Abstract
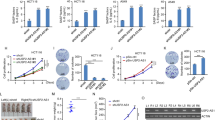
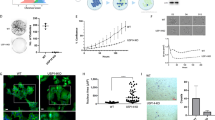
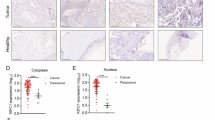
Oncogene-induced senescence (OIS) is a critical tumor-suppressor mechanism, which prevents hyper-proliferation and transformation of cells. c-Myc promotes OIS through the transcriptional activation of p14ARF followed by p53 activation. Although the oncogene-mediated transcriptional regulation of p14ARF has been well addressed, the post-translational modification of p14ARF regulated by oncogenic stress has yet to be investigated. Here, we found that c-Myc increased p14ARF protein stability by inducing the transcription of ubiquitin-specific protease 10 (USP10). USP10, in turn, mediated the deubiquitination of p14ARF, preventing its proteasome-dependent degradation. USP10-null mouse embryonic fibroblasts and human primary cells depleted of USP10 bypassed c-Myc-induced senescence via the destabilization of p14ARF, and these cells displayed accelerated hyper-proliferation and transformation. Clinically the c-Myc-USP10-p14ARF axis was disrupted in non-small cell lung cancer patients, resulting in significantly worse overall survival. Our studies indicate that USP10 induced by c-Myc has a crucial role in OIS by maintaining the stability of key tumor suppressor p14ARF.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Schmitt CA. Senescence, apoptosis and therapy–cutting the lifelines of cancer. Nat Rev Cancer. 2003;3:286–95.
Serrano M, Lin AW, McCurrach ME, Beach D, Lowe SW. Oncogenic ras provokes premature cell senescence associated with accumulation of p53 and p16INK4a. Cell. 1997;88:593–602.
Zindy F, Eischen CM, Randle DH, Kamijo T, Cleveland JL, Sherr CJ, et al. Myc signaling via the ARF tumor suppressor regulates p53-dependent apoptosis and immortalization. Genes Dev. 1998;12:2424–33.
Gil J, Peters G. Regulation of the INK4b-ARF-INK4a tumour suppressor locus: all for one or one for all. Nat Rev Mol Cell Biol. 2006;7:667–77.
Lin AW, Barradas M, Stone JC, van Aelst L, Serrano M, Lowe SW. Premature senescence involving p53 and p16 is activated in response to constitutive MEK/MAPK mitogenic signaling. Genes Dev. 1998;12:3008–19.
Zhang Y, Xiong Y, Yarbrough WG. ARF promotes MDM2 degradation and stabilizesp53: ARF-INK4a locus deletion impairs both the Rb and p53 tumor suppression pathways. Cell. 1998;92:725–34.
Honda R, Yasuda H. Association ofp19(ARF) with Mdm2 inhibits ubiquitin ligase activity of Mdm2 for tumor suppressor p53. EMBO J. 1999;18:22–27.
Weber JD, Taylor LJ, Roussel MF, Sherr CJ, Bar-Sagi D. Nucleolar Arf sequesters Mdm2 and activates p53. Nat Cell Biol. 1999;1:20–26.
Kubbutat MH, Jones SN, Vousden KH. Regulation of p53 stability by Mdm2. Nature. 1997;387:299–303.
Haupt Y, Maya R, Kazaz A, Oren M. Mdm2 promotes the rapid degradation of p53. Nature. 1997;387:296–9.
Eymin B, Claverie P, Salon C, Leduc C, Col E, Brambilla E, et al. p14ARF activates a Tip60-dependent and p53-independent ATM/ATR/CHK pathway in response to genotoxic stress. Mol Cell Biol. 2006;26:4339–50.
Fatyol K, Szalay AA. The p14ARF tumor suppressor protein facilitates nucleolar sequestration of hypoxia-inducible factor-1alpha (HIF-1alpha) and inhibits HIF-1-mediated transcription. J Biol Chem. 2001;276:28421–9.
Ko A, Han SY, Song J. Dynamics of ARF regulation that control senescence and cancer. BMB Rep. 2016;49:598–606.
Kamijo T, Bodner S, van de Kamp E, Randle DH, Sherr CJ. Tumor spectrum in ARF-deficient mice. Cancer Res. 1999;59:2217–22.
Matheu A, Pantoja C, Efeyan A, Criado LM, Martin-Caballero J, Flores JM, et al. Increased gene dosage of Ink4a/Arf results in cancer resistance and normal aging. Genes Dev. 2004;18:2736–46.
Iida S, Akiyama Y, Nakajima T, Ichikawa W, Nihei Z, Sugihara K, et al. Alterations and hypermethylation of thep14(ARF) gene in gastric cancer. Int J Cancer. 2000;87:654–8.
Hsu HS, Wang YC, Tseng RC, Chang JW, Chen JT, Shih CM, et al. 5′ cytosine-phospho-guanine island methylation is responsible for p14ARF inactivation and inversely correlates with p53 overexpression in resected non-small cell lung cancer. Clin Cancer Res: Off J Am Assoc Cancer Res. 2004;10:4734–41.
Silva J, Dominguez G, Silva JM, Garcia JM, Gallego I, Corbacho C, et al. Analysis of genetic and epigenetic processes that influence p14ARF expression in breast cancer. Oncogene. 2001;20:4586–90.
Lee M, Sup Han W, Kyoung Kim O, Hee Sung S, Sun Cho M, Lee SN, et al. Prognostic value of p16INK4a and p14ARF gene hypermethylation in human colon cancer. Pathol Res Pract. 2006;202:415–24.
Komori H, Enomoto M, Nakamura M, Iwanaga R, Ohtani K. Distinct E2F-mediated transcriptional program regulates p14ARF gene expression. EMBO J. 2005;24:3724–36.
Pollice A, Nasti V, Ronca R, Vivo M, Lo Iacono M, Calogero R, et al. Functional and physical interaction of the human ARF tumor suppressor with Tat-binding protein-1. J Biol Chem. 2004;279:6345–53.
Parisi T, Pollice A, Di Cristofano A, Calabro V, La Mantia G. Transcriptional regulation of the human tumor suppressorp14(ARF) by E2F1, E2F2, E2F3, and Sp1-like factors. Biochem Biophys Res Commun. 2002;291:1138–45.
Chen DL, Shan J, Zhu WG, Qin J, Gu W. Transcription-independent ARF regulation in oncogenic stress-mediated p53 responses. Nature. 2010;464:624–U193.
Ko A, Shin JY, Seo J, Lee KD, Lee EW, Lee MS, et al. Acceleration of gastric tumorigenesis through MKRN1-mediated posttranslational regulation of p14ARF. J Natl Cancer. 2012;104:1660–72.
Wang XW, Zha M, Zhao XC, Jiang P, Du WJ, Tam AYH, et al. Siva1 inhibits p53 function by acting as an ARF E3 ubiquitin ligase. Nat Commun. 2013;4:1551.
Yuan J, Luo KT, Zhang LZ, Cheville JC, Lou ZK. USP10 regulates p53 localization and stability by deubiquitinating p53. Cell. 2010;140:384–U121.
Boulkroun S, Ruffieux-Daidie D, Vitagliano JJ, Poirot O, Charles RP, Lagnaz D, et al. Vasopressin-inducible ubiquitin-specific protease 10 increases ENaC cell surface expression by deubiquitylating and stabilizing sorting nexin 3. Am J Physiol-Ren. 2008;295:F889–F900.
Bomberger JM, Barnaby RL, Stanton BA. The deubiquitinating enzyme USP10 regulates the post-endocytic sorting of cystic fibrosis transmembrane conductance regulator in airway epithelial cells. J Biol Chem. 2009;284:18778–89.
Deng M, Yang X, Qin B, Liu TZ, Zhang HX, Guo W, et al. Deubiquitination and activation of AMPK by USP10. Mol Cell. 2016;61:614–24.
Draker R, Sarcinella E, Cheung P. USP10 deubiquitylates the histone variant H2A.Z and both are required for androgen receptor-mediated gene activation. Nucleic Acids Res. 2011;39:3529–42.
Guturi KKN, Bohgaki M, Bohgaki T, Srikumar T, Ng D, Kumareswaran R, et al. RNF168 and USP10 regulate topoisomerase II alpha function via opposing effects on its ubiquitylation. Nat Commun. 2016;7:12638.
Zhang M, Hu C, Tong D, Xiang SY, Williams K, Bai WL, et al. Ubiquitin-specific peptidase 10 (USP10) deubiquitinates and stabilizes MutS Homolog 2 (MSH2) to regulate cellular sensitivity to DNA damage. J Biol Chem. 2016;291:10783–91.
Pomerantz J, Schreiber-Agus N, Liegeois NJ, Silverman A, Alland L, Chin L, et al. The Ink4a tumor suppressor gene product, p19Arf, interacts with MDM2 and neutralizes MDM2’s inhibition of p53. Cell. 1998;92:713–23.
Adhikary S, Eilers M. Transcriptional regulation and transformation by Myc proteins. Nat Rev Mol Cell Biol. 2005;6:635–45.
Wadhwa R, Sugihara T, Hasan MK, Taira K, Reddel RR, Kaul SC. A major functional difference between the mouse and human ARF tumor suppressor proteins. J Biol Chem. 2002;277:36665–70.
Jacobs JJ, Scheijen B, Voncken JW, Kieboom K, Berns A, van Lohuizen M. Bmi-1 collaborates with c-Myc in tumorigenesis by inhibiting c-Myc-induced apoptosis via INK4a/ARF. Genes Dev. 1999;13:2678–90.
Dang CV. MYC on the path to cancer. Cell. 2012;149:22–35.
Ozkara HA, Ozkara S, Topcu S, Criss WE. Amplification of the c-myc oncogene in non-small cell lung cancer. Tumori. 1999;85:508–11.
Mitani S, Kamata H, Fujiwara M, Aoki N, Tango T, Fukuchi K, et al. Analysis of c-myc DNA amplification in non-small cell lung carcinoma in comparison with small cell lung carcinoma using polymerase chain reaction. Clin Exp Med. 2001;1:105–11.
Kuo ML, den Besten W, Bertwistle D, Roussel MF, Sherr CJ. N-terminal polyubiquitination and degradation of the Arf tumor suppressor. Genes Dev. 2004;18:1862–74.
Sung YH, Jin Y, Kim S, Lee HW. Generation of knockout mice using engineered nucleases. Methods. 2014;69:85–93.
Acknowledgements
This work was supported by grants from the Basic Science Research Program of the National Research Foundation of Korea (NRF) funded by the Ministry of Science, ICT and Future Planning (2014R1A1A1002589) and Creative Research Initiative Program of NRF (2015R1A3A2066581) funded by the Ministry of Science, ICT.
Author contributions
JS and AK conceived and designed the projects and wrote the manuscript. AK and SYH performed the experiments. MSL, SYK and HWL provided technical and material support. CHC, HC, JSS, KH, SMH, and JYC performed the clinical analysis.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Edited by R.A. Knight
These authors contributed equally: Aram Ko, Su Yeon Han.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Ko, A., Han, S.Y., Choi, C.H. et al. Oncogene-induced senescence mediated by c-Myc requires USP10 dependent deubiquitination and stabilization of p14ARF. Cell Death Differ 25, 1050–1062 (2018). https://doi.org/10.1038/s41418-018-0072-0
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41418-018-0072-0
This article is cited by
-
c-Myc transactivates CFL1 to induce senescence-like phenotype and potentiate the bystander effects for the migration and proliferation in lung cancer cells
Cell Death Discovery (2026)
-
Ubiquitin-specific protease: an emerging key player in cardiomyopathy
Cell Communication and Signaling (2025)
-
Differential susceptibility and role for senescence in CART cells based on costimulatory domains
Molecular Cancer (2025)
-
Deubiquitinase USP10 promotes osteosarcoma autophagy and progression through regulating GSK3β-ULK1 axis
Cell & Bioscience (2024)
-
Stress-induced epinephrine promotes hepatocellular carcinoma progression via the USP10-PLAGL2 signaling loop
Experimental & Molecular Medicine (2024)