Abstract
Beta-blockers are recommended as a standard therapy for patients with heart failure (HF). However, beta-blockers are reportedly less effective in HF patients with atrial fibrillation (Af) compared with those with sinus rhythm (SR). Here, we investigated whether HR at discharge determined the cardiovascular outcomes in HF patients with Af treated with beta-blockers. In this analysis, we enrolled 97 HF patients with concomitant Af. These patients were divided into 6 groups according to beta-blocker use and tertiles of discharge HR: lowest <60 beats per minute (bpm), middle 61–70 bpm and highest >71 bpm. The primary endpoint was defined as a composite of rehospitalization due to worsening of HF and all-cause mortality. During a median follow-up of 772 days after discharge, the composite cardiovascular outcome occurred in 37 (61%) and 25 (69%) patients with or without beta-blockers, respectively. In the Cox proportional hazard analysis, the lowest HR tertile in patients with beta-blockers was associated with an increased risk of the composite outcome compared with the middle and highest tertiles in both the unadjusted model (hazard ratio: 2.568, 95% confidence interval (CI): 1.089–6.057, p = 0.031; hazard ratio: 2.024, 95% CI: 0.921–4.447, p = 0.079, respectively) and the model adjusted for potential confounders (hazard ratio: 2.631, 95% CI: 1.078–6.421, p = 0.034; hazard ratio: 2.876, 95% CI: 1.147–7.207, p = 0.024, respectively). In patients with HF and Af receiving beta-blockers, low HR adversely increased the risk of cardiovascular events. This fact may blunt the beneficial effects of beta-blockers in patients with HF and Af.
Similar content being viewed by others
Introduction
Atrial fibrillation (Af) is closely related to the increased risk of mortality or rehospitalization in patients with heart failure (HF) [1,2,3]. The severity of HF is correlated with the prevalence of Af, ranging from 4% in NYHA class I–II to 50% in NYHA class III–IV [4]. In Japan, more than one-third of hospitalized patients with HF have Af [5]. Although several studies have shown relatively lower hospital admission rates in Japanese patients with HF complicated with Af compared with Westerners [6], we need to take into account the existence of Af when we treat such patients, considering the deleterious effects of Af in HF patients [5].
On the other hand, beta-blockers are widely used as a recommended therapy for rate-control in HF patients with Af [7]. Although beta-blockers are known to be effective for HF with reduced ejection fraction (HFrEF), such beneficial effects are limited to HF patients with sinus rhythm (SR), and beta-blockers seem to be less effective in patients with coexisting Af [8]. These conflicting results led us to consider the underlying mechanisms. One likely explanation for the reduced effect of beta-blockers in HF patients with Af is the differences in the background characteristics of the study populations or the study designs [9]. Another explanation depends on the complex relationship between beta-blockers and heart rate (HR) in HF patients, particularly in those with Af. A study showed that beta-blockers decreased the risk of mortality regardless of HR in HFrEF patients with SR but not in those with Af [10]. Importantly, tight HR control was correlated with poor survival in patients with advanced chronic HF with Af [11], and a J-curve relationship may exist between HR and clinical outcomes in Af patients [12]. These lines of evidence support the idea that basal HR may determine whether there are beneficial effects of beta-blockers in HF patients with Af.
To test this idea, we investigated the association between HR at discharge following the occurrence of acute decompensated heart failure (ADHF) and cardiovascular outcomes in HF patients with Af treated with beta-blockers.
Methods
Study population
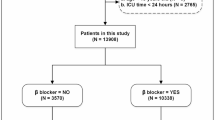
In total, 253 patients with ADHF were admitted to our hospital from 2005 to 2008. After excluding patients who were lost to follow up (n = 15) and patients with SR (n = 141), 97 patients with both HF and Af were eligible for analysis (Fig. 1). Af was confirmed by an expert team of cardiologists based on standard 12-lead electrocardiography performed at the time of hospital admission for ADHF. In this study, Af was defined as atrial fibrillation or atrial flutter. We retrospectively collected HR data from the medical records for the day of discharge. We then divided patients according to beta-blocker use at discharge into groups with or without beta-blockers. Furthermore, these groups were stratified based on the tertiles of discharge HR: HR ≤ 60 beats per minute (bpm), HR 61–70 bpm, and HR ≥ 71 bpm. The rationale for this categorization is as follows. Both the AFFIRM [13] and J-RHYTHM [14] studies targeted HR at rest for 60–80 bpm as the rate-control arms, so we categorized the patients with HRs less than 60 bpm as the bradycardia group. Second, the other patients were divided into two groups by the median value of 70 bpm for HR. The patients admitted to our hospital had strictly controlled, so the maximum HR at discharge was no more than 90 bpm. The estimated glomerular filtration rate (eGFR) was derived from the following equation used for the Japanese population: 194 × serum creatinine−1.094 × age−0.287 (× 0.739 for females). Left ventricular end-diastolic volume and end-systolic volume were obtained using the Teichholz formulae.
Flowchart of study design. ADHF, acute decompensated heart failure, HR, heart rate, bpm, beats per minute
Follow-up
The primary endpoint was defined as a composite of rehospitalization due to worsening of HF and all-cause mortality, including death from any cause or left ventricular assist device implantation, whichever occurred first. The secondary endpoint was defined as rehospitalization due to worsening of HF or all-cause mortality.
Ethics
This study was approved by the National Cerebral and Cardiovascular Center Research Ethics Committee. The committee decided that according to the Japanese Clinical Research Guidelines, the acquisition of informed consent from the chosen subjects was not required because it was a retrospective observational study. Instead, a public announcement was made in accordance with the Ethics Committee’s request and Japanese Clinical Research Guidelines.
Statistical analysis
For baseline characteristics, continuous variables are expressed as medians (interquartile ranges) and categorical variables as numbers (%). The HR tertiles were compared in each of the without beta-blockers and beta-blockers groups using the chi-square test for categorical variables and analysis of variance or Kruskal–Wallis test for continuous variables, as appropriate.
We selected potential confounders for inclusion in the multivariate analysis based on clinical judgment and a review of the relevant literature. Six covariates, namely, age, sex, brain natriuretic peptide (BNP) level at discharge, left atrial diameter (LAD), systolic blood pressure at discharge, and the use of cardiac resynchronization therapy, were included in the model. Missing data among baseline covariates were found for potassium level (n = 1), BNP level at discharge (n = 1) and LAD (n = 1).
We performed Kaplan–Meier survival curve analysis and the log-rank test to calculate the estimated event-free rates and to compare event rates between the HR tertiles. The hazard ratios for the primary and secondary endpoints were analyzed using Cox proportional hazard analysis. All of the tests were two-tailed, and p < 0.05 was considered statistically significant. We performed the statistical analysis using SPSS version 24.0 (SPSS, Chicago, IL, USA).
Results
The patient characteristics are presented in Tables 1 and 2. The baseline characteristics of the patients in the different HR tertiles at discharge, with or without beta-blockers, were compared. In patients without beta-blockers at discharge, there were significant differences among the tertiles with regard to angiotensin-converting enzyme inhibitor usage, interventricular septum thickness, posterior wall thickness and hemoglobin level. The stroke volume (SV) was significantly higher in the HR < 60 bpm group compared with the other groups (Table 1). Conversely, in patients with beta-blockers at discharge, there were no differences in baseline characteristics among the HR strata (Table 2). Furthermore, there were no differences among the tertiles in terms of the BNP level, left ventricular (LV) end-diastolic diameter, LV end-systolic diameter and fractional shortening in the groups with beta-blockers or without beta-blockers.
During a median follow-up of 772 days (197–2062 days) after hospital discharge, the composite outcome occurred in 37 (61%) patients with beta-blockers and 25 (69%) patients without beta-blockers. In the Kaplan–Meier analysis, there were no differences in the event-free rates among the tertiles of HR in patients without beta-blockers (Fig. 2a). In contrast, there were significant differences among the tertiles of HR with beta-blockers (log-rank test: p = 0.040) (Fig. 2b). In the secondary endpoint analysis, the event-free rates of rehospitalization due to worsening of HF (Fig. 3a, b) and all-cause mortality (Fig. 3c, d) were not significantly different among the tertiles of HR in HF patients with Af.
Kaplan–Meier curves for composite outcome (rehospitalization due to worsening of heart failure (HF) or all-cause mortality) in HF and atrial fibrillation patients without beta-blockers (a) or with beta-blockers (b). bpm, beats per minute
Kaplan–Meier curves for rehospitalization due to worsening of heart failure (HF) (a, b) and all-cause mortality (c, d) in HF and atrial fibrillation patients without or with beta-blockers. bpm, beats per minute
In the Cox proportional hazard analysis, there were no differences between the lowest and middle or highest HR tertiles for the composite outcome and secondary endpoints in HF and Af patients without beta-blockers (Table 3). In contrast, the unadjusted model showed that the lowest HR tertile in patients with beta-blockers was associated with an increased risk of the composite outcome compared with the middle and highest tertiles (hazard ratio: 2.568, 95% confidence interval (CI): 1.089–6.057, p = 0.031; hazard ratio: 2.024, 95% CI: 0.921–4.447, p = 0.079, respectively). Furthermore, in the adjusted analysis, the lowest HR tertile in patients with beta-blockers remained significantly associated with a higher risk of the composite outcome compared with the middle and highest HR tertiles (hazard ratio: 2.631, 95% CI: 1.078–6.421, p = 0.034; hazard ratio: 2.876, 95% CI: 1.147–7.207, p = 0.024, respectively).
In addition, in the multivariate analysis of secondary endpoints, the lowest HR tertile in patients with beta-blockers was also associated with an increased risk of rehospitalization due to the worsening of HF compared with the middle and highest tertiles (hazard ratio: 2.322, 95% CI: 0.866–6.229, p = 0.094; hazard ratio: 2.894, 95% CI: 1.011–8.286, p = 0.048, respectively). A similar association between the tertiles of HR and all-cause mortality was also observed (hazard ratio: 2.48, 95% CI: 0.786–7.823, p = 0.121; hazard ratio: 3.571, 95% CI: 1.110–11.495, p = 0.033, respectively) (Table 4).
Discussion
In this study, our investigation revealed that a low HR at discharge was associated with an increased risk of the composite outcome compared with the middle and highest HR tertiles in HF patients with Af who received beta-blockers. In contrast, this association was not observed for those without beta-blockers at discharge. The baseline HR contributes to the beneficial effects of beta-blockers in HF patients with Af.
Beta-blocker therapy in patients with concomitant HR and Af has been extensively investigated, but conclusive evidence is still lacking. A study showed that a reduction in mortality and HF hospitalization could not be achieved by beta-blocker therapy in HFrEF patients with Af [15]. This finding was re-emphasized in the study by Kotecha et al., which showed a neutral impact of beta-blockers on HFrEF patients with Af, in contrast to their SR counterparts [8]. However, another study reported that beta-blockers were associated with reduced mortality risk but not hospitalizations in patients with HFrEF and coexisting Af [16]. Similarly, a large nationwide cohort study revealed that the use of beta-blockers were associated with an approximately 25% risk reduction in all-cause mortality compared to the lack of the use of beta-blockers in HF patients with Af [17]. Furthermore, the Swedish Heart Failure Registry study also found that beta-blockers were still able to potently diminish the risk of mortality in patients with HFrEF with concomitant Af compared with those who did not receive beta-blockers [18]. Although the effect of beta-blockers on mortality is still incompletely understood in patients with HFrEF and Af, beta-blockers have been widely used to reduce HR [19].
The current study showed that low HR was associated with a worse prognosis in HF patients with Af receiving beta-blockers at discharge. The effect of beta-blockers in HF patients with a lower HR has rarely been evaluated because baseline HR < 60–68 bpm was used as an exclusion criterion in most randomized control trials [20,21,22,23,24] A meta-analysis of beta-blocker trials showed that the median HR at baseline in the Af group was 81 bpm, and HR reduction during follow-up was 12 bpm. They showed that HR was not correlated with all-cause mortality in HFrEF patients with Af, regardless of beta-blocker use. However, a careful review of the Kaplan–Meier curve showed that patients with HR ≤ 60 bpm in the beta-blockers group tended to have an increased risk of all-cause mortality compared with those with higher HRs at the interim visit [10]. In this study, the HR ≤ 60 bpm group without beta-blockers showed a significant increase in the SV and a similar incidence of cardiac events. In contrast, the HR ≤ 60 bpm group with beta-blockers showed similar SV and relatively poor prognosis. Cardiac output is determined by SV × HR, which means that the increased SV may compensate for bradycardia in patients with Af. Therefore, the excessive suppression of HR using beta-blockers may lead to the reduction of cardiac output, resulting in an increase in cardiac events. The deleterious effect of low HR in patients with HF with coexisting Af could also be explained through a physiological perspective. The loss of atrial systole in Af impairs LV filling and subsequently decreases cardiac output. Atrial systole can contribute up to 25% of cardiac output and up to 50% in patients with ventricular dysfunction [25]. Therefore, a higher ventricular rate is necessary as a response to the hemodynamic changes to maintain an equivalent cardiac output [4, 26].
Bradycardia is a common side effect during beta-blocker administration [27]. A study showed that the risk of symptomatic bradyarrhythmias in patients taking beta-blockers was associated with age [28]. Moreover, bradycardia is the most common cause of unplanned hospitalization related to the use of beta-blockers and digoxin in elderly patients [29]. Previous studies in the Japanese population have shown that beta-blockers effectively suppress HR in a dose-dependent manner without provoking excessive bradycardia in chronic Af patients [30, 31]. However, patients with chronic HF have sinus node remodeling, characterized by prolonged sinus node recovery and sinoatrial conduction [32], which may increase the likelihood of extreme bradycardia with beta-blocker therapy [27].
In addition to the effect of beta-blockers being partly modulated by HR reduction, beta-blockers also possess an HR-independent mechanism to improve prognosis in patients with HF through the attenuation of sympathetic overactivity, reduction of LV wall stress, exertion of anti-arrhythmic effects, facilitation of the recovering of physical capacity and protection against cardiac sudden death [33]. These properties might provide potential benefits if beta-blocker therapy is continued in patients with asymptomatic bradycardia [34]. However, the administration of beta-blockers in Af patients with bradycardia often results in the development of symptomatic bradycardia, requiring pacemaker implantation. A study showed that among patients with Af, a history of HF and permanent Af were associated with threefold increased odds of worsening bradycardia, requiring a permanent pacemaker [35] In addition, a study enrolled in the rate-control arms of AFFIRM and RACE reported that the strict rate-control in Af patients resulted in increased incidences of pacemaker implantations [36]. Thus, clinicians should make individualized decisions concerning beta-blocker usage, considering the risks and benefits, especially in HF patients with low HR.
In the present study, several limitations should be considered. First, this study was a single-center, observational study, which only included a small number of patients, causing low statistical power. Second, information about beta-blocker use was only provided in the discharge period; therefore, this study was conducted under the assumption of full adherence to outpatient beta-blocker therapy or nonmodified treatment in the group without beta-blockers at the time of discharge. Third, we did not collect information about the doses of beta-blockers at discharge or during follow-up. Fourth, we did not collect information about changes in HR during the follow-up period. Fifth, despite covariate adjustment, we could not completely exclude measured or unmeasured confounding factors. Sixth, we did not classify the types and duration of Af or stratify HF patients based on the ejection fraction. Despite these limitations, the present study provided clinically insightful results regarding the impact of HR at discharge on cardiovascular outcomes in patients with HF and Af treated with beta-blockers.
In conclusion, in patients with HF and Af receiving beta-blockers, low HR adversely increases the risk of cardiovascular events. Low HR may cause bradycardia that may worsen the severity of HF; therefore, we need to pay attention to HR during the administration of beta-blockers in patients with HF and Af.
References
Wang TJ, Larson MG, Levy D, Vasan RS, Leip EP, Wolf PA, et al. Temporal relations of atrial fibrillation and congestive heart failure and their joint influence on mortality: the Framingham Heart Study. Circulation . 2003;107:2920–5.
Koitabashi T, Inomata T, Niwano S, Nishii M, Takeuchi I, Nakano H, et al. Paroxysmal atrial fibrillation coincident with cardiac decompensation is a predictor of poor prognosis in chronic heart failure. Circ J. 2005;69:823–30.
Lip GY, Laroche C, Popescu MI, Rasmussen LH, Vitali-Serdoz L, Dan GA, et al. Heart failure in patients with atrial fibrillation in Europe: a report from the EURObservational Research Programme Pilot survey on Atrial Fibrillation. Eur J Heart Fail. 2015;17:570–82.
Ling LH, Kistler PM, Kalman JM, Schilling RJ, Hunter RJ. Comorbidity of atrial fibrillation and heart failure. Nat Rev Cardiol. 2016;13:131–47.
Hamaguchi S, Yokoshiki H, Kinugawa S, Tsuchihashi-Makaya M, Yokota T, Takeshita A, et al. Effects of atrial fibrillation on long-term outcomes in patients hospitalized for heart failure in Japan: a report from the Japanese Cardiac Registry of Heart Failure in Cardiology (JCARE-CARD). Circ J. 2009;73:2084–90.
Yamashita T. Recent mortality and morbidity rates of Japanese atrial fibrillation patients: racial differences and risk stratification. Circ J. 2013;77:864–8.
JCS Joint Working Group. Guidelines for Pharmacotherapy of Atrial Fibrillation (JCS 2013). Circ J. 2014;78:1997–2021.
Kotecha D, Holmes J, Krum H, Altman DG, Manzano L, Cleland JG, et al. Efficacy of beta blockers in patients with heart failure plus atrial fibrillation: an individual-patient data meta-analysis. Lancet . 2014;384:2235–43.
Khairy P, Roy D. Atrial fibrillation: Challenging the status quo: beta-blockers for HF plus AF. Nat Rev Cardiol. 2014;11:690–2.
Kotecha D, Flather MD, Altman DG, Holmes J, Rosano G, Wikstrand J, et al. Heart rate and rhythm and the benefit of beta-blockers in patients with heart failure. J Am Coll Cardiol. 2017;69:2885–96.
Rienstra M, Van Gelder IC, Van den Berg MP, Boomsma F, Hillege HL, Van Veldhuisen DJ. A comparison of low versus high heart rate in patients with atrial fibrillation and advanced chronic heart failure: effects on clinical profile, neurohormones and survival. Int J Cardiol. 2006;109:95–100.
Andrade JG, Roy D, Wyse DG, Tardif JC, Talajic M, Leduc H, et al. Heart rate and adverse outcomes in patients with atrial fibrillation: a combined AFFIRM and AF-CHF substudy. Heart rhythm. 2016;13:54–61.
Wyse DG, Waldo AL, DiMarco JP, Domanski MJ, Rosenberg Y, Schron EB, et al. A comparison of rate control and rhythm control in patients with atrial fibrillation. N Engl J Med. 2002;347:1825–33.
Yamashita T, Ogawa S, Aizawa Y, Atarashi H, Inoue H, Ohe T, et al. Investigation of the optimal treatment strategy for atrial fibrillation in Japan. Circ J. 2003;67:738–41.
Rienstra M, Damman K, Mulder BA, Van Gelder IC, McMurray JJ, Van Veldhuisen DJ. Beta-blockers and outcome in heart failure and atrial fibrillation: a meta-analysis. Jacc Heart Fail. 2013;1:21–8.
Cadrin-Tourigny J, Shohoudi A, Roy D, Talajic M, Tadros R, Mondesert B, et al. Decreased mortality with beta-blockers in patients with heart failure and coexisting atrial fibrillation: an AF-CHF substudy. Jacc Heart Fail. 2017;5:99–106.
Nielsen PB, Larsen TB, Gorst-Rasmussen A, Skjoth F, Lip GY. Beta-blockers in atrial fibrillation patients with or without heart failure: association with mortality in a Nationwide Cohort Study. Circ Heart Fail. 2016;9:e002597.
Li SJ, Sartipy U, Lund LH, Dahlstrom U, Adiels M, Petzold M, et al. Prognostic significance of resting heart rate and use of beta-blockers in atrial fibrillation and sinus rhythm in patients with heart failure and reduced ejection fraction: findings from the Swedish Heart Failure Registry. Circ Heart Fail. 2015;8:871–9.
Kirchhof P, Benussi S, Kotecha D, Ahlsson A, Atar D, Casadei B, et al. 2016 ESC Guidelines for the management of atrial fibrillation developed in collaboration with EACTS. Eur Heart J. 2016;37:2893–962.
Effect of metoprolol CR/XL in chronic heart failure: Metoprolol CR/XL Randomised Intervention Trial in Congestive Heart Failure (MERIT-HF). Lancet. 1999;353:2001–7.
The Cardiac Insufficiency Bisoprolol Study II (CIBIS-II): a randomised trial. Lancet. 1999;353:9–13.
Dargie HJ. Effect of carvedilol on outcome after myocardial infarction in patients with left-ventricular dysfunction: the CAPRICORN randomised trial. Lancet . 2001;357:1385–90.
Packer M, Bristow MR, Cohn JN, Colucci WS, Fowler MB, Gilbert EM, et al. The effect of carvedilol on morbidity and mortality in patients with chronic heart failure. U.S. Carvedilol Heart Failure Study Group. N Engl J Med. 1996;334:1349–55.
Packer M, Coats AJ, Fowler MB, Katus HA, Krum H, Mohacsi P, et al. Effect of carvedilol on survival in severe chronic heart failure. N Engl J Med. 2001;344:1651–8.
Rahimtoola SH, Ehsani A, Sinno MZ, Loeb HS, Rosen KM, Gunnar RM.Left atrial transport function in myocardial infarction. Importance of its booster pump function.Am J Med. 1975;59:686–94.
Rawles JM. What is meant by a “controlled” ventricular rate in atrial fibrillation? Br heart J. 1990;63:157–61.
Ko DT, Hebert PR, Coffey CS, Curtis JP, Foody JM, Sedrakyan A, et al. Adverse effects of beta-blocker therapy for patients with heart failure: a quantitative overview of randomized trials. Arch Intern Med. 2004;164:1389–94.
Lu HT, Kam J, Nordin RB, Khelae SK, Wang JM, Choy CN, et al. Beta-blocker use and risk of symptomatic bradyarrhythmias: a hospital-based case-control study. J Geriatr Cardiol: Jgc. 2016;13:749–59.
Marcum ZA, Amuan ME, Hanlon JT, Aspinall SL, Handler SM, Ruby CM, et al. Prevalence of unplanned hospitalizations caused by adverse drug reactions in older veterans. J Am Geriatr Soc. 2012;60:34–41.
Inoue H, Atarashi H, Okumura K, Yamashita T, Fukuzawa M, Shiosakai K, et al. Heart rate control by carvedilol in Japanese patients with chronic atrial fibrillation: The AF Carvedilol study. J Cardiol. 2017;69:293–301.
Yamashita T, Inoue H. Heart rate-reducing effects of bisoprolol in Japanese patients with chronic atrial fibrillation: results of the MAIN-AF study. J Cardiol. 2013;62:50–7.
Sanders P, Kistler PM, Morton JB, Spence SJ, Kalman JM. Remodeling of sinus node function in patients with congestive heart failure: reduction in sinus node reserve. Circulation . 2004;110:897–903.
Anzai T. Titration of beta-blockers for patients with heart failure. Circ J. 2013;77:902–3.
Gheorghiade M, Colucci WS, Swedberg K. Beta-blockers in chronic heart failure. Circulation . 2003;107:1570–5.
Barrett TW, Abraham RL, Jenkins CA, Russ S, Storrow AB, Darbar D. Risk factors for bradycardia requiring pacemaker implantation in patients with atrial fibrillation. Am J Cardiol. 2012;110:1315–21.
Van Gelder IC, Wyse DG, Chandler ML, Cooper HA, Olshansky B, Hagens VE. et al. Does intensity of rate-control influence outcome in atrial fibrillation? An analysis of pooled data from the RACE and AFFIRM studies. Europace. 2006;8:935–42.
Acknowledgements
This study was supported by a Grant from the Japan Cardiovascular Research Foundation. The sponsor of the present study had no role in the study design, data collection, data analysis, data interpretation, or writing of the report.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All co-authors of this manuscript have read and approved the submission of the manuscript. All of the authors have made important contributions to the study and are thoroughly familiar with the original data. All authors have completed and submitted the ICMJE Form for Disclosure of Potential Conflicts of Interest: Dr. Kitakaze reports grants and personal fees from Takeda during the study, grants from the Japanese government, grants from the Japan Heart Foundation, grants from the Japan Cardiovascular Research Foundation, grants and personal fees from Asteras, grants and personal fees from Sanofi, personal fees from Daiichi-sankyo, grants and personal fees from Pfizer, grants and personal fees from Ono, personal fees from Bayer, grants and personal fees from Novartis, personal fees from Bheringer, grants and personal fees from Tanabe-mitubishi, personal fees from Kowa, grants and personal fees from Kyowa-hakko-kirin, personal fees from Dainihon-sumitomo, personal fees from Sawai, personal fees from MSD, grants and personal fees from Abott, grants and personal fees from Otsuka, grants from Calpis, grants from Nihon Kohden, personal fees from Shionogi, personal fees from Astrazeneca, personal fees from Asahikasei Med., personal fees from Novo Nordisk, personal fees from Fuji-film RI, and personal fees from Japan Medical Data, none of which pertained to the submitted work.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Hudoyo, A., Fukuda, H., Imazu, M. et al. Heart rate determines the beneficial effects of beta-blockers on cardiovascular outcomes in patients with heart failure and atrial fibrillation. Hypertens Res 42, 1716–1725 (2019). https://doi.org/10.1038/s41440-019-0289-4
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41440-019-0289-4
Keywords
This article is cited by
-
Validation of an ambulatory blood pressure monitoring device employing a novel method to detect atrial fibrillation
Hypertension Research (2022)
-
Heart rate control using beta-blockers for heart failure with atrial fibrillation: more than enough is too much
Hypertension Research (2019)