Abstract
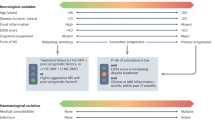
Current treatments for neuromyelitis optica spectrum disorder (NMOSD) highlight recurrence management, while little attention is paid to the relief of residual neurological dysfunction. Here we aimed to evaluate the safety and efficacy of human umbilical cord-derived mesenchymal stromal cells (hUC-MSCs) in reducing relapses and mitigating neurological impairments. This trial, hUC-MSC-NMOSD (ChiCTR-INR-16008037), a single-arm, dose-escalation, open-label study, included 31 NMOSD patients of three dose groups received four infusions every three months, with 15-month follow-up. Primary outcome was time to first recurrence; secondary outcomes focused on clinical scores, MRI lesions and exploratory findings. HUC-MSC infusion was well tolerated in all groups of patients. Adverse events were mostly mild, with urinary tract infections being the most common. Severe adverse events were rare and unrelated to the treatment. The median relapse-free interval increased significantly post-treatment from 305 (95%CI 226-382·5) to 760 (589-1016·5) (p < 0·001), especially in the medium- and high-dose groups. During the two years before and after therapy, the mean Annualized Relapse Rate (ARR) dropped considerably from 1 (0·75-1) to 0 (0-0·5) (p < 0·001). Clinical scores improved in the low and medium-dose groups. The total volume of high-signal white matter lesions in brain significantly decreased after therapy from 4144·5 (2857·2-5508·6) to 2914·4 (2453-3684·11) (p = 0·016). Exploratory single-cell RNA sequencing and metabolomics detection revealed a potential participation of thioredoxin and oxidative phosphorylation (OXPHOS)-mediated boosting of Treg differentiation and suppressive capacity. This trial indicates that intravenous hUC-MSC administration is safe and shows potential efficacy in treating NMOSD. Medium dose might be the best possible compromise between safety and effectiveness.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Code availability
ScRNA-sequencing data have been deposited to National Center for Biotechnology Information (NCBI) under the BioProject number PRJNA1402125. All data reported in our research will be shared by the lead contact upon request. Our research does not report original code. Any additional information required to reanalyze the data reported in this paper is available from the lead contact upon request.
References
Wingerchuk DM, Lucchinetti CF. Neuromyelitis optica spectrum disorder. N Engl J Med. 2022;387:631–9.
Jarius S, Ruprecht K, Kleiter I, Paul F, Aktas O, Asgari N, et al. MOG-IgG in NMO and related disorders: a multicenter study of 50 patients. Part 2: epidemiology, clinical presentation, radiological and laboratory features, treatment responses, and long-term outcome. J Neuroinflammation. 2016;13:280.
Mealy MA, Wingerchuk DM, Palace J, Greenberg BM, Levy M. Comparison of relapse and treatment failure rates among patients with neuromyelitis optica: multicenter study of treatment efficacy. JAMA Neurol. 2014;71:324–30.
Zhang C, Zhang M, Qiu W, Ma H, Zhang X, Zhu Z, et al. Safety and efficacy of tocilizumab versus azathioprine in highly relapsing neuromyelitis optica spectrum disorder (TANGO): an open-label, multicentre, randomised, phase 2 trial. Lancet Neurol. 2020;19:391–401.
Cree BAC, Kim HJ, Weinshenker BG, Bennett JL, Jiao Y, Paul F, et al. Safety and efficacy of inebilizumab for the treatment of neuromyelitis optica spectrum disorder: end-of-study results from the open-label period of the N-MOmentum trial. Lancet Neurol. 2024;23:588–602.
Ringelstein M, Asseyer S, Lindenblatt G, Schmidt-Pogoda A, Luessi F, Kraemer M, et al. Eculizumab use in neuromyelitis optica spectrum disorders: routine clinical care data from a European cohort. Neurology. 2024;103:e209888.
Kotani T, Saito T, Suzuka T, Matsuda S. Adipose-derived mesenchymal stem cell therapy for connective tissue diseases and complications. Inflamm Regen. 2024;44:35.
Song N, Scholtemeijer M, Shah K. Mesenchymal stem cell immunomodulation: mechanisms and therapeutic potential. Trends Pharmacol Sci. 2020;41:653–64.
Vellasamy S, Sandrasaigaran P, Vidyadaran S, George E, Ramasamy R. Mesenchymal stem cells of human placenta and umbilical cord suppress T-cell proliferation at G0 phase of cell cycle. Cell Biol Int. 2013;37:250–6.
Li X, Xu Z, Bai J, Yang S, Zhao S, Zhang Y, et al. Umbilical cord tissue-derived mesenchymal stem cells induce T lymphocyte apoptosis and cell cycle arrest by expression of indoleamine 2, 3-dioxygenase. Stem Cells Int. 2016;2016:7495135.
Chen QH, Wu F, Liu L, Chen HB, Zheng RQ, Wang HL, et al. Mesenchymal stem cells regulate the Th17/Treg cell balance partly through hepatocyte growth factor in vitro. Stem Cell Res Ther. 2020;11:91.
Franco da Cunha F, Andrade-Oliveira V, Candido de Almeida D, Borges da Silva T, Naffah de Souza Breda C, Costa Cruz M, et al. Extracellular vesicles isolated from mesenchymal stromal cells modulate CD4+ T lymphocytes toward a regulatory profile. Cells 2020;9:1059.
Corcione A, Benvenuto F, Ferretti E, Giunti D, Cappiello V, Cazzanti F, et al. Human mesenchymal stem cells modulate B-cell functions. Blood. 2006;107:367–72.
Asari S, Itakura S, Ferreri K, Liu CP, Kuroda Y, Kandeel F, et al. Mesenchymal stem cells suppress B-cell terminal differentiation. Exp Hematol. 2009;37:604–15.
Beyth S, Borovsky Z, Mevorach D, Liebergall M, Gazit Z, Aslan H, et al. Human mesenchymal stem cells alter antigen-presenting cell maturation and induce T-cell unresponsiveness. Blood. 2005;105:2214–9.
Jiang W, Zhu F, Xu H, Cheng M, Zhang Z, Wang Y, et al. CHI3L1 signaling impairs hippocampal neurogenesis and cognitive function in autoimmune-mediated neuroinflammation. Sci Adv. 2023;9:eadg8148.
Xue C, Yu H, Pei X, Yang Y, Chen R, Liu Y, et al. Efficacy of human umbilical cord mesenchymal stem cell in the treatment of neuromyelitis optica spectrum disorders: an animal study. Stem Cell Res Ther. 2025;16:51.
Zhang J, Buller BA, Zhang ZG, Zhang Y, Lu M, Morris D, et al. Exosomes derived from bone marrow mesenchymal stromal cells promote remyelination and reduce neuroinflammation in the demyelinating central nervous system. Exp Neurol. 2022;347:113895.
Li JF, Yin HL, Shuboy A, Duan HF, Lou JY, Li J, et al. Differentiation of hUC-MSC into dopaminergic-like cells after transduction with hepatocyte growth factor. Mol Cell Biochem. 2013;381:183–90.
Wang LT, Ting CH, Yen ML, Liu KJ, Sytwu HK, Wu KK, et al. Human mesenchymal stem cells (MSCs) for treatment towards immune- and inflammation-mediated diseases: review of current clinical trials. J Biomed Sci. 2016;23:76.
Yao XY, Xie L, Cai Y, Tang J, Zhu J, Qiu W, et al. Human umbilical cord mesenchymal stem cells to treat neuromyelitis optica spectrum disorder (hUC-MSC-NMOSD): a study protocol for a prospective, multicenter, randomized, placebo-controlled clinical trial. Front Neurol. 2022;13:860083.
Dura B, Choi JY, Zhang K, Damsky W, Thakral D, Bosenberg M, et al. scFTD-seq: freeze-thaw lysis based, portable approach toward highly distributed single-cell 3’ mRNA profiling. Nucleic Acids Res. 2019;47:e16.
Satija R, Farrell JA, Gennert D, Schier AF, Regev A. Spatial reconstruction of single-cell gene expression data. Nat Biotechnol. 2015;33:495–502.
Korsunsky I, Millard N, Fan J, Slowikowski K, Zhang F, Wei K, et al. Fast, sensitive and accurate integration of single-cell data with Harmony. Nat Methods. 2019;16:1289–96.
Yuan M, Breitkopf SB, Yang X, Asara JM. A positive/negative ion-switching, targeted mass spectrometry-based metabolomics platform for bodily fluids, cells, and fresh and fixed tissue. Nat Protoc. 2012;7:872–81.
Dunn WB, Broadhurst D, Begley P, Zelena E, Francis-McIntyre S, Anderson N, et al. Procedures for large-scale metabolic profiling of serum and plasma using gas chromatography and liquid chromatography coupled to mass spectrometry. Nat Protoc. 2011;6:1060–83.
Fu Y, Yan Y, Qi Y, Yang L, Li T, Zhang N, et al. Impact of autologous mesenchymal stem cell infusion on neuromyelitis optica spectrum disorder: a pilot, 2-year observational study. CNS Neurosci Ther. 2016;22:677–85.
Lu Z, Ye D, Qian L, Zhu L, Wang C, Guan D, et al. Human umbilical cord mesenchymal stem cell therapy on neuromyelitis optica. Curr Neurovasc Res. 2012;9:250–5.
Wang L, Zhang Z, Xu R, Yang X, Zhang L, Guan Y, et al. Human umbilical cord mesenchymal stem cell transfusion in immune non-responders with AIDS: a multicenter randomized controlled trial. Signal Transduct Target Ther. 2021;6:217.
Sengun E, Wolfs T, van Bruggen VLE, Maas M, van der Laan LJW, van Ijcken WFJ. Umbilical cord-mesenchymal stem cells induce a memory phenotype in CD4+ T cells. Front Immunol. 2023;14:1128359.
Wang D, Li J, Zhang Y, Zhang M, Chen J, Li X, et al. Umbilical cord mesenchymal stem cell transplantation in active and refractory systemic lupus erythematosus: a multicenter clinical study. Arthritis Res Ther. 2014;16:R79.
Cao Z, Wang D, Jing L, Wang L, Zhao J, Yang C, et al. Allogenic umbilical cord-derived mesenchymal stromal cells sustain long-term therapeutic efficacy compared with low-dose interleukin-2 in systemic lupus erythematosus. Stem Cells Transl Med. 2023;12:431–43.
Carnero Contentti E, Correale J. Neuromyelitis optica spectrum disorders: from pathophysiology to therapeutic strategies. J Neuroinflammation. 2021;18:208.
Li C, Wu F, Mao J, Dong N, Li M, Jiang T, et al. Mesenchymal stem cells-derived extracellular vesicles ameliorate lupus nephritis by regulating T and B cell responses. Stem Cell Res Ther. 2024;15:216.
You MJ, Bang M, Park HS, Lee HJ, Lee JY, Kim HT, et al. Human umbilical cord-derived mesenchymal stem cells alleviate schizophrenia-relevant behaviors in amphetamine-sensitized mice by inhibiting neuroinflammation. Transl Psychiatry. 2020;10:123.
Hu X, Liu L, Wang Y, Li Q, Xu H, Li W, et al. Human umbilical cord-derived mesenchymal stem cells alleviate acute lung injury caused by severe burn via secreting TSG-6 and inhibiting inflammatory response. Stem Cells Int. 2022;2022:8661689.
Dong N, Zhou PP, Li D, Zhao J, Li C, Chen Y, et al. Intratracheal administration of umbilical cord-derived mesenchymal stem cells attenuates hyperoxia-induced multi-organ injury via heme oxygenase-1 and JAK/STAT pathways. World J Stem Cells. 2022;14:556–76.
Angelin A, Gil-de-Gómez L, Dahiya S, Jiao J, Guo L, Levine MH, et al. Foxp3 reprograms T cell metabolism to function in low-glucose, high-lactate environments. Cell Metab. 2017;25:1282–93.
Mougiakakos D, Johansson CC, Jitschin R, Böttcher M, Kiessling R. Increased thioredoxin-1 production in human naturally occurring regulatory T cells confers enhanced tolerance to oxidative stress. Blood. 2011;117:857–61.
Bradford HF, McDonnell TCR, Stewart A, Lee JY, Graca L, Tsokos GC, et al. Thioredoxin is a metabolic rheostat controlling regulatory B cells. Nat Immunol. 2024;25:873–85.
Acknowledgements
We would like to thank all the staff and patients involved with the study. We would like to acknowledge the role of the Clinical Research Centre, Ren Ji Hospital, in supporting the ongoing delivery of the trial at the sites. We are grateful to all the staff of the Clinical Research Center of School of Medicine, Shanghai Jiao Tong University for their support in designing the present study protocol. This study was funded by National Natural Science Foundation of China (82071341, 72204161); New Quality Clinical Specialty Program of High-end Medical Disciplinary Construction in Shanghai Pudong New Area (2024-PWXZ-16); The Investigator-initiated Trial Program of Shanghai Pudong New Area Health Commission (the Medical and Industrial Integration Program, 2025-PWYC-06); Interdisciplinary Program of Shanghai Jiao Tong University (YG2023LC04); The Municipal Commission of Health and Family Planning Foundation of Shanghai Pudong New Area (PW2022E-01).
Author information
Authors and Affiliations
Contributions
X.Y.Y. designed the clinical trial, recruited the subjects and wrote the manuscript. Y.T.G. and B.Y.Q. conceptualized the study, provided administrative support and approved the design. H.J.Y. helped prepare hUC-MSCs, quality control and final application, designed and helped carry out the experiments, conducted and interpreted the statistical and bioinformatics analysis, revised the manuscript. X.Y.Y., Z.J.L., and N.Z. were responsible for clinical trial treatment procedure. L.X. provided statistical support. C.R.X. and Y.S.W. carried out patients’ follow-up visit and ensured data quality. X.Z.P. helped collect the clinical data and carried out the in vitro experiments. M.Z.Z., X.Y.W., and Y.C. collected the clinical data. Y.Z.(MD), WB.W., Y.L., and Y.D. helped conduct the treatment and administrative procedure. H.Z., LF.L., H.M.X. were responsible for hUC-MSCs’ quality and decided the dosage. J.D., YF.W., Z.W., YY.S., and K.W. helped recruit the patients and follow-up visit. H.Y.Q., Y.Z.(BS), Y.Y.J., L.P.N., J.L.Y., Q.G., J.H.X., and Y.F.M. provided hospitalization support for subjects. All authors approved the above contribution and the final version of manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yao, XY., Lu, ZJ., Zhao, N. et al. Human umbilical cord mesenchymal stromal cells therapy for neuromyelitis optica spectrum disorder: a phase 1/2a trial. Cell Death Differ (2026). https://doi.org/10.1038/s41418-026-01720-x
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41418-026-01720-x