Abstract
Mitochondrial dysfunction is one of the core drivers of aging. It is manifested by reactive oxygen species (ROS) accumulation, mitochondrial DNA (mtDNA) mutations, imbalanced energy metabolism, and abnormal biosynthesis. Mitochondrial autophagy maintains cellular homeostasis by selectively removing damaged mitochondria through mechanisms including the ubiquitin-dependent pathway (PINK1/Parkin pathway) and the ubiquitin-independent pathway (mediated by receptors such as BNIP3/FUNDC1). During aging, the decrease in mitochondrial autophagy efficiency leads to the accumulation of damaged mitochondria, forming a cycle of mitochondrial damage-ROS-aging damage and aggravating aging-related diseases such as neurodegenerative diseases and cardiovascular pathologies. The targeted regulation of mitochondrial autophagy (drug modulation and exercise intervention) can restore mitochondrial function and slow aging. However, autophagy has a double-edged sword effect; moderate activation is anti-aging, but excessive activation or dysfunction accelerates the pathological process. Therefore, targeting mitochondrial autophagy may be an effective anti-aging technique; however, future focus should be on the tissue-specific regulatory threshold and the dynamic balance mechanism to achieve precise intervention.

Similar content being viewed by others
Facts
-
Mitochondrial autophagy acts as both a shield and a spear in aging.
-
The balance between AMPK and mTOR is critical for mitochondrial autophagy and SASP.
-
NAD+ benefits mitochondrial autophagy, but prolonged elevation may disrupt mitochondrial homeostasis.
-
Mitochondrial gene editing in vivo requires further validation before clinical translation.
Introduction
Cellular aging involves a multilevel functional decline over time, and its pathological basis is closely related to the progressive dysfunction of mitochondria, the center of energy metabolism. As the power plants and signaling hubs of eukaryotic cells, mitochondria maintain cellular homeostasis through core functions such as oxidative phosphorylation (OXPHOS), calcium homeostasis regulation, and apoptosis execution. However, the accumulation of mitochondrial DNA (mtDNA) mutations, reactive oxygen species (ROS) burst, and kinetic imbalance (fusion/disintegration dysregulation) during aging form a cycle, which not only directly impairs ATP synthesis but also drives age-associated pathologies, such as neurodegeneration and cardiovascular diseases, by inhibiting autophagy flow and activating inflammatory pathways. Mitochondrial autophagy is a key quality control mechanism that selectively removes damaged mitochondria and constitutes the core defense against aging. This process is synergistically regulated by two sophisticated pathways: the ubiquitin-dependent pathway in which PINK1 kinase senses the dissipation of mitochondrial membrane potential and recruits the E3 ubiquitin ligase Parkin, which catalyzes the ubiquitin chain labeling of damaged mitochondria, and bridges LC3 molecules via autophagy receptors (OPTN/NDP52) to form autophagosomes; and the ubiquitin-independent pathway in which outer mitochondrial membrane proteins, such as BNIP3 and FUNDC1, anchor autophagy via the LC3 interaction motif (LIR) in response to hypoxia or stress signaling. Notably, autophagy efficiency decreases significantly with aging, leading to the accumulation of dysfunctional mitochondria, which further amplifies oxidative damage and energy crisis, whereas the overactivation of autophagy may lead to excessive mitochondrial clearance and exacerbate metabolic collapse, which is a double-edged sword that highlights the necessity of precise regulation. The current intervention strategies focus on restructuring mitochondrial homeostasis, including (1) pharmacological activation: urolithin A (UA) enhances the PINK1/Parkin pathway, and nicotinamide mononucleotide (NMN) facilitates SIRT3 (a sirtuin)-mediated deacetylation by elevating NAD⁺ levels; (2) gene editing: modulations of Mitochondrial-targeted Transcription Activator-Like Effector Nucleases (mitoTALEN) targets pathogenic mtDNA mutations; and (3) behavioral interventions: caloric restriction activates the AMPK/SIRT1 axis, and exercise training upregulates FUNDC1 expression to restore autophagic activity. This paper systematically analyzes the molecular mechanism, contradictory roles, and targeted intervention strategies of mitochondrial autophagy in aging, aiming to provide a theoretical basis for treatments that delay aging and related diseases.
The association between aging and mitochondrial dysfunction
Characteristics of aging and the core mitochondrial role
Characteristics of aging
The essence of aging is the cumulative multilevel functional decline of an organism over time. Its pathological basis stems from the progressive deterioration of 12 interrelated markers of aging in cells. These hallmarks include (Fig. 1) genomic instability, telomere damage, epigenetic alterations, the loss of protein arrest, the loss of macrophagy, the dysregulation of nutrient perception, mitochondrial dysfunction, cellular senescence, stem cell depletion, altered intercellular communication, chronic inflammation, and ecological dysregulation [1]. Among them, mitochondrial dysfunction and declining autophagy constitute a central hub. The accumulation of mitochondrial DNA mutations and the bursts of ROS in senescent cells further inhibit autophagic flow, leading to the aggregation of aberrant proteins and the accumulation of damaged organelles, and accelerating tissue degeneration [2].
Drivers and features of aging.
The current study found that aging hallmarks do not exist in isolation but constitute a dynamic network of interactions. For example, sustained DNA damage accelerates telomere depletion, triggering genomic rearrangements and further destabilization, leading to more DNA damage; and the overactivation of mammalian target of rapamycin (mTOR) inhibits autophagy and accelerates mitochondrial function deterioration [3]. Targeted interventions (enhancement of autophagy and epigenetic re-editing) for these features have become key strategies to delay aging and related diseases.
Core functions of mitochondria
As the energy metabolism center and multifunctional signaling hub of eukaryotic cells, mitochondria maintain cellular homeostasis through a precisely coordinated biochemical network. Their core functions include: (1) energy conversion: generating ATP through oxidative phosphorylation to drive cellular activities; (2) metabolic integration: regulating the tricarboxylic acid cycle, lipid metabolism, and ketone body synthesis; (3) calcium homeostasis regulation: acting as a calcium reservoir to buffer cytoplasmic calcium ions and regulating endoplasmic reticulum-mitochondria communication; (4) apoptosis execution: releasing cytochrome c to activate caspase cascade reactions; and (5) ROS signaling: physiological levels of ROS are involved in regulating cell proliferation and differentiation [4]. During aging, mitochondrial dysfunction is usually manifested as a breakdown of energy metabolism, the dissipation of membrane potential, and the accumulation of oxidative damage [5]. Mitochondrial dysfunction is a result of the synergistic effects of multiple mechanisms, involving structural damage caused by imbalances in mitochondrial dynamics (the downregulation of fusion proteins OPA1/MFN2 and the upregulation of the splitting protein Dynamin-related protein 1); metabolic derangement caused by the compensatory enhancement of glycolysis and insulin resistance induced by lipid accumulation; 10–20-fold increases in the mutation rate of mtDNA with age; genomic destabilization caused by the deletion of key subunits of the complex, for example, MT-ND4 genomic destabilization induced by deletion; and quality control failure triggered by decreased mitochondrial capacity due to defective clearance of the autophagy-lysosome system. These interacting mechanisms ultimately lead to the activation of cellular senescence markers [6, 7]. Therefore, mitochondrial function repair has become an important step in anti-aging interventions.
Molecular mechanisms associated with mitochondrial dysfunction and aging
ROS and oxidative stress
Studies have shown that most intracellular ROS originate mainly from mitochondria, and mitochondrial ROS (mtROS) are generated during OXPHOS that occurs on the mitochondrial electron transport chain (ETC) in the inner mitochondrial membrane [8]. The mitochondrial ETC is a redox hub that regulates cellular homeostasis by producing ATP and ROS. ETC dysfunction, especially decreases in mitochondrial energy production and increases ROS production, is related to the onset and progression of many biological changes leading to obesity and aging, as well as pathologies in all organ systems, including cardiovascular disease [9]. Studies have shown that Alzheimer’s disease (AD) patients have fewer intact mitochondria and impaired mitochondrial function attributed to the loss or dysfunction of specific ETC enzymes, and that dysfunctional ETCs trigger ROS accumulation and exacerbate oxidative stress [10]. In addition, excess ROS causes cellular damage and accelerates skin aging [11]. Therefore, the dynamic balance of ROS is a core aspect of maintaining cellular homeostasis, as well as a key driver of a variety of aging-related diseases. Thus, targeting the ROS regulatory network may provide a new strategy for intervening in related pathological processes.
mtDNA mutations
The mitochondrial genome is a small circular molecule of approximately 16,500 bp, with multiple copies in each cell (copy numbers range from 1000 to 10,000), and its encoded products are directly involved in the composition of the ETC complex. Studies have shown that mtDNA can lead to ETC dysfunction by accumulating oxidative damage, which, in turn, induces the aberrant generation of ROS, forming a positive feedback loop that ultimately accelerates the aging process. mtDNA damage is related to the p53 signaling pathway, telomere shortening, and other aging-related mechanisms, further exacerbating the cellular senescence phenotype [12]. Decreased mitochondrial autophagic activity in senescent individuals may be an important mechanism by which mtDNA mutations show age-dependent accumulation [13]. Further studies have shown that mitochondrial dysfunction and mtDNA damage are prevalent in the retinal pigment epithelium of patients with age-related macular degeneration (AMD), and this damage leads to oxidative stress, the collapse of energy metabolism, and inflammatory responses [14]. Additionally, mitochondrial damage leads to mtDNA leakage, the activation of the cGAS-NLRP3 inflammatory pathway and the complement system, the exacerbation of vitreous membrane wart deposition, and chronic inflammation, ultimately leading to the characteristic pathological changes in AMD [15]. mtDNA is associated with energy metabolism, and damage usually leads to an inadequate energy supply. For example, during aging, mtDNA defects cause a decrease in subcutaneous white adipose tissue volume, decreasing lipid storage capacity and triggering systemic lipid metabolic stress [16]. Meanwhile, mtDNA defects lead to excessive ROS and ATP depletion, shifting vascular smooth muscle cells from apoptosis to necrosis and accelerating atherosclerosis [17].
Abnormal energy metabolism
Adenosine monophosphate-activated protein kinase (AMPK) is a core regulator of cellular energy metabolism and plays multiple roles in the aging process. AMPK, as a central integrator of aging-related signals, is capable of regulating mitochondrial homeostasis, delaying aging-related damage, reprogramming energy metabolism, and exerting aging effects. AMPK is activated in all eukaryotic organisms with impaired mitochondrial function or ATP synthesis [18], which enhances oxidative metabolism, inhibits inefficient energy expenditure, reduces glycolysis-dependence in M1 macrophages, and attenuates mitochondrial oxidative damage. It also optimizes energy partitioning through central and peripheral tissue linkages [19].
AMPK activation decreases with aging, which exacerbates mitochondrial dysfunction and energy metabolism imbalances, leading to organismal aging [20]. When AMPK activity decreases, mTOR activity increases. mTOR is a downstream target molecule of AMPK phosphorylation, and the two maintain metabolic homeostasis antagonistically, forming a dynamic yin and yang regulatory network [21]. AMPK was reported to be activated under hypoxic or energetic stress conditions and regulate the secretory phenotype associated with aging by directly inhibiting the mTOR pathway [22]. The sustained activation of AMPK leads to increased ROS levels, dysregulated mitochondrial quality control, aberrant decreases in mtDNA copy numbers, and the excessive inhibition of mTOR, which impairs mitochondrial dynamic homeostasis and reduces mitochondrial stress adaptation [23]. Therefore, the relationship between AMPK and mitochondrial function shows a typical inverted U-shaped curve, moderate activation is beneficial, but weakened or sustained regulation may be harmful. AMPK not only occupies a key position in energy metabolism, but it is also a potential target for anti-aging interventions. Thus, AMPK should be regulated to circumvent its potential negative effects on mitochondrial function by precisely controlling tissue-specific differences, the degree and timing of activation, and dynamic monitoring of the cellular energy status.
Reduced biosynthesis
Mitochondrial biosynthesis refers to the process by which cells maintain their functions through mtDNA replication, respiratory chain protein synthesis, and mitochondrial proliferation. The core regulators include Peroxisome proliferator-activated receptor γ coactivator 1α (PGC-1α), Nuclear Respiratory Factor 1 (NRF1), and mitochondrial transcription factor A (TFAM). Among them, peroxisomal proliferation is the most important. PGC-1α is the master regulator of mitochondrial biosynthesis. It can activate NRF1 and TFAM, mtDNA transcription, and the gene expression of respiratory chain proteins, directly promoting mitochondrial generation and oxidative metabolism. However, PGC-1α expression was found to be downregulated during aging, directly leading to the overall repression of mitochondrial function-related genes and significantly disrupting neuronal energy homeostasis [24]. In addition, in the skeletal muscle of aged mice, PGC-1α overexpression was shown to reverse age-associated decline in mitochondrial translational capacity, restore ETC activity, and induce the transcription of mitochondrial unfolded protein response genes, which help to remove misfolded proteins and maintain mitochondrial protein homeostasis. In contrast, PGC-1α knockdown exacerbated dysfunction, leading to reduced biogenesis, decreased respiratory efficiency, ROS accumulation, and potentially accelerated aging and neurodegenerative pathologies [25].
Mitochondrial autophagy and its mechanisms
Autophagy process and its biological significance
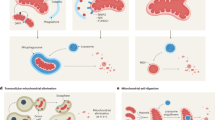
Mitochondrial autophagy (mitophagy) is a sophisticated quality control process in which cells selectively remove damaged mitochondria through autophagy. Its core function is to maintain the stability of the mitochondrial network and balance in the intracellular environment. The process follows a strict staged program, involving the dissipation of the membrane potential of mitochondria, the formation of mitochondrial autophagosomes, the fusion of autophagosomes with lysosomes, and the degradation of mitochondrial contents by lysosomes [26]. In addition, mitochondrial autophagy can specifically remove mitochondria with respiratory chain defects due to mtDNA mutations or ROS overload to achieve quality control, as well as the corresponding energy requirements or metabolic re-editing, and the removal of damaged mitochondria to regulate their numbers [27]. Moreover, mitochondrial dysfunction is a driver of aging, and decreases in mitochondrial autophagy directly drive aging pathology. For example, during aging, the decline in mitochondrial autophagy leads to the accumulation of damaged mitochondria, and these dysfunctional mitochondria produce excessive ROS, which not only causes oxidative damage but also acts as signaling molecules to participate in the regulation of aging, resulting in a cycle of mitochondrial damage-ROS production-more damage [28]. Therefore, mitochondrial autophagy is a central hub connecting organelle health and organism aging, and the precise analysis of its molecular mechanism will provide a revolutionary perspective for anti-aging therapy (Fig. 2).
Mitochondrial autophagy proceeds through four stages: membrane potential dissipation of mitochondria-mitochondrial autophagosome formation-autophagosome fusion with lysosome-mitochondrial contents are degraded by lysosomes. a PINK1-Parkin‐mediated mitophagy. When mitochondria are damaged, the input of PINK1 is hindered and accumulates on the outer mitochondrial membrane. PINK1 then phosphorylates Ub, which then binds to RING1, resulting in the release of RING2 and the exposure of the E2 interaction surface in RING1. RING2 then receives ubiquitin from E2 and transfers it to the substrate. Parkin is activated to ubiquitinate mitochondrial substrates on OMM, allowing LC3 attached to autophagosomes to be recruited through autophagy adaptors OPTN and NDP52, among others. b PINK-Parkin-independent mitophagy. FUNDC1, NIX, BNIP3, BCL2L13, and FKBP8 can bind LC3 alone to mediate mitochondrial autophagy. During reticulocyte maturation, the autophagic receptors NIX, FUNDC1, and BNIP3 are strongly activated, resulting in increased receptor levels on the OMM. Among them, BNIP3 can induce mitochondrial rupture and promote the separation of damaged mitochondria by promoting Drp1. At the same time, BNIP3 recruits Parkin to mitochondria and activates mitochondrial autophagy. Furthermore, hypoxia enhances PGAM5-mediated FUNDC1 dephosphorylation, while FUNDC1 dephosphorylation and mitochondrial fission mediated by FUNDC1-Drp1 complex binding jointly promote mitochondrial autophagy.
Autophagy mechanisms
Mitochondrial autophagy occurs through many different but interrelated mechanisms. These can be categorized into ubiquitin-dependent and ubiquitin-independent pathways.
Ubiquitin-dependent pathway
In the ubiquitin-dependent pathway, damaged mitochondria are recognized and cleared through the PINK1/Parkin signaling pathway, whose core components include PTEN-inducible kinase 1 (PINK1), Parkin, and the ubiquitin chain [29]. PINK1 is the initiating sensor and main regulator of the pathway, and utilizes kinase activity to convert mitochondrial damage signals into autophagy instructions [30]. In the normal state, PINK1 is imported into the inner mitochondrial membrane via the TOM/TIM complex. Upon the loss of mitochondrial membrane potential, PINK1 accumulates in the outer membrane and is activated to phosphorylate ubiquitin to recruit Parkin [31]. Loss-of-function mutations can lead to defective mitochondrial autophagy and early-onset Parkinson’s disease (PD) [32, 33]. Parkin, as a downstream effector of PINK1, consists of an N-terminal ubiquitin-like structural domain (UBL), a Parkin repressor element (REP), and a C-terminal really interesting new gene 2 (RING2) structural domain or C-terminal really interesting new gene 2 (RING2) structural domains [34]. Its activation relies on a PINK1-mediated phosphorylation cascade. Phosphorylation releases UBL and RING1 structural domains from autoinhibition, inhibits the catalytic activity of RING2, and ultimately leads to the construction of the ubiquitin chain by Parkin on the outer mitochondrial membrane protein [35, 36]. The ubiquitin chain marks damaged mitochondria as “to-be-degraded” targets in a specific modification pattern, and they are recognized and cleared by autophagy, which is essential for maintaining mitochondrial homeostasis [37]. Autophagy receptors (p62, NBR1, OPTN, and NDP52) ensure smooth autophagy by binding both cargo and ubiquitin chains [38]. Notably, NDP52 and OPTN can be directly recruited by PINK1 to achieve Parkin-independent mitochondrial autophagy [39]. Activation of the PINK1/Parkin pathway involves three key components: mitochondrial mass monitoring, ubiquitin signaling, and autophagosome recruitment [40]. In summary, PINK1 detects mitochondrial dysfunction and generates ubiquitin phosphate as an autophagy signal. Parkin amplifies and ubiquitinates outer mitochondrial membrane proteins, and ultimately directs the removal of damaged mitochondria by autophagy [41, 42].
Ubiquitin-independent pathway
The ubiquitin-independent pathway regulates mitochondrial autophagy through three mechanisms: (1) direct receptor recruitment to autophagosomes: outer mitochondrial membrane receptors, such as BNIP3, NIX, BCL2L13, and FKBP8, directly bind to the autophagy marker protein, LC3, through their LIR motifs (LC3-interacting region), bypassing the PINK1/Parkin ubiquitylation pathway, and mediate the clearance of damaged mitochondria; (2) stress-induced receptor activation: during hypoxia or mitochondrial damage, receptors (FUNDC1) undergo dephosphorylation or conformational changes to mitigate the inhibitory state to enhance LC3 binding capacity and initiate autophagy; and (3) receptor synergistic network regulation: some receptors (FKBP8 and FUNDC1) can form an interoperative network to finely regulate mitochondrial quality control [43]. Among them, in the BNIP3/NIX pathway, BNIP3 and NIX, which are homologous proteins of BCL2, directly recruit LC3 through LIR motifs without the involvement of ubiquitination. This pathway efficiently clears mitochondria in programmed autophagy (erythrocyte maturation) and in response to acute stress [44, 45]. In contrast, the FUNDC1 pathway anchors autophagosomes to mitochondria by binding LC3 via LIR, and its activity is dynamically regulated by phosphorylation, phosphorylation inhibits LC3 binding in the basal state, and dephosphorylation activates autophagy during hypoxia [46, 47].
Metabolic regulation of autophagy
In addition to the PINK1/Parkin signaling pathway and receptor-dependent pathways described above, metabolites such as iron, calcium, and NAD⁺ regulate mitochondrial autophagy by targeting key molecular nodes. Iron is the most prevalent metal in the mitochondrial matrix, contributing to the complex redox chemistry that facilitates the electron transport chain. Iron levels are closely linked to mitochondrial energy metabolism. Iron overload inhibits mitochondrial autophagy, and excess iron exacerbates ROS bursts, inhibits PINK1 stability and Parkin translocation, and leads to the accumulation of damaged mitochondria. In contrast, iron deficiency activates mitochondrial autophagy, and a low-iron environment induces FUNDC1 dephosphorylation, enhances the ability to bind the LIR motif to LC3, and promotes mitochondrial autophagy. In addition, iron chelators (Deferiprone) promote FIS1 localization in mitochondria through SENP3-mediated FIS1 de-SUMOization, driving mitochondrial division and autophagy [48, 49]. Calcium ions (Ca2+) are one of the most versatile signaling molecules and important regulators of mitochondria. RHOT1/RHOT2 (Miro proteins) act as Ca2+ sensors, coordinating mitochondrial motility and the PINK1/Parkin pathway. At normal Ca2+ concentrations, Miro proteins anchor mitochondria to kinesins and maintain organelle distribution. In cases of excess mitochondrial Ca2+ uptake, mitochondrial membrane potential collapses and inhibits mitochondrial autophagy, leading to cell death [50]. Nicotinamide adenine dinucleotide (NAD⁺) is a key coenzyme widely present in living cells. Its biological function is mainly reflected in two aspects. On the one hand, it acts as a coenzyme of oxidoreductase and participates in the electron transfer process, and on the other hand, it acts as a co-substrate for a variety of enzymes. Studies have shown that the cellular concentration of NAD⁺ changes significantly during the aging process, whereas the mitochondrial autophagy process can be affected by regulating the NAD⁺ metabolic pathway, which, in turn, has a modulating effect on lifespan [51]. NAD⁺ and mitochondrial autophagy are bidirectionally regulated. NAD⁺ promotes autophagosome formation and removes damaged mitochondria by activating nutrient-sensing pathways (AMPK/mTOR) and enhancing autophagic core proteins (ULK1 and the PI3K-III complex). Meanwhile, normal autophagy can maintain NAD⁺ homeostasis, whereas defective autophagy leads to mitochondrial dysfunction, triggering excessive ROS and DNA damage, and the overactivation of PARP/SIRT accelerates NAD⁺ depletion, ultimately leading to cell death. Further studies showed that supplementation with NAD⁺ precursors (NR/NMN) rescued the survival of autophagy-deficient cells and prolonged the healthy lifespan of model organisms [52]. The above metabolites dynamically regulate mitochondrial autophagy efficiency through energy-sensing organelle interactions. Although the specific mechanism of action has not been fully elucidated, these findings reveal the potential association between mitochondrial autophagy and cellular metabolism and provide a new research direction for treating mitochondrial autophagy dysfunction.
The association between mitochondrial autophagy and anti-aging
Double-edged sword effect of autophagy in aging
Mitochondrial autophagy maintains cellular homeostasis by selectively removing damaged mitochondria; however, it exhibits a loss of dynamic equilibrium during the aging process. The moderate activation of mitochondrial autophagy can precisely regulate the balance between mitochondrial quality and quantity to delay aging [53]. For example, ubiquitin-dependent autophagy mediated by the PINK1/Parkin pathway recognizes mitochondria with dissipated membrane potential and recruits LC3 to form an autophagosome via the OPTN/NDP52 receptor to remove defective respiratory chain units, while receptors such as BNIP3/FUNDC1 directly anchor LC3 via LIR motifs in response to cellular stress signals to maintain the balance of mitochondrial numbers [28]. In contrast, mitochondrial autophagy efficiency may decrease during aging, leading to the accumulation of damaged mitochondria, which exacerbates cellular damage, creating a vicious cycle. For example, defective mitochondrial autophagy in AD patients leads to the accumulation of damaged mitochondria, and the inability to efficiently scavenge dysfunctional mitochondria exacerbates oxidative damage and an energy crisis [54]. In addition, the over-activation of mitochondrial autophagy in some cases may lead to excessive mitochondrial clearance, affecting energy availability. For example, in Drosophila melanogaster mtDNA mutants, PINK1 overexpression triggered excessive autophagy that removed greater than 90% of the mitochondria, leading to a collapse of ATP synthesis with stagnant larval development [55]. In summary, mitophagy acts as both a shield (removing damage to maintain homeostasis) and a spear (accelerating pathology when dysregulated) in aging. Future studies need to focus on tissue-specific thresholds and dynamic equilibrium nodes to achieve precise interventions (Fig. 3).
Dual roles of autophagy: protective barrier and pathology driver.
Mechanisms of autophagy regulation during aging
Gene regulation
Mitochondrial gene regulation is critical for maintaining respiratory chain function and redox homeostasis, and its disruption accelerates aging and degenerative disease processes by disrupting energy metabolism and increasing oxidative damage [56, 57]. Gene regulation is multidimensionally dysregulated during aging (Fig. 4). At the epigenetic level, the episodic silencing of key mitochondrial genes (elevated DNA methylation) exacerbates energy metabolism disorders, ROS accumulation, and cellular senescence, further accelerating epigenetic drift [58], and disturbed methylation patterns can lead to transposon activation and DNA damage accumulation, exacerbating cellular functional decline [59]. Meanwhile, aberrant histone modifications (H3K27me3 deletion) directly lead to the aberrant activation of senescence-associated secretory phenotypic (SASP) genes pro-inflammatory factors such as interleukin (IL-6), driving chronic inflammation [60]. At the transcriptional level, compared with normal cells, the key autophagy-lysosomal master transcription factor (TFEB) retains the cytoplasm and is phosphorylated and inactivated more than 3-fold longer in senescent cells, leading to the expression of autophagy genes (ATG5 and LAMP1) is downregulated by 50%, causing a decrease in the expression of lysosomal and autophagy-related genes. Thus, erroneous proteins and damaged organelles cannot be effectively cleared, causing a cycle of metabolic disorders and dysregulated gene expression, which accelerate the occurrence of aging and related diseases [61]. Mitochondrial autophagy releases Ca²⁺, which induces TFEB activation and promotes nuclear translocation. TFEB entry into the nucleus directly upregulates PINK1 and Parkin expression and promotes mitochondrial biogenesis via the PPARα/PGC-1α axis [62]. Martini-Stoica et al. demonstrated that the activation of TFEB by drugs (alginin) promotes its nuclear translocation, restores lysosomal function, significantly reduces pathological inclusions of α-synuclein, and ameliorates behavioral deficits in animal models [63]; however, the over-activation of TFEB may lead to lysosomal overconsumption or autophagic cell death [64]. Researchers have found that DNA methylation changes are involved in senescence regulation, affecting cellular function through the methylation disruption of developmentally critical genes (imprinted genes), as well as inducing genomic instability (transposon activation) associated with senescence [65]. In contrast, the naked mole rat, which has specific epigenetic regulation, maintains an “ageless” phenotype through multiple mechanisms, including the LHX3-POU transcriptional network, the precise regulation of methylation of developmental genes, and transposon silencing [66]. Although methylation changes are widely observed during aging (small but widespread CpG/CpH site alterations), direct evidence that these changes are a driver rather than a concomitant phenomenon of aging is lacking [67].
Gene regulation.
Mitochondrial quality control
Mitochondria are important organelles within eukaryotic cells. Mitochondrial quality control (MQC) has evolved in response to a variety of physiological signals and external stimuli to maintain mitochondrial health and function through a multilayered synergistic mechanism. These include mitochondrial proteostasis maintenance, the regulation of mitochondrial dynamic homeostasis, mitochondria-derived vesicle formation, and mitochondrial autophagy (Fig. 5) [68]. Senescence leads to synergistic MQC failure and triggers cascading damage. The reduced expression or activity of PINK1 and Parkin in senescent cells restricts the clearance of damaged mitochondria, which triggers a variety of diseases. For example, in PD patients, PINK1/Parkin mutations cause impaired mitochondrial autophagy and mitochondrial accumulation in dopamine neurons, leading to oxidative stress and cell death [35]. The mitochondrial unfolded protein response (UPRmt) is a key mechanism for maintaining mitochondrial proteostasis, and mitochondria in aging organisms experience elevated proteotoxic stress (misfolded protein accumulation), UPRmt depletion, and the downregulation of molecular chaperones (HSPA9/HSPD1) and proteases (LONP1), which results in the accumulation of misfolded proteins in mitochondria that inhibit autophagy [69]. MQC can be positively regulated by mitochondrial autophagy, and autophagy cooperates with UPRmt to build a repair-clearance bipartite axis, UPRmt mainly repairs mitochondria damaged in the early stage, and if it fails to do so, it promotes the autophagy of PINK1/Parkin to clear irreparable mitochondria, which can help to restore OXPHOS function and maintain mitochondrial homeostasis. Parkin expression (or the lack of expression) does not interfere with the levels of activated UPRmt. Parkin expression and UPRmt activation are independently initiated but have complementary functions. Together, they constitute a double-insurance mechanism for mitochondrial quality control [70]. Wenshu Cong et al. reported that cobalt oxide nanoparticles modified with dimercaptosuccinic acid could serve as novel UPRmt activators. The metabolic regulation and protein homeostatic network enhancement by activating UPRmt significantly prolonged the healthy lifespan in the cryptic rod nematode (C. elegans) model [71]. The aging process is often accompanied by an imbalance in mitochondrial dynamics, which causes mitochondrial dysfunction, leading to cell fate abnormalities and a range of aging-related diseases [72]. Mitochondrial autophagy can couple with kinetic balances to optimize quality control. For example, autophagy targets the removal of excessive DRP1-mediated division products, and moderate fusion prevents autophagic overactivation [73]. Further studies revealed that mitochondrial GTP metabolism controls reproductive senescence in Cryptomeria hollisteri by promoting mitochondrial fission, preventing the aggregation of damaged mitochondria, and enhancing the efficiency of mitochondrial autophagy, which subsequently maintains mitochondrial mass in oocytes [74]. Thus, mitochondrial autophagy reverses senescence-associated MQC failure through the triple action of removing irreparable mitochondria, synergizing UPRmt repair, and regulating kinetic homeostasis.
a Healthy mitochondria. The damaged parts of mitochondria split from the healthy parts by split proteins and are encapsulated by autophagosomes for mitochondrial autophagy. While the healthy part of the mitochondria fuses with other mitochondria to achieve its anti-aging function. b Damaged mitochondria. Protein misfolding, mtROS and mtDNA leakage, and oxidative phosphorylation damage can all activate URPmt. URPmt initiation induces ATF4 to bind to CHOP, while ATF5 directly acts on the mitochondrial chaperone promoter, upregulating the expression of HSPA9 and LONP1 to rebuild homeostasis in mitochondria. In addition, PERK further activates the expression of ATF4 by phosphorylating eIF2α, thereby repairing oxidative phosphorylation damage. c Mitochondrial dysfunction. Aging causes decreased expression of PGC-1α and AMPK and impaired ETC, which in turn affects the normal transcription of autophagy genes, upregulates mTOR expression, and leaks ROS and mtDNA, ultimately exacerbating mitochondrial dysfunction.
Protein regulation
Proteostasis (protein homeostasis) maintains cellular function and delays aging by safeguarding proper protein folding and degradation. It is a key barrier to maintaining cellular function, and its decline with age is a central driver of aging (Fig. 6) [75]. The recession of long-life signaling pathways, such as Heat Shock Factor 1/Dauer Formation 16, during aging leads to the accumulation of misfolded proteins and increases protein damage, which accelerates aging [76]. In addition, the increased oxidative modification of proteins associated with aging and decreased clearance capacity synergize to cause protein aggregation problems, leading to the ineffective clearance of misfolded proteins and the development of functionally acquired toxicity disorders such as PD, Huntington’s disease, and AD [77]. Researchers found that mitochondrial autophagy improves protein homeostasis via a dual pathway. On the one hand, autophagy directly removes the sources of damage through protein mediation. For example, autophagy and beclin 1 regulator 1 (AMBRA1), a central hub for mitochondrial quality control and protein homeostasis, removes toxic protein-containing mitochondria by recruiting LC3 through PINK1/Parkin-dependent and non-dependent pathways. However, aging causes its dysfunction, leading to neuronal death and neurodegeneration [78]. On the other hand, autophagy can synergize with the sirtuin family (Table 1) to delay aging by regulating protein acetylation homeostasis, energy metabolism, and oxidative stress [79]. SIRT1, located in the nucleus and cytoplasm, regulates mitochondrial biosynthesis, and indirectly affects the number of autophagy substrates through deacetylating transcription factors (PGC-1α) [80]. Its downregulation enhances immune surveillance (inhibits the secretion of senescence associated secretory phenotype factors) during acute aging [150]. SIRT3, located in mitochondria, directly manages beta oxidation, ATP synthesis, and ROS scavenging [152]. Further studies revealed that SIRT3 overexpression significantly activated autophagy, reversed decreases in mitochondrial membrane potential induced by advanced glycosylation end products (AGEs), and maintained mitochondrial integrity [153]. Mitochondrial autophagy blocks the cycle of protein homeostasis through AMBRA1-mediated clearance and the synergistic repair of sirtuins, providing a new concept for improving mitochondrial quality as well as slowing the aging process.
Protein homeostasis network.
Activation of the inflammatory response
During aging, almost all cells in the body undergo senescence, and senescent cells drive chronic inflammation through a SASP: they secrete factors such as IL-6, tumor necrosis factor (TNF)-α, and transforming growth factor (TGF)-β, which recruit immune cells to remove senescent cells. However, when immune escape occurs, the continued release of SASP triggers “inflammaging”, leading to tissue fibrosis, organ damage, and age-related diseases (Alzheimer’s disease and atherosclerosis), while inflammatory signaling feedback promotes more cellular senescence, creating a self-reinforcing cycle [81, 82]. Mitochondria are central to this triangular cycle: dysfunctional mitochondria release molecules such as mtDNA and cardiolipin, which activate SASP and systemic inflammation via cGAS-STING and the NLRP inflammasome, a dual pathway that further damages mitochondria (Fig. 7) [2, 83, 84]. SASP is also associated with most of the non-autonomous effects observed in senescent cells, including inflammation, senescence enhancement, and paracrine senescence [85]. NLRP3, which is a mitochondrial damage sensor, is the most strongly associated with senescence in the many inflammatory vesicles (Table 2 Introduction to inflammatory vesicles) [86]. In contrast, defective mitochondrial autophagy induces ROS overproduction [87], mtDNA leakage, and the exposure of cardiolipin, which are signals that activate NLRP3, leading to the onset of inflammatory responses [88,89,90]. Thus, intact and functioning mitochondria are the primary gatekeepers controlling genetic, metabolic, and inflammatory homeostasis [3]. Mitochondrial autophagy removes damaged mitochondria, blocks mtDNA leakage, degrades oxidized phospholipids and removes inflammatory triggers [91]. The specific knockdown of mitochondrial transcription factor A (TFAM) decreased mtDNA synthesis in mouse macrophages and significantly inhibited NLRP3 activation, thus demonstrating that mtDNA synthesis is the rate-limiting step in NLRP3 activation [92]. In addition, lowering mtROS levels also inhibited TFAM-mediated mtDNA synthesis and NLRP3 activation [93]. Mitochondrial autophagy not only secures the ATP supply and prevents ATP/ADP imbalances to weaken NLRP3 autoinhibition [94], but also wraps cytoplasmic mtDNA to form mtDNA-trapping vesicles [95], which prevents it from contacting cGAS to block the cGAS-STING pathway, inhibiting the release of SASP factor [83, 84]. In conclusion, enhancing mitochondrial autophagy protects the normal function of mitochondria, which helps to ensure their integrity to avoid inflammation caused by the leakage of pro-inflammatory factors, and thus slows aging (Table 3).
Links between aging and inflammation.
Regulating mitochondrial autophagy to fight aging
Mitochondrial autophagy is a cellular quality control mechanism and may be a driver of pathological processes, depending mainly on factors such as the extent of damage, cell type, and the strength of pathway activation. Early in the process, aging can be slowed by removing damaged mitochondria; however, with age, mitochondrial autophagy may become a pro-aging process due to lysosomal decline or the erroneous degradation of key proteins [57]. Therefore, intervening in autophagy using rational means is extremely important for delaying aging (Table 4).
Pharmacological modulation
Chemical drugs
Urolithin A (UA) is a naturally occurring polyphenol produced in the intestine by ellagitannins and ellagic acid [96]. UA promotes mitochondrial autophagy by modulating the PINK1/Parkin pathway, as well as activating other receptor proteins and directly recruiting LC3 to promote autophagosome formation. Clinical studies have shown that all 13 mitochondria-related genes were significantly upregulated in the skeletal muscle of elderly people after 4 weeks of 500–1000 mg UA administration [97]. Further experiments revealed that the lifespan of the Cryptococcus hidradii nematode was significantly extended by 45.4% after treatment with 50 μM UA [98]. Sulforaphane (SFN), mainly derived from cruciferous vegetables, is an NRF2 (NFE2L2) activator. SFN was found to activate the PINK1/Parkin pathway by promoting NRF2 nuclear translocation in aging mice, which removes damaged mitochondria and reduces the aging phenotype [99]. Spermidine is an endogenous metabolite that slows or prevents the progression of neurodegenerative diseases with age [100]. Aging leads to a significant decline in brain levels of spermidine, and supplementation with spermidine activates mitochondrial autophagy, reducing oxidative damage and impaired energy metabolism within neurons [101]. For example, short-term memory and intermediate memory were significantly restored in aged Drosophila after spermidine supplementation [102]. Spermidine was shown to promote the formation of mitochondrial autophagosomes in the Aβ toxic environment of AD transgenic mice by activating the PINK1/Parkin pathway, which removes mitochondrial debris in AD models [103]. Metformin is a drug approved for treating diabetes, but it has been found to regulate aging by inhibiting inflammatory pathways and modulating oxidative stress, autophagy, and protein synthesis [104]. Previous studies demonstrated that the core mechanism of metformin in prolonging the lifespan is by activating AMPK, which directly phosphorylates ULK1 (autophagy-initiating kinase) and promotes the formation of autophagy precursors. It also upregulates the expression of PINK1, which promotes the recruitment of Parkin to the mitochondria and labels damaged mitochondria for clearance [105]. NAD⁺ is one of the several key markers that directly affect aging. During aging, NAD⁺ levels decrease significantly, leading to impaired mitochondrial autophagy and shortened lifespan, whereas nicotinamide mononucleotide (NMN), a direct precursor of NAD⁺, reversed this trend by upregulating the PINK1/Parkin pathway [106] and facilitating the clearance of damaged mitochondria (median lifespan was 15–20% longer in different strains of mice) [107]. In addition, NMN is closely related to sirtuins, reducing ROS damage and rebuilding the metabolic network of senescent cells by regulating the precise deacetylation of 39 mitochondrial proteins by SIRT3 [108]. NMN also ameliorates senescence-associated neurovascular function and cognitive deficits by upregulating SIRT1 activity. For example, in a nematode model of AD, NMN significantly ameliorated cognitive decline by elevating sirtuin levels and inducing neuronal mitochondrial autophagy [109]. Researchers found that insufficient NAD⁺ biosynthesis in rodent liver, adipose tissue, and skeletal muscle led to metabolic abnormalities associated with obesity and aging, which were ameliorated by NMN supplementation [110]. Increased levels of NAD⁺ may promote mitochondrial biosynthesis through pathways such as PGC-1α and alleviate energy crises in senescent cells [111]. NAD⁺ metabolites may likewise activate energy-sensing pathways and promote the expression of mitochondrial autophagy-related proteins (ULK1 and BNIP3) to retard senescence [112]. In addition, the NAD precursor nicotinamide riboside (NR) maintains mitochondrial homeostasis and alleviates the aging process by activating UPRmt to remove misfolded proteins [113]. Rapamycin was found to mimic developmental signals (meiotic initiation) to trigger programmed mitochondrial autophagy by inhibiting the mTORC1 pathway, which has very positive therapeutic significance for autophagy dysfunction caused by aging [114].
Traditional Chinese medicine and its active ingredients
Anti-aging has become a hot topic in Chinese medicine. Researchers have achieved many good anti-aging effects of flavonoids. Quercetin, a natural flavonoid found in a variety of fruits and vegetables, is a particularly promising treatment for various systemic or degenerative diseases due to its anti-aging, antioxidant, anti-inflammatory and anti-tumor properties [115]. Quercitin was shown to promote mitochondrial autophagy and scavenge damaged mitochondria by upregulating the PINK1/Parkin pathway [116]. Further mechanistic studies showed that quercetin greatly ameliorated senescence-associated proliferative disorders, reduced pro-senescence proteins, and exhibited significant anti-aging effects in a mesenchymal stem cell model of patients with Werner syndrome and Hutchinson-Gilford premature aging syndrome [117]. The combination of quercetin and other drugs also has great anti-aging potential. The median lifespan of premature aging model mice treated with the combination of quercetin and dasatinib was extended by 25%, which was superior to that of the single-agent group (quercetin 15%, dasatinib 10%) [118]. In addition to quercetin, goldfinch isoflavin, a soy isoflavone, has been shown to attenuate aging by modulating mitochondrial autophagy. Ovariectomized rats treated with goldfinch isoflavin showed activated mitochondrial autophagy and cleared abnormally functioning mitochondria, resulting in reduced ROS accumulation and DNA damage, as well as the downregulation of aging-associated secretory phenotypes [119]. Psoralen was found to activate mitochondrial autophagy by inhibiting the mTOR1 signaling pathway and significantly reducing fasting blood glucose, total cholesterol, low-density lipoprotein, and triglyceride levels in an aged mouse model. Improvements in these metabolic indexes were correlated with the enhancement of mitochondrial function, suggesting that it may slow aging by optimizing mitochondrial function [120]. The results showed that the lifespan of yeast, worms, and Drosophila was extended by treatment with the flavonoid 4,4′-dimethoxychalcone (DMC). DMC also slowed the aging process in human cell cultures [121]. Further studies revealed that DMC acts through GATA transcription factors to enhance autophagic flow and reduce p62 homologs in Drosophila brains [122]. The cGAS-STING pathway plays an important role in the pathogenesis of cellular inflammation and senescence diseases, making it an attractive drug target. Notably, STING signaling was aberrantly activated in senescent mouse model tissues, but the phosphorylation levels of STING and its downstream molecules were significantly reduced by ginkgetin treatment [123]. The results indicate that ginkgetin delays aging by specifically targeting STING, providing a new theoretical basis and potential drug candidate for anti-aging intervention.
Progress in clinical research and exploration of traditional Chinese medicine
In the current anti-aging research targeting mitochondrial autophagy, several clinical trials are actively exploring its translational potential. For example, UA, as a natural polyphenolic metabolite, has demonstrated its safety in human trials and has shown the potential to enhance mitochondrial gene expression and improve muscle function [124]. In addition, metformin is one of the most promising experimental anti-aging interventions at present, and clinical trials have shown that diabetic patients treated with metformin have a mild decline in cognitive function [125], but another trial showed that opposite result [126]. Overall, metformin is a very promising drug to slow down aging and prolong healthy life, with controllable side effects. NR is a NAD+ precursor substance, which shows good safety and tolerability during the treatment of PD patients, and provides a basis for the treatment of aging-related neurodegenerative diseases [127]. One of the pathways most obviously related to aging is the mTOR pathway, and clinical studies have found that the mTOR inhibitor RAD001 can improve the age-related decline of immune function in the elderly, confirming the scientific feasibility of targeting the mTOR pathway to improve immune function in the elderly [128, 129]. Dasatinib usually used in combination with the flavonol quercetin as senolytics, and relevant clinical trials have officially confirmed that this regimen has a systemic anti-aging effect, specifically inducing apoptosis of senescent cells, and simultaneously affecting aging markers in multiple tissues such as adipose tissue, skin, and circulatory system [130, 131]. Aging is a long-term change process accompanied by the occurrence of a variety of diseases. The western medicine used for anti-aging has long medication time and obvious side effects. Although traditional Chinese medicine has diverse ingredients and complex mechanisms, it has unique advantages in treating such diseases, which can meet medication needs and change clinical decisions. Astragali Radix has been proven effective in treating heart failure and activate mitochondrial autophagy to delay aging [132, 133]. Ginseng is believed to prolong life. Studies have confirmed that ginseng and its active ingredients can exert systemic anti-aging effects by regulating various aging-related pathways [134, 135]. Although several drugs have been proven to have good clinical efficacy, technological innovation and breakthroughs are still needed to ensure higher levels of safety, efficacy, and quality control for the high-quality development of Chinese medicines [136].
Gene and biological therapy
Genetic engineering is the science of artificially modifying the genome of an organism using molecular biology techniques to add new characteristics or to treat disease by cutting, splicing, inserting, or deleting DNA. The use of genetic engineering techniques for the genetic and epigenetic control of cells has brought very favorable information to biotechnology and medicine [137]. Genetic engineering is related to mitochondrial autophagy and anti-aging research, which can slow the aging process by precisely regulating the autophagy pathway to enhance the ability of cells to “self-clean.”
CRISPR-Cas9
CRISPR-Cas9 is derived from the adaptive immune system of bacteria and archaea as a defense against exogenous genetic material. Its core components include a CRISPR array of direct repeats and spacers, and the Cas gene, which has nuclease activity to encode the Cas protein. By designing guide RNAs to pair complementarily with the target DNA, Cas9 generates double-strand breaks at specific locations. The cell subsequently achieves knockouts or insertions via non-homologous end-joining or homologous recombination repair [137, 138]. For example, the main pathological features of human PD must be simulated in PD research, but cannot be simulated by mouse and pig models. However, a non-human primate model with Parkin gene deletion can be successfully constructed using CRISPR-Cas9 technology, which not only solves the modeling challenge but also confirms the critical role of Parkin phosphorylation defects in the pathogenesis of PD [139]. Protein Kinase B (AKT) can directly regulate mitochondrial biosynthesis and function, which is conducive to improving mitochondrial function [140]. After the target editing of miR-29b using CRISPR-Cas9 in mice, the protein levels of its downstream target genes, insulin-like growth factor 1 and phosphatidylinositol 3-kinase, were significantly restored, which are the upstream activators of the AKT pathway.
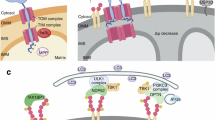
TALEN
In addition to using CRISPR-Cas9 technology, TALEN is the choice in scenarios lacking suitable targets and needing extremely high specificity (Fig. 8). TALEN is a gene editing tool that is on par with CRISPR-Cas9. However, unlike CRISPR-Cas9’s recognition mode, TALEN relies on a protein-DNA recognition mode, which consists of two fusion proteins and thus has a higher molecular weight. mitoTALEN, which is derived from TALEN to target mitochondrial DNA, has been used more frequently, both for the specific recognition and cleavage of mtDNA sequences carrying disease-causing mutations and for the preferential removal of mutant mtDNAs in heterogeneous cells containing a mixture of wild-type and mutant mtDNAs. MitoTALEN has been shown to effectively target two common mtDNA mutations that both accumulate during aging and lead to mitochondrial dysfunction. Moreover, the treated heterogeneous cells were able to restore normal respiratory function and significantly enhance their oxidative phosphorylase activity [141]. Further studies found that the MitoTALEN system significantly reduced the proportion of mutant mtDNA in a treated mouse model of heterogeneous mitochondrial DNA harboring the m.5024 C > T mutation by specifically cleaving the m.5024 C > T mutation in the tRNAAla gene, which provides direct evidence for gene editing treatment of mitochondrial heter [142]. Thus, MitoTALEN shows great potential as a novel anti-aging intervention.
Comparison of TALEN with CRISPR-Cas9.
Other modulations
A healthy life cannot be separated from good living habits, and diet and regular exercise in daily life are important signs of health. A reasonable diet includes the quantity, quality, and composition of food, as well as mealtime. Dietary regulation is more easily accepted by the public compared with drug treatment, and regulating nutritional availability will directly affect our health and life expectancy. Modifications include dietary interventions based on traditional eating habits, such as fasting, calorie restriction, the Mediterranean diet, and protein restriction, as well as newer dietary interventions, such as the ketogenic diet, glucose and carbohydrate restriction, amino acid interventions, and micronutrient interventions [143]. Caloric restriction was found to be effective in increasing the rate of mitochondrial protein synthesis in older individuals, primarily by activating the AMPK/SIRT1 signaling pathway, which improves the coordinated expression of nuclear and mitochondrial-encoded proteins [144]. Further studies have shown that caloric restriction significantly promoted mitochondrial biogenesis and improved mitochondrial electron transport chain activity, especially in the skeletal muscle and liver tissues of aging mice [145]. In addition, the ketogenic diet has been a hot topic in recent years, and researchers have found that the ketogenic diet not only promotes mitochondrial autophagy and protein renewal by inhibiting mTORC1 (35–50% reduction in activity) and the activation of AMPK (60–80% increase in activity), but also significantly enhances fat β-oxidation capacity by 40–60%, optimizes tricarboxylic acid cycle fluxes, and improves the aging-associated decline in mitochondrial metabolic efficiency [146].
Exercise conditioning is also important for healthy functioning and treating diseases, and exercise can significantly promote mitochondrial biogenesis to slow aging and ameliorate aging-related metabolic diseases [147]. Studies have shown that exercise training can restore the aging-induced decline in FUNDC1 protein levels, restore mitochondrial autophagy activity, and significantly delay the aging process of coronary artery endothelial cells in aged mice [148]. In addition, exercise was reported to upregulate PGC-1α expression, promote the dynamic balance between mitochondrial biogenesis and autophagy, and activate anti-inflammatory responses and neuroendocrine regulation [149]. These findings suggest that exercise can modulate mitochondrial autophagy in a multi-system and multi-targeted manner, and is one of the most promising non-pharmacological intervention strategies to slow aging.
Summary and prospects
This paper systematically elaborates the central position of mitochondrial dysfunction in aging and its involvement in regulating mitochondrial autophagy. Mitochondrial autophagy is a key anti-aging target, and removing damaged mitochondria blocks ROS accumulation, mtDNA leakage, and inflammatory cascades (inhibition of the cGAS-STING/NLRP3 pathway). Autophagy activators significantly extend model organism lifespan by enhancing the PINK1/Parkin or receptor pathway. For example, UA upregulates mitochondrial gene expression to promote autophagy, NMN repairs metabolic networks via SIRT3 deacetylation, and metformin activates the AMPK-ULK1 axis. In addition, gene editing and lifestyle can likewise restore mitochondrial quality control and optimize autophagy. However, the shortcomings of the study are that, on the one hand, clinical data on most drugs (UA and NMN) are limited to short-term trials, and the long-term safety and tissue-specific effects are unknown. On the other hand, the model of aging research is limited, and experiments are mostly dependent on nematode/mouse models, which have significant physiological differences from humans. The complexity of the effects of gender and genetic background on autophagy regulation has also not been sufficiently explored. Therefore, human safety studies of drugs such as UA and NMN should be advanced to assess long-term anti-aging benefits. Meanwhile, combining AI models to predict autophagy dynamic equilibrium nodes to guide personalized anti-aging interventions should be performed. In summary, mitochondrial autophagy provides a revolutionary perspective for anti-aging; however, its complexity requires cross-scale studies and interdisciplinary collaboration. In the future, a balance between the depth of mechanism and clinical feasibility should be pursued, ultimately realizing the precise removal of damage and the remodeling of metabolic youth.
References
López-Otín C, Blasco MA, Partridge L, Serrano M, Kroemer G. Hallmarks of aging: An expanding universe. Cell. 2023;186:243–78.
Guilbaud, E, Sarosiek, KA, Galluzzi L. Inflammation and mitophagy are mitochondrial checkpoints to aging. Nat Commun. 2024;15:3375–77.
López-Otín C, Pietrocola F, Roiz-Valle D, Galluzzi L, Kroemer G. Meta-hallmarks of aging and cancer. Cell Metab. 2023;35:12–35.
Monzel AS, Enríquez JA, Picard M. Multifaceted mitochondria: moving mitochondrial science beyond function and dysfunction. Nat Metab. 2023;5:546–62.
Miwa S, Kashyap S, Chini E, von Zglinicki T. Mitochondrial dysfunction in cell senescence and aging. J Clin Investig. 2022;132:e158447.
Amorim JA, Coppotelli G, Rolo AP, Palmeira CM, Ross JM, Sinclair DA. Mitochondrial and metabolic dysfunction in ageing and age-related diseases. Nat Rev Endocrinol. 2022;18:243–58.
Guo, Y, Guan, T, Shafiq, K, Yu, Q, Jiao, X, Na, D, et al. Mitochondrial dysfunction in aging. Ageing Res Rev. 2023;88:101955.
Sies H, Jones DP. Reactive oxygen species (ROS) as pleiotropic physiological signalling agents. Nat Rev Mol Cell Biol. 2020;21:363–83.
Nolfi-Donegan D, Braganza A, Shiva S. Mitochondrial electron transport chain: Oxidative phosphorylation, oxidant production, and methods of measurement. Redox Biol. 2020;37:101674.
Bai R, Guo J, Ye XY, Xie Y, Xie T. Oxidative stress: The core pathogenesis and mechanism of Alzheimer’s disease. Ageing Res Rev. 2022;77:101619.
Gu Y, Han J, Jiang C, Zhang Y. Biomarkers, oxidative stress and autophagy in skin aging. Ageing Res Rev. 2020;59:101036.
Lauri A, Pompilio G, Capogrossi MC. The mitochondrial genome in aging and senescence. Ageing Res Rev. 2014;18:1–15.
Sen A, Boix J, Pla-Martín D. Endosomal-dependent mitophagy coordinates mitochondrial nucleoid and mtDNA elimination. Autophagy. 2023;19:2609–10.
Kaarniranta K, Blasiak J, Liton P, Boulton M, Klionsky DJ, Sinha D. Autophagy in age-related macular degeneration. Autophagy. 2023;19:388–400.
Kaarniranta K, Uusitalo H, Blasiak J, Felszeghy S, Kannan R, Kauppinen A, et al. Mechanisms of mitochondrial dysfunction and their impact on age-related macular degeneration. Prog Retin Eye Res. 2020;79:100858.
Nguyen TT, Corvera S. Adipose tissue as a linchpin of organismal ageing. Nat Metab. 2024;6:793–807.
Grootaert MOJ, Moulis M, Roth L, Martinet W, Vindis C, Bennett MR, et al. Vascular smooth muscle cell death, autophagy and senescence in atherosclerosis. Cardiovasc Res. 2018;114:622–34.
Herzig S, Shaw RJ. AMPK: guardian of metabolism and mitochondrial homeostasis. Nat Rev Mol Cell Biol. 2018;19:121–35.
Townsend LK, Steinberg GR. AMPK and the Endocrine Control of Metabolism. Endocr Rev. 2023;44:910–33.
Salminen A, Kaarniranta K, Kauppinen A. Age-related changes in AMPK activation: Role for AMPK phosphatases and inhibitory phosphorylation by upstream signaling pathways. Ageing Res Rev. 2016;28:15–26.
González A, Hall MN, Lin SC, Hardie DG. AMPK and TOR: The Yin and Yang of Cellular Nutrient Sensing and Growth Control. Cell Metab. 2020;31:472–92.
van Vliet T, Varela-Eirin M, Wang B, Borghesan M, Brandenburg SM, Franzin R, et al. Physiological hypoxia restrains the senescence-associated secretory phenotype via AMPK-mediated mTOR suppression. Mol Cell. 2021;81:2041–52.e2046.
Burkewitz K, Zhang Y, Mair WB. AMPK at the nexus of energetics and aging. Cell Metab. 2014;20:10–25.
Huang T, Lin R, Su Y, Sun H, Zheng X, Zhang J, et al. Efficient intervention for pulmonary fibrosis via mitochondrial transfer promoted by mitochondrial biogenesis. Nat Commun. 2023;14:5781.
Souder DC, McGregor ER, Clark JP, Rhoads TW, Porter TJ, Eliceiri KW, et al. Neuron-specific isoform of PGC-1α regulates neuronal metabolism and brain aging. Nat Commun. 2025;16:2053.
Chen X, Luo Y, Zhu Q, Zhang J, Huang H, Kan Y, et al. Small extracellular vesicles from young plasma reverse age-related functional declines by improving mitochondrial energy metabolism. Nat Aging. 2024;4:814–38.
Lu Y, Li Z, Zhang S, Zhang T, Liu Y, Zhang L. Cellular mitophagy: Mechanism, roles in diseases and small molecule pharmacological regulation. Theranostics. 2023;13:736–66.
Youle RJ, Narendra DP. Mechanisms of mitophagy. Nat Rev Mol Cell Biol. 2011;12:9–14.
Campisi J, Kapahi P, Lithgow GJ, Melov S, Newman JC, Verdin E. From discoveries in ageing research to therapeutics for healthy ageing. Nature. 2019;571:183–92.
Gatica D, Lahiri V, Klionsky DJ. Cargo recognition and degradation by selective autophagy. Nat Cell Biol. 2018;20:233–42
Yin Z, Klionsky DJ. Pink1 balances reticulophagy and mitophagy by regulating distinct E3 ubiquitin ligases. Autophagy. 2024;20:983–4.
Harper JW, Ordureau A, Heo JM. Building and decoding ubiquitin chains for mitophagy. Nat Rev Mol Cell Biol. 2018;19:93–108.
Gan ZY, Callegari S, Cobbold SA, Cotton TR, Mlodzianoski MJ, Schubert AF, et al. Activation mechanism of PINK1. Nature. 2021;602:328–35.
Valente EM, Abou-Sleiman PM, Caputo V, Muqit MM, Harvey K, Gispert S, et al. Hereditary early-onset Parkinson’s disease caused by mutations in PINK1. Science. 2004;304:1158–60.
Kamienieva I, Duszyński J, Szczepanowska J. Multitasking guardian of mitochondrial quality: Parkin function and Parkinson’s disease. Transl Neurodegener. 2021;10:5.
Gladkova C, Maslen SL, Skehel JM, Komander D. Mechanism of parkin activation by PINK1. Nature. 2018;559:410–4.
Yamada T, Dawson TM, Yanagawa T, Iijima M, Sesaki H. SQSTM1/p62 promotes mitochondrial ubiquitination independently of PINK1 and PRKN/parkin in mitophagy. Autophagy. 2019;15:2012–8.
Vargas JNS, Hamasaki M, Kawabata T, Youle RJ, Yoshimori T. The mechanisms and roles of selective autophagy in mammals. Nat Rev Mol Cell Biol. 2023;24:167–85.
Lazarou M, Sliter DA, Kane LA, Sarraf SA, Wang C, Burman JL, et al. The ubiquitin kinase PINK1 recruits autophagy receptors to induce mitophagy. Nature. 2015;524:309–14.
Nguyen TN, Padman BS, Lazarou M. Deciphering the Molecular Signals of PINK1/Parkin Mitophagy. Trends Cell Biol. 2016;26:733–44.
Pickrell AM, Youle RJ. The roles of PINK1, parkin, and mitochondrial fidelity in Parkinson’s disease. Neuron. 2015;85:257–73.
Ashrafi G, Schwarz TL. The pathways of mitophagy for quality control and clearance of mitochondria. Cell Death Differ. 2013;20:31–42.
Wang S, Long H, Hou L, Feng B, Ma Z, Wu Y, et al. The mitophagy pathway and its implications in human diseases. Signal Transduct Target Ther. 2023;8:304.
Zhang J, Ney PA. Role of BNIP3 and NIX in cell death, autophagy, and mitophagy. Cell Death Differ. 2009;16:939–46.
Wilhelm LP, Zapata-Muñoz J, Villarejo-Zori B, Pellegrin S, Freire CM, Toye AM, et al. BNIP3L/NIX regulates both mitophagy and pexophagy. EMBO J. 2022;41:e111115.
Chen M, Chen Z, Wang Y, Tan Z, Zhu C, Li Y, et al. Mitophagy receptor FUNDC1 regulates mitochondrial dynamics and mitophagy. Autophagy. 2016;12:689–702.
Kuang Y, Ma K, Zhou C, Ding P, Zhu Y, Chen Q, et al. Structural basis for the phosphorylation of FUNDC1 LIR as a molecular switch of mitophagy. Autophagy. 2016;12:2363–73.
Ward DM, Cloonan SM. Mitochondrial Iron in Human Health and Disease. Annu Rev Physiol. 2019;81:453–82.
Wilkinson KA, Guo C. Iron chelation promotes mitophagy through SENP3-mediated deSUMOylation of FIS1. Autophagy. 2022;18:1743–5.
Zhang T, Liu Q, Gao W, Sehgal SA, Wu H. The multifaceted regulation of mitophagy by endogenous metabolites. Autophagy. 2021;18:1216–39.
Verdin E. NAD+ in aging, metabolism, and neurodegeneration. Science. 2015;350:1208–13.
Wilson N, Kataura T, Korsgen ME, Sun C, Sarkar S, Korolchuk VI. The autophagy-NAD axis in longevity and disease. Trends Cell Biol. 2023;33:788–802.
Kaushik S, Tasset I, Arias E, Pampliega O, Wong E, Martinez-Vicente M, et al. Autophagy and the hallmarks of aging. Ageing Res Rev. 2021;72:101468.
Pradeepkiran JA, Reddy PH. Defective mitophagy in Alzheimer’s disease. Ageing Res Rev. 2020;64:101191.
El Fissi N, Rosenberger FA, Chang K, Wilhalm A, Barton-Owen T, Hansen FM, et al. Preventing excessive autophagy protects from the pathology of mtDNA mutations in Drosophila melanogaster. Nat Commun. 2024;15:10719.
Childs BG, Gluscevic M, Baker DJ, Laberge RM, Marquess D, Dananberg J, et al. Senescent cells: an emerging target for diseases of ageing. Nat Rev Drug Discov. 2017;16:718–35.
Di Micco R, Krizhanovsky V, Baker D, d’Adda di Fagagna F. Cellular senescence in ageing: from mechanisms to therapeutic opportunities. Nat Rev Mol Cell Biol. 2021;22:75–95.
Zhang W, Qu J, Liu GH, Belmonte JCI. The ageing epigenome and its rejuvenation. Nat Rev Mol Cell Biol. 2020;21:137–50.
Wang K, Liu H, Hu Q, Wang L, Liu J, Zheng Z, et al. Epigenetic regulation of aging: implications for interventions of aging and diseases. Signal Transduct Target Ther. 2022;7:374.
Sen P, Shah PP, Nativio R, Berger SL. Epigenetic Mechanisms of Longevity and Aging. Cell. 2016;166:822–39.
Tan A, Prasad R, Lee C, Jho EH. Past, present, and future perspectives of transcription factor EB (TFEB): mechanisms of regulation and association with disease. Cell Death Differ. 2022;29:1433–49.
Abokyi S, Ghartey-Kwansah G, Tse DY-y. TFEB is a central regulator of the aging process and age-related diseases. Ageing Res Rev. 2023;89:101985.
Martini-Stoica H, Xu Y, Ballabio A, Zheng H. The Autophagy-Lysosomal Pathway in Neurodegeneration: A TFEB Perspective. Trends Neurosci. 2016;39:221–34.
Rusmini P, Cortese K, Crippa V, Cristofani R, Cicardi ME, Ferrari V, et al. Trehalose induces autophagy via lysosomal-mediated TFEB activation in models of motoneuron degeneration. Autophagy. 2019;15:631–51.
Greenberg MVC, Bourc’his D. The diverse roles of DNA methylation in mammalian development and disease. Nat Rev Mol Cell Biol. 2019;20:590–607.
Horvath S, Haghani A, Macoretta N, Ablaeva J, Zoller JA, Li CZ, et al. DNA methylation clocks tick in naked mole rats but queens age more slowly than nonbreeders. Nat Aging. 2022;2:46–59.
Unnikrishnan A, Freeman WM, Jackson J, Wren JD, Porter H, Richardson A. The role of DNA methylation in the epigenetics of aging. Pharm Ther. 2019;195:172–85.
Ng MYW, Wai T, Simonsen A. Quality control of the mitochondrion. Dev Cell. 2021;56:881–905.
Sutandy FXR, Gößner I, Tascher G, Münch C. A cytosolic surveillance mechanism activates the mitochondrial UPR. Nature. 2023;618:849–54.
Uoselis L, Lindblom R, Lam WK, Küng CJ, Skulsuppaisarn M, Khuu G, et al. Temporal landscape of mitochondrial proteostasis governed by the UPR(mt). Sci Adv. 2023;9:eadh8228.
Cong W, Wang Y, Yuan C, Xu M, Wang H, Hu Y, et al. Dietary cobalt oxide nanoparticles alleviate aging through activation of mitochondrial UPR in Caenorhabditis elegans. Theranostics. 2023;13:3276–89.
Chen W, Zhao H, Li Y. Mitochondrial dynamics in health and disease: mechanisms and potential targets. Signal Transduct Target Ther. 2023;8:333.
Wang Y, Dai X, Li H, Jiang H, Zhou J, Zhang S, et al. The role of mitochondrial dynamics in disease. MedComm (2020). 2023;4:e462.
Lee YT, Savini M, Chen T, Yang J, Zhao Q, Ding L, et al. Mitochondrial GTP metabolism controls reproductive aging in C. elegans. Dev Cell. 2023;58:2718–31.e2717.
Labbadia J, Morimoto RI. The biology of proteostasis in aging and disease. Annu Rev Biochem. 2015;84:435–64.
Hipp MS, Kasturi P, Hartl FU. The proteostasis network and its decline in ageing. Nat Rev Mol Cell Biol. 2019;20:421–35.
Bukau B, Weissman J, Horwich A. Molecular Chaperones and Protein Quality Control. Cell. 2006;125:443–51.
Di Rienzo M, Romagnoli A, Refolo G, Vescovo T, Ciccosanti F, Zuchegna C, et al. Role of AMBRA1 in mitophagy regulation: emerging evidence in aging-related diseases. Autophagy. 2024;20:2602–15.
Guarente L, Franklin H. Epstein Lecture: Sirtuins, aging, and medicine. N Engl J Med. 2011;364:2235–44.
Xu C, Wang L, Fozouni P, Evjen G, Chandra V, Jiang J, et al. SIRT1 is downregulated by autophagy in senescence and ageing. Nat Cell Biol. 2020;22:1170–9.
Li X, Li C, Zhang W, Wang Y, Qian P, Huang H. Inflammation and aging: signaling pathways and intervention therapies. Signal Transduct Target Ther. 2023;8:239.
Wang B, Han J, Elisseeff JH, Demaria M. The senescence-associated secretory phenotype and its physiological and pathological implications. Nat Rev Mol Cell Biol. 2024;25:958–78.
Gulen MF, Samson N, Keller A, Schwabenland M, Liu C, Glück S, et al. cGAS-STING drives ageing-related inflammation and neurodegeneration. Nature. 2023;620:374–80.
Victorelli S, Salmonowicz H, Chapman J, Martini H, Vizioli MG, Riley JS, et al. Apoptotic stress causes mtDNA release during senescence and drives the SASP. Nature. 2023;622:627–36
McHugh D, Durán I, Gil J. Senescence as a therapeutic target in cancer and age-related diseases. Nat Rev Drug Discov. 2024;24:57–71.
Strowig T, Henao-Mejia J, Elinav E, Flavell R. Inflammasomes in health and disease. Nature. 2012;481:278–86.
Green DR, Galluzzi L, Kroemer G. Mitochondria and the autophagy-inflammation-cell death axis in organismal aging. Science. 2011;333:1109–12.
Broz P, Dixit VM. Inflammasomes: mechanism of assembly, regulation and signalling. Nat Rev Immunol. 2016;16:407–20.
Guo H, Callaway JB, Ting JP. Inflammasomes: mechanism of action, role in disease, and therapeutics. Nat Med. 2015;21:677–87.
Paik S, Kim JK, Silwal P, Sasakawa C, Jo EK. An update on the regulatory mechanisms of NLRP3 inflammasome activation. Cell Mol Immunol. 2021;18:1141–60.
Poor TA, Chandel NS. Mitochondrial molecule controls inflammation. Nature. 2023;615:401–2.
Zhong Z, Liang S, Sanchez-Lopez E, He F, Shalapour S, Lin XJ, et al. New mitochondrial DNA synthesis enables NLRP3 inflammasome activation. Nature. 2018;560:198–203.
Zhao M, Wang Y, Li L, Liu S, Wang C, Yuan Y, et al. Mitochondrial ROS promote mitochondrial dysfunction and inflammation in ischemic acute kidney injury by disrupting TFAM-mediated mtDNA maintenance. Theranostics. 2021;11:1845–63.
Ma Q. Pharmacological Inhibition of the NLRP3 Inflammasome: Structure, Molecular Activation, and Inhibitor-NLRP3 Interaction. Pharm Rev. 2023;75:487–520.
Jiménez-Loygorri JI, Villarejo-Zori B, Viedma-Poyatos Á, Zapata-Muñoz J, Benítez-Fernández R, Frutos-Lisón MD, et al. Mitophagy curtails cytosolic mtDNA-dependent activation of cGAS/STING inflammation during aging. Nat Commun. 2024;15:830.
D’Amico D, Andreux PA, Valdés P, Singh A, Rinsch C, Auwerx J. Impact of the Natural Compound Urolithin A on Health, Disease, and Aging. Trends Mol Med. 2021;27:687–99.
Andreux PA, Blanco-Bose W, Ryu D, Burdet F, Ibberson M, Aebischer P, et al. The mitophagy activator urolithin A is safe and induces a molecular signature of improved mitochondrial and cellular health in humans. Nat Metab. 2019;1:595–603.
Ryu D, Mouchiroud L, Andreux PA, Katsyuba E, Moullan N, Nicolet-dit-Félix AA, et al. Urolithin A induces mitophagy and prolongs lifespan in C. elegans and increases muscle function in rodents. Nat Med. 2016;22:879–88.
Li D, Shao R, Wang N, Zhou N, Du K, Shi J, et al. Sulforaphane Activates a lysosome-dependent transcriptional program to mitigate oxidative stress. Autophagy. 2021;17:872–87.
Bhukel A, Madeo F, Sigrist SJ. Spermidine boosts autophagy to protect from synapse aging. Autophagy. 2017;13:444–5.
Sigrist SJ, Carmona-Gutierrez D, Gupta VK, Bhukel A, Mertel S, Eisenberg T, et al. Spermidine-triggered autophagy ameliorates memory during aging. Autophagy. 2014;10:178–9.
Gupta VK, Scheunemann L, Eisenberg T, Mertel S, Bhukel A, Koemans TS, et al. Restoring polyamines protects from age-induced memory impairment in an autophagy-dependent manner. Nat Neurosci. 2013;16:1453–60.
Freitag K, Sterczyk N, Wendlinger S, Obermayer B, Schulz J, Farztdinov V, et al. Spermidine reduces neuroinflammation and soluble amyloid beta in an Alzheimer’s disease mouse model. J Neuroinflammation. 2022;19:172.
Barzilai N, Crandall JP, Kritchevsky SB, Espeland MA. Metformin as a Tool to Target Aging. Cell Metab. 2016;23:1060–5.
Kulkarni AS, Gubbi S, Barzilai N. Benefits of Metformin in Attenuating the Hallmarks of Aging. Cell Metab. 2020;32:15–30.
Mills KF, Yoshida S, Stein LR, Grozio A, Kubota S, Sasaki Y, et al. Long-Term Administration of Nicotinamide Mononucleotide Mitigates Age-Associated Physiological Decline in Mice. Cell Metab. 2016;24:795–806.
Prolla TA, Denu JM. NAD+ deficiency in age-related mitochondrial dysfunction. Cell Metab. 2014;19:178–80.
Imai S. A possibility of nutriceuticals as an anti-aging intervention: activation of sirtuins by promoting mammalian NAD biosynthesis. Pharm Res. 2010;62:42–7.
Nadeeshani H, Li J, Ying T, Zhang B, Lu J. Nicotinamide mononucleotide (NMN) as an anti-aging health product - Promises and safety concerns. J Adv Res. 2022;37:267–78.
Yoshino M, Yoshino J, Kayser BD, Patti GJ, Franczyk MP, Mills KF, et al. Nicotinamide mononucleotide increases muscle insulin sensitivity in prediabetic women. Science. 2021;372:1224–9.
Fang T, Yang J, Liu L, Xiao H, Wei X. Nicotinamide mononucleotide ameliorates senescence in alveolar epithelial cells. MedComm. 2021;2:279–87.
Fang EF, Hou Y, Lautrup S, Jensen MB, Yang B, SenGupta T, et al. NAD(+) augmentation restores mitophagy and limits accelerated aging in Werner syndrome. Nat Commun. 2019;10:5284.
Zhang H, Ryu D, Wu Y, Gariani K, Wang X, Luan P, et al. NAD⁺ repletion improves mitochondrial and stem cell function and enhances life span in mice. Science. 2016;352:1436–43.
Palozzi JM, Hurd TR. The role of programmed mitophagy in germline mitochondrial DNA quality control. Autophagy. 2023;19:2817–8.
Yamaura K, Nelson AL, Nishimura H, Rutledge JC, Ravuri SK, Bahney C, et al. Therapeutic potential of senolytic agent quercetin in osteoarthritis: A systematic review and meta-analysis of preclinical studies. Ageing Res Rev. 2023;90:101989.
Han X, Xu T, Fang Q, Zhang H, Yue L, Hu G, et al. Quercetin hinders microglial activation to alleviate neurotoxicity via the interplay between NLRP3 inflammasome and mitophagy. Redox Biol. 2021;44:102010.
Geng L, Liu Z, Zhang W, Li W, Wu Z, Wang W, et al. Chemical screen identifies a geroprotective role of quercetin in premature aging. Protein Cell. 2019;10:417–35.
Xu M, Pirtskhalava T, Farr JN, Weigand BM, Palmer AK, Weivoda MM, et al. Senolytics improve physical function and increase lifespan in old age. Nat Med. 2018;24:1246–56.
Li M, Yu Y, Xue K, Li J, Son G, Wang J, et al. Genistein mitigates senescence of bone marrow mesenchymal stem cells via ERRα-mediated mitochondrial biogenesis and mitophagy in ovariectomized rats. Redox Biol. 2023;61:102649.
Wang TH, Tseng WC, Leu YL, Chen CY, Lee WC, Chi YC, et al. The flavonoid corylin exhibits lifespan extension properties in mouse. Nat Commun. 2022;13:1238.
Zimmermann A, Kainz K, Hofer SJ, Bauer MA, Schroeder S, Dengjel J, et al. 4,4’Dimethoxychalcone: a natural flavonoid that promotes health through autophagy-dependent and -independent effects. Autophagy. 2019;15:1662–4.
Carmona-Gutierrez D, Zimmermann A, Kainz K, Pietrocola F, Chen G, Maglioni S, et al. The flavonoid 4,4’-dimethoxychalcone promotes autophagy-dependent longevity across species. Nat Commun. 2019;10:651.
Liu Y, Ye J, Fan Z, Wu X, Zhang Y, Yang R, et al. Ginkgetin alleviates inflammation and senescence by targeting STING. Adv Sci. 2025;12:e2407222.
Singh A, D’Amico D, Andreux PA, Fouassier AM, Blanco-Bose W, Evans M, et al. Urolithin A improves muscle strength, exercise performance, and biomarkers of mitochondrial health in a randomized trial in middle-aged adults. Cell Rep Med. 2022;3:100633.
Samaras K, Makkar S, Crawford JD, Kochan NA, Wen W, Draper B, et al. Metformin Use Is Associated With Slowed Cognitive Decline and Reduced Incident Dementia in Older Adults With Type 2 Diabetes: The Sydney Memory and Ageing Study. Diab Care. 2020;43:2691–701.
Moore EM, Mander AG, Ames D, Kotowicz MA, Carne RP, Brodaty H, et al. Increased risk of cognitive impairment in patients with diabetes is associated with metformin. Diab Care. 2013;36:2981–7.
Brakedal B, Dölle C, Riemer F, Ma Y, Nido GS, Skeie GO, et al. The NADPARK study: A randomized phase I trial of nicotinamide riboside supplementation in Parkinson’s disease. Cell Metab. 2022;34:396–407.e396.
Mannick JB, Del Giudice G, Lattanzi M, Valiante NM, Praestgaard J, Huang B, et al. mTOR inhibition improves immune function in the elderly. Sci Transl Med. 2014;6:268ra179.
Mannick JB, Teo G, Bernardo P, Quinn D, Russell K, Klickstein L, et al. Targeting the biology of ageing with mTOR inhibitors to improve immune function in older adults: phase 2b and phase 3 randomised trials. Lancet Healthy Longev. 2021;2:e250–e262.
Hickson LJ, Langhi Prata LGP, Bobart SA, Evans TK, Giorgadze N, Hashmi SK, et al. Senolytics decrease senescent cells in humans: Preliminary report from a clinical trial of Dasatinib plus Quercetin in individuals with diabetic kidney disease. EBioMedicine. 2019;47:446–56.
Justice JN, Nambiar AM, Tchkonia T, LeBrasseur NK, Pascual R, Hashmi SK, et al. Senolytics in idiopathic pulmonary fibrosis: Results from a first-in-human, open-label, pilot study. EBioMedicine. 2019;40:554–63.
China Association of Chinese Medicine, Beijing, China. Top 10 research achievements of traditional Chinese medicine in 2023. Sci Tradit Chin Med. 2024;2:255-920.
Wan J, Zhang Z, Wu C, Tian S, Zang Y, Jin G, et al. Astragaloside IV derivative HHQ16 ameliorates infarction-induced hypertrophy and heart failure through degradation of lncRNA4012/9456. Signal Transduct Target Ther. 2023;8:414.
Fan W, Fan L, Wang Z, Mei Y, Liu L, Li L, et al. Rare ginsenosides: A unique perspective of ginseng research. J Adv Res. 2024;66:303–28.
Lee DY, Noren Hooten N, O'Connell JF, Lee BY, Kim Y. The Role of Ginseng and Its Bioactive Compounds in Aging: Cells and Animal Studies. Annu Rev Food Sci Technol. 2025;16:333–54.
Zhang X, He X, You L, Chen J, Ma J, Wang G, et al. Remarkable research achievements in traditional Chinese medicine during 2012-22. Sci Tradit Chin Med. 2024;2:5–13.
Hsu PD, Lander ES, Zhang F. Development and applications of CRISPR-Cas9 for genome engineering. Cell. 2014;157:1262–78.
Charpentier E, Doudna JA. Biotechnology: Rewriting a genome. Nature. 2013;495:50–1.
Han R, Wang Q, Xiong X, Chen X, Tu Z, Li B, et al. Deficiency of parkin causes neurodegeneration and accumulation of pathological α-synuclein in monkey models. J Clin Investig. 2024;134:e179633.
Li J, Wang L, Hua X, Tang H, Chen R, Yang T, et al. CRISPR/Cas9-Mediated miR-29b Editing as a Treatment of Different Types of Muscle Atrophy in Mice. Mol Ther. 2020;28:1359–72.
Hashimoto M, Bacman SR, Peralta S, Falk MJ, Chomyn A, Chan DC, et al. MitoTALEN: A General Approach to Reduce Mutant mtDNA Loads and Restore Oxidative Phosphorylation Function in Mitochondrial Diseases. Mol Ther. 2015;23:1592–9.
Bacman SR, Kauppila JHK, Pereira CV, Nissanka N, Miranda M, Pinto M, et al. Author Correction: MitoTALEN reduces mutant mtDNA load and restores tRNA(Ala) levels in a mouse model of heteroplasmic mtDNA mutation. Nat Med. 2018;24:1940.
Wu Q, Gao ZJ, Yu X, Wang P. Dietary regulation in health and disease. Signal Transduct Target Ther. 2022;7:252.
Johnson ML, Robinson MM, Nair KS. Skeletal muscle aging and the mitochondrion. Trends Endocrinol Metab. 2013;24:247–56.
Mitchell SJ, Madrigal-Matute J, Scheibye-Knudsen M, Fang E, Aon M, González-Reyes JA, et al. Effects of Sex, Strain, and Energy Intake on Hallmarks of Aging in Mice. Cell Metab. 2016;23:1093–112.
Wei SJ, Schell JR, Chocron ES, Varmazyad M, Xu G, Chen WH, et al. Ketogenic diet induces p53-dependent cellular senescence in multiple organs. Sci Adv. 2024;10:eado1463.
Jia D, Tian Z, Wang R. Exercise mitigates age-related metabolic diseases by improving mitochondrial dysfunction. Ageing Res Rev. 2023;91:101935.
Ma L, Li K, Wei W, Zhou J, Li Z, Zhang T, et al. Exercise protects aged mice against coronary endothelial senescence via FUNDC1-dependent mitophagy. Redox Biol. 2023;62:102693.
Neto IVdS, Pinto AP, Muñoz VR, de Cássia Marqueti R, Pauli JR, Ropelle ER, et al. Pleiotropic and multi-systemic actions of physical exercise on PGC-1α signaling during the aging process. Ageing Res Rev. 2023;87:101935.
Longo VD, Kennedy BK. Sirtuins in Aging and Age-Related Disease. Cell. 2006;126:257–68.
Wang Y, Yang J, Hong T, Chen X, Cui L. SIRT2: Controversy and multiple roles in disease and physiology. Ageing Res Rev. 2019;55:100961.
Ghosh S, Zhou Z. SIRTain regulators of premature senescence and accelerated aging. Protein Cell. 2015;6:322–33.
Xian Y, Liu B, Shen T, Yang L, Peng R, Shen H, et al. Enhanced SIRT3 expression restores mitochondrial quality control mechanism to reverse osteogenic impairment in type 2 diabetes mellitus. Bone Res. 2025;13:30.
Schwer B, Verdin E. Conserved metabolic regulatory functions of sirtuins. Cell Metab. 2008;7:104–12.
van de Ven RAH, Santos D, Haigis MC. Mitochondrial Sirtuins and Molecular Mechanisms of Aging. Trends Mol Med. 2017;23:320–31.
Raza U, Tang X, Liu Z, Liu B. SIRT7: the seventh key to unlocking the mystery of aging. Physiol Rev. 2024;104:253–80.
Zhang W, Zhang L, Fu S, Yan R, Zhang X, Song J, et al. Roles of NLRC4 inflammasome in neurological disorders: Mechanisms, implications, and therapeutic potential. Pharm Ther. 2025;267:108803.
Bauernfried S & Hornung V. Human NLRP1: From the shadows to center stage. J Exp Med. 2022;219.
Wang B, Bhattacharya M, Roy S, Tian Y, Yin Q. Immunobiology and structural biology of AIM2 inflammasome. Mol Asp Med. 2020;76:100869.
Barnett KC, Li S, Liang K, Ting JP. A 360° view of the inflammasome: Mechanisms of activation, cell death, and diseases. Cell. 2023;186:2288–312.
Acknowledgements
This work was supported by the Beijing Nova Program (20230484300).
Author information
Authors and Affiliations
Contributions
WJS and LFL were the major contributors in writing the manuscript; YLL, HL, and HJY helped revise and polish the wording of the article; RYT provided some valuable suggestions for revising the article. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Shan, W., Liu, Y., Tang, R. et al. Targeting mitochondrial autophagy for anti-aging. Cell Death Discov. 12, 78 (2026). https://doi.org/10.1038/s41420-025-02913-y
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41420-025-02913-y