Abstract
Chronic graft versus host disease is a major cause of morbidity after allogeneic haematopoietic cell transplantation. Belumosudil has recently been approved for the treatment of cGVHD refractory after two lines of treatment. However, few data are available to evaluate its efficacy and safety in real life. 68 patients with cGVHD received belumosudil through a compassionate access program in France. The median follow-up was 337 days from belumosudil initiation. Eighty-two percent of patients had severe cGVHD with a median of three organs involved. Patients had received a median of three prior treatment lines. Median treatment duration was 251 days. The best overall response rate (ORR) was 57.3%, including 14.7% complete remission (CR) and 42.6% partial response (PR). The ORR at three and six months was 47% and 45.6%, respectively. Liver and mouth involvement showed the highest response rates (72.7% and 70.4%), while lung involvement had the lowest (17.2%). Median failure-free survival (FFS) was not reached, with 6- and 12-month FFS rates of 89.1% and 80.4%, respectively. Nine patients died, mainly from GVHD (n = 5). Ten adverse events were reported, leading to treatment discontinuation in three cases. These results support the efficacy and safety of belumosudil in refractory cGVHD.
Similar content being viewed by others
Introduction
Allogeneic hematopoietic cell transplantation (alloHCT) is a cornerstone treatment for many hematologic malignancies and non-malignant disorders [1, 2]. AlloHCT can induce durable remissions and even cure hematopoietic neoplasms, owing to the graft-versus-leukemia (GVL) effect mediated by donor immune cells [3]. However, this curative potential is offset by the development of acute or chronic graft-versus-host disease (GVHD), characterized by the development of tissue damages induced by donor immune cells [4, 5]. Chronic GVHD (cGVHD) develops in 30–70% of patients after transplantation and remains one of the leading causes of late morbidity after alloHCT [6,7,8]. cGVHD is characterized by immune dysregulation, chronic inflammation, and tissue fibrosis with abnormal tissue repair, and can affect multiple organs, leading to significant disability and decreased quality of life [9].
The pathogenesis of cGVHD is complex and multifaceted. It involves a dysregulated interplay between donor-derived immune cells and host tissues, perpetuated by aberrant T-cell activation, B-cell dysfunction, and persistent inflammation. Acute inflammation associated with conditioning regimen, previous treatment, acute GvHD and interaction between host and microbes is considered as the first step that promote an abnormal B and T lymphopoiesis, leading to decreased production of regulatory cell and increased production of alloreactive donor-derived T and B cells [10,11,12,13,14]. Recent studies have highlighted the importance of the crosstalk between follicular helper T cell and germinal center B cell through IL-21 production, in the initiation of T and B-mediated alloreactive response in tissue [15, 16]. This process is thought to be directly involved in the peripheral activation of tissue macrophages by IL-17 and IL-21, leading to TGF-β production and tissue fibrosis [17, 18]. Central to this process is the role of Rho-associated coiled-coil kinase 2 (ROCK2), a key regulator of immune cell activity, cytokine production, and tissue fibrosis. ROCK2 modulates T-cell polarization, promoting a pro-inflammatory environment while impairing the balance of regulatory T cells (Tregs) and effector T cells. Furthermore, ROCK2 contributes to the activation of fibroblasts and the deposition of extracellular matrix, hallmark features of the fibrotic process in cGVHD. These insights have highlighted ROCK2 as a critical therapeutic target to address both the immune dysregulation and fibrotic remodeling that characterize cGVHD [19].
Standard first-line therapy for cGVHD consists of systemic corticosteroids, often combined with calcineurin inhibitors [20]. However, up to 50% of patients will develop corticosteroids-refractory cGVHD, requiring the use of additional systemic therapies [21, 22]. The management of steroid-refractory or multi-line refractory cGVHD remains challenging, with limited therapeutic options and suboptimal outcomes. Recently, the Reach3 clinical trial has demonstrated the interest of ruxolitinib, a Jak1/2 inhibitor, for steroid-resistant cGVHD, but fail to induce a complete or partial response in near to 50% of patients [23]. In addition, axatilimab, a CSF-R inhibitory monoclonal antibody, has recently demonstrated its safety and efficacy on the same population in a phase 2 clinical trial [24]. Recent advances in our understanding of cGVHD pathophysiology have also driven the development of novel targeted therapies, such as ROCK2 inhibitors.
Belumosudil, an oral selective ROCK2 inhibitor, represents a promising therapeutic advance for patients with refractory cGVHD. Its mechanism of action addresses key pathological processes in cGVHD by modulating T-cell responses, promoting the balance of pro-inflammatory and regulatory immune pathways, and reducing fibrosis [25]. Clinical evidence supporting the efficacy and safety of belumosudil comes primarily from two prospective studies, including the pivotal ROCKstar trial, which demonstrated an overall response rate (ORR) of 74% in patients with cGVHD who had received at least two prior lines of therapy [26, 27]. Responses were observed across multiple organ systems, including bronchiolitis obliterans during lung GvHD [28]. The durability of responses and a favorable safety profile further underscored its potential as a valuable addition to the therapeutic armamentarium for cGVHD.
In France, belumosudil became accessible to patients through a compassionate use program in 2022, offering an opportunity to assess its efficacy and safety in a real-world setting. This retrospective, multicenter analysis evaluates the outcomes, the efficacy and the safety of 68 patients with refractory cGVHD treated with belumosudil under this program. By examining the French experience, this study aims to contribute valuable real-world data to the growing body of evidence on belumosudil and its role in managing treatment-refractory cGVHD.
Patients and methods
Patients
Patients (n = 68) were included in the compassionate access program for belumosudil at the request of their referring physician between April 2022 and September 2024 from sixteen different transplantation centers in France, following verification of eligibility criteria by the French National Agency for Medicines and Health Products Safety (ANSM). The inclusion criteria were: age over 12 years, weight over 40 kg, performance status above 60%, diagnosis of moderate to severe chronic Graft-versus-Host Disease (cGVHD) according to NIH criteria [29], and having received at least two prior lines of treatment. Corticosteroid doses had to be stable for at least two weeks and below 1 mg/kg. Patients could not have signs of hematologic malignancy relapse or post-transplant lymphoproliferative disorder (PTLD) at treatment initiation, cytopenia (platelet count <50 G/L or neutrophil count <1.5 G/L), chronic renal insufficiency defined by a glomerular filtration rate (eGFR) below 30 mL/min/1.73 m², liver cytolysis (defined as ASAT or ALAT levels greater than three times the upper normal limit (ULN)), or bilirubin levels exceeding 1.5 ULN. Active viral infections with hepatitis B, hepatitis C, or HIV, and secondary cancers (except basal cell carcinoma) were also exclusion criteria. Women could not be pregnant or breastfeeding, and all participants had to agree to use contraception. Patient data were extracted from the EBMT Data registry (European Blood and Marrow Transplantation) and systematically reviewed and updated by their referring physician. Data were pseudonymized, and the following information was collected: donor and recipient age, weight, height, initial hematologic disease and pre-transplant status, donor and recipient CMV serology status, donor and recipient blood group, conditioning regimen, GVHD prophylaxis, stage and grade of acute and chronic GVHD, prior chronic GVHD treatments and response to each line according to NIH criteria, start and end dates of belumosudil, treatment response at 3 months, 6 months, relapse and status at the last follow-up. Treatment-related toxicities were documented as part of the compassionate access program and reported by clinicians.
Ethics approval and consent to participate
All patients gave a written informed consent that was validated by an ethical committee (IRB00003888) and by the Commission National Informatique et Liberté (number 2207763). This study was conducted in accordance with the Declaration of Helsinki, and was reviewed and approved by the scientific committee of the Société Francophone de Greffe de Moelle et de Thérapie Cellulaire (SFGM-TC) (number R2307) and declared to the Health Data Hub (F20231208135438).
Treatment
Patients received belumosudil at a dose of 200 mg per day. This dose was increased to 200 mg twice daily if co-administered with a proton pump inhibitor. The treatment could be combined with previously ongoing other immunosuppressive drugs, but the doses of these other treatments had to be stable for at least 15 days, to be considered in failure by the referring physician (stable disease or progression). Adverse events occurring under belumosudil were reported by the referring physicians irrespectively of the presumed relation to the treatment. Belumosudil treatment was maintained until failure (progression of chronic GVHD or initiation of a new treatment line), toxicity/intolerance, or decision to discontinue by the referring physician.
Response assessment and efficacy endpoint
Response to treatment and organ-specific cGvHD assessment were performed according to the NIH criteria at each line of treatment before belumosudil, at belumosudil onset and during follow-up at 3 and 6 months of treatment and at last follow-up. Best response was defined by the best overall grading or organ-specific response obtained at any time after treatment. Complete response was defined by the resolution of any sign of cGVHD, partial response by the decreased staging of at least one involved organ, stable disease by the absence of changes in organ-specific staging and progression by the increased organ-specific staging or the involvement of a new organ. Overall response rate was defined by the sum of complete and partial response. Time to response was defined by the time between belumosudil onset and the best response (CR or PR), duration of response (DOR) was defined by the time from the best response to progression of cGvHD or new line of treatment. Overall Survival (OS) was defined as time from belumosudil onset to death from any cause. Failure free survival (FFS) was defined from the date of belumosudil onset to death, relapse, progression of cGVHD or introduction of a new line of treatment. Relapse was defined by ≥5% bone marrow blasts or reappearance of the underlying disease.
Statistical analysis
Categorical and continuous variables were analyzed using Fisher’s exact and Kruskal–Wallis tests, respectively. Survival probabilities (OS, FFS) were calculated using the Kaplan–Meier method. Univariate analyses were performed using the log-rank test for FFS [30], with a significance at p < 0.05. Analyses were conducted with R software (version 4.1.2), survfit2 and survminer packages, data visualization were performed with ggplot2 and networkD3 [31]. Figure were prepared with InkScape v1.4.
Results
Patients’ characteristic
Between April 2022 and September 2024, 68 patients received belumosudil in France through the compassionate access program. Median follow-up from transplantation date was 46 months and median follow-up from belumosudil onset was 337 days (11–830). Most patients were male (n = 46, 67%), and 15 patients had gender mismatch with donors (female into male, n = 15, 22%). Most patients underwent an alloHCT for a hematologic malignancy, mainly for acute leukemia (n = 34, 50%), non-Hodgkin lymphoma (n = 10, 15%), myeloproliferative neoplasm (n = 8, 12%) and myelodysplastic syndrome (n = 7, 10%). Reduced conditioning regimen was used in 63% of cases. GVHD prophylaxis was mainly based on the combination of ciclosporin A (CSA) with methotrexate (n = 26, 39%), mycophenolate mofetil (n = 27, 41%) or mycophenolate mofetil, post-transplant cyclophosphamide (n = 11, 17%) and 28 patients (41%) received ATG. Acute GVHD was diagnosed in 71% of patients, among which grade II-IV aGVHD and grade III-IV represented 61% (n = 41) and 28.4% (n = 19) of patients, respectively. Full patients’ characteristics are detailed in Table 1.
cGVHD characteristics and treatments
Chronic GVHD characteristics and treatments are described in Table 2. The median delay between transplantation and cGVHD onset was 6 months (1–50). Most patients had moderate or severe cGVHD at onset of symptoms (n = 64, 94%). First line therapy was mostly based on corticosteroids alone (n = 35, 51%) or in combination with other drugs (n = 23, 34%), mainly including calcineurin inhibitors or extracorporeal photopheresis. Overall, patients received a median number of 3 lines of treatment before belumosudil onset. All patients received steroids and most patients were exposed to ruxolitinib at any line of treatment (n = 65, 95.5%), followed by ECP (n = 36, 53%) and MMF (n = 16, 23.5%). Median delay between cGVHD diagnosis and belumosudil onset was 21.5 months. At belumosudil onset, most patients had severe cGVHD (n = 56, 82%) with a median of 3 organs involved (1–5), the most frequently being skin, mouth, eyes and lung. 66% of patients received belumosudil at 200 mg BID due to the combination with proton pump inhibitors. Median duration of treatment was 251 days (11–830). 27 patients stopped treatment, mainly due to stable disease or progression (n = 12, 44.5%). The median delay to progression or new line of treatment was 140 days (22–506) (Table 3).
Survival and response to belumosudil
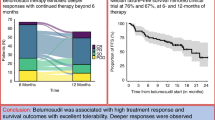
Median overall survival (OS) from transplantation date was not reached and 6-, 12- and 48-months OS were 97.1%, 95.6% and 90.6% respectively. Six-months and one-year overall survival from belumosudil onset were 89.1% and 84.7%, respectively (Fig. 1A). Failure free survival (FFS) at 6 and 12 months were 89.1% and 80.4% respectively and median FFS was not reached (Fig. 1B). FFS was not significantly different between patients receiving belumosudil in third line of treatment and those with more than 3 lines of treatment (Fig. 1C). Best overall response rate (ORR) was 57.3% with 14.7% of complete remission (CR) and 42.6% of partial response (PR). The median delay from treatment to best response was 183 days (14–560). ORR at 3 months was 47% with 42.6% experiencing PR and 4.4% with CR. ORR at 6 months was 45.6% with 35.3% experiencing PR and 10.3% with CR (Fig. 1D). At 6 months, only 19.1% and 35.3% of patients had moderate and severe cGVHD, respectively (Fig. 1E). All patients with CR maintained this response until the end of the follow-up. Among patients with stable or progressive disease at 3 months (n = 29, 42.7%), 9 reached partial or complete response at 6 months (31%) (Fig. 1F). Best response by organ is represented in Fig. 1G. Liver and mouth cGVHD were associated with the best response rate, with an ORR of 72.7% and 70.4% respectively. Conversely, lung was associated with the lowest response rate (17.2%).
a Overall survival from belumosudil onset (OS). b Failure-free survival (FFS) for the entire cohort (n = 68) with a median follow-up of 337 days. c FFS for patients receiving belumosudil as a third-line therapy (yellow) or after 3 lines of treatment (blue). Statistical analyses were performed at one year using a log-rank test and a significance defined for p < 0.05. d Stacked histogram with response to belumosudil at 3 months, 6 months and best response during follow-up. e Sankey diagram depicting the evolution of cGVHD grading according to NIH criteria, at GVHD onset, belumosudil onset, 3 months and 6 months. f Sankey diagram illustrating the evolution of response at 3 and 6 months, according to initial cGVHD grading at diagnosis and belumosudil onset. g Stacked diagram with best response per organ.
Safety and adverse event
Overall, 9 patients died, mainly from GVHD (n = 5). Other causes of death were secondary neoplasm (n = 1), relapse of acute myeloid leukemia (n = 1), severe bacterial infection (n = 1) and cardiac arrythmia (n = 1). Only 2 patients relapsed after belumosudil onset from the primary disease and one patient died of relapse. Ten adverse events were reported by physicians (Table 4). None of them were associated with death, but belumosudil was stopped in 3 cases (cholecystitis, diarrhea and one case of severe kidney insufficiency due to acute tubular necrosis). After belumosudil stop, a new line of treatment was started for 16 patients, including lung transplantation for severe bronchiolitis obliterans (n = 1), etanercept (n = 1), ATG (n = 1), methotrexate (n = 1), MMF (n = 1), dasatinib (n = 1), ibrutinib (n = 3), ruxolitinib (n = 4) or ECP (n = 3).
Discussion
Chronic GVHD is a frequent and severe complication following alloHCT, which not only jeopardizes the survival of patients but also significantly impacts their quality of life in all its aspects [32]. In addition, cGVHD management raised the question of economic burden and of cost-effectiveness analyses of new therapeutic strategies [33, 34]. Although significant progress has been made in patient care, particularly in managing complications associated with chronic GVHD, the development of new targeted curative treatments remains a major challenge in patient management [20, 35]. In the past decade, new drugs have emerged for treating corticosteroid-resistant forms, notably ruxolitinib and ibrutinib, which have been approved by the FDA for this indication, and even more recently with axatilimab [24, 36]. However, with 50% of cases being corticosteroid-resistant, ruxolitinib achieved an overall response rate of only 49.7% at 24 months, with a failure-free survival (FFS) of approximately 60% [23]. Thus, nearly half of the patients receiving ruxolitinib for chronic GVHD will require the introduction of a third-line treatment.
In this study, we reported multicenter real-world data from the compassionate access program (CAP) of belumosudil in France among 68 patients who had received at least two prior lines of treatment for chronic GVHD. Our data show that belumosudil is a safe therapeutic option, achieving an overall response in 57.3% of patients. Currently, few real-world data are available to evaluate the efficacy and safety of belumosudil. In this study, patients accessed the treatment under a CAP, with eligibility criteria similar to those of the ROCKstar trial. Overall, patients had severe chronic GVHD and many treatment lines had failed previously. Nevertheless, a comparison of patients who received belumosudil as a third-line treatment with those who received more than three lines shows comparable treatment efficacy in terms of FFS. These results suggest that belumosudil remains an effective therapeutic alternative, even for heavily pretreated patients.
Two prospective phase 2 trials have evaluated the efficacy of belumosudil in patients with chronic GVHD. The first phase IIa trial, KD025-208, assessed the efficacy and safety of belumosudil in 54 patients who had received 1–3 prior lines of treatment for chronic GVHD, comparing three different dosing regimens: 200 mg once daily, 200 mg twice daily (BID), and 400 mg once daily [27]. This trial achieved an overall response rate of 65% and showed that the 200 mg daily dose was safe and as effective as the other two arms of the trial. A second prospective phase 2 trial, ROCKstar, evaluated belumosudil in 132 patients who had received 2–5 lines of treatment, comparing two dosing regimens of 200 mg daily and 200 mg BID [26]. In this trial, the best overall response rates were 74% and 77% for the two experimental arms, respectively. Our results are therefore similar in terms of ORR, though slightly lower. This difference can be attributed to the severity of the patients from the CAP, in which 82.4% of patients had a severe disease, with 36.7% presenting with pulmonary involvement. Recently, these results have been confirmed by a retrospective real-life study from Germany and Switzerland on 33 patients, with an ORR of 42% [37].
One of the major current challenges in the curative management of chronic GVHD is the prevention of tissue fibrosis lesions, which are responsible for the severity of chronic GVHD and its impact on quality of life. Due to its dual immunosuppressive and anti-fibrotic mechanisms, Rock2 inhibition is a relevant strategy to prevent the onset of tissue fibrosis lesions [19, 25]. Recently, a study reanalyzed the organ-specific response data from both phase II trials [28]. Among 59 patients who developed bronchiolitis obliterans syndrome (BOS), the overall response rate was 32%, which was primarily associated with the severity of pulmonary involvement, suggesting that early intervention with belumosudil could effectively block the onset of fibrosis lesions. Currently, a phase 3 trial is ongoing to evaluate the benefit of combining corticosteroids with belumosudil versus corticosteroids alone as a first-line treatment (NCT06143891). In our cohort, response rates for pulmonary involvement were lower, with an ORR of 17.4%. However, stage 3 pulmonary involvement affected 15 out of 25 patients with BOS, which may also explain the lower ORR. Among these 15 patients, only one experienced a partial response in pulmonary staging, while three out of ten patients with stage 1 or 2 pulmonary involvement improved their pulmonary severity scores. Additionally, we did not had access to Lee Symptom Scale (LSS) in this cohort, but a recent publication based on the two phase 2 trial of belumosudil revealed a good correlation between patient-reported outcome and NIH response criteria [38].
Finally, our data confirmed the safety profile associated with the use of belumosudil. Although our data may be biased by underreporting of adverse events by physicians, the compassionate access program involved heightened monitoring of adverse events by the French Medicines Agency (ANSM) and has not identified any safety alerts related to the use of belumosudil to date. These findings are corroborated by high survival rates and a limited number of deaths among heavily pretreated patients. In our series, no deaths could be attributed to adverse effects of belumosudil, with chronic GVHD being the main cause of death. However, the safety data for belumosudil are still recent, and constant monitoring for potential toxicities is essential as this treatment becomes increasingly used outside of clinical trials.
In conclusion, our real-world data confirm the efficacy of belumosudil for treating patients who have already received at least two lines of treatment. These findings also confirm the safety of this treatment. With the emergence of new targeted therapies for chronic GVHD, the question arises as to the therapeutic strategy and the place of these treatments within the broader context of available options. Currently, corticosteroids remain the reference as first-line treatment, although new therapeutic combinations with other targeted therapies that could early reduce fibrotic lesions would be relevant to evaluate. In cases of corticosteroid resistance, the reference treatment is ruxolitinib, based on the results of the REACH3 trial. In case of ruxolitinib failure, three targeted drugs are available, including belumosudil, ibrutinib, and axatilimab. The choice between these three drugs can be guided by tolerance, efficacy, and the mode of administration. Although no comparative studies are currently available, new prospective phase 3 studies should soon help optimize the management algorithm for patients with chronic GVHD after alloHCT.
Data availability
Raw data and source code to reproduce the analyses, figures, and tables described in this manuscript are available on a Git repository: https://github.com/TIgit-lab/Belumosudil/tree/main.
References
Passweg JR, Baldomero H, Ciceri F, de la Cámara R, Glass B, Greco R, et al. Hematopoietic cell transplantation and cellular therapies in Europe 2022. CAR-T activity continues to grow; transplant activity has slowed: a report from the EBMT. Bone Marrow Transpl. 2024;59:803–12.
McDonald GB, Sandmaier BM, Mielcarek M, Sorror M, Pergam SA, Cheng G-S, et al. Survival, Nonrelapse Mortality, and Relapse-Related Mortality After Allogeneic Hematopoietic Cell Transplantation: Comparing 2003-2007 Versus 2013-2017 Cohorts. Ann Intern Med. 2020;172:229–39.
Negrin RS. Graft-versus-host disease versus graft-versus-leukemia. Hematol Am Soc Hematol Educ Program. 2015;2015:225–30.
Hill GR, Betts BC, Tkachev V, Kean LS, Blazar BR. Current Concepts and Advances in Graft-Versus-Host Disease Immunology. Annu Rev Immunol. 2021;39:19–49.
Socie G, Michonneau D. Milestones in acute GVHD pathophysiology. Front Immunol. 2022;13:1079708.
Langer R, Lelas A, Rittenschober M, Piekarska A, Sadowska-Klasa A, Sabol I et al. Retrospective analysis of the incidence and outcome of late acute and chronic graft-versus-host disease—an analysis from transplant centers across Europe. Front Transplant. 2024;3. https://doi.org/10.3389/frtra.2024.1332181.
Kitko CL, Pidala J, Schoemans HM, Lawitschka A, Flowers ME, Cowen EW, et al. National Institutes of Health Consensus Development Project on Criteria for Clinical Trials in Chronic Graft-versus-Host Disease: IIa. The 2020 Clinical Implementation and Early Diagnosis Working Group Report. Transpl Cell Ther. 2021;27:545–57.
Bachier CR, Aggarwal SK, Hennegan K, Milgroom A, Francis K, Dehipawala S, et al. Epidemiology and Treatment of Chronic Graft-versus-Host Disease Post-Allogeneic Hematopoietic Cell Transplantation: A US Claims Analysis. Transplant Cell Ther. 2021;27:504.e1–504.e6.
Zeiser R, Blazar BR. Pathophysiology of Chronic Graft-versus-Host Disease and Therapeutic Targets. N. Engl J Med. 2017;377:2565–79.
Matsuoka K, Kim HT, McDonough S, Bascug G, Warshauer B, Koreth J, et al. Altered regulatory T cell homeostasis in patients with CD4+ lymphopenia following allogeneic hematopoietic stem cell transplantation. J Clin Invest. 2010;120:1479–93.
Greinix HT, Pohlreich D, Kouba M, Körmöczi U, Lohmann I, Feldmann K, et al. Elevated Numbers of Immature/Transitional CD21− B Lymphocytes and Deficiency of Memory CD27+ B Cells Identify Patients with Active Chronic Graft-versus-Host Disease. Biol Blood Marrow Transplant. 2008;14:208–19.
Sarantopoulos S, Stevenson KE, Kim HT, Cutler CS, Bhuiya NS, Schowalter M, et al. Altered B-cell homeostasis and excess BAFF in human chronic graft-versus-host disease. Blood. 2009;113:3865–74.
Poe JC, Fang J, Zhang D, Lee MR, DiCioccio RA, Su H, et al. Single-cell landscape analysis unravels molecular programming of the human B cell compartment in chronic GVHD. JCI Insight. 2023;8:e169732.
Bordenave J, Gajda D, Michonneau D, Vallet N, Chevalier M, Clappier E, et al. Deciphering bone marrow engraftment after allogeneic stem cell transplantation in humans using single-cell analyses. J Clin Invest. 2024;134:e180331.
Flynn R, Du J, Veenstra RG, Reichenbach DK, Panoskaltsis-Mortari A, Taylor PA, et al. Increased T follicular helper cells and germinal center B cells are required for cGVHD and bronchiolitis obliterans. Blood. 2014;123:3988–98.
Forcade E, Kim HT, Cutler C, Wang K, Alho AC, Nikiforow S, et al. Circulating T follicular helper cells with increased function during chronic graft-versus-host disease. Blood. 2016;127:2489–97.
Alexander KA, Flynn R, Lineburg KE, Kuns RD, Teal BE, Olver SD, et al. CSF-1-dependant donor-derived macrophages mediate chronic graft-versus-host disease. J Clin Invest. 2014;124:4266–80.
Hill GR, Olver SD, Kuns RD, Varelias A, Raffelt NC, Don AL, et al. Stem cell mobilization with G-CSF induces type 17 differentiation and promotes scleroderma. Blood. 2010;116:819–28.
Flynn R, Paz K, Du J, Reichenbach DK, Taylor PA, Panoskaltsis-Mortari A, et al. Targeted Rho-associated kinase 2 inhibition suppresses murine and human chronic GVHD through a Stat3-dependent mechanism. Blood. 2016;127:2144–54.
Penack O, Marchetti M, Aljurf M, Arat M, Bonifazi F, Duarte RF, et al. Prophylaxis and management of graft-versus-host disease after stem-cell transplantation for haematological malignancies: updated consensus recommendations of the European Society for Blood and Marrow Transplantation. Lancet Haematol. 2024;11:e147–e159.
Pidala J, Onstad L, Martin PJ, Hamilton BK, Cutler C, Kitko CL, et al. Initial therapy for chronic graft-versus-host disease: analysis of practice variation and failure-free survival. Blood Adv. 2021;5:4549–59.
Chen GL, Onstad L, Martin PJ, Carpenter P, Pidala J, Arai S, et al. Durable discontinuation of systemic therapy in patients affected by chronic graft-versus-host disease. Haematologica. 2023;108:483–9.
Zeiser R, Polverelli N, Ram R, Hashmi SK, Chakraverty R, Middeke JM, et al. Ruxolitinib for Glucocorticoid-Refractory Chronic Graft-versus-Host Disease. N Engl J Med. 2021;385:228–38.
Wolff D, Cutler C, Lee SJ, Pusic I, Bittencourt H, White J, et al. Axatilimab in Recurrent or Refractory Chronic Graft-versus-Host Disease. N Engl J Med. 2024;391:1002–14.
Zanin-Zhorov A, Blazar BR. ROCK2, a critical regulator of immune modulation and fibrosis has emerged as a therapeutic target in chronic graft-versus-host disease. Clin Immunol. 2021;230:108823.
Cutler C, Lee SJ, Arai S, Rotta M, Zoghi B, Lazaryan A, et al. Belumosudil for chronic graft-versus-host disease after 2 or more prior lines of therapy: the ROCKstar Study. Blood. 2021;138:2278–89.
Jagasia M, Lazaryan A, Bachier CR, Salhotra A, Weisdorf DJ, Zoghi B, et al. ROCK2 Inhibition With Belumosudil (KD025) for the Treatment of Chronic Graft-Versus-Host Disease. J Clin Oncol. 2021;39:1888–98.
DeFilipp Z, Kim HT, Yang Z, Noonan J, Blazar BR, Lee SJ, et al. Clinical response to belumosudil in bronchiolitis obliterans syndrome: a combined analysis from 2 prospective trials. Blood Adv. 2022;6:6263–70.
Jagasia MH, Greinix HT, Arora M, Williams KM, Wolff D, Cowen EW, et al. National Institutes of Health Consensus Development Project on Criteria for Clinical Trials in Chronic Graft-versus-Host Disease: I. The 2014 Diagnosis and Staging Working Group report. Biol Blood Marrow Transpl. 2015;21:389–401.e1.
Fine JP, Gray RJ. A Proportional Hazards Model for the Subdistribution of a Competing Risk. J Am Stat Assoc. 1999;94:496–509.
R: The R Project for Statiscal Computing. 2023. Available from: https://www.r-project.org/.
Fiuza-Luces C, Simpson RJ, Ramírez M, Lucia A, Berger NA. Physical function and quality of life in patients with chronic GvHD: a summary of preclinical and clinical studies and a call for exercise intervention trials in patients. Bone Marrow Transpl. 2016;51:13–26.
Yalniz FF, Murad MH, Lee SJ, Pavletic SZ, Khera N, Shah ND, et al. Steroid Refractory Chronic Graft-Versus-Host Disease: Cost-Effectiveness Analysis. Biol Blood Marrow Transpl. 2018;24:1920–7.
Michonneau D, Quignot N, Jiang H, Reichenbach D, Kelly M, Burrell A, et al. Clinical and economic burden associated with graft-versus-host disease following allogeneic hematopoietic cell transplantation in France. Bone Marrow Transpl. 2023;58:514–25.
Salhotra A, Sandhu K, O’Hearn J, Ali H, Nakamura R, Modi BG. A critical review of belumosudil in adult and pediatric patients with chronic graft-versus-host disease. Expert Rev Clin Immunol. 2023;19:241–51.
Martini DJ, Chen Y-B, DeFilipp Z. Recent FDA Approvals in the Treatment of Graft-Versus-Host Disease. Oncologist. 2022;27:685–93.
Heidenreich S, Egger-Heidrich K, Halter JP, Jost L, Stölzel F, Perl M et al. Safety and efficacy of the ROCK-2-inhibitor Belumosudil in cGvHD treatment - a retrospective, German-Swiss multicenter real-world data analysis. Bone Marrow Transplant. 2025. https://doi.org/10.1038/s41409-024-02507-9.
Lee SJ, Cutler C, Blazar BR, Tu A, Yang Z, Pavletic SZ. Correlation of Patient-Reported Outcomes with Clinical Organ Responses: Data from the Belumosudil Chronic Graft-versus-Host Disease Studies. Transplant Cell Ther. 2022;28:700.e1–700.e6.
Acknowledgements
The authors thanks all physicians and nurses who contributed to health care, the Société Francophone de Greffe de Moelle et de Thérapie Cellulaire (SFGM-TC) for its support, and all patients and their families. No funding or financial support were provided for this study.
Author information
Authors and Affiliations
Contributions
DM designed the study; DM, FM, SLG, LM, MDA, JJT, NV, MC, NM, CCL, FS, QC, AH, ALM, EF, HLW, NR, and ML provided patients’ data, DM collected data, DM and SLG analyzed data and interpreted results, DM and SLG prepared figures, DM wrote the original version of the manuscript. All authors contributed to reviewing and editing the manuscript and approved the final version.
Corresponding author
Ethics declarations
Competing interests
DM received research grants from Novartis, Sanofi and CSL Behring, consulting fees from Novartis, Incyte, Sanofi, Jazz Pharmaceuticals, and Mallinckrodt. FM received honoraria from Therakos/Mallinckrodt, Janssen, Biocodex, Sanofi, JAZZ Pharmaceuticals, Gilead, Novartis, Priothera, and Astellas. EF received speaker fees and honoraria for advisory board from Novartis, Sanofi, Gilead, Alexion, MSD, Astellas, Jazz, Pfizer, GSK and Sobi. ML received research grant from Janssen, consulting honoraria from Abbvie, Alexion, Bristol Myers Squibb, Gilead, Glaxo Smith Kline, Incyte, Jazz, Kartos, Mallinkrodt, Novartis, Roche, Sanofi, Takeda. Other authors did not declare any competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Michonneau, D., Malard, F., Le Grand, S. et al. Efficacy and safety of belumosudil for treatment of cGVHD: multicenter retrospective analysis of the French cohort of the compassionate use program, on behalf of the French Society of Bone Marrow Transplantation and Cellular Therapy. Bone Marrow Transplant 60, 779–786 (2025). https://doi.org/10.1038/s41409-025-02554-w
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41409-025-02554-w
This article is cited by
-
Real world experience using belumosudil for treatment of chronic graft versus host disease in children and young adults
Bone Marrow Transplantation (2026)
-
Efficacy and safety of belumosudil for refractory chronic graft-versus-host disease in routine practice
Annals of Hematology (2026)
-
Real-world experience of belumosudil and belumosudil/ruxolitinib combination in steroid-refractory chronic graft-versus-host disease
Bone Marrow Transplantation (2025)
-
Real-world Canadian data on belumosudil therapy in heavily pretreated patients with steroid-refractory chronic graft-versus-host disease: treatment outcomes and risk factor analysis for failure-free survival
Annals of Hematology (2025)