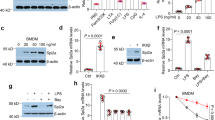
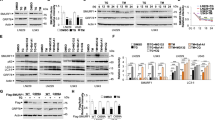
Abstract
Sepsis, a devastating microbe-induced inflammatory response, culminates in multi-organ dysfunction, with pyroptosis mediated by the non-canonical inflammasome being a pivotal factor. The mouse Caspase-11, central to this pathway, is directly activated by cytoplasmic lipopolysaccharide (LPS). Although ubiquitination is known to tightly regulate the inflammatory response in pyroptosis, its role in modulating the non-canonical inflammasome remains enigmatic. In this study, we unveil that the E3 ubiquitin ligase Smurf1 is a critical negative regulator of the non-canonical inflammasome pathway. Smurf1 orchestrates K48-linked polyubiquitination of Caspase-11 at K245 and K247 residues, leading to its degradation via the 26S proteasome. This process is further amplified by ERK phosphorylation of Smurf1 at the S148 site. In parallel, Caspase-11 modulates Smurf1 protein content through cleavage. Notably, macrophage-specific Smurf1 deficiency exacerbates sepsis-induced mortality in mice, attributed to the hyperactivation of the non-canonical inflammasome. Conversely, targeted supplementation of Smurf1 in macrophages mitigates the high mortality and inflammatory response associated with sepsis. Thus, Smurf1 emerges as a key player in modulating the activation of the non-canonical inflammasome in response to Gram-negative bacterial infections.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
Data availability
Data supporting the present study are available from the corresponding author upon reasonable request.
References
Kaukonen KM, Bailey M, Pilcher D, Cooper DJ, Bellomo R. Systemic inflammatory response syndrome criteria in defining severe sepsis. N Engl J Med. 2015;372:1629–38.
Kumar V. Inflammasomes: Pandora’s box for sepsis. J Inflamm Res. 2018;11:477–502.
Ramachandran G. Gram-positive and gram-negative bacterial toxins in sepsis: a brief review. Virulence. 2014;5:213–8.
Kayagaki N, Stowe IB, Lee BL, O’Rourke K, Anderson K, Warming S, et al. Caspase-11 cleaves gasdermin D for non-canonical inflammasome signalling. Nature. 2015;526:666–71.
Yang D, He Y, Muñoz-Planillo R, Liu Q, Núñez G. Caspase-11 requires the Pannexin-1 channel and the purinergic P2X7 pore to mediate pyroptosis and endotoxic shock. Immunity. 2015;43:923–32.
Moretti J, Jia B, Hutchins Z, Roy S, Yip H, Wu J, et al. Caspase-11 interaction with NLRP3 potentiates the noncanonical activation of the NLRP3 inflammasome. Nat Immunol. 2022;23:705–17.
Tang Y, Wang X, Li Z, He Z, Yang X, Cheng X, et al. Heparin prevents caspase-11-dependent septic lethality independent of anticoagulant properties. Immunity. 2021;54:454–456.e6.
Silva CMS, Wanderley CWS, Veras FP, Sonego F, Nascimento DC, Gonçalves AV, et al. Gasdermin D inhibition prevents multiple organ dysfunction during sepsis by blocking NET formation. Blood. 2021;138:2702–13.
Deng M, Tang Y, Li W, Wang X, Zhang R, Zhang X, et al. The endotoxin delivery protein HMGB1 mediates caspase-11-dependent lethality in sepsis. Immunity. 2018;49:740–753.e7.
Bhoj VG, Chen ZJ. Ubiquitylation in innate and adaptive immunity. Nature. 2009;458:430–7.
Heaton SM, Borg NA, Dixit VM. Ubiquitin in the activation and attenuation of innate antiviral immunity. J Exp Med. 2016;213:1–13.
Pickart CM. Mechanisms underlying ubiquitination. Annu Rev Biochem. 2001;70:503–33.
Sluimer J, Distel B. Regulating the human HECT E3 ligases. Cell Mol Life Sci. 2018;75:3121–41.
Shah SS, Kumar S. Adaptors as the regulators of HECT ubiquitin ligases. Cell Death Differ. 2021;28:455–72.
Cockram PE, Kist M, Prakash S, Chen SH, Wertz IE, Vucic D. Ubiquitination in the regulation of inflammatory cell death and cancer. Cell Death Differ. 2021;28:591–605.
Bednash JS, Mallampalli RK. Regulation of inflammasomes by ubiquitination. Cell Mol Immunol. 2016;13:722–8.
Song H, Liu B, Huai W, Yu Z, Wang W, Zhao J, et al. The E3 ubiquitin ligase TRIM31 attenuates NLRP3 inflammasome activation by promoting proteasomal degradation of NLRP3. Nat Commun. 2016;7:13727.
Guan K, Wei C, Zheng Z, Song T, Wu F, Zhang Y, et al. MAVS promotes inflammasome activation by targeting ASC for K63-linked ubiquitination via the E3 Ligase TRAF3. J Immunol. 2015;194:4880–90.
Zhang L, Liu Y, Wang B, Xu G, Yang Z, Tang M, et al. POH1 deubiquitinates pro-interleukin-1β and restricts inflammasome activity. Nat Commun. 2018;9:4225.
Liu Q, Zhang S, Sun Z, Guo X, Zhou H. E3 ubiquitin ligase Nedd4 is a key negative regulator for non-canonical inflammasome activation. Cell Death Differ. 2019;26:2386–99.
Lin Z, Li S, Xiao H, Xu Z, Li C, Zeng J, et al. The degradation of TGR5 mediated by Smurf1 contributes to diabetic nephropathy. Cell Rep. 2023;42:112851.
Souza-Costa LP, Andrade-Chaves JT, Andrade JM, Costa VV, Franco LH. Uncovering new insights into the role of the ubiquitin ligase Smurf1 on the regulation of innate immune signaling and resistance to infection. Front Immunol. 2023;14:1185741.
Roskoski R Jr. ERK1/2 MAP kinases: structure, function, and regulation. Pharmacol Res. 2012;66:105–43.
Lucas RM, Luo L, Stow JL. ERK1/2 in immune signalling. Biochem Soc Trans. 2022;50:1341–52.
Wen M, Ma X, Cheng H, Jiang W, Xu X, Zhang Y, et al. Stk38 protein kinase preferentially inhibits TLR9-activated inflammatory responses by promoting MEKK2 ubiquitination in macrophages. Nat Commun. 2015;6:7167.
Zhao X, Xie J, Duan C, Wang L, Si Y, Liu S, et al. ADAR1 protects pulmonary macrophages from sepsis-induced pyroptosis and lung injury through miR-21/A20 signaling. Int J Biol Sci. 2024;20:464–85.
Ma Y, Zhao R, Guo H, Tong Q, Langdon WY, Liu W, et al. Cytosolic LPS-induced caspase-11 oligomerization and activation is regulated by extended synaptotagmin 1. Cell Rep. 2023;42:112726.
Zheng J, Shi Z, Yang P, Zhao Y, Tang W, Ye S, et al. ERK-Smurf1-RhoA signaling is critical for TGFβ-drived EMT and tumor metastasis. Life Sci Alliance. 2022;5:e202101330.
Kayagaki N, Wong MT, Stowe IB, Ramani SR, Gonzalez LC, Akashi-Takamura S, et al. Noncanonical inflammasome activation by intracellular LPS independent of TLR4. Science. 2013;341:1246–9.
Kang WS, Kwon JS, Kim HB, Jeong HY, Kang HJ, Jeong MH, et al. A macrophage-specific synthetic promoter for therapeutic application of adiponectin. Gene Ther. 2014;21:353–62.
Xu J, Gao C, He Y, Fang X, Sun D, Peng Z, et al. NLRC3 expression in macrophage impairs glycolysis and host immune defense by modulating the NF-κB-NFAT5 complex during septic immunosuppression. Mol Ther. 2023;31:154–73.
Choudhury SR, Babes L, Rahn JJ, Ahn BY, Goring KR, King JC, et al. Dipeptidase-1 is an adhesion receptor for neutrophil recruitment in lungs and liver. Cell. 2019;178:1205–1221.e17.
Shi J, Zhao Y, Wang Y, Gao W, Ding J, Li P, et al. Inflammatory caspases are innate immune receptors for intracellular LPS. Nature. 2014;514:187–92.
Cao Y, Zhang L. A Smurf1 tale: function and regulation of an ubiquitin ligase in multiple cellular networks. Cell Mol Life Sci. 2013;70:2305–17.
Orvedahl A, Sumpter R Jr, Xiao G, Ng A, Zou Z, et al. Image-based genome-wide siRNA screen identifies selective autophagy factors. Nature. 2011;480:113–7.
Franco LH, Nair VR, Scharn CR, Xavier RJ, Torrealba JR, Shiloh MU, et al. The Ubiquitin Ligase Smurf1 functions in selective autophagy of mycobacterium tuberculosis and anti-tuberculous host defense. Cell Host Microbe. 2017;21:59–72.
Lee YS, Park JS, Kim JH, Jung SM, Lee JY, Kim SJ, et al. Smad6-specific recruitment of Smurf E3 ligases mediates TGF-β1-induced degradation of MyD88 in TLR4 signalling. Nat Commun. 2011;2:460.
Qian G, Ren Y, Zuo Y, Yuan Y, Zhao P, Wang X, et al. Smurf1 represses TNF-α production through ubiquitination and destabilization of USP5. Biochem Biophys Res Commun. 2016;474:491–6.
Wang P, Li Y, Sun Y, Xu T. EFHD2 cooperates with E3 ubiquitin ligase Smurf1 to facilitate virus infection by promoting the degradation of TRAF6 in teleost fish. J Virol. 2024;98:e0117623.
Yang W, Bai X, Luan X, Min J, Tian X, Li H, et al. Delicate regulation of IL-1β-mediated inflammation by cyclophilin A. Cell Rep. 2022;38:110513.
Eltobgy MM, Zani A, Kenney AD, Estfanous S, Kim E, Badr A, et al. Caspase-4/11 exacerbates disease severity in SARS-CoV-2 infection by promoting inflammation and immunothrombosis. Proc Natl Acad Sci USA. 2022;119:e2202012119.
Harvey KF, Harvey NL, Michael JM, Parasivam G, Waterhouse N, Alnemri ES, et al. Caspase-mediated cleavage of the ubiquitin-protein ligase Nedd4 during apoptosis. J Biol Chem. 1998;273:13524–30.
Song L, Luo ZQ. Post-translational regulation of ubiquitin signaling. J Cell Biol. 2019;218:1776–86.
Persaud A, Alberts P, Mari S, Tong J, Murchie R, Maspero E, et al. Tyrosine phosphorylation of NEDD4 activates its ubiquitin ligase activity. Sci Signal. 2014;7:ra95.
Singer M, Deutschman CS, Seymour CW, Shankar-Hari M, Annane D, Bauer M, et al. The third international consensus definitions for Sepsis and Septic Shock (Sepsis-3). JAMA. 2016;315:801–10.
Martínez-Zamudio RI, Dewald HK, Vasilopoulos T, Gittens-Williams L, Fitzgerald-Bocarsly P, Herbig U. Senescence-associated β-galactosidase reveals the abundance of senescent CD8+ T cells in aging humans. Aging Cell. 2021;20:e13344.
Zhu CL, Xie J, Liu Q, Wang Y, Li HR, Yu CM, et al. PD-L1 promotes GSDMD-mediated NET release by maintaining the transcriptional activity of Stat3 in sepsis-associated encephalopathy. Int J Biol Sci. 2023;19:1413–29.
Matute-Bello G, Downey G, Moore BB, Groshong SD, Matthay MA, Slutsky AS, et al. An official American Thoracic Society workshop report: features and measurements of experimental acute lung injury in animals. Am J Respir Cell Mol Biol. 2011;44:725–38.
Funding
This work was supported by the National Natural Science Foundation of China (No. 32400760, 82572494), Shanghai Industrial Collaborative Innovation Project (No. HCXBCY-2023-041, HCXBCY-2024-033, XTCX-KJ-2024-39), Shanghai Oriental Talents Program (Outstanding Talents Project), 2024 Medical Technology Key Research Project of the Chinese Medical Education Association (No. 2024KTZ011), and 2024 Basic Medicine Innovation Open Project of Naval Medical University (supported by National Key Laboratory of Immunity and Inflammation, No. JCKFKT-MS-002), Naval Medical University Key Cultivation Program for Outstanding Doctoral Dissertations, and partner laboratory of basic medicine school of Naval Medical University (JCHZJL-009).
Author information
Authors and Affiliations
Contributions
ZZ, YY, SX, and YT designed and supervised the research. CZ, WZ, YL, RJ, LC, YW, TZ, and YT performed research. CZ, WZ, and YL collected clinical samples and performed analyses. LC and RJ performed bioinformatics analyses. ZZ, YY, SX, and YT provided financial support. ZZ, YY, SX, and CZ analyzed data and wrote the paper. CZ, WZ, and YL contributed equally.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
All animal experiments were approved by the Animal Care Committee of Naval Medical University. PBMC sample collection was approved by Biomedical Research Ethics Committee of Naval Medical University. The informed consent was obtained from all participants. This study was conducted in accordance with all relevant guidelines and regulations.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhu, C., Zhang, W., Liao, Y. et al. ERK/Smurf1 regulates non-canonical pyroptosis by prompting Caspase-11 ubiquitination. Cell Death Differ (2026). https://doi.org/10.1038/s41418-025-01654-w
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41418-025-01654-w