Abstract
Objectives
To review the trend of microbial isolates for postoperative endophthalmitis (POE) in the United Kingdom (UK) and determine the sensitivity to current empirical intravitreal antibiotic treatment.
Methods
We conducted a long term multicentre consecutive case review of POE across 3 geographically distant tertiary eye centres in the UK: Sunderland Eye Infirmary (2000–2022), Oxford Eye Hospital (2016–2022), and Southampton General Hospital (2016–2022). Data on the microbial samples taken and results including sensitivities to antibiotics agents given were collected. Poisson regression was used to analyse microbial trends and outcomes were considered statistically significant at a level of p < 0.05.
Results
179 consecutive eyes of 177 patients with POE met our inclusion criteria. The most common primary procedure was phacoemulsification and IOL insertion followed by intravitreal injections. 104 (58.1%) were culture positive and most were Gram-positive bacteria (85, 81.7%). The microbial trend consistently showed Staphylococcus epidermidis and unspecified coagulase-negative Staphylococci to be the most prevalent pathogens. Poisson regression showed no statistically significant change in any of the bacterial isolates over our study period. Antibiotic sensitivity data was available for 74% of the culture positive samples (77/104). All Gram-positive bacteria (68/68, 100%) and most (8/9, 88.9%) Gram-negative bacteria were sensitive to the empirical antibiotics (Vancomycin and Ceftazidime/Amikacin) given at presentation.
Conclusions
Most of the bacterial isolates causing POE in the UK are Gram-positive bacteria, and the trend has remained stable over more than two decades. Current empirical treatment with intravitreal Vancomycin and Ceftazidime/Amikacin provides effective broad coverage for the vast majority of cases.
Similar content being viewed by others
Introduction
Postoperative endophthalmitis (POE) is a rare sight-threatening complication that may occur following any intraocular procedure. It is characterised by severe inflammation of the inner layers of the eye secondary to infection. The severity and clinical course of POE depend on the type and virulence of inoculated pathogens, as well as the time to diagnosis and treatment [1, 2].
Over the last 20 years, there has been a reduction in the incidence of POE following cataract surgery, thought to be associated with the consistent use of antiseptics and the introduction of intracameral antibiotics [3,4,5,6]. However, at the same time, with the success of anti-vascular endothelial growth factor (VEGF) treatments for a range of common retinal diseases, there has been a rapid increase in the number of intravitreal injections delivered over the past two decades [7, 8]. Whilst the overall incidence of POE has remained low, the sheer increase in number of injections performed has made it one of the major causes of POE [9, 10]. Reassuringly, a recent study reported a substantial reduction in the incidence of POE following any intraocular procedure by nearly 75% over the past two decades [11].
The current management of POE is mainly informed by the landmark Endophthalmitis Vitrectomy Study (EVS) [12], reinforced by guidelines from learned societies [13]. Vitreous and/or aqueous samples were first obtained for microbiologic testing, followed by empirical intravitreal injection of antibiotics. At present, the most commonly used antibiotic combination is Vancomycin and Ceftazidime or Amikacin [14]. However, there is growing concern regarding increasing bacterial resistance to antibiotics globally [15, 16] and this might also impact on the optimal management of POE.
In this study, we aimed to review the trend of microbial isolates for POE and determine the sensitivity to current empirical intravitreal antibiotic treatment using data from 3 large tertiary ophthalmic units in the United Kingdom (UK).
Methods
This was a multicentre study involving 3 geographically distant tertiary eye centres in the UK: Sunderland Eye Infirmary, Oxford Eye Hospital and Southampton General Hospital. The study was classed as a continuous departmental audit and surveillance as part of clinical governance requirements, and was therefore exempt from requiring formal ethics approval.
A database was established in January 2000 in Sunderland Eye Infirmary to prospectively record all patients with endophthalmitis presenting to the unit. Data was collected from this database up to 31st July 2022. In the other two units (Oxford Eye Hospital and Southampton General Hospital), retrospective review of all patients presenting with endophthalmitis were conducted from 1st January 2016 to 31st July 2022 using departmental electronic medical record systems or paper records.
We included all consecutive patients presenting with acute-onset POE presenting within 6 weeks of any primary intraocular procedure. The initial diagnosis was made clinically, based on signs and symptoms associated with endophthalmitis such as pain, redness, loss of vision, hypopyon, fibrin and vitritis, supported by adjunctive B-scan ultrasonography. Patients with delayed-onset endophthalmitis (more than 6 weeks after primary intraocular procedure), endogenous endophthalmitis and those associated with trauma were excluded [10].
Data collected included patient demographics, details of the primary ophthalmic procedure, timing of presentation, microbiological samples taken and results including sensitivities to antimicrobial agents given, initial management, and visual acuity (VA) at presentation and final follow-up (at least 3 months following acute-onset POE).
Across all 3 units, initial management of POE was either a ‘tap and inject’ procedure (obtaining samples of intraocular fluids followed by intravitreal antibiotic injection) or patients proceeding directly to pars plana vitrectomy with intravitreal antibiotic cover. For tap and inject, aqueous samples were obtained through peripheral clear cornea paracentesis using 30-gauge needle attached to a 1 ml plastic sterile disposable tuberculin syringe inserted parallel to the iris plane. Vitreous samples were taken via vitreous taps with needle aspiration using a 23- or 25-gauge needle, or mechanically via vitreous cutter, depending on local protocols. The intravitreal antibiotics of choice were Vancomycin (1–2 mg/0.1 mL) and Ceftazidime (2 mg/0.1 mL). Intravitreal Amikacin (0.4 mg/0.1 mL) was used in place of Ceftazidime in those allergic to penicillin or cephalosporins, depending on local treatment protocols. Choice of topical antibiotics, steroids, cycloplegics, and oral antibiotics and steroids were prescribed at the discretion of the treating clinicians.
All data collected were anonymised and recorded in a standardised Microsoft Excel spreadsheet (Microsoft Corporation, Redmond, WA). VA values were converted to the appropriate logarithm of the minimum angle of resolution (logMAR) equivalent for analyses. VA of counting fingers (CF), hand movements (HM), perception of light (PL), and no perception of light (NPL) were assigned logMAR values of 2.1, 2.4, 2.7, and 3.0 respectively, as per the National Ophthalmology Database audit [17].
Descriptive data were presented using tabular and graphical summaries. Statistical analyses were performed using IBM SPSS Statistics, version 29 (IBM Corporation, Armonk, NY, USA). Outcomes were considered statistically significant at a level of p < 0.05. Quantitative variables were presented as frequencies with percentages. Non-parametric continuous variables were reported as median, interquartile range (IQR) and range. The Chi-squared test was used to evaluate associations between categorical variables. We expressed results using odds ratios (OR) and their 95% confidence intervals (CI). Multivariate analysis by stepwise logistic regression was used to examine variables associated with a poor visual outcome (defined as logMAR 1.0 or worse), and Poisson regression was used to analyse microbial trends.
Results
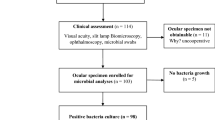
We identified a total of 179 eyes of 177 patients with POE that met our inclusion criteria (106 eyes of 104 patients from Sunderland Eye Infirmary, 35 from Oxford Eye Hospital and 38 from Southampton General Hospital), following exclusion of 6 cases of delayed-onset endophthalmitis, 5 trauma-related endophthalmitis and 15 endogenous endophthalmitis.
Demographic details of patients in our cohort are summarised in Table 1. The median age was 76.8 years (range 27–94), with slight female preponderance (53.7%). The median presenting VA was 2.40 logMAR (or HM).
The primary ophthalmic intervention is shown in Table 2. The most common primary procedure was phacoemulsification and IOL insertion comprising 89 (49.7%) of cases followed by intravitreal injections (67, 37.4%). The aetiology of POE over the course of the study is illustrated in Supplementary Fig. 1.
All patients underwent aqueous and/or vitreous sampling as part of their initial management before intravitreal injection of antibiotics. The majority (161/179, 89.9%) underwent a tap and inject procedure at presentation, while the remainder (18/179, 10.1%) underwent initial pars plana vitrectomy. The microbiological sampling technique used is shown in Supplementary Table 1. Treating clinicians have aimed to obtain vitreous samples from all eyes. However, this was not possible in 10 eyes due to reasons such as dry vitreous tap or silicone oil-filled vitreous cavity; consequently, only anterior chamber (AC) taps were successfully performed in these cases. All samples were sent for microbiological testing which included microscopy and gram staining as well as subsequent culture and sensitivities of identified microbes.
Of all the samples sent, 104 (58.1%) were culture positive. The majority of the cultures were of a single type of bacterium (94/104; 90.4%) and the remaining 10 (9.6%) grew more than one type. Most of the culture positive cases were Gram-positive bacteria (85, 81.7%). Subgroup analysis showed no statistically significant difference across the three centres (p = 0.87; Supplementary Table 2). Table 3 outlines the microbiological profile of the culture specimens.
The microbial trend over more than two decades consistently showed Staphylococcus epidermidis and unspecified coagulase-negative Staphylococci to be the most prevalent pathogens (Fig. 1). Poisson regression showed no statistically significant change in any of the bacterial isolates over our study period. (Supplementary Table 3).
Area plot showing the trends of pathogens causing acute postoperative endophthalmitis (POE) in 104 culture positive cases.
The microbial yield of the aqueous samples was significantly lower at 32.4% compared to 55.6% in vitreous samples (p < 0.05). Adding an aqueous sample on top of the vitreous sample marginally improved the microbial yield to 58.1% (p = 0.64), (Supplementary Table 4). Antibiotic sensitivity data was not available for a quarter of the culture positive samples (27/104, 26.0%). For the remaining 77 samples, all Gram-positive bacteria (68/68, 100%) and most (8/9, 88.9%) Gram-negative bacteria were sensitive to the empirical antibiotics given at presentation. One eye grew Gram-negative Serratia marcescens which was resistant to these antibiotics and proceeded to vitrectomy. Nonetheless, the eye developed recurrent retinal detachments requiring multiple vitrectomies and subsequently became phthisical with NPL vision. Overall, 98.7% (76/77) of our bacterial isolates were susceptible to Vancomycin and Ceftazidime/Amikacin.
Two patients in our series had bilateral endophthalmitis. One had same day bilateral intravitreal anti-VEGF injection for diabetic macular oedema. Samples from both eyes had polymicrobial growth of Staphylococcus epidermidis, Staphylococcus hominis and Streptococcus mitis. Another patient had sequential cataract surgery 5 months apart and unfortunately developed POE in both eyes. The first eye grew Streptococcus pneumoniae and the second eye was culture negative. Nasolacrimal duct obstruction was identified as a potential risk factor for the patient developing bilateral sequential POE.
Overall, median VA at final follow up was 0.8 logMAR (Snellen equivalent 6/38), with 40.1% eyes having 1.0 logMAR (Snellen equivalent 6/60) vision or worse. Univariable analysis demonstrated that poor presenting visual acuity (p < 0.001) and positive microbiological culture (p = 0.002) were significantly associated with a poor visual outcome (Supplementary Table 5). When we performed multivariable logistic regression, we found that presenting VA (OR 4.69, 95% CI 2.13–10.33) was the only statistically significant predictor for a poor visual outcome (p < 0.001), after adjusting for other variables including age, gender, primary ophthalmic procedure, days since primary procedure, microbiological yield (culture positive), Gram stain results, presence of multiple bacterial strains and antibiotics susceptibility (Supplementary Table 6).
Discussion
Antimicrobial resistance (AMR) has been identified as one of the most pressing global challenges with the World Health Organisation (WHO) declaring AMR as one of the top 10 global public health threats [18]. This is also relevant in management of endophthalmitis as there are limited antibiotic alternatives to Vancomycin and Ceftazidime known to be safe for intraocular use [19]. In our study of 77 culture positive eyes with antibiotic sensitivity data, all Gram-positive bacteria (68/68, 100%) were sensitive to Vancomycin and 8 of the 9 (88.9%) Gram-negative bacteria were sensitive to Ceftazidime/Amikacin. Chen et al. [20] recently summarised the published literature of bacterial isolates and their in-vitro antibiotics susceptibility globally. While Vancomycin has excellent coverage for Gram-positive bacteria with around 96–100% susceptibility, Ceftazidime and Amikacin were reported to have much lower susceptibility, ranging from 38 to 100% and 64 to 100% respectively for Gram-negative bacteria. Though our study showed excellent antibiotic susceptibility for bacterial isolates in our cohort, there is a need for continuous monitoring of antibiotic resistance patterns and explore other potential antibiotic alternatives to treat POE.
The management of endophthalmitis relies on the initiation of appropriate empirical antibiotic therapy that targets the most common causative organisms. It is therefore important to understand the regional microbial spectrum as it has been recognised that there is geographical variation in the microbial profile of POE [21]. In our study, the spectrum of identified microbes showed that 81.7% of culture positive cases were due to Gram-positive bacteria, with coagulase-negative Staphylococci (mostly Staphylococcus epidermidis) being the most prevalent species causing POE. This is largely similar to other USA, European and Australian studies. The EVS study [12] conducted in the USA in 1995 showed that 94% of the bacterial isolates were Gram-positive with 70% of them being coagulase-negative Staphylococci. Similarly, in a multicentre European study conducted by the European Society of Cataract & Refractive Surgeons (ESCRS) in 2007, all culture-positive endophthalmitis cases were due to Gram-positive bacteria, with 40% being Staphylococcus epidermidis [22]. Moloney et al. in 2014 reported on their 15-year results of culture-proven endophthalmitis in Australia where around 85% were Gram-positive organisms [23].
In contrast, a review over two decades in India has shown that Gram-positive organisms accounted for a much lower proportion for POE, varying from 37.6% to 64.8% [24]. Another study in North-East India also reported that only around 46% of their exogenous endophthalmitis cases were caused by Gram-positive organisms, and the majority (27%) were in fact due to Gram-negative Pseudomonas species [25]. The POE isolates and their antibiotic sensitivity from different countries/regions is summarised in Table 4.
In terms of microbial trends, we found that the microbial profile of culture positive POE cases has remained largely stable within the three geographically distant UK centres, with no major shifts in bacterial isolates. Our findings align with similar studies conducted internationally. A 10-year review in the USA [26] and a 25-year study in India [27] both reported stable microbial profiles over their respective study periods. This suggests a potential consistency in the types of bacteria associated with POE within individual geographical regions.
Our study showed that phacoemulsification and IOL implant accounted for nearly half of the POE in our cohort (49.7%), followed by intravitreal injections (37.4%). Nevertheless, there has been a change in trend with intravitreal injections overtaking phacoemulsification as the leading cause of POE in recent years (Supplementary Fig. 1), agreeing with other published reports [28, 29].
Two patients in our series had bilateral endophthalmitis. One patient was known to be diabetic and another had nasolacrimal duct obstruction, both of which were recognised risk factors for POE [6]. Previous study also suggested a potential underlying genetic predisposition increasing individual patients’ risks of developing endophthalmitis [30].
We found that our microbial yield from vitreous samples was significantly higher than aqueous samples. This is consistent with multiple studies highlighting the superiority of vitreous over aqueous fluid in improving microbiological yield [31,32,33]. In our cohort, we managed to obtain vitreous samples from the majority of the cases (169/179, 94.4%). Overall, 58.1% of our POE cohort had positive microbial cultures, similar to previous published UK studies [31, 34].
The issue of a high culture-negative rate could potentially be addressed with the advancement in diagnostic technologies, such as polymerase chain reaction (PCR) testing, metagenomic and whole genome sequencing (WGS) [35, 36]. These methods allow for direct, large-scale parallel sequencing of single DNA molecules, providing detailed identification of pathogens from intraocular fluid samples. This enhanced capability allows for a timely and accurate diagnosis of endophthalmitis, facilitating more targeted treatment strategies. Lee et al. [36] in their prospective cohort study demonstrated that WGS has helped to identify potential pathogens in 33% of their culture-negative cases. It was also reported that higher bacterial DNA load other than Staphylococcus epidermidis is associated with worse outcomes, suggesting this could function as a prognostic marker. Despite the advantages, PCR assays can be limited by their inability to distinguish contaminants from true infections, leading to ambiguous results [37]. A recent review also found that these techniques are not always readily available, require advanced bioinformatic analysis and are expensive [38]. Further research is therefore needed to ascertain the optimal approach in incorporating these new technologies to complement the diagnostic process.
Although obtaining intraocular fluid samples for microbiologic testing has conventionally been a standard part of initial management of POE, the necessity for sampling has recently been contended with studies showing culture results having limited effect on subsequent clinical management [39, 40]. We intend to explore this interesting controversy in more detail in a separate study.
Our study has several limitations inherent to retrospective analyses that could affect the robustness of the outcomes and conclusions. The quality and completeness of the documented information were variable, particularly in the earlier chronological cases from the early 2000s. Some original records also no longer exist, making further analysis impossible in these instances. Additionally, we only included data from centres in the UK, which may limit the generalisability of our findings to other countries with potential differences in the spectrum of causative pathogens. Nevertheless, we believe that the large numbers of patients from 3 separate and geographically distant tertiary eye centres across the UK provides a robust overview of the state of play for POE in this region.
Conclusions
In summary, we provide a comprehensive analysis of the microbial profile of acute-onset POE and the antibiotic sensitivity patterns across three geographically distant UK tertiary eye centres over an extended period. The stability in the trend of our POE pathogens, coupled with their excellent antibiotic susceptibility, suggests that current empirical treatment with intravitreal Vancomycin and Ceftazidime/Amikacin provides effective broad coverage for the vast majority of cases. It is likely that this antibiotic combination will remain relevant in the future, assuming no rapid development of antibiotic resistance. Continuous surveillance of POE trends would help to identify any shifts in microbial profiles or resistance patterns.
Summary
What was known before
-
There is geographical variation in pathogens causing postoperative endophthalmitis with growing concerns of antimicrobial resistance especially in Gram-negative bacteria.
What this study adds
-
Majority of bacterial isolates causing postoperative endophthalmitis in the UK are Gram-positive bacteria with no major shifts in the spectrum of causative pathogens.
-
Empirical treatment using intravitreal Vancomycin and Ceftazidime/Amikacin remained effective with low levels of resistance in our cohort.
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Behndig A, Cochener B, Güell JL, Kodjikian L, Mencucci R, Nuijts RM, et al. Endophthalmitis prophylaxis in cataract surgery: overview of current practice patterns in 9 European countries. J Cataract Refract Surg. 2013;39:1421–31.
Sadaka A, Durand ML, Gilmore MS. Bacterial endophthalmitis in the age of outpatient intravitreal therapies and cataract surgeries: host-microbe interactions in intraocular infection. Prog Retin Eye Res. 2012;31:316–31.
Nentwich MM, Ta CN, Kreutzer TC, Li B, Schwarzbach F, Yactayo-Miranda YM, et al. Incidence of postoperative endophthalmitis from 1990 to 2009 using povidone-iodine but no intracameral antibiotics at a single academic institution. J Cataract Refract Surg. 2015;41:58–66.
Asencio MA, Huertas M, Carranza R, Tenias JM, Celis J, Gonzalez-Del Valle F. A case-control study of post-operative endophthalmitis diagnosed at a Spanish hospital over a 13-year-period. Epidemiol Infect. 2015;143:178–83.
Daien V, Papinaud L, Gillies MC, Domerg C, Nagot N, Lacombe S, et al. Effectiveness and Safety of an Intracameral Injection of Cefuroxime for the Prevention of Endophthalmitis After Cataract Surgery With or Without Perioperative Capsular Rupture. JAMA Ophthalmol. 2016;134:810–6.
Pathengay A, Khera M, Das T, Sharma S, Miller D, Flynn HW Jr. Acute Postoperative Endophthalmitis Following Cataract Surgery: A Review. Asia Pac J Ophthalmol. 2012;1:35–42.
Chopra R, Preston GC, Keenan TDL, Mulholland P, Patel PJ, Balaskas K, et al. Intravitreal injections: past trends and future projections within a UK tertiary hospital. Eye. 2022;36:1373–8.
Keenan TD, Wotton CJ, Goldacre MJ. Trends over time and geographical variation in rates of intravitreal injections in England. Br J Ophthalmol. 2012;96:413–8.
Malmin A, Syre H, Ushakova A, Utheim TP, Forsaa VA. Twenty years of endophthalmitis: Incidence, aetiology and clinical outcome. Acta Ophthalmologica. 2021;99:e62–e9.
Ong AY, Rigaudy A, Toufeeq S, Robins J, Shalchi Z, Bindra MS, et al. Intravitreal injections as a leading cause of acute postoperative endophthalmitis-a regional survey in England. Eye. 2023;37:163–9.
VanderBeek BL, Chen Y, Tomaiuolo M, Deaner JD, Syed ZA, Acharya B, et al. Endophthalmitis Rates and Types of Treatments After Intraocular Procedures. JAMA Ophthalmol. 2024;142:827–34.
Results of the Endophthalmitis Vitrectomy Study. A randomized trial of immediate vitrectomy and of intravenous antibiotics for the treatment of postoperative bacterial endophthalmitis. Endophthalmitis Vitrectomy Study Group Arch Ophthalmol. 1995;113:1479–96.
Barry P, Cordovés L, Gardner S. ESCRS guidelines for prevention and treatment of endophthalmitis following cataract surgery: data, dilemmas and conclusions. European Society of Cataract and Refractive Surgeons. 2013, pp. 1–44. https://www.escrs.org/media/uljgvpn1/english_2018_updated.pdf
Das T, Joseph J, Simunovic MP, Grzybowski A, Chen KJ, Dave VP, et al. Consensus and controversies in the science of endophthalmitis management: Basic research and clinical perspectives. Progress Retinal Eye Res. 2023;97:101218.
Relhan N, Pathengay A, Schwartz SG, Flynn HW Jr. Emerging Worldwide Antimicrobial Resistance, Antibiotic Stewardship and Alternative Intravitreal Agents for the Treatment of Endophthalmitis. Retina. 2017;37:811–8.
Liu C, Ding B, Ji J, Wang Z, Chen H, Cao W. Microbial Spectrum and Resistance Patterns in Ocular Infections: A 15-Year Review in East China. Infect Drug Resist. 2021;14:2165–71.
Day AC, Donachie PH, Sparrow JM, Johnston RL. The Royal College of Ophthalmologists’ National Ophthalmology Database study of cataract surgery: report 1, visual outcomes and complications. Eye. 2015;29:552–60.
EclinicalMedicine. Antimicrobial resistance: a top ten global public health threat. eClinicalMedicine. 2021;41:101221.
Velez-Montoya R, Monroy-Esquivel L, Ortiz-Guevara R, Quiroz-Mercado H, Fromow-Guerra J, Mexican Endophthalmitis Study Group. ALTERNATIVE INTRAVITREAL ANTIBIOTICS: A Systematic Review for Consideration in Recalcitrant or Resistant Endophthalmitis. Retina. 2023;43:1433–47.
Chen KJ, Sun MH, Hou CH, Chen HC, Chen YP, Wang NK, et al. Susceptibility of bacterial endophthalmitis isolates to vancomycin, ceftazidime, and amikacin. Sci Rep. 2021;11:15878.
Clarke B, Williamson TH, Gini G, Gupta B. Management of bacterial postoperative endophthalmitis and the role of vitrectomy. Surv Ophthalmol. 2018;63:677–93.
Seal D, Reischl U, Behr A, Ferrer C, Alió J, Koerner RJ, Barry P, ESCRS Endophthalmitis Study G. Laboratory diagnosis of endophthalmitis: comparison of microbiology and molecular methods in the European Society of Cataract & Refractive Surgeons multicenter study and susceptibility testing. J Cataract Refract Surg. 2008;34:1439–50.
Moloney TP, Park J. Microbiological isolates and antibiotic sensitivities in culture-proven endophthalmitis: a 15-year review. Brit J Ophthalmol. 2014;98:1492–7.
Lalitha P, Sengupta S, Ravindran RD, Sharma S, Joseph J, Ambiya V, et al. A literature review and update on the incidence and microbiology spectrum of postcataract surgery endophthalmitis over past two decades in India. Indian J Ophthalmol. 2017;65:673–7.
Bhattacharjee H, Bhattacharjee K, Gogoi K, Singh M, Singla BG, Yadav A. Microbial profile of the vitreous aspirates in culture proven exogenous endophthalmitis: A 10-year retrospective study. Indian J Med Microbiol. 2016;34:153–8.
Schimel AM, Miller D, Flynn HW Jr. Endophthalmitis isolates and antibiotic susceptibilities: a 10-year review of culture-proven cases. Am J Ophthalmol. 2013;156:50–2.e1.
Joseph J, Sontam B, Guda SJM, Gandhi J, Sharma S, Tyagi M, et al. Trends in microbiological spectrum of endophthalmitis at a single tertiary care ophthalmic hospital in India: a review of 25 years. Eye. 2019;33:1090–5.
Simunovic MP, Rush RB, Hunyor AP, Chang AA. Endophthalmitis following intravitreal injection versus endophthalmitis following cataract surgery: clinical features, causative organisms and post-treatment outcomes. Br J Ophthalmol. 2012;96:862–6.
Ong AP, Angbue Te Te N, Zagora SL, Symes RJ, Yates W, Chang AA, et al. Post-surgical versus post-intravitreal injection endophthalmitis: changing patterns in causative flora. Clinical Exp Ophthalmol. 2019;47:57–62.
Zhang DA, DeGeus DS, Hogden DM, et al. The association between endophthalmitis risk following a variety of intra-ocular procedures and defensin gene profile. AJO Int. 2024;1:100034.
Mollan SP, Gao A, Lockwood A, Durrani OM, Butler L. Postcataract endophthalmitis: Incidence and microbial isolates in a United Kingdom region from 1996 through 2004. J Cataract Refract Surg. 2007;33:265–8.
Feng HL, Robbins CB, Fekrat S. A Nine-Year Analysis of Practice Patterns, Microbiologic Yield, and Clinical Outcomes in Cases of Presumed Infectious Endophthalmitis. Ophthalmol Retina. 2020;4:555–9.
Sjoholm-Gomez de Liano C, Soberon-Ventura VF, Salcedo-Villanueva G, Santos-Palacios A, Guerrero-Naranjo JL, Fromow-Guerra J, et al. Sensitivity, specificity and predictive values of anterior chamber tap in cases of bacterial endophthalmitis. Eye Vis. 2017;4:18.
Kamalarajah S, Silvestri G, Sharma N, Khan A, Foot B, Ling R, et al. Surveillance of endophthalmitis following cataract surgery in the UK. Eye. 2004;18:580–7.
Kirstahler P, Bjerrum SS, Friis-Møller A, la Cour M, Aarestrup FM, Westh H, et al. Genomics-Based Identification of Microorganisms in Human Ocular Body Fluid. Sci Rep. 2018;8:4126.
Lee CS, Hong B, Kasi SK, Aderman C, Talcott KE, Adam MK, et al. Prognostic Utility of Whole-Genome Sequencing and Polymerase Chain Reaction Tests of Ocular Fluids in Postprocedural Endophthalmitis. Am J Ophthalmol. 2020;217:325–34.
Deshmukh D, Joseph J, Chakrabarti M, Sharma S, Jayasudha R, Sama KC, et al. New insights into culture negative endophthalmitis by unbiased next generation sequencing. Sci Rep. 2019;9:844.
Naik P, Gandhi J, Joseph J. Recent Advances and Ongoing Challenges in the Diagnosis of Culture Negative Endophthalmitis. Semin Ophthalmol. 2023;38:92–8.
Patel SN, Storey PP, Pancholy M, Obeid A, Wibbelsman TD, Levin H, et al. Changes in Management Based on Vitreous Culture in Endophthalmitis After Intravitreal Anti-vascular Endothelial Growth Factor Injection. Am J Ophthalmol. 2019;207:224–31.
Patel SN, Storey PP, Levin H, Pancholy M, Obeid A, Wibbelsman TD, et al. Endophthalmitis after Cataract Surgery: Changes in Management Based on Microbiologic Cultures. Ophthalmol Retina. 2021;5:16–22.
Author information
Authors and Affiliations
Contributions
BLT, AYO, DHS, JS and PCI designed the study. BLT, AYO, AM, VH and CS collected data. BLT, AYO, PCI, JS and DHS analysed and interpreted data. BLT, AYO and AM drafted the manuscript. DHS, JS, AJL, CS and PCI revised manuscript critically with approval of the final version. All authors substantively revised the paper.
Corresponding author
Ethics declarations
Competing interests
DHS has received consulting fees from Alcon, Gyroscope Therapeutics (a Novartis company), Alimera, Complement therapeutics, BVI, and Roche, and research funding from Alcon, Gyroscope Therapeutics, Boehringer Ingelheim, DORC, Roche, and Bayer – all unrelated to this project. The other authors declare that they have no conflict of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Teh, B.L., Ong, A.Y., Mehta, A. et al. Long term analysis of microbiological isolates and antibiotic susceptibilities in acute-onset postoperative endophthalmitis: a UK multicentre study. Eye 39, 1470–1475 (2025). https://doi.org/10.1038/s41433-025-03673-w
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41433-025-03673-w