Abstract
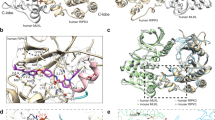
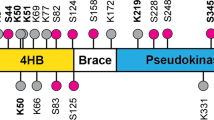
Receptor-interacting protein kinase 3 (RIPK3) has emerged as a central player in necroptosis and apoptosis activation in specific scenarios, concurrently modulating inflammatory responses. Here, we reveal that direct activation of RIPK3 concomitantly triggers mixed lineage kinase domain-like (MLKL) phosphorylation, caspase activation, and gasdermin cleavage within individual cells, inducing PANoptotic cell death. This process is orchestrated by the formation of RIPK3-MLKL-RIPK1-FADD-Caspase-8 complexes on progressively polymerized RIPK3 homo-aggregates, achieved through sequential recruitment dictated by the differential affinities of MLKL and Receptor-interacting protein kinase 1 (RIPK1) for distinct oligomeric states of RIPK3. In this process, MLKL- and GSDMD-mediated membrane rupture is respectively inhibited by Caspase-3-dependent cleavage of RIPK3 and GSDMD cleavage, while the pro-necrotic kinase activity of RIPK3 impedes RIPK1 recruitment and attenuates caspase activation. Cross-regulation between pathways results in unique cellular morphology, altered damage-associated molecular patterns (DAMPs) release profiles and distinct chemokine secretion paradigms that differ fundamentally from classical necroptosis, apoptosis and pyroptosis. This work highlights a common mechanism unveiling RIPK3 as a multimolecular platform to modulate and integrate different programmed cell death (PCD) pathways, thus providing a framework for targeting inflammatory cell death in disease.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
Data availability
All data and materials supporting the conclusions of this paper are presented in the main text, figures, supplementary data figures, and attachment files. The uncropped Western blots are included in the Supplementary Material. Further data can be received from the corresponding author on reasonable request. RNA-sequencing data were deposited into the Gene Expression Omnibus (GEO) database under accession code GSE320510. Source data are provided with this paper.
References
Ai Y, Meng Y, Yan B, Zhou Q, Wang X. The biochemical pathways of apoptotic, necroptotic, pyroptotic, and ferroptotic cell death. Mol Cell. 2024;84:170–9. https://doi.org/10.1016/j.molcel.2023.11.040.
Newton K, Strasser A, Kayagaki N, Dixit VM. Cell death. Cell. 2024;187:235–56. https://doi.org/10.1016/j.cell.2023.11.044.
Yuan J, Ofengeim D. A guide to cell death pathways. Nat Rev Mol Cell Biol. 2024;25:379–395. https://doi.org/10.1038/s41580-023-00689-6.
He S, Wang L, Miao L, Wang T, Du F, Zhao L, et al. Receptor-interacting protein kinase-3 determines cellular necrotic response to TNF-alpha. Cell. 2009;137:1100–11. https://doi.org/10.1016/j.cell.2009.05.021.
Zhang DW, Shao J, Lin J, Zhang N, Lu BJ, Lin SC, et al. RIP3, an energy metabolism regulator that switches TNF-induced cell death from apoptosis to necrosis. Science. 2009;325:332–6. https://doi.org/10.1126/science.1172308.
Cho YS, Challa S, Moquin D, Genga R, Ray TD, Guildford M, et al. Phosphorylation-driven assembly of the RIP1-RIP3 complex regulates programmed necrosis and virus-induced inflammation. Cell. 2009;137:1112–23. https://doi.org/10.1016/j.cell.2009.05.037.
Jouan-Lanhouet S, Arshad MI, Piquet-Pellorce C, Martin-Chouly C, Le Moigne-Muller G, Van Herreweghe F, et al. TRAIL induces necroptosis involving RIPK1/RIPK3-dependent PARP-1 activation. Cell Death Differ. 2012;19:2003–14. https://doi.org/10.1038/cdd.2012.90.
Strilic B, Yang L, Albarran-Juarez J, Wachsmuth L, Han K, Muller UC, et al. Tumour-cell-induced endothelial cell necroptosis via death receptor 6 promotes metastasis. Nature. 2016;536:215–8. https://doi.org/10.1038/nature19076.
He S, Liang Y, Shao F, Wang X. Toll-like receptors activate programmed necrosis in macrophages through a receptor-interacting kinase-3-mediated pathway. Proc Natl Acad Sci USA. 2011;108:20054–59. https://doi.org/10.1073/pnas.1116302108.
Kaiser WJ, Sridharan H, Huang C, Mandal P, Upton JW, Gough PJ, et al. Toll-like receptor 3-mediated necrosis via TRIF, RIP3, and MLKL. J Biol Chem. 2013;288:31268–79. https://doi.org/10.1074/jbc.M113.462341.
Rebsamen M, Heinz LX, Meylan E, Michallet MC, Schroder K, Hofmann K, et al. DAI/ZBP1 recruits RIP1 and RIP3 through RIP homotypic interaction motifs to activate NF-kappaB. EMBO Rep. 2009;10:916–22. https://doi.org/10.1038/embor.2009.109.
Upton JW, Kaiser WJ, Mocarski ES. DAI/ZBP1/DLM-1 complexes with RIP3 to mediate virus-induced programmed necrosis that is targeted by murine cytomegalovirus vIRA. Cell Host Microbe. 2012;11:290–7. https://doi.org/10.1016/j.chom.2012.01.016.
Thapa RJ, Ingram JP, Ragan KB, Nogusa S, Boyd DF, Benitez AA, et al. DAI senses influenza A virus genomic RNA and activates RIPK3-dependent cell death. Cell Host Microbe. 2016;20:674–81. https://doi.org/10.1016/j.chom.2016.09.014.
Newton K, Wickliffe KE, Maltzman A, Dugger DL, Strasser A, Pham VC, et al. RIPK1 inhibits ZBP1-driven necroptosis during development. Nature. 2016;540:129–33. https://doi.org/10.1038/nature20559.
Lin J, Kumari S, Kim C, Van TM, Wachsmuth L, Polykratis A, et al. RIPK1 counteracts ZBP1-mediated necroptosis to inhibit inflammation. Nature. 2016;540:124–8. https://doi.org/10.1038/nature20558.
Zhang T, Yin C, Boyd DF, Quarato G, Ingram JP, Shubina M, et al. Influenza virus Z-RNAs induce ZBP1-mediated necroptosis. Cell. 2020;180:1115–29.e1113. https://doi.org/10.1016/j.cell.2020.02.050.
Jiao H, Wachsmuth L, Kumari S, Schwarzer R, Lin J, Eren RO, et al. Z-nucleic-acid sensing triggers ZBP1-dependent necroptosis and inflammation. Nature. 2020;580:391–5. https://doi.org/10.1038/s41586-020-2129-8.
Huang Z, Wu SQ, Liang Y, Zhou X, Chen W, Li L, et al. RIP1/RIP3 binding to HSV-1 ICP6 initiates necroptosis to restrict virus propagation in mice. Cell Host Microbe. 2015;17:229–42. https://doi.org/10.1016/j.chom.2015.01.002.
Sun X, Yin J, Starovasnik MA, Fairbrother WJ, Dixit VM. Identification of a novel homotypic interaction motif required for the phosphorylation of receptor-interacting protein (RIP) by RIP3. J Biol Chem. 2002;277:9505–11. https://doi.org/10.1074/jbc.M109488200.
Li J, McQuade T, Siemer AB, Napetschnig J, Moriwaki K, Hsiao YS, et al. The RIP1/RIP3 necrosome forms a functional amyloid signaling complex required for programmed necrosis. Cell. 2012;150:339–50. https://doi.org/10.1016/j.cell.2012.06.019.
Sun L, Wang H, Wang Z, He S, Chen S, Liao D, et al. Mixed lineage kinase domain-like protein mediates necrosis signaling downstream of RIP3 kinase. Cell. 2012;148:213–27. https://doi.org/10.1016/j.cell.2011.11.031.
Murphy JM, Czabotar PE, Hildebrand JM, Lucet IS, Zhang JG, Alvarez-Diaz S, et al. The pseudokinase MLKL mediates necroptosis via a molecular switch mechanism. Immunity. 2013;39:443–53. https://doi.org/10.1016/j.immuni.2013.06.018.
Wang H, Sun L, Su L, Rizo J, Liu L, Wang LF, et al. Mixed lineage kinase domain-like protein MLKL causes necrotic membrane disruption upon phosphorylation by RIP3. Mol Cell. 2014;54:133–46. https://doi.org/10.1016/j.molcel.2014.03.003.
Dondelinger Y, Declercq W, Montessuit S, Roelandt R, Goncalves A, Bruggeman I, et al. MLKL compromises plasma membrane integrity by binding to phosphatidylinositol phosphates. Cell Rep. 2014;7:971–81. https://doi.org/10.1016/j.celrep.2014.04.026.
Cai Z, Jitkaew S, Zhao J, Chiang HC, Choksi S, Liu J, et al. Plasma membrane translocation of trimerized MLKL protein is required for TNF-induced necroptosis. Nat Cell Biol. 2014;16:55–65. https://doi.org/10.1038/ncb2883.
Mandal P, Berger SB, Pillay S, Moriwaki K, Huang C, Guo H, et al. RIP3 induces apoptosis independent of pronecrotic kinase activity. Mol Cell. 2014;56:481–95. https://doi.org/10.1016/j.molcel.2014.10.021.
Newton K, Dugger DL, Wickliffe KE, Kapoor N, de Almagro MC, Vucic D, et al. Activity of protein kinase RIPK3 determines whether cells die by necroptosis or apoptosis. Science. 2014;343:1357–60. https://doi.org/10.1126/science.1249361.
Nogusa S, Thapa RJ, Dillon CP, Liedmann S, Oguin TH 3rd, et al. RIPK3 activates parallel pathways of MLKL-driven necroptosis and FADD-mediated apoptosis to protect against influenza A virus. Cell Host Microbe. 2016;20:13–24. https://doi.org/10.1016/j.chom.2016.05.011.
Shubina M, Tummers B, Boyd DF, Zhang T, Yin C, Gautam A, et al. Necroptosis restricts influenza A virus as a stand-alone cell death mechanism. J Exp Med. 2020;217. https://doi.org/10.1084/jem.20191259
Li D, Chen J, Guo J, Li L, Cai G, Chen S, et al. A phosphorylation of RIPK3 kinase initiates an intracellular apoptotic pathway that promotes prostaglandin(2alpha)-induced corpus luteum regression. Elife. 2021 https://doi.org/10.7554/eLife.67409
Lawlor KE, Khan N, Mildenhall A, Gerlic M, Croker BA, D’Cruz AA, et al. RIPK3 promotes cell death and NLRP3 inflammasome activation in the absence of MLKL. Nat Commun. 2015;6:6282. https://doi.org/10.1038/ncomms7282.
Dondelinger Y, Aguileta MA, Goossens V, Dubuisson C, Grootjans S, Dejardin E, et al. RIPK3 contributes to TNFR1-mediated RIPK1 kinase-dependent apoptosis in conditions of cIAP1/2 depletion or TAK1 kinase inhibition. Cell Death Differ. 2013;20:1381–92. https://doi.org/10.1038/cdd.2013.94.
Zheng M, Karki R, Vogel P, Kanneganti TD. Caspase-6 is a key regulator of innate immunity, inflammasome activation, and host defense. Cell. 2020;181:674–87 e613. https://doi.org/10.1016/j.cell.2020.03.040.
Banoth B, Tuladhar S, Karki R, Sharma BR, Briard B, Kesavardhana S, et al. ZBP1 promotes fungi-induced inflammasome activation and pyroptosis, apoptosis, and necroptosis (PANoptosis). J Biol Chem. 2020;295:18276–83. https://doi.org/10.1074/jbc.RA120.015924.
Lee S, Karki R, Wang Y, Nguyen LN, Kalathur RC, Kanneganti TD. AIM2 forms a complex with pyrin and ZBP1 to drive PANoptosis and host defence. Nature. 2021;597:415–19. https://doi.org/10.1038/s41586-021-03875-8.
Malireddi RKS, Gurung P, Kesavardhana S, Samir P, Burton A, Mummareddy H et al. Innate immune priming in the absence of TAK1 drives RIPK1 kinase activity-independent pyroptosis, apoptosis, necroptosis, and inflammatory disease. J Exp Med. 2020. https://doi.org/10.1084/jem.20191644
Sundaram B, Pandian N, Kim HJ, Abdelaal HM, Mall R, Indari O, et al. NLRC5 senses NAD(+) depletion, forming a PANoptosome and driving PANoptosis and inflammation. Cell. 2024;187:4061–77.e4017. https://doi.org/10.1016/j.cell.2024.05.034.
Sundaram B, Pandian N, Mall R, Wang Y, Sarkar R, Kim HJ, et al. NLRP12-PANoptosome activates PANoptosis and pathology in response to heme and PAMPs. Cell. 2023;186:2783–801.e2720. https://doi.org/10.1016/j.cell.2023.05.005.
Pandeya A, Kanneganti TD. Therapeutic potential of PANoptosis: innate sensors, inflammasomes, and RIPKs in PANoptosomes. Trends Mol Med. 2024;30:74–88. https://doi.org/10.1016/j.molmed.2023.10.001.
Han X, Li F, Fang Z, Gao Y, Li F, Fang R, et al. Transdifferentiation of lung adenocarcinoma in mice with Lkb1 deficiency to squamous cell carcinoma. Nat Commun. 2014;5:3261. https://doi.org/10.1038/ncomms4261.
Zhang W, Fan W, Guo J, Wang X. Osmotic stress activates RIPK3/MLKL-mediated necroptosis by increasing cytosolic pH through a plasma membrane Na(+)/H(+) exchanger. Sci Signal. 2022;15:eabn5881. https://doi.org/10.1126/scisignal.abn5881.
Burg MB, Ferraris JD, Dmitrieva NI. Cellular response to hyperosmotic stresses. Physiol Rev. 2007;87:1441–74. https://doi.org/10.1152/physrev.00056.2006.
Ofengeim D, Yuan J. Regulation of RIP1 kinase signalling at the crossroads of inflammation and cell death. Nat Rev Mol Cell Biol. 2013;14:727–36. https://doi.org/10.1038/nrm3683.
Holler N, Zaru R, Micheau O, Thome M, Attinger A, Valitutti S, et al. Fas triggers an alternative, caspase-8-independent cell death pathway using the kinase RIP as effector molecule. Nat Immunol. 2000;1:489–95. https://doi.org/10.1038/82732.
Degterev A, Hitomi J, Germscheid M, Ch’en IL, Korkina O, Teng X, et al. Identification of RIP1 kinase as a specific cellular target of necrostatins. Nat Chem Biol. 2008;4:313–21. https://doi.org/10.1038/nchembio.83.
Nadella V, Kanneganti TD. Inflammasomes and their role in PANoptosomes. Curr Opin Immunol. 2024;91:102489. https://doi.org/10.1016/j.coi.2024.102489.
Mompean M, Li W, Li J, Laage S, Siemer AB, Bozkurt G, et al. The structure of the necrosome RIPK1-RIPK3 core, a human hetero-amyloid signaling complex. Cell. 2018;173:1244–53.e1210. https://doi.org/10.1016/j.cell.2018.03.032.
Chen X, Zhu R, Zhong J, Ying Y, Wang W, Cao Y, et al. Mosaic composition of RIP1-RIP3 signalling hub and its role in regulating cell death. Nat Cell Biol. 2022;24:471–82. https://doi.org/10.1038/s41556-022-00854-7.
Tran HT, Kratina T, Coutansais A, Michalek D, Hogan BM, Lawlor KE, et al. RIPK3 cleavage is dispensable for necroptosis inhibition but restricts NLRP3 inflammasome activation. Cell Death Differ. 2024;31:662–71. https://doi.org/10.1038/s41418-024-01281-x.
Newton K, Wickliffe KE, Maltzman A, Dugger DL, Webster JD, Guo H, et al. Caspase cleavage of RIPK3 after Asp(333) is dispensable for mouse embryogenesis. Cell Death Differ. 2024;31:254–62. https://doi.org/10.1038/s41418-023-01255-5.
He K, Wan T, Wang D, Hu J, Zhou T, Tao W, et al. Gasdermin D licenses MHCII induction to maintain food tolerance in small intestine. Cell. 2023;186:3033–48 e3020. https://doi.org/10.1016/j.cell.2023.05.027.
Li M, Yang D, Yan H, Tang Z, Jiang D, Zhang J, et al. Gasdermin D maintains bone mass by rewiring the endo-lysosomal pathway of osteoclastic bone resorption. Dev Cell. 2022;57:2365–80.e2368. https://doi.org/10.1016/j.devcel.2022.09.013.
Orning P, Weng D, Starheim K, Ratner D, Best Z, Lee B, et al. Pathogen blockade of TAK1 triggers caspase-8-dependent cleavage of gasdermin D and cell death. Science. 2018;362:1064–69. https://doi.org/10.1126/science.aau2818.
Sarhan J, Liu BC, Muendlein HI, Li P, Nilson R, Tang AY, et al. Caspase-8 induces cleavage of gasdermin D to elicit pyroptosis during Yersinia infection. Proc Natl Acad Sci USA. 2018;115:E10888–E10897. https://doi.org/10.1073/pnas.1809548115.
DeMarco B, Grayczyk JP, Bjanes E, Le Roy D, Tonnus W, Assenmacher CA, et al. Caspase-8-dependent gasdermin D cleavage promotes antimicrobial defense but confers susceptibility to TNF-induced lethality. Sci Adv. 2020. https://doi.org/10.1126/sciadv.abc3465
Schwarzer R, Jiao H, Wachsmuth L, Tresch A, Pasparakis M. FADD and caspase-8 regulate Gut homeostasis and inflammation by controlling MLKL- and GSDMD-mediated death of intestinal epithelial cells. Immunity. 2020;52:978–93.e976. https://doi.org/10.1016/j.immuni.2020.04.002.
Zheng Z, Deng W, Bai Y, Miao R, Mei S, Zhang Z, et al. The lysosomal rag-ragulator complex licenses RIPK1 and caspase-8-mediated pyroptosis by Yersinia. Science. 2021. https://doi.org/10.1126/science.abg0269
Orozco S, Yatim N, Werner MR, Tran H, Gunja SY, Tait SW, et al. RIPK1 both positively and negatively regulates RIPK3 oligomerization and necroptosis. Cell Death Differ. 2014;21:1511–21. https://doi.org/10.1038/cdd.2014.76.
Vringer E, Tait SWG. Mitochondria and cell death-associated inflammation. Cell Death Differ. 2023;30:304–12. https://doi.org/10.1038/s41418-022-01094-w.
Zhu K, Liang W, Ma Z, Xu D, Cao S, Lu X, et al. Necroptosis promotes cell-autonomous activation of proinflammatory cytokine gene expression. Cell Death Dis. 2018;9:500. https://doi.org/10.1038/s41419-018-0524-y.
Zu R, Yu Z, Zhao J, Lu X, Liang W, Sun L, et al. Quantitative analysis of phosphoproteome in necroptosis reveals a role of TRIM28 phosphorylation in promoting necroptosis-induced cytokine production. Cell Death Dis. 2021;12:994. https://doi.org/10.1038/s41419-021-04290-7.
Lin CC, Mabe NW, Lin YT, Yang WH, Tang X, Hong L, et al. RIPK3 upregulation confers robust proliferation and collateral cystine-dependence on breast cancer recurrence. Cell Death Differ. 2020;27:2234–47. https://doi.org/10.1038/s41418-020-0499-y.
Wu E, He W, Wu C, Chen Z, Zhou S, Wu X, et al. HSPA8 acts as an amyloidase to suppress necroptosis by inhibiting and reversing functional amyloid formation. Cell Res. 2023;33:851–66. https://doi.org/10.1038/s41422-023-00859-3.
Wang W, Prokopec JS, Zhang Y, Sukhoplyasova M, Shinglot H, Wang MT, et al. Sensing plasma membrane pore formation induces chemokine production in survivors of regulated necrosis. Dev Cell. 2022;57:228–45.e226. https://doi.org/10.1016/j.devcel.2021.12.015.
Yu X, Ma H, Li B, Ji Y, Du Y, Liu S, et al. A novel RIPK1 inhibitor reduces GVHD in mice via a nonimmunosuppressive mechanism that restores intestinal homeostasis. Blood. 2023;141:1070–86. https://doi.org/10.1182/blood.2022017262.
Li X, Zhong CQ, Wu R, Xu X, Yang ZH, Cai S, et al. RIP1-dependent linear and nonlinear recruitments of caspase-8 and RIP3 respectively to necrosome specify distinct cell death outcomes. Protein Cell. 2021;12:858–76. https://doi.org/10.1007/s13238-020-00810-x.
He W, Xu J, Mu R, Li Q, Lv DL, Huang Z, et al. High-salt diet inhibits tumour growth in mice via regulating myeloid-derived suppressor cell differentiation. Nat Commun. 2020;11:1732. https://doi.org/10.1038/s41467-020-15524-1.
Acknowledgements
The authors thank D Xiao, Y Lei, H Pan, S Qiu, and L Chen for reagents. We thank Drs. S He, M Zheng, X Yang, B Han, and H Wang for helpful discussion. We thank X Dong for the identification of GSDMD-deficient clones. The authors thank the Chemical Biology Core Facility at SIBCB for technical assistance in time-lapse imaging.
Funding
This study is supported by the grants from the National Natural Science Foundation of China (82072573, 82273335, 8240113215 and 82403114), Shanghai Municipal Health Commission (2022LJ015, 2022YQ039 and 20224Y0186).
Author information
Authors and Affiliations
Contributions
YY, RZ, and HZ conceptualized the study. YY, YuW, and EW designed the methodology. YY and YuW performed the majority of experiments and data analysis. YaW performed the confocal microscopy and image analysis. LS did the mass spectrometry analysis. HC performed the bioinformatics analysis. CZ, SC, and JL housed the mice and conducted the experiments in the animal facility. LZ and HQ generated all the plasmids. YL, RQ, and SZ discussed the results and provided valuable expertise. YY wrote the manuscript with input from all authors. YZ and HZ acquired the funding.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics statement
All animal experimental procedures were approved by the Institutional Animal Care and Use Committee (IACUC) of Shanghai Jiao Tong University (Ethical Approval No. KS(Y)24008), and tumor dimensions complied with IACUC-specified limits. No human material was used.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yang, Y., Wang, Y., Wang, Y. et al. RIPK3 sequentially recruits MLKL and RIPK1 to induce PANoptosis and chemokine production. Cell Death Differ (2026). https://doi.org/10.1038/s41418-026-01737-2
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41418-026-01737-2