Abstract
Background
The optimal number of induction chemotherapy (IC) cycles for locoregionally advanced nasopharyngeal carcinoma (LA-NPC) is debated. This study investigates if early cell-free Epstein-Barr virus (cfEBV) DNA clearance can personalise treatment.
Methods
We included 1590 LA-NPC patients treated with IC+ chemoradiotherapy (2010–2023) with complete early cfEBV DNA data. COX regression identified independent prognostic factors, and receiver operating characteristic curves assessed predictive accuracy. Propensity score matching (PSM) balanced covariates between groups receiving different IC cycles. The primary outcome, progression-free survival (PFS) was analysed using Kaplan-Meier and log-rank tests.
Results
After the first IC cycle, 45.5% of patients had undetectable cfEBV DNA. Combining cfEBV DNA clearance, N-stage, and overall stage yielded the highest AUC for 5-year PFS (0.632). Patients were stratified into high- and low-risk groups (p < 0.001). Post-matching, low-risk patients receiving three IC cycles had better 5-year PFS than those receiving two (85.2% vs. 76.0%, p = 0.024), with no significant increase in grade 3–4 toxicities (30.36% vs. 24.29%, p = 0.157). High-risk patients showed no PFS benefit from additional cycles (63.2% vs. 61.0%, p = 0.960).
Discussion
Early cfEBV DNA clearance predicts LA-NPC outcomes. Low-risk patients may benefit from an additional cycle of IC, while high-risk patients may require alternative strategies like immunotherapy or earlier chemoradiotherapy.
Similar content being viewed by others
Introduction
Nasopharyngeal carcinoma (NPC), a malignancy arising from the nasopharyngeal epithelium, demonstrates distinct geographical clustering, with over 70% of the global cases concentrated in East and Southeast Asia [1, 2]. Clinically, NPC is characterised by an insidious onset, leading to delayed diagnosis. Consequently, approximately 60–70% of patients present with locoregionally advanced disease (LA-NPC; T3-4/N1–3/M0) at initial diagnosis [3]. Platinum-based concurrent chemoradiotherapy (CCRT) is the therapeutic cornerstone for LA-NPC, which leverages chemotherapy-induced radio-sensitisation to enhance local control [4,5,6]. However, distant metastasis persists as the predominant cause of treatment failure, accounting for 70% of cancer-specific mortality [7,8,9]. Therefore, induction chemotherapy (IC) prior to CCRT has been adopted to address micro-metastatic dissemination, with landmark Phase III trials demonstrating significant survival benefits [10,11,12]. IC followed by CCRT achieved absolute 5-year survival increments of 7.9–9.1% for overall survival (OS) [10,11,12], securing its position as a Category 1A recommendation in the National Comprehensive Cancer Network (NCCN) and Chinese Society of Clinical Oncology (CSCO) guidelines [13, 14].
Despite these advances, the optimal number of IC cycles remains unknown. Current protocols variably employ 2-4 cycles [11, 15, 16], whereas escalating cycles may paradoxically compromise outcomes. Additionally, prolonged IC administration increases the risk of radiotherapy delay, potentially accelerating tumour repopulation and compromising radiotherapy tolerance due to cumulative toxicities, a therapeutic postponement consistently associated with survival detriment [10, 17,18,19]. This underscores the need for personalised IC cycle selection.
Moreover, accurate prognostic stratification is pivotal for optimising therapeutic strategies. While TNM staging provides a foundational framework, plasma cell-free Epstein-Barr virus DNA (cfEBV DNA) has emerged as a validated biomarker for NPC [20, 21]. Consequently, pretreatment cfEBV DNA levels, reflecting tumour burden, are frequently integrated with TNM staging for risk stratification or guide IC cycle selection [22,23,24]. Nonetheless, static baseline measurements fail to capture dynamic treatment responses. Furthermore, although serial imaging offers insights into tumour volume changes, frequent radiographic assessments impose logistic and financial burdens [25]. In this regard, recent evidence suggests that dynamic cfEBV DNA kinetics during therapy may predict outcomes and elucidate tumour biology [26,27,28], offering a cost-effective alternative to imaging [29]. However, early-phase cfEBV DNA changes (e.g., post-first IC cycle) remain underexplored as predictive tools for IC cycle optimisation in LA-NPC.
Therefore, this study investigated the early dynamic cfEBV DNA changes (pretreatment and post-first IC cycle) in patients with LA-NPC receiving 2–3 IC cycles followed by CCRT, with or without adjuvant therapy (AT). We aimed to establish a biomarker-driven framework for personalised IC cycle selection by correlating these kinetics with survival outcomes, ultimately refining risk-adapted therapeutic strategies in this high-risk population.
Materials and methods
Patient selection
This retrospective study enroled consecutive patients with LA-NPC treated at Sun Yat-sen University Cancer Center (SYSUCC) between January 2013 and December 2023. The inclusion criteria were defined as follows: (1) histopathologically confirmed non-keratinising NPC classified as stage III–IVa (8th edition American Joint Committee on Cancer/Union for International Cancer Control TNM stage system) [30]; (2) completion of 2–3 cycles of platinum-based IC followed by definitive intensity-modulated radiotherapy (IMRT) with concurrent platinum-based chemotherapy (CCRT), and adjuvant chemotherapy permitted at clinicians’ discretion; (3) availability of pre- and post-first IC cycle plasma cfEBV DNA quantitation and pretreatment EBV DNA > 0 copies/mL; (4) absence of pregnancy, lactation, or history of synchronous/metachronous malignancies; and (5) preserved organ function (hematopoietic, hepatic, and renal). The patient enrolment workflow is summarised in eFig. 1.
All participants underwent standardised pretreatment evaluations, including comprehensive physical examination, EBV serological profiling, electrocardiography, nasopharyngeal fiberoptic endoscopy with biopsy, contrast-enhanced magnetic resonance imaging (MRI) of the nasopharynx and neck, chest/abdominal contrast-enhanced computed tomography (CT), and bone scan, or whole-body fluorodeoxyglucose positron emission tomography to exclude distant metastases.
Treatment
All patients included in this study received 2 or 3 cycles of IC combined with CCRT, with or without AC. The common IC regimens included: a) GP, consisting of gemcitabine (1 g/m² on days 1 and 8) and cisplatin (80 mg/m² on day 1); b) TPF, comprising docetaxel (60 mg/m² on day 1), cisplatin (60 mg/m² on day 1), and 5-fluorouracil (500–600 mg/m² on days 1–5); c) TP, consisting of docetaxel (75 mg/m² on day 1) and cisplatin (75 mg/m² on day 1); d) PF, consisting of 5-fluorouracil (800–1000 mg/m², 120-h continuous infusion) and cisplatin (75 mg/m² on day 1); e) TPC, comprising docetaxel (60 mg/m² on day 1), cisplatin (60 mg/m² on day 1), and capecitabine (1000 mg/m², twice daily, days 1–14). IC was administered at 3-week intervals. All patients received radical radiotherapy using IMRT techniques. Target volume delineation and dose constraints for organs at risk (OARs) were performed according to our institutional protocol [7, 31]. The CCRT regimens were platinum-based, primarily using cisplatin. Common agents and dosing regimens included: cisplatin/nedaplatin (80–100 mg/m², day 1), carboplatin (400 mg/m², day 1), or loplatin (80–100 mg/m², day 1), administered every 3 weeks for a total of 2–3 cycles. The AC regimens in this study primarily used capecitabine (1000 mg/m², BID, days 1–14) or S-1 (40–60 mg, BID, days 1–14), administered every 3 weeks, with at least one cycle completed within 3 months after CCRT.
Plasma cfEBV DNA quantification
Plasma cfEBV DNA levels were analysed via real-time quantitative polymerase chain reaction (qPCR) targeting the BamHI-W genomic region, according to a previously validated protocol [32, 33]. Specifically, plasma samples were collected in ethylenediaminetetraacetic acid (EDTA)-anticoagulant tubes, with 5 mL of peripheral blood obtained from each participant. The blood samples were centrifuged at 1600 × g to separate plasma, which was then carefully aliquoted into polypropylene tubes and stored at -80 °C until subsequent processing. Genomic DNA extraction from plasma specimens was performed using the QIAamp Blood Kit (Qiagen, Hilden, Germany) in accordance with the manufacturer’s blood and body fluid extraction protocol. For each extraction column, 500–1000 μL of plasma was used, and the extracted DNA was eluted in a final volume of 50 μL; the exact volume of plasma input and eluate was recorded for the subsequent calculation of EBV DNA copy concentration. For real-time quantitative polymerase chain reaction (qPCR) targeting EBV DNA, the BamHI-W region of the EBV genome was selected as the detection locus, a well-validated target for EBV DNA quantification in nasopharyngeal carcinoma. The qPCR assay was established with specific amplification primers: W-44F (5’-AGT CTC TGC CTC CAG GCA-3’) and W-119R (5’-ACA GAG GGC CTG TCC ACC G-3’), alongside a dual-labelled fluorescent probe W-67T (5’-[FAM] CAC TGT CTG TAA AGT CCA GCC TCC [TAMRA]-3’). To verify the amplifiability of plasma DNA and exclude extraction or PCR inhibition, the β-actin gene was used as an internal control. The β-actin primers were designed as forward: 5’-ACA GGC ACCA GGG CGT GAT GG-3’ and reverse: 5’-CTC CAT GTC GTC CCA GTT GGT-3’, with a dual-labelled probe (5’-[FAM] CAT CCT CAC CCT GAA GTA CCC CAT C [TAMRA]-3’) [34,35,36]. Longitudinal monitoring of viral load dynamics was systematically implemented across four critical phases, as described in a previous study [37]: (1) baseline assessment conducted 2 weeks prior to IC initiation; (2) post-first IC cfEBV DNA levels collected within 1 week before 2nd IC cycle; (3) post-IC levels assessed within 1 week before the initiation of CCRT; and (4) post-treatment levels assessed within 1 week upon CCRT completion. The cutoff value for pre-treatment cfEBV DNA was defined as 4000 copies/ml, consistent with our previously validated clinical threshold [12, 38,39,40,41].
Follow-up and outcomes
Following the completion of CCRT, patients were typically instructed to undergo scheduled follow-up assessments quarterly during the first 2 years, semi-annually for the subsequent 3 years, and annually thereafter until death. Standard surveillance incorporated nasopharyngoscopy, head-neck contrast-enhanced MRI, chest X-ray or CT, abdominal ultrasound or CT, and plasma cfEBV DNA quantification.
The primary endpoint was progression-free survival (PFS), calculated from treatment initiation to disease progression, death from any cause, or censoring at the last follow-up. Secondary endpoints comprised OS, calculated from the time of treatment initiation to death. Distant metastasis-free survival (DMFS) as the duration until first distant metastasis or death, and locoregional recurrence-free survival (LRRFS) as the interval until initial locoregional recurrence or death. Censoring was applied at the final documented follow-up for event-free patients. Acute adverse events during IC were graded according to the Common Terminology Criteria for Adverse Events (Version 5.1).
Statistical analysis
All statistical analyses were conducted using SPSS 26.0 (IBM Corp.) and R 4.3.2 (R Foundation). Categorical variables were analysed using χ² or Fisher’s exact tests, with continuity correction applied where appropriate. Survival analyses employed Kaplan–Meier methodology with log-rank testing for between-group comparisons. Propensity score matching (PSM) (1:1 ratio) was implemented using the nearest neighbour algorithm (caliper = 0.1 SD) to mitigate selection bias between the 2-cycle and 3-cycle IC groups. The matching covariates encompassed: demographic parameters (age, sex), disease characteristics (T/N/overall stage, KPS), virological profiles (pre-treatment, post-first IC/post-IC/post-CCRT cfEBV DNA), and treatment details (IC regimen, CCT cycles, AT administration).
Multivariate Cox proportional hazards regression models were applied to identify independent prognostic factors. The resulting regression coefficients were used to calculate individual prognostic risk scores. The predictive accuracy of the prognostic model was further evaluated using receiver operating characteristic (ROC) curve analysis, with area under the curve (AUC) values calculated at 5-year timepoints to quantify discrimination ability. Based on the median value of these risk scores, patients were classified into high-risk and low-risk subgroups. All statistical tests were two-sided, with p < 0.05 being considered statistically significant.
Results
Patient characteristics
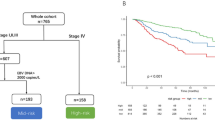
After rigorous screening, we identified 1590 patients with LA-NPC from a total of 79,899 newly diagnosed NPC cases who had received 2–3 cycles of IC followed by CCRT and had complete cfEBV DNA data (eFig. 1, Table 1). Analysis of demographic and clinical characteristics revealed a median age of 46 (range: 18–76) years with a significant male predominance (1184 patients, 74.5%). Stage III disease was present in 1107 cases (69.6%). The cohort demonstrated balanced distribution of IC cycles with 857 patients (53.9%) receiving 2 cycles and 733 (46.1%) patients receiving 3 cycles. Subsequent CCRT cycles followed a similar pattern, with 972 patients (61.1%) completing 2 cycles and 618 (38.9%) patients undergoing 3 cycles. Post-CCRT AT was administered to 173 patients (10.9%). Pretreatment cfEBV DNA analysis identified 612 patients (38.5%) with elevated viral load ≥4000 copies/mL. All patients had positive pretreatment cfEBV DNA; 45.5% of patients achieved undetectable cfEBV DNA after the first IC cycle, 67.5% had cleared cfEBV DNA before radiotherapy, and the proportion reached 94.2% at the end of CCRT (eFig. 2).
Outcomes of different cycles in the whole cohort
During a median follow-up duration of 33.1 months (IQR: 21.37–51.33), the 3- and 5-year PFS rates for the whole cohort were 81.3% and 74.1%, respectively. Survival outcomes in the entire cohort were analysed based on IC cycles. Before PSM, patients receiving 3-cycle IC showed no significant improvement in any survival outcome (all p > 0.05) compared to those receiving 2-cycle IC (eFig. 3A, C, E, G). Subgroup analyses revealed that patients with higher pre-treatment cfEBV DNA levels who received three IC cycles achieved a borderline significant PFS advantage (p = 0.054). In contrast, other early-treatment characteristics did not show significant differences in outcomes after two or three IC cycles (all p > 0.05), as shown in eFig. 4A.
PSM resulted in 491 patients in each group. Patients who received 3-cycle IC had significantly better PFS (HR = 0.72, 95% CI: 0.55–0.96, p = 0.023; eFig. 3B), and the same in OS and DMFS (all p < 0.05, details shown in eFig. 3D, F). Notably, LRRFS did not show significant difference between the different IC-cycle groups (p = 0.11; eFig. 3H). Subgroup analyses demonstrated that among patients with a higher N stage (p = 0.023), an advanced overall stage (p = 0.018), or higher pre-treatment cfEBV DNA levels (p = 0.007), those who received 3-cycle IC experienced a notable improvement in PFS. Specifically, patients who achieved cfEBV DNA clearance after the first cycle of IC also showed a more favourable PFS outcome when they underwent 3-cycle IC (p = 0.007). Additional subgroup analyses details are shown in eFig. 4B.
Prognosis values of post-first IC cfEBV DNA in the whole cohort
Both univariate and multivariate Cox regression analyses (Table 2) verified that a higher N stage (p = 0.008), an advanced overall stage (p = 0.030), and persistent cfEBV DNA after the first IC cycle (p = 0.014), after all IC cycles (before CCRT, p < 0.001), and after CCRT (p < 0.001) were negative prognostic factors for the PFS of the entire cohort.
We selected three early-stage clinical parameters, the N stage, overall stage, and post-first IC cfEBV DNA, which were significantly associated with PFS, to develop a prognostic model and guide IC cycle decisions. ROC curve analysis (eFig. 5A) demonstrated superior discriminative ability for PFS at the 60-month timepoint: the combined model (AUC = 0.632) outperformed single parameters (all p < 0.001) and dual-parameter combinations (AUCN stage+cfEBV DNA = 0.617, p = 0.029; AUCoverall stage+cfEBV DNA = 0.605, p = 0.002). This predictive advantage also extended to secondary outcomes (eFig. 5B-D).
High and low-risk stratification based on prognostic scoring model
Subsequently, we constructed a prognostic scoring model based on the Cox regression coefficients for N stage (β = 0.5118), overall stage (β = 0.2702), and post-first IC cfEBV DNA (β = 0.3465), calculated as: Prognostic Score = (0.5118 × N stage) + (0.2702 × overall stage) + (0.3465 × post-first IC cfEBV DNA). Using the median prognostic score of 2.451 (range: 1.669–2.797) as the threshold, patients were stratified into low-risk (≤2.451, n = 889) and high-risk (>2.451, n = 701) groups (eTable 1). The high-risk group exhibited significantly inferior outcomes compared to the low-risk group (all p < 0.05, eFig. 6). Particularly, survival rates at 3- and 5-years demonstrated marked disparities, with PFS rates of 87.3% and 81.4% in the low-risk group versus 73.8% and 65.1% in the high-risk group, respectively. Multivariate Cox analysis confirmed this risk stratification to be an independent prognostic factor for PFS (adjusted HR = 1.60, 95% CI: 1.24–2.07, p < 0.001) (eFig. 7).
Optimal IC cycles for high- and low-risk groups
Next, we assessed the differential efficacy of IC cycles across risk-stratified cohorts. In the low-risk group, 520 and 369 patients underwent 2-cycle and 3-cycle IC, respectively. While the initial unmatched analysis revealed no survival advantage for extended IC cycles across endpoints (all p > 0.05, eFig. 8A-D), an imbalance in baseline prognostic factors was observed (all p < 0.05) (eTable 2). Therefore, PSM was performed to address these confounders (n = 247 for each group). Post-PSM, the 3-cycle regimen demonstrated clinically meaningful benefits: PFS (HR = 0.59, 95% CI: 0.37–0.94, p = 0.024, Fig. 1a), OS (HR = 0.38, 95% CI: 0.16–0.90, p = 0.024, Fig. 1b), DMFS (HR = 0.53, 95% CI: 0.31–0.91, p = 0.019, Fig. 1c). Meanwhile, LRRFS remained comparable between groups (HR = 0.64, 95% CI: 0.35–1.20, p = 0.157, Fig. 1d). Multivariate Cox regression corroborated these findings, confirming a 41% reduction in progression risk with additional IC cycles (p = 0.029) within the matched cohort (eTable 3, eFig. 9A).
a Progression-free survival after PSM; b Overall survival after PSM; c Distant metastasis-free survival after PSM; d Locoregional recurrence-free survival after PSM.
In contrast, the high-risk cohort, comprising 337 and 364 patients who underwent 2-cycle and 3-cycle IC, exhibited no differences in survival outcomes (eTable 4). Pre-PSM comparisons showed overlapping survival curves for all outcomes (all p > 0.05, eFig. 10A-D). Post-PSM analysis (213 patients for each group) maintained these findings, with comparable PFS (HR = 1.01, 95% CI: 0.69–1.47, p = 0.960, Fig. 2a), OS (HR = 0.65, 95% CI: 0.36–1.18, p = 0.156, Fig. 2b), DMFS (HR = 0.95, 95% CI: 0.63–1.43, p = 0.808, Fig. 2c), and LRRFS (HR = 0.96, 95% CI: 0.61–1.48, p = 0.840, Fig. 2d). Cox regression modelling confirmed that the CCT cycles (p = 0.041), rather than IC cycles (p = 0.984), was the prognostic factor for PFS in the high-risk group (eTable 5, eFig. 9B).
a Progression-free survival after PSM; b Overall survival after PSM; c Distant metastasis-free survival after PSM; d Locoregional recurrence-free survival after PSM.
Adverse events during IC between different IC cycle arms in high- and low-risk groups
No grade 5 adverse events occurred in the PSM-matched low-risk and high-risk cohorts (Table 3). Patients receiving 3-cycle IC showed a numerically higher, but non-significant, incidence of grade 3–4 toxicities compared with those receiving 2-cycle IC (30.36% [75/247] vs. 24.29% [60/247]; p = 0.157). Haematologic toxicities were the predominant severe adverse events, with neutropenia showing the highest frequency (3-cycle vs. 2-cycle: 24.7% vs. 18.2%), followed by leukopenia (3-cycle vs. 2-cycle: 11.7% vs. 9.7%). Mild-to-moderate anaemia was the most frequent low-grade toxicity (3-cycle vs. 2-cycle: 66.4% vs. 60.3%).
Similarly, in the PSM-matched high-risk group, the incidence of grade 3–4 adverse events was comparable between the 3-cycle (31.5% [67/213]) and 2-cycle (28.2% [60/213]) regimens (p = 0.525). Neutropenia was the most common grade 3–4 adverse event (3-cycle vs. 2-cycle: 24.4% vs. 22.1%), while anaemia was the most frequent mild adverse event (3-cycle vs. 2-cycle: 74.2% vs. 68.5%). Notably, none of the adverse events showed significant differences between the 3- and 2-cycle regimens, except for grade 1–2 hypoalbuminemia, which was significantly more frequent in the 3-cycle group (61.0%) than in the 2-cycle group (49.3%; p = 0.027).
Discussion
Our findings demonstrate that integrating post-first IC cfEBV DNA with N stage and overall disease stage enabled effective patient stratification into distinct prognostic cohorts. Particularly, although the low-risk patients derived clinical benefit from three IC cycles without significant toxicity escalation, the high-risk counterparts failed to gain survival advantages from additional chemotherapy cycles, suggesting the necessity for alternative therapeutic strategies in this subpopulation. This differential response pattern underscores the clinical utility of early biomarker-guided treatment intensification while highlighting the biological heterogeneity of chemotherapy-resistant cases.
To the best of our knowledge, our study represents the first systematic integration of post-first IC cfEBV DNA into prognostic stratification and therapeutic decision-making frameworks for LA-NPC. Previous studies have established pretreatment cfEBV DNA as a valuable complement to clinical staging [42, 43] and demonstrated its synergistic prognostic value when combined with clinicopathological parameters [44, 45]. However, existing models relying on baseline cfEBV DNA measurements fail to address two fundamental biological realities, the dynamic clonal evolution during treatment and the need for adaptive therapeutic strategies guided by real-time tumour response [46]. In the present study, we focused on the dynamic change of cfEBV DNA during treatment, and demonstrated that post-first IC cfEBV DNA rather than pre-treatment cfEBV DNA, was the significant prognostic factor for PFS of LA-NPC. This observation aligns with previous study. Lv et al. [37]. pioneered a kinetic profiling approach to stratify patients into distinct prognostic clusters based on longitudinal cfEBV DNA dynamics during treatment. Their analysis revealed that patients achieving immediate clearance after the first IC cycle, termed as “early molecular responders,” exhibited significantly superior disease-free survival compared to non-responders (HR = 2.71, 95% CI: 1.82–4.03, p < 0.001), thereby validating the platinum-sensitive phenotype within this subgroup. However, Lv et al. did not construct an exploration for optimal IC cycles in this manuscript.
Although IC is an important component in the treatment of LA-NPC [47,48,49], our study and previous evidence [50] confirm that the main benefit of IC lies in the improvement of OS/DMFS rather than LRRFS, which may be due to the high locoregional control rate (>90%) achieved within modern IMRT/CCRT [6, 51]. However, the optimal number of cycles remains controversial. Our analysis of 1591 patients revealed clinically meaningful PFS improvements favouring 3-cycle IC, contrasting with prior PSM studies [25, 52] reporting equivalent outcomes between 2-/3-cycle regimens. This discrepancy stems from three distinctions: (1) enhanced statistical power (triple the sample size of prior cohorts); (2) incorporation of longitudinal cfEBV DNA profiling (post-IC/post-CCRT [32, 53]); and (3) enriched representation of stage III patients (66% vs. 36–56% historically), better reflecting LA-NPC’s biological heterogeneity. Although Jiang et al. [54] concluded that 3 cycles of IC could benefit N2-3 patients or responders after 2 cycles, conventional imaging was unable to detect micrometastases leading to systemic progression. Similarly, Lv et al. [26] found that extended IC was ineffective for persistent cfEBV DNA positivity after 2 IC cycles. Our study advances this by utilising cfEBV DNA after the first IC cycle for early decision-making. Post-PSM, low-risk patients (limited tumour burden, platinum-sensitive) derived survival benefits from 3-cycle IC via micrometastasis eradication and tumour volume reduction. In contrast, high-risk patients (bulky disease, chemoresistance) showed no benefit, likely due to delayed RT enabling resistant clones to proliferate [19] and exacerbated toxicity (e.g., higher hypoalbuminemia incidence). Persistent cfEBV DNA after the first IC cycle signals chemoresistance, warranting timely transition to RT or immunotherapy rather than prolonged IC. As Lv et al. demonstrated, RT enhances tumour clearance in non-responders, underscoring its role in overcoming chemoresistance.
In contrast to the study by Lv et al. [37], which determined the administration of 3 or 4 cycles of IC based on whether cfEBV DNA was cleared after 2 cycles of IC, the present study advanced the observation time point to after the first cycle of IC and explored whether patients with LA-NPC in different risk stratifications were more suitable for 2 or 3 cycles of IC. On the one hand, 2–3 cycles of IC are more consistent with clinical practice; most importantly, the earlier time window reduced patients’ waiting time for radiotherapy—a known poor prognostic factor, and alleviated the adverse reactions, economic burden, and psychological stress caused by unnecessary chemotherapy. Second, while Lv et al. [26] merely defined patients with cfEBV DNA clearance after the first cycle of IC as “early responders”, our study achieved clinical translation of this biomarker for the first time. We demonstrated that cfEBV DNA detection after the first cycle of IC, combined with N stage and overall stage (AUC = 0.632), was sufficient to support decision-making regarding IC cycles. Furthermore, the sample size of Lv et al.’s 2019 study was 673 patients, and our sample size was more than twice that of theirs. This not only enhanced the statistical power to validate the value of early cfEBV DNA clearance after the first cycle of IC as a prognostic biomarker but also clarified the efficacy differences between 2 and 3 cycles of IC through rigorous propensity score matching—a key comparison that lacked persuasiveness in previous small-sample studies. In summary, while validating the prognostic value of cfEBV DNA identified in previous studies, the present study proposed an optimised IC strategy that aligns with standard clinical practice, is clinically actionable, and can effectively control toxicity, addressing the key limitations of the aforementioned literature.
Furthermore, our toxicity analysis yielded clinically reassuring findings: low-risk patients receiving 3-cycle IC exhibited adverse event rates comparable to those receiving 2-cycle IC, with grade 3–4 toxicities closely mirroring historical data from randomised clinical trials [10, 11, 55]. This favourable safety profile may be attributed to the preserved integrity of the tumour immune microenvironment in low-risk patients, which could enhance chemosensitivity while mitigating off-target toxicities [56]. In contrast, high-risk patients receiving 3-cycle IC experienced a significantly higher incidence of hypoalbuminemia compared to those receiving 2-cycle IC. This disparity probably results from two mechanisms: (1) accelerated catabolism due to heavy tumour burden and (2) aggravated nitrogen imbalance from prolonged IC exposure [57]. Consequently, these findings underscore the necessity of risk-adapted toxicity monitoring, particularly in patients with persistent post-first IC cfEBV DNA, to optimise treatment efficacy while minimising undue toxicity.
Nonetheless, our study has some limitations that warrant consideration. First, while the qPCR-based assay for EBV DNA quantification is inherently prone to inter-laboratory variability, which may partially limit the universal applicability of some conclusions, this study mitigates such concerns by enroling patients uniformly treated at a single institution (Sun Yat-sen University Cancer Center). Moreover, prior work has demonstrated robust quality control for this assay, with minimal within-run (<10%) and between-day (<20%) variation [34]. Additionally, it is important to acknowledge that harmonising EBV DNA copy number cutoffs across different studies and institutions remains an ongoing effort that requires collaborative advancement from the entire field. Second, the exclusive enrolment of patients from endemic regions may limit the generalisability of our findings to non-endemic populations, where EBV-negative histopathological subtypes are more prevalent. Third, the single-centre design necessitates external validation across diverse ethnic and geographic cohorts to confirm the broader applicability of our prognostic model. Finally, although rigorous PSM was employed to mitigate confounding variables, the inherent selection bias of retrospective observational studies cannot be entirely eliminated.
Conclusion
This study established a novel paradigm for risk-adapted treatment in LA-NPC, utilising post-first IC cfEBV DNA levels in conjunction with conventional staging. This approach enabled the stratification of patients into distinct prognostic groups, highlighting the potential of tailored IC regimens. While three cycles of IC demonstrated benefit in low-risk patients, high-risk patients showed no significant survival advantage with additional chemotherapy, suggesting the need for alternative treatment strategies. Therefore, future research should explore the role of cfEBV DNA kinetics in the current and emerging era of immunotherapy for LA-NPC, to further refine and personalise treatment approaches.
Data availability
The datasets supporting the conclusions of this article are not publicly available to protect patient privacy. Data may be accessed upon reasonable request by contacting the first author Wan-Ping Guo, [guowp@sysucc.org.cn]) or the corresponding author (Ling Guo, [guoling@sysucc.org.cn]), subject to institutional approval and compliance with data protection regulations.
References
Chen YP, Chan ATC, Le QT, Blanchard P, Sun Y, Ma J. Nasopharyngeal carcinoma. Lancet. 2019;394:64–80.
Su ZY, Siak PY, Lwin YY, Cheah SC. Epidemiology of nasopharyngeal carcinoma: current insights and future outlook. Cancer Metastasis Rev. 2024;43:919–39.
Mao YP, Xie FY, Liu LZ, Sun Y, Li L, Tang LL, et al. Re-evaluation of 6th edition of AJCC staging system for nasopharyngeal carcinoma and proposed improvement based on magnetic resonance imaging. Int J Radiat Oncol Biol Phys. 2009;73:1326–34.
Chan AT, Leung SF, Ngan RK, Teo PM, Lau WH, Kwan WH, et al. Overall survival after concurrent cisplatin-radiotherapy compared with radiotherapy alone in locoregionally advanced nasopharyngeal carcinoma. J Natl Cancer Inst. 2005;97:536–9.
Zhang L, Zhao C, Peng PJ, Lu LX, Huang PY, Han F, et al. Phase III study comparing standard radiotherapy with or without weekly oxaliplatin in treatment of locoregionally advanced nasopharyngeal carcinoma: preliminary results. J Clin Oncol. 2005;23:8461–8.
Lin JC, Jan JS, Hsu CY, Liang WM, Jiang RS, Wang WY. Phase III study of concurrent chemoradiotherapy versus radiotherapy alone for advanced nasopharyngeal carcinoma: positive effect on overall and progression-free survival. J Clin Oncol. 2003;21:631–7.
Lai SZ, Li WF, Chen L, Luo W, Chen YY, Liu LZ, et al. How does intensity-modulated radiotherapy versus conventional two-dimensional radiotherapy influence the treatment results in nasopharyngeal carcinoma patients? Int J Radiat Oncol Biol Phys. 2011;80:661–8.
Lee AW, Ng WT, Chan LL, Hung WM, Chan CC, Sze HC, et al. Evolution of treatment for nasopharyngeal cancer-success and setback in the intensity-modulated radiotherapy era. Radiother Oncol. 2014;110:377–84.
Sun XS, Liu SL, Luo MJ, Li XY, Chen QY, Guo SS, et al. The association between the development of radiation therapy, image technology, and chemotherapy, and the survival of patients with nasopharyngeal carcinoma: a cohort study from 1990 to 2012. Int J Radiat Oncol Biol Phys. 2019;105:581–90.
Sun Y, Li WF, Chen NY, Zhang N, Hu GQ, Xie FY, et al. Induction chemotherapy plus concurrent chemoradiotherapy versus concurrent chemoradiotherapy alone in locoregionally advanced nasopharyngeal carcinoma: a phase 3, multicentre, randomised controlled trial. Lancet Oncol. 2016;17:1509–20.
Zhang Y, Chen L, Hu GQ, Zhang N, Zhu XD, Yang KY, et al. Gemcitabine and cisplatin induction chemotherapy in nasopharyngeal carcinoma. N Engl J Med. 2019;381:1124–35.
Zhang Y, Chen L, Hu GQ, Zhang N, Zhu XD, Yang KY, et al. Final overall survival analysis of gemcitabine and cisplatin induction chemotherapy in nasopharyngeal carcinoma: a multicenter, randomized phase III trial. J Clin Oncol. 2022;40:2420–5.
National Comprehensive Cancer Network. Head and neck cancers. version 2.2025. https://www.nccn.org/guidelines/guidelines-detail?category=1&id=1437.
Chinese Society of Clinical Oncology (CSCO) Clinical Guidelines Committee. CSCO clinical guidelines for the diagnosis and treatment of nasopharyngeal carcinoma. Cancer Commun (London, England). 2021;41:1023–52.
Li WZ, Lv X, Hu D, Lv SH, Liu GY, Liang H, et al. Effect of induction chemotherapy with paclitaxel, cisplatin, and capecitabine vs cisplatin and fluorouracil on failure-free survival for patients with stage iva to ivb nasopharyngeal carcinoma: a multicenter phase 3 randomized clinical trial. JAMA Oncol. 2022;8:706–14.
Xie WH, Xiao WW, Chang H, Xu MJ, Hu YH, Zhou TC, et al. Four cycles of docetaxel plus cisplatin as neoadjuvant chemotherapy followed by concurrent chemoradiotherapy in stage N2-3 nasopharyngeal carcinoma: phase 3 multicentre randomised controlled trial. BMJ. 2025;389:e081557.
Davis AJ, Tannock JF. Repopulation of tumour cells between cycles of chemotherapy: a neglected factor. Lancet Oncol. 2000;1:86–93.
Chen YP, Mao YP, Zhang WN, Chen L, Tang LL, Li WF, et al. Prognostic value of wait time in nasopharyngeal carcinoma treated with intensity modulated radiotherapy: a propensity matched analysis. Oncotarget. 2016;7:14973–82.
Liang H, Xiang YQ, Lv X, Xie CQ, Cao SM, Wang L, et al. Survival impact of waiting time for radical radiotherapy in nasopharyngeal carcinoma: a large institution-based cohort study from an endemic area. Eur J Cancer. 2017;73:48–60.
Pramanik R, Arora S, Sharma P, Biswas A, Nayak B, Thakar A, et al. Cell-free EBV DNA as a biomarker during clinical management of nasopharyngeal carcinoma in a nonendemic region. J Med Virol. 2022;94:720–8.
Chan KCA, Woo JKS, King A, Zee BCY, Lam WKJ, Chan SL, et al. Analysis of plasma Epstein-Barr Virus DNA to screen for nasopharyngeal cancer. N Engl J Med. 2017;377:513–22.
Weng Y, Cai S, Li C, Xu Y, Pan Y, Huang Z, et al. Selection of induction chemotherapy cycles for stage N3 nasopharyngeal carcinoma based on pre-treatment plasma EBV DNA. Sci Rep. 2024;14:24484.
Ding C, Dai DY, Luo ZK, Wang GY, Dong Z, Qin GJ, et al. Evaluation of a novel model incorporating serological indicators into the conventional TNM staging system for nasopharyngeal carcinoma. Oral Oncol. 2024;151:106725.
Peng L, Chen YP, Xu C, Tang LL, Chen L, Lin AH, et al. A novel scoring model to predict benefit of additional induction chemotherapy to concurrent chemoradiotherapy in stage II-IVa nasopharyngeal carcinoma. Oral Oncol. 2018;86:258–65.
He Y, Zhao Z, Wang Y, Chai J, He J, Wang J, et al. Optimizing number of cycles of induction chemotherapy for patients with nasopharyngeal carcinoma: retrospective survival analysis. Head Neck. 2020;42:2067–76.
Lv J, Xu LX, Li ZX, Lin L, Wu CF, Quan TQ, et al. Longitudinal on-treatment circulating tumor DNA as a biomarker for real-time dynamic risk monitoring in cancer patients: the EP-SEASON study. Cancer Cell. 2024;42:1401–14 e1404.
Lee AWM, Lee VHF, Ng WT, Strojan P, Saba NF, Rinaldo A, et al. A systematic review and recommendations on the use of plasma EBV DNA for nasopharyngeal carcinoma. Eur J Cancer. 2021;153:109–22.
Wu CF, Lin L, Mao YP, Deng B, Lv JW, Zheng WH, et al. Liquid biopsy posttreatment surveillance in endemic nasopharyngeal carcinoma: a cost-effective strategy to integrate circulating cell-free Epstein-Barr virus DNA. BMC Med. 2021;19:193.
Tan R, Phua SKA, Soong YL, Oon LLE, Chan KS, Lucky SS, et al. Clinical utility of Epstein-Barr virus DNA and other liquid biopsy markers in nasopharyngeal carcinoma. Cancer Commun. 2020;40:564–85.
Du XJ, Wang GY, Zhu XD, Han YQ, Lei F, Shen LF, et al. Refining the 8th edition TNM classification for EBV-related nasopharyngeal carcinoma. Cancer Cell. 2024;42:464–73 e463.
Li WF, Sun Y, Chen M, Tang LL, Liu LZ, Mao YP, et al. Locoregional extension patterns of nasopharyngeal carcinoma and suggestions for clinical target volume delineation. Chin J Cancer. 2012;31:579–87.
Chen FP, Huang XD, Lv JW, Wen DW, Zhou GQ, Lin L, et al. Prognostic potential of liquid biopsy tracking in the posttreatment surveillance of patients with nonmetastatic nasopharyngeal carcinoma. Cancer. 2020;126:2163–73.
Huang CL, Wang GY, Lou JH, Chen L, Li QJ, Li KP, et al. Oral chemotherapy versus observation alone in nasopharyngeal carcinoma patients with persistently detected circulating cell-free Epstein-Barr virus DNA during follow-up. Radiother Oncol. 2024;190:110032.
Shao JY, Li YH, Gao HY, Wu QL, Cui NJ, Zhang L, et al. Comparison of plasma Epstein-Barr virus (EBV) DNA levels and serum EBV immunoglobulin A/virus capsid antigen antibody titers in patients with nasopharyngeal carcinoma. Cancer. 2004;100:1162–70.
Tang LQ, Chen QY, Fan W, Liu H, Zhang L, Guo L, et al. Prospective study of tailoring whole-body dual-modality [18F]fluorodeoxyglucose positron emission tomography/computed tomography with plasma Epstein-Barr virus DNA for detecting distant metastasis in endemic nasopharyngeal carcinoma at initial staging. J Clin Oncol. 2013;31:2861–9.
An X, Wang FH, Ding PR, Deng L, Jiang WQ, Zhang L, et al. Plasma Epstein-Barr virus DNA level strongly predicts survival in metastatic/recurrent nasopharyngeal carcinoma treated with palliative chemotherapy. Cancer. 2011;117:3750–7.
Lv J, Chen Y, Zhou G, Qi Z, Tan KRL, Wang H, et al. Liquid biopsy tracking during sequential chemo-radiotherapy identifies distinct prognostic phenotypes in nasopharyngeal carcinoma. Nat Commun. 2019;10:3941.
Guo SS, Yang JH, Sun XS, Liu LZ, Yang ZC, Liu LT, et al. Reduced-dose radiotherapy for Epstein-Barr virus DNA-selected staged III nasopharyngeal carcinoma: a single-arm, phase 2 trial. Eur J Cancer. 2023;194:113336.
Li XY, Luo DH, Guo L, Mo HY, Sun R, Guo SS, et al. Deintensified chemoradiotherapy for pretreatment Epstein-Barr Virus DNA-selected low-risk locoregionally advanced nasopharyngeal carcinoma: a phase II randomized noninferiority trial. J Clin Oncol. 2022;40:1163–73.
Tang LL, Guo R, Zhang N, Deng B, Chen L, Cheng ZB, et al. Effect of radiotherapy alone vs radiotherapy with concurrent chemoradiotherapy on survival without disease relapse in patients with low-risk nasopharyngeal carcinoma: a randomized clinical trial. JAMA. 2022;328:728–36.
Tang SQ, Chen L, Li WF, Chan ATC, Huang SH, Chua MLK, et al. Identifying optimal clinical trial candidates for locoregionally advanced nasopharyngeal carcinoma: Analysis of 9468 real-world cases and validation by two phase 3 multicentre, randomised controlled trial. Radiother Oncol. 2022;167:179–86.
Leung SF, Zee B, Ma BB, Hui EP, Mo F, Lai M, et al. Plasma Epstein-Barr viral deoxyribonucleic acid quantitation complements tumor-node-metastasis staging prognostication in nasopharyngeal carcinoma. J Clin Oncol. 2006;24:5414–8.
Chen FP, Lin L, Liang JH, Tan SH, Ong EHW, Luo YS, et al. Development of a risk classification system combining TN-categories and circulating EBV DNA for non-metastatic NPC in 10,149 endemic cases. Ther Adv Med Oncol. 2021;13:17588359211052417.
Tang, LQ, Li, CF, Li, J, Chen, WH, Chen, QY, Yuan, LX, et al. Establishment and validation of prognostic nomograms for endemic nasopharyngeal carcinoma. J Natl Cancer Inst. 2016;108.
Wang G, Dong Z, Huang C, Du X, Chen L, Li K, et al. The value of integrating tumor volume and plasma Epstein-Barr virus DNA load during sequential chemoradiotherapy for prognostic prediction and therapeutic guidance in high-risk locoregionally advanced nasopharyngeal carcinoma. Oral Oncol. 2023;145:106500.
Lv J, Wu C, Li J, Chen F, He S, He Q, et al. Improving on-treatment risk stratification of cancer patients with refined response classification and integration of circulating tumor DNA kinetics. BMC Med. 2022;20:268.
Chen YP, Ismaila N, Chua MLK, Colevas AD, Haddad R, Huang SH, et al. Chemotherapy in combination with radiotherapy for definitive-intent treatment of stage II-IVA nasopharyngeal carcinoma: CSCO and ASCO guideline. J Clin Oncol. 2021;39:840–59.
Jiang W, Lv JW, Tang LL, Sun Y, Chen YP, Ma J. Enhancing efficacy and reducing toxicity: therapeutic optimization in locoregionally advanced nasopharyngeal carcinoma. Cell Rep Med. 2024;5:101594.
Zhang B, Li MM, Chen WH, Zhao JF, Chen WQ, Dong YH, et al. Association of chemoradiotherapy regimens and survival among patients with nasopharyngeal carcinoma: a systematic review and meta-analysis. JAMA Netw Open. 2019;2:e1913619.
OuYang PY, Xie C, Mao YP, Zhang Y, Liang XX, Su Z, et al. Significant efficacies of neoadjuvant and adjuvant chemotherapy for nasopharyngeal carcinoma by meta-analysis of published literature-based randomized, controlled trials. Ann Oncol. 2013;24:2136–46.
Lee N, Xia P, Quivey JM, Sultanem K, Poon I, Akazawa C, et al. Intensity-modulated radiotherapy in the treatment of nasopharyngeal carcinoma: an update of the UCSF experience. Int J Radiat Oncol Biol Phys. 2002;53:12–22.
Jiang Y, Chen K, Yang J, Liang Z, Qu S, Li L, et al. Optimize the number of cycles of induction chemotherapy for locoregionally advanced nasopharyngeal carcinoma: a propensity score matching analysis. J Cancer. 2022;13:426–35.
Chen FP, Luo YS, Chen K, Li JY, Huo LQ, Shi L, et al. Circulating Epstein-Barr virus DNA level post induction chemotherapy contributes to prognostication in advanced-stage nasopharyngeal carcinoma. Eur J Cancer. 2021;151:63–71.
Jiang YT, Chen KH, Liang ZG, Yang J, Qu S, Li L, et al. Individualized number of induction chemotherapy cycles for locoregionally advanced nasopharyngeal carcinoma patients based on early tumor response. Cancer Med. 2023;12:4010–22.
Cao SM, Yang Q, Guo L, Mai HQ, Mo HY, Cao KJ, et al. Neoadjuvant chemotherapy followed by concurrent chemoradiotherapy versus concurrent chemoradiotherapy alone in locoregionally advanced nasopharyngeal carcinoma: A phase III multicentre randomised controlled trial. Eur J Cancer. 2017;75:14–23.
Gong L, Kwong DL, Dai W, Wu P, Li S, Yan Q, et al. Comprehensive single-cell sequencing reveals the stromal dynamics and tumor-specific characteristics in the microenvironment of nasopharyngeal carcinoma. Nat Commun. 2021;12:1540.
Miao J, Wang L, Ong EHW, Hu C, Lin S, Chen X, et al. Effects of induction chemotherapy on nutrition status in locally advanced nasopharyngeal carcinoma: a multicentre prospective study. J Cachexia Sarcopenia Muscle. 2023;14:815–25.
Funding
This study was funded by grants from the National Key Research and Development Program of China (2022YFC2505800, 2022YFC2705005), Noncommunicable Chronic Diseases-National Science and Technology Major Project (2024ZD0520706), National Natural Science Foundation of China (Nos. 82173287, 82372980, 82222050, 82303873, 32200651, 82203776, 82203125, 82272739, 82272882, 82373258, 82361168664, 82473396, 82473038), Guangdong Basic and Applied Basic Research Foundation (2023B1515120092), Guangdong Natural Science Foundation (2025A1515010068), Science and Technology Program of Guangzhou (2023A04J1956, 2024B03J1248), Sun Yat-sen University Clinical Research 5010 Program (Nos. 2015021, 2017010), and China Postdoctoral Science Foundation (2024M763792).
Author information
Authors and Affiliations
Contributions
Conceptualisation: Ling Guo, Sai-Lan Liu, Dong-Hua Luo. Data curation: Wan-Ping Guo, Xuan Yu, Zi-Jian Lu. Formal analysis: Wan-Ping Guo, Xuan Yu, Chun Wu. Investigation: Wan-Ping Guo, Xuan Yu, Yu-Chen Li. Methodology: Sai-Lan Liu. Project administration: Ling Guo, Dong-Hua Luo. Resources: Ling Guo, Sai-Lan Liu, Dong-Hua Luo. Supervision: Ling Guo, Dong-Hua Luo. Validation: Chun Wu, Li-Wen Gu, Jing-Na Cao. Visualisation: Yu-Chen Li, Li-Wen Gu, Xiao-Yun Li. Writing-original draft: Wan-Ping Guo. Writing-review & editing: Ling-Guo, Sai-Lan Liu, Dong-Hua Luo.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval and consent to participate
This retrospective study was approved by the Institutional Review Board of Sun Yat-sen University Cancer Center (B2025-634-01). As a retrospective analysis of anonymized clinical data, the need for individual patient consent was waived by the ethics committee. The study was conducted in accordance with the principles of the Declaration of Helsinki.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Guo, WP., Yu, X., Lu, ZJ. et al. Incorporating early cfEBV DNA clearance into clinical risk stratification to tailor induction chemotherapy cycles for locoregionally advanced nasopharyngeal carcinoma. Br J Cancer (2026). https://doi.org/10.1038/s41416-026-03401-5
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41416-026-03401-5