Abstract
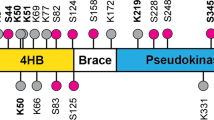
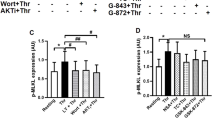
Necroptosis has been definitively confirmed as a caspase-deficient, non-apoptotic cellular mechanism that exhibits a profound connection to inflammatory disorders. The receptor-interacting protein kinase 1 (RIPK1), RIPK3, and mixed-lineage kinase domain-like protein (MLKL Cys86) have been recognized as three main targets for necroptosis for many years. Here, we report HSPBP1 Cys201 and MLKL Cys184 as new cellular targets for necroptosis in human cells. Parthenolide, a natural sesquiterpene lactone, was first confirmed to have anti-necroptotic activity and effectively alleviated the necroptosis-induced systemic inflammatory response syndrome and abdominal aortic aneurysm (AAA) in mice. In the elastase-induced mouse AAA model, MLKL deficiency is highlighted as attenuating AAA formation. HSPBP1 Cys201 was identified to be an upstream target contributing to the anti-necroptotic activity. Co-incubated with purified HSPBP1, followed by mass spectrometry analysis, confirmed that PTL binds to HSPBP1 at Cys201, while HSPBP1 knockdown conferred a certain degree of resilience to necroptosis. Human MLKL Cys184 was discovered as another novel anti-necroptotic target in human HT-29 cells. The human MTRP and molecular dynamics results suggested that Cys184 is the potential binding site between PTL and MLKL. Our co-incubation experiments of PTL with MLKL further demonstrated that PTL can interact with the sulfhydryl group of MLKL Cys184 via covalent modification. These findings yield important insights into the complex regulatory mechanisms of necroptosis and, concurrently, underscore the therapeutic potential of PTL and its derivatives for treating AAA.
Similar content being viewed by others
Data availability
The data underlying this article are available in its online Supplementary Materials. The mRNA sequencing data of mouse abdominal aortic aneurysm were deposited in the Figshare database with the https://doi.org/10.6084/m9.figshare.32052066.
References
Pasparakis M, Vandenabeele P. Necroptosis and its role in inflammation. Nature. 2015;517:311–20.
Sun L, Wang X. A new kind of cell suicide: mechanisms and functions of programmed necrosis. Trends Biochem Sci. 2014;39:587–93.
Degterev A, Huang Z, Boyce M, Li Y, Jagtap P, Mizushima N, et al. Chemical inhibitor of nonapoptotic cell death with therapeutic potential for ischemic brain injury. Nat Chem Biol. 2005;1:112–9.
Degterev A, Hitomi J, Germscheid M, Ch’en IL, Korkina O, Teng X, et al. Identification of RIP1 kinase as a specific cellular target of necrostatins. Nat Chem Biol. 2008;4:313–21.
He S, Wang L, Miao L, Wang T, Du F, Zhao L, et al. Receptor interacting protein kinase-3 determines cellular necrotic response to TNF-alpha. Cell. 2009;137:1100–11.
Zhang DW, Shao J, Lin J, Zhang N, Lu BJ, Lin SC, et al. RIP3, an energy metabolism regulator that switches TNF-induced cell death from apoptosis to necrosis. Science. 2009;325:332–6.
Cho YS, Challa S, Moquin D, Genga R, Ray TD, Guildford M, et al. Phosphorylation-driven assembly of the RIP1-RIP3 complex regulates programmed necrosis and virus-induced inflammation. Cell. 2009;137:1112–23.
Liao DH, Sun LM, Liu WL, He SD, Wang XD, Lei XG. Necrosulfonamide inhibits necroptosis by selectively targeting the mixed lineage kinase domain-like protein. MedChemComm. 2014;5:333–7.
Sun L, Wang H, Wang Z, He S, Chen S, Liao D, et al. Mixed lineage kinase domain-like protein mediates necrosis signaling downstream of RIP3 kinase. Cell. 2012;148:213–27.
Zhuang C, Chen F. Small-molecule inhibitors of necroptosis: current status and perspectives. J Med Chem. 2020;63:1490–510.
Bai Y, Lam HC, Lei X. Dissecting programmed cell death with small molecules. Acc Chem Res. 2020;53:1034–45.
Gardner CR, Davies KA, Zhang Y, Brzozowski M, Czabotar PE, Murphy JM, et al. From (Tool)Bench to Bedside: the potential of necroptosis inhibitors. J Med Chem. 2023;66:2361–85.
Samson AL, Zhang Y, Geoghegan ND, Gavin XJ, Davies KA, Mlodzianoski MJ, et al. MLKL trafficking and accumulation at the plasma membrane control the kinetics and threshold for necroptosis. Nat Commun. 2020;11:3151.
Atanasov AG, Zotchev SB, Dirsch VM, Supuran CTThe International Natural Product Sciences Taskforce Natural products in drug discovery: advances and opportunities. Nat Rev Drug Discov. 2021;20:200–16.
Zhu Y, Ouyang Z, Du H, Wang M, Wang J, Sun H, et al. New opportunities and challenges of natural products research: When target identification meets single-cell multiomics. Acta Pharm Sin B. 2022;12:4011–39.
Li D, Li C, Li L, Chen S, Wang L, Li Q, et al. Natural product Kongensin A is a non-canonical HSP90 inhibitor that blocks RIP3-dependent necroptosis. Cell Chem Biol. 2016;23:257–66.
Jacobsen AV, Lowes KN, Tanzer MC, Lucet IS, Hildebrand JM, Petrie EJ, et al. HSP90 activity is required for MLKL oligomerisation and membrane translocation and the induction of necroptotic cell death. Cell Death Dis. 2016;7:e2051.
Chen L, Fan H, Chu H, Du F, Chen Y, Hu L, et al. The HSP90 inhibitor 17-DMAG alleviates primary biliary cholangitis via cholangiocyte necroptosis prevention. J Cell Biochem. 2022;123:1857–72.
Wu Z, Geng Y, Lu X, Shi Y, Wu G, Zhang M, et al. Chaperone-mediated autophagy is involved in the execution of ferroptosis. Proc Natl Acad Sci USA. 2019;116:2996–3005.
Jacobsen AV, Silke J. The importance of being chaperoned: HSP90 and necroptosis. Cell Chem Biol. 2016;23:205–7.
Pareek A, Suthar M, Rathore GS, Bansal V. Feverfew (Tanacetum parthenium L.): a systematic review. Pharmacogn Rev. 2011;5:103–10.
Xu L, Zhuang C. Profiling of small-molecule necroptosis inhibitors based on the subpockets of kinase-ligand interactions. Med Res Rev. 2023;43:1974–2024.
Duprez L, Takahashi N, Van Hauwermeiren F, Vandendriessche B, Goossens V, Vanden Berghe T, et al. RIP kinase-dependent necrosis drives lethal systemic inflammatory response syndrome. Immunity. 2011;35:908–18.
Quan D, Hou R, Shao H, Zhang X, Yu J, Zhang W, et al. Structure-based design of novel alkynyl thio-benzoxazepinone receptor-interacting protein kinase-1 inhibitors: extending the chemical space from the allosteric to ATP binding pockets. J Med Chem. 2023;66:3073–87.
Wang Q, Liu Z, Ren J, Morgan S, Assa C, Liu B. Receptor-interacting protein kinase 3 contributes to abdominal aortic aneurysms via smooth muscle cell necrosis and inflammation. Circ Res. 2015;116:600–11.
Meng Y, Garnish SE, Davies KA, Black KA, Leis AP, Horne CR, et al. Phosphorylation-dependent pseudokinase domain dimerization drives full-length MLKL oligomerization. Nat Commun. 2023;14:6804.
Maurais AJ, Weerapana E. Reactive-cysteine profiling for drug discovery. Curr Opin Chem Biol. 2019;50:29–36.
Tian C, Sun R, Liu K, Fu L, Liu X, Zhou W, et al. Multiplexed thiol reactivity profiling for target discovery of electrophilic natural products. Cell Chem Biol. 2017;24:1416–27.e5.
Intagliata S, Salerno L, Ciaffaglione V, Leonardi C, Fallica AN, Carota G, et al. Heme oxygenase-2 (HO-2) as a therapeutic target: activators and inhibitors. Eur J Med Chem. 2019;183:111703.
Consoli V, Sorrenti V, Pittala V, Greish K, D’Amico AG, Romeo G, et al. Heme oxygenase modulation drives ferroptosis in TNBC cells. Int J Mol Sci. 2022;23:5709.
Raynes DA, Guerriero V Jr. Inhibition of Hsp70 ATPase activity and protein renaturation by a novel Hsp70-binding protein. J Biol Chem. 1998;273:32883–8.
Wu E, He W, Wu C, Chen Z, Zhou S, Wu X, et al. HSPA8 acts as an amyloidase to suppress necroptosis by inhibiting and reversing functional amyloid formation. Cell Res. 2023;33:851–66.
Shomura Y, Dragovic Z, Chang HC, Tzvetkov N, Young JC, Brodsky JL, et al. Regulation of Hsp70 function by HspBP1: structural analysis reveals an alternate mechanism for Hsp70 nucleotide exchange. Mol Cell. 2005;17:367–79.
Petrie EJ, Birkinshaw RW, Koide A, Denbaum E, Hildebrand JM, Garnish SE, et al. Identification of MLKL membrane translocation as a checkpoint in necroptotic cell death using monobodies. Proc Natl Acad Sci USA. 2020;117:8468–75.
Wang H, Sun L, Su L, Rizo J, Liu L, Wang LF, et al. Mixed lineage kinase domain-like protein MLKL causes necrotic membrane disruption upon phosphorylation by RIP3. Mol Cell. 2014;54:133–46.
Pradhan AJ, Lu D, Parisi LR, Shen S, Berhane IA, Galster SL, et al. Protein acylation by saturated very long chain fatty acids and endocytosis are involved in necroptosis. Cell Chem Biol. 2021;28:1298–309.e7.
Mathema VB, Koh YS, Thakuri BC, Sillanpaa M. Parthenolide, a sesquiterpene lactone, expresses multiple anti-cancer and anti-inflammatory activities. Inflammation. 2012;35:560–5.
Guzman ML, Rossi RM, Karnischky L, Li X, Peterson DR, Howard DS, et al. The sesquiterpene lactone parthenolide induces apoptosis of human acute myelogenous leukemia stem and progenitor cells. Blood. 2005;105:4163–9.
Kwok BH, Koh B, Ndubuisi MI, Elofsson M, Crews CM. The anti-inflammatory natural product parthenolide from the medicinal herb Feverfew directly binds to and inhibits IkappaB kinase. Chem Biol. 2001;8:759–66.
Liu M, Xiao C, Sun M, Tan M, Hu L, Yu Q. Parthenolide inhibits STAT3 signaling by covalently targeting janus kinases. Molecules. 2018;23:1478.
Ding Y, Xue Q, Liu S, Hu K, Wang D, Wang T, et al. Identification of parthenolide dimers as activators of pyruvate kinase M2 in xenografts of glioblastoma multiforme in vivo. J Med Chem. 2020;63:1597–611.
Liu X, Wang C, Li S, Qu L, Yin F, Lu D, et al. Parthenolide derivatives as PKM2 activators showing potential in colorectal cancer. J Med Chem. 2021;64:17304–25.
Bedoui S, Herold MJ, Strasser A. Emerging connectivity of programmed cell death pathways and its physiological implications. Nat Rev Mol Cell Biol. 2020;21:678–95.
Youn CK, Lee JH, Hariharasudhan G, Kim HB, Kim J, Lee S, et al. HspBP1 is a dual function regulatory protein that controls both DNA repair and apoptosis in breast cancer cells. Cell Death Dis. 2022;13:309.
Romanova EA, Yurkina DM, Yashin DV, Sashchenko LP, Georgiev GP. HspBP1 in complex with the peptide of the innate immunity protein Tag7 is able to lyse tumor cells carrying TNFR1 receptor. Dokl Biochem Biophys. 2024;515:36–40.
Quarato G, Guy CS, Grace CR, Llambi F, Nourse A, Rodriguez DA, et al. Sequential engagement of distinct MLKL phosphatidylinositol-binding sites executes necroptosis. Mol Cell. 2016;61:589–601.
Liu S, Liu H, Johnston A, Hanna-Addams S, Reynoso E, Xiang Y, et al. MLKL forms disulfide bond-dependent amyloid-like polymers to induce necroptosis. Proc Natl Acad Sci USA. 2017;114:E7450–9.
Dondelinger Y, Declercq W, Montessuit S, Roelandt R, Goncalves A, Bruggeman I, et al. MLKL compromises plasma membrane integrity by binding to phosphatidylinositol phosphates. Cell Rep. 2014;7:971–81.
Su LJ, Quade B, Wang HY, Sun LM, Wang XD, Rizo J. A plug release mechanism for membrane permeation by MLKL. Structure. 2014;22:1489–500.
Murphy JM, Czabotar PE, Hildebrand JM, Lucet IS, Zhang JG, Alvarez-Diaz S, et al. The Pseudokinase MLKL mediates necroptosis via a molecular switch mechanism. Immunity. 2013;39:443–53.
Huang D, Zheng X, Wang ZA, Chen X, He WT, Zhang Y, et al. The MLKL channel in necroptosis is an octamer formed by tetramers in a dyadic process. Mol Cell Biol. 2017;37:e00497–16.
Pradhan AJ, Chitkara S, Ramirez RX, Monje-Galvan V, Sancak Y, Atilla-Gokcumen GE. Acylation of MLKL impacts its function in necroptosis. ACS Chem Biol. 2024;19:407–18.
Ma B, Marcotte D, Paramasivam M, Michelsen K, Wang T, Bertolotti-Ciarlet A, et al. ATP-competitive MLKL binders have no functional impact on necroptosis. PLoS ONE. 2016;11:e0165983.
Rubbelke M, Fiegen D, Bauer M, Binder F, Hamilton J, King J, et al. Locking mixed-lineage kinase domain-like protein in its auto-inhibited state prevents necroptosis. Proc Natl Acad Sci USA. 2020;117:33272–81.
Xue C, Zhao G, Zhao Y, Chen YE, Zhang J. Mouse abdominal aortic aneurysm model induced by perivascular application of elastase. J Vis Exp. 2022. https://doi.org/10.3791/63608.
Babon JJ, Murphy JM. In vitro JAK kinase activity and inhibition assays. Methods Mol Biol. 2013;967:39–55.
Percher A, Thinon E, Hang H. Mass-tag labeling using acyl-PEG exchange for the determination of endogenous protein S-fatty acylation. Curr Protoc Protein Sci. 2017;89:14.7.1–11.
Acknowledgements
This work was funded by grants from the National Key R&D Program of China (2021YFA1302200), National Natural Science Foundation of China (82204266, 82022065), and the Zhuoyue and Jingying Programs of Second Military Medical University. The project was also supported by the Open Research Fund of the School of Chemistry and Chemical Engineering, Henan Normal University (2024Y05). The authors thank Prof. Tao Pang (State Key Laboratory of Natural Medicines), New Drug Screening Center, Key Laboratory of Drug Quality Control and Pharmacovigilance (Ministry of Education, China Pharmaceutical University, Nanjing 210009, China) for providing the valuable discussion on target identification.
Author information
Authors and Affiliations
Contributions
CZ conceived and designed the research; HS, JW, QH, LX, PD, RW, JL, YH, RH, and YC performed experiments; WW and ZC provided material; HS, JW, QH, PD, JL, PW, LX, TH, and CZ analyzed data. HS, PD, LX, and CZ wrote the manuscript. PW and TH discussed and edited the manuscript. CZ and TH provided funding. All authors contributed with productive discussions and knowledge to the final version of this manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Edited by Professor Massimiliano Agostini
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Shao, H., Wu, J., Han, Q. et al. Identification of human MLKL Cys184 and HSPBP1 Cys201 as novel cellular targets for necroptosis. Cell Death Dis (2026). https://doi.org/10.1038/s41419-026-08764-4
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41419-026-08764-4