Abstract
Circulating saturated fatty acids (SFAs) have been associated with cardiovascular disease. However, little is known about the relationship of SFAs with the risk of pregnancy-induced hypertension (PIH). We conducted a nested case–control study to examine the associations between circulating SFAs and the risk of PIH. A total of 92 PIH cases were matched to 184 controls by age (±2 years) and infant sex from a birth cohort study conducted in Wuhan, China. Levels of circulating fatty acids in plasma were measured using gas chromatography–mass spectrometry. Conditional logistic regressions were conducted to calculate odds ratios (ORs) and 95% confidence intervals (95% CIs). Even-chain SFAs, including myristic acid (14:0) and palmitic acid (16:0), were positively associated with the risk of PIH [ORs (95% CIs): 2.92 (1.27, 6.74) for 14:0 and 2.85 (1.18, 6.89) for 16:0, % by wt]. In contrast, higher levels of very-long-chain SFAs, including arachidic acid (20:0), behenic acid (22:0), and lignoceric acid (24:0), were associated with a lower risk of PIH [ORs (95% CIs): 0.40 (0.17, 0.92) for 20:0, 0.30 (0.12, 0.71) for 22:0 and 0.26 (0.11, 0.64) for 24:0, μg/mL]. For odd-chain SFAs, including pentadecanoic acid (15:0) and heptadecanoic acid (17:0), no significant difference was observed. Our results provided convincing evidence that different subclasses of SFAs showed diverse effects on the risk of PIH. This suggests that dietary very-long-chain SFAs may be a novel means by which to prevent hypertension. Future studies are required to confirm these associations and elucidate the underlying mechanisms.
Similar content being viewed by others
Introduction
Pregnancy-induced hypertension (PIH) is one of the leading causes of maternal and fetal morbidity and mortality from conception to birth [1,2,3]. Increasing evidence has shown that PIH can lead to multiple irreversible health issues for mothers and their infants. For mothers, it can increase the risk of essential hypertension and cardiovascular and renal diseases later in life [4,5,6,7]. For infants, PIH is associated with adverse outcomes, including preterm birth, low birth weight, and neonatal death, and may even cause the future development of cardiovascular disease in childhood [7,8,9,10]. Therefore, the prevention of PIH by feasible intervention strategies is very valuable for the health of mothers and their infants.
Saturated fatty acids (SFAs) are a unique group of fatty acids due to the absence of double bonds between the carbons. It is generally believed that SFAs have adverse effects on human health and has been suggested that reducing the intake of dietary SFAs may prevent the occurrence and development of some chronic noncommunicable diseases [11, 12]. However, the evidence supporting the adverse effects of high SFA intake remains inconsistent and inadequate [13]. Some studies have even observed no association between reduced SFA consumption and incident stroke, diabetes, hypertension, and coronary heart disease [14,15,16]. Therefore, these inconsistencies prompted us to doubt whether all kinds of SFAs exert adverse effects on human health. Recently, a growing number of studies have indicated that different subclasses of SFAs classified by their numbers of carbon atoms have disparate impacts on human health [17,18,19]. Several human and animal studies have revealed positive associations of circulating levels of even-chain SFAs, including myristic acid (14:0), palmitic acid (16:0), and stearic acid (18:0), with levels of total cholesterol and risk of diabetes, hypertension, and atherosclerosis [20,21,22]. Findings from two large prospective cohort studies showed that higher circulating levels of odd-chain SFAs, including pentadecanoic acid (15:0) and heptadecanoic acid (17:0), might be associated with lower risks of type 2 diabetes and pancreatic cancer [23, 24]. In addition, emerging evidence indicated that very-long-chain SFAs with 20 or more carbon atoms, including arachidic acid (20:0), behenic acid (22:0), and lignoceric acid (24:0), may be conducive to reducing the occurrence of metabolic disorders such as diabetes, cardiovascular disease, and cancer [25,26,27]. Many studies have explored the relationships between circulating SFAs and cardiovascular diseases, such as coronary heart disease and stroke [26, 28]. To our knowledge, only a few studies have investigated the effects of circulating fatty acids on blood pressure or hypertension, and they focused on polyunsaturated fatty acids [29,30,31,32,33,34]. Epidemiologic evidence on the role of SFAs in hypertension, especially during pregnancy, is sparse.
Thus, we carried out a nested case–control study in the Hubei Province of China to investigate the associations between levels of circulating SFAs with different chain lengths and the risk of PIH among pregnant women.
Methods
Study design and population
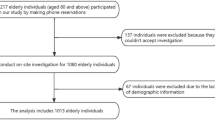
We performed a nested case–control study of PIH based on a prospective birth cohort study conducted at Wuhan Medical & Healthcare Center for Women and Children in Wuhan, China. From October 2013 to September 2016, a total of 4297 pregnant women were recruited at their first prenatal visit in this hospital. Eligibility criteria included residents of Wuhan who had no Chinese communication problem and who decided to deliver in the study hospital. All participants were invited to attend face-to-face interviews and provide urine and blood samples during the routine antenatal examinations. Of the pregnant women, we excluded 527 with hypertension, diabetes, or renal disease before pregnancy; with a family history of hypertension or diabetes; and who did not provide a blood sample for fatty acid measurements, leaving 3770 participants for the final analysis, among whom 92 PIH cases were identified based on a doctor's diagnosis identified in medical records. PIH, including gestational hypertension and preeclampsia, was defined according to the criteria of the International Society for the Study of Hypertension [35]. Controls were selected randomly from the remaining participants who were free of PIH, and they were individually 2:1 matched to PIH cases by age (±2 years) and infant sex. Thus, a total of 92 cases and 184 controls were included in the study. All participants provided written informed consent at enrollment. The study was approved by the ethics committees of Tongji Medical College, Huazhong University of Science and Technology and the study hospital.
Plasma fatty acid measurements
Fasting blood samples of the participants were collected early in the pregnancy (mean ± standard deviation: 13.3 ± 1.2 weeks) to separate plasma and were then stored at −80 °C until further analysis.
Concentrations of fatty acids in plasma were determined by gas chromatography–mass spectrometry (GC–MS, 5975C MS/7890A GC, Agilent Technologies, Santa Clara, CA). Plasma lipids were extracted using the modified Folch method [36]. Briefly, methanol (2 ml) and chloroform (4 ml) were added to plasma samples diluted with physiological saline. The mixture was vortexed and left for 5 min and then centrifuged at 1700 g for 10 min. The lower phase (chloroform) was collected and dried under a mild nitrogen stream. Then, the residue was derivated by adding 2 mL of 1% sulfuric acid/methanol to prepare fatty acid methyl esters (FAMEs). After heptane extraction, the FAMEs were transferred into a glass insert in a vial with a cap for GC–MS analysis. FAMEs were separated on an Agilent DB-23 capillary column (60 m × 0.25 mm; film thickness 0.25 μm) at a helium flow of 1.2 mL/min using the following programmed temperature conditions: the initial oven temperature was set at 50 °C and maintained at this temperature for 1 min, increased to 155 °C at 20 °C/min and maintained at 155 °C for 2 min, and then increased to 230 °C at 5 °C/min and maintained at 230 °C for 6 min. The sample was injected by a syringe in split mode (20:1), and the temperature of the injector port was set at 250 °C. In mass spectrometry, electron impact ionization mode was applied to keep the ion source temperature at 230 °C. The quadrupole and transfer line temperatures were set at 150 °C and 230 °C, respectively. The full scan in the 40–400 m/z range and selected ion monitoring mode were used for qualitative and quantitative analyses of fatty acids. The selected ions and retention times for fatty acid detection are shown in Supplementary Table 1. The absolute concentrations of fatty acids were determined in accordance with established working curves of commercial standards and the amount of internal standard. To minimize the effect of blood lipid levels on fatty acids, we calculated the percentage of total fatty acids by weight (% by wt) based on the absolute concentrations (μg/ml). Matched samples from cases and controls were processed and assayed in the same batch. For each batch of plasma samples, we included quality control samples and procedural and reagent blanks. The coefficients of variation of fatty acids, which were assessed by measuring quality control samples, ranged from 0.71 to 9.52%.
Covariates
Baseline information on demographic and socioeconomic characteristics (e.g., maternal age, occupation, education, and household income), lifestyle factors (e.g., active smoking, passive smoking, alcohol use, and physical activity during pregnancy) and nutritional supplements (calcium, iron, and multivitamin supplementation during pregnancy) was obtained from interviews administered by well-trained nurses within three days before or after delivery. Medical information including parity, type of delivery, family history, and medical history of illness, and infant’s birth date, sex, birth weight (in g), and birth length (in cm) were obtained from medical records. Gestational age was calculated in weeks based on the dates of last menstrual period (LMP) and the infant’s birth. Preterm delivery was defined as delivery prior to 37 weeks gestational age. Low birth weight (LBW) was defined as a birth weight less than 2500 g. Small for gestational age (SGA) was defined as a birth weight below the 10th percentile for gestational age by infant sex [37]. The prepregnancy body mass index (BMI) was calculated using the self-reported prepregnancy body weight in kilograms and height in meters, which was measured by a stadiometer in the hospital. The gestational week of plasma collection was calculated based on plasma collection date and the LMP. The diagnosis of gestational diabetes mellitus (GDM) was conducted between 24 and 28 weeks gestation for all pregnant women according to the recommended criteria of the International Association of Diabetes and Pregnancy Study Group [38].
Statistical analysis
General characteristics between controls and cases were compared using Student’s t tests for continuous variables and Chi-square tests for categorical variables. Medians (25th–75th percentiles) of plasma SFAs expressed as absolute concentrations and percentages of weight of total fatty acids are described. The Wilcoxon rank sum test was performed to compare the differences between controls and cases.
We used conditional logistic regression models to calculate odds ratios (ORs) and 95% confidence intervals (95% CIs) for the associations of SFAs with the risk of PIH. Individual and subclasses of SFAs were analyzed as categorical variables based on their tertile distributions among controls (the lowest tertile was defined as the referent group). All models were adjusted for parity, education level, prepregnancy BMI, passive smoking, physical activity, iron supplementation, and gestational week of blood collection based on the significant differences in the bivariate analyses and data published in previous studies [39,40,41,42,43]. Active smoking and alcohol use during pregnancy were not included in the models because few pregnant women reported the use of these two. Tests of linear trends were conducted by modeling the median value of each tertile of the SFAs as continuous variables. The false discovery rate correction was performed on p values for trends to account for multiple tests [44]. Considering the significant association of prepregnancy BMI with the risk of PIH [42, 45], we further estimated model parameters using unconditional logistic regression models in analyses stratified by prepregnancy BMI to minimize the effect of prepregnancy BMI on the association of SFAs and risk of PIH. Due to the limited number of underweight or overweight pregnant women, the model tests were only performed among pregnant women with normal prepregnancy BMI in the stratified analysis. In view of the similar risk factors for PIH and GDM, sensitivity analysis after excluding the participants with GDM was also conducted. In the sensitivity analyses, the matched set was excluded if the case or any one of the two matched controls were identified as having GDM.
All of the tests were two-sided, and statistical significance levels were set at α = 0.05. Statistical analyses were conducted using SAS version 9.4 (SAS Institute Inc., Cary, NC, USA).
Results
General characteristics of study participants
Table 1 presents the general characteristics of PIH cases and controls. The average age of the 276 participants was 29.4 years, and 67.4% of them had male infants. As expected, PIH cases had significantly higher prepregnancy BMIs than controls (P < 0.0001). The proportions of pregnant women who were overweight or obese in the PIH case and control groups were 39.1% and 7.1%, respectively. PIH cases had a younger gestational age at delivery and were more likely than controls to deliver infants before 37 weeks and to deliver LBW or SGA infants (P < 0.05). No significant differences were observed between the cases and controls with regard to education level, parity, passive smoking, nutritional supplements, or physical activity during pregnancy (P > 0.05).
Levels of plasma SFAs in PIH cases and controls
The levels of plasma fatty acids (μg/mL and % by wt) in PIH cases and controls are shown in Table 2. In both PIH cases and controls, the SFAs with the highest levels in plasma were 16:0 (median: 490.13 μg/mL in the controls and 516.9 μg/mL in the cases), and the lowest was 24:0 (median: 2.07 μg/mL in the controls and 1.46 μg/mL in the cases). With respect to the composition of fatty acids, ~30% of total fatty acids were SFAs, with the largest contributors being even-chain SFAs (16:0: nearly 22%; 18:0: nearly 6.5%), while odd-chain SFAs and very-long-chain SFAs were present in low relative abundance (all < 0.6%). Compared with controls, PIH cases had significantly lower levels of very long chain SFAs (including 20:0, 22:0, 24:0 and sum of very-long-chain SFAs, % by wt and μg/mL) and odd-chain SFAs (including 17:0 and sum of odd-chain SFAs, % by wt) but higher levels of even-chain SFAs (including 14:0, 16:0, and sum of even-chain SFAs, % by wt) (Table 2).
Associations between maternal plasma SFA levels and the risk of pregnancy-induced hypertension
In general, the risk of PIH was negatively associated with very-long-chain SFAs and was positively associated with even-chain SFAs (Table 3). For very long chain SFAs, higher levels of circulating 20:0, 22:0, and 24:0 were significantly associated with a lower risk of PIH. The adjusted ORs (95% CIs) from the comparison of tertile 3 versus tertile 1 were 0.40 (95% CI: 0.17, 0.92; p-Trend = 0.02) for 20:0, 0.30 (95% CI: 0.12, 0.71; p-Trend = 0.007) for 22:0, and 0.26 (95% CI: 0.11, 0.64; p-Trend = 0.002) for 24:0. A similar association was also observed for the sum of very-long-chain SFAs [OR (95% CI) = 0.29 (0.12, 0.70); p-Trend = 0.005]. In addition, 24:0 expressed as a percentage was associated with a 61% decreased risk of PIH [OR (95% CI) = 0.39 (0.17, 0.92); p-Trend = 0.03], comparing tertile 3 to tertile 1. After multiple testing correction, these negative associations remained significant (p-FDR < 0.05). In contrast, a positive association of even-chain SFAs with the risk of PIH was observed. The adjusted ORs (95% CIs) of PIH comparing tertile 3 to tertile 1 of fatty acid percentages were 2.92 (95% CI: 1.27, 6.74; p-Trend = 0.02) for 14:0 and 2.85 (95% CI: 1.18, 6.89; p-Trend = 0.02) for 16:0. For odd-chain SFAs, no significant difference was observed in the adjusted model. When calculating the sum of all SFAs, we did not find a significant association with the risk of PIH.
Sensitivity analyses
Among pregnant women with normal prepregnancy BMI, very-long-chain SFAs were negatively associated with the risk of PIH, whereas even-chain SFAs were positively associated with the risk of PIH. For example, the results of adjusted models showed that the ORs (95% CIs) of PIH from the comparison of tertile 3 to tertile 1 were 0.36 (95% CI: 0.14, 0.97; p-Trend = 0.03) for 22:0, and 2.75 (95% CI: 1.06, 7.14; p-Trend = 0.03) for 16:0 (Supplementary Table 2). After excluding the participants with GDM, significant associations of very-long-chain SFAs with the risk of PIH were still observed (Supplementary Table 3).
Discussion
In the present study, we found that different subclasses of SFAs had distinct patterns of association with the risk of PIH. All individual very-long-chain SFAs and the sum of very-long-chain SFAs were negatively correlated with the risk of PIH, whereas 14:0, 16:0, and the sum of even-chain SFAs were positively correlated with the risk of PIH. No significant relationships were observed for 15:0, 17:0, or the sum of odd-chain SFAs.
For studies of fatty acids and blood pressure or hypertension, many researchers have focused on the effect of PUFAs, especially n-3 and n-6 PUFAs [29,30,31,32,33]. For example, one study from a birth cohort found that higher levels of plasma n-3 PUFAs were associated with a lower risk of PIH [32]. However, to date, the role of SFAs in blood pressure or hypertension has not been reported as a special topic. As far as we know now, only one study explored the association of SFAs with blood pressure among pregnant women and showed a positive association between serum total SFA concentrations and systolic blood pressure levels [46]. However, our study did not find a relationship between the sum of SFAs and the risk of PIH. A prospective study in US men also reported that SFA intake was uncorrelated with incident hypertension [16]. The discrepancy may be due to differences in the participant characteristics, biological samples, and study designs. Interestingly, consistent with previous studies among nonpregnant populations [21, 46], we also observed that even-chain SFAs, including 14:0 and 16:0, were positively correlated with the risk of hypertension. A community study of middle-aged adults in the USA reported that the baseline levels of 16:0 in plasma cholesterol esters were significantly higher in hypertensive patients than normotensive patients [21]. Similarly, a retrospective study indicated that positive linear associations of 14:0 and 16:0 with blood pressure existed in middle-aged adults [47]. For very-long-chain SFAs, we found that elevated levels of 20:0, 22:0, 24:0, and the sum of very-long-chain SFAs were significantly associated with a lower risk of PIH. Sensitivity analysis after excluding the participants with GDM also showed the same direction of association. Although no other studies have investigated the relationship of very-long-chain SFAs with hypertension, numerous recent studies have shown negative associations of very-long-chain SFAs with multiple health outcomes, including diabetes, cardiovascular diseases, and cancers [17, 25,26,27]. Taken together, these studies suggest potential beneficial effects of very long chain SFAs and provide a great incentive for further research on this subclass of SFAs.
The biological mechanisms underlying the different associations of SFA subclasses with the risk of PIH are unknown. Increasing evidence in vitro and in vivo suggests that inflammation and apoptosis play important roles in the pathophysiological processes of hypertension, which can potentially cause vascular endothelial injury and peripheral vascular resistance, further leading to hypertension [48, 49]. Considering these pathophysiological factors, various plausible mechanisms may support our findings. Multiple studies have affirmed that very-long-chain SFAs are associated with the resolution of inflammation, whereas even-chain SFAs can promote inflammation [50,51,52]. Additional findings have also demonstrated that high levels of very-long-chain SFAs may indirectly reflect high activity of peroxisome proliferator activated receptor α (PPARα), which may trigger the synthesis of very-long-chain SFAs [53, 54]. After PPARα activation, the inflammatory process could be restrained by blocking the transcription of genes related to inflammation and by inhibiting de novo lipogenesis, resulting in lower endogenous even-chain SFA production [53,54,55]. Therefore, different SFAs could cause changes in PIH risk by modulating inflammation. In addition, data from in vitro studies indicated that ceramides with distinct chain lengths of fatty acids play different roles in apoptosis [50, 56]. It is well known that apoptosis in vascular endothelial cells can promote the occurrence and progression of hypertension [48, 57]. The evidence from several groups suggests that 16:0 ceramides may induce apoptosis, whereas 22:0 and 24:0 ceramides may prevent apoptosis [50, 58]. Accordingly, it is reasonable for us to assume that ceramides containing different SFAs may affect the risk of PIH by regulating the apoptosis of vascular endothelial cells. As discussed above, the diverse direction of the effects of SFA subclasses on the risk of PIH may be attributed to their different biological actions, including modulation of inflammation and vascular endothelial apoptosis.
Our study has several strengths. The nested case–control design enabled us to select all PIH cases and matched control subjects to avoid selection bias. In addition, we objectively evaluated levels of circulating fatty acids by measuring fatty acids in plasma instead of by dietary survey, which may result in reporting bias. More importantly, circulating fatty acids are derived from exogenous and endogenous sources. As the exogenous source, dietary intake of fatty acids could not reflect all circulating fatty acid levels. Finally, the detailed record of information on sociodemographic characteristics and medical records may provide reliable data on potential confounders and reduce recall bias. However, some limitations should also be mentioned. First, causality cannot be established due to the observational design. In addition, we cannot control the effect of other unmeasured factors on the results. For example, detailed dietary factors, dietary salt intake and biochemical indicators were not considered, which may partly affect the relationship of fatty acids with the risk of PIH. Last, since the participants in our study came from a birth cohort in China, the research findings may not be generalizable to other ethnic and racial groups.
Our results suggest that pregnant women with higher levels of very-long-chain SFAs may have a lower risk of PIH, whereas higher levels of even-chain SFAs may increase the risk of PIH. These novel findings provide convincing epidemiological evidence that different circulating SFAs exert potential diverse effects on the risk of PIH. The conventional attitudes toward the health effects of SFAs should be updated. Dietary very-long-chain SFAs may be a novel means by which to prevent hypertension. Future studies are required to confirm these associations in other larger populations and elucidate the underlying mechanisms.
References
Khan KS, Wojdyla D, Say L, Gulmezoglu AM, Van Look PF. WHO analysis of causes of maternal death: a systematic review. Lancet. 2006;367:1066–74.
Ghulmiyyah L, Sibai B. Maternal mortality from preeclampsia/eclampsia. Semin Perinatol. 2012;36:56–9.
Jwa SC, Arata N, Sakamoto N, Watanabe N, Aoki H, Kurauchi-Mito A, et al. Prediction of pregnancy-induced hypertension by a shift of blood pressure class according to the JSH 2009 guidelines. Hypertens Res. 2011;34:1203–8.
Kristensen JH, Basit S, Wohlfahrt J, Damholt MB, Boyd HA. Pre-eclampsia and risk of later kidney disease: nationwide cohort study. BMJ. 2019;365:l1516.
Behrens I, Basit S, Melbye M, Lykke JA, Wohlfahrt J, Bundgaard H, et al. Risk of post-pregnancy hypertension in women with a history of hypertensive disorders of pregnancy: nationwide cohort study. BMJ. 2017;358:j3078.
Bellamy L, Casas JP, Hingorani AD, Williams DJ. Pre-eclampsia and risk of cardiovascular disease and cancer in later life: systematic review and meta-analysis. BMJ. 2007;335:974.
Mol BWJ, Roberts CT, Thangaratinam S, Magee LA, de Groot CJM, Hofmeyr GJ. Pre-eclampsia. Lancet 2016;387:999–1011.
Hu J, Li Y, Zhang B, Zheng T, Li J, Peng Y, et al. Impact of the 2017 ACC/AHA guideline for high blood pressure on evaluating gestational hypertension-associated risks for newborns and mothers. Circ Res 2019;125:184–94.
Alsnes IV, Vatten LJ, Fraser A, Bjorngaard JH, Rich-Edwards J, Romundstad PR, et al. Hypertension in pregnancy and offspring cardiovascular risk in young adulthood: prospective and sibling studies in the HUNT study (Nord-Trondelag Health Study) in Norway. Hypertension. 2017;69:591–8.
Lane-Cordova AD, Gunderson EP, Carnethon MR, Catov JM, Reiner AP, Lewis CE, et al. Pre-pregnancy endothelial dysfunction and birth outcomes: The Coronary Artery Risk Development in Young Adults (CARDIA) study. Hypertens Res. 2018;41:282–9.
Lichtenstein AH, Appel LJ, Brands M, Carnethon M, Daniels S, Franch HA, et al. Diet and lifestyle recommendations revision 2006: a scientific statement from the American Heart Association Nutrition Committee. Circulation. 2006;114:82–96.
Eyre H, Kahn R, Robertson RM. Preventing cancer, cardiovascular disease, and diabetes: a common agenda for theAmerican Cancer Society, the American Diabetes Association, and the American Heart Association. CA Cancer J Clin. 2004;54:190–207.
Micha R, Mozaffarian D. Saturated fat and cardiometabolic risk factors, coronary heart disease, stroke, and diabetes: a fresh look at the evidence. Lipids. 2010;45:893–905.
de Souza RJ, Mente A, Maroleanu A, Cozma AI, Ha V, Kishibe T, et al. Intake of saturated and trans unsaturated fatty acids and risk of all cause mortality, cardiovascular disease, and type 2 diabetes: systematic review and meta-analysis of observational studies. BMJ. 2015;351:h3978.
Howard BV, Van Horn L, Hsia J, Manson JE, Stefanick ML, Wassertheil-Smoller S, et al. Low-fat dietary pattern and risk of cardiovascular disease: the Women's Health Initiative Randomized Controlled Dietary Modification Trial. JAMA. 2006;295:655–66.
Ascherio A, Rimm EB, Giovannucci EL, Colditz GA, Rosner B, Willett WC, et al. A prospective study of nutritional factors and hypertension among US men. Circulation. 1992;86:1475–84.
Fretts AM, Mozaffarian D, Siscovick DS, Djousse L, Heckbert SR, King IB, et al. Plasma phospholipid saturated fatty acids and incident atrial fibrillation: the Cardiovascular Health Study. J Am Heart Assoc. 2014;3:e000889.
Zheng JS, Sharp SJ, Imamura F, Koulman A, Schulze MB, Ye Z, et al. Association between plasma phospholipid saturated fatty acids and metabolic markers of lipid, hepatic, inflammation and glycaemic pathways in eight European countries: a cross-sectional analysis in the EPIC-InterAct study. BMC Med. 2017;15:203.
Forouhi NG, Koulman A, Sharp SJ, Imamura F, Kroger J, Schulze MB, et al. Differences in the prospective association between individual plasma phospholipid saturated fatty acids and incident type 2 diabetes: the EPIC-InterAct case–cohort study. Lancet Diabetes Endocrinol. 2014;2:810–8.
Patel PS, Sharp SJ, Jansen E, Luben RN, Khaw KT, Wareham NJ, et al. Fatty acids measured in plasma and erythrocyte-membrane phospholipids and derived by food-frequency questionnaire and the risk of new-onset type 2 diabetes: a pilot study in the European Prospective Investigation into Cancer and Nutrition (EPIC)-Norfolk cohort. Am J Clin Nutr. 2010;92:1214–22.
Zheng ZJ, Folsom AR, Ma J, Arnett DK, McGovern PG, Eckfeldt JH. Plasma fatty acid composition and 6-year incidence of hypertension in middle-aged adults: the Atherosclerosis Risk in Communities (ARIC) study. Am J Epidemiol. 1999;150:492–500.
Afonso MS, Lavrador MS, Koike MK, Cintra DE, Ferreira FD, Nunes VS, et al. Dietary interesterified fat enriched with palmitic acid induces atherosclerosis by impairing macrophage cholesterol efflux and eliciting inflammation. J Nutr Biochem. 2016;32:91–100.
Matejcic M, Lesueur F, Biessy C, Renault AL, Mebirouk N, Yammine S, et al. Circulating plasma phospholipid fatty acids and risk of pancreatic cancer in a large European cohort. Int J Cancer. 2018;143:2437–48.
Santaren ID, Watkins SM, Liese AD, Wagenknecht LE, Rewers MJ, Haffner SM, et al. Serum pentadecanoic acid (15:0), a short-term marker of dairy food intake, is inversely associated with incident type 2 diabetes and its underlying disorders. Am J Clin Nutr. 2014;100:1532–40.
Chiu YH, Bertrand KA, Zhang S, Laden F, Epstein MM, Rosner BA, et al. A prospective analysis of circulating saturated and monounsaturated fatty acids and risk of non-Hodgkin lymphoma. Int J Cancer. 2018;143:1914–22.
Malik VS, Chiuve SE, Campos H, Rimm EB, Mozaffarian D, Hu FB, et al. Circulating very-long-chain saturated fatty acids and incident coronary heart disease in US men and women. Circulation. 2015;132:260–8.
Lemaitre RN, Fretts AM, Sitlani CM, Biggs ML, Mukamal K, King IB, et al. Plasma phospholipid very-long-chain saturated fatty acids and incident diabetes in older adults: the Cardiovascular Health Study. Am J Clin Nutr. 2015;101:1047–54.
Yamagishi K, Iso H, Yatsuya H, Tanabe N, Date C, Kikuchi S, et al. Dietary intake of saturated fatty acids and mortality from cardiovascular disease in Japanese: the Japan Collaborative Cohort Study for Evaluation of Cancer Risk (JACC) Study. Am J Clin Nutr. 2010;92:759–65.
Filipovic MG, Aeschbacher S, Reiner MF, Stivala S, Gobbato S, Bonetti N, et al. Whole blood omega-3 fatty acid concentrations are inversely associated with blood pressure in young, healthy adults. J Hypertens. 2018;36:1548–54.
Bonaa KH, Bjerve KS, Straume B, Gram IT, Thelle D. Effect of eicosapentaenoic and docosahexaenoic acids on blood pressure in hypertension. A population-based intervention trial from the Tromso study. N. Engl J Med. 1990;322:795–801.
Dokholyan RS, Albert CM, Appel LJ, Cook NR, Whelton P, Hennekens CH. A trial of omega-3 fatty acids for prevention of hypertension. Am J Cardiol. 2004;93:1041–3.
Lim WY, Chong M, Calder PC, Kwek K, Chong YS, Gluckman PD, et al. Relations of plasma polyunsaturated Fatty acids with blood pressures during the 26th and 28th week of gestation in women of Chinese, Malay, and Indian ethnicity. Medicine. 2015;94:e571.
Begg DP, Sinclair AJ, Stahl LA, Premaratna SD, Hafandi A, Jois M, et al. Hypertension induced by omega-3 polyunsaturated fatty acid deficiency is alleviated by alpha-linolenic acid regardless of dietary source. Hypertens Res. 2010;33:808–13.
Qiao GH, Sun XZ. Increased plasma fatty acid binding protein 4 concentration at the first prenatal visit and its relevance to preeclampsia. Hypertens Res. 2018;41:763–9.
Brown MA, Lindheimer MD, de Swiet M, Van Assche A, Moutquin JM. The classification and diagnosis of the hypertensive disorders of pregnancy: statement from the International Society for the Study of Hypertension in Pregnancy (ISSHP). Hypertens Pregnancy. 2001;20:Ix–xiv.
Folch J, Lees M, Sloane Stanley GH. A simple method for the isolation and purification of total lipides from animal tissues. J Biol Chem. 1957;226:497–509.
Mikolajczyk RT, Zhang J, Betran AP, Souza JP, Mori R, Gulmezoglu AM, et al. A global reference for fetal-weight and birthweight percentiles. Lancet. 2011;377:1855–61.
Association AD. Diagnosis and classification of diabetes mellitus. Diabetes Care. 2011;34(Suppl 1):S62–9.
Davenport MH, Ruchat SM, Poitras VJ, Jaramillo Garcia A, Gray CE, Barrowman N, et al. Prenatal exercise for the prevention of gestational diabetes mellitus and hypertensive disorders of pregnancy: a systematic review and meta-analysis. Br J Sports Med. 2018;52:1367–75.
Leng B, Jin Y, Li G, Chen L, Jin N. Socioeconomic status and hypertension: a meta-analysis. J Hypertens. 2015;33:221–9.
Simon JA, Fong J, Bernert JT Jr., Browner WS. Relation of smoking and alcohol consumption to serum fatty acids. Am J Epidemiol. 1996;144:325–34.
Lane-Cordova AD, Tedla YG, Carnethon MR, Montag SE, Dude AM, Rasmussen-Torvik LJ. Pre-pregnancy blood pressure and body mass index trajectories and incident hypertensive disorders of pregnancy. Pregnancy Hypertens. 2018;13:138–40.
Iwama N, Metoki H, Nishigori H, Mizuno S, Takahashi F, Tanaka K, et al. Association between alcohol consumption during pregnancy and hypertensive disorders of pregnancy in Japan: the Japan Environment and Children's Study. Hypertens Res. 2019;42:85–94.
Benjamini Y, Hochberg Y. Controlling the false discovery rate: a practical and powerful approach to multiple testing. J R Stat Soc Ser B. 1995;57:289–300.
Zhou A, Xiong C, Hu R, Zhang Y, Bassig BA, Triche E, et al. Pre-pregnancy BMI, gestational weight gain, and the risk of hypertensive disorders of pregnancy: a cohort study in Wuhan, China. PLoS ONE. 2015;10:e0136291.
Lepsch J, Farias DR, Eshriqui I, Rebelo F, Dos Santos Vaz J, Adegboye AA, et al. Serum fatty acids are positively associated with changes in systemic blood pressure throughout pregnancy. Pregnancy Hypertens. 2018;13:7–13.
Kim SR, Jeon SY, Lee SM. The association of cardiovascular risk factors with saturated fatty acids and fatty acid desaturase indices in erythrocyte in middle-aged Korean adults. Lipids Health Dis. 2015;14:133.
Granger JP, Alexander BT, Bennett WA, Khalil RA. Pathophysiology of pregnancy-induced hypertension. Am J Hypertens. 2001;14:178s–85s.
Ali SM, Khalil RA. Genetic, immune and vasoactive factors in the vascular dysfunction associated with hypertension in pregnancy. Exp Opin Ther Targets. 2015;19:1495–515.
Grosch S, Schiffmann S, Geisslinger G. Chain length-specific properties of ceramides. Prog Lipid Res. 2012;51:50–62.
Kihara A. Very long-chain fatty acids: elongation, physiology and related disorders. J Biochem. 2012;152:387–95.
Harvey KA, Walker CL, Pavlina TM, Xu Z, Zaloga GP, Siddiqui RA. Long-chain saturated fatty acids induce pro-inflammatory responses and impact endothelial cell growth. Clin Nutr. 2010;29:492–500.
Jump DB, Tripathy S, Depner CM. Fatty acid-regulated transcription factors in the liver. Annu Rev Nutr. 2013;33:249–69.
Wahli W, Michalik L. PPARs at the crossroads of lipid signaling and inflammation. Trends Endocrinol Metab. 2012;23:351–63.
Siler SQ, Neese RA, Hellerstein MK. De novo lipogenesis, lipid kinetics, and whole-body lipid balances in humans after acute alcohol consumption. Am J Clin Nutr. 1999;70:928–36.
Crowder CM. Cell biology. Ceramides-friend or foe in hypoxia? Science. 2009;324:343–4.
Ngene NC, Moodley J. Role of angiogenic factors in the pathogenesis and management of pre-eclampsia. Int J Gynaecol Obstet. 2018;141:5–13.
Hartmann D, Lucks J, Fuchs S, Schiffmann S, Schreiber Y, Ferreiros N, et al. Long chain ceramides and very long chain ceramides have opposite effects on human breast and colon cancer cell growth. Int J Biochem Cell Biol. 2012;44:620–8.
Acknowledgements
We thank all the participants, staff, and students involved in the birth cohort study.
Funding
This study was supported by grants from the National Key Research and Development Plan (2016YFC0206700, 2016YFC0206203), the National Natural Science Foundation of China (91743103, 21437002, 91643207), the Fundamental Research Funds for the Central Universities, Huazhong University of Science and Technology (2018KFYXMPT00), and the Program for HUST Academic Frontier Youth Team (2018QYTD12).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Li, X., Huang, Y., Zhang, W. et al. Association of circulating saturated fatty acids with the risk of pregnancy-induced hypertension: a nested case–control study. Hypertens Res 43, 412–421 (2020). https://doi.org/10.1038/s41440-019-0383-7
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41440-019-0383-7
Keywords
This article is cited by
-
Characteristics of lipid metabolism dysfunction in hypertensive disorders of pregnancy
BMC Pregnancy and Childbirth (2025)
-
Association Between Circulating Fatty Acids and Blood Pressure: A Review
Current Nutrition Reports (2025)
-
Comprehensive fatty acid fractionation profilling in preeclampsia: a case control study with multivariable analysis
BMC Pregnancy and Childbirth (2022)