Abstract
Maternally transmitted symbionts such as Wolbachia can alter sex allocation in haplodiploid arthropods. By biasing population sex ratios towards females, these changes in sex allocation may facilitate the spread of symbionts. In contrast to symbiont-induced cytoplasmic incompatibility (CI), the mechanisms that underpin sex allocation distortion remain poorly understood. Using a nuclear genotype reference panel of the haplodiploid mite Tetranychus urticae and a single Wolbachia variant that is able to simultaneously induce sex allocation distortion and CI, we unraveled the mechanistic basis of Wolbachia-mediated sex allocation distortion. Host genotype was an important determinant for the strength of sex allocation distortion. We further show that sex allocation distortion by Wolbachia in haplodiploid mites is driven by increasing egg size, hereby promoting egg fertilization. This change in reproductive physiology was also coupled to increased male and female adult size. Our results echo previous work on Cardinium symbionts, suggesting that sex allocation distortion by regulating host investment in egg size is a common strategy among symbionts that infect haplodiploids. To better understand the relevance that sex allocation distortion may have for the spread of Wolbachia in natural haplodiploid populations, we parametrized a model based on generated phenotypic data. Our simulations show that empirically derived levels of sex allocation distortion can be sufficient to remove invasion thresholds, allowing CI to drive the spread of Wolbachia independently of the initial infection frequency. Our findings help elucidate the mechanisms that underlie the widespread occurrence of symbionts in haplodiploid arthropods and the evolution of sex allocation.
Similar content being viewed by others
Introduction
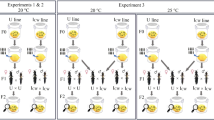
Maternally inherited symbionts can spread rapidly within arthropod populations by inducing a variety of reproductive phenotypes in their host (Engelstädter and Hurst 2009). These reproductive phenotypes increase the proportion of infected females that transmit the symbiont in host populations. Cytoplasmic incompatibility (CI) is currently recognized as the most common reproductive manipulation. CI interferes with the development of uninfected host embryos that were fertilized by infected males (Beckmann et al. 2019; Shropshire et al. 2020). In arthropod hosts with a haplodiploid mode of reproduction (unfertilized eggs develop into haploid males and fertilized eggs into diploid females), fertilized eggs that suffer from symbiont-induced CI have two developmental outcomes. Embryos either die before reaching adulthood (Female Mortality CI (FM‐CI)) or develop into viable males (Male Development CI (MD‐CI)) (Vavre et al. 2001; Perrot-Minnot et al. 2002) (Fig. 1). Previous work showed that a single CI cross in haplodiploids can simultaneously result in FM‐CI and MD‐CI (Vavre et al. 2001; Wybouw et al. 2022) (Fig. 1). As CI is rescued when males and females have an identical infection state, the reproductive phenotype provides a selective advantage to transmitting females. In haplodiploid arthropods, parthenogenesis induction is another classic and well-studied invasion strategy of symbionts (Ma and Schwander 2017). In contrast to CI, symbiont-induced parthenogenesis does not rely on egg fertilization and causes a diploidization of unfertilized eggs, generating parthenogenetic females instead of males that do not transmit the symbiont (Weeks and Breeuwer 2001; Ma and Schwander 2017). Another, much less understood, strategy of maternally transmitted symbionts to increase the proportion of infected females in haplodiploid host populations consists of directly altering sex allocation (i.e. the parental investment of resources to female vs. male offspring).
As Tetranychus mites have a haplodiploid mode of reproduction, unfertilized eggs develop into haploid males, while fertilized eggs generate diploid females (control, top left). Wolbachia-mediated sex allocation distortion facilitates successful egg fertilization and subsequent female development, biasing population sex ratios towards females. In CI crosses, fertilized eggs suffer from higher mortality rates (Female Mortality CI, FM-CI), or a proportion of fertilized eggs reach adulthood as males (Male Development CI, MD-CI). A single CI cross can lead to a mixture of FM-CI and MD-CI. Here, CI strength varies across the illustrative CI crosses.
As for CI, egg fertilization by males is essential for sex allocation distortion. However, in contrast to CI, sex allocation distortion occurs in crosses with infected females and is not contingent on the infection state of males (Fig. 1). These changes in sex allocation result in female-biased broods of infected females compared to those of uninfected females. To our knowledge, this reproductive phenotype was first described in a strain of the haplodiploid spider mite Tetranychus urticae that was infected with Wolbachia (Vala et al. 2000, 2003). In addition to Wolbachia, sex allocation distortion is now associated with other bacterial symbionts. Rickettsia, Hamiltonella, and Arsenophonus influence offspring sex ratio of infected females in haplodiploid whitefly species (Himler et al. 2011; Shan et al. 2019; Wang et al. 2020). Similarly, Cardinium also influence sex allocation in infected Kelly’s citrus thrips Pezothrips kellyanus in favor of female offspring (Katlav et al. 2022). Haplodiploid arthropods, including Tetranychus mites and Pezothrips insects, allocate egg fertilization and female development to larger eggs (Macke et al. 2011; Katlav et al. 2021). Katlav et al. 2022 show that Cardinium affect sex allocation in P. kellyanus by increasing egg size investment, hereby facilitating egg fertilization (and thus female development). Cardinium-mediated increase in egg size also led to progeny that were characterized by a higher survival rate and larger adult body size. In contrast, we do not understand how Wolbachia induce sex allocation distortion. The host genotype modulates the expression of many Wolbachia-induced reproductive phenotypes (Fujii et al. 2001; Kaur et al. 2021; Wybouw et al. 2022), raising the question whether sex allocation distortion is also contingent on host genetics.
Unraveling the mechanistic underpinnings of Wolbachia-induced sex allocation distortion is not only crucial for our fundamental understanding of host-Wolbachia interactions but also for the development of integrated pest management that relies on Wolbachia. A number of pest management programs take advantage of the CI drive system of Wolbachia to replace native pest populations with Wolbachia-infected populations that carry favorable traits, such as reduced virus transmission (Ross et al. 2019). However, CI strength can be weakened by varying population and environmental effects (Ross et al. 2017; Wybouw et al. 2022), threatening a collapse of the pest control technique. Previous theoretical work predicts that sex allocation distortion could reinforce the effect of CI and promote the invasion of Wolbachia from low initial infection frequencies (Egas et al. 2002). Yet, the actual contribution of sex allocation distortion to the spread of Wolbachia in natural pest populations remains unclear. Indeed, this previous model was not parametrized using empirical data of Wolbachia variants that cause both CI and sex allocation distortion. Moreover, in conflict with recent findings (Wybouw et al. 2022), the previous model did not allow for a simultaneous expression of FM-CI and MD-CI in single CI crosses. Understanding whether and how sex allocation of haplodiploid pests can be altered during population replacement events is critical to predict the outcome of Wolbachia-based pest control.
Here, we use the haplodiploid Tetranychus system to study a Wolbachia variant that simultaneously induces CI and sex allocation distortion. We show that sex allocation distortion is determined by the host background using a nuclear genotype reference panel of T. urticae. We further examined whether this reproductive phenotype is associated with changes in oviposition and immature survival rate. We tested whether, in analogy with Cardinium, Wolbachia also control sex allocation of haplodiploid hosts via egg size provisioning, and whether sex allocation distortion is associated with changes in adult size. Our results reveal signatures of convergent mechanisms of sex allocation distortion across two divergent host-symbiont systems. Finally, we parametrized a mathematical model to determine whether our observed levels of sex allocation distortion would contribute to the invasion of Wolbachia in natural haplodiploid populations.
Materials and methods
Mite lines and maintenance
We selected six T. urticae near-isogenic lines from the nuclear genotype reference panel described in Wybouw et al. (2022); Beis-w, Beis-c, LonX-w, LonX-c, Stt-w, and Stt-c. These lines are composed of three different near-isogenic nuclear genotypes (Beis, LonX, and Stt) that were transferred by seven rounds of iterative paternal introgression into a single cytoplasm that was originally recovered from the Wolbachia-infected Scp-w line. All lines share a single mitochondrion and were either infected with a single CI-inducing Wolbachia variant (indicated by “-w”) or were cured of the infection by antibiotic treatment (indicated by “-c”) (Wybouw et al. 2022). After antibiotic curing, the cured lines were propagated for ~18 generations before the onset of the experiments. Selection of the three nuclear genotypes from the reference panel was based on CI strength and phenotype. In addition, we also used the original Stt near-isogenic line that was naturally uninfected and did not undergo any antibiotic treatment. Infection states of Wolbachia and other maternally transmitted symbionts were confirmed before the start of the experiments by diagnostic PCR assays, as previously described (Wybouw et al. 2022). Mite lines were propagated by serial passage on detached bean leaves (Phaseolus vulgaris L. cv ‘Prelude’) at 24 °C, 60% RH, and a 16:8 light:dark photoperiod.
Effects of Wolbachia on sex allocation
To obtain estimates of sex ratio and immature stage survival (ISS), we generated age cohorts of the mite lines by allowing 75 randomly collected adult females to oviposit for 24 h on a detached bean leaf. At the end of offspring development (between 9 and 11 days post-oviposition), ~50 females were isolated on a single day as teleiochrysalids (the last immobile molting stage before reaching adulthood). These females were paired with ~30 one- to three-day old adult males of their own line on a detached bean leaf, allowing for compatible within-line matings. Three days were given to the females to emerge as virgin adults, and for matings to occur (we confirmed that the near-isogenic lines exhibit high copulation propensity under laboratory conditions; Supplementary information Box S1). Replicates were established by pooling five females (males were discarded) and allowing oviposition on 16 cm2 leaf discs for 3 days. Females were removed from the leaf discs and eggs were counted. Male and female offspring were collected and counted upon reaching adulthood. For Beis-w, Beis-c, LonX-w, LonX-c, Stt-w, and Stt-c, a total of 14 replicates were performed per line across three different batches (experimental blocks, each corresponding to a different age cohort). To fully ensure that the observed decrease in sex ratio in Stt-c vs. Stt-w was not associated with long-term deleterious effects of the antibiotic treatment, we expanded the experimental set-up for this host genotype by adding the original uninfected near-isogenic Stt line. Here, following the same rearing and mating protocol as described above, five replicates for Stt (uninfected), Stt-w (infected), and Stt-c (cured) were performed in a single batch.
To verify that the effect of Wolbachia on the sex ratio of Stt-w is not caused by Wolbachia-mediated parthenogenesis, feminization, or male killing, we isolated ~50 teleiochrysalid females from age cohorts of Stt-c and Stt-w as described above. We allowed the teleiochrysalids to hatch into virgin adult females and feed on a detached bean leaf for 3 days. As above, replicates were established by pooling five females on 16 cm2 leaf discs and allowing oviposition for 3 days. Females were removed from the leaf discs and eggs were counted. Male and female offspring were collected and counted upon reaching adulthood. For both Stt-c and Stt-w, a total of nine replicates were performed across two different batches.
Effects of Wolbachia on offspring number and size
To obtain accurate estimates of oviposition rates, we isolated ~50 teleiochrysalid females from age cohorts of Beis-w, Beis-c, LonX-w, LonX-c, Stt-w, and Stt-c, created as described above. These females were paired with ~30 one- to three-day old adult males on a detached bean leaf and were allowed to mate for 3 days. Replicates were established by pooling two females and allowing oviposition on 16 cm2 leaf discs for 24 h. Two hours after creating the replicates, all experimental females were examined to ensure they survived the transfer. Replicates with one or two dead females were removed from further analyses. After the oviposition interval, females were removed from the leaf discs and eggs were counted. The number of replicates per mite line ranged between 25 and 43 and were performed across four different batches.
To determine the effect of Wolbachia infection on Tetranychus egg size, eggs were randomly sampled from the leaf discs that were used to estimate oviposition rates for Beis-w, Beis-c, LonX-w, LonX-c, Stt-w, and Stt-c. Here, we focused on eggs that were oviposited across three batches of the oviposition tests. Eggs were photographed under a binocular microscope (6x 10x) using a Leica M50 camera (5 MP HD Microscope Camera Leica MC170 HD). Between 125 and 197 eggs were analyzed for each mite line (with a total of 971 eggs). To confirm that sex allocation is mediated by egg size in T. urticae (Macke et al. 2011), between 21 and 37 photographed eggs from Beis-c, Beis-w, LonX-c, and LonX-w were isolated and transferred to separate 4 cm2 leaf discs (with a total of 124 eggs). Sex was determined at the sexually dimorphic teleiochrysalid stage. Photos were analyzed using Natsumushi image measuring software (Tanahashi and Fukatsu 2018). For each egg, Natsumushi generated a smoothed outline based on eight manually specified demarcating points and automatically extracted the projected egg area. The total egg surface was calculated by assuming a spherical shape, following previous studies (Macke et al. 2011, 2012).
To determine the effect of Wolbachia infection on Tetranychus adult size, ~75 adult females were isolated from age cohorts of LonX-w, LonX-c, Stt-w, and Stt-c, and allowed to oviposit on a detached bean leaf for 24 h. Larval offspring were collected upon hatching and 12 larvae were pooled on 16 cm2 leaf discs to ensure equal population densities. As adult mites are highly mobile and are difficult to position in equal planes, we used the projected body surface of teleiochrysalids as a proxy for adult size. Both male and female teleiochrysalids were isolated, placed on a flat perspex slide, and photographed as described above. Using Natsumushi image measuring software (Tanahashi and Fukatsu 2018), the projected teleiochrysalid surface was measured by manually specifying ten demarcating points. Between 55 and 66 teleiochrysalids were analyzed for each mite line. In contrast to egg size, adult size was not expressed as total surface, but as the projected surface area.
Statistical analyses
All analyses were carried out using R (version 4.1.3) (R Core Team 2021). Raw data and the R script are publicly accessible as Supplementary Files and the statistical models are described in Table S1. Sex ratio was calculated as the proportion of females among the progeny that successfully reached adulthood. Immature stage survival (ISS) included embryonic and juvenile survival and was defined as the proportion of adult offspring over the total number of eggs. Daily oviposition was computed as the number of eggs per alive female (oviposition was allowed for 24 h only). All data, except copulation latency (Supplementary information Box S1), were analyzed using generalized linear mixed models (glmmTMB function of the “glmmTMB” package) (Brooks et al. 2017). Proportional data, sex ratio and ISS, were examined with a Binomial (or Betabinomial to account for overdispersion) error distribution, with a logit link. Continuous data, oviposition, egg surface, male and female adult size were fitted with a Gaussian error distribution (Table S1). In the case of non-normal error distribution, the response variables were linearized using a Box-cox transformation prior to analysis (boxcox function of the “MASS” package was used to find the appropriate value of lambda) (Crawley 2007). For most of the statistical models, genotype, infection state and their interaction were fit as fixed explanatory variables, whereas batch was considered as a random explanatory variable. To analyze sex ratio within the Stt background (Stt, Stt-c, and Stt-w), only the infection state was used as a fixed explanatory variable. For the analysis of female and male egg surface, offspring sex along with all two- and three-way interactions with the genotype and/or infection state were added as fixed explanatory variables. Maximal models, including all higher order interactions, were simplified by sequentially eliminating non-significant terms and interactions to establish minimal models. The significance of the explanatory variables was established using chi-square tests (Anova function of the “car” package). The reported significant chi-squared values are for the minimal model while the non-significant values were obtained before removing the variable from the minimal model (Crawley 2007). When interactions or factors with more than two levels were significant, differences among levels were analyzed using multiple comparisons (pairs function of the “emmeans” package (Lenth et al. 2022)), with Bonferroni corrections to account for multiple testing.
Modeling Wolbachia invasion in Tetranychus populations
To determine how sex allocation distortion, combined with varying CI phenotype (FM- and/or MD-CI) and strength, may affect the spread of a Wolbachia variant in divergent Tetranychus populations, we extended a mathematical model previously developed by Vavre et al. (2000) and Egas et al. (2002). The proportion of females (ft) and males (mt) that are infected at generation t, are given by the following equations:
where most of the parameters correspond to those in Vavre et al. (2000). F is the relative fecundity of infected vs. uninfected females, ISS the relative immature stage survival of infected vs. uninfected offspring, and µ the proportion of uninfected eggs produced by infected females (transmission efficiency is thus given by 1 – µ). k corresponds to the fertilization rate of eggs (i.e., estimated as the female to adult ratio in control crosses), and Sd to the sex allocation distortion induced by Wolbachia in the brood of infected females (i.e., the ratio of female to adult in the brood of infected females relative to uninfected females in control crosses). Finally, CI is the total proportion of eggs affected by CI (i.e., total CI strength), computed as a function of MD and FM, which correspond to the MD‐ and FM-CI phenotypes, respectively (MD and FM are equal to the corrected indexes MDcorr and FMcorr, respectively) (Poinsot et al. 1998; Cattel et al. 2018; Zélé et al. 2020; Cruz et al. 2021; Wybouw et al. 2022). Assuming that the effects of both CI phenotypes are independent, total CI should be computed as \({{\mathrm{CI}}}={{\mathrm{MD}}}+{{\mathrm{FM}}}\cdot (1-{{\mathrm{MD}}})\). In the current study, however, we computed total CI as CI = MD + FM, as the implemented FMcorr indexes already account for a potential decrease in female production due to MD-CI (i.e., these indexes were already corrected using MDcorr; Wybouw et al. 2022).
In this model, Wolbachia infection frequency in females is calculated by the same formula as when only MD- or FM-CI is considered (Egas et al. 2002, and Vavre et al. 2000 for Sd = 1). The difference lies in how the CI parameter is computed. As only MD-CI directly affects the proportion of infected males, the formula for Wolbachia infection frequency in males is the same as previous models that only consider MD-CI, and when MD = 0, it is also the same as those that only consider FM-CI (Vavre et al. 2000 for Sd = 1). Note, however, that the current formula differs from that of Egas et al. 2002 for MD-CI, as one category of uninfected males produced by uninfected females, is missing in the denominator of the previous model. The category corresponds to uninfected eggs fertilized by the sperm of infected males that develop as males instead of females due to MD-CI, given by \((1-{f}_{t})(k\cdot {{\mathrm{MD}}}\cdot {m}_{t})\).
As in Egas et al. (2002) and Vavre et al. (2000), when Wolbachia induce CI, the model can yield three different equilibria: (1) an unstable polymorphic equilibrium, which corresponds to ‘the threshold infection frequency for Wolbachia invasion’ known in diploid species (Hoffmann et al. 1990; Turelli 1994); (2) a stable polymorphic equilibrium, which is reached when the initial infection frequency is above the unstable equilibrium (note that only perfectly transmitted Wolbachia can fully invade a host population; i.e., this equilibrium can equal 1 only when µ = 0), and (3) an equilibrium where Wolbachia infection is purged, which is reached when the initial infection frequency is below the unstable equilibrium. When Wolbachia do not induce CI, only one polymorphic equilibrium can exist that is either stable or unstable when Wolbachia are beneficial or costly for the host, respectively. Analytical solutions and main equilibrial conditions are provided in the Supplementary Box S2.
Results
Sex allocation in Tetranychus relies on an interaction between host and Wolbachia
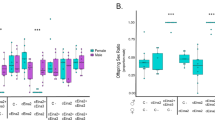
To study Wolbachia-induced sex allocation distortion in Tetranychus, we selected three nuclear genotypes, Beis, LonX, and Stt that were either infected with a single Wolbachia variant (‘-w’ suffix) or cured via antibiotic treatment (‘-c’ suffix) (Wybouw et al. 2022). Selection of these nuclear genotypes was based on varying CI strength and phenotype. Whereas Beis is typified by complete CI, LonX and Stt exhibit intermediate CI strength. The CI phenotype is characterized by a combination of MD-CI and FM-CI in LonX, while only consisting of FM-CI in Beis and Stt (Wybouw et al. 2022). We performed compatible within-line crosses for these Wolbachia-infected and cured sister lines. The interaction between nuclear genotype and the Wolbachia infection state significantly affected the female-biased sex ratios (χ22 = 14.6286, p = 0.0007) (Fig. 2). Within-genotype a posteriori comparisons revealed that the average sex ratio increased by ~11% when Stt females were infected with Wolbachia (t-ratio = 4.272, p = 0.0005), whereas the sex ratios were not significantly different within Beis nor LonX (t-ratio = 1.125, p = 1.0000 and t-ratio = 1.847, p = 0.6176, respectively) (Fig. 2 and Table S2). In a second experiment, we included the naturally uninfected Stt line to rule out any potential long-term interference of the antibiotic treatment on sex ratio. Infection state determined the female-biased sex ratio (χ22 = 18.9370, p < 0.0001). No significant difference was found between uninfected Stt and cured Stt-c (t-ratio = 0.110, p = 1.0000), whereas the sex ratio of infected Stt-w was higher to both Wolbachia-free lines (vs. Stt-c: t-ratio = 3.964, p = 0.0056 and vs. Stt: t-ratio = 3.864, p = 0.0068) (Fig. 2). Together with the non-differential sex ratios between the infected and cured sister lines of the Beis and LonX genotype (Fig. 2), this is strong evidence that the antibiotic treatment had no long-term effects on observed sex ratios.
A Sex ratio from compatible within-line crosses for the infected and cured lines of the Beis, LonX, and Stt host genotype. Sex ratio was calculated as the proportion of females among the progeny that successfully reached adulthood. Host genotypes are ordered according to decreasing CI strength. Statistical significance within each host genotype is outlined above: *p < 0.05, while ns, not significantly different at the 5% level. B Sex ratio from compatible within-line crosses for the uninfected, infected, and cured line of the Stt host genotype. Identical superscripts outline non-significant differences at the 5% level. For both panels, boxplots and replicates are color coded based on the infection state (see bottom middle). Each boxplot represents the 25%, 50%, and 75% quantile.
Host genotype, infection state, and their interaction did not significantly affect immature stage survival (ISS). ISS is therefore not associated with the change in sex ratio within the Stt background (Fig. S1 and Table S3). We also investigated the offspring sex ratio and ISS of virgin Stt-w and Stt-c females to test whether the Wolbachia variant induces parthenogenesis, feminization, or male killing within the Stt background. All offspring were male, ruling out Wolbachia-mediated parthenogenesis and feminization. As no significant difference in ISS was observed between the infected and cured line (χ21 = 0.0000, p = 0.9949), the hypothesis of Wolbachia-induced male killing was also rejected (Fig. S1 and Table S3). This further shows that Wolbachia infection does not cause any particular survival costs in the focal nuclear genotypes during offspring development. However, the interaction between host genotype and the Wolbachia infection state significantly determined daily oviposition (χ22 = 9.0006, p = 0.0111) (Fig. 3 and Table S4). Comparisons within host nuclear backgrounds uncovered that the daily oviposition rates were only significantly different between the infected and cured line of the LonX genotype (t-ratio = 5.704, p < 0.0001). For the LonX genotype, Wolbachia infection decreased daily oviposition (Fig. 3).
Daily oviposition from fertilized females for the infected and cured lines of the Beis, LonX, and Stt host genotype. Host genotypes are ordered according to decreasing CI strength. Statistical significance within each host genotype is outlined above: *p < 0.05, whereas ns, not significantly different at the 5% level. Boxplots and replicates are color coded based on the infection state (see bottom right). Each boxplot represents the 25%, 50%, and 75% quantile.
Wolbachia control host sex allocation by regulating egg provisioning
In Tetranychus mites, the probability of egg fertilization (and subsequent female development) positively covaries with egg size (Macke et al. 2011). Here, we confirm that sex allocation is mediated by egg size by tracking the offspring of Beis-w, Beis-c, LonX-w, and LonX-c. Statistical analyses revealed that female egg surface was significantly larger (χ21 = 28.8573, p < 0.0001), and also varied depending on the interaction between nuclear genotype and the infection state (χ23 = 11.1822, p = 0.0008) (Fig. S2). In haplodiploid Pezothrips insects, Cardinium manipulate sex allocation by increasing egg size, hereby facilitating egg fertilization (Katlav et al. 2022). We tested whether Wolbachia achieve higher female-biased sex ratios in Stt mites by inducing similar changes in the host reproductive physiology. We quantified egg size across the infected and cured lines of the three T. urticae genotypes and again observed that the interaction between host genotype and the Wolbachia infection state significantly affected egg surface (χ22 = 97.0092, p < 0.0001) (Fig. 4). Although we observed that Wolbachia infection only biased the sex ratio of Stt mites, we found that for both LonX and Stt mites, Wolbachia-infected females oviposited larger eggs compared to cured females (t-ratio = 7.030, p < 0.0001 and t-ratio = 12.606, p < 0.0001, respectively). However, on average, Wolbachia infection increased egg surface by ~11% in Stt mites and by only ~5% in LonX mites (Fig. 4 and Table S5). In contrast, egg size was not significantly different within the Beis genotype (t-ratio = 0.541, p = 1.0000).
Egg size from fertilized females for the infected and cured lines of the Beis, LonX, and Stt host nuclear genotype. Host genotypes are ordered according to decreasing CI strength. Statistical significance within each host genotype is outlined above: *p < 0.05, whereas ns, not significantly different at the 5% level. Boxplots and replicates are color coded based on the infection state (see bottom right). Each boxplot represents the 25%, 50%, and 75% quantile.
Egg size positively covaries with adult size in Tetranychus mites (Macke et al. 2011), raising the question whether the Wolbachia-mediated increase in egg size also results in larger adults for the LonX and Stt genotypes. Using the projected body surface of teleiochrysalids (final molting stage before adulthood) as a proxy, our results show that the interaction between host genotype and the Wolbachia infection state significantly affected the adult size for both males and females (males: χ21 = 14.5653, p = 0.0001 and females: χ21 = 11.4769, p = 0.0007) (Fig. 5). Wolbachia infection significantly increased male and female adult size within the Stt genotype (males: t-ratio = 4.310, p = 0.0002 and females: t-ratio = 3.255, p = 0.0058), but no significant differences were observed for LonX (males: t-ratio = 0.998, p = 1.0000 and females: t-ratio = 1.542, p = 0.5021) (Fig. 5). For the Stt genotype, the projected body area of teleiochrysalid males and females were ~10% and ~5% larger in response to Wolbachia infection, respectively (Fig. 5 and Table S6).
The projected body surface of teleiochrysalids as a proxy for adult size for the infected and cured lines of the LonX and Stt host nuclear genotype (with males and females in panel A and B, respectively). Statistical significance within each host genotype is outlined above: *p < 0.05, whereas ns, not significantly different at the 5% level. Boxplots and replicates are color coded based on the infection state (see middle bottom). Each boxplot represents the 25%, 50%, and 75% quantile.
Sex allocation distortion removes the threshold for Wolbachia invasion
We predicted the spread of Wolbachia in the three T. urticae nuclear genotypes and assessed the effect of sex allocation distortion on infection dynamics by adapting previously developed deterministic models for haplodiploids (Vavre et al. 2000; Egas et al. 2002). As observed for the LonX genotype, a single CI cross can lead to a mixture of FM-CI and MD-CI (Wybouw et al. 2022), an outcome that is now incorporated into the revised version of the model (Table S7 provides the full comparison between our and previous models).
For the Beis and LonX genotypes, simulations for Wolbachia invasion revealed that estimated fitness costs of Wolbachia infection result in unstable equilibria under which infection is lost after a number of host generations (i.e., the threshold infection frequency for Wolbachia invasion) (Fig. 6). In particular, the high fecundity costs estimated in LonX leads to the highest threshold (~46% vs. ~16% in Beis; Fig. 6). Above the threshold frequency, Wolbachia are predicted to spread the fastest in populations of the Beis nuclear genotype, in which total CI strength is the strongest (~100% in Beis, ~65% in LonX, and ~50% in Stt; Table S8). In contrast, despite similar fitness costs in Stt and Beis and weaker CI strength in the former, the model does not predict a threshold frequency for Wolbachia invasion in Stt (Fig. 6). Additional simulations that revoke the effect of sex allocation distortion in the Stt genotype further show that the absence of an infection threshold is solely due to the Wolbachia-induced female-biased sex ratio (Fig. S3). Indeed, without sex allocation distortion in the Stt genotype, only an initial infection frequency above ~24% would allow for Wolbachia invasion (Fig. S3). In agreement with these results, there is no longer a threshold frequency for Wolbachia invasion in the Beis genotype after introducing the same level of sex allocation distortion as observed for Stt (Fig. S3, the unstable equilibrium becomes ~0%). In contrast, the introduction of sex allocation distortion in the LonX genotype has only little effect (the invasion threshold decreases from ~46% to ~43%) (Fig. S3). Finally, revoking CI in each of the three genotypes shows that Wolbachia should be purged from Beis and LonX but should still spread in Stt where sex allocation distortion compensates for the infection costs (Fig. S3). Consistent with these observations, even though the Wolbachia variant induces weaker CI in Stt than in LonX (~50% vs. ~65% total CI strength), the infection is predicted to spread above the threshold frequency faster in Stt due to sex allocation distortion (Fig. 6). Our model parametrization thus shows that, in some host genotypes, sex allocation distortion can strengthen the effect of CI and ensure the spread of Wolbachia independently of the initial infection frequencies. Together, these results corroborate previous theoretical predictions of the functional importance of sex allocation distortion for maternally inherited symbionts in haplodiploid hosts (Egas et al. 2002).
Wolbachia infection frequencies throughout host generations for initial infection frequencies ranging from 0 to 1. Host genotypes are ordered according to decreasing CI strength. Dashed red lines represent the unstable polymorphic equilibrium, which corresponds to ‘the threshold infection frequency for Wolbachia invasion’. Blue lines represent the stable polymorphic equilibrium, which is reached when the initial infection frequency is above the unstable equilibrium. Our parametrized model does not predict a threshold infection frequency in Stt despite a lower oviposition estimate upon Wolbachia infection. Additional simulations that add/remove sex allocation distortion in Beis, LonX, and Stt further support the contribution of sex allocation distortion to Wolbachia invasion (Fig. S3).
Discussion
Maternally inherited symbionts commonly infect haplodiploid arthropods and spread through these host populations by inducing distinct reproductive phenotypes (Weinert et al. 2015; Ma and Schwander 2017; Shropshire et al. 2020; Kaur et al. 2021). A growing body of evidence indicates that sex allocation distortion could be a widespread, previously neglected, reproductive phenotype across divergent haplodiploid host-symbiont systems (Vala et al. 2003; Wang et al. 2020; Katlav et al. 2022). Yet, several questions about the mechanisms that underpin sex allocation distortion are still outstanding, especially for Wolbachia.
The current results on the Stt nuclear genotype confirm that Wolbachia can cause higher female-biased sex ratios by regulating host sex allocation in Tetranychus mites (Vala et al. 2003). Here, Wolbachia infection increases female production by ~11%, whereas Vala et al. 2003 reports an increase of ~18% in the T. urticae C-strain. As for other symbiont-mediated phenotypes, this difference in phenotypic strength can be attributed to genetic variation in Wolbachia and/or the arthropod host (Turelli 1994; Engelstädter and Hurst 2009; Beckmann et al. 2021; Wybouw et al. 2022). The Wolbachia variant of the current study does not induce quantifiable sex allocation distortion in the Beis and LonX genotype, suggestive of strong host modulation by Tetranychus. These findings are consistent with a previous hypothesis that T. urticae evolves compensatory mechanisms that suppress Wolbachia-induced sex allocation distortion (Vala et al. 2003). However, this hypothesis implies that sex allocation distortion is deleterious for Tetranychus hosts and that suppressor systems evolved in the Beis and LonX genotype to counteract the reproductive manipulations induced by Wolbachia.
As the focal Wolbachia variant also induces CI in incompatible matings (Wybouw et al. 2022), we show that Wolbachia are able to simultaneously induce CI and sex allocation distortion. These observations add to a growing body of literature showing that symbiont genomes often carry the genetic architecture of multiple reproductive manipulations (Fujii et al. 2001; Kaur et al. 2021; Katlav et al. 2022). Previous work uncovered that CI strength is also determined by host modulation within the T. urticae nuclear genotype reference panel (Wybouw et al. 2022). The penetrance of sex allocation distortion and CI appears uncoupled across the host genotypes. The Wolbachia variant only causes intermediate CI in Stt and LonX (~50% and ~65%, respectively), compared to complete CI in Beis (100%). We can therefore speculate that the host modifier systems act independently on the two Wolbachia-induced reproductive phenotypes. Host modulation of CI is largely manifested in infected Tetranychus males (Wybouw et al. 2022). As the reproductive physiology of haplodiploid females is a strong determinant of the sex ratio of their offspring (Overmeer and Harrison 1969; Mitchell 1972; Takafuji and Ishii 1989; Katlav, Nguyen, et al. 2021) and sex allocation distortion appears independent of the infection state of males, it is likely that host modulation of sex allocation distortion occurs in females. Together, this further indicates that the host modifier systems of haplodiploids might have sex-specific phenotypic effects.
In this study, we also gathered strong evidence that Wolbachia-induced sex allocation distortion in Tetranychus mites is mediated by regulating egg size. For the Stt genotype, Wolbachia-infected females oviposit larger eggs compared to the Wolbachia-free sister line, increasing the probability of successful fertilization and female development (Macke et al. 2011; Katlav, Cook, et al. 2021). We also noted a weaker, but significant, increase in egg size for the LonX genotype. Here, Wolbachia infection does not cause sex allocation distortion, likely because the egg size effect is too weak. As egg size also varied across the three T. urticae nuclear genotypes regardless of Wolbachia infection, our results identify egg size investment as a reproductive trait that is modulated by both the host and symbiont genotype. In Tetranychus females, a physiological trade-off controls resource allocation into egg number and size (Macke et al. 2012). Here, Wolbachia infection influences egg number within the LonX nuclear background, but not within Stt. However, only a narrow 24 h time interval of the oviposition period was monitored within our experimental set-up, raising the possibility that, conversely to Macke et al. 2012, we missed the full trade-off effect of increased egg size on total egg number. Alternatively, this trade-off may be altered if Wolbachia infection causes an increase in available resources for ovipositing females. Many bacterial symbionts provide nutrients for their invertebrate host, including Wolbachia (Hosokawa et al. 2010; Kaur et al. 2021). Based on dietary supplementation experiments, symbiont-mediated provisioning of B vitamins is implicated in sex ratio distortion in haplodiploid whitefly species (Wang et al. 2020). In addition to egg size regulation, other Wolbachia-mediated changes in the female reproductive physiology could further contribute to sex allocation distortion. To increase the likelihood of successful egg fertilization, Wolbachia could facilitate the flow of sperm from the spermatheca to the ovary and the entry of sperm cells into the ovarian cavity (Helle 1967; Helle and Sabelis 1985). As sperm cells tend to accumulate in the dorsal region of the ovarian cavity in Tetranychus mites, Wolbachia could also promote sperm-egg contact and subsequent egg fertilization by causing a more even sperm distribution within the cavity (Feiertag-Koppen and Pijnacker 1982; Helle and Sabelis 1985).
Previous studies in haplodiploids uncovered a positive correlation between egg size, immature survival rate, and adult body size (Macke et al. 2011; Katlav, Cook, et al. 2021). Consistent with these studies, Cardinium-mediated increase in egg size is shown to be linked to higher juvenile survival rate and adult size in P. kellyanus (Katlav et al. 2022). Although Wolbachia-infected LonX-w and Stt-w females tended to have broods with higher immature survival rates than cured LonX-c and Stt-c females, we could not statistically demonstrate that Wolbachia infection positively affects this trait in these two genotypes. However, we found that Wolbachia infection increases male and female adult size within the Stt genotype, an effect that may lead to a higher reproductive success for infected adults (Li and Zhang 2018). In Tetranychus, competition for mates among males can be intense, resulting in aggressive fighting behavior. Larger males have an advantage in these competitive interactions, and are more likely to fertilize virgin females under certain conditions (Potter et al. 1976; Enders 1993). A higher fertilization success of Wolbachia-infected males may strengthen the effect of CI, further driving Wolbachia infection frequencies through host populations. In contrast, egg size difference does not persist into the adult stage in LonX mites, which is likely explained by the relatively weak egg size effect of Wolbachia infection in this nuclear genotype.
In haplodiploid Pezothrips insects, Cardinium symbionts also cause host sex allocation distortion (Katlav et al. 2022). The common strategy within the Wolbachia-Tetranychus and Cardinium-Pezothrips systems to distort sex allocation by regulating egg size raises the question of whether these symbionts share the same genetic basis for sex allocation distortion or evolved this reproductive phenotype independently. CI is also induced by both Cardinium and Wolbachia and is manifested by remarkably similar cytological defects (Gebiola et al. 2017). Yet, despite a nearly identical cellular phenotype, CI appears to be underpinned by different genetic architectures in these intracellular bacteria (Gotoh et al. 2007; Penz et al. 2012; LePage et al. 2017; Beckmann et al. 20172019; Shropshire et al. 2020). Our results suggest that convergent mechanisms also underpin sex allocation distortion across divergent biological systems. To dissect the underlying mechanisms of sex allocation distortion, comparative genomics and transcriptomics data sets can be leveraged, for instance, the differentially expressed gene set of T. urticae embryos upon Wolbachia infection (Bing et al. 2020).
Many haplodiploid arthropods, including thrips, whiteflies, and spider mites, are pathogen-carrying pests that threaten agricultural crop production (Robertson and Carroll 1988; Jones 2005; Kitajima et al. 2010; Navas-Castillo et al. 2011). Pest control programs are being developed that rely on the driving ability of artificial Wolbachia infections that reduce pathogen transmission (Ross et al. 2019; Gong et al. 2020). For successful population replacement, infected individuals must be released at a rate high enough to exceed threshold infection frequencies (Ross et al. 2019). Here, using a parametrized model, we show that sex allocation distortion could facilitate the spread of Wolbachia within haplodiploid pest populations by removing threshold infection frequencies and increasing invasion rates. Therefore, this reproductive phenotype may be instrumental to increase the efficacy of pest control measures. Our results underscore the importance of studying the impact and underlying mechanisms of natural and artificial Wolbachia infections on host sex allocation.
Data archiving
All raw data and R scripts are available at the Dryad repository (https://doi.org/10.5061/dryad.1vhhmgqzk).
References
Beckmann JF, Bonneau M, Chen H, Hochstrasser M, Poinsot D, Merçot H et al. (2019) The toxin–antidote model of cytoplasmic incompatibility: genetics and evolutionary implications. Trends Genet 35:175–185
Beckmann JF, Ronau JA, Hochstrasser M (2017) A Wolbachia deubiquitylating enzyme induces cytoplasmic incompatibility. Nat Microbiol 2:17007
Beckmann JF, Van Vaerenberghe K, Akwa DE, Cooper BS (2021) A single mutation weakens symbiont-induced reproductive manipulation through reductions in deubiquitylation efficiency. Proc Natl Acad Sci USA 118:e2113271118
Bing X, Lu Y, Xia C, Xia X, Hong X (2020) Transcriptome of Tetranychus urticae embryos reveals insights into Wolbachia‐induced cytoplasmic incompatibility. Insect Mol Biol 29:193–204
Brooks ME, Kristensen K, van Benthem KJ, Magnusson A, Berg CW, Nielsen A, et al. (2017) glmmTMB balances speed and flexibility among packages for zero-inflated generalized linear mixed modeling. R J 9:378–400
Cattel J, Nikolouli K, Andrieux T, Martinez J, Jiggins F, Charlat S et al. (2018) Back and forth Wolbachia transfers reveal efficient strains to control spotted wing drosophila populations (J Beggs, Ed.). J Appl Ecol 55:2408–2418
Crawley M (2007) The R book. John Wiley & Sons: Hoboken, NJ
Cruz MA, Magalhães S, Sucena É, Zélé F (2021) Wolbachia and host intrinsic reproductive barriers contribute additively to postmating isolation in spider mites. Evolution 75:2085–2101
Egas M, Vala F, Hans Breeuwer JAJ (2002) On the evolution of cytoplasmic incompatibility in haplodiploid species. Evolution 56:1101–1109
Enders MM (1993) The effect of male size and operational sex ratio on male mating success in the common spider mite, Tetranychus urticae Koch (Acari: Tetranychidae). Anim Behav 46:835–846
Engelstädter J, Hurst GDD (2009) The ecology and evolution of microbes that manipulate host reproduction. Annu Rev Ecol Evol Syst 40:127–149
Feiertag-Koppen CCM, Pijnacker LP (1982) Development of the female germ cells and process of internal fertilization in the two-spotted spider mite Tetranychus urticae koch (Acariformes: Tetranychidae). Int J Insect Morphol Embryol 11:271–284
Fujii Y, Kageyama D, Hoshizaki S, Ishikawa H, Sasaki T (2001) Transfection of Wolbachia in Lepidoptera: the feminizer of the adzuki bean borer Ostrinia scapulalis causes male killing in the Mediterranean flour moth Ephestia kuehniella. Proc R Soc Lond B 268:855–859
Gebiola M, Giorgini M, Kelly SE, Doremus MR, Ferree PM, Hunter MS (2017) Cytological analysis of cytoplasmic incompatibility induced by Cardinium suggests convergent evolution with its distant cousin Wolbachia. Proc R Soc B 284:20171433
Gong J-T, Li Y, Li T-P, Liang Y, Hu L, Zhang D et al.(2020) Stable Introduction of Plant-Virus-Inhibiting Wolbachia into Planthoppers for Rice Protection. Curr Biol. 30:4837–4845.e5
Gotoh T, Noda H, Ito S (2007) Cardinium symbionts cause cytoplasmic incompatibility in spider mites. Heredity 98:13–20
Helle W (1967) Fertilization in the two-spotted spider mite (Tetranychus urticae: ACARI). Entomol Exp Appl 10:103–110
Helle W, Sabelis MW (1985) Spider mites. Their biology, natural enemies, and control. Elsevier, Amsterdam
Himler AG, Adachi-Hagimori T, Bergen JE, Kozuch A, Kelly SE, Tabashnik BE et al. (2011) Rapid spread of a bacterial symbiont in an invasive whitefly is driven by fitness benefits and female bias. Science 332:254–256
Hoffmann AA, Turelli M, Harshman LG (1990) Factors affecting the distribution of cytoplasmic incompatibility in Drosophila simulans. Genetics 126:933–948
Hosokawa T, Koga R, Kikuchi Y, Meng X-Y, Fukatsu T (2010) Wolbachia as a bacteriocyte-associated nutritional mutualist. Proc Natl Acad Sci 107:769–774
Jones DR (2005) Plant viruses transmitted by thrips. Eur J Plant Pathol 113:119–157
Katlav A, Cook JM, Riegler M (2021) Egg size‐mediated sex allocation and mating‐regulated reproductive investment in a haplodiploid thrips species (T Houslay, Ed.). Funct Ecol 35:485–498
Katlav A, Cook JM, Riegler M (2022) Common endosymbionts affect host fitness and sex allocation via egg size provisioning. Proc R Soc B 289:20212582
Katlav A, Nguyen DT, Cook JM, Riegler M (2021) Constrained sex allocation after mating in a haplodiploid thrips species depends on maternal condition. Evolution 75:1525–1536
Kaur R, Shropshire JD, Cross KL, Leigh B, Mansueto AJ, Stewart V et al. (2021) Living in the endosymbiotic world of Wolbachia: a centennial review. Cell Host Microbe 29:879–893
Kitajima EW, Rodrigues JCV, Freitas-Astua J (2010) An annotated list of ornamentals naturally found infected by Brevipalpus mite-transmitted viruses. Sci agric (Piracicaba, Braz) 67:348–371
Lenth R, Singmann H, Love J, Buerkner P, Herve M (2022) emmeans: estimated marginal means, aka least-squares means. R Package. https://github.com/rvlenth/emmeans
LePage DP, Metcalf JA, Bordenstein SR, On J, Perlmutter JI, Shropshire JD et al. (2017) Prophage WO genes recapitulate and enhance Wolbachia-induced cytoplasmic incompatibility. Nature 543:243–247
Li G, Zhang Z-Q (2018) Does size matter? Fecundity and longevity of spider mites (Tetranychus urticae) in relation to mating and food availability. Syst Appl Acarol 23:1796
Ma W-J, Schwander T (2017) Patterns and mechanisms in instances of endosymbiont-induced parthenogenesis. J Evolut Biol 30:868–888
Macke E, Magalhães S, Do-Thi Khanh H, Frantz A, Facon B, Olivieri I (2012) Mating Modifies Female Life History in a Haplodiploid Spider Mite. Am Naturalist 179:E147–E162
Macke E, Magalhães S, Khan HD-T, Luciano A, Frantz A, Facon B et al. (2011) Sex allocation in haplodiploids is mediated by egg size: evidence in the spider mite Tetranychus urticae Koch. Proc R Soc B 278:1054–1063
Mitchell R (1972) The sex ratio of the spider mite Tetranychus urticae. Entomol Exp Appl 15:299–304
Navas-Castillo J, Fiallo-Olivé E, Sánchez-Campos S (2011) Emerging virus diseases transmitted by whiteflies. Annu Rev Phytopathol 49:219–248
Overmeer W, Harrison R (1969) Notes on the control of the sex ratio in populations of the two-spotted spider mite, Tetranychus urticae Koch (Acarina: Tetranychidae). NZ J Sci 12:920–928
Penz T, Schmitz-Esser S, Kelly SE, Cass BN, Müller A, Woyke T et al. (2012) Comparative genomics suggests an independent origin of cytoplasmic incompatibility in Cardinium hertigii (NA Moran, Ed.). PLoS Genet 8:e1003012
Perrot-Minnot M-J, Cheval B, Migeon A, Navajas M (2002) Contrasting effects of Wolbachia on cytoplasmic incompatibility and fecundity in the haplodiploid mite Tetranychus urticae: Contrasting effects of Wolbachia in a haplodiploid mite. J Evolut Biol 15:808–817
Poinsot D, Bourtzis K, Markakis G, Savakis C, Merçot H (1998) Wolbachia transfer from Drosophila melanogaster into D. simulans: host effect and cytoplasmic incompatibility relationships. Genetics 150:227–237
Potter DA, Wrensch DL, Johnston DE (1976) Guarding, aggressive behavior, and mating success in male two-spotted spider mites. Ann Entomological Soc Am 69:707–711
R Core Team (2021). R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. URL http://www.R-project.org
Robertson NL, Carroll TW (1988) Virus-Like Particles and a Spider Mite Intimately Associated with a New Disease of Barley. Science 240:1188–1190
Ross PA, Turelli M, Hoffmann AA (2019) Evolutionary ecology of Wolbachia Releases for disease control. Annu Rev Genet 53:93–116
Ross PA, Wiwatanaratanabutr I, Axford JK, White VL, Endersby-Harshman NM, Hoffmann AA (2017) Wolbachia Infections in Aedes aegypti differ markedly in their response to cyclical heat stress (EA McGraw, Ed.). PLoS Pathog 13:e1006006
Shan H-W, Luan J-B, Liu Y-Q, Douglas AE, Liu S-S (2019) The inherited bacterial symbiont Hamiltonella influences the sex ratio of an insect host. Proc R Soc B 286:20191677
Shropshire JD, Leigh B, Bordenstein SR (2020) Symbiont-mediated cytoplasmic incompatibility: what have we learned in 50 years? eLife 9:e61989
Takafuji A, Ishii T (1989) Inheritance of sex ratio in the Kanzawa spider mite, Tetranychus kanzawai Kishida. Popul Ecol 31:123–128
Tanahashi M, Fukatsu T (2018) Natsumushi: Image measuring software for entomological studies: Image measuring software for entomology. Entomol Sci 21:347–360
Turelli M (1994) Evolution of incompatibility-inducing microbes and their hosts. Evolution 48:1500–1513
Vala F, Breeuwer JAJ, Sabelis MW (2000) Wolbachia–induced ‘hybrid breakdown’ in the two–spotted spider mite Tetranychus urticae Koch. Proc R Soc Lond B 267:1931–1937
Vala F, Van Opijnen T, Breeuwer JAJ, Sabelis MW (2003) Genetic conflicts over sex ratio: mite‐endosymbiont interactions. Am Naturalist 161:254–266
Vavre F, Dedeine F, Quillon M, Fouillet P, Fleury F, Boulétreau M (2001) Within-species diversity of Wolbachia-induced cytoplasmic incompatibility in haplodiploid insects. Evolution 55:1710–1714
Vavre F, Fleury F, Varaldi J, Fouillet P, Bouleatreau M (2000) Evidence for female mortality in Wolbachia-mediated cytoplasmic incompatibility in haplodiploid insects: Epidemiologic and evolutionary consequences. Evolution 54:191–200
Wang Y-B, Ren F-R, Yao Y-L, Sun X, Walling LL, Li N-N et al. (2020) Intracellular symbionts drive sex ratio in the whitefly by facilitating fertilization and provisioning of B vitamins. ISME J 14:2923–2935
Weeks AR, Breeuwer JAJ (2001) Wolbachia–induced parthenogenesis in a genus of phytophagous mites. Proc R Soc Lond B 268:2245–2251
Weinert LA, Araujo-Jnr EV, Ahmed MZ, Welch JJ (2015) The incidence of bacterial endosymbionts in terrestrial arthropods. Proc R Soc B: Biol Sci 282:20150249–20150249
Wybouw N, Mortier F, Bonte D (2022) Interacting host modifier systems control Wolbachia‐induced cytoplasmic incompatibility in a haplodiploid mite. Evol Lett 6:255–265
Zélé F, Santos I, Matos M, Weill M, Vavre F, Magalhães S (2020) Endosymbiont diversity in natural populations of Tetranychus mites is rapidly lost under laboratory conditions. Heredity 124:603–617
Acknowledgements
We thank Masahiko Tanahashi for assisting with the Natsumushi analyses, Sara Magalhães and Fabrice Vavre for useful discussions, Martijn Egas for confirming the error in Egas et al. 2002, and Guillaume Martin for his help with identifying the equilibria of the mathematical model. We also thank Bouwe Cattrysse for generating exploratory pilot data for the LonX-c and LonX-w lines. NW was supported by a BOF post-doctoral fellowship (Ghent University, 01P03420) and by a Research Foundation-Flanders (FWO) Research Grant (1513719N). This work was further supported by the FWO Research Network EVENET. This is contribution ISEM-2023-125 of the Institute of Evolutionary Science of Montpellier (ISEM).
Author information
Authors and Affiliations
Contributions
NW conceived and designed the experiments. NW, EVR, and JZ performed the experiments. FZ extended the model and performed the simulations. NW, FZ, and DB analyzed the data. NW wrote the manuscript with input from FZ and DB. All authors read and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Associate editor: Darren Obbard.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wybouw, N., Van Reempts, E., Zarka, J. et al. Egg provisioning explains the penetrance of symbiont-mediated sex allocation distortion in haplodiploids. Heredity 131, 221–229 (2023). https://doi.org/10.1038/s41437-023-00638-1
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41437-023-00638-1
This article is cited by
-
Wolbachia infection facilitates adaptive increase in male egg size in response to environmental changes
Scientific Reports (2025)
-
Cardinium symbionts are pervasive in Iranian populations of the spider mite Panonychus ulmi despite inducing an infection cost and no demonstrable reproductive phenotypes when Wolbachia is a symbiotic partner
Experimental and Applied Acarology (2023)