Abstract
Europe’s Beating Cancer Plan is a substantial European Union (EU) investment into cancer prevention and treatment. Integration of genetic services towards personalised cancer prevention and care is a flagship of this plan. Genetic counselling is critical to this integration, facilitating informed patient decision making and improved clinical management. However, growing demands for genetic testing and concurrently increasing workforce shortages necessitate new strategies to equitably ensure sustainable access to counselling across the EU. This project aimed to inform future European activities by identifying priority European strategies for addressing common European genetic literacy, workforce, and reimbursement barriers to genetic counselling in cancer noted in prior work. A Delphi survey was conducted, with genetics, oncology, and patient stakeholders invited from all EU Member States. The response rate was 62% (124 total invitations). Over three phases, 77 participants – 28 geneticists; 14 oncologists; 18 genetic counsellors; 16 patient representatives; 1 otherwise qualified expert – rated 19 strategies according to their Importance, Urgency, and Feasibility and selected their top three priority strategies. Five strategies met pre-defined consensus thresholds and received a clear plurality of priority ratings: (1) EU-wide genetic counsellor recognition; (2) Including genetics expertise in oncology guideline creation; (3) Shared EU genetic counsellor registration/education with legal weight; (4) Mandatory counselling reimbursement when clinical guidelines are met; (5) Mandatory inclusion of genetics in oncology fellowship/continuing education. Results provide a roadmap of European actions which promise to sustainably improve access to genetic counselling in cancer care. Upcoming and ongoing EU projects promise to advance their implementation.
Similar content being viewed by others
Introduction
The inauguration of Europe’s Beating Cancer Plan (EBCP) in 2021 marked the beginning of a new era of European Union (EU) investment into coordinated activities to improve cancer prevention, diagnosis, treatment, and survivorship [1]. EBCP is organised around ten flagship initiatives, of which two relate specifically to genetic/genomic technologies: Flagship 6 ‘Cancer Diagnostics and Treatments for All’ includes an aim to “improve access to innovative cancer diagnosis and treatments [including] ‘next generation sequencing’ for quick and efficient genetic profiles.”; and Flagship 7 ‘The European Initiative to Understand Cancer (UNCAN.eu)’ includes an aim to facilitate personalised approaches to cancer prevention and care through increasing integration of polygenic risk scores [1]. The prominent feature of genetics in the EBCP underscores the growing importance of genetic technologies in guiding targeted cancer treatments and prevention strategies, in particular to address the estimated 5–15% of cancer cases linked to genetic cancer predisposition syndromes [2].
Funded under the EBCP, the CAN.HEAL project (https://canheal.eu) ran from November 2022 – April 2025 with the broad objective to ‘Build the EU Cancer and Public Health Genomics Platform’ and provide a critical foundation for the realisation of EBCP Flagships 6 and 7. Included amongst the activities of CAN.HEAL was a work program related to genetic counselling, in recognition of its essential and often legally mandated [3] role in helping patients and their families understand and make informed decisions about genetic testing and subsequent measures [4].
Ideally, genetic counselling is delivered by health practitioners with clinical/medical genetics training and expertise, including clinical/medical geneticist physicians and/or genetic counsellors, but also non-genetics physicians and health professionals with genetics competencies [3, 5]. Genetic counselling delivered by individuals with insufficient genetics expertise has been linked to negative impacts on care through avoidable costs (e.g. unnecessary genetic tests), suboptimal clinical management, and negative psychosocial outcomes in patients and family members [6].
An increasing demand for genetic testing related to the push towards personalised cancer medicine across paediatric and adult patient populations [7] is also leading to increasing demands for genetic counselling by appropriate experts. Rapid advances in knowledge and methods (e.g. evolving gene panels, multi-omics analyses, polygenic risk scores) further fuel needs for both broad and further specialised genetics expertise to meet clinical demands. However, shortages in both the general healthcare and clinical/medical genetics workforce across EU Member States already exist and are predicted to further grow over the coming decades [8, 9]. New strategies are needed to ensure that quality genetic counselling can be provided in sufficient volumes and in a reasonable timeframe to meet increasing genetic testing demands across the EU.
To guide the development of these new strategies, preliminary work identified common barriers to the delivery of high-quality genetic counselling present across Member States: genetic literacy (of patients and non-genetics health professionals; a noted barrier in all 27 Member States); workforce capacity (a noted barrier in 25 Member States); and insurance reimbursement (a noted barrier in 13 Member States) [3, 10]. These common barriers were identified in similar proportions by both expert health professionals and cancer patient organisation representatives [3, 10].
The three identified European barriers to genetic counselling in cancer provide clear targets for new strategies and indicate that European-level action is likely to most efficiently address these shared challenges. Accordingly, we aimed to identify, through structured consultation with key genetics, oncology, and patient stakeholders from across the EU, priority European strategies to address these three common European barriers to genetic counselling in cancer and guide EU policy and initiatives.
Subjects and methods
Participants
Invitations to participate in a Delphi survey were sent to 1–3 individuals from each of the following four stakeholder groups per Member State (n = 124 total individuals) by the lead author (JMM) in consultation with the corresponding author (AKB): clinical/medical geneticist physicians; clinical/medical oncologist physicians; genetic counsellors; and cancer patient organisation representatives. Invitations were sent to at least one clinical/medical geneticist physician, clinical/medical oncologist physician, and cancer patient organisation representative from all 27 Member States; at least one genetic counsellor (specialised allied health professional, qualifications defined according to European Board of Medical Genetics registration criteria [11]) was invited from the 15 Member States in which genetic counsellors are known to be active [12]. Individuals across stakeholder groups were invited because they were in a leadership position of their national genetics/oncology society or national patient organisation (defined according to membership in the Association of European Cancer Leagues) and/or recognised as possessing nationally-recognised expertise in genetic counselling in cancer (e.g. through prior work with the CAN.HEAL consortium [3, 10]). In some cases, multiple individuals from a given stakeholder group and Member State were invited to collectively achieve the desired combination of expertise and national leadership. This approach was favoured over a broader ‘snowball’ approach to participant recruitment because it avoided responses from individuals with unknown expertise and national representativeness and allowed us to quantify response rates.
Delphi survey
A modified ‘Policy Delphi’ process was used to identify priority European strategies to address common barriers to genetic counselling in cancer [13]. Given the large number of invited stakeholders (n = 124), a structured approach was favoured in all three Phases described below, forgoing an initial phase of open-ended questions. All Phase I participants were invited to contribute to both subsequent Phases (II and III) [14]. Individual participant responses and identities were kept confidential throughout the Delphi process [13]. Thresholds for consensus ‘high’ Importance and Urgency ratings for a given strategy were defined a priori and as per prior Delphi studies as an average rating ≥7 out of 9 [13]. The following descriptors were defined to contextualise average Feasibility ratings: low feasibility (1–3); moderate feasibility (4–6); high feasibility (7–9).
Phase I
Participants were first asked to rate (1-9 Likert scale [13]) nine European strategies proposed by the research team (JMM, TR, JR, AKB)(Table 1) according to their ‘Importance (i.e. impact potential)’, ‘Urgency (i.e. need for swift implementation)’, and ‘Feasibility (i.e. ease of implementation)’.
Participants had the option to provide additional text comments for each strategy, as well as propose and rate (Importance, Urgency, and Feasibility criteria; 1-9 Likert scale) up to three of their own strategies.
Phase II
Participants were presented with a summary of all Phase I ratings, and asked to rate 10 additional strategies (Importance, Urgency, and Feasibility criteria; 1-9 Likert scale) which were aggregated around common themes from 64 strategies proposed by participants in Phase I. Additionally, participants were asked to designate their #1, #2, and #3 priority strategies from the 19 strategies presented in Phase I and Phase II to help define a focused set of strategies to guide future action.
Phase III
To further refine the relative prioritisation of the highest ranked strategies from Phase II, participants were asked to again designate their #1, #2, and #3 priority strategies from five strategies which received a clear plurality of priority rankings in Phase II.
Statistical analyses
Priority rankings were weighted using the Borda count developed for ranked choice voting (i.e. Priority #1 ranking = 3 points; Priority #2 ranking = 2 points; Priority #3 ranking = 1 point) [15]. The non-parametric Kruskal-Wallis 1-way ANOVA was used to test for significant differences in Importance, Urgency, and Feasibility ratings and weighted priority rankings between the four stakeholder groups: clinical/medical geneticists; clinical/medical oncologists; genetic counsellors; and cancer patient organisation representatives. When significant main effects were found, pairwise comparisons between professional groups were performed using the non-parametric Dunn’s test and Bonferroni correction for multiple comparisons. The Kruskal-Wallis 1-way ANOVA with post-hoc Dunn’s tests with Bonferroni correction were also used to test for significant differences in response rates across the four stakeholder groups and 27 EU Member States. Differences in weighted priority rankings between strategies were tested using the non-parametric Friedman test; pairwise post-hoc comparisons were conducted using Wilcoxon signed rank tests with the Bonferroni correction for multiple comparisons when significant main effects were observed. Significance was set at α = 0.05 for all statistical tests. All statistical analyses were performed in SPSS version 29.0 (IBM Corporation).
Role of funding source
The European Commission had no role in the study design, collection/analysis/interpretation of data, or writing of the report, nor in the decision to submit the paper for publication.
Results
A total of 77 individuals accepted invitations to participate in Phase I of the Delphi survey from 124 total invitations (62% overall response rate). Response rates for each stakeholder group were: clinical/medical geneticists (29 of 44, 66%); clinical/medical oncologists (13 of 33, 39%); genetic counsellors (18 of 20, 90%); and cancer patient organisations (16 of 27, 59%). A total of 34 participants were from Southern/Eastern European countries (44%), with 43 from Western/Northern European countries (56%), as classified by EuroVoc [16]. Response rates were significantly different across stakeholder groups (χ2(3) = 13.5; p = 0.01) but not EU Member States (χ2(26) = 19.2; p = 0.86). Pairwise post-hoc comparisons showed significant differences in response rates between clinical/medical oncologists and genetic counsellors (p = 0.004), but no other significant pairwise differences (p > 0.32). The majority of Phase I participants were aged 41–60 (n = 57; 74%) and female (n = 50; 65%). In Phases II and III, 66 and 68 individuals participated, respectively (86–88% retention rate). See Table 2 for a breakdown of participants by Member State, Phase, and professional group.
Phase I
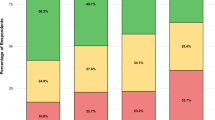
Six out of nine strategies met consensus criteria (average rating ≥7 out of 9) for both Importance and Urgency, with another two strategies meeting consensus criteria for Importance only (Fig. 1). Average ratings were higher for Importance than Urgency across all strategies. Significant differences in ratings between professional stakeholder groups were found for strategies P1, P2, P7, and P8 (8.7≤χ2(3)≤16.6; p < 0.05). Post-hoc pairwise comparisons showed significantly higher Importance and Urgency ratings of strategies P1 (genetic counsellor recognition) and P2 (genetic counsellor education/accreditation) by Genetic Counsellors compared to Clinical/Medical Geneticists and, to a lesser extent, Clinical/Medical Oncologists (p < 0.02; Table 3). Additionally, Cancer Patient Organisation representatives rated genetics education programs for secondary students (P8) as being significantly more Important and Urgent than genetic counsellors (p < 0.03).
Error bars represent standard error. Consensus Importance and Urgency was defined a priori as an average rating ≥7. # - significant differences between professional groups, Importance rating (p < 0.05). * - significant differences between professional groups, Urgency rating (p < 0.05).
Phase II
A total of 10 additional strategies (Table 4) were aggregated from 64 total proposals from 30 Delphi participants (13 clinical/medical geneticists; 1 clinical/medical oncologist; 9 genetic counsellors; 6 cancer patient organisation representatives; 1 otherwise qualified expert) in Phase I and presented to participants for rating in Phase II.
Importance and Urgency ratings for five out of ten aggregated strategies met consensus criteria (average rating ≥7 out of 9); Importance but not Urgency ratings met consensus criteria for two additional aggregated strategies (Fig. 2). Average importance ratings were higher for Importance than Urgency across all strategies.
Error bars represent standard error. Consensus Importance and Urgency was defined a priori as an average rating ≥7. # - significant differences between professional groups, Importance rating (p < 0.05). * - significant differences between professional groups, Urgency rating (p < 0.05). ^ - significant differences between professional groups, Feasibility rating (p < 0.05).
Cancer patient organisation representatives were overall the most positive regarding the Importance, Urgency, and Feasibility of Aggregated solutions – significant main effects of professional stakeholder group for strategies A1, A2, A3, A4, A5, A8, and A10 (8.8≤χ2(3)≤24.1; p < 0.04) were mostly driven by the more positive ratings of cancer patient organisation representatives compared to one or more professional groups (p < 0.05; Table 3). The exception is strategy A8 related to EU funding for genetic counselling training, which genetic counsellors rated as being significantly more Urgent than clinical/medical geneticists (p < 0.04).
No single strategy received priority votes from a majority of Delphi participants. However, five strategies received a clear plurality of priority rankings, ranked as Priority #1, #2 or #3 by at least 15 participants (weighted Borda count range: 32 – 67): P1, P2, P4, P7, P9. All other strategies received Priority rankings from 8 or fewer participants (weighted Borda count ≤ 15).
Phase III
Priority rankings for the five strategies receiving a plurality of Phase II priority rankings are displayed in Fig. 3. A statistically significant difference in priority rankings between these five strategies was observed (main effect: χ2(4) = 18.611, p < 0.001). Post-hoc tests showed that strategy P1 (EU genetic counsellor recognition) received significantly higher weighted rankings than strategies P2 (EU-wide genetic counsellor education/accreditation; p = 0.03), P4 (full reimbursement when guidelines are met; p = 0.02), and P7 (genetics continuing education for oncologists; p = 0.01). No other post-hoc differences between priority rankings of strategies were found (p > 0.34).
$ - significant differences in prioritisation between professional groups (p < 0.05). Weighted Borda count totals corresponding to vote totals for each strategy are as follows (maximum possible score = 204): P1 = 119; P2 = 69; P4 = 64; P7 = 62; P9 = 88.
Significant differences in priority rankings between professional stakeholder groups were present for strategies P1, P2, P7, and P9 (genetics expertise in oncology guideline development)(13.1≤χ2(3)≤19.2; p < 0.01). Post hoc tests revealed that this significant main effect was driven for all four strategies by significant differences in the priority rankings of genetic counsellors compared to other professional groups: higher prioritisation of strategy P1 compared to all other groups (p = 0.02); higher prioritisation of strategy P2 compared to geneticist and oncology physicians (p = 0.03); and a lower prioritisation of strategies P7 and P9 compared to geneticist physicians and patient organisation representatives (p < 0.04).
Discussion
Integrating the perspectives of members of four key stakeholder groups including representation from all 27 EU Member States, this Delphi survey identified five priority European strategies for addressing common barriers to genetic counselling in cancer identified in preliminary work [3, 10]. All five priority strategies were judged by consensus to be both Important and Urgent and received a clear plurality of priority rankings amongst 19 evaluated strategies. Of these five priority strategies, strategy P1 – EU-wide recognition of genetic counsellors as allied health professionals and appropriately qualified experts to deliver genetic counselling – was ranked as highest priority, albeit with notable differences in its relative prioritisation across stakeholder groups. Similar differences in relative prioritisation, as well as Importance and Urgency ratings, were noted for most priority strategies, with implications for future implementation. Individual discussion of each priority strategy follows in order of relative prioritisation (Phase III Borda count) below, with a focus on implementation pathways and potential hurdles.
Strategy P1 – EU-wide recognition of genetic counsellors as allied health professionals and appropriately qualified experts to deliver genetic counselling (Moderate Feasibility: 6.6/9 in Phase I)
Recognition of genetic counsellors throughout EU Member States has been proposed previously, with work in this area currently underway within the European Board of Medical Genetics, Genetic Nurse and Genetic Counsellor Branch [17]. Genetic counsellors, typically Master’s degree qualified allied health professionals, have been demonstrated to deliver quality genetic counselling services in multiple clinical scenarios, and are recognised and well-integrated into health systems in France and several countries outside the EU, e.g. Australia, the United Kingdom, and the United States [18]. An estimated 570 trained genetic counsellors currently exist in Europe [12]. However, their integration into the delivery of genetic services in the EU is limited by a lack of recognition of genetic counselling as an independent health profession in all Member States except France [3]. An estimated 340 of these 570 genetic counsellors (60%) are present in 14 EU countries outside France; recognition thus presents a first step towards addressing immediate genetic counselling workforce capacity barriers [12].
Implementing genetic counsellor recognition across the EU will require broad support from, in particular, clinical/medical geneticists – recent positive experiences in Iceland and Malaysia and challenges in Malta related to professional genetic counsellor recognition within the health systems have noted that the support, or lack thereof, of geneticist physicians has been decisive [19, 20]. Additional support from cancer patient organisations can provide further momentum.
Significant differences in stakeholder perspectives in this Delphi survey indicate that additional dialogues between clinical/medical geneticists and genetic counsellors are needed to build a broad mandate for timely genetic counsellor recognition; considering only the responses of clinical/medical geneticists, Importance (7.2/9) but not Urgency (6.9/9) ratings would have met consensus criteria. The reasons underpinning these discrepant perspectives were not captured in this Delphi study. However, and encouragingly, dialogues between genetic counsellors and clinical/medical geneticists have recently advanced genetic counsellor recognition in German-speaking countries [21]. The European Board of Medical Genetics and European Society of Human Genetics present appropriate forums for such dialogue on a European level, as both organisations already integrate both clinical/medical geneticists and genetic counsellors into their membership. The upcoming EU Joint Action on Personalised Cancer Medicine includes support for such dialogues within its project objectives. These upcoming European dialogues can then provide context and momentum for national discussions critical to furthering recognition efforts in relevant Member States.
Strategy P9 – Mandatory inclusion of genetics expertise in the creation and revision of national/EU clinical oncology guidelines (High Feasibility: 7.6/9 in Phase I)
Including a genetics expert in the process of creating and revising national and European oncology guidelines presents a straightforward solution rated as being highly Important, Urgent, and Feasible across stakeholder groups. Several Delphi survey participants noted that genetics expertise is already included in national oncology guideline processes in their Member States. An EU-wide mandate would expand these practices to help facilitate equitable integration of genetics into oncology practice across Member States.
Immediate next steps towards implementation are planned within the upcoming EU Joint Action on Personalised Cancer Medicine – an initial mapping of the present inclusion of genetics expertise in the creation and revision of national oncology guidelines across Member States, followed by direct dialogue with the relevant national health authorities and/or oncology societies to facilitate procedural updates and explore the possibilities for such a ‘hard’ or ‘soft’ EU mandate.
Strategy P2 – Establish an EU system of registration, education and accreditation for genetic counsellors (allied health professional) with legal weight in Member States (Moderate Feasibility: 6.3/9 in Phase I)
Our preliminary work noted concerns related to the high burden of establishing national registration/accreditation and education frameworks to serve low numbers of genetic counsellors within a given Member State [3]. While understandable, this also leads to a genetic workforce ‘chicken and egg’ problem, wherein low numbers of genetic counsellors preclude the establishment of national registration/accreditation/education frameworks, but the absence of these frameworks inhibits the training and registration of greater numbers of genetic counsellors.
Accordingly, a distributed system for ensuring adequate training and practice standards across Member States is envisioned as a means of supporting increased integration of genetic counsellors without disproportionate national burdens. Existing EU regulations provide guidance on how such a distributed system could function. Directly establishing a supra-national mandatory European system for the education, registration, and accreditation of genetic counsellors is likely a legal non-starter, given that primary responsibility for organising and delivering medical care and health services is explicitly the domain of Member States [22].
However, once the genetic counselling profession is officially recognised across multiple Member States (i.e Strategy P1 as an essential first step), the existing Directive 2005/36/EU would permit genetic counselling qualifications to be immediately transferred across these Member States through the ‘general system’ for recognition of qualifications. This ability to transfer qualifications across borders would de facto support a distributed system for genetic counsellor education; such a system will be particularly critical to support the initial growth of the recognised profession across the EU, as Masters programs accredited by the European Board of Medical Genetics are presently only available in France, Spain, Portugal, Italy, and Austria. A collaborative Nordic Masters program is also planned to undergo accreditation procedures and start accepting students in 2027. Key to ensuring the smooth operation of a distributed education system will be the continued standardisation of Masters of Genetic Counselling qualifications – e.g. according to the core curriculum established by the European Board of Medical Genetics, Genetic Nurse and Genetic Counsellor Branch [23] – to simplify mutual recognition procedures. National genetic counsellor registration procedures should be similarly standardised to support reciprocal arrangements between Member States and international mobility; here the voluntary European registration system supported by the Genetic Nurse and Genetic Counsellor Branch also already provides a framework [11].
Should growth of the recognised genetic counsellor profession follow strong patterns seen in Australia and the United States [12], the development of additional Masters programs will rapidly become more financially viable for universities across the EU. In this case, care will need to be taken to ensure sufficient educational opportunities are developed in Eastern European countries to support equitable expertise distribution across Member States. Additionally, proportional increases in available genetic counselling jobs will be required across Member States to realise the benefits of increased European genetic counselling training capabilities within the healthcare workforce. Over the longer-term, genetic counsellors could aim to eventually join streamlined ‘automatic recognition’ procedures under EU Directive 2005/36/EU currently available to seven established ‘sectoral professions’ to further promote mobility: medical doctors, nurses, midwives, dental practitioners, pharmacists, architects, and veterinary surgeons [24].
Strategy P4 – Mandate full reimbursement of genetic counselling when conducted according to national or European guidelines (Moderate Feasibility: 6.2/9 in Phase I)
Our preliminary work noted that reimbursement of/payment for genetic counselling services was at least partly limited for health practitioners and/or patients in 13 of 27 Member States broadly distributed across Southern, Eastern, and Western European regions [3]. This is despite the widespread inclusion of genetic counselling in clinical practice guidelines and national legislation – in several Member States, incomplete reimbursement for counselling persists despite the existence of legal mandates for pre- and/or post-test genetic counselling [3]. Such reimbursement challenges are hardly new, as a European Society of Human Genetics Public and Professional Policy Committee workshop in the year 2000 also emphasised ‘financial coverage’ as being a key issue impacting genetic service provision [25].
Prior study suggests that the unclear economic utility of genetic counselling is likely at least partly responsible for ongoing reimbursement challenges [26], with further work needed to ensure the universal inclusion of genetic counselling within emerging personalised medicine reimbursement models [27]. Accordingly, while a European reimbursement mandate promises to address a key genetic counselling barrier, the broad uptake of such a mandate across Member States would be likely predicated on clear demonstrations of long-term economic utility and sustainability. A 2022 review of economic evaluations of genomic/genetic tests and related genetic counselling notes that the evidence base upon which economic analyses can be conducted is presently ‘under-developed’ [28]. A planned multi-country cost-effectiveness analysis of genetic counselling in cancer prevention and care contexts in the upcoming EU Joint Action on Personalised Cancer Medicine will provide a first step towards closing this evidence gap.
Strategy P7 – Mandatory education for clinical oncologists, continuing and as a part of fellowship training, regarding screening for cancer predisposition syndromes (e.g. when to refer, how to screen) (High Feasibility: 7.1/9 in Phase I)
Global recommendations for oncology fellowship curricula from the European Society for Medical Oncology (ESMO) and American Society for Clinical Oncology (ASCO) have noted that genetics competencies are required to deliver best-practice medical oncology since 2016 [29]. Further, the most recent (2023) recommendations include an entire section dedicated to competencies related to genetic testing and counselling [30]. However, both our prior work and the results of this Delphi survey indicate that present educational programs for medical oncologists do not sufficiently support the acquisition of these competencies [3].
Tailored education specific to the needs of oncologists – i.e. as compared to generalised training in cancer predisposition syndromes – best supports genetics competency acquisition and integration [31]. However, the availability of such tailored education is presently sparse [32], including 2-day courses delivered by the European Society for Paediatric Oncology, as well as ESMO in conjunction with the European Reference Network on Genetic Tumour Risk Syndromes (ERN GENTURIS).
Further, the most recent available data indicate that ESMO/ASCO curricular recommendations are presently only fully implemented in fellowship training in four Member States, with these recommendations noted as being ‘adapted’ or ‘partially integrated’ in a further 11 Member States [30]. Whether and how genetics information is included in fellowship training in circumstances were ESMO/ASCO recommendations are adapted, partially integrated, or not observed has not been chronicled.
Recently started and upcoming EU projects present first steps towards enhancing the availability of educational opportunities: the Joint Action on Networks of Expertise in Cancer (‘JANE-2’); Joint Action EUnetCCC (Comprehensive Cancer Centers); and Joint Action on Personalised Cancer Medicine. These Joint Actions plan to map the current inclusion of genetics in core education, fellowship training, and continuing education of relevant physicians and health professionals, including but not limited to oncologists; genetic literacy needs are, of course, not constrained to oncologists but also include the full breadth of practitioners involved in cancer care pathways, e.g., surgeons, gynaecologists, general practitioners, nurses. Based on identified gaps, new educational content and/or courses will be developed for inclusion in general medical education, fellowship/residency, and continuing education programmes, in collaboration with European and national stakeholders.
Limitations
The robust size and multistakeholder composition of our Delphi survey cohort is one of its strengths – our cohort of 77 participants is in line with recommended sample sizes for ‘highly replicable’ results from multistakeholder Delphi surveys [33]. However, the potential impact of an unequal distribution of participants across stakeholder groups on results should be noted, as well as geographic asymmetries. While unsurprising given the topic area, the relative over-representation of clinical/medical geneticists compared, in particular, to clinical/medical oncologists emphasises the need for further interdisciplinary dialogue to guide the broad implementation and, where necessary, adaptation of identified strategies. Such dialogue can help account for perspectives which may not have been represented in this Delphi survey. Similar sensitivities should also be integrated into interdisciplinary dialogues related to the implementation of priority strategies at the national level – while all Member States were represented in our Delphi cohort, not all stakeholder groups were represented from each Member State.
Summary and conclusions
The five priority strategies identified present a clear strategic roadmap to address common European barriers to genetic counselling in cancer care. Upcoming and recently started European projects provide opportunities to push these priorities toward implementation, in collaboration with national and European cancer patient organisations and genetics and oncology societies.
Data availability
The de-identified data presented in this manuscript are available upon reasonable request.
References
European Commission. Europe’s Beating Cancer Plan. https://health.ec.europa.eu/system/files/2022-02/eu_cancer-plan_en_0.pdf; 2021.
Ding L, Bailey MH, Porta-Pardo E, Thorsson V, Colaprico A, Bertrand D, et al. Perspective on oncogenic processes at the end of the beginning of cancer genomics. Cell. 2018;173:305–20.e10.
McCrary JM, Van Valckenborgh E, Poirel HA, de Putter R, van Rooij J, Horgan D, et al. Genetic counselling legislation and practice in cancer in EU Member States. Eur J Public Health. 2024:34;ckae093.
Resta R, Biesecker BB, Bennett RL, Blum S, Estabrooks Hahn S, Strecker MN, et al. A new definition of genetic counseling: National Society of Genetic Counselors’ task force report. J Genet Counsel. 2006;15:77–83.
Rantanen E, Hietala M, Kristoffersson U, Nippert I, Schmidtke J, Sequeiros J, et al. What is ideal genetic counselling? A survey of current international guidelines. Eur J Hum Genet. 2008;16:445–52.
Bensend TA, Veach PM, Niendorf KB. What’s the harm? Genetic counselor perceptions of adverse effects of genetics service provision by non-genetics professionals. J Genet Counsel. 2014;23:48–63.
Dragojlovic N, Kopac N, Borle K, Tandun R, Salmasi S, Ellis U, et al. Utilization and uptake of clinical genetics services in high-income countries: a scoping review. Health Policy. 2021;125:877–87.
Abacan M, Alsubaie L, Barlow-Stewart K, Caanen B, Cordier C, Courtney E, et al. The global state of the genetic counseling profession. Eur J Hum Genet. 2019;27:183–97.
Dragojlovic N, Borle K, Kopac N, Ellis U, Birch P, Adam S, et al. The composition and capacity of the clinical genetics workforce in high-income countries: a scoping review. Genet Med. 2020;22:1437–49.
McCrary JM, Bergmann AK CAN.HEAL Deliverable 5.4: Regulations and recommendations related to remote and face-to-face genetic counselling quality, access, and sustainability. https://doi.org/10.5281/zenodo.14893028; 2024.
Paneque M, Moldovan R, Cordier C, Serra-Juhé C, Feroce I, Lambert D, et al. Development of a registration system for genetic counsellors and nurses in health-care services in Europe. Eur J Hum Genet. 2016;24:312–4.
Ormond KE, Abad PJ, MacLeod R, Nishigaki M, Wessels T-M The global status of genetic counselors in 2023: What has changed in the past 5 years? Genet Med Open. 2024:2;101887.
Khodyakov D, Grant S, Kroger J, Bauman M RAND methodological guidance for conducting and critically appraising Delphi panels: RAND; 2023.
Boel A, Navarro-Compán V, Landewé R, van der Heijde D. Two different invitation approaches for consecutive rounds of a Delphi survey led to comparable final outcome. J Clin Epidemiol. 2021;129:31–9.
Saari DG. Selecting a voting method: the case for the Borda count. Const Polit Econ. 2023;34:357–66.
Publications Office of the European Union. EuroVoc 2025 [Available from: https://eur-lex.europa.eu/browse/eurovoc.html?params=72,7206,912#arrow_912.
Paneque M, Liehr T, Serra Juhé C, Moog U, Melegh B, Carreira I. The need for recognition of core professional groups in genetics healthcare services in Europe. Eur J Hum Genet. 2022;30:639–40.
Skirton H, Cordier C, Ingvoldstad C, Taris N, Benjamin C. The role of the genetic counsellor: a systematic review of research evidence. Eur J Hum Genet. 2015;23:452–8.
Stefansdottir V, Thorolfsdottir E, Hognason H, editors. The (long) Road to Licensure. 5th World Congress on Genetic Counselling; 2025; Hinxton, UK.
Cham B, editor The state of genetic counselling in Asia. 5th World Congress on Genetic Counselling; 2025; Hinxton, UK.
Schwaninger G, Heidemann S, Rudnik-Schöneborn S, Zschocke J. The current state of the genetic counselor profession in the German-speaking countries. Genet Med Open. 2024;2:101849.
Official Journal of the European Communities. Treaty on European Union. 1992.
Skirton H, Barnoy S, Ingvoldstad C, Van Kessel I, Patch C, O’connor A, et al. A Delphi study to determine the European core curriculum for Master programmes in genetic counselling. Eur J Hum Genet. 2013;21:1060–6.
Directive 2005/36/EC, 2005.
Godard B, Kääriäinen H, Kristoffersson U, Tranebjaerg L, Coviello D, Aymé S. Provision of genetic services in Europe: current practices and issues. Eur J Hum Genet. 2003;11:S13–S48.
Payne K, Eden M. Measuring the economic value of genetic counselling. Eur J Med Genet. 2019;62:385–9.
Koleva-Kolarova R, Buchanan J, Vellekoop H, Huygens S, Versteegh M, Mölken MR-v, et al. Financing and reimbursement models for personalised medicine: a systematic review to identify current models and future options. Appl Health Econ Health Policy. 2022;20:501–24.
Bouttell J, Heggie R, Oien K, Romaniuk A, VanSteenhouse H, von Delft S, et al. Economic evaluation of genomic/genetic tests: a review and future directions. Int J Technol Assess Health Care. 2022;38:e67.
Dittrich C, Kosty M, Jezdic S, Pyle D, Berardi R, Bergh J, et al. ESMO/ASCO recommendations for a global curriculum in medical oncology edition 2016. ESMO Open. 2016;1:e000097.
Cufer T, Kosty M, Osterlund P, Jezdic S, Pyle D, Awada A, et al. Current landscape of ESMO/ASCO Global Curriculum adoption and medical oncology recognition: a global survey. ESMO Open. 2021;6:100219.
Rahman B, McEwen A, Phillips JL, Tucker K, Goldstein D, Jacobs C. Genetic and genomic learning needs of oncologists and oncology nurses in the era of precision medicine: A scoping review. Personalized Med. 2022;19:139–53.
Hoxhaj I, Beccia F, Calabrò GE, Boccia S. A web screening on training initiatives in cancer genomics for healthcare professionals. Genes. 2022;13:430.
Manyara AM, Purvis A, Ciani O, Collins GS, Taylor RS. Sample size in multistakeholder Delphi surveys: at what minimum sample size do replicability of results stabilize?. J Clin Epidemiol. 2024;174:111485.
Acknowledgements
We would like to thank all Delphi survey participants for their valuable contributions to this project, including but certainly not limited to the 11 who requested to remain anonymous. We would also like to thank Prof. Kelly Ormond for her feedback on the draft version of the Delphi survey.
Funding
The CAN.HEAL Consortium is funded by the European Union (Grant number 101080009). Views and opinions expressed are, however, those of the author(s) only and do not necessarily reflect those of the European Union or HaDEA. Neither the European Union nor the granting authority can be held responsible for them. Ramūnas Janavičius would like to also acknowledge support from Joint Action PreventNCD (EU4H-2022-JA-02). Open Access funding enabled and organized by Projekt DEAL.
Author information
Authors and Affiliations
Contributions
Conceptualisation: McCrary, Bergmann. Formal Analysis: McCrary, Bergmann. Funding acquisition: Bergmann, Colas, Dalmas, Genuardi, Horgan, Krajc, Mazzarella, Poirel, Ripperger, Stamatopoulos, Van Den Bulcke, Van Valckenborgh. Data collection/analysis: All authors. Methodology: McCrary, Ripperger, Ronez, Bergmann. Project administration/supervision: McCrary, Bergmann. Writing – original draft: McCrary, Bergmann. Writing – review & editing: All authors
Corresponding author
Ethics declarations
Competing interests
J. Matt McCrary, Els Van Valckenborgh, Denis Horgan, Chrystelle Colas, Miriam Dalmas, Maurizio Genuardi, Mateja Krajc, Hélène A. Poirel, Kostas Stamatopoulos, Joelle Ronez, Tim Ripperger, Marc Van Den Bulcke, and Anke Katharina Bergmann are members of the CAN.HEAL consortium, which supported this project (funded by the European Union – grant number 101080009). Evandro de Azambuja reports grants paid to his institution from Roche/GNE, Astra-Zeneca, GSK/Novartis, Gilead Sciences, and Seagen/Pfizer, honoraria paid to his institution from Roche/GNE, Novartis, SeaGen, Zodiac, Libbs, Pierre Fabre, Lilly, Astra-Zeneca, MSD, and Gilead Sciences, travel support paid to himself from Astra-Zeneca and Gilead Sciences, and his participation on a Data Safety Monitoring Board/Advisory Board of Roche and Lilly. Robin de Putter reports grants from AstraZeneca to his institution. Kostas Stamatopoulos reports grants paid to his institution from Johnson & Johnson and AstraZeneca, consulting fees paid to himself from AbbVie and Johnson & Johnson, honoraria paid to himself from AbbVie, Johnson& Johnson, AstraZeneca and BeiGene, and travel support paid to himself from Johnson & Johnson and AstraZeneca.
Ethical approval
This is a collaborative project in which all contributors (i.e. Delphi participants) were invited to review and endorse the final manuscript as named co-authors. Accordingly, ethical approval was not required.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
McCrary, J.M., Van Valckenborgh, E., Horgan, D. et al. Priority European strategies for sustainable access to high-quality genetic counselling in cancer: A Delphi study. Eur J Hum Genet (2026). https://doi.org/10.1038/s41431-026-02015-y
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41431-026-02015-y