 regulates HIV Tat and morphine-mediated neuronal dysfunction. Cell Death Dis. 2012;3:e381.
Hu G, Niu F, Liao K, Periyasamy P, Sil S, Liu J, et al. HIV-1 Tat-Induced astrocytic extracellular vesicle miR-7 impairs synaptic architecture. J Neuroimmune Pharmacol. 2020;15:538–53.
Johnston JB, Zhang K, Silva C, Shalinsky DR, Conant K, Ni W, et al. HIV-1 Tat neurotoxicity is prevented by matrix metalloproteinase inhibitors. Ann Neurol. 2001;49:230–41.
Datta G, Miller NM, Chen X. 17α-Estradiol Protects against HIV-1 Tat-Induced endolysosome dysfunction and dendritic impairments in neurons. Cells. 2023;12:813.
Hui L, Chen X, Haughey NJ, Geiger JD. Role of endolysosomes in HIV-1 Tat-induced neurotoxicity. ASN Neuro. 2012;4:243–52.
Arjona SP, Allen CNS, Santerre M, Gross S, Soboloff J, Booze R, et al. Disruption of Mitochondrial-associated ER membranes by HIV-1 tat protein contributes to premature brain aging. CNS Neurosci Ther. 2023;29:365–77.
Haughey NJ, Nath A, Mattson MP, Slevin JT, Geiger JD. HIV-1 Tat through phosphorylation of NMDA receptors potentiates glutamate excitotoxicity. J Neurochem. 2001;78:457–67.
Weber E, Morgan EE, Iudicello JE, Blackstone K, Grant I, Ellis RJ, et al. Substance use is a risk factor for neurocognitive deficits and neuropsychiatric distress in acute and early HIV infection. J Neurovirol. 2013;19:65–74.
Kanmogne GD, Schall K, Leibhart J, Knipe B, Gendelman HE, Persidsky Y. HIV-1 gp120 compromises blood-brain barrier integrity and enhances monocyte migration across blood-brain barrier: implication for viral neuropathogenesis. J Cereb Blood Flow Metab. 2007;27:123–34.
Huang J, Zhang R, Wang S, Zhang D, Leung CK, Yang G, et al. Methamphetamine and HIV-Tat protein synergistically induce oxidative stress and blood-brain barrier damage via transient receptor potential melastatin 2 channel. Front Pharmacol. 2021;12:619436.
Kaul M, Lipton SA. Chemokines and activated macrophages in HIV gp120-induced neuronal apoptosis. Proc Natl Acad Sci USA. 1999;96:8212–6.
El-Hage N, Wu G, Wang J, Ambati J, Knapp PE, Reed JL, et al. HIV-1 Tat and opiate-induced changes in astrocytes promote chemotaxis of microglia through the expression of MCP-1 and alternative chemokines. Glia. 2006;53:132–46.
Persidsky Y, Ghorpade A, Rasmussen J, Limoges J, Liu XJ, Stins M, et al. Microglial and astrocyte chemokines regulate monocyte migration through the blood-brain barrier in human immunodeficiency virus-1 encephalitis. Am J Pathol. 1999;155:1599–611.
Pillai PP, Kannan M, Sil S, Singh S, Thangaraj A, Chivero ET, et al. Involvement of lncRNA TUG1 in HIV-1 Tat-Induced Astrocyte Senescence. Int J Mol Sci. 2023;24:4330.
Shah A, Kumar S, Simon SD, Singh DP, Kumar A. HIV gp120- and methamphetamine-mediated oxidative stress induces astrocyte apoptosis via cytochrome P450 2E1. Cell Death Dis. 2013;4:e850.
Zou S, Balinang JM, Paris JJ, Hauser KF, Fuss B, Knapp PE. Effects of HIV-1 Tat on oligodendrocyte viability are mediated by CaMKIIβ-GSK3β interactions. J Neurochem. 2019;149:98–110.
Liu H, Liu J, Xu E, Tu G, Guo M, Liang S, et al. Human immunodeficiency virus protein Tat induces oligodendrocyte injury by enhancing outward K+ current conducted by KV1.3. Neurobiol Dis. 2017;97:1–10.
Scutari R, Alteri C, Perno CF, Svicher V, Aquaro S. The Role of HIV Infection in Neurologic Injury. Brain Sci. 2017;7:38.
Sreeram S, Ye F, Garcia-Mesa Y, Nguyen K, El Sayed A, Leskov K, et al. The potential role of HIV-1 latency in promoting neuroinflammation and HIV-1-associated neurocognitive disorder. Trends Immunol. 2022;43:630–9.
Maezawa I, Jin LW. Rett syndrome microglia damage dendrites and synapses by the elevated release of glutamate. J Neurosci. 2010;30:5346–56.
Suvieri C, Mondanelli G, Orabona C, Pallotta MT, Panfili E, Rossini S, et al. Sensing of an HIV-1-Derived Single-Stranded RNA-Oligonucleotide induces arginase 1-Mediated tolerance. Cells. 2024;13:1088.
Campbell GR, To RK, Hanna J, Spector SA. SARS-CoV-2, SARS-CoV-1, and HIV-1 derived ssRNA sequences activate the NLRP3 inflammasome in human macrophages through a non-classical pathway. iScience. 2021;24:102295.
Alter G, Suscovich TJ, Teigen N, Meier A, Streeck H, Brander C, et al. Single-stranded RNA derived from HIV-1 serves as a potent activator of NK cells. J Immunol. 2007;178:7658–66.
Chang J, Lindsay RJ, Kulkarni S, Lifson JD, Carrington M, Altfeld M. Polymorphisms in interferon regulatory factor 7 reduce interferon-α responses of plasmacytoid dendritic cells to HIV-1. AIDS. 2011;25:715–7.
Martinez-Meza S, Premeaux TA, Cirigliano SM, Friday CM, Michael S, Mediouni S, et al. Antiretroviral drug therapy does not reduce neuroinflammation in an HIV-1 infection brain organoid model. J Neuroinflammation. 2025;22:66.
Donadoni M, Cakir S, Bellizzi A, Swingler M, Sariyer IK. Modeling HIV-1 infection and NeuroHIV in hiPSCs-derived cerebral organoid cultures. J Neurovirol. 2024;30:362–79.
Maldini CR, Claiborne DT, Okawa K, Chen T, Dopkin DL, Shan X, et al. Dual CD4-based CAR T cells with distinct costimulatory domains mitigate HIV pathogenesis in vivo. Nat Med. 2020;26:1776–87.
Niessl J, Baxter AE, Mendoza P, Jankovic M, Cohen YZ, Butler AL, et al. Combination anti-HIV-1 antibody therapy is associated with increased virus-specific T cell immunity. Nat Med. 2020;26:222–7.
Fromentin R, DaFonseca S, Costiniuk CT, El-Far M, Procopio FA, Hecht FM, et al. PD-1 blockade potentiates HIV latency reversal ex vivo in CD4+ T cells from ART-suppressed individuals. Nat Commun. 2019;10:814.
Herskovitz J, Hasan M, Patel M, Blomberg WR, Cohen JD, Machhi J, et al. CRISPR-Cas9 Mediated Exonic Disruption for HIV-1 Elimination. EBioMedicine. 2021;73:103678.
Ye F, Alvarez-Carbonell D, Nguyen K, Leskov K, Garcia-Mesa Y, Sreeram S, et al. Recruitment of the CoREST transcription repressor complexes by Nerve Growth factor IB-like receptor (Nurr1/NR4A2) mediates silencing of HIV in microglial cells. PLoS Pathog. 2022;18:e1010110.
Alvarez-Carbonell D, Ye F, Ramanath N, Dobrowolski C, Karn J. The glucocorticoid receptor is a critical regulator of HIV latency in human microglial cells. J Neuroimmune Pharmacol. 2019;14:94–109.
Carrillo-de Sauvage MÁ, Maatouk L, Arnoux I, Pasco M, Sanz Diez A, Delahaye M, et al. Potent and multiple regulatory actions of microglial glucocorticoid receptors during CNS inflammation. Cell Death Differ. 2013;20:1546–57.
Nguyen K, Dobrowolski C, Shukla M, Cho WK, Luttge B, Karn J. Inhibition of the H3K27 demethylase UTX enhances the epigenetic silencing of HIV proviruses and induces HIV-1 DNA hypermethylation but fails to permanently block HIV reactivation. PLoS Pathog. 2021;17:e1010014.
Schwartz C, Bouchat S, Marban C, Gautier V, Van Lint C, Rohr O, et al. On the way to find a cure: Purging latent HIV-1 reservoirs. Biochem Pharmacol. 2017;146:10–22.
Chen J, Zhou T, Zhang Y, Luo S, Chen H, Chen D, et al. The reservoir of latent HIV. Front Cell Infect Microbiol. 2022;12:945956.
Kaushik A, Yndart A, Atluri V, Tiwari S, Tomitaka A, Gupta P, et al. Magnetically guided non-invasive CRISPR-Cas9/gRNA delivery across blood-brain barrier to eradicate latent HIV-1 infection. Sci Rep. 2019;9:3928.
Xu W, Huang Y, Zhou R. NLRP3 inflammasome in neuroinflammation and central nervous system diseases. Cell Mol Immunol. 2025;22:341–55.
Naeem A, Prakash R, Kumari N, Ali Khan M, Quaiyoom Khan A, Uddin S, et al. MCC950 reduces autophagy and improves cognitive function by inhibiting NLRP3-dependent neuroinflammation in a rat model of Alzheimer’s disease. Brain Behav Immun. 2024;116:70–84.
Lonnemann N, Hosseini S, Marchetti C, Skouras DB, Stefanoni D, D’Alessandro A, et al. The NLRP3 inflammasome inhibitor OLT1177 rescues cognitive impairment in a mouse model of Alzheimer’s disease. Proc Natl Acad Sci USA. 2020;117:32145–54.
Ventyx Biosciences, Inc. Ventyx Biosciences announces positive topline phase 1 data for its peripheral NLRP3 inhibitor VTX2735. 2022. https://ir.ventyxbio.com/news-releases/news-release-details/ventyx-biosciences-announces-positive-topline-phase-1-data-its.
Coll RC, Schröder K. Inflammasome components as new therapeutic targets in inflammatory disease. Nat Rev Immunol. 2025;25:22–41.
NodThera, I NodThera announces positive phase 1 study readouts for the NLRP3 inflammasome inhibitors NT-0796 and NT-0249. 2022. https://www.nodthera.com/news/nodthera-announces-positive-phase-1-study-readouts-for-the-nlrp3-inflammasome-inhibitors-nt-0796-and-nt-0249/.
Omeragic A, Kayode O, Hoque MT, Bendayan R. Potential pharmacological approaches for the treatment of HIV-1 associated neurocognitive disorders. Fluids Barriers CNS. 2020;17:42.
Ashraf T, Jiang W, Hoque MT, Henderson J, Wu C, Bendayan R. Role of anti-inflammatory compounds in human immunodeficiency virus-1 glycoprotein120- mediated brain inflammation. J Neuroinflammation. 2014;11:91.
Sacktor N, Miyahara S, Deng L, Evans S, Schifitto G, Cohen BA, et al. Minocycline treatment for HIV-associated cognitive impairment: results from a randomized trial. Neurology. 2011;77:1135–42.
Sun B, Wang X, Ji Z, Li R, Xia T. NLRP3 inflammasome activation induced by engineered nanomaterials. Small. 2013b;9:1595–607.
Lahooti B, Chhibber T, Bagchi S, Varahachalam SP, Jayant RD. Therapeutic role of inflammasome inhibitors in neurodegenerative disorders. Brain Behav Immun. 2021;91:771–83.
Acknowledgements
This work was supported by the Science and Technology Innovation Committee of Shenzhen Municipality (No. JCYJ20220531102202005), the Natural Science Foundation of Guangdong Province (No. 2024A1515012118).
Author information
Authors and Affiliations
Contributions
Jiaye Liu designed the work and reviewed and edited the manuscript; Jinfang Zhao participated in the original draft preparation; Fan Bu, Hui Wu and Jiayi He helped to review the manuscript. All authors have read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
All authors claim that there are no conficts of interest. Figures were created with BioRender software (https://biorender.com/).
Consent to publish
All authors consented to publication.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Zhao, J., Bu, F., Wu, H. et al. Neuroinflammation and NeuroHIV: understanding the role of HIV-1 related factors in microglial activation. Transl Psychiatry 16, 194 (2026). https://doi.org/10.1038/s41398-026-03941-7
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41398-026-03941-7