Abstract
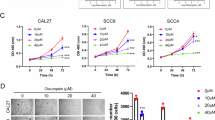
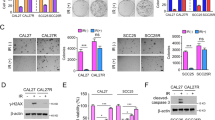
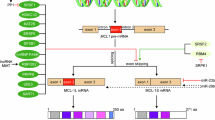
Oral squamous cell carcinoma (OSCC) is a prevalent and aggressive malignancy of the oral cavity, with increasing incidence and poor prognosis. Myeloid leukemia 1 (Mcl-1), an anti-apoptotic protein in the BCL-2 family, is critical for tumor development and progression. In this study, we investigated Dioscin, a natural compound, as a potential therapeutic agent for OSCC. Our results demonstrated that Dioscin significantly inhibits cell viability and colony formation in OSCC cell lines. Mechanistically, Dioscin induced intrinsic apoptosis by promoting the ubiquitination and degradation of Mcl-1. Further analysis revealed that Dioscin enhances the interaction between the E3 ligase β-TRCP and Mcl-1 by inhibiting the Akt/GSK3β signaling pathway, resulting in increased phosphorylation of Mcl-1 at Ser159, which drives its destabilization. In vivo, Dioscin notably suppressed OSCC tumor growth, including in sensitive and radioresistant cells, by reducing Mcl-1 levels. These findings highlight the therapeutic potential of Dioscin for OSCC treatment, offering new insights for overcoming radioresistance and improving clinical outcomes in OSCC patients.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
Data availability
The datasets used and analyzed in this study are available from the corresponding author (W. L.) upon request.
References
Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, et al. Global Cancer Statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 Cancers in 185 countries. CA Cancer J Clin. 2021;71:209–49.
Tan Y, Wang Z, Xu M, Li B, Huang Z, Qin S, et al. Oral squamous cell carcinomas: state of the field and emerging directions. Int J Oral Sci. 2023;15:44.
Siegel RL, Miller KD, Jemal A. Cancer statistics, 2020. CA Cancer J Clin. 2020;70:7–30.
Fan T, Wang X, Zhang S, Deng P, Jiang Y, Liang Y, et al. NUPR1 promotes the proliferation and metastasis of oral squamous cell carcinoma cells by activating TFE3-dependent autophagy. Signal Transduct Target Ther. 2022;7:130.
Zhang C, Cai Q, Ke J. Poor prognosis of oral squamous cell carcinoma correlates with ITGA6. Int Dent J. 2023;73:178–85.
Park SJ, Kim MJ, Kim YK, Kim SM, Park JY, Myoung H. Combined cetuximab and genistein treatment shows additive anti-cancer effect on oral squamous cell carcinoma. Cancer Lett. 2010;292:54–63.
Lee YM, Hsu CL, Chen YH, Ou DL, Hsu C, Tan CT. Genomic and transcriptomic landscape of an oral squamous cell carcinoma mouse model for immunotherapy. Cancer Immunol Res. 2023;11:1553–67.
Diao P, Jiang Y, Li Y, Wu X, Li J, Zhou C, et al. Immune landscape and subtypes in primary resectable oral squamous cell carcinoma: prognostic significance and predictive of therapeutic response. J Immunother Cancer. 2021;9:e002434.
Weckx A, Riekert M, Grandoch A, Schick V, Zöller JE, Kreppel M. Time to recurrence and patient survival in recurrent oral squamous cell carcinoma. Oral Oncol. 2019;94:8–13.
Wang R, Li X, Gan Y, Liao J, Han S, Li W, et al. Dioscin inhibits non-small cell lung cancer cells and activates apoptosis by downregulation of survivin. J Cancer. 2024;15:1366–77.
Zhou L, Yu X, Li M, Gong G, Liu W, Li T, et al. Cdh1-mediated Skp2 degradation by dioscin reprogrammes aerobic glycolysis and inhibits colorectal cancer cells growth. EBioMedicine. 2020;51:102570.
Li M, Gao F, Li X, Gan Y, Han S, Yu X, et al. Stabilization of MCL-1 by E3 ligase TRAF4 confers radioresistance. Cell Death Dis. 2022;13:1053.
Li XY, Gao F, Wang XC, Liu LL, Gan Y, Han SZ, et al. Curcumol exerts antitumor effect via inhibiting EGFR-Akt-Mcl-1 signaling. Am J Chin Med. 2023;51:741–60.
Wang H, Guo M, Wei H, Chen Y. Targeting MCL-1 in cancer: current status and perspectives. J Hematol Oncol. 2021;14:67.
Mittal P, Singh S, Sinha R, Shrivastava A, Singh A, Singh IK. Myeloid cell leukemia 1 (MCL-1): structural characteristics and application in cancer therapy. Int J Biol Macromol. 2021;187:999–1018.
Gerdes J, Schwab U, Lemke H, Stein H. Production of a mouse monoclonal antibody reactive with a human nuclear antigen associated with cell proliferation. Int J Cancer. 1983;31:13–20.
Wang M, Guo FS, Hou DS, Zhang HL, Chen XT, Shen YX, et al. Glutamine signaling specifically activates c-Myc and Mcl-1 to facilitate cancer cell proliferation and survival. Protein Cell. 2025;16:968–84.
Beltrán-Visiedo M, Jiménez-Alduán N, Díez R, Cuenca M, Benedi A, Serrano-Del Valle A, et al. Dinaciclib synergizes with BH3 mimetics targeting BCL-2 and BCL-X(L) in multiple myeloma cell lines partially dependent on MCL-1 and in plasma cells from patients. Mol Oncol. 2023;17:2507–25.
Munkhbaatar E, Dietzen M, Agrawal D, Anton M, Jesinghaus M, Boxberg M, et al. MCL-1 gains occur with high frequency in lung adenocarcinoma and can be targeted therapeutically. Nat Commun. 2020;11:4527.
Wei LH, Kuo ML, Chen CA, Chou CH, Cheng WF, Chang MC, et al. The anti-apoptotic role of interleukin-6 in human cervical cancer is mediated by up-regulation of Mcl-1 through a PI 3-K/Akt pathway. Oncogene. 2001;20:5799–809.
Bonneaud TL, Lefebvre CC, Nocquet L, Basseville A, Roul J, Weber H, et al. Targeting of MCL-1 in breast cancer-associated fibroblasts reverses their myofibroblastic phenotype and pro-invasive properties. Cell Death Dis. 2022;13:787.
Weng JR, Bai LY, Ko HH, Tsai YT. Cyclocommunol induces apoptosis in human oral squamous cell carcinoma partially through a Mcl-1-dependent mechanism. Phytomedicine. 2018;39:25–32.
Maji S, Samal SK, Pattanaik L, Panda S, Quinn BA, Das SK, et al. Mcl-1 is an important therapeutic target for oral squamous cell carcinomas. Oncotarget. 2015;6:16623–37.
Wang R, Li X, Wang J. Butein inhibits oral squamous cell carcinoma growth via promoting MCL-1 ubiquitination. J Cancer. 2024;15:3173–82.
Maji S, Shriwas O, Samal SK, Priyadarshini M, Rath R, Panda S, et al. STAT3- and GSK3β-mediated Mcl-1 regulation modulates TPF resistance in oral squamous cell carcinoma. Carcinogenesis. 2019;40:173–83.
Palve V, Mallick S, Ghaisas G, Kannan S, Teni T. Overexpression of Mcl-1L splice variant is associated with poor prognosis and chemoresistance in oral cancers. PLoS One. 2014;9:e111927.
Bandopadhyay S, Anand U, Gadekar VS, Jha NK, Gupta PK, Behl T, et al. Dioscin: a review on pharmacological properties and therapeutic values. Biofactors. 2022;48:22–55.
Li X, Shen D, Zhu Z, Lyu D, He C, Sun Y, et al. Dual roles of demethylation in cancer treatment and cardio-function recovery. Redox Biol. 2023;64:102785.
Li XL, Ma RH, Ni ZJ, Thakur K, Cespedes-Acuña CL, Wang S, et al. Dioscin inhibits human endometrial carcinoma proliferation via G0/G1 cell cycle arrest and mitochondrial-dependent signaling pathway. Food Chem Toxicol. 2021;148:111941.
Chiu CH, Chou YC, Lin JP, Kuo CL, Lu HF, Huang YP, et al. Chloroform extract of solanum lyratum induced G0/G1 arrest via p21/p16 and induced apoptosis via reactive oxygen species, caspases and mitochondrial pathways in human oral cancer cell lines. Am J Chin Med. 2015;43:1453–69.
Lv L, Zheng L, Dong D, Xu L, Yin L, Xu Y, et al. Dioscin, a natural steroid saponin, induces apoptosis and DNA damage through reactive oxygen species: a potential new drug for treatment of glioblastoma multiforme. Food Chem Toxicol. 2013;59:657–69.
Radović T, Manzey D. Effects of complexity and similarity of an interruption task on resilience toward interruptions in a procedural task with sequential constraints. J Exp Psychol Hum Percept Perform. 2022;48:159–73.
Mao Z, Han X, Chen D, Xu Y, Xu L, Yin L, et al. Potent effects of dioscin against hepatocellular carcinoma through regulating TP53-induced glycolysis and apoptosis regulator (TIGAR)-mediated apoptosis, autophagy, and DNA damage. Br J Pharm. 2019;176:919–37.
Xun J, Zhou S, Lv Z, Wang B, Luo H, Zhang L, et al. Dioscin modulates macrophages polarization and MDSCs differentiation to inhibit tumorigenesis of colitis-associated colorectal cancer. Int Immunopharmacol. 2023;117:109839.
Rehbein P, Saxena K, Schlepckow K, Schwalbe H. Protocol for aerosol-free recombinant production and NMR analysis of prion proteins. J Biomol NMR. 2014;59:111–7.
Deng H, Han Y, Liu L, Zhang H, Liu D, Wen J, et al. Targeting myeloid leukemia-1 in cancer therapy: advances and directions. J Med Chem. 2024;67:5963–98.
Liu S, Qiao X, Wu S, Gai Y, Su Y, Edwards H, et al. c-Myc plays a critical role in the antileukemic activity of the Mcl-1-selective inhibitor AZD5991 in acute myeloid leukemia. Apoptosis. 2022;27:913–28.
Zhong Q, Gao W, Du F, Wang X. Mule/ARF-BP1, a BH3-only E3 ubiquitin ligase, catalyzes the polyubiquitination of Mcl-1 and regulates apoptosis. Cell. 2005;121:1085–95.
Robinson EJ, Aguiar S, Smidt MP, van der Heide LP. MCL1 as a therapeutic target in Parkinson’s disease? Trends Mol Med. 2019;25:1056–65.
Ren H, Koo J, Guan B, Yue P, Deng X, Chen M, et al. The E3 ubiquitin ligases β-TrCP and FBXW7 cooperatively mediates GSK3-dependent Mcl-1 degradation induced by the Akt inhibitor API-1, resulting in apoptosis. Mol Cancer. 2013;12:146.
Maurer U, Charvet C, Wagman AS, Dejardin E, Green DR. Glycogen synthase kinase-3 regulates mitochondrial outer membrane permeabilization and apoptosis by destabilization of MCL-1. Mol Cell. 2006;21:749–60.
Li Y, Shen Z, Jiang X, Wang Y, Yang Z, Mao Y, et al. Mouse mesenchymal stem cell-derived exosomal miR-466f-3p reverses EMT process through inhibiting AKT/GSK3β pathway via c-MET in radiation-induced lung injury. J Exp Clin Cancer Res. 2022;41:128.
Jere SW, Houreld NN, Abrahamse H. Role of the PI3K/AKT (mTOR and GSK3β) signalling pathway and photobiomodulation in diabetic wound healing. Cytokine Growth Factor Rev. 2019;50:52–9.
Lin J, Song T, Li C, Mao W. GSK-3β in DNA repair, apoptosis, and resistance of chemotherapy, radiotherapy of cancer. Biochim Biophys Acta Mol Cell Res. 2020;1867:118659.
Smyth EC, Lagergren J, Fitzgerald RC, Lordick F, Shah MA, Lagergren P, et al. Oesophageal cancer. Nat Rev Dis Prim. 2017;3:17048.
Ford PJ, Rich AM. Tobacco use and oral health. Addiction. 2021;116:3531–40.
Hübbers CU, Akgül B. HPV and cancer of the oral cavity. Virulence. 2015;6:244–8.
Palve VC, Teni TR. Association of anti-apoptotic Mcl-1L isoform expression with radioresistance of oral squamous carcinoma cells. Radiat Oncol. 2012;7:135.
Sulkshane P, Pawar SN, Waghole R, Pawar SS, Rajput P, Uthale A, et al. Elevated USP9X drives early-to-late-stage oral tumorigenesis via stabilisation of anti-apoptotic MCL-1 protein and impacts outcome in oral cancers. Br J Cancer. 2021;125:547–60.
Sun Y, Nie W, Qiu B, Yang Q, Zhao H. FBXW7 affects autophagy through MCL1 in oral squamous cell carcinoma. Oral Dis. 2023;29:3259–67.
Thomas LW, Lam C, Edwards SW. Mcl-1; the molecular regulation of protein function. FEBS Lett. 2010;584:2981–9.
Ding Q, He X, Hsu JM, Xia W, Chen CT, Li LY, et al. Degradation of Mcl-1 by beta-TrCP mediates glycogen synthase kinase 3-induced tumor suppression and chemosensitization. Mol Cell Biol. 2007;27:4006–17.
Wolfsperger F, Hogh-Binder SA, Schittenhelm J, Psaras T, Ritter V, Bornes L, et al. Deubiquitylating enzyme USP9x regulates radiosensitivity in glioblastoma cells by Mcl-1-dependent and -independent mechanisms. Cell Death Dis. 2016;7:e2039.
Liu J, Wei L, Miao Q, Zhan S, Chen P, Liu W, et al. MDM2 drives resistance to Osimertinib by contextually disrupting FBW7-mediated destruction of MCL-1 protein in EGFR mutant NSCLC. J Exp Clin Cancer Res. 2024;43:302.
Kim E, Kim YJ, Ji Z, Kang JM, Wirianto M, Paudel KR, et al. ROR activation by Nobiletin enhances antitumor efficacy via suppression of IκB/NF-κB signaling in triple-negative breast cancer. Cell Death Dis. 2022;13:374.
Aumsuwan P, Khan SI, Khan IA, Ali Z, Avula B, Walker LA, et al. The anticancer potential of steroidal saponin, dioscin, isolated from wild yam (Dioscorea villosa) root extract in invasive human breast cancer cell line MDA-MB-231 in vitro. Arch Biochem Biophys. 2016;591:98–110.
Kim EA, Jang JH, Lee YH, Sung EG, Song IH, Kim JY, et al. Dioscin induces caspase-independent apoptosis through activation of apoptosis-inducing factor in breast cancer cells. Apoptosis. 2014;19:1165–75.
Hsieh MJ, Yang SF, Hsieh YS, Chen TY, Chiou HL. Autophagy inhibition enhances apoptosis induced by dioscin in huh7 cells. Evid Based Complement Altern Med. 2012;2012:134512.
Zhang G, Zeng X, Zhang R, Liu J, Zhang W, Zhao Y, et al. Dioscin suppresses hepatocellular carcinoma tumor growth by inducing apoptosis and regulation of TP53, BAX, BCL2 and cleaved CASP3. Phytomedicine. 2016;23:1329–36.
Tantawy SI, Sarkar A, Hubner S, Tan Z, Wierda WG, Eldeib A, et al. Mechanisms of MCL-1 protein stability induced by MCL-1 antagonists in B-Cell malignancies. Clin Cancer Res. 2023;29:446–57.
Zhou H, Zhou L, Guan Q, Hou X, Wang C, Liu L, et al. Skp2-mediated MLKL degradation confers cisplatin-resistant in non-small cell lung cancer cells. Commun Biol. 2023;6:805.
Funding
This work was supported by the Wisdom Accumulation and Talent Cultivation Project of the Third Xiangya Hospital of Central South University (BJ202203) and the Medical and Health Industry Joint Fund Project of Hunan Provincial Natural Science Foundation (2024JJ9261).
Author information
Authors and Affiliations
Contributions
Qi Liang and Wei Li conceived and designed the project. Xuecheng Wu, Dongyu Li and Yiwei Liu administered the project and developed its methodology. Ruirui Wang, Xiaoying Li and Pengfei Guo analyzed and interpreted the data. Qi Liang wrote the original draft. Wei Li supervised the manuscript and acquired funding. All authors have read and agreed to the published version.
Corresponding author
Ethics declarations
Competing interests
The authors have declared no Competing interest.
Ethics Approval
All animal experiments were approved by the Institutional Animal Care and Use Committee, the Third Xiangya Hospital of Central South University (Changsha, China). For experiments using human samples, all samples were anonymously coded following local ethical guidelines (as stipulated by the Declaration of Helsinki). Written informed consent was obtained from patients, and the protocol was approved by the Ethical Review Board of Central South University. All methods were performed in accordance with the relevant guidelines and regulations.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Liang, Q., Wu, X., Li, D. et al. Dioscin suppresses tumorigenesis and overcomes radioresistance by promoting ubiquitination-mediated degradation of Mcl-1. Cancer Gene Ther (2026). https://doi.org/10.1038/s41417-026-01022-x
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41417-026-01022-x