Abstract
The role of extranodal (EN) sites as potential sanctuary regions resistant to CD19-directed chimeric antigen receptor T-cell (CAR-T) therapy in large B-cell lymphoma (LBCL) remains unclear. To investigate this, we retrospectively analyzed 283 adults treated with commercial CD19 CAR-T therapy, assessing 958 PET-CT scans across four time points: pre-apheresis, pre-lymphodepletion, best response, and relapse. EN involvement prior to CAR-T therapy was common (76%). Outcomes for patients with exclusive EN disease were similar to those with nodal (ND) disease alone; however, patients with concomitant EN and ND disease (EN + ND) had lower complete response rates and shorter progression-free survival. Site-specific outcomes varied: lungs/pleura/pericardium and gastrointestinal/peritoneum involvement had the lowest local response rates (48% and 51%, respectively). Notably, the risk of same-site relapse was highest in the lungs/pleura/pericardium (hazard ratio [HR] 7.8) and gastrointestinal/peritoneum (HR 5.97). Among patients relapsing after CAR-T, two-year overall survival rates from time of relapse were significantly lower in those with EN relapse (23% for exclusive EN; 25% for EN + ND) compared to exclusive ND relapse (64%; p = 0.008). These findings underscore the high prevalence of EN disease in CAR-T recipients and its site-specific impact on outcomes, highlighting the need for organ-targeted strategies to enhance treatment efficacy.

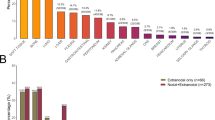
Differential site-specific response and relapse/progression risk according to pre-CAR-T therapy anatomical site involvement in Large B-cell Lymphoma. Risk of site-specific relapse or progression was not evaluable for CNS/orbital/cranial sinuses, adrenal/genitourinary, hepatobiliary/pancreas, and spleen due to insufficient number of events.
Similar content being viewed by others
Introduction
Chimeric antigen receptor T-cell (CAR-T) therapy has transformed the care of relapsed or refractory large B-cell lymphoma (LBCL) [1,2,3]. While CAR-T therapy achieves significant remission rates, up to 60% of patients experience relapse or refractory disease, highlighting ongoing challenges [4,5,6]. These outcomes reflect a complex interplay among host factors, CAR-T product attributes, and disease-specific characteristics [7,8,9,10]. Among these, tumor-related features—such as disease burden and lymphoma-specific genomic alterations—have emerged as important determinants of response to CAR-T therapy [1,2,3, 11,12,13]. However, variability in disease control among CAR-T recipients remains poorly understood, necessitating further investigation.
The inherent heterogeneity of LBCL is further compounded by variations in disease involvement across anatomical sites [14,15,16]. Differences in organ tropism, driven by genetic and phenotypic heterogeneity, may significantly influence treatment response [17,18,19,20,21]. Extranodal involvement has been widely recognized as an adverse prognostic factor in the first-line treatment of LBCL, with certain regions—such as the central nervous system (CNS), respiratory tract, liver, and pancreas—associated with particularly poor outcomes [15,16,17]. However, data on the influence of anatomical disease distribution in patients receiving CAR-T therapy are limited, with most studies focusing primarily on CNS involvement [22,23,24,25]. The broader role of anatomical sites in shaping treatment response remains unclear and warrants further investigation.
In this retrospective observational study, we hypothesized that specific anatomical sites may act as protective sanctuaries from CAR-T therapy. To test this hypothesis, we analyzed LBCL site involvement patterns before and after CAR-T therapy and evaluated their implications on key clinical outcomes.
Methods
Patient cohort and design
We conducted a retrospective analysis of 283 adult patients (age ≥18 years) with relapsed or refractory LBCL who received CAR-T therapy at Memorial Sloan Kettering Cancer Center (MSK) from January 2018 to September 2023. Patients with primary CNS involvement and patients with no evidence of disease at the time of pre-apheresis assessment were excluded from the study. CAR-T products were administered as standard of care (axicabtagene ciloleucel [axi-cel], tisagenlecleucel [tisa-cel], and lisocabtagene maraleucel [liso-cel]), except for liso-cel infusions, some of which were performed as part of a clinical trial (NCT02631044) [3].
Patient data were recorded and entered into a REDCap database [26].
Treatment response and outcome
Treatment response was assessed according to Lugano classification [27]. The best overall response was defined as the most favorable response, achieved at any time from CAR-T cell infusion up to day 365. Overall survival (OS) and progression-free survival (PFS) were measured from the time of CAR-T cell infusion; corresponding events were death for OS and death, relapse, or disease progression for PFS. Patients without events were censored at their date of last follow-up. Relapse and progression were estimated using cumulative incidence functions with death as a competing risk. Site-specific relapse or progression was also analyzed using cumulative incidence functions; however, for these outcomes, competing risks were death along with relapse or progression in a site other than the one under investigation. Site-specific complete response to CAR-T therapy was defined as the absence of disease at previously involved sites during the best response assessment. Cytokine release syndrome (CRS) and immune effector cell-associated neurotoxicity syndrome (ICANS) were defined according to the American Society of Transplantation and Cellular Therapy criteria [28].
Evaluation of site-specific disease involvement
Disease involvement was systematically evaluated at four key time points: prior to apheresis, prior to lymphodepletion (LD), at best response, and at relapse or progression (Fig. 1A). A total of 958 18F-fluorodeoxyglucose positron emission tomography/computed tomography (PET-CT) scans were analyzed, including 281 pre-apheresis, 256 pre-LD, 273 at best response, and 148 at relapse or progression. Data extracted from PET-CT reports included the number and location of lesions, classification of sites as nodal (ND) or extranodal (EN), and disease staging. ND sites were defined according to the Revised European-American Lymphoma (REAL) classification, encompassing lymph nodes, spleen, thymus, and Waldeyer’s ring [15].
A Disease involvement was assessed at four time points using PET-CT and additional modalities, as outlined in the flow diagram. B Definitions for clustering of nodal and extranodal disease sites and localization by supradiaphragmatic and infradiaphragmatic nodes for ND disease. * Of 256 patients with available PET-CTs following apheresis, 231 were evaluated after bridging therapy. LBCL Large B-cell Lymphoma, LD lymphodepletion, PET-CT positron emission tomography/computed tomography, CNS central nervous system, MRI magnetic resonance imaging, BM bone marrow.
CNS involvement was evaluated using magnetic resonance imaging (MRI), with 351 scans reviewed, including 213 conducted prior to CAR-T therapy. CNS imaging was performed based on a clinical indication or suspicion, or for screening purposes. Response in patients with CNS involvement was assessed based on radiographic evaluation by a radiologist using PET-CT and MRI.
Disease involvement was categorized into three groups before CAR-T therapy: exclusively ND disease, exclusively EN disease, and concomitant EN and ND disease. Bone marrow involvement was not included in these categories since it was assessed in a small fraction of patients (n = 43). Bone disease was determined exclusively based on FDG uptake using standard radiographic criteria and considered as EN disease.
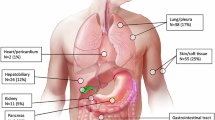
Anatomical site involvement was clustered as follows (Fig. 1B): CNS with orbital and cranial sinuses, lungs with pleura and pericardium, gastrointestinal with peritoneum, hepatobiliary with pancreas, adrenal glands with kidneys and ovarian, and bone with soft tissue. Additionally, nodal involvement was classified into supradiaphragmatic regions (cervical, supraclavicular, axillary, mediastinal, and hilar/paratracheal lymph nodes) and infradiaphragmatic regions (mesenteric/paraaortic, iliac, and inguinal lymph nodes). Spleen involvement was considered separately from the other nodal regions.
Metabolic tumor volume (MTV) and standardized uptake value (SUV) were calculated as previously described using PET-CT scans [29]. Same-site relapse or progression was defined as the reappearance or worsening of metabolic activity in the same anatomical location as prior tumors, based on radiologists assessment of increased SUV or other indicators of disease activity; death was considered a competing event.
Sequencing
Integrated Molecular Profiling and Analysis for Clinical Tumors (IMPACT) sequencing was used to identify somatic alterations in tumor samples. DNA was extracted from formalin-fixed, paraffin-embedded tumor tissues and matched non-involved samples, which were available for 118 patients prior to CAR-T therapy. Barcoded libraries were prepared, and sequencing targeted all exons and selected introns of approximately 400 cancer-related genes using a custom gene panel. Sequencing data were processed using an established bioinformatics pipeline to detect somatic mutations, gene fusions, and copy number alterations [30]. Genomic data were curated and uploaded to the cBioPortal platform for integrative analysis [31].
Statistical analysis
Continuous variables were summarized using the median and interquartile range, while categorical data were presented as frequencies and percentages.Tests of association were based on a Fisher’s exact test, Chi-square test, or a Wilcoxon rank-sum test, as appropriate. Cox proportional hazards models were used to analyze associations with OS and PFS, while logistic regression models were applied to grade 2–4 CRS, grade 2–4 ICANS, response to bridging therapy, and complete response (CR) to CAR-T therapy. Cause-specific Cox regression models estimated univariable and multivariable models of overall relapse and site-specific relapse. P-values below 0.05, including adjusted p-values, were considered statistically significant. All analyses were conducted using R statistical software (version 4.3, R Foundation for Statistical Computing).
Ethics approval and consent to participate
This retrospective study was approved by the Memorial Sloan Kettering Cancer Center Institutional Review Board (IRB), approval number 19–373. All methods were performed in accordance with relevant guidelines and regulations. The requirement for informed consent was waived by the IRB, as this retrospective study involved minimal risk to participants and did not adversely affect their rights or welfare.
Results
Population characteristics
We included 283 patients with a median age of 66 years (IQR: 56–72) who were treated with commercial CAR-T cell therapy (58% axi-cel, 24% tisa-cel, and 19% liso-cel) (Table 1). Diffuse large B-cell lymphoma Not Otherwise Specified was the most common diagnosis (77%), followed by high-grade B-cell lymphoma (16%) and other LBCL subtypes (7%). Transformed LBCL was reported in 36% of patients. At the last assessment before CAR-T infusion, 40% of patients had elevated LDH, and the median MTV was 23 mL (IQR: 1–141). Bridging therapy was administered to 82% of patients. CAR-T therapy was used as a third-line or later treatment in 80% of patients.
CRS occurred in 42% of patients, with grade ≥2 CRS in 29%. ICANS was observed in 29%, with grade ≥2 ICANS in 20%. The overall response rate (ORR) to CAR-T therapy was 80%, with a CR rate of 64%. With a median follow-up of 26 months (IQR: 13–49), 2-year PFS and OS were 36% (95% CI: 30–43%) and 53% (47–60%), respectively. At 2 years, the cumulative incidence of relapse or progression was 58% (51–64%), and non-relapse mortality was 7.4% (4.5–11%).
Extranodal disease status and response to bridging
At apheresis, 79 patients (25%) had ND disease only, 67 patients (24%) had exclusive EN disease, and 137 patients (51%) had concomitant EN and ND disease. Among the 231 patients who received bridging therapy, 64% underwent systemic treatment (with or without localized treatment, such as radiotherapy or intrathecal chemotherapy), while 18% received exclusively localized monotherapy.
To minimize bias related to the selection of bridging modalities, analyses were restricted to patients receiving systemic bridging therapy (Fig. S1). Among patients with evaluable post-bridging disease assessments (n = 175), the ORR to systemic bridging therapy was 47% (9% CR and 38% partial response). For patients with exclusive ND disease involvement pre-apheresis, the ORR to bridging therapy was 46% (CR 14%). ORR for patients with exclusive EN disease was 54% (CR 9%), and for those with concomitant EN and ND disease was 45% (CR 7%). In univariable logistic regression, neither exclusive EN disease nor concomitant EN and ND disease was significantly associated with overall response to bridging therapy (OR 0.73 [95% CI: 0.29–1.79] and OR 1.04 [0.25–2.15], respectively).
Patients with concomitant extranodal and nodal disease have unfavorable outcomes
At the last disease assessment before CAR-T infusion, 263 out of 283 (93%) had evidence of disease; 64 out of 263 patients (24%) had exclusive ND disease, while the remaining 199 patients (76%) exhibited some EN involvement. EN disease typically co-occurred with ND disease (133/199, 67%), with a smaller subset presenting with exclusive EN disease (66/199, 33%).
Patients with concomitant EN and ND involvement exhibited adverse prognostic factors such as lower Karnofsky performance status, elevated LDH levels pre-lymphodepletion, and higher MTV (Table 1). The most frequently observed mutations (Fig. S2, Table S1) in the patients with any EN disease involvement were KMT2D (40%), TP53 (38%), and CDKN2A (24%).
Rates of grade ≥ 2 CRS occurred in 50% of exclusive EN cases, 47% of concomitant EN and ND cases, and 31% of exclusive ND cases (p = 0.062). Grade ≥ 2 ICANS was more frequent in patients with EN involvement, appearing in 27% of both exclusive EN and combined EN and ND groups, compared to 6% in the exclusive ND group (p = 0.002). For patients with CNS/orbital/cranial sinus involvement at the last assessment, 38% experienced ICANS, versus 20% among those without CNS involvement (p = 0.093).
CR rates to CAR-T therapy were 68% for patients with exclusive ND disease, 70% for those with exclusive EN disease, and 59% for those with concomitant EN and ND disease. In multivariable logistic regression, adjusted for age, transformed lymphoma, KPS, pre-LD LDH, bridging therapy, and CAR-T product, patients with concomitant EN and ND disease had a significantly higher risk of not attaining CR compared to those with exclusive ND disease (OR 2.13 [1.10–4.23], p = 0.028). In contrast, patients with exclusive EN disease did not exhibit a significant difference (OR 1.17 [0.54–2.53], p = 0.7).
PFS at 2 years post-CAR-T infusion was highest in the exclusive ND group (51% [95% CI: 40–60%]), followed by 39% (27–55%) in the exclusive EN group, and 21% (15–31%) in the concomitant EN and ND group (p < 0.001) (Fig. 2A). In multivariable Cox regression adjusted for the same variables as above, concomitant EN and ND disease was associated with significantly lower PFS (HR 1.72 [1.12–2.65], p = 0.014) compared to exclusive ND. In contrast, there was no evidence that the PFS risk was increased with exclusive EN disease (HR 1.23 [0.76–1.98], p = 0.4).
Progression-free survival (A), and overall survival (B), relapse (C), and non-relapse mortality (NRM) (D) by nodal or extranodal disease status at last assessment before CAR-T therapy.
Two-year OS was also highest in the exclusive ND group (67% [55%-81%]), followed by 53% (41–70%) for exclusive EN, and 41% (33–52%) for concomitant EN and ND (p = 0.003) (Fig. 2B). In multivariable Cox regression, neither concomitant EN and ND disease (HR 1.52 [0.91–2.53], p = 0.11) nor exclusive EN disease (HR 1.30 [0.73-2.31], p = 0.4) were associated with a significantly shorter OS compared to exclusive ND.
Cumulative incidence of 2-year relapse or progression were 42% (29–54%) for ND versus 55% (40–68%) for exclusive EN versus 71% (62–79%) for concomitant EN-ND (p < 0.001, Fig. 2C). In multivariable cause-specific Cox regression, concomitant EN and ND disease had a higher risk of relapse or progression (HR 1.98 [1.23-3.12], p = 0.003) compared to exclusive ND disease. In contrast, the risk was not increased with exclusive EN disease (HR 1.32 [0.79–2.21]) compared to ND disease. The cumulative incidence of non-relapse mortality at 2 years post-infusion was 7.8% (3.2–15%) for exclusive ND, 9.1% (4.5–16%) for concomitant EN and ND, and 3.5% (0.63–11%) for exclusive EN (Fig. 2D)
In a sensitivity analysis replacing LDH with MTV in multivariable models, concomitant EN + ND remained associated with a higher risk of relapse or progression compared to exclusive ND disease (HR 1.72 [1.05–2.81], p = 0.003). Exclusive EN was associated with shorter OS (HR 1.92 [1.03–3.56], p = 0.04) and a numerically higher, but not statistically significant, risk of not achieving CR (OR 1.82 [0.75–4.52], p = 0.2). Concomitant EN + ND showed a trend toward worse PFS but did not reach statistical significance (HR 1.40 [0.88–2.22], p = 0.2).
CAR-T outcome is related to the number of extranodal sites
Next, we investigated the relationship between the number of EN sites at the last assessment before CAR-T and CAR-T outcomes, irrespective of nodal involvement. In the exclusive EN group, 49% had a single EN site, 15% had two sites, and 36% had three or more. In the concomitant EN + ND group, the corresponding rates were 26%, 20%, and 53% (p = 0.01).
Patients with one, two, and three or more EN disease sites before CAR-T therapy had CR rates of 62%, 72%, and 43%, respectively. Involvement of three or more sites was strongly related to shorter OS and PFS and higher relapse, while these were comparable between patients with one or two sites of disease (Tables S2 and S3).
Differential response rates across disease sites
Infradiaphragmatic and supradiaphragmatic regions were the most frequently involved sites, with ≥45% of patients affected both before apheresis and at the last disease assessment prior to CAR-T therapy (Fig. 3A). Commonly involved EN sites at both time points included bone/soft tissue (40–45%), gastrointestinal/peritoneum (22–23%), lungs/pleura/pericardium (18–20%), and hepatobiliary/pancreas (10–11%). CNS/orbital/cranial sinuses involvement was less frequent, observed in 8–9% of patients.
A Distribution of disease involvement sites at pre-apheresis, last assessment, and relapse/progression. B Circus diagram showing the relative frequency and pairwise co-occurrence of anatomical sites involved with lymphoma before apheresis. Arc length represents the frequency of site involvement, while ribbon width indicates the percentage of patients with co-occurrence at a second site. C Pairwise association analysis between anatomical sites involved before apheresis using logistic regression. The color represents the z-statistics from the model. Values above 1.96 correspond to frequently co-occurring pairs of sites, and values below -1.96 correspond to infrequently co-occurring pairs. The red * represents statistical significance (p < 0.05) using the z-statistic. D Site-specific response rates to CAR-T therapy. Response is assessed relative to disease involvement at last disease assessment before CAR-T therapy. BM bone marrow, GI gastrointestinal, CNS central nervous system.
At pre-apheresis disease assessment, strong associations between specific sites were identified: supradiaphragmatic nodes were correlated with bone/soft tissue and lungs/pleura/pericardium; infradiaphragmatic nodes were associated with the spleen; lungs/pleura/pericardium correlated with adrenal/genitourinary involvement; and the spleen correlated with hepatobiliary/pancreas involvement (Fig. 3B, C).
Site-specific CR rates to CAR-T therapy varied significantly. The highest responses were observed in bone/soft tissue (67%), supradiaphragmatic nodes (66%), and spleen (61%). In contrast, lower CR rates were noted for adrenal/genitourinary sites (48%), lungs/pleura/pericardium (51%), and CNS/orbital/cranial sinuses (58%) (Fig. 3D). Notably, in a subanalysis considering CNS involvement, without combining it with orbital or cranial sinuses involvement, site-specific CR rates were 50% among 16 evaluable patients.
Next, we evaluated whether site involvement at the last assessment before CAR-T therapy influenced overall patient-level outcomes. Univariable logistic regression identified specific sites associated with failure to achieve a CR, and these were incorporated into a multivariable model (Table 2). Adrenal/genitourinary, lungs/pleura/pericardium, and infradiaphragmatic nodes emerged as independent risk factors for not achieving a CR.
Using the same analytical framework, we examined PFS and OS. Adrenal/genitourinary, CNS/orbital/cranial sinuses, and hepatobiliary/pancreas involvement were associated with an increased risk of shorter PFS. Similarly, adrenal/genitourinary, hepatobiliary/pancreas, CNS/orbital/cranial sinuses, and gastrointestinal/peritoneum involvement were linked to shorter OS.
We performed a subanalysis considering CNS involvement at the last disease assessment before CAR-T, without combining it with orbital or cranial sinusoidal involvement. In multivariable Cox regression models, CNS involvement was associated with shorter PFS (HR 4.05 [95% CI: 2.14–7.67], p < 0.001) and OS (HR 3.60 [1.84–7.06], p < 0.001) compared to no CNS involvement.
Patterns of site-specific relapse and progression following CAR-T therapy
At the time of relapse or progression, the most frequently involved sites were infradiaphragmatic nodes (53%), supradiaphragmatic nodes (50%), and bone/soft tissue (48%) (Fig. 3A). The cumulative incidence of relapse or progression at a specific site at 2 years was highest for infradiaphragmatic nodes (31%), supradiaphragmatic nodes (29%), and lungs/pleura/pericardium (17%) (Fig. 4A).
A Cumulative incidence of relapse and progression after CAR-T by disease site. B Cumulative incidence of nodal and extranodal relapse and progression after CAR-T therapy. C Overall survival measured from the time of relapse or progression after CAR-T therapy.
The likelihood of relapse or progression in a specific site post-CAR-T was strongly associated with prior disease involvement at that location. Lungs/pleura/pericardium (HR 7.80, [95% CI: 4.14-14.7], p < 0.001), gastrointestinal/peritoneum (HR 5.97, [3.07–11.6], p < 0.001), and bone/soft tissue (HR 5.03, [2.72–9.29], p < 0.001) had the highest risk of same-site involvement after CAR-T therapy compared to no baseline involvement in that site (Table 3). Comparative risk of same-site relapse or progression was not evaluable for CNS/orbital/cranial sinuses, adrenal/genitourinary, hepatobiliary/pancreas, and spleen due to an insufficient number of events. However, there was a significant association between pre-CAR-T involvement at these sites and relapse or progression at the same location (p < 0.001 for all).
Patients with EN disease involvement after CAR-T therapy have shorter overall survival
The 1-year cumulative incidence of exclusive ND, exclusive EN, and concomitant EN and ND disease relapse or progression was 43% (95% CI: 32–54%), 48% (35–60%), and 60% (50–68%) (Fig. 4B). Importantly, in univariable Cox regression, patients with exclusive EN disease (HR 2.14, 95% CI: 1.10–4.17) or concomitant ND and EN (HR 2.52, 95% CI: 1.38–4.60, p = 0.003) at relapse or progression had a shorter OS from the time of relapse or progression compared to those with exclusive ND (Fig. 4C). Corresponding 1-year OS after relapse were 64% for ND (48–85%), 30% for EN (95% CI: 18–51%) and 30% for concomitant EN and ND (21%–42%).
Discussion
In this comprehensive analysis of over 900 PET-CT scans from 283 patients with LBCL, we observed a high burden of EN disease prior to CAR-T therapy, exceeding 75%. Importantly, our findings indicate that concomitant EN and nodal disease, rather than isolated EN disease, is associated with poorer outcomes after CAR-T therapy. We also identified site-specific variations in treatment response and outcomes at both the patient and organ levels, suggesting that certain anatomical sites may act as sanctuary regions from CAR-T activity. Finally, patients experiencing relapse or progression with any EN involvement after CAR-T therapy have shorter survival than those with ND relapse. These observations underscore the need for personalized treatment strategies to address and minimize EN disease both before and after CAR-T therapy.
The current understanding of EN disease impact in CAR-T therapy remains limited, with few studies specifically examining this aspect. The largest report to date,d5 involving 126 patients with LBCL treated primarily with tisa-cel (79%), described reduced PFS in those with three or more EN sites before CAR-T therapy. Our analysis is considerably larger and includes patients treated across all commercially available CAR-T products, offering a more detailed understanding of EN disease’s role in CAR-T therapy. Our analysis provides several novel insights important to patient care. First, EN disease is a negative prognostic factor but primarily in the presence of concurrent nodal disease. Second, certain EN sites, such as adrenal/genitourinary, CNS, and hepatobiliary/pancreas are linked with shorter PFS and OS compared to other sites. Third, EN sites tend to persist despite bridging therapy and recur in previously involved locations. Finally, when patients relapse with EN involvement, their survival is considerably shorter than that of counterparts who relapse or progress without EN disease. Therefore, strategies prioritizing comprehensive disease debulking, particularly for EN involvement before CAR-T therapy, are warranted.
Beyar Katz et al. [25] reported a tendency for higher rates of CRS and ICANS in patients with EN disease (CRS 76% vs. 57%, p = 0.06), consistent with our findings of increased toxicity in this population. Grade ≥2 ICANS was more frequent in patients with EN or EN + ND disease compared to ND alone. Among patients with CNS involvement, the frequency and severity of ICANS were similar to those without CNS involvement, aligning with studies suggesting that CNS involvement does not increase the risk of severe ICANS [32,33,34]. However, our study may have been underpowered to detect the difference. The increased toxicity observed in EN disease highlights the potential need for prophylactic strategies. While anakinra has been explored for ICANS prevention, its role remains investigational, and further studies are warranted to define its impact on patient outcomes [35].
Data on site-specific disease dynamics remain limited. For instance, a study of 32 patients reported poorer PFS and OS associated with pre-CAR-T soft tissue infiltration but lacked detailed data on other types of involvement [17]. In our cohort, however, bone or soft tissue involvement did not correlate with worse outcomes. Similarly, a study of 24 patients with gastrointestinal involvement found no difference in outcomes compared to 106 patients without such involvement before CAR-T [36]. In contrast, our analysis identified gastrointestinal or peritoneal involvement as being associated with shorter OS. Finally, our finding of shorter PFS and OS in patients with CNS involvement before CAR-T therapy aligns with previous reports identifying this site as high risk for CAR-T treatment failure [37, 38]. CNS-directed interventions may improve outcomes in this population [39].
We identified potential anatomical sites contributing to CAR-T therapy resistance, with low response rates observed in regions such as the adrenal/genitourinary system (48%), lungs/pleura/pericardium (51%), and central nervous system (58%). Disease involvement in these areas—particularly adrenal/genitourinary, hepatobiliary/pancreas, and CNS—was associated with reduced survival. These findings highlight the importance of disease localization in predicting CAR-T outcomes and tailoring management strategies. Site-specific radiotherapy may help reduce tumor burden in EN lesions before CAR-T therapy, addressing the limitations of traditional chemotherapy in accessing these sanctuary sites [6, 39,40,41].
EN disease burden in newly diagnosed LBCL is approximately 30% [42, 43]. However, its prevalence exceeds 75% in our cohort before CAR-T therapy, reflecting a more aggressive disease profile in these patients [15, 16], which may contribute to their enrichment in second-line or later treatments. Unique genomic and molecular features drive the tropism of LBCL to EN sites, with recurrent mutations in genes like MYD88, CD79B, and CDKN2A commonly described in CNS, skin, breast, and ear-nose-throat sites [20, 21, 44,45,46]. These shared mutations suggest molecular similarities among EN lymphomas across various locations. Molecular findings in our cohort align with previous studies, demonstrating a higher frequency of TP53 and KMT2D mutations in EN disease [47, 48]. Given that biopsies typically target a single site, the full extent of intra-patient heterogeneity in EN LBCL may also be underappreciated.
This study has notable limitations, including its retrospective and single-center design. The extent of EN involvement may be underestimated, as brain MRIs were conducted in only 67% of patients, and bone marrow assessments were performed too infrequently to be included in the analysis. However, PET-CT, the standard imaging modality for LBCL evaluation, was available for all patients, ensuring a consistent assessment of disease burden. Additionally, not all patients underwent biopsies to confirm relapse, which may affect the precision of relapse data. In addition, the association between specific EN sites and poorer survival or relapse risk, while significant, could be influenced by unmeasured factors such as treatment history and prior therapies. Future studies with multi-site sampling and longitudinal designs could enhance our understanding of EN disease dynamics in LBCL and provide a more comprehensive view of disease heterogeneity.
In conclusion, this study demonstrates the significant impact of disease localization on CAR-T therapy outcomes in lymphoma, with high rates of treatment failure in cases involving adrenal/genitourinary, hepatobiliary/pancreas, and CNS sites. The adverse prognosis associated with EN disease, along with the increased incidence and severity of treatment-related toxicities, calls for potentially personalized treatment strategies and vigilant monitoring in this vulnerable group. Future research should aim to elucidate the biological mechanisms driving these disparities and develop innovative approaches to enhance outcomes for patients with EN disease.
Data availability
The datasets supporting the findings of this study, including imaging and sequencing data, are available upon reasonable request from the corresponding author, subject to institutional and ethical approvals.
References
Neelapu SS, Jacobson CA, Ghobadi A, Miklos DB, Lekakis LJ, Oluwole OO, et al. Five-year follow-up of ZUMA-1 supports the curative potential of axicabtagene ciloleucel in refractory large B-cell lymphoma. Blood. 2023;141:2307–15. https://doi.org/10.1182/blood.2022018893
Schuster SJ, Bishop MR, Tam CS, Waller EK, Borchmann P, McGuirk JP, et al. Tisagenlecleucel in adult relapsed or refractory diffuse large B-Cell Lymphoma. N. Engl J Med. 2019;380:45–56. https://doi.org/10.1056/NEJMoa1804980
Abramson, Palomba JS, Gordon LI ML, Lunning MA, Wang M, Arnason J, et al. Lisocabtagene maraleucel for patients with relapsed or refractory large B-cell lymphomas (TRANSCEND NHL 001): a multicentre seamless design study. Lancet. 2020;396:839–52. https://doi.org/10.1016/S0140-6736(20)31366-0
Alarcon Tomas A, Fein JA, Fried S, Flynn JR, Devlin SM, Fingrut WB, et al. Outcomes of first therapy after CD19-CAR-T treatment failure in large B-cell lymphoma. Leukemia. 2023;37:154–63. https://doi.org/10.1038/s41375-022-01739-2
Di Blasi R, Le Gouill S, Bachy E, Cartron G, Beauvais D, Le Bras F, et al. Outcomes of patients with aggressive B-cell lymphoma after failure of anti-CD19 CAR T-cell therapy: a DESCAR-T analysis. Blood. 2022;140:2584–93. https://doi.org/10.1182/blood.2022016945
Figura NB, Robinson TJ, Sim AJ, Wang X, Cao B, Chavez JC, et al. Patterns and predictors of failure in recurrent or refractory Large B-cell lymphomas after chimeric antigen receptor T-cell therapy. Int J Radiat Oncol Biol Phys. 2021;111:1145–54. https://doi.org/10.1016/j.ijrobp.2021.06.038
Lindo L, Wilkinson LH, Hay KA. Befriending the hostile tumor microenvironment in CAR T-cell therapy. Front Immunol. 2020;11:618387 https://doi.org/10.3389/fimmu.2020.618387
Kankeu Fonkoua LA, Sirpilla O, Sakemura R, Siegler EL, Kenderian SS. CAR T cell therapy and the tumor microenvironment: Current challenges and opportunities. Mol Ther Oncol. 2022;25:69–77. https://doi.org/10.1016/j.omto.2022.03.009
Scholler N, Perbost R, Locke FL, Jain MD, Turcan S, Danan C, et al. Tumor immune contexture is a determinant of anti-CD19 CAR T cell efficacy in large B cell lymphoma. Nat Med. 2022;28:1872–82. https://doi.org/10.1038/s41591-022-01916-x
Roschewski M, Phelan JD, Jaffe ES Primary Large B-cell Lymphomas of Immune-Privileged Sites. Blood. Published online April 18, 2024:blood.2023020911. https://doi.org/10.1182/blood.2023020911
Shouval R, Alarcon Tomas A, Fein JA, Flynn JR, Markovits E, Mayer S, et al. Impact of TP53 genomic alterations in large B-cell lymphoma treated with CD19-Chimeric antigen receptor T-cell therapy. J Clin Oncol J Am Soc Clin Oncol. 2022;40:369–81. https://doi.org/10.1200/JCO.21.02143
Dean EA, Mhaskar RS, Lu H, Mousa MS, Krivenko GS, Lazaryan A, et al. High metabolic tumor volume is associated with decreased efficacy of axicabtagene ciloleucel in large B-cell lymphoma. Blood Adv. 2020;4:3268–76. https://doi.org/10.1182/bloodadvances.2020001900
Sworder BJ, Kurtz DM, Alig SK, Frank MJ, Shukla N, Garofalo A, et al. Determinants of resistance to engineered T cell therapies targeting CD19 in large B cell lymphomas. Cancer Cell. 2023;41:210–225.e5. https://doi.org/10.1016/j.ccell.2022.12.005
Rivas‐Delgado A, López C, Clot G, Nadeu F, Grau M, Frigola G, et al. Testicular large B‐cell lymphoma is genetically similar to PCNSL and distinct from nodal DLBCL. HemaSphere. 2024;8:e70024 https://doi.org/10.1002/hem3.70024
Castillo JJ, Winer ES, Olszewski AJ. Sites of extranodal involvement are prognostic in patients with diffuse large B-cell lymphoma in the rituximab era: an analysis of the Surveillance, Epidemiology and End Results database. Am J Hematol. 2014;89:310–4. https://doi.org/10.1002/ajh.23638
Takahashi H, Tomita N, Yokoyama M, Tsunoda S, Yano T, Murayama K, et al. Prognostic impact of extranodal involvement in diffuse large B-cell lymphoma in the rituximab era. Cancer. 2012;118:4166–72. https://doi.org/10.1002/cncr.27381
Zhou L, Li P, Ye S, Tang X, Wang J, Liu J, et al. Different sites of extranodal involvement may affect the survival of patients with relapsed or refractory non-Hodgkin lymphoma after chimeric antigen receptor T cell therapy. Front Med. 2020;14:786–91. https://doi.org/10.1007/s11684-020-0751-3
Li P, Chai J, Chen Z, Liu Y, Wei J, Liu Y, et al. Genomic Mutation Profile of Primary Gastrointestinal Diffuse Large B-Cell Lymphoma. Front Oncol. 2021;11. Accessed February 12, 2024. https://www.frontiersin.org/journals/oncology/articles/10.3389/fonc.2021.622648
López-Guillermo A, Colomo L, Jiménez M, Bosch F, Villamor N, Arenillas L, et al. Diffuse large B-cell lymphoma: clinical and biological characterization and outcome according to the nodal or extranodal primary origin. J Clin Oncol J Am Soc Clin Oncol. 2005;23:2797–804. https://doi.org/10.1200/JCO.2005.07.155
Cao XX, Li J, Cai H, Zhang W, Duan MH, Zhou DB. Patients with primary breast and primary female genital tract diffuse large B cell lymphoma have a high frequency of MYD88 and CD79B mutations. Ann Hematol. 2017;96:1867–71. https://doi.org/10.1007/s00277-017-3094-7
Ollila TA, Olszewski AJ. Extranodal diffuse large B Cell lymphoma: molecular features, prognosis, and risk of central nervous system recurrence. Curr Treat Options Oncol. 2018;19:38 https://doi.org/10.1007/s11864-018-0555-8
Bobillo S, Khwaja J, Ferreri AJM, Cwynarski K. Prevention and management of secondary central nervous system lymphoma. Haematologica. 2023;108:673–89. https://doi.org/10.3324/haematol.2022.281457
Chen DH, O’Reilly M, Cheok KP, Low R, Puranik R, Clark S, et al. CD19-targeting chimeric antigen receptor T-cell therapy is safe and effective for intra-cardiac B cell non-Hodgkin lymphoma. eJHaem. n/a(n/a). https://doi.org/10.1002/jha2.1020
Choquet S, Soussain C, Azar N, Morel V, Metz C, Ursu R, et al. CAR T-cell therapy induces a high rate of prolonged remission in relapsed primary CNS lymphoma: Real-life results of the LOC network. Am J Hematol. 2024;99:1240–9. https://doi.org/10.1002/ajh.27316
Beyar Katz O, Perry C, Grisariu-Greenzaid S, Yehudai-Ofir D, Luttwak E, Avni B, et al. Response rates of extra-nodal diffuse large B cell lymphoma to anti-CD19-CAR T cells: A real word retrospective multicenter study. Eur J Haematol. 2023;111:63–71. https://doi.org/10.1111/ejh.13968
Harris PA, Taylor R, Minor BL, Elliott V, Fernandez M, O’Neal L, et al. The REDCap consortium: Building an international community of software platform partners. J Biomed Inf. 2019;95:103208 https://doi.org/10.1016/j.jbi.2019.103208
Cheson BD, Fisher RI, Barrington SF, Cavalli F, Schwartz LH, Zucca E, et al. Recommendations for initial evaluation, staging, and response assessment of Hodgkin and non-Hodgkin lymphoma: the Lugano classification. J Clin Oncol J Am Soc Clin Oncol. 2014;32:3059–68. https://doi.org/10.1200/JCO.2013.54.8800
Lee DW, Santomasso BD, Locke FL, Ghobadi A, Turtle CJ, Brudno JN, et al. ASTCT Consensus Grading for Cytokine Release Syndrome and Neurologic Toxicity Associated with Immune Effector Cells. Biol Blood Marrow Transpl J Am Soc Blood Marrow Transpl. 2019;25:625–38. https://doi.org/10.1016/j.bbmt.2018.12.758
Leithner D, Flynn JR, Devlin SM, Mauguen A, Fei T, Zeng S, et al. Conventional and novel [18F]FDG PET/CT features as predictors of CAR-T cell therapy outcome in large B-cell lymphoma. J Hematol OncolJ Hematol Oncol. 2024;17:21 https://doi.org/10.1186/s13045-024-01540-x
Cheng DT, Mitchell TN, Zehir A, Shah RH, Benayed R, Syed A, et al. Memorial Sloan Kettering-integrated mutation profiling of actionable cancer targets (MSK-IMPACT): A hybridization capture-based next-generation sequencing clinical assay for solid tumor molecular oncology. J Mol Diagn. 2015;17:251–64. https://doi.org/10.1016/j.jmoldx.2014.12.006
Cerami E, Gao J, Dogrusoz U, Gross BE, Sumer SO, Aksoy BA, et al. The cBio cancer genomics portal: an open platform for exploring multidimensional cancer genomics data. Cancer Discov. 2012;2:401–4. https://doi.org/10.1158/2159-8290.CD-12-0095
Velasco R, Mussetti A, Villagrán-García M, Sureda A CAR T-cell-associated neurotoxicity in central nervous system hematologic disease: Is it still a concern? Front Neurol. 2023;14. Accessed December 27, 2023. https://www.frontiersin.org/articles/10.3389/fneur.2023.1144414
Ayuk F, Gagelmann N, von Tresckow B, Wulf G, Rejeski K, Stelljes M, et al. Real-world results of CAR T-cell therapy for large B-cell lymphoma with CNS involvement: a GLA/DRST study. Blood Adv. 2023;7:5316–9. https://doi.org/10.1182/bloodadvances.2023010336
Yuen CA, Hsu JM, Van Besien K, Reshef R, Iwamoto FM, Haggiagi A, et al. Axicabtagene Ciloleucel in patients ineligible for ZUMA-1 because of CNS Involvement and/or HIV: A Multicenter Experience. J Immunother. 2022;45:254 https://doi.org/10.1097/CJI.0000000000000416
Park JH, Nath K, Devlin SM, Sauter CS, Palomba ML, Shah G, et al. CD19 CAR T-cell therapy and prophylactic anakinra in relapsed or refractory lymphoma: phase 2 trial interim results. Nat Med. 2023;29:1710–7. https://doi.org/10.1038/s41591-023-02404-6
Cortes-Bullich A, Perez A, Bachmeier C, Mhaskar R, Chavez JC, Shah B, et al. Outcomes of CD19 chimeric antigen receptor T cell therapy in patients with gastrointestinal tract involvement of large B cell lymphoma. Transpl Cell Ther. 2021;27:768.e1–768.e6. https://doi.org/10.1016/j.jtct.2021.05.018
Cook MR, Dorris CS, Makambi KH, Luo Y, Munshi PN, Donato M, et al. Toxicity and efficacy of CAR T-cell therapy in primary and secondary CNS lymphoma: a meta-analysis of 128 patients. Blood Adv. 2023;7:32–39. https://doi.org/10.1182/bloodadvances.2022008525
Alsouqi A, Ahmed G, Wang J, Cassanello G, Szabo A, Rojek AE, et al. Chimeric antigen receptor T-cell therapy in secondary central nervous system lymphoma: A multicenter analysis. Am J Hematol. 2024;99:1624–7. https://doi.org/10.1002/ajh.27354
Cederquist GY, Schefflein J, Devlin SM, Shah GL, Shouval R, Hubbeling H, et al. CNS bridging radiotherapy achieves rapid cytoreduction before CAR T-cell therapy for aggressive B-cell lymphomas. Blood Adv. 2024;8:5192–9. https://doi.org/10.1182/bloodadvances.2024013393
Kim DW, Lee G, Lee H, Mahal AR, Lam MB, Ng AK. Response to hyperfractionated accelerated radiotherapy in chemotherapy-refractory non-Hodgkin lymphoma. Leuk Lymphoma. 2020;61:1428–34. https://doi.org/10.1080/10428194.2020.1719096
Saifi O, Breen WG, Lester SC, Rule WG, Stish B, Rosenthal A, et al. Does bridging radiation therapy affect the pattern of failure after CAR T-cell therapy in non-Hodgkin lymphoma? Radiother Oncol J Eur Soc Ther Radio Oncol. 2022;166:171–9. https://doi.org/10.1016/j.radonc.2021.11.031
Møller MB, Pedersen NT, Christensen BE. Diffuse large B-cell lymphoma: clinical implications of extranodal versus nodal presentation-a population-based study of 1575 cases. Br J Haematol. 2004;124:151–9. https://doi.org/10.1046/j.1365-2141.2003.04749.x
Otter R, Gerrits WB, vd, Sandt MM, Hermans J, Willemze R. Primary extranodal and nodal non-Hodgkin’s lymphoma. A survey of a population-based registry. Eur J Cancer Clin Oncol. 1989;25:1203–10. https://doi.org/10.1016/0277-5379(89)90416-1
Radke J, Ishaque N, Koll R, Gu Z, Schumann E, Sieverling L, et al. The genomic and transcriptional landscape of primary central nervous system lymphoma. Nat Commun. 2022;13:2558 https://doi.org/10.1038/s41467-022-30050-y
Weissinger SE, Dugge R, Disch M, Barth TF, Bloehdorn J, Zahn M, et al. Targetable alterations in primary extranodal diffuse large B-cell lymphoma. EJHaem. 2022;3:688–97. https://doi.org/10.1002/jha2.428
Ngo VN, Young RM, Schmitz R, Jhavar S, Xiao W, Lim KH, et al. Oncogenically active MYD88 mutations in human lymphoma. Nature. 2011;470:115–9. https://doi.org/10.1038/nature09671
Bolen CR, Klanova M, Trneny M, Sehn LH, He J, Tong J, et al. Prognostic impact of somatic mutations in diffuse large B-cell lymphoma and relationship to cell-of-origin: data from the phase III GOYA study. Haematologica. 2020;105:2298–307. https://doi.org/10.3324/haematol.2019.227892
Ma G, Gao Y, Jing X, He C, Liu H, Wu X, et al. Targeted sequencing reveals the relationship between mutations and patients’ clinical indicators, blood cell counts and early progression in diffuse large-B cell lymphoma. Leuk Lymphoma. 2023;64:140–50. https://doi.org/10.1080/10428194.2022.2131427
Acknowledgements
The reported research was supported in part by the National Institutes of Health/National Cancer Institute (NIH/NCI) award number P01CA023766 and Memorial Sloan Kettering Cancer Center Support Grant (P30 CA008748). RS reports grant support from an NIH-NCI K08CA282987, the Long Island Sound Chapter, Swim Across America, The Lymphoma Research Foundation, and Comedy vs. Cancer. The work of AL has been funded by the grant FEHH/Gilead 2022 from the Spanish Society of Hematology. The work of MC has been funded by the grant Alfonso Martin Escudero 2022. The Graphical Abstract and Fig. 1 were created using Biorender.
Author information
Authors and Affiliations
Contributions
AL, SD, MAP, RS: literature research, interpretation of data, conceived and designed the manuscript topics to be covered as well as conception of figures; SD: designed and supervised the statistical methods behind the work. SD, JF, RS, KR: performed the statistical analysis and data visualization. AL, RS: writing the manuscript and abstract. KR, MC, EL, AR, IL, GC, MG, SR, PD, RL, AP, MP, GS, MS, AA, DL, AB, HK, BI, GS, HP and MAP played an important role in interpreting results, revised the manuscript, approved the final version, and agreed to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Corresponding author
Ethics declarations
Competing interests
AL reports research funding from Gilead. KR: Kite/Gilead: Research Funding, Consultancy, Honoraria and travel support; Novartis: Honoraria; BMS/Celgene: Consultancy, Honoraria; Pierre-Fabre: travel support. MAP reports honoraria from Adicet, Allogene, Caribou Biosciences, Celgene, Bristol-Myers Squibb, Equilium, Exevir, ImmPACT Bio, Incyte, Karyopharm, Kite/Gilead, Merck, Miltenyi Biotec, MorphoSys, Nektar Therapeutics, Novartis, Omeros, OrcaBio, Sanofi, Syncopation, Takeda, VectivBio AG, and Vor Biopharma. He serves on DSMBs for Cidara Therapeutics and Sellas Life Sciences, and the scientific advisory board of NexImmune. He has ownership interests in NexImmune, Omeros and OrcaBio. He has received institutional research support for clinical trials from Allogene, Incyte, Kite/Gilead, Miltenyi Biotec, Nektar Therapeutics, and Novartis. RS: Received speaker honorarium from Incyte and Sanofi. GS Consultancy: AbbVie, BeiGene, BMS, Debiopharm, Epizyme, Genentech/Roche, Genmab, Innate Pharma, Incyte, Ipsen, Janssen, Kite/Gilead, Merck, Modex, Molecular Partners, Nurix, Orna Therapeutics, Pfizer, Treeline. Research funding: AbbVie, Genentech, Genmab, Janssen, Ipsen, and Nurix. MS served as a paid consultant for McKinsey & Company, Angiocrine Bioscience, Inc., and Omeros Corporation; received research funding from Angiocrine Bioscience, Inc., Omeros Corporation, Amgen Inc., Bristol Myers Squibb, and Sanofi; served on ad hoc advisory boards for Kite – A Gilead Company, and Miltenyi Biotec; and received honoraria from i3Health, Medscape, CancerNetwork, and IDEOlogy. The rest of co-authors declare no COIs.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This work has been presented in part as an oral presentation at the 2024 annual meeting of the European Blood and Marrow Transplantation.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Luna, A., Devlin, S.M., Rejeski, K. et al. CAR T-cell therapy response varies by extranodal disease site in large B-cell lymphoma. Blood Cancer J. 15, 64 (2025). https://doi.org/10.1038/s41408-025-01273-1
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41408-025-01273-1
This article is cited by
-
Anatomical patterns at first confirmed relapse or refractory disease predict outcomes in diffuse large B-cell lymphoma
International Journal of Hematology (2025)