Abstract
To explore the effect of repetitive transcranial magnetic stimulation (rTMS) on influenza-induced sleep disorders, verify its adjuvant value, and reveal the “influenza-immune inflammation-abnormal brain function-sleep disorder” pathway, a retrospective cohort study enrolled 55 adults (18-40 years, no comorbidities) with acute influenza (2023-2025, First Affiliated Hospital of Air Force Medical University). They were divided into positive control group (PC, n = 20, conventional treatment) and rTMS group (n = 16, conventional + rTMS: 10 Hz, 1x/day, 20-30 mins, 3000 pulses, 100% motor threshold, 3 days; split into pre-treatment [BT-rTMS] and post-treatment [AT-rTMS] subgroups). Twenty healthy people served as negative controls group (NC). Assessments included polysomnography (PSG), Sleep Disorder-Visual Analog Scale (SD-VAS), functional near-infrared spectroscopy (fNIRS) for frontal oxygenated hemoglobin, and ELISA for 9 blood indicators. Two-sample Mendelian randomization (TSMR), ROC models, and t-tests (P < 0.05) were used. TSMR showed influenza correlated with insomnia (IVW:P = 0.047, 95% OR = 1.001-1.134) and 27 brain structural changes (frontal most affected, n = 10). Serum IgM (IVW:P = 0.048, 95% OR = 1.001-1.193) and neutrophils (IVW:P = 0.003, 95% OR = 1.018-1.089) mediated sleep disorders, linking to SD-VAS and frontal gray matter. Left inferior frontal gyrus and right orbital gyrus were key. BT-rTMS had abnormal sleep (duration/efficiency/stages) vs NC, with higher right orbitofrontal cortex (R-OFC) and left ventrolateral prefrontal cortex (L-VLPFC) oxygenated hemoglobin (P < 0.05). AT-rTMS improved sleep vs PC, with lower R-OFC/L-VLPFC oxygenated hemoglobin (P < 0.05). A 7-indicator ROC model had AUC = 0.8571. rTMS improves influenza-related sleep disorders short-term by inhibiting frontal overactivation (R-OFC, L-VLPFC) and regulating immunity (IgM,neutrophils), offering a safe, rapid new approach for acute influenza sleep management.
Similar content being viewed by others
Introduction
Influenza is an acute respiratory infectious disease caused by influenza A, B, and C viruses. The virus invades respiratory epithelial cells via hemagglutinin (HA) and neuraminidase (NA), triggering local and systemic inflammatory responses. Globally, influenza causes 3 to 5million severe cases and 290,000 to 650,000 respiratory-related deaths annually, with the Asia region (especially East and Southeast Asia) bearing the heaviest disease burden. As a populous country, China experiences a surge in outpatient and emergency visits during seasonal influenza epidemics, which continuously impacts the public health system,and is increasingly recognized as a trigger for acute sleep disorders—often persisting beyond the resolution of fever and respiratory symptoms [1,2,3].In addition to typical respiratory symptoms, influenza infection is often accompanied by systemic symptoms such as headache, myalgia, fatigue, and sleep disorders. These symptoms may persist for more than 2 weeks, severely impairing patients’ work efficiency and quality of life. The risks are particularly significant for occupational groups requiring high precision and concentration, such as healthcare workers, drivers, and engineers [4, 5].Current clinical management of influenza mainly relies on neuraminidase inhibitors (e.g., oseltamivir), symptomatic treatment, and supportive care. Although these measures can effectively shorten the disease course and reduce the risk of complications, they have limited efficacy in improving virus infection-related neuropsychiatric symptoms, especially sleep disorders. Guidelines from the World Health Organization (WHO) point out that existing treatment regimens lack targeted effects on influenza-associated complications, indicating promising prospects for exploring relevant adjuvant therapeutic approaches [6, 7].
In recent years, non-pharmacological interventions have attracted increasing attention in the management of complications of infectious diseases such as influenza infection, and neuromodulation techniques possess unique advantages due to their precision and safety [8]. As a depression treatment technology approved by the FDA, Repetitive Transcranial Magnetic Stimulation (rTMS) induces action potentials in cortical neurons through time-varying magnetic fields. It can regulate the functional connectivity of the default mode network and improve sleep disorders associated with various neurological diseases [9, 10], a recent systematic review supports the potential efficacy of rTMS for insomnia it also highlights critical gaps—including the lack of mechanistic insights into how rTMS modulates brain–immune interactions in specific insomnia subtypes, such as post-infectious cases [11]. Relevant studies have shown that rTMS can affect the sleep-wake cycle by regulating the neural circuit of the prefrontal cortex-thalamus-limbic system, providing a theoretical basis for the treatment of mental and sleep-related complications caused by infectious diseases such as influenza infection. Although rTMS has abundant evidence in the field of neuropsychiatric diseases, there is a knowledge gap in its application in complications of acute infectious diseases [12, 13]. Most existing literatures focus on chronic neurological diseases, with insufficient research on transient sleep disorders during the acute and recovery phases of viral infection. In particular, there is a lack of clinical evidence for rTMS-related intervention therapy, which limits the exertion of its potential value in the comprehensive management of influenza virus infection.
To further explore this area, our study adopted a retrospective cohort design to systematically analyze the intervention effect of rTMS on sleep disorder symptoms following influenza virus infection. Patients diagnosed with influenza virus infection at the First Affiliated Hospital of Air Force Medical University (PLA) between 2023 and 2025 were included. Clinical data of patients who received conventional treatment alone or conventional treatment combined with rTMS were screened and analyzed retrospectively. Key outcome measures included the Polysomnography (PSG), Sleep Disorder-Visual Analog Scale (SD-VAS), and functional near-infrared spectroscopy (fNIRS). These were used to assess improvements in sleep parameters within 72 h after intervention.Potential confounding factors such as underlying diseases, medication use, and viral subtypes were strictly controlled in the study. By applying neuromodulation technology to symptom management during the acute phase of influenza, this study aimed to verify the efficacy of rTMS as an adjuvant therapy for influenza-related sleep disorders and evaluate the feasibility of this intervention in real-world clinical settings. It is expected to provide new insights into the management of acute-phase complications of influenza virus infection and promote the application of neuromodulation technology in the field of acute infectious diseases.
Our study hypothesizes that rTMS can rapidly improve the sleep quality of influenza patients in the short term, which is of great significance for enhancing recovery efficiency, reducing losses in social productivity, and preventing adverse events related to daily life and occupation [14] (Fig. 1).
Influenza infection leads to multi-system dysfunction, including respiratory, hematological, and nervous system alterations. In this study, we integrated clinical data—including blood biomarkers, brain structural information from magnetic resonance imaging (MRI), polysomnography, Sleep Disorder-Visual Analog Scale (SD-VAS), and functional near-infrared spectroscopy (fNIRS)—to retrospectively evaluate the effect of rTMS on post-influenza sleep disorders. Two-sample Mendelian randomization analysis (MRA) was applied to GWAS-derived datasets to infer causal relationships between influenza exposure, brain structural changes (localized using MRI-based anatomical atlases), immune markers, and insomnia symptoms.
Methods
Criteria for screening and grouping of the study population
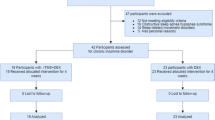
A total of 258 patients diagnosed with influenza virus infection were enrolled from the First Affiliated Hospital of Air Force Medical University of the Chinese People’s Liberation Army between 2023 and 2025, and based on previous clinical treatment pathways, patient case data that met the study needs were gradually screened; the patients were divided into the Positive Control Group (PC group, N = 20), in which patients received only conventional treatment regimens, the Repetitive Transcranial Magnetic Stimulation Group (rTMS group, N = 16), in which patients were given rTMS intervention therapy in addition to conventional treatment regimens, and the rTMS group was further divided into the BT-rTMS group (Before rTMS Therapy Group) and the AT-rTMS group (After rTMS Therapy Group) by time points according to the differences before and after treatment. The inclusion criteria were: aged 18–40 years, regardless of gender, no history of chronic diseases such as hypertension or diabetes, no history of major surgery or trauma, no other infectious diseases, no history of other neuropsychiatric diseases, first influenza virus infection in the past 6 months and in the acute phase of infection (Within 72 h after the onset of symptoms and upon confirmation through rapid antigen testing), no use of hypnotics, sedatives, antidepressants, antipsychotics, or other psychoactive medications known to affect sleep architecture within two weeks prior to enrollment, etc. The screening criteria were: patients were gradually screened and excluded according to the degree of completion of key outcome measures, such as whether they had completed MRI examination, fNIRS, sleep monitoring and rTMS treatment. The entire retrospective study analysis was conducted over a 5-day period, which was restricted to the acute phase of influenza virus infection. Meanwhile, 20 individuals who were not infected with influenza virus and met the above inclusion criteria were selected as the Negative Control Group (NC group, N = 20) for comparative analysis (Fig. 2, S1).Given the retrospective nature of this study, neither patients nor treating clinicians were blinded to rTMS administration. However, all outcome assessments—including polysomnography (PSG) scoring, functional near-infrared spectroscopy (fNIRS) data analysis, and ELISA measurements—were performed by independent investigators who were blinded to group allocation. All human samples were obtained with written informed consent from all participants.
All patients included in the study analysis were enrolled based on strict inclusion and exclusion criteria, and divided into the following groups according to the analysis needs:Negative Control Group(NC Group):Healthy control population without influenza infection;Positive Control Group (PC Group): Patients with influenza who received conventional treatment;Repetitive Transcranial Magnetic Stimulation Treatment Group (rTMS Group): Patients with influenza who received conventional treatment combined with rTMS intervention;Subgroups of the rTMS Treatment Group:BT-rTMS Group:Patients with influenza before receiving rTMS treatment;AT-rTMS Group: Patients with influenza after receiving rTMS treatment.
rTMS treatment protocol
Both patients in the rTMS group and PC group received conventional symptomatic treatment, and patients in the rTMS group additionally underwent rTMS therapy on this basis. Patients in the rTMS group received one session of rTMS treatment each day from day 1 to day 3. The treatment equipment used was the YRD Magnetic Stimulator (Model: YRD-CCY-II), produced by Wuhan Yiruidi Medical Equipment New Technology Co., Ltd. An 8-shaped coil was adopted to achieve double-sided and bidirectional stimulation. The rTMS treatment protocol employed low-frequency stimulation (10 Hz), administered once daily. Each single treatment session lasted 20–30 min, containing 3000 pulses per session, with the stimulation intensity set at 100% of the motor threshold (MT) (Fig. 3).
a.The Sleep Disorder-Visual Analog Scale (SD-VAS) was used to assess the severity of sleep disorders mainly characterized by insomnia symptoms in influenza patients, with a quantitative range of 0 to 10 points. A lower score indicates milder insomnia symptoms, while a higher score indicates more severe insomnia symptoms;b.Examination and treatment procedures for patients in the rTMS group, PC group, and NC group. Day 0: The day when the subjects began to undergo a series of examinations. Day 1: The subjects received the appropriate treatment measures. Day 4: The subjects underwent a series of follow-up examinations.
Polysomnography (PSG) and sleep disorder visual analog scale (SD-VAS)
All subjects underwent PSG and SD-VAS assessments. Both the PC group and the rTMS group completed one sleep monitoring session and one sleep disorder visual analog scale assessment on the 1st day and the 5th day, respectively. The PSG monitoring period is from 10 p.m. to 8 a.m. the next day. Polysomnographs were used for sleep monitoring, which could simultaneously monitor multiple parameters including electroencephalography(EEG), electrocardiography (ECG), respiratory airflow, snoring, blood oxygen saturation, and body position, enabling a more comprehensive and accurate evaluation of sleep status. Meanwhile, for the comparison between the rTMS group and the NC group, the SD-VAS adopted a “0-10 point continuous scale” to conduct a rapid subjective assessment of the patients’ recent sleep quality: scores leaning toward the left end (0 points) indicated “the worst sleep quality” (e.g., being awake all night, extreme fatigue), while scores leaning toward the right end (10 points) indicated “the best sleep quality” (e.g., falling asleep quickly, deep sleep, and feeling energetic after waking up).
Blood test and ELISA assay
All subjects underwent relevant blood tests including routine blood test, blood biochemistry, and immunoglobulin detection at the First Affiliated Hospital of Air Force Medical University of the Chinese People’s Liberation Army. According to the study requirements, focus was placed on blood indices potentially associated with influenza infection (serum IL-1β, IL-8, IgA, IgM, IgG, CRP, PTGSE2, blood red cell count, and neutrophil count), and ELISA experiments were conducted for these indices. The nine biomarkers were selected based on their established roles in influenza-related immune responses, prior links to neuroinflammation or sleep disruption, and routine inclusion in our hospital’s standard clinical panel for acute viral infections [15]. All ELISA kits were purchased from Elabscience Biotechnology Co.,Ltd, and the experiments were performed at the Animal Experiment Center of Air Force Medical University. Meanwhile, a comparative analysis was conducted on the relevant blood ELISA detection indices of the NC group(S2).
Mendelian randomization analyses (MRA)
In this study, all MRA employed Two-Sample Mendelian Randomization (TSMR) as the analytical tool. R-Studio (version 4.4.3) was used to analyze datasets related to influenza virus infection, blood cells, inflammatory factors, immunoglobulins, and brain structure, which were screened from Genome-Wide Association Study (GWAS) databases. Genetic variations were used as instrumental variables, with screening criteria set as follows: p = 1e-5, clump=TRUE, r2 = 0.001, and kb=10000. Inverse Variance Weighting (IVW) was adopted as the primary analytical method, while Weighted Median method and MR-Egger regression were used for sensitivity analysis to explore the potential associated factors and pathways of sleep disorders dominated by insomnia symptoms caused by influenza virus(S7).
Brain region visualization with BrainNet viewer in MATLAB
MATLAB is a core programming environment in the field of scientific computing. In neuroscience, it is commonly used for preprocessing of brain imaging data, calculation of functional connectivity, graph theory analysis, and other tasks. BrainNet Viewer is an open-source brain network visualization tool developed based on MATLAB, which focuses on the three-dimensional presentation of neuroimaging data and can intuitively display brain region functional connections, structural networks, etc. When drawing brain region maps, we combined the BrainNet Viewer program (version 1.7-Released-20191031-Matlab Code) with MATLAB software (version R2023a), selected the BrainMesh-Ch2 with Cerebellum module in the SurfTemplate tool as the basic brain region layout, sorted out the relevant data results of Mendelian randomization analysis, and then drew and visually displayed the brain region maps through BrainNet Viewer [16].
Brodmann brain regions
The distribution of Brodmann areas(BA) refers to 52 distinct regions divided by the German neurologist Korbinian Brodmann in the early 20th century through his research on the cytoarchitecture (cellular structure) of the cerebral cortex. These regions represent areas of the cerebral cortex with similar cellular structures and functions. In this study, the brain structural regions potentially associated with insomnia symptoms caused by influenza virus infection, which were identified and localized via Mendelian Randomization Analysis (MRA), were matched with the Brodmann area parcellation. Furthermore, the specific brain regions were accurately localized by integrating the analytical results of near-infrared brain functional imaging(S8).
Functional near-infrared spectroscopy (fNIRS)
The fNIRS instrument used in this study was the NIRSIT near-infrared multi-channel device manufactured by OBELAB Inc (South Korea), which is a device for measuring changes in cerebral blood oxygen saturation. It achieves this function by irradiating the cerebral cortex with near-infrared light beams at wavelengths of 780 nm and 850 nm. MATLAB software (version R2020a) combined with the Near-Infrared Brain Function Imaging Atlas Analysis Tool (NIRSIT Analysis Tool, version 3.6.1) was used to measure and visually display the relative concentration changes of oxygenated hemoglobin in the frontal lobe regions of patients. According to the different detection points of the relative concentration of oxygenated hemoglobin in the frontal lobe regions in the near-infrared brain function imaging atlas, the detection was divided into 48 channels, corresponding to a total of 8 frontal lobe regions in the left and right hemispheres. The results were displayed as 3D brain maps and 48-channel maps, which could intuitively show the differences in the relative concentration changes of oxygenated hemoglobin in brain regions among different patients. Given accumulating evidence that sleep-disordered breathing and insomnia are associated with dysregulation of prefrontal cortical activity—as demonstrated by neuroimaging studies including fNIRS and fMRI [17]—we focused our fNIRS measurements on the bilateral prefrontal cortex (channels covering BA9, BA10, and BA46) to capture task-free hemodynamic alterations relevant to sleep pathology.
Clinical prediction model
R-Studio (version 4.4.3) was used to construct a clinical risk prediction model, with the ELISA test results of the subjects’ blood samples (such as C-reactive protein, immunoglobulin M, neutrophil count, etc.) and influenza virus infection status as risk factors, and insomnia symptoms as the outcome (N = 75). The “nomogram” function in the “rms” package was utilized to create a nomogram, and the “pROC” package was employed to plot the ROC curve for the clinical risk prediction.
Statistical methods and graph plotting
In this study, all data difference analyses was performed using two-sample t-test in R-Studio (version 4.4.3) and Graphpad Prism (version 10.1.2). A P-value < 0.05 was considered statistically significant, indicating meaningful results. The “grid” and “forestploter” packages in R-Studio were used to draw forest plots, the “ggplot2” and “ggradar” packages to generate radar charts, the “pheatmap” and “viridis” packages to create heatmaps, and the “ggpubr” and “ggpmisc” packages to plot differential scatter plots for result visualization. Additionally, Graphpad Prism was used to draw bar graphs for the difference analysis of blood indicators and sleep monitoring data.
Results
We employed the MRA approach to explore the association between influenza virus infection and insomnia symptoms.Mendelian randomization estimates of the causal association are shown odds ratios (OR) with 95% confidence intervals; an OR > 1 indicates that genetically predicted influenza infection is associated with an increased risk of elevated IgM or neutrophil levels, while OR < 1 suggests a protective effect. Significance was defined as P < 0.05 (IVW method).Results of TSMR analysis on influenza virus infection and sleep disorder-related datasets from the GWAS database indicated a significant correlation between influenza virus infection (as an exposure factor) and insomnia symptoms (IVW method: P = 0.047, 95%OR = 1.001–1.134) (Fig. 4a).TSMR analysis of datasets related to influenza virus infection and brain region structure revealed that influenza virus infection (as an exposure factor) was also significantly associated with structural changes in 27 brain regions (IVW method: P < 0.05) (Fig. 4b). Among the brain region structural changes affected by influenza infection, the frontal lobe was the most affected (N = 10), followed by the temporal lobe (N = 6), parietal lobe (N = 5), cingulate gyrus (N = 5), insula (N = 1), and occipital lobe (N = 0) (Fig. 4c–d).Differential analysis was performed on blood test results of patients in the rTMS group, PC group, and NC group. Nine blood indicators related to influenza virus infection were selected. The results showed that seven blood indicators differed significantly between the infected populations (rTMS group and PC group) and the non-infected population (NC group): serum interleukin-1β (IL-1β), immunoglobulin A (IgA), immunoglobulin M (IgM), C-reactive protein (CRP), prostaglandin E synthase 2 (PTGES2) levels, neutrophil count, and red blood cell count (all P < 0.05) (Fig. 4e).Meanwhile, TSMR analysis was conducted between the nine blood indicators and brain region structure-related datasets. It was found that blood neutrophil count had the widest impact on brain region structure, with the frontal lobe being the most strongly affected brain region (N = 14) (Fig. 4f–g). Additionally, influenza infection showed a clear association with two of the above seven blood indicators: serum IgM level (IVW method: P = 0.048, 95% OR = 1.001–1.193) and blood neutrophil count (IVW method: P = 0.003, 95% OR = 1.018–1.089) (Fig. 4h).Results of TSMR analysis on serum IgM level, neutrophil count, and frontal lobe brain region structure showed that IgM and neutrophil count had a certain correlation with the structure of 15 brain regions (IVW method: P < 0.05) (Fig. 4i). Among them, neutrophil count exerted the greatest impact on frontal lobe brain region structure (N = 14) (Fig. 5b–c).Subsequently, TSMR was used to conduct an analysis with insomnia as the exposure factor and the structure of the aforementioned 15 associated brain regions as the outcome factors. Ultimately, the key frontal lobe brain regions associated with insomnia symptoms caused by influenza virus infection were identified as the left inferior frontal gyrus and the right orbital gyrus(Fig. 5a).Based on the obtained blood indicator data and sleep disorder score data, we developed a clinical prediction model nomogram for predicting insomnia symptoms caused by influenza virus infection. Using the 7 differential blood indicators as predictors, the resulting receiver operating characteristic (ROC) curve demonstrated good predictive performance (AUC = 0.8571, N = 75).
aThere was a certain correlation between influenza infection and insomnia (a symptom related to sleep disorders)(IVW method:P = 0.047,95%OR = 1.001-1.134);b. Influenza infection was correlated with 27 brain regions (IVW method:P < 0.05);c, d.Radar chart and brain map of the correlation between influenza infection and brain regions;e.Serum ELISA analysis showed that there were significant differences in the levels of some influenza-related blood factors between patients in the rTMS group+PC group and those in the NC group;f, g.Correlation between blood factors and brain regions;h. The results of MRA on the relationship between influenza infection and the levels of differential blood factors indicated that influenza infection had a certain correlation with serum IgM(IVW method: P = 0.048, 95%OR = 1.001-1.193) and blood neutrophil count (IVW method: P = 0.003, 95% OR = 1.018-1.089);i.The MRA results about the relationship between IgM, neutrophil count and frontal lobe brain regions showed that IgM and neutrophil count had a certain correlation with 15 brain regions (IVW method: P < 0.05.
a A certain correlation between insomnia and brain regions by MRA;b, c. Radar charts and brain maps of the correlation between serum IgM, neutrophil count and brain regions;d.Nomogram and ROC curve assessing the predictive value of baseline blood marker levels for insomnia onset in patients with acute influenza infection. The area under the curve (AUC) is presented with a 95% confidence interval estimated using DeLong’s method;e.Scatter plot of the correlation between influenza infection-related blood factors and SD-VAS(S6).
To ensure the rigor and accuracy of the analysis, we simultaneously performed a correlation analysis between 9 influenza virus infection-related blood indices and the SD-VAS data of the screened influenza virus-infected patients (N = 55)(S6). The results revealed that changes in four blood indices were significantly correlated with the patients’ SD-VAS scores, specifically: serum IL-1β level (P = 3.9e-4, R = 0.46, Fig. 5e1), IgM level (P = 9.9e-12, R = 0.77, Fig. 5e8), IgA level (P = 4.9e-9, R = 0.69, Fig. 5e6), and blood neutrophil count (P = 4.4e-7, R = 0.62, Fig. 5e3).Subsequently, a correlation analysis was conducted between changes in these 4 blood indices and changes in gray matter volume of brain regions. The results showed that in the influenza virus-infected population, serum IgM level (P = 6.5e-6, R = 0.63, Fig. 6a) and blood neutrophil count (P = 6.1e-7, R = 0.68, Fig. 7g) each exhibited a strong correlation with frontal lobe gray matter volume. In contrast, although IgA level (Fig. 7a-f) and IL-1β level (Fig. 6g-i) also showed certain correlations with the gray matter volume of the frontal lobe and other brain regions, the correlation strength was relatively weak(S3).
a-fScatter plots of the correlation between serum IgM levels and gray matter volume in various brain regions; g–l. Scatter plots of the correlation between serum IL-1b levels and gray matter volume in various brain regions.(S3).
a-fScatter plots of the correlation between serum IgA levels and gray matter volume in various brain regions; g-l. Scatter plots of the correlation between blood neutrophil levels and gray matter volume in various brain regions.(S3).
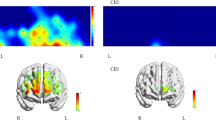
From the analysis of sleep monitoring data between patients in the BT-rTMS group and those in the NC group, there were significant differences in effective sleep duration, sleep efficiency, and the proportion of different sleep stages between the two groups (Fig. 8a, S4).The monitoring region of fNIRS focused on the frontal lobe of the brain, which was divided into a total of 8 regions across the left and right hemispheres. Based on the MRA results and Brodmann Area(BA) distribution (Fig. 8b), the key frontal lobe regions affected by influenza virus infection were identified as the right orbitofrontal cortex (R-OFC) and the left ventrolateral prefrontal cortex (L-VLPFC). Within these two regions, fNIRS measurements showed that the relative concentration changes of oxygenated hemoglobin in brain tissue were higher in the BT-rTMS group than in the NC group (relative concentration value > 0, Fig. 8c-d). This finding suggests that after influenza infection, the activity of brain neurons in the frontal cortex is higher than that in healthy individuals, and this elevated neuronal activity is one of the factors contributing to the occurrence of insomnia symptoms in patients.In addition, a cross-comparison of the relative concentrations of oxygenated hemoglobin in the frontal lobe between the two groups revealed that whether in the R-OFC or L-VLPFC, the relative concentration in the BT-rTMS group was significantly higher than that in the NC group (relative concentration value > 0, Fig.8e-f).
aAnalysis of sleep monitoring data between influenza patients before treatment(BT-rTMS) and the negative control group(NC)(S4);b. Division of frontal lobe functional regions in near-infrared brain functional imaging maps, including 4 regions in each of the left and right hemispheres, Dorsolateral Prefrontal Cortex(DLPFC) (BA9/BA46),Ventrolateral Prefrontal Cortex(VLPFC) (BA45/BA47),Orbitofrontal Cortex(OFC) (BA11/BA47),Frontopolar Prefrontal Cortex(FPC) (BA10),with a total of 48 analysis channels;c, d.Brain region channel maps showing the distribution of differences in relative oxygenated hemoglobin concentration in the frontal cortex between the BT-rTMS group and the NC group (N = 1);e.Overall cross-comparative analysis of differences in relative oxygenated hemoglobin concentration in the frontal cortex between the BT-rTMS group and the NC group (R-OFC region);f.Overall cross-comparative analysis of differences in relative oxygenated hemoglobin concentration in the frontal cortex between the BT-rTMS group and the NC group (L-VLPFC region).
After rTMS treatment, significant differences were observed in the sleep monitoring data between the AT-rTMS group and the PC group (which only received conventional treatment). These differences were mainly reflected in the improvement of sleep efficiency and the four sleep stages (Fig. 9a, S5).When comparing the differences in the relative concentrations of oxygenated hemoglobin across the 8 frontal lobe regions between the two groups of patients, a decreasing trend was observed in most regions, with more prominent decreases in the R-OFC and L-VLPFC (Fig. 9b).fNIRS measurements showed that the relative concentration changes of oxygenated hemoglobin in brain tissue were lower in the AT-rTMS group than in the PC group (relative concentration value < 0, Fig. 9c-d). This indicates that after rTMS treatment, the degree of decrease in the relative concentration of oxygenated hemoglobin in the frontal lobe of the AT-rTMS group was greater than that of the PC group (which only received conventional treatment), and this reduction played a certain role in promoting the improvement of insomnia symptoms in patients after influenza infection.In addition, a cross-comparison of the relative concentrations of oxygenated hemoglobin in the frontal lobe between the two groups revealed that whether in the R-OFC or L-VLPFC, the relative concentration in the AT-rTMS group was significantly lower than that in the PC group (relative concentration value < 0, Fig. 9e-f).
a Analysis of sleep monitoring data between influenza patients after treatment (AT-rTMS) and the positive control (PC) group(S5);b.Analysis of differences in relative hemoglobin concentration in 8 frontal lobe channel regions between the AT-rTMS group and the PC group;c, d.Brain region channel maps showing the distribution of differences in relative oxygenated hemoglobin concentration in the frontal cortex between the AT-rTMS group and the PC group (N = 1);e.Overall cross-comparative analysis of differences in relative oxygenated hemoglobin concentration in the frontal cortex between the AT-rTMS group and the PC group (R-OFC region);f.Overall cross-comparative analysis of differences in relative oxygenated hemoglobin concentration in the frontal cortex between the AT-rTMS group and the PC group (L-VLPFC region).
Discussion
This study employed a retrospective cohort design and integrated multi-dimensional assessment approaches such as MRA, PSG, and fNIRS to systematically investigate the intervention effect of rTMS therapy on sleep disorders following influenza virus infection. It verified the value of neuromodulation technology in improving virus-related sleep disorder complications, and revealed the potential association pathway of “influenza infection - immune inflammation - abnormal brain region function - sleep disorder”. This provides new theoretical basis and clinical strategies for the management of complications during the acute phase of influenza.Sleep disorders are highly prevalent in acute respiratory viral infections and may exacerbate adverse infection outcomes. Previous studies have confirmed that poor sleep quality is closely associated with the severity of COVID-19, influenza, and upper respiratory tract infections. Additionally, influenza virus can induce narcolepsy-like sleep disturbances by affecting sleep-wake regulatory neurons, which lays an important foundation for this study [18, 19].
Through two-sample Mendelian randomization analysis, this study clearly identified influenza infection as a potential risk factor for sleep disorders (with insomnia as the core). This finding is consistent with the epidemiological conclusion that “the incidence of sleep disorders increases significantly within 2 weeks after influenza infection”. Moreover, the use of genetic instrumental variables excluded confounding factors, thereby strengthening the evidence for a causal association.More crucially, this study is the first to identify serum IgM levels and blood neutrophil count as key mediators of influenza-induced sleep disorders. Both factors showed a strong positive correlation with the SD-VAS and were significantly associated with changes in gray matter volume in the frontal lobe. These results suggest that influenza may activate humoral immunity (elevated IgM) and innate immunity (increased neutrophils), triggering central nervous system inflammation, which in turn leads to abnormal brain structure and function, and ultimately induces sleep disorders [20]. This provides clear targets for targeted interventions.Notably, neutrophil-related inflammatory indicators may also affect the response to neuromodulation therapy. Although our study did not formally test the predictive value of the neutrophil–lymphocyte ratio (NLR), emerging evidence suggests that systemic inflammation may modulate response to neuromodulation. It is plausible that a pro-inflammatory state—reflected by high NLR—could also attenuate therapeutic efficacy by impairing cortical plasticity. Future studies should prospectively evaluate baseline NLR as a potential biomarker for both safety and clinical response to rTMS in sleep and mood disorders [21].
Results of MRA showed that influenza infection was associated with structural changes in 27 brain regions, with the frontal lobe being the most prominently affected, followed by the temporal lobe and parietal lobe. This finding was highly consistent with the functional results of fNIRS: before treatment, patients with influenza in the BT-rTMS group had significantly higher concentrations of oxygenated hemoglobin in the R-OFC and L-VLPFC of the frontal lobe compared with the NC group (relative concentration value > 0), suggesting excessive activation of neurons in these two brain regions.fNIRS has been widely used in studies on sleep-related diseases. For instance, it has been used to detect abnormalities in the prefrontal functional network in adult patients with obstructive sleep apnea [17], and can also capture changes in brain region activity under sleep deprivation [22], providing methodological support for the analysis of brain mechanisms in this study.Further MRA analysis with insomnia as the exposure factor confirmed that the left inferior frontal gyrus and right orbital gyrus are key regulatory brain regions for influenza-related insomnia. The R-OFC is involved in emotional regulation and cortical regulation of the sleep-wake cycle, while the L-VLPFC, as a core node of the default mode network, has functional abnormalities that are closely associated with difficulty falling asleep and sleep maintenance disorders.Previous studies have mostly focused on brain region mechanisms in chronic neurological diseases (e.g., depression). Our findings suggest that rTMS may not only directly modulate prefrontal hyperactivity—particularly in the right orbitofrontal cortex (R-OFC) and left ventrolateral prefrontal cortex (L-VLPFC)—but also indirectly influence peripheral immune responses, such as IgM and neutrophil levels. Conversely, influenza-induced inflammation may exacerbate cortical dysregulation, creating a bidirectional “brain–immune” loop that perpetuates sleep disruption. Although our retrospective design does not establish causality in this interaction, the concurrent improvements in both neural activity and immune markers following rTMS support a synergistic mechanism.This study is the first to link brain region abnormalities to sleep disorders related to acute viral infection, filling the gap in the “infection-brain-sleep” research field and laying a foundation for subsequent studies.
Combining rTMS with conventional treatment significantly improved sleep quality in influenza patients in the short term. Compared with the PC group that received only conventional treatment, PSG monitoring showed that the AT-rTMS group had a significantly increased sleep efficiency and a more reasonable proportion of each stage of non-rapid eye movement (NREM) sleep. fNIRS results revealed that the concentrations of oxygenated hemoglobin in the R-OFC and L-VLPFC of the AT-rTMS group were significantly lower than those of the PC group (relative concentration value < 0), suggesting that rTMS can effectively inhibit the excessive activation of the frontal lobe and restore the functional balance of brain regions. A large number of studies have confirmed the improvement effect of rTMS on sleep disorders. For example, in primary sleep disorders, rTMS can improve sleep structure by regulating cortical activity [23]; for patients with chronic insomnia, rTMS targeting the DLPFC can restore abnormal sleep electroencephalogram (EEG) power [24] and reshape functional connectivity with the hippocampus [25, 26]. Regarding safety, patients in the rTMS group did not report common adverse reactions such as headache, scalp discomfort, or dizziness during or after the 3-day treatment course. No serious adverse events were observed. These findings suggest that short-term, high-frequency rTMS is well tolerated in patients with acute influenza-associated sleep disturbances. The present study further confirms the effectiveness of rTMS in sleep disorders related to acute infection, expanding its application scenarios.Meanwhile, as a sensitive tool for evaluating the efficacy of rTMS, fNIRS has been used in multiple studies to monitor its regulatory effect on cortical activity [27], providing reliable reference for verifying the efficacy in this study.
The timeliness and safety of rTMS intervention make it an ideal adjuvant treatment for sleep disorders during the acute phase of influenza, and it is particularly suitable for professional groups who need to quickly regain attention, such as engineers, medical staff, and drivers, as it can reduce the risk of occupation-related adverse events caused by sleep disorders and minimize the loss of social productivity; from the perspective of the mechanism of action, the effects of rTMS may be achieved through two pathways: the first is directly modulating the excitability of frontal cortical neurons, inhibiting the overactivated R-OFC and L-VLPFC, and restoring the balance of cortical regulation in the sleep-wake cycle, which is consistent with previous conclusions that rTMS improves sleep and cognitive functions by regulating cortical activity [28], elevated IgM and neutrophils may contribute to prefrontal dysfunction through complementary pathways. Although IgM does not cross the intact blood–brain barrier, it can activate complement (e.g., C5a), triggering neuroinflammation and impairing GABA ergic inhibition. Neutrophils release reactive oxygen species and myeloperoxidase, which may disrupt barrier integrity and promote oxidative stress in cortical regions. Together, these immune-mediated effects could lead to orbitofrontal hyperactivity and sleep disruption observed in our patients; the second is indirectly affecting the immune-inflammatory response,although this study did not directly detect the impact of rTMS on IgM and neutrophils, previous studies have shown that rTMS can inhibit the release of peripheral inflammatory factors by regulating the prefrontal-thalamic-limbic system circuit [29], so it is speculated that rTMS may reduce influenza related immune inflammatory levels, alleviate brain region damage, and thereby improve sleep, and the “neuro-immune” dual regulation hypothesis requires verification in subsequent prospective studies. Notably, the parallel improvements in prefrontal cortical activity, peripheral immune markers, and sleep quality suggest a potential bidirectional interaction between central neuromodulation and systemic immunity. On one hand, rTMS-induced suppression of hyperactivity in the right orbitofrontal cortex, a region implicated in emotional and autonomic regulation—may enhance top-down inhibitory control over peripheral immune responses via autonomic outflow. On the other hand, the resolution of acute infection–related immune activation (as reflected by declining IgM and neutrophil levels) may reduce neuroinflammatory signaling, thereby alleviating cortical hyperexcitability and promoting sleep stability. Together, these reciprocal processes may form a self-reinforcing loop through which rTMS contributes to multisystem recovery.Although our study focused on sleep outcomes, it is worth noting that rTMS may concurrently alleviate other influenza-related neuropsychiatric symptoms. rTMS improves both sleep and memory, suggesting potential broader cognitive benefits. Given that viral infections like influenza can impair hippocampal-prefrontal circuitry and lead to transient memory deficits, future studies should evaluate whether the observed sleep improvements with rTMS are accompanied by cognitive recovery in this population [28].
As a depression treatment technology approved by the FDA, rTMS has been proven effective in sleep disorders associated with chronic neurological diseases; for example, in neuropsychiatric disorders, rTMS targeting the DLPFC can improve symptoms in multiple domains, including sleep [30], and several systematic reviews and meta-analyses also support its value in insomnia treatment [31, 32]. However, the application of rTMS in acute infectious diseases remains a gap—existing literature mostly focuses on chronic insomnia or psychiatric disorders, with no involvement in transient sleep disorders during the acute phase of viral infection. In addition, other non-invasive brain stimulation technologies (such as transcranial direct current stimulation [tDCS] and transcranial alternating current stimulation [tACS]) have also shown potential in sleep disorder treatment: for instance, alpha-frequency tACS applied to the medial parietal lobe can improve chronic insomnia [33], and the efficacy of tDCS combined with rTMS may be superior to monotherapy [34], but comparisons of efficacy between different technologies still need further exploration [35].While tACS has shown promise in modulating sleep rhythms, rTMS offers distinct advantages in the context of acute post-infectious sleep disorders. Unlike tACS—which primarily entrains endogenous oscillations—rTMS can directly modulate cortical excitability and induce longer-lasting neuroplastic changes in prefrontal regions implicated in both immune regulation and sleep control. Given the pronounced prefrontal hyperactivation and inflammatory state observed in influenza-related insomnia, rTMS’s capacity for targeted neuromodulation may render it particularly suitable for this acute, inflammation-driven subtype. This study is the first to apply rTMS to influenza patients, confirming its feasibility and effectiveness in acute infection scenarios, expanding the application boundaries of neuromodulation technology, and also providing a reference for the management of sleep complications related to other acute respiratory viral infections such as COVID-19 and respiratory syncytial virus infections.Our study employed a fixed rTMS protocol (10 Hz, 5 consecutive days) targeting the prefrontal cortex. While this regimen demonstrated rapid efficacy in alleviating influenza-related sleep disturbances, we acknowledge that optimal parameters—such as stimulation frequency (e.g., 5 Hz vs. 10 Hz), treatment duration (e.g., 3 vs. 5 days), or target localization—may vary depending on specific insomnia phenotypes (e.g., sleep-onset vs. sleep-maintenance difficulties). Future studies should explore parameter titration and symptom-guided target selection to advance personalized rTMS therapy for post-infectious sleep disorders.
Unlike traditional clinical studies that rely solely on subjective assessments (e.g., SD-VAS) or a single objective indicator (e.g.,PSG), this study integrated MRA(for causal inference), fNIRS(for brain function), blood immune indicators (for inflammatory status), PSG (for sleep structure), and a clinical prediction model to form a multi-dimensional evaluation system: MRA excluded the interference of confounding factors on the “influenza-sleep disorder” association, fNIRS revealed the dynamic changes of abnormal brain region function, blood indicators identified key mediating factors, and the clinical prediction model quickly screened high-risk patients with influenza-related insomnia using 7 blood indicators—providing a tool for clinical precise intervention and significantly enhancing the reliability of results and their clinical translation value. Non-invasive brain stimulation technology holds broad prospects in sleep medicine; in the future, its efficacy is expected to be improved by precisely locating targets and optimizing stimulation parameters [36], and while improving sleep, it may also exert a positive impact on accompanying psychosocial symptoms [37], offering a new direction for the comprehensive management of patients with acute infections. As a non-invasive intervention with short-term efficacy,rTMS can be applied in hospital emergency departments, infectious disease departments, or community health service centers during influenza seasons to provide rapid intervention for patients with prominent sleep disorder symptoms.Given the relatively rapid improvement in sleep continuity and prefrontal cortical regulation observed in our cohort, rTMS may offer a time-efficient intervention for individuals in high-demand occupations where swift cognitive recovery is critical. However, this potential advantage remains hypothetical, as our study did not assess occupational subgroups or occupation-specific insomnia phenotypes. Future trials should stratify by professional context to evaluate whether therapeutic efficacy varies across such groups.
Our study has certain limitations: A notable limitation of this study is the absence of a sham-rTMS control group. As this was a retrospective analysis of real-world clinical practice during acute influenza infection, the implementation of sham stimulation was neither ethically justifiable nor practically feasible. However, the observed improvements were not limited to subjective reports; they were corroborated by objective measures including polysomnography (e.g., increased sleep efficiency and REM duration), reduced oxygenated hemoglobin in prefrontal regions on fNIRS, and normalization of inflammatory markers (e.g., IgM and neutrophil counts). These multimodal biological changes are unlikely to be fully explained by placebo effects alone, lending support to a genuine neuromodulatory impact of rTMS. In addition, as a retrospective cohort study, it had a small sample size (N = 55 for infected population, N = 16 for the rTMS group, N = 20 for the PC group, and N = 20 for the NC group); additionally, it did not adopt a randomized grouping design, which may lead to selection bias. The study only included patients aged 18-40 years without underlying diseases, excluding high-risk populations such as the elderly and those with comorbid chronic diseases (e.g., hypertension, diabetes), which limits the extrapolation of the results. However, the consistent direction and magnitude of effects across multimodal outcomes—including polysomnography, fNIRS-derived cortical activity, and systemic immune markers—support the presence of a biologically plausible signal. It should be noted that gender was not included as a stratification factor in our analysis, and the potential influence of sex differences on rTMS response remains unexplored. In the future, large-sample, multi-center, randomized controlled trials are needed, with the inclusion of a broader population, to further verify the efficacy of rTMS. Meanwhile, this study did not conduct an in-depth exploration of the mechanism pathways; in subsequent research, it is necessary to expand the scope of brain region monitoring and clarify the molecular mechanisms underlying rTMS-induced improvement of influenza-related insomnia through animal experiments or in vitro cell experiments. Furthermore, the inhibitory effect of rTMS on the frontal cortex may exert its effects by influencing brain network connectivity [38]; in the future, brain network analysis techniques can be combined to further explore its mechanism of action.
In conclusion, our study confirms that rTMS can improve sleep disorders following influenza virus infection by regulating the function of frontal lobe brain regions, and for the first time reveals the mediating role of serum IgM and neutrophils in the “influenza-sleep disorder” association. Despite its limitations, this study still provides a new strategy for the treatment and management of sleep disorder complications related to acute infectious diseases, and also lays a foundation for the application of neuromodulation technology in the field of infectious diseases.
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Paget J, Spreeuwenberg P, Charu V, Taylor RJ, Iuliano AD, Bresee J, et al. Global mortality associated with seasonal influenza epidemics: new burden estimates and predictors from the GLaMOR Project. Lancet. 2019;391:1285–95.
Diamond C, Gong H, Sun FY, Liu Y, Quilty BJ, Jit M, et al. Regional-based within-year seasonal variations in influenza-related health outcomes across mainland China: a systematic review and spatio-temporal analysis. BMC Med. 2022;20:324.
Lafond KE, Porter RM, Whaley MJ, Suizan Z, Ran Z, Aleem MA, et al. Global burden of influenza-associated lower respiratory tract infections and hospitalizations among adults: A systematic review and meta-analysis. PLoS Med. 2021;18:e1003550.
García-Azorín D, Santana-López L, Lozano-Alonso JE, Ordax-Díez A, González-Osorio Y, Rojo-Rello S, et al. InfluenCEF study: Clinical phenotype and duration of headache attributed to influenza infection. Cephalalgia. 2023;43:3331024231212900.
Bolek H, Ozisik L, Caliskan Z, Tanriover MD. Clinical outcomes and economic burden of seasonal influenza and other respiratory virus infections in hospitalized adults. J Med Virol. 2023;95:e28153.
Lasselin J, Ingre M, Regenbogen C, Olsson MJ, Garke M, Brytting M, et al. Sleep during naturally occurring respiratory infections: A pilot study. Sleep Med. 2019;56:101–7.
Byrd-Leotis L, Cummings RD, Steinhauer DA. The interplay between the host receptor and influenza virus hemagglutinin and neuraminidase. Int J Mol Sci. 2017;18:1541.
Majeed A, Quint JK, Bhatt S, Davies F, Islam N. Non - pharmaceutical interventions: evaluating challenges and priorities for future health shocks. BMJ. 2024;387:e080528.
Wu H, Lv J, Liu M, Wu Y, Qu Y, Ji L, et al. The long - term effect of repetitive transcranial magnetic stimulation in the treatment of intractable insomnia. J Clin Neurosci. 2022;101:251–6.
Jiang CG, Zhang T, Yue FG, Yi ML, Gao D. Efficacy of repetitive transcranial magnetic stimulation in the treatment of patients with chronic primary insomnia. J Psychiatr Res. 2023;157:260–7.
Sun N, He Y, Wang Z, Zou W, Liu X. The effect of repetitive transcranial magnetic stimulation for insomnia: a systematic review and meta-analysis. Sleep Med. 2021;77:226–37.
Wei L, Zhang Y, Wang J, Xu L, Yang K, Lv X, et al. Parietal-hippocampal rTMS improves cognitive function in Alzheimer’s disease and increases dynamic functional connectivity of default mode network. Psychiatry Res. 2022;315:114721.
Nardone R, Sebastianelli L, Versace V, Brigo F, Golaszewski S, Pucks-Faes E, et al. Effects of repetitive transcranial magnetic stimulation in subjects with sleep disorders. Sleep Med. 2020;71:113–21.
Jiang S, Li H, Zhang L, Mu W, Zhang Y, Chen T, et al. Generic diagramming platform (GDP): a comprehensive database of high-quality biomedical graphics. Nucleic Acids Res. 2025;53:D1670–D1676.
Irwin MR. Sleep and inflammation: partners in sickness and in health. Nat Rev Immunol. 2019;19:702–15.
Xia M, Wang J, He Y. BrainNet viewer: a network visualization tool for human brain connectomics. PLoS ONE. 2013;8:e68910.
Mingming Z, Wenhong C, Xiaoying M, Yang J, Liu HH, Lingli S, et al. Abnormal prefrontal functional network in adult obstructive sleep apnea: A resting-state fNIRS study. J Sleep Res. 2024;33:e14033.
Jones SE, Maisha FI, Strausz SJ, Lammi V, Cade BE, Tervi A, et al. The public health impact of poor sleep on severe COVID-19, influenza and upper respiratory infections. EBioMedicine. 2023;93:104630 https://doi.org/10.1016/j.ebiom.2023.104630.
Tesoriero C, Codita A, Zhang MD, Cherninsky A, Karlsson H, Grassi-Zucconi G, et al. H1N1 influenza virus induces narcolepsy-like sleep disruption and targets sleep-wake regulatory neurons in mice. Proc Natl Acad Sci USA. 2016;113:E368–77.
Zenaro E, Pietronigro E, Della Bianca V, Piacentino G, Marongiu L, Budui S, et al. Neutrophils promote Alzheimer’s disease-like pathology and cognitive decline via LFA-1 integrin. Nat Med. 2015;21:880–6.
Koparal B, Temizkan HN, Aksu MH, Karadağ RF. Higher neutrophil-lymphocyte ratio predicts poor response to repetetive transcranial magnetic stimulation(rTMS) in treatment resistant depression. Eur Arch Psychiatry Clin Neurosci. 2025;275:2547–55.
Pan Y, Borragán G, Peigneux P. Applications of functional near-infrared spectroscopy in fatigue, sleep deprivation, and social cognition. Brain Topogr. 2019;32:998–1012.
Lanza G, Fisicaro F, Cantone M, Pennisi M, Cosentino FII, Lanuzza B, et al. Repetitive transcranial magnetic stimulation in primary sleep disorders. Sleep Med Rev. 2023;67:101735 https://doi.org/10.1016/j.smrv.2022.101735.
Zhao X, Liu J, Shao Z, Liu X, Wang Z, Yuan K, et al. Restoration of abnormal sleep EEG power in patients with insomnia disorder after 1Hz rTMS over left DLPFC. Front Psychiatry. 2024;15:1431837.
Liu X, Wang S, Niu X, Liu Z, Meng X. The effects of transcranial magnetic stimulation on patients with chronic insomnia: a prospective functional near-infrared spectroscopy study. Sleep Med. 2025;131:106517.
Li M, Zhu Y, Zhang X, Yang H, Zhang S, Liu J, et al. 1Hz rTMS over left DLPFC rewired the coordination with hippocampus in insomnia patients: A pilot study. Brain Stimul. 2022;15:437–40.
Chen SY, Tsou MH, Chen KY, Liu YC, Lin MT, et al. Impact of repetitive transcranial magnetic stimulation on cortical activity: a systematic review and meta-analysis utilizing functional near-infrared spectroscopy evaluation. J Neuroeng Rehabil. 2024;21:108.
Malkani RG, Zee PC. Brain stimulation for improving sleep and memory. Sleep Med Clin. 2022;17:505–21.
Geiser T, Hertenstein E, Fehér K, Maier JG, Schneider CL, Züst MA, et al. Targeting arousal and sleep through noninvasive brain stimulation to improve mental health. Neuropsychobiology. 2020;79:284–92.
Kan RLD, Padberg F, Giron CG, Lin TTZ, Zhang BBB, Brunoni AR, et al. Effects of repetitive transcranial magnetic stimulation of the left dorsolateral prefrontal cortex on symptom domains in neuropsychiatric disorders: a systematic review and cross-diagnostic meta-analysis. Lancet Psychiatry. 2023;10:252–9.
Krone LB, Fehér KD, Rivero T, Omlin X. Brain stimulation techniques as novel treatment options for insomnia: A systematic review. J Sleep Res. 2023;32:e13927.
Ma H, Lin J, He J, Lo DHT, Tsang HWH. Effectiveness of TES and rTMS for the treatment of insomnia: meta-analysis and meta-regression of randomized sham-controlled trials. Front Psychiatry. 2021;12:744475.
Wang L, Chen Y, Piao Z, Gu X, Liu H, Wang D, et al. Medial parietal alpha-frequency transcranial alternating current stimulation for chronic insomnia: a randomized sham-controlled trial. Psychol Med. 2025;55:e102.
Zhou Q, Liu Z, Yu C, Wang Q, Zhuang W, Tang Y, et al. Effect of combined treatment with transcranial direct current stimulation and repetitive transcranial magnetic stimulation compared to monotherapy for the treatment of chronic insomnia: a randomised, double-blind, parallel-group, controlled trial. BMC Med. 2024;22:538.
Shao Z, Guo Y, Yue L, Liu X, Liu J, Zhao X, et al. Comparisons of transcranial alternating current stimulation and repetitive transcranial magnetic stimulation treatment therapy for insomnia: a pilot study. Gen Psychiatr. 2024;37:e101184.
Krone LB, Song SH, Jaramillo V, Violante IR. The future of non-invasive brain stimulation in sleep medicine. J Sleep Res. 2025;34:e70071.
Luff CE, de Lecea L. Can neuromodulation improve sleep and psychiatric symptoms?. Curr Psychiatry Rep. 2024;26:650–8.
Freedman M, Binns MA, Meltzer JA, Hashimi R, Chen R. Enhanced mind-matter interactions following rTMS induced frontal lobe inhibition. Cortex. 2024;172:222–33.
Funding
This work was supported by the National Natural Science Foundation of China (No. 82201774,No. 82500922,No. 82503662), the China Postdoctoral Science Foundation (2024M754265 and 2024T171196).
Author information
Authors and Affiliations
Contributions
H.D and X.M were primarily responsible for writing the full manuscript, conducting the collection and analysis of core data, designing and creating key figures such as brain region visualizations and ROC curves, assisting in writing the manuscript and sample selection, participating in the processing of IgM and fNIRS data, and generating auxiliary figures related to sleep parameters and blood indicators. C.Z was responsible for the follow-up of SD-VAS scores and ELISA experiments, and participated in the analysis of sleep parameters and the writing of parts of the results section. Y.Y, J.B, K.H, Y.W, and others participated in the collection and filing of clinical data, with divided responsibilities including adverse reaction monitoring, MRI data collation, and proofreading of manuscript details. L.G, Y.Z, and H.Z assisted in patient screening and medical record retrieval, analyzed the clinical applicability of rTMS based on outpatient experience, and provided translational references. L.B, L.Y, and Y.G assisted in analyzing the infection-brain function mechanism, supplemented relevant literature, and interpreted the functional significance of key brain regions. H.W provided guidance on fNIRS technology, demonstrated the mechanism of rTMS, reviewed the academic quality of the manuscript, and communicated matters related to ethics. K.Z designed and coordinated the study, applied for funding, interpreted the significance of the prediction model, and coordinated work division to solve problems. W.Q reviewed the rTMS protocol, analyzed the advantages of combined therapy, and provided guidance on manuscript submission and revision.
Corresponding authors
Ethics declarations
Competing interests
All authors involved in this study declare that there are no conflicts of interest.The ethics of this study have been reviewed and approved by review approval document of the medical ethics committee of the first affiliated hospital of the AirForce Medical University (Ethics Document No. KY20252111-F-1).
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Du, H., Meng, X., Zhang, C. et al. rTMS for rapid relief of sleep disorders induced by influenza virus infection: a clinical retrospective study. Transl Psychiatry 16, 218 (2026). https://doi.org/10.1038/s41398-026-03988-6
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41398-026-03988-6