Abstract
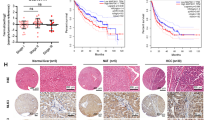
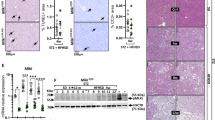
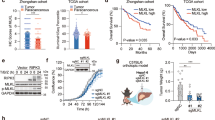
The pseudokinase mixed lineage kinase domain-like (MLKL) is an essential component of the activation of the necroptotic pathway. Emerging evidence suggests that MLKL plays a key role in liver disease. However, how MLKL contributes to hepatocarcinogenesis has not been fully elucidated. Herein, we report that MLKL is upregulated in a diethylnitrosamine (DEN)-induced murine HCC model and is associated with human hepatocellular carcinomas. Hepatocyte-specific MLKL knockout suppresses the progression of hepatocarcinogenesis. Conversely, MLKL overexpression aggravates the initiation and progression of DEN-induced HCC. Mechanistic study reveals that deletion of MLKL significantly increases the activation of autophagy, thereby protecting against hepatocarcinogenesis. MLKL directly interacts with AMPKα1 and inhibits its activity independent of its necroptotic function. Mechanistically, MLKL serves as a bridging molecule between AMPKα1 and protein phosphatase 1B (PPM1B), thus enhancing the dephosphorylation of AMPKα1. Consistently, MLKL expression correlates negatively with AMPKα1 phosphorylation in HCC patients. Taken together, our findings highlight MLKL as a novel AMPK gatekeeper that plays key roles in inhibiting autophagy and driving hepatocarcinogenesis, suggesting that the MLKL-AMPKα1 axis is a potential therapeutic target for HCC.
Similar content being viewed by others
Log in or create a free account to read this content
Gain free access to this article, as well as selected content from this journal and more on nature.com
or
Data availability
The authors declare that the data are present in the paper and/or the supplementary information. Additional data supporting the present study are available from the corresponding author upon reasonable request.
References
Siegel RL, Miller KD, Fuchs HE, Jemal A. Cancer statistics, 2022. CA Cancer J Clin. 2022;72:7–33.
Pinyol R, Torrecilla S, Wang H, Montironi C, Piqué-Gili M, Torres-Martin M, et al. Molecular characterisation of hepatocellular carcinoma in patients with non-alcoholic steatohepatitis. J Hepatol. 2021;75:865–78.
Qian H, Chao X, Williams J, Fulte S, Li T, Yang L, et al. Autophagy in liver diseases: a review. Mol Aspects Med. 2021;82:100973.
Alim Al-Bari A, Ito Y, Thomes PG, Menon MB, García-Macia M, Fadel R, et al. Emerging mechanistic insights of selective autophagy in hepatic diseases. Front Pharmacol. 2023;14:1149809.
Yao J, Tang S, Shi C, Lin Y, Ge L, Chen Q, et al. Isoginkgetin, a potential CDK6 inhibitor, suppresses enhancer activity to induce AMPK-ULK1-mediated cytotoxic autophagy in hepatocellular carcinoma. Autophagy. 2023;19:1221–38.
He L, Li H, Li C, Liu ZK, Lu M, Zhang RY, et al. HMMR alleviates endoplasmic reticulum stress by promoting autophagolysosomal activity during endoplasmic reticulum stress-driven hepatocellular carcinoma progression. Cancer Commun. 2023;43:981–1002.
Kim J, Kundu M, Viollet B, Guan KL. AMPK and mTOR regulate autophagy through direct phosphorylation of Ulk1. Nat Cell Biol. 2011;13:132–41.
Park JM, Kim DH. A paradigm shift: AMPK negatively regulates ULK1 activity. Autophagy. 2024;20:960–2.
Guo S, Zhang C, Zeng H, Xia Y, Weng C, Deng Y, et al. Glycolysis maintains AMPK activation in sorafenib-induced Warburg effect. Mol Metab. 2023;77:101796.
Zhang J, Yang Y, He W, Sun L. Necrosome core machinery: MLKL. Cell Mol Life Sci. 2016;73:2153–63.
Wu J, Huang Z, Ren J, Zhang Z, He P, Li Y, et al. Knockout mice demonstrate the indispensable role of Mlkl in necroptosis. Cell Res. 2013;23:994–1006.
Zhang X, Fan C, Zhang H, Zhao Q, Liu Y, Xu C, et al. MLKL and FADD are critical for suppressing progressive lymphoproliferative disease and activating the NLRP3 inflammasome. Cell Rep. 2016;16:3247–59.
Zhao Q, Yu X, Li M, Liu Y, Han Y, Zhang X, et al. MLKL attenuates colon inflammation and colitis-tumorigenesis via suppression of inflammatory responses. Cancer Lett. 2019;459:100–11.
Cao M, Wu Z, Lou Q, Lu W, Zhang J, Li Q, et al. Dectin-1-induced RIPK1 and RIPK3 activation protects host against infection. Cell Death Differ. 2019;26:2622–36.
Zhao Q, Cheng X, Guo J, Bi Y, Kuang L, Ren J, et al. MLKL inhibits intestinal tumorigenesis by suppressing STAT3 signaling pathway. Int J Biol Sci. 2021;17:869–81.
Guo R, Jia X, Ding Z, Wang G, Jiang M, Li B, et al. Loss of MLKL ameliorates liver fibrosis by inhibiting hepatocyte necroptosis and hepatic stellate cell activation. Theranostics. 2022;12:5220–36.
Xu H, Du X, Liu G, Huang S, Du W, Zou S, et al. The pseudokinase MLKL regulates hepatic insulin sensitivity independently of inflammation. Mol Metab. 2019;23:14–23.
Wu X, Poulsen KL, Sanz-Garcia C, Huang E, McMullen MR, Roychowdhury S, et al. MLKL-dependent signaling regulates autophagic flux in a murine model of non-alcohol-associated fatty liver and steatohepatitis. J Hepatol. 2020;73:616–27.
Jiang X, Deng W, Tao S, Tang Z, Chen Y, Tian M, et al. A RIPK3-independent role of MLKL in suppressing parthanatos promotes immune evasion in hepatocellular carcinoma. Cell Discov. 2023;9:7.
Sun L, Wang H, Wang Z, He S, Chen S, Liao D, et al. Mixed lineage kinase domain-like protein mediates necrosis signaling downstream of RIP3 kinase. Cell. 2012;148:213–27.
Qiu B, Lawan A, Xirouchaki CE, Yi JS, Robert M, Zhang L, et al. MKP1 promotes nonalcoholic steatohepatitis by suppressing AMPK activity through LKB1 nuclear retention. Nat Commun. 2023;14:5405.
Lee A, Kondapalli C, Virga DM, Lewis TL Jr, Koo SY, Ashok A, et al. Aβ42 oligomers trigger synaptic loss through CAMKK2-AMPK-dependent effectors coordinating mitochondrial fission and mitophagy. Nat Commun. 2022;13:4444.
Fu X, Wan S, Lyu YL, Liu LF, Qi H. Etoposide induces ATM-dependent mitochondrial biogenesis through AMPK activation. PLoS ONE. 2008;3:e2009.
Xu X, Ding G, Liu C, Ding Y, Chen X, Huang X, et al. Nuclear UHRF1 is a gate-keeper of cellular AMPK activity and function. Cell Res. 2022;32:54–71.
Wakayama K, Kamiyama T, Yokoo H, Orimo T, Shimada S, Einama T, et al. Huge hepatocellular carcinoma greater than 10 cm in diameter worsens prognosis by causing distant recurrence after curative resection. J Surg Oncol. 2017;115:324–9.
Yang B, Li CL, Guo WH, Qin TQ, Jiao H, Fei ZJ, et al. Intra-arterial ethanol embolization augments response to TACE for treatment of HCC with portal venous tumor thrombus. BMC Cancer. 2018;18:101.
Zhang T, Wang Y, Inuzuka H, Wei W. Necroptosis pathways in tumorigenesis. Semin Cancer Biol. 2022;86:32–40.
Geng L, Gao W, Saiyin H, Li Y, Zeng Y, Zhang Z, et al. MLKL deficiency alleviates neuroinflammation and motor deficits in the α-synuclein transgenic mouse model of Parkinson’s disease. Mol Neurodegener. 2023;18:94.
Zhan C, Huang M, Yang X, Hou J. MLKL: Functions beyond serving as the executioner of necroptosis. Theranostics. 2021;11:4759–69.
Nakatsuka T, Tateishi R. Development and prognosis of hepatocellular carcinoma in patients with diabetes. Clin Mol Hepatol. 2023;29:51–64.
Foerster F, Gairing SJ, Müller L, Galle PR. NAFLD-driven HCC: safety and efficacy of current and emerging treatment options. J Hepatol. 2022;76:446–57.
Galluzzi L, Pietrocola F, Bravo-San Pedro JM, Amaravadi RK, Baehrecke EH, Cecconi F, et al. Autophagy in malignant transformation and cancer progression. EMBO J. 2015;34:856–80.
Li J, Zhan H, Ren Y, Feng M, Wang Q, Jiao Q, et al. Sirtuin 4 activates autophagy and inhibits tumorigenesis by upregulating the p53 signaling pathway. Cell Death Differ. 2023;30:313–26.
Li GM, Li L, Li MQ, Chen X, Su Q, Deng ZJ, et al. DAPK3 inhibits gastric cancer progression via activation of ULK1-dependent autophagy. Cell Death Differ. 2021;28:952–67.
Guha P, Tyagi R, Chowdhury S, Reilly L, Fu C, Xu R, et al. IPMK mediates activation of ULK signaling and transcriptional regulation of autophagy linked to liver inflammation and regeneration. Cell Rep. 2019;26:2692–703.
Jang HJ, Lee YH, Dao T, Jo Y, Khim KW, Eom HJ, et al. Thrap3 promotes nonalcoholic fatty liver disease by suppressing AMPK-mediated autophagy. Exp Mol Med. 2023;55:1720–33.
Ponnusamy L, Natarajan SR, Thangaraj K, Manoharan R. Therapeutic aspects of AMPK in breast cancer: Progress, challenges, and future directions. Biochim Biophys Acta Rev Cancer. 2020;1874:188379.
Wang Y, Yang Z, Zheng G, Yu L, Yin Y, Mu N, et al. Metformin promotes autophagy in ischemia/reperfusion myocardium via cytoplasmic AMPKα1 and nuclear AMPKα2 pathways. Life Sci. 2019;225:64–71.
Huang Z, Fang W, Liu W, Wang L, Liu B, Liu S, et al. Aspirin induces Beclin-1-dependent autophagy of human hepatocellular carcinoma cell. Eur J Pharmacol. 2018;823:58–64.
Zong Y, Zhang CS, Li M, Wang W, Wang Z, Hawley SA, et al. Hierarchical activation of compartmentalized pools of AMPK depends on severity of nutrient or energy stress. Cell Res. 2019;29:460–73.
Voss M, Paterson J, Kelsall IR, Martín-Granados C, Hastie CJ, Peggie MW, et al. Ppm1E is an AMP-activated protein kinase phosphatase. Cell Signal. 2011;23:114–24.
Ruiz A, Xu X, Carlson M. Ptc1 protein phosphatase 2C contributes to glucose regulation of SNF1/AMP-activated protein kinase (AMPK) in Saccharomyces cerevisiae. J Biol Chem. 2013;288:31052–8.
Tang Y, Fang G, Guo F, Zhang H, Chen X, An L, et al. Selective inhibition of STRN3-containing PP2A phosphatase restores hippo tumor-suppressor activity in gastric cancer. Cancer Cell. 2020;38:115–28.
Tasdelen I, van Beekum O, Gorbenko O, Fleskens V, van den Broek NJ, Koppen A, et al. The serine/threonine phosphatase PPM1B (PP2Cβ) selectively modulates PPARγ activity. Biochem J. 2013;451:45–53.
Guo FX, Wu Q, Li P, Zheng L, Ye S, Dai XY, et al. The role of the LncRNA-FA2H-2-MLKL pathway in atherosclerosis by regulation of autophagy flux and inflammation through mTOR-dependent signaling. Cell Death Differ. 2019;26:1670–87.
Günther C, He GW, Kremer AE, Murphy JM, Petrie EJ, Amann K, et al. The pseudokinase MLKL mediates programmed hepatocellular necrosis independently of RIPK3 during hepatitis. J Clin Invest. 2016;126:4346–60.
Lawlor KE, Khan N, Mildenhall A, Gerlic M, Croker BA, D’Cruz AA, et al. RIPK3 promotes cell death and NLRP3 inflammasome activation in the absence of MLKL. Nat Commun. 2015;6:6282.
Zhou H, Zhou L, Guan Q, Hou X, Wang C, Liu L, et al. Skp2-mediated MLKL degradation confers cisplatin-resistant in non-small cell lung cancer cells. Commun Biol. 2023;6:805.
Liu Z, Dagley LF, Shield-Artin K, Young SN, Bankovacki A, Wang X, et al. Oligomerization-driven MLKL ubiquitylation antagonizes necroptosis. EMBO J. 2021;40:e103718.
Funding
This work was supported by the National Natural Science Foundation of China (81902852, 82003237), Natural Science Foundation of Hubei Province of China (2022CFB481), Natural Science Foundation of Hubei Provincial Department of Education (T2022021), Projects of International Cooperation and Exchanges (G2022027004L, G2022027012L) and the Advantages Discipline Group (Medicine) Project in Higher Education of Hubei Province (2021-2025) (2024XKQY26, 2024BMXKQY2). The authors appreciate Professor Quentin Qiang Liu and Professor Mian Wu for their helpful suggestions on this study. The authors thank the Biomedical Research Institute of Hubei University of Medicine for instrument support.
Author information
Authors and Affiliations
Contributions
XY and QZ conceived the project and designed the research studies. QZ, XY, MF, JG, and HW performed most of the experiments described. JY performed structure analysis. AZ, and JW provided help with animal and technical assistance in the mouse experiments. YH, ZS and YL provided conceptual advice and helpful discussion. XY, QZ and MF analyzed data. XY and QZ prepared the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
The study was approved by the Science and Technology Ethics Committee of Hubei University of Medicine (No. 2023-EER-05). Animal experiments were approved by the the Animal Care and Use Committee of Hubei Medicine University (No. 2022-021) and animal care was conducted in accordance with institutional guidelines.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yu, X., Feng, M., Guo, J. et al. MLKL promotes hepatocarcinogenesis through inhibition of AMPK-mediated autophagy. Cell Death Differ 31, 1085–1098 (2024). https://doi.org/10.1038/s41418-024-01314-5
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41418-024-01314-5
This article is cited by
-
MLKL depletion enhances chemotherapy-induced apoptosis in colorectal cancer by prolonged retention of TNFR-I in endosomes
Apoptosis (2026)
-
Regulated necrosis at the crossroads of liver inflammation and cancer development
Nature Reviews Gastroenterology & Hepatology (2025)