Abstract
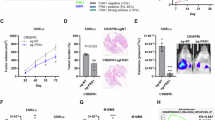
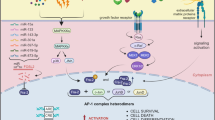
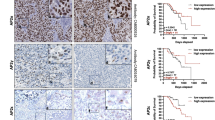
Metastatic melanoma remains a challenging disease. Understanding the molecular mechanisms how melanoma becomes metastatic is therefore of interest. Herein we show that downregulation of the AP-1 transcription factor member Fra-2 in melanoma cells is associated with an aggressive melanoma phenotype in vitro and in vivo. In vitro, Fra-2 knockdown in melanoma cells promoted cell migration and invasion associated with increased Snail-1, Twist-1/2, and matrix metalloproteinase-2 (MMP-2) expression. In vivo, Fra-2 knockdown in a melanoma cell line led to increased metastasis into the lungs and liver. The increased metastatic potential of Fra-2 knockdown melanoma cells was likely due to an accelerated cell cycle transition and increased tissue angiogenesis. Using Fra-2 knockdown cell lines microarray analysis, we identified the protein Fam212b (family with sequence similarity 212 member B) as a downstream target of Fra-2. By additional knockdown of Fam212b in Fra-2 mutant cells, we mitigated the cell migration, invasion, and cell cycle transition phenotype induced by Fra-2 knockdown. Furthermore, Fam212b overexpression enhanced β-catenin pathway. Finally, Fam212b expression is correlated with increased melanoma metastasis and poor clinical outcomes in human patients. In summary, these findings reveal the Fra-2-Fam212b axis as a new pathway of melanoma metastasis, which can be in the future used as potential marker of the metastatic properties of melanoma.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
References
Bhatia S, Tykodi SS, Thompson JA. Treatment of metastatic melanoma: an overview. Oncol (Williston Park). 2009;23:488–96.
Beaver JA, Theoret MR, Mushti S, He K, Libeg M, Goldberg K, et al. FDA approval of nivolumab for the first-line treatment of patients with BRAFV600 wild-type unresectable or metastatic melanoma. Clin Cancer Res. 2017;23:3479–83.
Caramel J, Papadogeorgakis E, Hill L, Browne GJ, Richard G, Wierinckx A, et al. A switch in the expression of embryonic EMT-inducers drives the development of malignant melanoma. Cancer Cell. 2013;24:466–80.
Li FZ, Dhillon AS, Anderson RL, McArthur G, Ferrao PT. Phenotype switching in melanoma: implications for progression and therapy. Front Oncol. 2015;5:31.
Eferl R, Wagner EF. AP-1: a double-edged sword in tumorigenesis. Nat Rev Cancer. 2003;3:859–68.
Yang S, McNulty S, Meyskens FL Jr. During human melanoma progression AP-1 binding pairs are altered with loss of c-Jun in vitro. Pigment Cell Res. 2004;17:74–83.
Obenauf AC, Zou Y, Ji AL, Vanharanta S, Shu W, Shi H, et al. Therapy-induced tumour secretomes promote resistance and tumour progression. Nature. 2015;520:368–72.
Ramsdale R, Jorissen RN, Li FZ, Al-Obaidi S, Ward T, Sheppard KE, et al. The transcription cofactor c-JUN mediates phenotype switching and BRAF inhibitor resistance in melanoma. Sci Signal. 2015;8:ra82.
Verfaillie A, Imrichova H, Atak ZK, Dewaele M, Rambow F, Hulselmans G, et al. Decoding the regulatory landscape of melanoma reveals TEADS as regulators of the invasive cell state. Nat Commun. 2015;6:6683.
Carro MS, Lim WK, Alvarez MJ, Bollo RJ, Zhao X, Snyder EY, et al. The transcriptional network for mesenchymal transformation of brain tumours. Nature. 2010;463:318–25.
Milde-Langosch K, Janke S, Wagner I, Schroder C, Streichert T, Bamberger AM, et al. Role of Fra-2 in breast cancer: influence on tumor cell invasion and motility. Breast Cancer Res Treat. 2008;107:337–47.
Milde-Langosch K, Bamberger AM, Rieck G, Grund D, Hemminger G, Muller V, et al. Expression and prognostic relevance of activated extracellular-regulated kinases (ERK1/2) in breast cancer. Br J Cancer. 2005;92:2206–15.
Nakayama T, Higuchi T, Oiso N, Kawada A, Yoshie O. Expression and function of FRA2/JUND in cutaneous T-cell lymphomas. Anticancer Res. 2012;32:1367–73.
Yang S, Meyskens FL Jr. Alterations in activating protein 1 composition correlate with phenotypic differentiation changes induced by resveratrol in human melanoma. Mol Pharm. 2005;67:298–308.
Iwasaki Y, Yumoto T, Sakakibara S. Expression profiles of inka2 in the murine nervous system. Gene Expr Patterns. 2015;19:83–97.
Yun CY, You ST, Kim JH, Chung JH, Han SB, Shin EY, et al. p21-activated kinase 4 critically regulates melanogenesis via activation of the CREB/MITF and beta-catenin/MITF pathways. J Invest Dermatol. 2015;135:1385–94.
Shin G, Kang TW, Yang S, Baek SJ, Jeong YS, Kim SY. GENT: gene expression database of normal and tumor tissues. Cancer Inf. 2011;10:149–57.
Barrett T, Wilhite SE, Ledoux P, Evangelista C, Kim IF, Tomashevsky M, et al. NCBI GEO: archive for functional genomics data sets-update. Nucleic Acids Res. 2013;41:D991–995.
Kwong LN, Davies MA. Targeted therapy for melanoma: rational combinatorial approaches. Oncogene. 2014;33:1–9.
Talantov D, Mazumder A, Yu JX, Briggs T, Jiang Y, Backus J, et al. Novel genes associated with malignant melanoma but not benign melanocytic lesions. Clin Cancer Res. 2005;11:7234–42.
Riker AI, Enkemann SA, Fodstad O, Liu S, Ren S, Morris C, et al. The gene expression profiles of primary and metastatic melanoma yields a transition point of tumor progression and metastasis. BMC Med Genomics. 2008;1:13.
Xu L, Shen SS, Hoshida Y, Subramanian A, Ross K, Brunet JP, et al. Gene expression changes in an animal melanoma model correlate with aggressiveness of human melanoma metastases. Mol Cancer Res. 2008;6:760–9.
Rappa G, Fodstad O, Lorico A. The stem cell-associated antigen CD133 (Prominin-1) is a molecular therapeutic target for metastatic melanoma. Stem Cells. 2008;26:3008–17.
Poste G, Doll J, Hart IR, Fidler IJ. In vitro selection of murine B16 melanoma variants with enhanced tissue-invasive properties. Cancer Res. 1980;40:1636–44.
Fidler IJ. Biological behavior of malignant melanoma cells correlated to their survival in vivo. Cancer Res. 1975;35:218–24.
Gene Ontology Consortium. The Gene Ontology (GO) project in 2006. Nucleic Acids Res. 2006;34:D322–6.
Draghici S, Khatri P, Tarca AL, Amin K, Done A, Voichita C, et al. A systems biology approach for pathway level analysis. Genome Res. 2007;17:1537–45.
Baskaran Y, Ang KC, Anekal PV, Chan WL, Grimes JM, Manser E, et al. An in cellulo-derived structure of PAK4 in complex with its inhibitor Inka1. Nat Commun. 2015;6:8681.
Kappelmann M, Bosserhoff A, Kuphal S. AP-1/c-Jun transcription factors: regulation and function in malignant melanoma. Eur J Cell Biol. 2014;93:76–81.
Yamanishi DT, Buckmeier JA, Meyskens FL Jr. Expression of c-jun, jun-B, and c-fos proto-oncogenes in human primary melanocytes and metastatic melanomas. J Invest Dermatol. 1991;97:349–53.
Smalley KS, Fedorenko IV. Inhibition of BRAF and BRAF+MEK drives a metastatic switch in melanoma. Mol Cell Oncol. 2015;2:e1008291.
Chiappinelli KB, Strissel PL, Desrichard A, Li H, Henke C, Akman B, et al. Inhibiting DNA methylation causes an interferon response in cancer via dsRNA including endogenous retroviruses. Cell. 2017;169:361.
Wang J, Sun D, Wang Y, Ren F, Pang S, Wang D, et al. FOSL2 positively regulates TGF-beta1 signalling in non-small cell lung cancer. PLoS ONE. 2014;9:e112150.
Nakayama T, Hieshima K, Arao T, Jin Z, Nagakubo D, Shirakawa AK, et al. Aberrant expression of Fra-2 promotes CCR4 expression and cell proliferation in adult T-cell leukemia. Oncogene. 2008;27:3221–32.
Gupta S, Kumar P, Kaur H, Sharma N, Saluja D, Bharti AC, et al. Selective participation of c-Jun with Fra-2/c-Fos promotes aggressive tumor phenotypes and poor prognosis in tongue cancer. Sci Rep. 2015;5:16811.
Schroder C, Schumacher U, Muller V, Wirtz RM, Streichert T, Richter U, et al. The transcription factor Fra-2 promotes mammary tumour progression by changing the adhesive properties of breast cancer cells. Eur J Cancer. 2010;46:1650–60.
Tkach V, Tulchinsky E, Lukanidin E, Vinson C, Bock E, Berezin V. Role of the Fos family members, c-Fos, Fra-1 and Fra-2, in the regulation of cell motility. Oncogene. 2003;22:5045–54.
Hanna SC, Krishnan B, Bailey ST, Moschos SJ, Kuan PF, Shimamura T, et al. HIF1alpha and HIF2alpha independently activate SRC to promote melanoma metastases. J Clin Invest. 2013;123:2078–93.
Luther J, Ubieta K, Hannemann N, Jimenez M, Garcia M, Zech C, et al. Fra-2/AP-1 controls adipocyte differentiation and survival by regulating PPARgamma and hypoxia. Cell Death Differ. 2014;21:655–64.
Deng X, Gao F, May WS Jr. Bcl2 retards G1/S cell cycle transition by regulating intracellular ROS. Blood. 2003;102:3179–85.
Nekrasova T, Minden A. PAK4 is required for regulation of the cell-cycle regulatory protein p21, and for control of cell-cycle progression. J Cell Biochem. 2011;112:1795–806.
Overwijk WW, Restifo NP. B16 as a mouse model for human melanoma. Curr Protoc Immunol. 2001;Chapter 20:Unit 20 21.
Chen GL, Luo Y, Eriksson D, Meng X, Qian C, Bauerle T, et al. High fat diet increases melanoma cell growth in the bone marrow by inducing osteopontin and interleukin 6. Oncotarget. 2016;7:26653–69.
Zhu J, Sanborn JZ, Benz S, Szeto C, Hsu F, Kuhn RM, et al. The UCSC cancer genomics browser. Nat Methods. 2009;6:239–40.
Salt MB, Bandyopadhyay S, McCormick F. Epithelial-to-mesenchymal transition rewires the molecular path to PI3K-dependent proliferation. Cancer Discov. 2014;4:186–99.
Acknowledgements
We thank technicians from Shanghai Zuo Cheng Bio Co., Ltd. for providing excellent support for the immunohistochemistry experiments. This study was supported by grants from the Shanghai Science and Technology Committee, Shanghai International Communication Key Project (14430712000, 14430712001, and 14430712002), the National Natural Science Foundation of China (Nos. 81771729, Nos. 81802362, Nos. 81801593), and the Deutsche Forschungs-gemeinschaft (IZKF projekt D23, A01-CRC1181, microBone-SPP1468; BO3811/1-5, BO3811/1-6, BO3811/1-7, and Emmy Noether).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Edited by S. Fulda
Supplementary information
Rights and permissions
About this article
Cite this article
Chen, GL., Li, R., Chen, XX. et al. Fra-2/AP-1 regulates melanoma cell metastasis by downregulating Fam212b. Cell Death Differ 28, 1364–1378 (2021). https://doi.org/10.1038/s41418-020-00660-4
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41418-020-00660-4
This article is cited by
-
Role of Fra-2 in cancer
Cell Death & Differentiation (2024)
-
Decoding the metastatic potential and optimal postoperative adjuvant therapy of melanoma based on metastasis score
Cell Death Discovery (2023)
-
Age-related self-DNA accumulation may accelerate arthritis in rats and in human rheumatoid arthritis
Nature Communications (2023)
-
Xanthine dehydrogenase as a prognostic biomarker related to tumor immunology in hepatocellular carcinoma
Cancer Cell International (2021)