Abstract
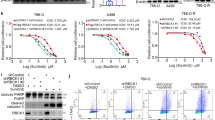
Although the use of tyrosine kinase inhibitors (TKIs), such as sunitinib, has led to impressive advancements in the treatment of clear cell renal cell carcinoma (ccRCC), primary or acquired resistance to sunitinib remains elusive. Here, we report that death receptor 5 (DR5) is upregulated in ccRCC tissues and sunitinib-resistant cells, and is associated with poor outcomes and sunitinib resistance. Gain- and loss-of-function experiments revealed that DR5 promotes sunitinib resistance both in vitro and in vivo. Mechanistically, DR5 enhances the activation of NF-κB signalling by reducing the ubiquitin-mediated proteasomal degradation of p65 via competitive binding to the CUL4B-DDB1 E3 ligase complex linker protein WDR12, leading to the transcriptional upregulation of DR5 and BCL2. The positive feedback loop between DR5 and p65 contributes to the upregulation of BCL2 expression, which in turn modulates sunitinib resistance in ccRCC. Notably, targeting the DR5/NF-κB/BCL2 axis sensitizes ccRCC cells to sunitinib both in vitro and in vivo. Clinically, ccRCC patients with high DR5 expression show decreased responsiveness to TKI-based therapy. Collectively, these results highlight the importance of the positive feedback loop involving the DR5/NF-κB axis in sunitinib resistance and provide an effective therapeutic strategy for overcoming resistance.

This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
Data availability
The data analyzed in this study were obtained from the Gene Expression Omnibus (GEO) databases at GSE54052 and GSE68629. The sequence data generated in this study have been deposited in the NCBI SRA database (SRA PRJNA1236818).
References
Young M, Jackson-Spence F, Beltran L, Day E, Suarez C, Bex A, et al. Renal cell carcinoma. Lancet. 2024;404:476–91.
Cotta BH, Choueiri TK, Cieslik M, Ghatalia P, Mehra R, Morgan TM, et al. Current landscape of genomic biomarkers in clear cell renal cell carcinoma. Eur Urol. 2023;84:166–75.
Ghoreifi A, Vaishampayan U, Yin M, Psutka SP, Djaladat H. Immune checkpoint inhibitor therapy before nephrectomy for locally advanced and metastatic renal cell carcinoma: a review. JAMA Oncol. 2024;10:240–8.
Ryan CW, Tangen CM, Heath EI, Stein MN, Meng MV, Alva AS, et al. Adjuvant everolimus after surgery for renal cell carcinoma (EVEREST): a double-blind, placebo-controlled, randomised, phase 3 trial. Lancet. 2023;402:1043–51.
Motzer RJ, Jonasch E, Agarwal N, Alva A, Bagshaw H, Baine M, et al. NCCN guidelines(R) insights: kidney cancer, version 2. J Natl Compr Canc Netw 2024. 2024;22:4–16.
Jin J, Xie Y, Zhang JS, Wang JQ, Dai SJ, He WF, et al. Sunitinib resistance in renal cell carcinoma: from molecular mechanisms to predictive biomarkers. Drug Resist Updat. 2023;67:100929.
Yu H, Lin L, Zhang Z, Zhang H, Hu H. Targeting NF-kappaB pathway for the therapy of diseases: mechanism and clinical study. Signal Transduct Target Ther. 2020;5:209.
Zhang T, Ma C, Zhang Z, Zhang H, Hu H. NF-KappaB signaling in inflammation and cancer. MedComm. 2021;2:618–53.
Tanaka K, Yu HA, Yang S, Han S, Selcuklu SD, Kim K, et al. Targeting Aurora B kinase prevents and overcomes resistance to EGFR inhibitors in lung cancer by enhancing BIM- and PUMA-mediated apoptosis. Cancer Cell. 2021;39:1245–61.
Li Z, Ning K, Zhao D, Zhou Z, Zhao J, Long X, et al. Targeting the metabolic enzyme PGAM2 overcomes enzalutamide resistance in castration-resistant prostate cancer by inhibiting BCL2 signaling. Cancer Res. 2023;83:3753–66.
Hata AN, Niederst MJ, Archibald HL, Gomez-Caraballo M, Siddiqui FM, Mulvey HE, et al. Tumour cells can follow distinct evolutionary paths to become resistant to epidermal growth factor receptor inhibition. Nat Med. 2016;22:262–9.
Collins PE, Mitxitorena I, Carmody RJ The ubiquitination of NF-kappaB subunits in the control of transcription. Cells. 2016;5:23.
Ren C, Han X, Lu C, Yang T, Qiao P, Sun Y, et al. Ubiquitination of NF-kappaB p65 by FBXW2 suppresses breast cancer stemness, tumourigenesis, and paclitaxel resistance. Cell Death Differ. 2022;29:381–92.
Cong M, Wang Y, Yang Y, Lian C, Zhuang X, Li X, et al. MTSS1 suppresses mammary tumour-initiating cells by enhancing RBCK1-mediated p65 ubiquitination. Nat Cancer. 2020;1:222–34.
Higa LA, Wu M, Ye T, Kobayashi R, Sun H, Zhang H. CUL4-DDB1 ubiquitin ligase interacts with multiple WD40-repeat proteins and regulates histone methylation. Nat Cell Biol. 2006;8:1277–83.
Green DR The death receptor pathway of apoptosis. Cold Spring Harb Perspect Biol. 2022;14:a041053.
Deng Q, Chen L, Zhang G, Liu L, Luo SM, Gao X. TRIAL-based combination therapies in cancers. Int Immunopharmacol. 2024;138:112570.
Oh YT, Sun SY Regulation of cancer metastasis by TRAIL/death receptor signaling. Biomolecules. 2021;11:499.
Chaudhary PM, Eby M, Jasmin A, Bookwalter A, Murray J, Hood L. Death receptor 5, a new member of the TNFR family, and DR4 induce FADD-dependent apoptosis and activate the NF-kappaB pathway. Immunity. 1997;7:821–30.
Hu WH, Johnson H, Shu HB. Tumour necrosis factor-related apoptosis-inducing ligand receptors signal NF-kappaB and JNK activation and apoptosis through distinct pathways. J Biol Chem. 1999;274:30603–10.
von Karstedt S, Conti A, Nobis M, Montinaro A, Hartwig T, Lemke J, et al. Cancer cell-autonomous TRAIL-R signaling promotes KRAS-driven cancer progression, invasion, and metastasis. Cancer Cell. 2015;27:561–73.
Zhang S, Chen Z, Shi P, Fan S, He Y, Wang Q, et al. Downregulation of death receptor 4 is tightly associated with positive response of EGFR mutant lung cancer to EGFR-targeted therapy and improved prognosis. Theranostics. 2021;11:3964–80.
Xiong Z, Yuan C, Shi J, Xiong W, Huang Y, Xiao W, et al. Restoring the epigenetically silenced PCK2 suppresses renal cell carcinoma progression and increases sensitivity to sunitinib by promoting endoplasmic reticulum stress. Theranostics. 2020;10:11444–61.
Pan Y, Lu X, Shu G, Cen J, Lu J, Zhou M, et al. Extracellular vesicle-mediated transfer of LncRNA IGFL2-AS1 confers sunitinib resistance in renal cell carcinoma. Cancer Res. 2023;83:103–16.
Tao W, Wang BY, Luo L, Li Q, Meng ZA, Xia TL, et al. A urine extracellular vesicle lncRNA classifier for high-grade prostate cancer and increased risk of progression: a multi-center study. Cell Rep Med. 2023;4:101240.
Zhang L, Wang X, Bullock AJ, Callea M, Shah H, Song J, et al. Anti-S1P antibody as a novel therapeutic strategy for VEGFR TKI-resistant renal cancer. Clin Cancer Res. 2015;21:1925–34.
Wang H, Xiong A, Chen X, Guo J, Tang Z, Wu C, et al. CXCR1(+) neutrophil infiltration orchestrates response to third-generation EGFR-TKI in EGFR mutant non-small-cell lung cancer. Signal Transduct Target Ther. 2024;9:342.
Zhu Y, Liu H, Xu L, An H, Liu W, Liu Y, et al. p21-activated kinase 1 determines stem-like phenotype and sunitinib resistance via NF-kappaB/IL-6 activation in renal cell carcinoma. Cell Death Dis. 2015;6:e1637.
Liu J, Liu K, Wang Y, Shi Z, Xu R, Zhang Y, et al. Death receptor 5 is required for intestinal stem cell activity during intestinal epithelial renewal at homoeostasis. Cell Death Dis. 2024;15:27.
Li S, Lv J, Li Z, Zhang Q, Lu J, Huo X, et al. Overcoming multi-drug resistance in SCLC: a synergistic approach with venetoclax and hydroxychloroquine targeting the lncRNA LYPLAL1-DT/BCL2/BECN1 pathway. Mol Cancer. 2024;23:243.
Wang L, Jin H, Jochems F, Wang S, Lieftink C, Martinez IM, et al. cFLIP suppression and DR5 activation sensitize senescent cancer cells to senolysis. Nat Cancer. 2022;3:1284–99.
Zhang J, Wu T, Simon J, Takada M, Saito R, Fan C, et al. VHL substrate transcription factor ZHX2 as an oncogenic driver in clear cell renal cell carcinoma. Science. 2018;361:290–5.
Sanchez A, Tripathy D, Yin X, Luo J, Martinez JM, Grammas P. Sunitinib enhances neuronal survival in vitro via NF-kappaB-mediated signaling and expression of cyclooxygenase-2 and inducible nitric oxide synthase. J Neuroinflamm. 2013;10:93.
Giuliano S, Dufies M, Ndiaye PD, Viotti J, Borchiellini D, Parola J, et al. Resistance to lysosomotropic drugs used to treat kidney and breast cancers involves autophagy and inflammation and converges in inducing CXCL5. Theranostics. 2019;9:1181–99.
Zeng X, Chen Z, Zhu Y, Liu L, Zhang Z, Xiao Y, et al. O-GlcNAcylation regulation of RIPK1-dependent apoptosis dictates sensitivity to sunitinib in renal cell carcinoma. Drug Resist Updat. 2024;77:101150.
Wen S, Huang X, Xiong L, Zeng H, Wu S, An K, et al. WDR12/RAC1 axis promoted proliferation and anti-apoptosis in colorectal cancer cells. Mol Cell Biochem. 2024;479:3341–54.
Li JL, Chen C, Chen W, Zhao LF, Xu XK, Li Y, et al. Integrative genomic analyses identify WDR12 as a novel oncogene involved in glioblastoma. J Cell Physiol. 2020;235:7344–55.
Powles T, Plimack ER, Soulieres D, Waddell T, Stus V, Gafanov R, et al. Pembrolizumab plus axitinib versus sunitinib monotherapy as first-line treatment of advanced renal cell carcinoma (KEYNOTE-426): extended follow-up from a randomised, open-label, phase 3 trial. Lancet Oncol. 2020;21:1563–73.
Yang Y, Li S, Wang Y, Zhao Y, Li Q. Protein tyrosine kinase inhibitor resistance in malignant tumours: molecular mechanisms and future perspective. Signal Transduct Target Ther. 2022;7:329.
Cruz-Gordillo P, Honeywell ME, Harper NW, Leete T, Lee MJ ELP-dependent expression of MCL1 promotes resistance to EGFR inhibition in triple-negative breast cancer cells. Sci Signal. 2020;13:9820.
Apicella M, Giannoni E, Fiore S, Ferrari KJ, Fernandez-Perez D, Isella C, et al. Increased lactate secretion by cancer cells sustains non-cell-autonomous adaptive resistance to MET and EGFR targeted therapies. Cell Metab. 2018;28:848–65.
Jeong WJ, Park JC, Kim WS, Ro EJ, Jeon SH, Lee SK, et al. WDR76 is a RAS binding protein that functions as a tumour suppressor via RAS degradation. Nat Commun. 2019;10:295.
Zhang H, Chen G, Feng X, Song H, Meng L, Fu Y, et al. Targeting WDxR motif reprograms immune microenvironment and inhibits hepatocellular carcinoma progression. EMBO Mol Med. 2023;15:e15924.
Carneiro BA, El-Deiry WS. Targeting apoptosis in cancer therapy. Nat Rev Clin Oncol. 2020;17:395–417.
Deng D, Shah K. TRAIL of hope meeting resistance in cancer. Trends Cancer. 2020;6:989–1001.
Shrader M, Pino MS, Lashinger L, Bar-Eli M, Adam L, Dinney CP, et al. Gefitinib reverses TRAIL resistance in human bladder cancer cell lines via inhibition of AKT-mediated X-linked inhibitor of apoptosis protein expression. Cancer Res. 2007;67:1430–5.
Correction: The Multikinase Inhibitor Sorafenib Potentiates TRAIL lethality in human leukemia cells in association with Mcl-1 and cFLIPL down-regulation. Cancer Res. 2018;78:1575.
Ding W, Cai T, Zhu H, Wu R, Tu C, Yang L, et al. Synergistic antitumour effect of TRAIL in combination with sunitinib in vitro and in vivo. Cancer Lett. 2010;293:158–66.
Mahalingam D, Carew JS, Espitia CM, Cool RH, Giles FJ, de Jong S, et al. Heightened JNK activation and reduced XIAP levels promote TRAIL and sunitinib-mediated apoptosis in colon cancer models. Cancers. 2019;11:895.
Shadman M. Diagnosis and treatment of chronic lymphocytic leukemia: a review. JAMA. 2023;329:918–32.
DiNardo CD, Jonas BA, Pullarkat V, Thirman MJ, Garcia JS, Wei AH, et al. Azacitidine and venetoclax in previously untreated acute myeloid leukemia. N Engl J Med. 2020;383:617–29.
Oppermann S, Ylanko J, Shi Y, Hariharan S, Oakes CC, Brauer PM, et al. High-content screening identifies kinase inhibitors that overcome venetoclax resistance in activated CLL cells. Blood. 2016;128:934–47.
Tang Y, Song T, Gao L, Mao F. Venetoclax synergizes sunitinib in renal cell carcincoma through inhibition of BCL-2. Anticancer Agents Med Chem. 2023;23:2027–34.
Acknowledgements
We sincerely thank Prof. Xiaoping Zhang for providing the sunitinib-resistant RCC cells. We thank all the patients involved in this study.
Funding
This work was supported by grants from the Minimally Invasive Innovation Team of the Urology Department of the Chinese PLA General Hospital (X. Zhang), the Priority Specialty in Medicine (X. Zhang), Development of Key Clinical Specialties (X. Zhang), the Pioneer Program for Discipline Innovation and Development of the Third Medical Center of PLA General Hospital (2024BJ-11 to Y. Huang), the Youth Fund of Chinese PLA General Hospital (22QNCZ022 to Y. Huang), National Natural Science Foundation of China (82373233 to T. Shi) and Key Research and Development Project of Hainan Provincial Science and Technology Department (ZDYF2021SHFZ056 to T. Shi).
Author information
Authors and Affiliations
Contributions
W. Tao: Conceptualization, data curation, formal analysis, visualization, investigation, methodology, writing-original draft, writing-review and editing. Y. Dong: Data curation, investigation, and methodology. S. Zuo: Formal analysis, investigation, methodology. H. Wang: Investigation and methodology. Q. Liang: Formal analysis and investigation. T. Cai: Formal analysis and investigation. X. Chen: Investigation. W. Wei: Formal analysis. C. Zhang: Investigation. S. Tian: Investigation. C. Wang: Formal analysis. H. Li: Data curation and resources. B. Wang: Data curation and resources. X. Ma: Conceptualization, data curation, resources. Q. Huang: Conceptualization, data curation, resources. T. Shi: Conceptualization, data curation, funding acquisition. Y. Huang: Conceptualization, data curation, formal analysis, investigation, funding acquisition, resources, supervision, project administration, writing-review and editing. X. Zhang: Conceptualization, data curation, formal analysis, investigation, funding acquisition, resources, supervision, project administration and writing-review.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval
Ethical approval for the animal experiments was obtained from the Ethics Committee of the Chinese People’s Liberation Army (PLA) General Hospital. The collection of human tissues was approved by the Ethics and Research Committee of the Chinese PLA General Hospital and written informed consent was obtained from the patients before surgery.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Tao, W., Dong, Y., Zuo, S. et al. DR5/WDR12 balances p65 stability promoting sunitinib resistance in renal cell carcinoma. Cell Death Differ (2026). https://doi.org/10.1038/s41418-026-01723-8
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41418-026-01723-8