Abstract
The pathological hallmark of schistosomiasis is egg-induced granulomatous inflammation and the subsequent fibrosis in the liver; and mesenchymal stem cells have been explored for the treatment of schistosomiasis fibrosis. However, the underlying mechanisms are largely unknown. We demonstrated here that IFN-γ/ LPS-primed MSCs alleviate schistosomiasis fibrosis through promoting conversion of Ly6ChiCX3CR1lo to pro-resolving Ly6CloCX3CR1hi. Importantly, primed MSCs promoted macrophage efferocytosis in the infected mice, and in vitro experiments confirmed the direct role of primed MSC on enhancement of macrophage efferocytosis, as well as its conversion to pro-resolving type. Mechanistically, primed MSC promoted β2 integrins(Itgb2) expression within macrophages; and Itgb2 blockade not only inhibited GTPase Rac1 activity, a key regulator of actin filament organization during efferocytosis, but also abolished the enhancement of primed MSCs on macrophage efferocytosis, suggesting that primed MSCs enhance efferocytosis via Itgb2-Rac1 axis. Moreover, either Itgb2 blockade or Rac1 inhibition within macrophages reversed the regulation of primed MSCs on macrophage subset switching, suggesting that primed MSCs promote macrophage subset conversion dependent on efferocytosis pathway. Taken together, this study demonstrates that primed MSCs attenuate schistosomiasis liver fibrosis by enhancing macrophage subset switch and efferocytosis via Itgb2-Rac1 axis, which offers novel insights into the therapeutic targets of MSC-based anti-fibrotic therapy.
Similar content being viewed by others
Introduction
Schistosomiasis is one of the world’s major public health problems in terms of morbidity and mortality [1]. Although praziquantel (PZQ) has been widely used to kill or damage the adult worms, the development of effective therapies for the regression of egg-induced granuloma and fibrosis remains a challenge [2]. Considering the similarities of immunopathological characteristics between human and mice schistosomiasis, murine schistosomiasis model has been extensively used for various aspects of human schistosomiasis [3]. With deposition of eggs in the liver 4-6 weeks after infection, a local Th2-driven granulomas inflammation is induced, a collection of inflammatory cells including eosinophils, neutrophils, lymphocytes and macrophages [1, 4,5,6], among which, upon infection-associated removal of KCs, recruited bone marrow monocyte-derived macrophages (MoMϕs) play a key role through connecting with inflammatory cells and hepatic stellate cells (HSCs), and as a result, with excessive accumulation of extracellular matrix (ECM), fibrosis develops nearly at 7-8 weeks postinfection [7, 8].
Based on cell surface glycoprotein Ly-6C expression, at least two subsets of MoMϕ are defined in mice, Ly6Chi and Ly6Clo subsets. It is generally considered that Ly6Chi MoMϕs have a proinflammatory phenotype (similar to classical M1 phenotype), which can aggravate liver injury and fibrosis by releasing proinflammatory factors and fibrogenic cytokines, such as TNF-α, IL-1, CCL2, CCL5, TGF-β1, PDGF, while Ly6Clo macrophages promote inflammation or fibrosis resolution by secreting anti-inflammatory factors or matrix metalloproteinases (MMPs) and inducing HSC apoptosis [9,10,11].
In murine schistosomasis model, bone marrow derived Ly6Chi monocytes can be robustly recruited and responsible for Th2 associated host-protective macrophage accumulation [12, 13]; Additionally, it is reported that inducing pro-resolving phenotype (Ly6Clo MoMϕs) through overexpression of miR-130a-3p in mice infected with S. japonicum promoted regression of fibrosis through decreasing tissue inhibitor of metalloproteinases (TIMP) 1 and increasing matrix metalloproteinase (MMP) 2 [14]. Thus, enhancement of phenotypic switching from Ly6Chi to Ly6Clo may confer anti-fibrotic benefits for liver schistosomiasis.
Recently, the accumulating evidence shows that MSCs has become a promising cyto-therapy for treating a variety of liver fibrosis [15,16,17,18,19,20,21,22,23,24,25]. There have been relatively consistent results showing that MSCs may alleviate hepatic fibrosis through promoting phenotypic conversion from Ly6Chi toward pro-resolving Ly6Clo macrophages in CCl4 [15, 23, 24] and MCD diet fibrosis model [25]; however, research on schistosomiasis fibrosis treatment with MSCs are still limited. A few reports have indicated that MSCs exhibited preliminary anti-fibrotic efficacy regarding the histopathological evaluation, yet it remains to be verified whether MSCs exert anti-fibrotic effects by regulating Ly6Chi /Ly6Clo phenotype conversion [19, 26,27,28,29]. More importantly, the significant pathological features dominated by the Th2 or M2 response in the schistosomiasis fibrosis stage renders this issue of particular interest [30, 31].
Our previous research have shown that toll like receptor (TLR)4 agonist and IFN-γprimed MSCs alleviated hepatic fibrosis through modulating Th1/Th2 responses [32], here we further investigate whether and how primed MSCs play anti-fibrotic role through regulation of Ly6Chi /Ly6Clo phenotype during chronic schistosomiasis, which will provide new evidence for MSC therapy of chronic inflammatory fibrosis.
Results
IFN-γ + LPS-primed MSCs alleviated liver pathology
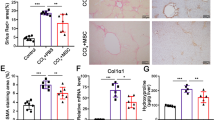
In our previous study, we showed that TLR4 agonist +IFN-γ primed MSCs (their identification results showed in Fig. 1s) alleviated liver pathology [32]. In this work, we confirmed the therapeutic potential of IFN-γ + LPS-primed MSCs in Schistosoma infection model again; at the end of the 2-week observation period after MSCs therapy, the primed MSCs group exhibited a liver with a grossly more reddish appearance and softer texture (Fig. 1A) and showed higher body weight as well, compared with none-treated control or unprimed MSCs group, respectively (Fig. 1B); and more significantly, the primed MSCs group showed alleviation in liver fibrosis at 1 week posttreatment, manifested by lower percentage of egg granulomas area and fibrosis area, compared with unprimed MSCs group, and even more clear improvement in liver fibrosis at 2 weeks posttreatment, compared with both non-treated control and unprimed MSCs group (Fig. 1C, D). Accordingly, collagen type III (COL-3) levels [33] in primed MSCs group significantly decreased both at 1 week and 2 weeks after treatment, compared with none-treated control or unprimed MSCs group, respectively, and similarly decreased for α-SMA (a key activation marker for HSC)at 1 week after treatment (Fig. 1E). Thus, only primed MSCs but not unprimed MSCs showed the therapeutic potential for schistosomiasis fibrosis.
Mice were infected with 16 ± 2 cercariae and treated with praziquantel (PZQ) at 6 weeks post-infection, followed by treatment with MSCs and then sacrificed at 1, 2 weeks after MSCs treatment, respectively. At 2 weeks after treatment, gross liver appearance was examined (A), and weekly body weight measurements were taken and compared (B). Histological change was evaluated. Representative granulomatous was showed by HE staining and by Masson staining (C). The percentage of egg granulomas area and fibrosis area were compared among different groups (D). Fibrosis-related genes Col3a1 and Acta2 were assessed by semi-quantitative PCR, normalized to GAPDH (E). Data are presented as the means ± SEM (n = 4–5). *P < 0.05, **P < 0.01, ****P < 0.0001 and n.s. not significant. Comparing each time among PBS, none primed MSCs and primed MSCs group.
Since the fibrosis development have been generally considered to be related to the phenotypes of macrophages; and two phenotypes of bone marrow/monocyte-derived macrophages (MoMϕs), either pro-inflammatory Ly6Chigh or pro-restorative Ly6Clow MoMϕs have been described to play key role in the granulomatous inflammation of schistosomiasis [11, 34], we next focused on the alteration of phenotypic switching of Ly6Chigh/Ly6Clow over time following treatment with MSCs.
IFN-γ + LPS-primed MSCs led to lower Ly6ChiCX3CR1lo and higher Ly6CloCX3CR1hi levels in mice infected with Schistosoma japonicum
Two kinds of bone marrow-derived MoMϕs, named as Ly6ChiCX3CR1lo and Ly6CloCX3CR1hi phenotypes in the liver were detected by FACS at 1 week and 2 weeks after MSCs treatment, respectively. First, non-MSCs control showed higher Ly6ChiCX3CR1lo (compared with normal group) at the first observation timepoint, followed by comparable Ly6ChiCX3CR1lo and significantly lower Ly6CloCX3CR1hi levels (compared with normal group) at the second observation timepoint (Fig. 2A, B). The primed MSCs group exhibited similar Ly6ChiCX3CR1lo levels compared with non-MSCs control (despite a downward trend) but lower Ly6ChiCX3CR1lo levels compared with unprimed MSCs group, and higher Ly6CloCX3CR1hi levels (compared with both non-MSCs control and unprimed MSCs group) one week after treatment, with higher Ly6CloCX3CR1hi persisting into week 2(compared with non-MSCs control). In contrast, the unprimed MSCs group showed persistently higher Ly6ChiCX3CR1lo levels (compared with untreated control) both in weeks 1 and 2 after treatment, in spite of a higher Ly6CloCX3CR1hi levels (compared with untreated control) in week 2 (Fig. 2A, B). Thus, primed MSCs exhibited therapeutic effects, accompanied by a downward trend in Ly6ChiCX3CR1lo and a persistent increase in Ly6CloCX3CR1hi levels during observation period, whereas non-primed MSCs showed no significant therapeutic effect, accompanied by a persistent increase in Ly6ChiCX3CR1lo, in spite of a higher Ly6CloCX3CR1hi levels at later stage of treatment.
Representative percentages of Ly6ChiCX3CR1lo subset and Ly6CloCX3CR1hi subset as a proportion of MoMϕs (gated on CD11b+ F4/80int), analyzed by flow cytometry (A) and quantification of each subset as a proportion of MoMϕs (B) demonstrating the dynamics alteration of hepatic Ly6Chi and Ly6Clo in week1, 2 after MSCs treatment in infected mice, respectively. Data are presented as the means ± SEM, n = 4–5. *P < 0.05, **P < 0.01, ****P < 0.0001 and n.s. not significant. Comparing each time among PBS, none primed MSCs and primed MSCs group.
Since Ly6Chi MoMϕ or Ly6Clo MoMϕs have been described as pro-inflammatory or pro-resolving phenotype [6, 8], to further disclose their functional characteristics, representative pro-inflammatory or anti-inflammatory factors of macrophages were further detected in infected mice.
IFN-γ + LPS -primed MSCs induced lower levels of inflammatory factors and a moderate pro-resolving profile in the liver of infected mice
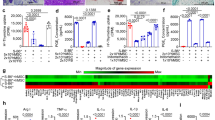
The key pro-inflammatory factors (TNF-α, IL-1β and iNOS) and inflammatory chemokines(CCL2, CX3CL1,CXCL1) were detected using the semi-quantitative PCR method, and so were anti-inflammatory factors (IL-10 and Arg-1). The primed MSCs group showed the lowest levels of TNF-α, IL-1βand iNOS among all the infected groups, as well as the lowest levels of chemokines (CCL2, CX3CL1,CXCL1) in week 1 after MSCs treatment, and continuously maintained lower levels of iNOS and CX3CL1 (compared with non-MSCs control) in week 2. By contrast, non-primed MSCs group showed either higher levels of IL-1βand iNOS or comparable levels of CCL2, CX3CL1 and CXCL1 to those in non-MSCs control in week 1 after MSCs treatment, followed by persistently higher levels of IL-1βand CCL2 (compared with non-MSC control)in week 2 (Fig. 3).
Fifty to 100 mg of liver tissue was removed from infected mice at certain timepoints, and the expression of Nos2, Il1b, Tnf, Ccl2, Cx3cl1, Cxcl1, Arg1 and Il10 mRNA were assessed by semi-quantitative PCR analysis, normalized to GAPDH. Data are presented as the means ± SEM, n = 4-5. ***P < 0.001**P < 0.01, *P < 0.05.
As for anti-inflammatory or pro-resolving factors, consistent with most reports, control group exhibited pathological features dominated by a TH2 or M2 response, with persistently higher Arg-1 levels at both observation timepoints; primed MSCs group showed comparable Arg-1 levels to those in non-MSCs control in week 1 after MSCs treatment, followed by lower Arg-1 levels in week 2; by contrast, non-primed MSCs group showed persistently lower Arg-1 both in week1,2. Additionally, primed MSCs group moderately increasing levels of IL-10 among all the infected groups, whereas, non-primed MSCs group showed the highest IL-10 levels among all groups in week 1 (Fig. 3).
Collectively, primed MSC treatment led to reductions in pro-inflammatory cytokines, chemokines, and iNOS, while maintaining similarly high levels of Arg-1 as the control group at 1 week posttreatment, followed by lower levels of iNOS and CX3CL1 at 2 weeks; in contrast, treatment with non-primed MSCs increased pro-inflammatory cytokines and iNOS while decreasing Arg-1, followed by persistently higher levels of IL-1βand CCL2 at 2 weeks. This finding was just in accord with the results of macrophage subset analysis, in which primed MSCs induced a persistent increase in Ly6CloCX3CR1hi levels without increase of Ly6ChiCX3CR1lo, whereas non-primed MSCs led to a persistent increase in Ly6ChiCX3CR1lo, in spite of a higher Ly6CloCX3CR1hi levels at later stage of treatment.
To further confirm whether primed MSCs induced increase of Ly6CloCX3CR1hi levels through promoting Ly6Chi/Ly6Clo conversion, we blocked recruitment of Ly6Chi monocytes/macrophages using CCR2 antagonist before MSCs treatment, then identified Ly6Clo MoMϕs percentage at 2 weeks posttreatment.
IFN-γ + LPS -primed MSCs ameliorate schistosomiasis fibrosis through promoting conversion of Ly6Chi/ Ly6Clo MoMϕs
We used RS102895 to block CCR2-dependent recruitment of Ly6Chi monocytes/macrophages at the early stage of treatment with primed MSCs and further investigated subtypes switching of MoMϕs at 2 weeks posttreatment. As expected, the primed MSCs group exhibited higher levels of Ly6Clo MoMϕs and the same level of Ly6Chi MoMϕs, compared to the control group, while blocking CCR2 led to decrease of both Ly6Chi MoMϕs and Ly6Clo MoMϕs at 2 weeks posttreatment in infected mice (Fig. 4C, D), with a corresponding deteriorated liver pathology (Fig. 4A, B). This blocking assay confirmed that IFN-γ + LPS-primed MSCs enhanced Ly6Clo subtypes dependent on recruitment of Ly6Chi monocytes/macrophages at earlier stage post-treatment, and in other words, primed MSCs promoted conversion of Ly6Chi/ Ly6Clo MoMϕs.
Histological change was evaluated at 2 weeks after treatment in infected mice, which were injected with PBS, primed MSCs or activated MSC + RS102895, respectively. Representative granulomatous was shown by HE staining and by Masson staining (A). The percentage of fibrosis area were measured by computer-assisted morphometric analysis (B). Representative percentages of Ly6ChiCX3CR1lo subset and Ly6CloCX3CR1hi subset as a proportion of MoMϕs, analyzed by flow cytometry (C) and quantification of each subset as a proportion of MoMϕs (D). Data are presented as mean ± SEM; n = 3–4 mice for each group. (*P < 0.05, **P < 0.01 and n.s. not significant).
Next, we ask how MSCs promote switching of Ly6Chi/Ly6Clo subtypes in schistosomiasis model. It has been reported that cytokines in the microenvironment (such as IL-4 and IL-10) can promote macrophage phenotype switching [15, 35,36,37], but this is clearly not the case in our experiment, where the primed MSCs group showed positive therapeutic effects with moderate increase of IL-10, while non-primed MSCs group showed no anti-fibrotic efficacy despite inducing the highest IL-10 levels. Recently, more and more studies have noted that the synergy between macrophages and neutrophils affects post-liver injury repair; and the phagocytosis of dying neutrophils by macrophages, known as efferocytosis, served not only as clearance of the apoptotic cells [38, 39] but also promoted a pro-resolving phenotype (Ly6Clow MoMϕs) in liver injury model [40,41,42]. Because of the activation and recruitment of macrophages and neutrophils within the schistosomiasis granulomas, leading to amplified inflammatory response, we suspect there is a possibility that primed MSCs promote macrophage subtype switching through efferocytosis of apoptotic neutrophils. Therefore, the efferocytosis-mediated mechanism among different groups was investigated.
IFN-γ + LPS-primed MSCs promoted the phagocytosis of neutrophils by macrophages in infected mice
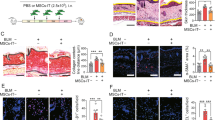
Since TAM proteins Mertk is one of the most universally expressed receptors in the phagocytic macrophages to mediate apoptotic neutrophils recognition [43], double staining of Mertk and neutrophils marker Ly6G are used to show engulfment of apoptotic neutrophils by macrophages. As our expectation, primed MSCs group showed significant higher engulfment ratio of neutrophils both at 1 week and 2 weeks posttreatment (compared with control group or non-primed MSCs group, respectively), which is exactly consistent with the higher Ly6Clo macrophage in primed MSCs group (Fig. 5A–C). Thus, primed MSCs promote macrophage to phagocytose neutrophils in vivo, suggesting a correlation between macrophage subtype switching and efferocytosis. To further investigate the direct role of primed MSCs on efferocytosis and macrophage subtype conversion, in vitro co-culture of macrophages and apoptotic neutrophils was performed.
Mice were infected with 16 ± 2 cercariae and treated with praziquantel (PZQ) at 6 weeks post-infection, followed by treatment with MSCs. Efferocytosis was examined by dual immunofluorescence for MerTK (red) and Ly6G (green) in liver sections. Representative images of macrophage phagocytosis at 1 week (A), 2 weeks post-treatment (B), respectively (Arrows indicate phagocytosing macrophages with neutrophils inside). Scale bars, 100 µm and 400 µm, respectively. The number of efferocytosis cells per field (C). n = 4-5 per group. Data are presented as mean ± SEM (*P < 0.05, ***P < 0.001, ****P <0.0001).
IFN-γ + LPS-primed MSCs directly promoted macrophage efferocytosis, accompanied by decreased inflammatory cytokines and increased pro-resolving factors in vitro
To investigate the direct role of primed MSCs on engulfment of apoptotic neutrophils, we first incubated bone marrow derived macrophage with non-primed MSCs or primed MSCs, respectively, and then added Dil-labeled apoptotic neutrophils (induced by staurosporine). As expected, primed MSCs significantly increased macrophage phagocytic efficiency (compared with control group or non-activated MSC group, respectively) (Fig. 6A1, A2). Meanwhile, primed MSCs led to a decrease in pro-inflammatory cytokine mRNA Il1b, Tnf and Cx3cl1, and an increase in pro-resolving factors mRNA including Cx3cr1 (a key marker of pro-resolving macrophage), Il10 and Mmp2 in macrophages (Fig. 6B). In addition, protein detection in the co-culture system showed consistent results, lower TNF and higher IL10 level in the activated MSC group, compared with control group. However, non-activated MSC group showed comparable TNF levels to those of the control group, but higher IL-10 levels (Fig. 6C). It has been reported that the Ly6Chi/Ly6Clo subset switch not only blocked the source of inflammatory factors, but also triggered pro-resolving factors IL-10 and MMPs production for inflammation or fibrosis resolution [15, 16]. Taken together, our results showed that IFN-γ + LPS-activated MSC directly promoted macrophage efferocytosis, as well as its subtypes switching. Next, the mechanisms underpinning efferocytosis were further investigated.
Prior to the start of efferocytosis, MoMϕs from infected mice were first incubated with MSCs or primed MSCs, respectively, and then added Dil-labeled apoptotic neutrophils (induced by staurosporine). Macrophages and culture supernatant were collected after co-incubation for 1 hour or 12 hours, respectively, and the efferocytosis of macrophages was detected by immunofluorescence for MerTK and Dil-labeled apoptotic neutrophils and observed under a fluorescence microscope. Representative images of efferocytosis cells (A1), Scale bars, 100 µm, and the percentage of macrophages containing at least one ingested apoptotic neutrophil or multiple apoptotic neutrophils, presented as rate of efferocytosis or rate of multiple efferocytosis, respectively (A2); the mRNA levels of Il1b, Tnf, Cx3cl1, Cx3cr1, Il10 and Mmp2 were detected by qPCR (B); the protein levels of IL10 and TNF-ɑ in the culture supernatant were detected by ELISA(C). n = 3; Data are presented as mean ± SEM (*P < 0.05, **P < 0.01 and n.s. not significant).
IFN-γ + LPS-primed MSCs promoted macrophage efferocytosis mediated by Itgb2 -Rac1 activated pathways
For triggering efferocytosis process, macrophages first need to find and recognize the apoptotic cells through interactions between their receptors and ligands exposed on apoptotic cells (such as phosphatidylserine, PS). Therefore, we first investigated the effects of MSCs on engulfment receptors that recognize PS [39, 44]. Our results showed that primed MSCs specially promoted Itgb2 expression, while non-primed MSCs reduced those levels (Fig. 7A); regarding other receptors, including TAM family Mertk, Axl and Gas6, BAI1, Timd4 and integrin Itgb5, there were no significant differences between control group and primed MSCs group (Fig. 2s).
MoMϕs from infected mice were first incubated with MSCs or primed MSCs, respectively, and then added apoptotic neutrophils (or Dil-labeled apoptotic neutrophils). Macrophages were collected at different efferocytosis times, and the mRNA levels of Itgb2 and Rac1were detected by qPCR (A) after 12 h phagocytosis; prior to the start of phagocytosis, macrophages were incubated with CD18 neutralizing antibody(2.5 μg/ml) for 3 h to block Itgb2 signal pathway, and then the mRNA levels of Rac-1 were detected by qPCR after 12 h phagocytosis and activity of Rac1 in macrophages was determined by G-LISA after 1 h of phagocytosis (B); after blocking Itgb2 signal pathway, the efferocytosis efficiency of macrophages was detected by immunofluorescence for MerTK (green) and Dil-labeled apoptotic neutrophils (red), Representative images of efferocytosis cells (C1), Scale bars, 100 µm, and the percentage of macrophages engulfing apoptotic neutrophils (C2). n = 3; Data are presented as mean ± SEM (*P < 0.05, **P < 0.01 and n.s. not significant).
Following the ligand-receptor interaction, cytoskeletal rearrangement of macrophages can be triggered, a key step for the subsequent phagocytic activities, in which GTPase Rac1 plays an important role in the organization of actin filament networks and in membrane ruffling [39, 44]. Indeed, our results demonstrate that both mRNA levels of Rac1 and Rac1 activity of macrophages were significantly increased in the primed MSCs group, while decreased in the non-primed MSCs group (Fig. 7B); treatment of the primed MSCs group with Itgb2 blocking antibody significantly reduced both mRNA levels of Rac1 and Rac1 activity of macrophages (Fig. 7B). Furthermore, consistent with reduced Rac1 activity of macrophage in the blocking group, a significant decrease in the macrophage efferocytosis efficiency was observed in the blocking group as well (Fig. 7C1, C2).
Collectively, our results indicated that primed MSCs enhanced macrophage efferocytosis via Itgb2-Rac1 pathway, and combined with related reports that efferocytosis process can trigger macrophage switching from inflammatory subsets to anti-inflammatory or pre-resolving subsets [15, 16, 41], blocking experiment (blockade of Itgb2-Rac1 pathway) was performed to investigate whether primed MSCs promoted macrophage phenotypic switching via efferocytosis pathway.
Blocking key pathways of efferocytosis reversed the regulation of IFN-γ + LPS -primed MSCs on macrophage subset switching
Either CD18 blockade or inhibition Rac1 activation led to increased mRNA levels of Il1band Tnf, and decreased both Cx3cr1(key marker of reparative macrophages) and Mmp2/9 within macrophages. (Fig. 8). Thus, Itgb2-Rac1 pathway mediate effects of primed MSCs on macrophage function skewing. However, the regulation of IL-10 and TGF-βpresents a different scenario. Primed MSCs led to a decrease of Tgfb levels within macrophages compared with control group; CD18 blockade failed to increase Tgfb expression (in spite of an increasing trend), while Rac1 inhibitor NSC23766 could (Fig. 8). Most surprisingly, the neutralizing CD18 antibody failed to inhibit the effect of primed MSCs in promoting anti-inflammatory Il10 expression, while Rac1 inhibitor NSC23766 could (Fig. 8). Collectively, our data demonstrated that blocking key pathway Rac1 of efferocytosis reversed the regulation of IFN-γ + LPS -primed MSCs on macrophage subset switching, while Itgb2 blockade abolished most of the pro-reparative functional characteristics of macrophages, except for regulation of IL-10 expression by primed MSCs. The enhancement of IL-10 expression by primed MSCs may involve other pathways-triggered efferocytosis beyond Itgb2 activation.
MoMϕs from infected mice were first incubated with primed MSCs or without MSCs for 24 h (for blocking test, macrophages were still exposed to neutralizing antibody against CD18 or Rac1 inhibitor NSC23766 3 h prior to the start of efferocytosis), and then apoptotic neutrophils were added. Macrophages were collected after 12 h phagocytosis, and the mRNA levels of Rac1, IL-1β, Tnf-ɑ, TFG-β, Cx3cr1, Mmp2, Mmp9, and Il10 were detected by qPCR. n = 3; Data are presented as mean ± SEM (*P < 0.05, **P < 0.01, ***P < 0.001 and n.s. not significant).
Discussion
Macrophages are the key regulators of liver fibrosis and its resolution [34, 45, 46]; and an increasing number of studies have noted that recruited Ly6Chi monocytes/macrophages and their descendants Ly6Clo macrophage cooperate to influences the outcome of fibrosis in liver damage model [11, 40, 47, 48]. However, whether IFN-γ + LPS-primed MSCs alleviate schistosomiasis fibrosis through modulating Ly6Chi/Ly6Clo subtypes conversion is waiting to be clarified. Therefore, based on our previous finding that toll like receptor (TLR)4 agonist and IFN-γ primed MSCs alleviated hepatic fibrosis through modulating Th1/Th2 responses [32], this study focuses on the regulation of primed MSCs on macrophage subsets, as well as the underpinning mechanisms, aiming to provide new clues for the precision application of MSCs in fibrosis treatment.
This study ensured the same positive effects of primed MSCs in improving fibrosis as our previous report [32], with a simultaneous increase in Ly6CloCX3CR1hi without increase of Ly6ChiCX3CR1lo throughout the 2-week period after treatment. By contrast, non-primed MSC even aggravated fibrosis at 1 week post treatment with persistently higher levels of Ly6ChiCX3CR1lo throughout the 2-week post-treatment period.
In accordance with the analysis of macrophage subtypes, activated MSC group exhibited a restorative profile manifested by reducing pro-inflammatory cytokines (such as iNOS, TNF-ɑ, L-1β, CCL2, CX3CL1 and CXCL1), and moderately increasing levels of IL-10 and maintaining similarly high levels of Arg-1 as the control group at acute stage, followed by lower levels of iNOS and CX3CL1 at 2 weeks posttreatment; whereas non-primed MSCs group exhibited the strongest inflammation state manifested by increasing pro-inflammatory cytokines iNOS, TNF-ɑ, L-1β, CCL2, CX3CL1 and CXCL1 and IL-10, and simultaneously maintaining the lowest levels of Arg-1 among different groups at early stage of treatment, followed by persistently higher levels of IL-1βand CCL2 at 2 weeks posttreatment. Taken together, it is suggested that primed MSCs exert the beneficial effect on schistosomiasis fibrosis through inducing pro-resolving Ly6Clo subsets in a finely tuned manner. Furthermore, the finding that blocking recruitment of Ly6Chi MoMϕ led to reduced percentage of Ly6Clo MoMϕs, with a corresponding deteriorated liver pathology, confirmed that primed MSCs exhibited therapeutic effects through promoting conversion of Ly6Chi to Ly6Clo MoMϕs.
The main pathology of schistosomiasis is characterized by schistosome eggs induced Th2 or M2-rich granulomas, which marked by significant Arg1, IL-10 expression, etc [1, 4,5,6], though M2 macrophages is still considered to suppress Th2-dependent inflammation [49]. This raises confusion regarding the improvement of fibrosis through regulating macrophage subset conversion. In line with the definition of M2 polarization, this study demonstrated that primed MSCs induced an M2-dominant microenvironment at 1 week posttreatment, followed by the reduced Arg-1 level and lower inflammatory factors levels at 2 weeks. Therefore, primed MSCs did not induce a sustained M2 macrophage polarization state, but rather induced Ly6Clo phenotypic switch to attenuate fibrosis during the 2-week observation period. Next, the mechanism underlying Ly6Chi /Ly6Clo subtypes conversion was further explored.
Paracrine mechanisms, such as IL-4, IL-13, IL-10, PGE2, TSG-6, etc [10, 15, 35,36,37], have been reported to mediate effects of MSCs on phenotypic switching of Ly6Chi/lo macrophages. But this is clearly not the case in our experiment where the non-primed MSCs group showed no significant enhancement on Ly6Clo subtypes switching at 1 week posttreatment, in spite of the highest levels of IL-10 among all groups. In addition, another study from our laboratory found that upregulation of host interleukin-10 (IL-10) expression counteract clearance of schistosomes by macrophages [50], thus the overall benefit of high levels of IL-10 in the treatment of schistosomiasis warrants prudent assessment. Recently, it is reported that the phagocytosis of dying neutrophils by macrophages, known as efferocytosis, not only prevents secondary lysis and spillage of noxious neutrophil substances, but also triggers an anti-inflammatory and pro-resolving response in macrophages [40,41,42, 51]. Meanwhile, considering the close interaction between macrophages and neutrophils within the schistosomiasis granulomas, we attempted to explore efferocytosis-mediated mechanisms.
First, we observed that significant higher engulfment ratio of neutrophils both at 1 week and 2 weeks posttreatment in primed MSCs group (compared with control group or non-primed MSCs group, respectively), which is exactly consistent with the higher Ly6Clo macrophage in primed MSCs group (Figs. 5A–C). Furthermore, in vitro coculture experiment directly showed enhancement effects of primed MSCs on efferocytosis, accompanied by decreased inflammatory cytokines(IL-1β, Tnf-ɑ and CX3CL1 mRNA) and increased pro-resolving factors(CX3CR1,IL-10 and MMP2 [41]). Taken together, our results showed that primed MSCs promoted macrophage efferocytosis, as well as pro-inflammatory/pro-resolving subset switching in schistosomiasis mice.
There have been some similar reports in CCl4-induced fibrosis models, indicating that MSCs treatment promoted the macrophage transition from Ly6Chi to Ly6Clo population or induction of anti-inflammatory macrophages, while enhancing engulfment of apoptotic bodies [15] or the lysosomal function of macrophage [23]. Another report further disclosed that small extracellular vesicles (sEVs) derived from interferon-γ (IFN-γ) pre-conditioned MSCs exerted superior therapeutic effects to those of non-preconditioned MSCs therapy, which was related to induction of anti-inflammatory macrophages with increased phagocytic ability of bioparticles [24]. Here, we clearly showed that IFN-γ + LPS-primed MSCs promoted macrophage efferocytosis of neutrophils, as well as its subtype switching in schistosomiasis mice. Next, the potential targets of primed MSCs acting on during efferocytosis process were further identified.
We screened for engulfment receptors that can recognize PS (ligands exposed on apoptotic cells), including TAM family Mertk, Axl and Gas6, BAI1, Timd4 and integrin Itgb2,5 [39, 44]. Surprisingly, except that primed MSCs specially promoted levels of Itgb2 and the non-primed MSCs reduced Itgb2, no significant differences in other receptors were observed among different groups (Fig. 2s). Following engagement of eat-me signals with their receptors, downstream signaling pathways are activated, in which, GTPase Rac1 plays an important role in the organization of actin filament networks and in membrane ruffling to help macrophages complete the phagocytic program [39, 44]. Our results showed that primed MSCs specially promoted mRNA of Rac1, as well as Rac1protein activity at 1 hour after efferocytosis; Additionally, blocking Itgb2 ligation led to reduced Rac1 expression and Rac1 activity, as well as the decreased efferocytosis efficiency. Furthermore, both blocking Itgb2 ligation and inhibition Rac1 activation reverse promotion effects of primed MSCs in macrophage subtypes switching. Thus, our data suggest that primed MSCs promote Ly6Chi /Ly6Clo switching in a manner dependent on itgb2-Rac-1 mediated efferocytosis process.
Some reports have disclosed the significance of LFA-1(αLβ2) or itgb2 (CD18)) within human monocyte derived macrophage in the efferocytosis and its association with inflammation trajectory [52, 53]; a diminished efferocytosis was observed in CD18−/− mouse model, which drive inflammatory bone loss of periodontitis and related upregulation of interleukin (IL)-23 and IL-17 [53]. More interestingly, it has been indicated that CD18 controls the proliferation and maturation of monocytes in schistosomiasis mice; and low CD18 expression affects the accumulation of Ly6Chigh CD11b+ monocytes in the liver and their subsequent polarization to pro-resolving macrophages, thus interfere tissue repair during chronic schistosomiasis [54]. Our results provide application potential for this finding, suggesting that MSC therapy targeting to enhance Itgb2-Rac1 activation in a meticulous manner may be beneficial for schistosomiasis fibrosis treatment. Anyway, we still propose that other crosstalk signaling pathways may jointly contribute to efferocytosis process. In-depth mechanistic research and its applied validation studies in this field are indispensable.
Conclusion
In conclusion, our data demonstrated IFN-γ + LPS-primed MSCs promoted macrophage efferocytosis of neutrophil, as well as macrophage subset switching to participate in fibrosis resolution; and more importantly, macrophage efferocytosis is closely related to Itgb2-Rac1 pathway (Fig. 9). Although the mechanisms by which primed MSCs modulate Itgb2 expression remain to be elucidated, it offers novel insights into the therapeutic targets of MSC-based anti-fibrotic therapy.
Schematic of the mechanisms by which IFN-γ + LPS-primed MSCs regulate macrophage phenotypic switching through efferocytosis pathway.
Materials and methods
Host animals
BALB/c male mice, 6–8 weeks old, were obtained from the Laboratory Animal Center of Guangdong province (Guangzhou, China) and housed in the Biosafety Level-2 (BSL-2) laboratory of Sun Yat-sen University at 5 mice per cage on a 12-h light/dark cycle with 21–26 °C room temperature, 40–70% humidity, and ad libitum access to food and water. Experiments were performed as a non-blinded approach, and all animal work was performed according to the recommendations of the Laboratory Animal Welfare and Ethics Committee (LAWEC) of China and approved by the LAWEC of South China University of Technology.
Isolation and culture of mouse MSCs
Mouse MSCs were isolated from the bone marrow of 6-week-old C57BL/6 mice according to the improved low-density culture method reported previously [32]. In brief, femurs and tibiae were removed and bone marrow cavity was flushed with L-Dulbecco’s modified essential medium (DMEM) complete medium, and individual cells were obtained. After red blood cells were removed by ammonium chloride lysis, the remaining cells were cultured at the low density of 5ⅹ104 cells/cm2. Through continuous passage, nonadherent cells were gradually removed and the remaining adherent cells at 3 passages were collected and identified. Those positive for Sca-1and CD105, but negative for CD45, CD11b and MHC-Ⅰ, Ⅱ, were used for in vitro and in vivo experiment.
Liver fibrosis models induced by schistosomiasis
BALB/c male mice were divided randomly into 4 groups: normal group (uninfected), infected control group, MSC group, and IFN-γ plus LPS pretreated MSC group using a random number table. The number of animals in each group was indicated in the figure legend, which was determined by reference to our study experience and related researches [32, 55, 56]. Mice were percutaneously infected with 16 ± 2 pairs cercaria collected from S. japonicum-infected snails (bought from the Jiangsu Institute of Parasitic Diseases, Wuxi, China), followed by 5 days-long treatment with praziquantel (PZQ), once per day, oral gavage(i.g.) of 150 mg/kg, at 6 weeks post-infection, and then mice received 1ⅹ 105MSC via the tail vein or pretreated MSC suspended in 0.2 ml of saline. Animals were euthanized by cervical dislocation under anesthesia at 1 and 2 weeks after MSC treatment (or at the corresponding observation period in non-MSC control group). Thus, the effects of MSC treatment for the hepatic fibrosis induced by schistosomiasis are investigated.
When inhibition of monocyte recruitment was required, mice received intraperitoneal (i.p.) injection of 500 µLCCR2 antagonist (RS102895, KKL-med, USA) (15 mg/kg/day) The injections were performed three times a day for 1 week, prior to MSC treatment.
Hepatic histologic evaluation
Liver samples were obtained at 1 week, 2 weeks after MSC treatment, respectively. The remaining right liver lobe of each mouse (others were used for FACS analysis and RNA extraction, respectively) was fixed in 4% paraformaldehyde, embedded in paraffin, and cut into about 5-μm sections. The sections were stained with hematoxylin and eosin (H&E) and Masson to show granuloma formation and the deposition of collagen fibers in liver. Representative sections from each animal were scanned under ×200 magnifications with a compound microscope, and the degree of liver fibrosis was quantified by calculating the percentage of fibrosis area using AxioVision Rel 4.7 (Carl Zeiss GmbH, Jena, Germany).
Flow cytometry detection
Percentages of Ly6ChighCX3CR1low and Ly6ClowCX3CR1high macrophages in the liver of infected mice at 1 week and 2 weeks after treatment with MSC, respectively, were detected by flow cytometry. Liver mononuclear cells were isolated from the livers of mice as previously described [32]. Briefly, mouse livers were minced through a 100 μm cell strainer (BD Falcon) and re-suspended in PBS (pH 7.2) and centrifuged at 50 × g for 5 min. Supernatants containing liver mononuclear cells were collected and washed in PBS. The cell pellets were re-suspended in 35% Percoll solution (Sigma-Aldrich) and gently overlaid onto 70% Percoll. After centrifuged (600 × g) for 30 min at room temperature, the inter-phase containing liver mononuclear cells were collected and washed twice in PBS. These cells were subsequently used for surface staining.
The following antibodies were used for flow cytometry analysis: FITC-anti-mouse CD11b (11-0112-41, Thermofisher, San Diego, CA, USA), PE-cyanine 7-anti-mouse F4/80 (25-4801-82, Thermofisher), Percp-cyanine5.5-anti-mouse Ly-6C (128012, BioLegend, San Diego, CA, USA), PE-anti-mouse CD192(CCR2, 160105, BioLegend), APC/Cyanine7-anti-mouse CX3CR1(149047, BioLegend). Stained cells were detected by flow cytometry (CytoFLEX S, Beckman, USA), and the data were analyzed using FlowJo software (TreeStar, Ashland, OR, USA).
Real-time polymerase chain reaction
Total RNA was extracted with the TRIzol reagent (Invitrogen, CA, USA), and reverse transcription (RT) was performed using Evo M-MLV RT master mix (Accurate biology, China). The cDNA thus obtained was subjected to real-time PCR with the SYBR Green reagent (Roche Life Science, Switzerland) using Primers specific for the following genes listed in Table 1s. The housekeeping gene GADPH was used as an internal control and Quantitation of relative mRNA expression was calculated using the 2−ΔΔCt method
Liver immunofluorescence staining
About 4-mm-thick paraffin sections were dewaxed and incubated, according to the manufacturer’s instructions. The sections banded with MERTK (EPR17534-139, Abcam, USA; diluted at 1:1000) and Ly6G (EPR22909-135, Abcam, USA; diluted at 1:1000)) antibodies at 4 °C overnight and then were incubated with the corresponding secondary antibodies for 45 min. Nuclei were counterstained with 4’, 6-diamidino-2-phenylindole (DAPI, 564907, BD Pharmingen, USA; diluted at 1:1000). Observation and photo-taking were conducted by a confocal laser-scanning microscope (LSM700, Carl Zeiss, Germany)
Generation of bone marrow-derived macrophages (BMDMs) and preparation
CD45+CD11b+ monocytes were isolated from bone marrow cells by FACS sorting (using BD FACSAria II); and the enriched monocyte fraction was cultured in medium (Dulbecco’s modified Eagle’s medium/F12 media; Thermo Fisher Scientific, Waltham, MA) containing 10 ng/ml CSF-1 (Recombinant Murine M-CSF; Peprotech Inc., Rocky Hill, NJ) at 37 °C in 5% CO2. After 7 days-incubation, BMDMs were collected and identified. These cells were subsequently incubated without or with unpretreated MSC, or TLR4 + IFN-γ-treated MSC for 24 h at 37 °C in 5% CO2, and then used for coculture with apoptotic neutrophils.
Generation of apoptotic neutrophils
Bone marrow neutrophils were using Histopaque 11191 and 10771 (Sigma-Aldrich) as previously described [56]. Isolated neutrophils were stimulated with staurosporine (1 μM, 8 h) for induction of apoptosis and labeled with 2 μM Celltracker CM-DiI following the manufacturer’s instructions (Yeasen Biotechnology, Shanghai). The percentage of apoptosis was detected by FACS (>90%).
Assessment of efferocytosis in vitro
In vitro efferocytosis assay was performed as previously described [57], by co-culturing BMDMs with apoptotic neutrophils labeled with CM-DiI in a proportion of three apoptotic neutrophils: one macrophage. After co-incubation for 1 hour, cells were washed and fixed with 4% PFA and further stained for MERTK (EPR17534-139, Abcam, USA; diluted at 1:500). The efferocytosis was examined with a confocal laser-scanning microscope (LSM700, Carl Zeiss, Germany), and efferocytosis index was determined on 300 cells as the percentage of BMDMs containing at least one ingested apoptotic neutrophil. Digital images were processed using ImageJ software (National Institutes of Health, Bethesda, MD, USA).
ELISA
The levels of cytokine in the culture supernatants were determined according to the instruction of the ELISA Kits (eBioscience). In brief, mouse TNF and IL-10 were detected by biotinylated monoclonal antibodies, which were evidenced by avidin-conjugated horseradish peroxidase followed by incubation with TMB substrate. OD values at 450 nm were recorded using MK3 microplate reader.
Macrophage Rac1 activity G-LISA assays
BMDMs were cocultured with apoptotic neutrophils in 24-well plate, as described above. After co-coculture for indicated times, BMDMs were collected, and active Rac1 was measured using the Rac1 G-LISA Activation Assay kit (BK126, Cytoskeleton) according to the manufacturer’s instructions. For blocking test, before adding apoptotic neutrophils, BMDMs were first incubated with neutralizing anti-CD18 antibody (2.5ug/ml, eBioscience™) for 3 h. Background control values from cells pre-treated with Rac1 inhibitor NSC23766 (100 µm, Biofount) were subtracted from sample values to get Rac1 activity values.
Statistical analysis
All graphs and statistical analyses were performed using GraphPad Prism 8.0 (GraphPad Software Inc., San Diego, CA, USA). All data are shown as mean ± SEM. Multiple comparisons were analyzed by two-way ANOVA with Dunnett’s posttest for comparisons of three or more groups at different time points after treatment, or One-way ANOVA with Tukey’s multiple comparisons for comparisons of three groups at certain time point. All data were assessed for Gaussian distribution using the D’Agostino-Pearson normality test before analysis with these parametric tests).
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
McManus DP, Dunne DW, Sacko M, Utzinger J, Vennervald BJ, Zhou XN. Schistosomiasis. Nat Rev Dis Primers. 2018;4:13.
Niu X, Hu T, Hong Y, Li X, Shen Y. The role of praziquantel in the prevention and treatment of fibrosis associated with schistosomiasis: a review. J Trop Med. 2022;2022:1413711.
Abdul-Ghani RA, Hassan AA. Murine schistosomiasis as a model for human schistosomiasis mansoni: similarities and discrepancies. Parasitol Res. 2010;107:1–8.
Angeles JMM, Mercado VJP, Rivera PT. Behind enemy lines: immunomodulatory armamentarium of the schistosome parasite. Front Immunol. 2020;11:1018.
Llanwarne F, Helmby H. Granuloma formation and tissue pathology in Schistosoma japonicum versus Schistosoma mansoni infections. Parasite Immunol. 2021;43:e12778.
Fairfax K, Nascimento M, Huang SC, Everts B, Pearce EJ. Th2 responses in schistosomiasis. Semin Immunopathol. 2012;34:863–71.
Nascimento M, Huang SC, Smith A, Everts B, Lam W, Bassity E, et al. Ly6Chi monocyte recruitment is responsible for Th2 associated host-protective macrophage accumulation in liver inflammation due to schistosomiasis. PLoS Pathog. 2014;10:e1004282.
Barron L, Wynn TA. Macrophage activation governs schistosomiasis-induced inflammation and fibrosis. Eur J Immunol. 2011;41:2509–14.
Zwicker C, Bujko A, Scott CL. Hepatic macrophage responses in inflammation, a function of plasticity, heterogeneity or both?. Front Immunol. 2021;12:690813.
Wen Y, Lambrecht J, Ju C, Tacke F. Hepatic macrophages in liver homeostasis and diseases-diversity, plasticity and therapeutic opportunities. Cell Mol Immunol. 2021;18:45–56.
Li YH, Zhang Y, Pan G, Xiang LX, Luo DC, Shao JZ. Occurrences and functions of Ly6Chi and Ly6Clo macrophages in health and disease. Front Immunol. 2022;13:901672.
Daemen S, Gainullina A, Kalugotla G, He L, Chan MM, Beals JW, et al. Dynamic shifts in the composition of resident and recruited macrophages influence tissue remodeling in NASH. Cell Rep. 2021;34:108626.
Rückerl D, Cook PC. Macrophages assemble! But do they need IL-4R during schistosomiasis?. Eur J Immunol. 2019;49:996–1000.
Liu L, Wang P, Wang YS, Zhang YN, Li C, Yang ZY, et al. MiR-130a-3p alleviates liver fibrosis by suppressing HSCs activation and skewing macrophage to Ly6Clo phenotype. Front Immunol. 2021;12:696069.
Li YH, Shen S, Shao T, Jin MT, Fan DD, Lin AF, et al. Mesenchymal stem cells attenuate liver fibrosis by targeting Ly6Chi/lo macrophages through activating the cytokine-paracrine and apoptotic pathways. Cell Death Discov. 2021;7:239.
Yang X, Li Q, Liu W, Zong C, Wei L, Shi Y, et al. Mesenchymal stromal cells in hepatic fibrosis/cirrhosis: from pathogenesis to treatment. Cell Mol Immunol. 2023;20:583–99.
Yao L, Hu X, Dai K, Yuan M, Liu P, Zhang Q, et al. Mesenchymal stromal cells: promising treatment for liver cirrhosis. Stem Cell Res Ther. 2022;13:308.
Hu C, Wu Z, Li L. Mesenchymal stromal cells promote liver regeneration through regulation of immune cells. Int J Biol Sci. 2020;16:893–903.
Alsulami M, Abdel-Gaber R. Cell therapy as a new approach on hepatic fibrosis of murine model of Schistosoma mansoni-infection. Acta Parasitol. 2021;66:136–45.
Malmir A, Farivar S, Rezaei R, Tokhanbigli S, Hatami B, Mazhari S, et al. The effect of mesenchymal stem cells and imatinib on macrophage polarization in rat model of liver fibrosis. Cell Biol Int. 2023;47:135–43.
Torabi S, Zarrabi M, Shekari F, Poorkazem H, Lotfinia M, Bencina S, et al. Wharton’s Jelly mesenchymal stem cell-derived extracellular vesicles induce liver fibrosis-resolving phenotype in alternatively activated macrophages. J Cell Mol Med. 2024;28:e18507.
Yao Y, Zhang L, Cheng F, Jiang Q, Ye Y, Ren Y, et al. PPARγ-dependent hepatic macrophage switching acts as a central hub for hUCMSC-mediated alleviation of decompensated liver cirrhosis in rats. Stem Cell Res Ther. 2023;14:184.
Zhou J, Feng X, Zhu J, Feng B, Yao Q, Pan Q, et al. Mesenchymal stem cell treatment restores liver macrophages homeostasis to alleviate mouse acute liver injury revealed by single-cell analysis. Pharmacol Res. 2022;179:106229.
Takeuchi S, Tsuchiya A, Iwasawa T, Nojiri S, Watanabe T, Ogawa M, et al. Small extracellular vesicles derived from interferon-γ pre-conditioned mesenchymal stromal cells effectively treat liver fibrosis. NPJ Regen Med. 2021;6:19.
Watanabe T, Tsuchiya A, Takeuchi S, Nojiri S, Yoshida T, Ogawa M, et al. Development of a non-alcoholic steatohepatitis model with rapid accumulation of fibrosis, and its treatment using mesenchymal stem cells and their small extracellular vesicles. Regen Ther. 2020;14:252–61.
Abou Rayia DM, Ashour DS, Abo Safia HS, Abdel Ghafar MT, Amer RS, Saad AE. Human umbilical cord blood mesenchymal stem cells as a potential therapy for schistosomal hepatic fibrosis: an experimental study. Pathog Glob Health. 2023;117:190–202.
Hegab MH, Abd-Allah SH, Badawey MS, Saleh AA, Metwally AS, Fathy GM, et al. Therapeutic potential effect of bone marrow-derived mesenchymal stem cells on chronic liver disease in murine Schistosomiasis Mansoni. J Parasit Dis. 2018;42:277–86.
Dong L, Pu Y, Chen X, Qi X, Zhang L, Xu L, et al. hUCMSC-extracellular vesicles downregulated hepatic stellate cell activation and reduced liver injury in S. japonicum-infected mice. Stem Cell Res Ther. 2020;11:21.
Hammam OA, Elkhafif N, Attia YM, Mansour MT, Elmazar MM, Abdelsalam RM, et al. Wharton’s jelly-derived mesenchymal stem cells combined with praziquantel as a potential therapy for Schistosoma mansoni-induced liver fibrosis. Sci Rep. 2016;6:21005.
Li YQ, Chen YF, Dang YP, Wang Y, Shang ZZ, Ma Q, et al. Corilagin counteracts IL-13Rα1 signaling pathway in macrophages to mitigate schistosome egg-induced hepatic fibrosis. Front Cell Infect Microbiol. 2017;7:443.
Xie Y, Wen H, Yan K, Wang S, Wang X, Chen J, et al. Toxoplasma gondii GRA15(II) effector-induced M1 cells ameliorate liver fibrosis in mice infected with Schistosomiasis japonica. Cell Mol Immunol. 2018;15:120–34.
Liu C, Zhang YS, Chen F, Wu XY, Zhang BB, Wu ZD, et al. Immunopathology in schistosomiasis is regulated by TLR2,4- and IFN-γ-activated MSC through modulating Th1/Th2 responses. Stem Cell Res Ther. 2020;11:217.
Karsdal MA, Daniels SJ, Holm Nielsen S, Bager C, Rasmussen DGK, Loomba R, et al. Collagen biology and non-invasive biomarkers of liver fibrosis. Liver Int. 2020;40:736–50.
Cheng D, Chai J, Wang H, Fu L, Peng S, Ni X. Hepatic macrophages: Key players in the development and progression of liver fibrosis. Liver Int. 2021;41:2279–94.
Choi H, Lee RH, Bazhanov N, Oh JY, Prockop DJ. Anti-inflammatory protein TSG-6 secreted by primed MSCs attenuates zymosan-induced mouse peritonitis by decreasing TLR2/NF-κB signaling in resident macrophages. Blood. 2011;118:330–8.
Melief SM, Geutskens SB, Fibbe WE, Roelofs H. Multipotent stromal cells skew monocytes towards an anti-inflammatory interleukin-10-producing phenotype by production of interleukin-6. Haematologica. 2013;98:888–95.
Dal-Secco D, Wang J, Zeng Z, Kolaczkowska E, Wong CH, Petri B, et al. A dynamic spectrum of monocytes arising from the in situ reprogramming of CCR2+ monocytes at a site of sterile injury. J Exp Med. 2015;212:447–56.
Park SY, Kim IS. Engulfment signals and the phagocytic machinery for apoptotic cell clearance. Exp Mol Med. 2017;49:1–10.
Greenlee-Wacker MC. Clearance of apoptotic neutrophils and resolution of inflammation. Immunol Rev. 2016;273:357–70.
Graubardt N, Vugman M, Mouhadeb O, Caliari G, Pasmanik-Chor M, Reuveni D, et al. Ly6Chi monocytes and their macrophage descendants regulate neutrophil function and clearance in acetaminophen-induced liver injury. Front Immunol. 2017;8:626.
Yang W, Tao Y, Wu Y, Zhao X, Ye W, Zhao D, et al. Neutrophils promote the development of reparative macrophages mediated by ROS to orchestrate liver repair. Nat Commun. 2019;10:1076.
Kang JW, Lee SM. Resolvin D1 protects the liver from ischemia/reperfusion injury by enhancing M2 macrophage polarization and efferocytosis. Biochim Biophys Acta. 2016;1861:1025–35.
Bouchery T, Harris N. Neutrophil-macrophage cooperation and its impact on tissue repair. Immunol Cell Biol. 2019;97:289–98.
Moon B, Yang S, Moon H, Lee J, Park D. After cell death: the molecular machinery of efferocytosis. Exp Mol Med. 2023;55:1644–51.
Sun C, Matsukawa A. Role of macrophages in liver fibrosis. Acta Med Okayama. 2024;78:1–8.
Wang Z, Du K, Jin N, Tang B, Zhang W. Macrophage in liver fibrosis: identities and mechanisms. Int Immunopharmacol. 2023;120:110357.
Ramachandran P, Pellicoro A, Vernon MA, Boulter L, Aucott RL, Ali A, et al. Differential Ly-6C expression identifies the recruited macrophage phenotype, which orchestrates the regression of murine liver fibrosis. Proc Natl Acad Sci USA. 2012;109:E3186–95.
Yang W, Zhao X, Tao Y, Wu Y, He F, Tang L. Proteomic analysis reveals a protective role of specific macrophage subsets in liver repair. Sci Rep. 2019;9:2953.
Pesce JT, Ramalingam TR, Mentink-Kane MM, Wilson MS, El Kasmi KC, Smith AM, et al. Arginase-1-expressing macrophages suppress Th2 cytokine-driven inflammation and fibrosis. PLoS Pathog. 2009;5:e1000371.
Wang L, Zhu Z, Liao Y, Zhang L, Yu Z, Yang R, et al. Host liver-derived extracellular vesicles deliver miR-142a-3p induces neutrophil extracellular traps via targeting WASL to block the development of Schistosoma japonicum. Mol Ther. 2022;30:2092–107.
Soliman AM, Soliman M, Shah SSH, Baig HA, Gouda NS, Alenezi BT, et al. Molecular dynamics of inflammation resolution: therapeutic implications. Front Cell Dev Biol. 2025;13:1600149.
Kristóf E, Zahuczky G, Katona K, Doró Z, Nagy É, Fésüs L. Novel role of ICAM3 and LFA-1 in the clearance of apoptotic neutrophils by human macrophages. Apoptosis. 2013;18:1235–51.
Stark MA, Huo Y, Burcin TL, Morris MA, Olson TS, Ley K. Phagocytosis of apoptotic neutrophils regulates granulopoiesis via IL-23 and IL-17. Immunity. 2005;22:285–94.
Souza COS, Elias-Oliveira J, Pastore MR, Fontanari C, Rodrigues VF, Rodriguez V, et al. CD18 controls the development and activation of monocyte-to-macrophage axis during chronic schistosomiasis. Front Immunol. 2022;13:929552.
Zhang B, Wu X, Li J, Ning A, Zhang B, Liu J, et al. Hepatic progenitor cells promote the repair of schistosomiasis liver injury by inhibiting IL-33 secretion in mice. Stem Cell Res Ther. 2021;12:546.
Liao Y, Zhu Z, Liu Y, Wu J, Li D, Li Z, et al. Schistosome egg-derived extracellular vesicles deliver Sja-miR-71a inhibits host macrophage and neutrophil extracellular traps via targeting Sema4D. Cell Commun Signal. 2023;21:366.
Negreiros-Lima GL, Lima KM, Moreira IZ, Jardim BLO, Vago JP, Galvão I, et al. Cyclic AMP regulates key features of macrophages via PKA: recruitment, reprogramming and efferocytosis. Cells. 2020;9:128.
Acknowledgements
The study was supported by the “National Natural Science Foundation of China (General Program; Grant number: 82372277).
Author information
Authors and Affiliations
Contributions
JL and ZW conceived the study and revised the paper. JL designed the experiments, acquired the data and wrote the paper. YR performed and analyzed the data for most of the experiments. ZC, WH, and MX assisted with performing experiments or analyzing data.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
Animal experiments and procedures were approved by the Laboratory Animal Welfare and Ethics Committee of South China University of Technology on 02/26/2021(title of the approve project: Role of TLR4 and IFN-γ-primed MSCs in the treatment of liver fibrosis; Approval number:2021069). The manuscript reporting adheres to the ARRIVE (Animal Research: Reporting of In Vivo Experiments) 2.0 guidelines.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Lei, J., Ren, Y., Chen, Z. et al. Primed mesenchymal stem cells attenuate schistosomiasis fibrosis by enhancing macrophage subset switching and efferocytosis via Itgb2-Rac1 axis. Cell Death Discov. 12, 97 (2026). https://doi.org/10.1038/s41420-026-02947-w
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41420-026-02947-w