Abstract
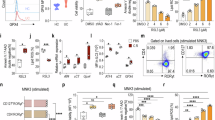
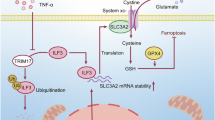
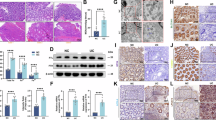
The critical role of group 2 innate lymphoid cells (ILC2s) in host defense and mucosal inflammation has been well established, yet the mechanisms underlying ILC2 survival and death remain unknown. Here, we report that ILC2s are vulnerable to ferroptosis, as evidenced by an abundance of ferroptosis signature genes and the accumulation of lipid peroxidation in ILC2s. Ablation of glutathione peroxidase 4 (GPX4) in ILC2s (Il5Cre/+Gpx4f/f mice) resulted in ferroptosis of ILC2s and diminished their responses. The increase in IFN-γ signaling-enhanced oxidative stress represents the mechanism underlying GPX4 ablation-induced ILC2 ferroptosis. Importantly, Il5Cre/+Gpx4f/f mice exhibited increased susceptibility to DSS-induced colitis. Clinically, ILC2s from inflammatory bowel disease (IBD) patients exhibit reduced GPX4 expression and increased lipid peroxidation. These observations revealed a previously unrecognized role of ferroptosis in ILC2 homeostasis, providing a promising therapeutic opportunity for mucosal inflammatory disorders.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Data availability
Colonic scRNA-seq data from Il5Cre/+Gpx4f/f and Il5Cre/+Gpx4+/+ immune cells under homeostasis are shown in Figs. 1 and 3 and are openly available from the China National Genomics Data Center (accession number OMIX009034). Bulk RNA-seq data for ILC2s treated with DMSO or RSL3 (Fig. S3) are openly available from the China National Genomics Data Center (accession number OMIX009038).
References
Spits H, Mjosberg J. Heterogeneity of type 2 innate lymphoid cells. Nat Rev Immunol. 2022;22:701–12. https://doi.org/10.1038/s41577-022-00704-5.
Serafini N, Vosshenrich CA, Di Santo JP. Transcriptional regulation of innate lymphoid cell fate. Nat Rev Immunol. 2015;15:415–28. https://doi.org/10.1038/nri3855.
von Moltke J, Ji M, Liang HE, Locksley RM. Tuft-cell-derived IL-25 regulates an intestinal ILC2-epithelial response circuit. Nature. 2016;529:221–5. https://doi.org/10.1038/nature16161.
Xu H, Ding J, Porter CBM, Wallrapp A, Tabaka M, Ma S, et al. Transcriptional atlas of intestinal immune cells reveals that neuropeptide alpha-CGRP modulates group 2 innate lymphoid cell responses. Immunity. 2019;51:696–708.e699. https://doi.org/10.1016/j.immuni.2019.09.004.
Wallrapp A, Riesenfeld SJ, Burkett PR, Abdulnour RE, Nyman J, Dionne D, et al. The neuropeptide NMU amplifies ILC2-driven allergic lung inflammation. Nature. 2017;549:351–6. https://doi.org/10.1038/nature24029.
Cao Y, Li Y, Wang X, Liu S, Zhang Y, Liu G, et al. Dopamine inhibits group 2 innate lymphoid cell-driven allergic lung inflammation by dampening mitochondrial activity. Immunity. 2023;56:320–35.e329. https://doi.org/10.1016/j.immuni.2022.12.017.
Sui P, Wiesner DL, Xu J, Zhang Y, Lee J, Van Dyken S, et al. Pulmonary neuroendocrine cells amplify allergic asthma responses. Science. 2018;360. https://doi.org/10.1126/science.aan8546.
Talbot S, Abdulnour RE, Burkett PR, Lee S, Cronin SJ, Pascal MA, et al. Silencing nociceptor neurons reduces allergic airway inflammation. Neuron. 2015;87:341–54. https://doi.org/10.1016/j.neuron.2015.06.007.
Laffont S, Blanquart E, Savignac M, Cenac C, Laverny G, Metzger D, et al. Androgen signaling negatively controls group 2 innate lymphoid cells. J Exp Med. 2017;214:1581–92. https://doi.org/10.1084/jem.20161807.
Liu G, Chen Y, Wang Y, Deng X, Xiao Q, Zhang L, et al. Angiotensin II enhances group 2 innate lymphoid cell responses via AT1a during airway inflammation. J Exp Med.2022;219. https://doi.org/10.1084/jem.20211001.
Cui W, Nagano Y, Morita S, Tanoue T, Yamane H, Ishikawa K, et al. Diet-mediated constitutive induction of novel IL-4+ ILC2 cells maintains intestinal homeostasis in mice. J Exp Med. 2023;220. https://doi.org/10.1084/jem.20221773.
Hodge SH, Krauss MZ, Kaymak I, King JI, Howden AJM, Panic G, et al. Amino acid availability acts as a metabolic rheostat to determine the magnitude of ILC2 responses. J Exp Med. 2023;220. https://doi.org/10.1084/jem.20221073.
Nadjsombati MS, McGinty JW, Lyons-Cohen MR, Jaffe JB, DiPeso L, Schneider C, et al. Detection of Succinate by Intestinal Tuft Cells Triggers a Type 2 Innate Immune Circuit. Immunity. 2018;49:33–41.e37. https://doi.org/10.1016/j.immuni.2018.06.016.
Schneider C, O’Leary CE, von Moltke J, Liang HE, Ang QY, Turnbaugh PJ, et al. A metabolite-triggered tuft cell-ILC2 circuit drives small intestinal remodeling. Cell. 2018;174:271–84.e214. https://doi.org/10.1016/j.cell.2018.05.014.
Nussbaum JC, Van Dyken SJ, von Moltke J, Cheng LE, Mohapatra A, Molofsky AB, et al. Type 2 innate lymphoid cells control eosinophil homeostasis. Nature. 2013;502:245–8. https://doi.org/10.1038/nature12526.
Lei AH, Xiao Q, Liu GY, Shi K, Yang Q, Li X, et al. ICAM-1 controls development and function of ILC2. J Exp Med. 2018;215:2157–74. https://doi.org/10.1084/jem.20172359.
Tamari M, Del Bel KL, Ver Heul AM, Zamidar L, Orimo K, Hoshi M, et al. Sensory neurons promote immune homeostasis in the lung. Cell. 2024;187:44–61.e17. https://doi.org/10.1016/j.cell.2023.11.027.
Liu GY, Wang Y, Yi XF, Wang JY, Zhang J, Feng XX, et al. BACH2 drives the development and function of group 2 innate lymphoid cells. Sci Adv. 2025;11. https://doi.org/10.1126/sciadv.ads4323.
Zang Y, Liu S, Rao Z, Wang Y, Zhang B, Li H, et al. Retinoid X receptor gamma dictates the activation threshold of group 2 innate lymphoid cells and limits type 2 inflammation in the small intestine. Immunity. 2023;56:2542–2554.e2547. https://doi.org/10.1016/j.immuni.2023.08.019.
Monticelli LA, Osborne LC, Noti M, Tran SV, Zaiss DM, Artis D. IL-33 promotes an innate immune pathway of intestinal tissue protection dependent on amphiregulin-EGFR interactions. Proc Natl Acad Sci USA. 2015;112:10762–7. https://doi.org/10.1073/pnas.1509070112.
Uddin J, Yano H, Parkhurst CN, Zhang W, Ahmed A, Ribeiro de Godoy V, et al. CGRP-related neuropeptide adrenomedullin 2 promotes tissue-protective ILC2 responses and limits intestinal inflammation. Nature Immunology. 2025;26:1516–26. https://doi.org/10.1038/s41590-025-02243-2.
Zaiss DMW, Pearce EJ, Artis D, McKenzie ANJ, Klose CSN. Cooperation of ILC2s and T(H)2 cells in the expulsion of intestinal helminth parasites. Nat Rev Immunol. 2023. https://doi.org/10.1038/s41577-023-00942-1.
Neill DR, Wong SH, Bellosi A, Flynn RJ, Daly M, Langford TK, et al. Nuocytes represent a new innate effector leukocyte that mediates type-2 immunity. Nature. 2010;464:1367–70. https://doi.org/10.1038/nature08900.
McKenzie GJ, Fallon PG, Emson CL, Grencis RK, McKenzie AN. Simultaneous disruption of interleukin (IL)-4 and IL-13 defines individual roles in T helper cell type 2-mediated responses. J Exp Med. 1999;189:1565–72. https://doi.org/10.1084/jem.189.10.1565.
Madden KB, Whitman L, Sullivan C, Gause WC, Urban JF Jr., Katona IM, et al. Role of STAT6 and mast cells in IL-4- and IL-13-induced alterations in murine intestinal epithelial cell function. J Immunol. 2002;169:4417–22. https://doi.org/10.4049/jimmunol.169.8.4417.
Hasnain SZ, Evans CM, Roy M, Gallagher AL, Kindrachuk KN, Barron L, et al. Muc5ac: a critical component mediating the rejection of enteric nematodes. J Exp Med. 2011;208:893–900. https://doi.org/10.1084/jem.20102057.
Arifuzzaman M, Won TH, Yano H, Uddin J, Emanuel ER, Hu E, et al. Dietary fiber is a critical determinant of pathologic ILC2 responses and intestinal inflammation. J Exp Med. 2024;221. https://doi.org/10.1084/jem.20232148.
Tsou AM, Yano H, Parkhurst CN, Mahlakõiv T, Chu C, Zhang W, et al. Neuropeptide regulation of nonredundant ILC2 responses at barrier surfaces. Nature. 2022;611:787–93. https://doi.org/10.1038/s41586-022-05297-6.
Liu G, Huang H, Wang Y, Han Y, Wang J, Shi M, et al. ILC2 instructs neural stem and progenitor cells to potentiate neurorepair after stroke. Neuron. 2025. https://doi.org/10.1016/j.neuron.2025.03.014.
Lei G, Zhuang L, Gan B. Targeting ferroptosis as a vulnerability in cancer. Nat Rev Cancer. 2022;22:381–96. https://doi.org/10.1038/s41568-022-00459-0.
Jiang X, Stockwell BR, Conrad M. Ferroptosis: mechanisms, biology and role in disease. Nat Rev Mol Cell Biol. 2021;22:266–82. https://doi.org/10.1038/s41580-020-00324-8.
Chen X, Zeh HJ, Kang R, Kroemer G, Tang D. Cell death in pancreatic cancer: from pathogenesis to therapy. Nat Rev Gastroenterol Hepatol. 2021;18:804–23. https://doi.org/10.1038/s41575-021-00486-6.
Yang WS, SriRamaratnam R, Welsch ME, Shimada K, Skouta R, Viswanathan VS, et al. Regulation of ferroptotic cancer cell death by GPX4. Cell. 2014;156:317–31. https://doi.org/10.1016/j.cell.2013.12.010.
Qiu B, Zandkarimi F, Bezjian CT, Reznik E, Soni RK, Gu W, et al. Phospholipids with two polyunsaturated fatty acyl tails promote ferroptosis. Cell. 2024;187:1177–1190.e1118. https://doi.org/10.1016/j.cell.2024.01.030.
Oh SJ, Ikeda M, Ide T, Hur KY, Lee MS. Mitochondrial event as an ultimate step in ferroptosis. Cell Death Discov. 2022;8:414 https://doi.org/10.1038/s41420-022-01199-8.
Marchi S, Guilbaud E, Tait SWG, Yamazaki T, Galluzzi L. Mitochondrial control of inflammation. Nat Rev Immunol. 2023;23:159–73. https://doi.org/10.1038/s41577-022-00760-x.
Song X, Long D. Nrf2 and ferroptosis: a new research direction for neurodegenerative diseases. Front Neurosci. 2020;14:267 https://doi.org/10.3389/fnins.2020.00267.
Xu S, He Y, Lin L, Chen P, Chen M, Zhang S. The emerging role of ferroptosis in intestinal disease. Cell Death Dis. 2021;12:289. https://doi.org/10.1038/s41419-021-03559-1.
Xu M, Tao J, Yang Y, Tan S, Liu H, Jiang J, et al. Ferroptosis involves in intestinal epithelial cell death in ulcerative colitis. Cell Death Dis. 2020;11:86. https://doi.org/10.1038/s41419-020-2299-1.
Panda SK, Peng V, Sudan R, Ulezko Antonova A, Di Luccia B, Ohara TE, et al. Repression of the aryl-hydrocarbon receptor prevents oxidative stress and ferroptosis of intestinal intraepithelial lymphocytes. Immunity. 2023;56:797–812.e794. https://doi.org/10.1016/j.immuni.2023.01.023.
Wang S, Liu W, Wang J, Bai X. Curculigoside inhibits ferroptosis in ulcerative colitis through the induction of GPX4. Life Sci. 2020;259:118356. https://doi.org/10.1016/j.lfs.2020.118356.
Li Z, Ferguson L, Deol KK, Roberts MA, Magtanong L, Hendricks JM, et al. Ribosome stalling during selenoprotein translation exposes a ferroptosis vulnerability. Nat Chem Biol. 2022;18:751–61. https://doi.org/10.1038/s41589-022-01033-3.
Doll S, Proneth B, Tyurina YY, Panzilius E, Kobayashi S, Ingold I, et al. ACSL4 dictates ferroptosis sensitivity by shaping cellular lipid composition. Nat Chem Biol. 2016;13:91–8. https://doi.org/10.1038/nchembio.2239.
Wang Y, Tian Q, Hao Y, Yao W, Lu J, Chen C, et al. The kinase complex mTORC2 promotes the longevity of virus-specific memory CD4(+) T cells by preventing ferroptosis. Nat Immunol. 2022;23:303–17. https://doi.org/10.1038/s41590-021-01090-1.
von Krusenstiern AN, Robson RN, Qian N, Qiu B, Hu F, Reznik E, et al. Identification of essential sites of lipid peroxidation in ferroptosis. Nat Chem Biol. 2023;19:719–30. https://doi.org/10.1038/s41589-022-01249-3.
Nakamura T, Hipp C, Santos Dias Mourao A, Borggrafe J, Aldrovandi M, Henkelmann B, et al. Phase separation of FSP1 promotes ferroptosis. Nature. 2023;619:371–7. https://doi.org/10.1038/s41586-023-06255-6.
Mayassi T, Li C, Segerstolpe A, Brown EM, Weisberg R, Nakata T, et al. Spatially restricted immune and microbiota-driven adaptation of the gut. Nature. 2024;636:447–56. https://doi.org/10.1038/s41586-024-08216-z.
Hardman CS, Panova V, McKenzie AN. IL-33 citrine reporter mice reveal the temporal and spatial expression of IL-33 during allergic lung inflammation. Eur J Immunol. 2013;43:488–98. https://doi.org/10.1002/eji.201242863.
Cayrol C, Girard JP. IL-33: an alarmin cytokine with crucial roles in innate immunity, inflammation and allergy. Curr Opin Immunol. 2014;31:31–7. https://doi.org/10.1016/j.coi.2014.09.004.
Barrat FJ, Crow MK, Ivashkiv LB. Interferon target‒gene expression and epigenomic signatures in health and disease. Nature Immunology. 2019;20:1574–83. https://doi.org/10.1038/s41590-019-0466-2.
De Benedetti F, Prencipe G, Bracaglia C, Marasco E, Grom AA. Targeting interferon-γ in hyperinflammation: opportunities and challenges. Nat Rev Rheumatol. 2021;17:678–91. https://doi.org/10.1038/s41584-021-00694-z.
Zhao J, Jia Y, Mahmut D, Deik AA, Jeanfavre S, Clish CB, et al. Human hematopoietic stem cell vulnerability to ferroptosis. Cell. 2023;186:732–47.e716. https://doi.org/10.1016/j.cell.2023.01.020.
Waddell A, Vallance JE, Hummel A, Alenghat T, Rosen MJ. IL-33 induces murine intestinal goblet cell differentiation indirectly via innate lymphoid cell IL-13 secretion. J Immunol. 2019;202:598–607. https://doi.org/10.4049/jimmunol.1800292.
Yang D, Jacobson A, Meerschaert KA, Sifakis JJ, Wu M, Chen X, et al. Nociceptor neurons direct goblet cells via a CGRP-RAMP1 axis to drive mucus production and gut barrier protection. Cell. 2022;185:4190–205.e4125. https://doi.org/10.1016/j.cell.2022.09.024.
Zhang W, Lyu M, Bessman NJ, Xie Z, Arifuzzaman M, Yano H, et al. Gut-innervating nociceptors regulate the intestinal microbiota to promote tissue protection. Cell. 2022;185:4170–89.e4120. https://doi.org/10.1016/j.cell.2022.09.008.
Yao Y, Chen Z, Zhang H, Chen C, Zeng M, Yunis J, et al. Selenium–GPX4 axis protects follicular helper T cells from ferroptosis. Nature Immunology. 2021;22:1127–39. https://doi.org/10.1038/s41590-021-00996-0.
Huang LJ, Mao XT, Li YY, Liu DD, Fan KQ, Liu RB, et al. Multiomics analyses reveal a critical role of selenium in controlling T-cell differentiation in Crohn’s disease. Immunity. 2021;54:1728–44.e1727. https://doi.org/10.1016/j.immuni.2021.07.004.
Haberman Y, Karns R, Dexheimer PJ, Schirmer M, Somekh J, Jurickova I, et al. Ulcerative colitis mucosal transcriptomes reveal mitochondriopathy and personalized mechanisms underlying disease severity and treatment response. Nat Commun. 2019;10:38. https://doi.org/10.1038/s41467-018-07841-3.
Lyu M, Suzuki H, Kang L, Gaspal F, Zhou W, Goc J, et al. ILC3s select microbiota-specific regulatory T cells to establish tolerance in the gut. Nature. 2022;610:744–51. https://doi.org/10.1038/s41586-022-05141-x.
Li J, Jiang JL, Chen YM, Lu WQ. KLF2 inhibits colorectal cancer progression and metastasis by inducing ferroptosis via the PI3K/AKT signaling pathway. J Pathol Clin Res. 2023;9:423–35. https://doi.org/10.1002/cjp2.325.
Karagiannis F, Masouleh SK, Wunderling K, Surendar J, Schmitt V, Kazakov A, et al. Lipid-Droplet Formation Drives Pathogenic Group 2 Innate Lymphoid Cells in Airway Inflammation. Immunity. 2020;52:620–34.e626. https://doi.org/10.1016/j.immuni.2020.03.003.
Rao Z, Liu S, Li Z, Wang Q, Gao F, Peng H, et al. Alarmin-loaded extracellular lipid droplets induce airway neutrophil infiltration during type 2 inflammation. Immunity. 2024;57:2514–29.e2517. https://doi.org/10.1016/j.immuni.2024.09.001.
Zhang Y, Swanda RV, Nie L, Liu X, Wang C, Lee H, et al. mTORC1 couples cyst(e)ine availability with GPX4 protein synthesis and ferroptosis regulation. Nat Commun. 2021;12:1589. https://doi.org/10.1038/s41467-021-21841-w.
Li X, He J, Gao X, Zheng G, Chen C, Chen Y, et al. GPX4 restricts ferroptosis of NKp46+ILC3s to control intestinal inflammation. Cell Death Dis. 2024;15. https://doi.org/10.1038/s41419-024-07060-3.
Kato A, Kita H. The immunology of asthma and chronic rhinosinusitis. Nat Rev Immunol. 2025;25:569–87. https://doi.org/10.1038/s41577-025-01159-0.
Lambrecht BN, Ahmed E, Hammad H. The immunology of asthma. Nat Immunol. 2025;26:1233–45. https://doi.org/10.1038/s41590-025-02212-9.
Liu G-Y, Deng X-H, Li X, Cao Y-J, Xing Y-F, Zhou P, et al. Expansion of group 2 innate lymphoid cells in patients with end-stage renal disease and their clinical significance. J Immunol. 2020;205:36–44. https://doi.org/10.4049/jimmunol.1901095.
Wang Y, Wang J, Liu G, Yi X, Wu J, Cao H, et al. NRP1 instructs IL-17-producing ILC3s to drive colitis progression. Cell Mol Immunol. 2025;22:161–75. https://doi.org/10.1038/s41423-024-01246-7.
Picelli S, Bjorklund AK, Faridani OR, Sagasser S, Winberg G, Sandberg R. Smart-seq2 for sensitive full-length transcriptome profiling in single cells. Nat Methods. 2013;10:1096–8. https://doi.org/10.1038/nmeth.2639.
Hao Y, Stuart T, Kowalski MH, Choudhary S, Hoffman P, Hartman A, et al. Dictionary learning for integrative, multimodal and scalable single-cell analysis. Nat Biotechnol. 2024;42:293–304. https://doi.org/10.1038/s41587-023-01767-y.
Acknowledgements
This work was supported by the following grants: National Key Research and Development Program of China (No. 2025YFA1309100 to JZ), National Natural Science Foundation of China (No. 82525104, 82430055, 82130049 to JZ; 82321001 to YY; and 82225015, 82171284 to QL, 82201917 to GL). This work was also supported by the New Cornerstone Science Foundation through the XPLORER PRIZE (to QL). China Postdoctoral Science Foundation (No. 2022M713571 to GL).
Author information
Authors and Affiliations
Contributions
JZ conceived and supervised this study and wrote the manuscript with input from all the authors. QL cosupervised this study. GL, YW, and JW performed most of the experiments. GL analyzed the data and wrote the manuscript. HH analyzed the bioinformatic data. JY, YZ, YJ and PZ participated in the animal experiments. JW and HC collected the samples and clinical information of the IBD patients and paired healthy individuals. YY and CC provided suggestions.
Corresponding authors
Ethics declarations
Competing interests
The authors declare that they have no competing financial interests. JZ is an editorial board member of Cellular & Molecular Immunology; she has not been involved in the peer review or the decision-making of the manuscript.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Liu, G., Wang, Y., Huang, H. et al. GPX4-dependent ferroptosis governs ILC2 homeostasis and colitis progression. Cell Mol Immunol 23, 367–382 (2026). https://doi.org/10.1038/s41423-026-01388-w
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41423-026-01388-w