Abstract
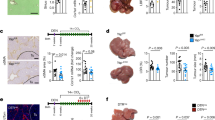
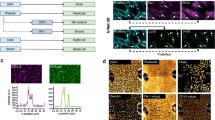
Growing evidence suggests a mechanistic link between steatohepatitis and hepatocellular carcinoma (HCC). However, the lack of representative animal models hampers efforts to understand pathophysiological mechanisms underlying steatohepatitis-related HCC. We found that liver-specific deletion of Ssu72 phosphatase in mice, leads to a high incidence of nonalcoholic fatty liver disease and nonalcoholic steatohepatitis, but not HCC. However, loss of Ssu72 drastically increased the probability of HCC developing, as well as the population of hepatic progenitors, in various chemical and metabolic syndrome-induced HCC models. Importantly, hepatic Ssu72 loss resulted in the induction of mature hepatocyte-to-progenitor cell conversion, by dedifferentiation orchestrated by Ssu72-mediated hypo-phosphorylation of hepatocyte nuclear factor 4α (HNF4α), a master regulator of hepatocyte function. Our findings suggest that Ssu72-mediated HNF4α transcription contributes to the progression of steatohepatitis-associated HCC by regulating the dedifferentiation potential of hepatocytes. Thus, targeting the Ssu72-mediated HNF4α signaling that underlies the pathogenesis of steatohepatitis-associated HCC development could be a novel therapeutic intervention for steatohepatitis-associated HCC.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
Data availability
All data included in this work are presented in the paper and Supplementary Figures and Tables. Additional data relevant to this paper may be provided by the corresponding authors upon request.
References
Cohen JC, Horton JD, Hobbs HH. Human fatty liver disease: old questions and new insights. Science. 2011;332:1519–23.
Niu ZS, Niu XJ, Wang WH. Genetic alterations in hepatocellular carcinoma: an update. World J Gastroenterol. 2016;22:9069–95.
Febbraio MA, Reibe S, Shalapour S, Ooi GJ, Watt MJ, Karin M. Preclinical models for studying NASH-driven HCC: how useful are they? Cell Metab. 2019;29:18–26.
Severi T, van Malenstein H, Verslype C, van Pelt JF. Tumor initiation and progression in hepatocellular carcinoma: risk factors, classification, and therapeutic targets. Acta Pharmacol Sin. 2010;31:1409–20.
Gaudio E, Carpino G, Cardinale V, Franchitto A, Onori P, Alvaro D. New insights into liver stem cells. Digestive Liver Dis. 2009;41:455–62.
Kitade M, Kaji K, Yoshiji H. Relationship between hepatic progenitor cell-mediated liver regeneration and non-parenchymal cells. Hepatol Res. 2016;46:1187–93.
Tarlow BD, Pelz C, Naugler WE, Wakefield L, Wilson EM, Finegold MJ, et al. Bipotential adult liver progenitors are derived from chronically injured mature hepatocytes. Cell Stem Cell. 2014;15:605–18.
He GB, Dhar D, Nakagawa H, Font-Burgada J, Ogata H, Jiang YH, et al. Identification of liver cancer progenitors whose malignant progression depends on autocrine IL-6 signaling. Cell. 2013;155:384–96.
Font-Burgada J, Shalapour S, Ramaswamy S, Hsueh B, Rossell D, Umemura A, et al. Hybrid periportal hepatocytes regenerate the injured liver without giving rise to cancer. Cell. 2015;162:766–79.
Miyaoka Y, Miyajima A. To divide or not to divide: revisiting liver regeneration. Cell Div. 2013;8:8.
Fan B, Malato Y, Calvisi DF, Naqvi S, Razumilava N, Ribback S, et al. Cholangiocarcinomas can originate from hepatocytes in mice. J Clin Investig. 2012;122:2911–5.
Sekiya S, Suzuki A. Intrahepatic cholangiocarcinoma can arise from Notch-mediated conversion of hepatocytes. J Clin Investig. 2012;122:3914–8.
Kim HS, Baek KH, Ha GH, Lee JC, Kim YN, Lee J, et al. The hsSsu72 phosphatase is a cohesin-binding protein that regulates the resolution of sister chromatid arm cohesion. Embo J. 2010;29:3544–57.
Kim HS, Kim SH, Park HY, Lee J, Yoon JH, Choi S, et al. Functional interplay between Aurora B kinase and Ssu72 phosphatase regulates sister chromatid cohesion. Nat Commun. 2013;4:2631.
Kim SH, Jeon Y, Kim HS, Lee JK, Lim HJ, Kang D, et al. Hepatocyte homeostasis for chromosome ploidization and liver function is regulated by Ssu72 protein phosphatase. Hepatology. 2016;63:247–59.
Subramanian A, Tamayo P, Mootha VK, Mukherjee S, Ebert BL, Gillette MA, et al. Gene set enrichment analysis: A knowledge-based approach for interpreting genome-wide expression profiles. Proc Natl Acad Sci USA. 2005;102:15545–50.
Aizarani N, Saviano A, Sagar, Mailly L, Durand S, Herman JS, et al. A human liver cell atlas reveals heterogeneity and epithelial progenitors. Nature. 2019;572:199–204.
Lim E, Wu D, Pal B, Bouras T, Asselin-Labat ML, Vaillant F, et al. Transcriptome analyses of mouse and human mammary cell subpopulations reveal multiple conserved genes and pathways. Breast Cancer Res. 2010;12:1–14.
Walesky C, Edwards G, Borude P, Gunewardena S, O’Neil M, Yoo B, et al. Hepatocyte nuclear factor 4 alpha deletion promotes diethylnitrosamine-induced hepatocellular carcinoma in rodents. Hepatology. 2013;57:2480–90.
Sun K, Montana V, Chellappa K, Brelivet Y, Moras D, Maeda Y, et al. Phosphorylation of a conserved serine in the deoxyribonucleic acid binding domain of nuclear receptors alters intracellular localization. Mol Endocrinol. 2007;21:1297–311.
Vető B, Bojcsuk D, Bacquet C, Kiss J, Sipeki S, Martin L, et al. The transcriptional activity of hepatocyte nuclear factor 4 alpha is inhibited via phosphorylation by ERK1/2. PLoS ONE. 2017;12:e0172020.
Guo HT, Gao CJ, Mi ZY, Wai PY, Kuo PC. Phosphorylation of Ser(158) regulates inflammatory redox-dependent hepatocyte nuclear factor-4 alpha transcriptional activity (Retracted article. See vol. 461, pg. 347, 2014). Biochem J. 2006;394:379–87.
Chellappa K, Jankova L, Schnabl JM, Pan SQ, Brelivet Y, Fung CLS, et al. Src tyrosine kinase phosphorylation of nuclear receptor HNF4 alpha correlates with isoform-specific loss of HNF4 alpha in human colon cancer. Proc Natl Acad Sci USA. 2012;109:2302–7.
Wang ZY, Salih E, Burke PA. Quantitative analysis of cytokine-induced hepatocyte nuclear factor-4 alpha phosphorylation by mass spectrometry. Biochemistry. 2011;50:5292–300.
Fujii M, Shibazaki Y, Wakamatsu K, Honda Y, Kawauchi Y, Suzuki K, et al. A murine model for non-alcoholic steatohepatitis showing evidence of association between diabetes and hepatocellular carcinoma. Med Mol Morphol. 2013;46:141–52.
Kang JS, Wanibuchi H, Morimura K, Gonzalez FJ, Fukushima S. Role of CYP2E1 in diethylnitrosamine-induced hepatocarcinogenesis in vivo. Cancer Res. 2007;67:11141–6.
Halpern KB, Shenhav R, Matcovitch-Natan O, Toth B, Lemze D, Golan M, et al. Single-cell spatial reconstruction reveals global division of labour in the mammalian liver. Nature. 2017;542:352–6.
Miyajima A, Tanaka M, Itoh T. Stem/progenitor cells in liver development, homeostasis, regeneration, and reprogramming. Cell Stem Cell. 2014;14:561–74.
Tatetsu H, Kong NR, Chong G, Amabile G, Tenen DG, Chai L. SALL4, the missing link between stem cells, development and cancer. Gene. 2016;584:111–9.
Oikawa T, Kamiya A, Kakinuma S, Zeniya M, Nishinakamura R, Tajiri H, et al. Sall4 Regulates Cell Fate Decision in Fetal Hepatic Stem/Progenitor Cells. Gastroenterology. 2009;136:1000–11.
Lu H. Crosstalk of HNF4α with extracellular and intracellular signaling pathways in the regulation of hepatic metabolism of drugs and lipids. Acta Pharm Sin B. 2016;6:393–408.
Yeh MM, Bosch DE, Daoud SS. Role of hepatocyte nuclear factor 4-alpha in gastrointestinal and liver diseases. World J Gastroenterol. 2019;25:4074–91.
Grohmann M, Wiede F, Dodd GT, Gurzov EN, Ooi GJ, Butt T, et al. Obesity Drives STAT-1-Dependent NASH and STAT-3-Dependent HCC. Cell. 2018;175:1289–306.e20.
Guest RV, Boulter L, Kendall TJ, Minnis-Lyons SE, Walker R, Wigmore SJ, et al. Cell lineage tracing reveals a biliary origin of intrahepatic cholangiocarcinoma. Cancer Res. 2014;74:1005–10.
Mu X, Español-Suñer R, Mederacke I, Affò S, Manco R, Sempoux C, et al. Hepatocellular carcinoma originates from hepatocytes and not from the progenitor/biliary compartment. J Clin Investig. 2015;125:3891–903.
Seehawer M, Heinzmann F, D’Artista L, Harbig J, Roux PF, Hoenicke L, et al. Necroptosis microenvironment directs lineage commitment in liver cancer. Nature. 2018;562:69–75.
Bellanti F, di Bello G, Iannelli G, Pannone G, Pedicillo MC, Boulter L, et al. Inhibition of nuclear factor (erythroid-derived 2)-like 2 promotes hepatic progenitor cell activation and differentiation. Npj Regen Med. 2021;6:1–12.
Ning BF, Ding J, Yin C, Zhong W, Wu K, Zeng X, et al. Hepatocyte nuclear factor 4 alpha suppresses the development of hepatocellular carcinoma. Cancer Res. 2010;70:7640–51.
Yin C, Lin Y, Zhang X, Chen YX, Zeng X, Yue HY, et al. Differentiation therapy of hepatocellular carcinoma in mice with recombinant adenovirus carrying hepatocyte nuclear factor-4alpha gene. Hepatology. 2008;48:1528–39.
Acknowledgements
We would like to thank Dr. Valentina Factor (National Cancer institute, Bethesda, MD) and Dr. Shunsuke Matsuo (Gunma University, Japan) for kindly providing A6 antibody and Tfr2 promoter constructs, respectively.
Funding
This work was supported by grants [2017R1A2B3006776 (CWL) and 2018R1D1A1A02085616 (HSK)] of National Research Foundation (NSF) funded by the Ministry of Education, Science, and Technology (MEST), Republic of Korea.
Author information
Authors and Affiliations
Contributions
HSK and JSY designed the studies, performed and analyzed data, and wrote the parts of paper; YJ and HL generated conditional KO mice and analyzed data; EJP, JKL, SC, JYP, and HG contributed to specific experiments; CWL designed the studies, supervised the overall project, wrote the paper, and performed the final paper preparation. All authors provided feedback and agreed on the final paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval
All animal experiments were conducted in accordance with guidelines of the IACUC 2018-05-16-2, IACUC 2019-04-31-1, IACUC 2020-08-33-1 and IACUC 2021-02-41-1 of Sungkyunkwan University, an institution accredited by the Association for Assessment and Accreditation of Laboratory Animal Care International. Research with human liver specimens was approved by the Asan Medical Center Institutional Review Board (2019-0636).
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Edited by S. Kumar
Rights and permissions
About this article
Cite this article
Kim, HS., Yoon, JS., Jeon, Y. et al. Ssu72-HNF4α signaling axis classify the transition from steatohepatitis to hepatocellular carcinoma. Cell Death Differ 29, 600–613 (2022). https://doi.org/10.1038/s41418-021-00877-x
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41418-021-00877-x
This article is cited by
-
Identification of alternative splicing events related to fatty liver formation in duck using full-length transcripts
BMC Genomics (2023)
-
Ssu72 phosphatase is essential for thermogenic adaptation by regulating cytosolic translation
Nature Communications (2023)
-
Protein phosphatases regulate the liver microenvironment in the development of hepatocellular carcinoma
Experimental & Molecular Medicine (2022)