Abstract
We investigated the antihypertensive effects of maximakinin (MK) on spontaneously hypertensive rats (SHRs). The effects of MK on arterial blood pressure in SHRs were observed, and flow cytometry and 4,5-diaminofluorescein-2 staining were used to examine MK-induced nitric oxide (NO) release in human umbilical vein endothelial cells (HUVECs). Western blotting was used to analyze the effects of MK on the expression of AMP-activated protein kinase (AMPK), Akt, Connexin 43, ERK1/2, p38, and p-eNOS in HUVECs. The results showed that MK induced a more significant antihypertensive effect on SHRs than bradykinin (BK). MK induced significant increases in endothelial nitric oxide synthase (eNOS) phosphorylation and NO release in HUVECs. MK also significantly increased the phosphorylation of Akt and AMPK in HUVECs. The AMPK inhibitor compound C blocked the effect of MK on the generation of NO. MK induced the phosphorylation of ERK1/2, p38, and Connexin 43. The expression of p-Connexin 43 was significantly decreased in the presence of the ERK1/2 inhibitor U0126 but not the p38 inhibitor SB203580. The effects of MK on the phosphorylation of AMPK and ERK1/2 were significantly decreased by the BK B2 receptor inhibitor HOE-140. In summary, MK can significantly reduce blood pressure in SHRs. The antihypertensive effect might be mediated through the activation of the BK B2 receptor, while the downstream AMPK/PI3K/Akt/eNOS/NO and ERK1/2/Connexin 43 signaling pathways play additional roles.
Similar content being viewed by others
Introduction
Maximakinin (MK), a novel N-terminally extended form of bradykinin (BK) (DLPKINRKGPRPPGFSPFR) extracted from the skin venom of the Chinese toad (Bombina maxima), displays enhanced activity compared with that of BK and shows a highly selective 50-fold increase in molar potency for the BK B2 receptor (B2R) in mammalian arterial smooth muscle [1]. Intravenous MK dose dependently induced hypotension, vasodilation, and tachycardia in anesthetized rats, and the effects were antagonized by pretreatment with the BK inhibitor icatibant but not pyrilamine or enalaprilat [2]. However, there have been no reports on the effects of MK on hypertensive animals.
Endothelial nitric oxide synthase (eNOS) is an important regulator of cardiovascular homeostasis because it is the major source of nitric oxide (NO) production in vascular endothelial cells (VECs). Since NO release from VECs can lead to the relaxation of vascular smooth muscle, eNOS plays a crucial role in blood vessel vasodilation and blood pressure regulation. Several laboratories have reported that eNOS is phosphorylated in endothelial cells (ECs) at Ser1177 (bovine sequence) by the protein kinase Akt, resulting in an approximately twofold increase in eNOS catalytic activity [3, 4].
At the vascular level, AMP-activated protein kinase (AMPK) is expressed in both ECs and vascular smooth muscle cells (VSMCs). In cultured ECs, AMPK induces eNOS phosphorylation, thus increasing NO release [5, 6]. However, the relationship between AMPK, Akt, and eNOS is still not clear, and whether the phosphorylation of these three proteins is associated with the activation of B2R has not yet been reported.
A gap junction is a type of membrane channel structure that typically exists between adjacent cells, and its basic unit is known as connexin (Cx). Electrical or chemical signals can enter VSMCs from ECs through Cxs, further inducing the activation of calcium-dependent potassium channels, hyperpolarizing the cell membrane, and causing vasodilation [7]. At present, 21 Cxs have been identified, among which the expression of Cx43 is most closely associated with hypertension [8]. One study showed that in VSMCs and ECs, the activation of extracellular signal kinase (ERK1/2) promoted the phosphorylation of Cx43 [9]. Another study showed that MK induced the phosphorylation of ERK1/2 by activating B2R [10]. Therefore, we hypothesized that the antihypertensive effect of MK might be associated with the promotion of ERK1/2 phosphorylation, which further increases the activation of Cx43.
Herein, we describe in detail for the first time the antihypertensive effect of MK on spontaneously hypertensive rats (SHRs) and the effect of MK on the AMPK/Akt/eNOS/NO and ERK1/2/Cx43 signaling pathways.
Methods
Maximakinin and reagents
MK (DLPKINRKGPRPPGFSPFR) was obtained from the School of Biomedical Sciences, University of Ulster, Northern Ireland, UK.
L-NAME (NOS inhibitor), LY294002 (Akt inhibitor), compound C (AMPK inhibitor), U0126 (ERK1/2 inhibitor), and HOE-140 (B2 receptor inhibitor) were purchased from Biomol (Plymouth Meeting, PA, USA). Rabbit anti-Akt, p-Akt (Ser473), eNOS, p-eNOS (Ser1177), AMPK, p-AMPK (Thr172), ERK1/2, and p-ERK1/2 antibodies were obtained from Cell Signaling Technology (Boston, MA, USA). All other cell culture reagents were obtained from Sigma Chemical (St. Louis, MO, USA). MK was dissolved in DMSO such that the final concentration of DMSO was <0.1% in all experiments.
Animals and treatments
Adult male SHRs (weighing 250–300 g) were obtained from the animal production unit of the animal house in the Department of Pharmaceutical Sciences, Shenyang Pharmaceutical University, China. All animals were kept under constant temperature (22 ± 2 °C) conditions with a 12 h light/12 h dark cycle and free access to food and water and were cared for in accordance with the Guide for the Care and Use of Laboratory Animals (Eighth Edition, 2011, The National Academies Press, https://www.ncbi.nlm.nih.gov/pubmed/21595115).
The SHRs were equally divided into five groups: the control group, 4 mg/kg BK group and 2, 4, and 8 mg/kg MK group. After the SHRs were intravenously administered the indicated treatments, changes in arterial blood pressure were measured using an automatic sphygmomanometer (BP-98A; Softron, Tokyo, Japan). Before the measurement was performed, the rats were placed at 37 °C for 10 min until their heart rates were stable. The blood pressure measurements were repeated three times, and the average value of the three measurements was reported.
Cell culture and treatments
Human umbilical vein endothelial cells (HUVECs) were obtained from Key GEN Biotech (Nanjing, China). HUVECs were cultured in DMEM (HyClone, Logan, UT, USA) supplemented with 10% heat-inactivated (56 °C, 30 min) fetal bovine serum (Beijing Yuanheng Shengma Research Institution of Biotechnology, Beijing, China), 100 μg/mL streptomycin, 100 U/mL penicillin, and 0.03% L-glutamine and maintained at 37 °C with 5% CO2 in a humidified atmosphere.
NO release was quantified using 4,5-diaminofluorescein-2 (DAF-2), a NO-sensitive fluorescent probe. DAF-2 (5 μM) was added to the HUVECs and incubated for 1 h at 37 °C. Then, the cells were treated with MK (0.1 μM) for 0–2, 5, and 10 min. The fluorescence levels of the supernatant were measured using a multifunctional detection microplate reader (Tecan, Genios-Pro, Austria) at an excitation wavelength of 485 nm and an emission wavelength of 520 nm.
For Western blotting, total proteins were extracted from HUVECs after the administration of the indicated treatments, and then the analyses were carried out. The cell pellets were resuspended in lysis buffer consisting of 50 mmol/L HEPES pH 7.4, 1% Triton X-100, 2 mmol/L sodium orthovanadate, 100 mmol/L sodium fluoride, 1 mmol/L edetic acid, 1 mmol/L PMSF, 10 mg/L aprotinin (Sigma, MO, USA), and 10 mg/L leupeptin (Sigma, MO, USA) and then lysed at 4 °C for 1 h. After centrifugation at 12,000 × g for 15 min, the protein content of the supernatant was determined by a Bio-Rad DC protein assay (Bio-Rad Laboratories, Hercules, CA, USA). Equal amounts of the total protein were separated by 12% SDS–PAGE and transferred to nitrocellulose membranes, and the membranes were soaked in blocking buffer (5% skimmed milk). Proteins were detected using anti-rabbit, anti-mouse, or peroxidase (HRP)-conjugated anti-goat secondary antibodies and visualized using an ECL substrate kit.
Statistical analysis
All data represent at least three independent experiments and are expressed as the mean ± SEM. The data were analyzed by ANOVA using Statistics Package for Social Science software (version 13.0; SPSS, Chicago, IL, USA), and an LSD post hoc test was used to assess the statistical significance of differences between the control and treatment groups. P values < 0.05 were considered statistically significant.
Results
MK decreases arterial blood pressure in SHRs
To identify the acute effects of MK on arterial blood pressure, MK (2, 4, and 8 μg/kg) was intravenously administered to SHRs. The results showed that 4 and 8 μg/kg MK rapidly reduced the blood pressure of SHRs. The systolic blood pressure (SBP), diastolic blood pressure (DBP), and mean arterial pressure (MAP) began to drop 2 min after intravenous administration of MK, dropped to the lowest level at 5 min, and recovered to a normal level at 10 min (Fig. 1A–C).
Variations in SBP, DBP, and MAP in the experimental groups in the presence of MK. SBP (A), DBP (B), and MAP (C) were measured by the direct method using a BP-98A. The results are shown as the means ± SEM. n = 10. *P < 0.05 vs. the bradykinin group
MK increases NO release by activating eNOS in HUVECs
Production of the vasodilator NO by HUVECs was analyzed by DAF-2 (an NO-sensitive fluorescent probe). After DAF-2 (5 μM) was added to HUVECs and incubated for 1 h at 37 °C, MK was added to the HUVECs and incubated for 0–2, 5, and 10 min. The amount of NO produced was detected using a multifunctional microplate reader. The results showed that NO release was the highest at 5 min (Fig. 2A).
MK increases NO production in HUVECs. A MK (0.1 μM) treatment without inhibitors. B After 5 min, MK (0.1 μM) was coincubated with the eNOS inhibitor L-NAME (30 mM). NO production was quantified using DAF-2. The values are expressed as the means ± SEM (n = 3). *P < 0.05, **P < 0.01 vs. the control group, and #P < 0.05 vs. the MK group
eNOS is the major source of NO production in VECs. After HUVECs were pretreated with the eNOS inhibitor L-NAME for 30 min, MK was added to the cells and incubated for 5 min, after which NO production was measured. The results showed that MK did not promote the release of NO in ECs after eNOS activation was blocked with L-NAME (Fig. 2B).
MK increases the phosphorylation of eNOS, Akt, and AMPK
Enhanced phosphorylation of eNOS at Ser1177 is known to stimulate eNOS activity. Akt activation was measured by analyzing the phosphorylation of the Akt-Ser473 residue [11]. The activation of AMPK was monitored by determining the phosphorylation of the AMPK-Thr172 residue [12]. After treatment with MK, the phosphorylation levels of eNOS-Ser1177 (Fig. 3A), Akt-Ser473 (Fig. 3B), and AMPK-Thr172 (Fig. 3C) were increased in HUVECs. In comparison, the total protein levels of eNOS, Akt, and AMPK remained unchanged. These results suggested that MK had direct stimulatory effects on the enzymatic activities of eNOS, Akt, and AMPK.
Effects of MK on the phosphorylation of eNOS, Akt, and AMPK in HUVECs. HUVECs were growth arrested in 10% fetal bovine serum in DMEM for 24 h and then treated with MK (0.1 μM) for 0–2, 5, and 10 min. The phosphorylation levels of eNOS (A), Akt (B), and AMPK (C) were analyzed by Western blotting. The results are shown as the means ± SEM (n = 3). *P < 0.05 compared with the control
The relationship between AMPK and Akt
The phosphorylation of eNOS at Ser1177, which is one of the most prominent upstream determinants of endothelial NO production, is well known. In cultured ECs, it is not known whether the eNOS-Ser1177 site can be phosphorylated by AMPK or Akt, and the upstream and downstream relationships between AMPK and Akt remain unclear. To further confirm the relationship between AMPK and Akt, we added LY294002 (a PI3K/Akt inhibitor) or compound C (an AMPK inhibitor) to HUVECs to investigate the effects of MK on p-eNOS, p-AKT, and p-AMPK levels.
LY294002 (30 µM) was added to HUVECs and incubated for 30 min, and then MK (0.1 µM) was added and incubated for an additional 5 min. The western blot results showed that LY294002 significantly reduced the MK-induced expression of p-eNOS in HUVECs, but no significant difference in AMPK phosphorylation was observed (Fig. 4A, B). To further confirm whether the AMPK signaling pathway is involved in the phosphorylation of Akt and eNOS, we added compound C to investigate the effect of MK on p-eNOS and p-Akt. HUVECs were incubated with compound C (30 µM) for 30 min, and then MK was added and incubated for 5 min. Western blot analysis showed that compound C significantly reduced the MK-induced phosphorylation of eNOS and Akt in HUVECs (Fig. 4C, D).
The relationship between AMPK and Akt. Role of Akt in the AMPK/Akt/eNOS signaling pathway in endothelial cells. HUVECs were exposed to LY294002 (30 μM) for 30 min before the administration of MK for 5 min. Then, the phosphorylation of AMPK (A) and eNOS (B) was analyzed by western blotting. The role of AMPK in the AMPK/Akt/eNOS signaling pathway in endothelial cells. HUVECs were exposed to compound C (30 μM) for 30 min before the administration of MK for 5 min. Then, the phosphorylation of Akt (C) and eNOS (D) was analyzed by western blotting. The results are shown as the means ± SEM (n = 3). #P < 0.05 vs. the control and *P < 0.05 vs. the MK group
Taken together, these results indicate that activation of the AMPK signaling pathway is necessary for MK-induced activation of eNOS and Akt and that Akt is upstream of eNOS.
The effect of MK on the phosphorylation of Cx43 and ERK1/2
The phosphorylation of the Cx (Cx43) is associated with hypertension. We hypothesized that ERK1/2 may promote Cx43 phosphorylation and regulate the function of Cx43. Thus, we investigated the effect of MK on the expression levels of Cx43 and ERK1/2. MK was added to HUVECs and incubated for 0–2, 5, and 10 min, and then western blot analysis was carried out. The results showed that 0.1 μM MK significantly increased the phosphorylation of Cx43 and ERK1/2 at 5 min, while MK did not affect the total levels of the two proteins (Fig. 5).
Effects of MK on the phosphorylation of Cx43 and ERK1/2 in HUVECs. HUVECs were treated with MK (0.1 μM) for 0–2, 5, 10 min. The phosphorylation levels of Cx43 (A) and ERK1/2 (B) were analyzed by western blotting. The results are shown as the means ± SEM (n = 3). *P < 0.05 compared with the control
The effect of ERK1/2 on the phosphorylation of Cx43
Although ERK1/2 is a regulatory protein associated with Cx43 phosphorylation, the relationship between ERK1/2 and Cx43 in MK-treated HUVECs is still unclear. The ERK1/2 inhibitor U0126 was used to investigate the interactions between ERK1/2 and Cx43 (Fig. 6). The western blot results showed that the addition of U0126 significantly reversed the MK-induced phosphorylation of Cx43.
Role of ERK1/2 in the ERK1/2/Cx43 signaling pathway in HUVECs. HUVECs were exposed to U0126 (30 μM) for 30 min before the administration of MK for 5 min. Then, the phosphorylation of Cx43 was analyzed by western blotting. The results are shown as the means ± SEM (n = 3). #P < 0.05 vs. the control and *P < 0.05 vs. the MK group
The effect of the B2 receptor inhibitor HOE-140 on the phosphorylation of AMPK and ERK1/2
To further confirm whether MK induces the phosphorylation of AMPK and ERK1/2 by activating BK B2R, we investigated the effect of MK on p-AMPK and p-ERK1/2 in the presence of HOE-140, a BK B2R inhibitor. After HUVECs were incubated with HOE-140 (0.05 µg/μL) for 30 min, MK was added to the HUVECs and incubated for 5 min. The results showed that HOE-140 significantly reduced the MK-induced phosphorylation of AMPK and ERK1/2 in HUVECs (Fig. 7A, B). These results suggest that MK plays a stimulatory role in the AMPK/Akt/eNOS and ERK1/2/Cx43 signaling pathways by activating BK B2R.
Role of B2R in the phosphorylation of AMPK and ERK1/2 in HUVECs. HUVECs were exposed to the B2R inhibitor HOE-140 (0.05 μg/μL) for 30 min before the addition of MK for 5 min. Then, the phosphorylation levels of AMPK (A) and ERK1/2 (B) were analyzed by western blotting. The results are shown as the means ± SEM (n = 3). #P < 0.05 vs. the control group and *P < 0.05 vs. the MK group
Discussion
BK is a nonapeptide with a wide range of biological effects that have been extensively studied in mammals. A major physiological role of BK in mammals, including humans, is in the maintenance of systemic blood pressure through modulating the dilation of arterial smooth muscle to effectively counteract angiotensin-II-induced constriction [13]. MK is a 19-residue peptide from the amphibian Bombina maxima that incorporates the full sequence of BK at its C-terminus and has a hydrophilic 10-residue N-terminal extension. Although in vitro experiments have shown that the relaxation effect of MK on Wistar rat tail artery smooth muscle was 50 times greater than that of BK, the in vivo effect of MK is still unclear. To further investigate the effect of MK, we first selected SHRs to examine the antihypertensive effect of MK in vivo. The results showed that intravenously administered BK (4 μg/kg) had no effect on arterial blood pressure in SHRs, while intravenously administered MK (4 and 8 μg/kg) significantly reduced arterial blood pressure in SHRs. SBP, DBP, and MAP began to drop 2 min after intravenous administration of MK and decreased to their lowest levels at 5 min. Therefore, MK has robust antihypertensive activity in vivo, similar to its effects in vitro, and MK induces its antihypertensive effects more rapidly than BK in SHRs.
ECs contribute to the regulation of blood pressure and blood flow by releasing vasodilators such as NO [14]. ECs produce NO, a heteronuclear diatomic free radical product generated through the oxidation of L-arginine to L-citrulline by eNOS. In this study, MK enhanced eNOS phosphorylation, leading to increased NO availability in cultured HUVECs, which likely explains how MK can reduce blood pressure in hypertensive rats.
The AMPK pathway is considered to be an important regulator of metabolism and is crucial for the maintenance of endothelial function and redox balance. AMPK exerts its antihypertensive effects by influencing various signaling cascades, resulting in improved NO bioavailability, attenuated free radical generation, and the activation of angiogenic factors. Activated AMPK enhances eNOS activity by phosphorylating the eNOS-Ser1177 residue.
Protein kinase B, also known as Akt, is a serine/threonine-specific protein kinase that plays a key role in multiple cellular processes. Some studies have shown that the Akt pathway is closely associated with the occurrence and development of cardiovascular diseases [15]. As an important target gene downstream of the PI3K/AKT signaling pathway, eNOS is activated by Akt and then catalyzes NO release. Therefore, we investigated the regulation of AMPK and Akt in MK-treated ECs. The western blot results showed that 0.1 μM MK significantly promoted the phosphorylation of AMPK and Akt, suggesting that the NO release induced by eNOS phosphorylation may be regulated by AMPK and Akt.
However, it is still not clear whether there is a relationship between AMPK and Akt in ECs. The AMPK inhibitor compound C and the Akt inhibitor LY294002 were used to further investigate the connection between AMPK and Akt. The results showed that the addition of LY294002 significantly inhibited the MK-induced phosphorylation of eNOS, while it did not affect the phosphorylation of AMPK. The addition of the AMPK inhibitor compound C significantly inhibited the MK-induced phosphorylation of eNOS and Akt. These results suggest that AMPK is located upstream of Akt and that AMPK regulates eNOS phosphorylation by activating Akt. The observed effect was associated with an increase in AMPK that led to the activation of eNOS via the signal transduction pathway involving PI3K/Akt. Thus, MK activates the AMPK/PI3K/Akt-eNOS-NO signaling pathway in ECs.
Bawolak et al. [10] reported that MK could also promote ERK1/2 phosphorylation [10]. ERK1/2 belongs to the mitogen-activated protein kinase family. Activation of ERK1/2 can promote the phosphorylation of Cx43 [9]. As a member of the Cx family, Cx43 phosphorylation increases electrical or chemical signal sensing in VSMCs from ECs through Cxs, further regulating blood vessel tension, dilating blood vessels, and lowering blood pressure [7]. Thus, we investigated the effect of MK on the phosphorylation of ERK1/2 and Cx43 in ECs. MK (0.1 μM) notably increased the phosphorylation of ERK1/2 and Cx43 in HUVECs, suggesting that the antihypertensive effect of MK might partly be achieved by stimulating the phosphorylation of ERK1/2 and activating Cx43. Furthermore, the addition of U0126 effectively blocked this effect. These results showed that MK activated Cx43 through upstream ERK1/2 phosphorylation, and activated Cx43 further promoted vasodilation and decreased blood pressure.
The biological effects of BK on mammalian tissues are thought to be mediated by two different receptor subtypes, designated B1 and B2, which differ in their pharmacological profiles and their species-specific tissue distributions. B2Rs are mainly expressed in ECs, VSMCs, and cardiomyocytes and mediate most of the functions of BK, while B1 receptors are expressed at low levels under physiological conditions, and their effects remain controversial [16, 17]. One study showed that MK exhibited 8–12-fold lower potency than BK toward mammalian B2R [2]. However, this effect was primarily observed in VSMCs, and no specific report has described the effects of MK in VECs. Thus, we investigated the mechanism by which MK stimulates AMPK and ERK1/2 phosphorylation in VECs. The addition of the B2R inhibitor HOE-140 obviously abrogated the MK-induced phosphorylation of AMPK and ERK1/2, suggesting that MK regulates the AMPK and ERK1/2 signaling pathways through the activation of B2R in VECs.
Conclusions
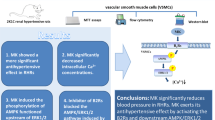
Herein, the in vivo pharmacological activity of MK was reported for the first time. We found that MK rapidly and effectively reduced blood pressure in SHRs. In an in vitro study, we found that VECs may be one of the targets of MK. In the HUVEC membrane, MK activated B2R, activated the downstream AMPK-Akt-eNOS-NO and ERK1/2-Cx43 intracellular signaling pathways, promoted Cx43 phosphorylation, and increased NO production, leading to dilated blood vessels, reduced peripheral resistance, and decreased blood pressure (Fig. 8).
The antihypertensive effect of MK may occur through the AMPK/Akt/eNOS/NO and ERK1/2/Cx43 signaling pathways
References
O’Rourke M, Chen T, Hirst DG, Rao P, Shaw C. The smooth muscle pharmacology of maximakinin, a receptor-selective, bradykinin-related nonadecapeptide from the venom of the Chinese toad, Bombina maxima. Regul Pept. 2004;121:65–72.
Charest-Morin X, Bachelard H, Jean M, Marceau F. Species-specific pharmacology of maximakinin, an amphibian homologue of bradykinin: putative prodrug activity at the human B2 receptor and peptidase resistance in rats. PeerJ. 2017;5:e2911.
Dimmeler S, Fleming I, Fisslthaler B, Hermann C, Busse R, Zeiher AM. Activation of nitric oxide synthase in endothelial cells by Akt-dependent phosphorylation. Nature. 1999;399:601–5.
Decker B, Pumiglia K. mTORc1 activity is necessary and sufficient for phosphorylation of eNOS(S1177). Physiol Rep. 2018;6:e13733.
Feng L, Ren J, Li Y, Yang G, Kang L, Zhang S, et al. Resveratrol protects against isoproterenol induced myocardial infarction in rats through VEGF-B/AMPK/eNOS/NO signalling pathway. Free Radic Res. 2019;53:82–93.
Youn JY, Wang T, Cai H. An ezrin/calpain/PI3K/AMPK/eNOSs1179 signaling cascade mediating VEGF-dependent endothelial nitric oxide production. Circ Res. 2009;104:50–9.
Figueroa XF, Isakson BE, Duling BR. Vascular gap junctions in hypertension. Hypertension. 2006;48:804–11.
Arishiro K, Hoshiga M, Ishihara T, Kondo K, Hanafusa T. Connexin 43 expression is associated with vascular activation in human radial artery. Int J Cardiol. 2010;145:270–2.
Ogut O, Brozovich FV. The potential role of MLC phosphatase and MAPK signalling in the pathogenesis of vascular dysfunction in heart failure. J Cell Mol Med. 2008;12:2158–64.
Bawolak MT, Roy C, Gera L, Marceau F. Prolonged signalling and trafficking of the bradykinin B2 receptor stimulated with the amphibian peptide maximakinin: insight into the endosomal inactivation of kinins. Pharmacol Res. 2012;65:247–53.
Freudlsperger C, Horn D, Weissfuss S, Weichert W, Weber KJ, Saure D, et al. Phosphorylation of AKT(Ser473) serves as an independent prognostic marker for radiosensitivity in advanced head and neck squamous cell carcinoma. Int J Cancer. 2015;136:2775–85.
Le LT, Couvet M, Favier B, Coll JL, Nguyen CH, Molla A. Discovery of benzo[e]pyridoindolones as kinase inhibitors that disrupt mitosis exit while erasing AMPK-Thr172 phosphorylation on the spindle. Oncotarget. 2015;6:22152–66.
Ma J, Luo Y, Ge L, Wang L, Zhou M, Zhang Y, et al. Ranakinestatin-PPF from the skin secretion of the Fukien gold-striped pond frog, Pelophylax plancyi fukienensis: a prototype of a novel class of bradykinin B2 receptor antagonist peptide from ranid frogs. The Sci World J. 2014;2014:564839.
Cines DB, Pollak ES, Buck CA, Loscalzo J, Zimmerman GA, McEver RP, et al. Endothelial cells in physiology and in the pathophysiology of vascular disorders. Blood. 1998;91:3527–61.
Ning WH, Zhao K. Propionyl-L-carnitine induces eNOS activation and nitric oxide synthesis in endothelial cells via PI3 and Akt kinases. Vasc Pharmacol. 2013;59:76–82.
Schmaier AH. The plasma kallikrein-kinin system counterbalances the renin-angiotensin system. J Clin Investig. 2002;109:1007–9.
Xu J, Carretero OA, Sun Y, Shesely EG, Rhaleb NE, Liu YH, et al. Role of the B1 kinin receptor in the regulation of cardiac function and remodeling after myocardial infarction. Hypertension. 2005;45:747–53.
Acknowledgements
We thank Renee Mosi, Ph.D., from Liwen Bianji, Edanz Editing China (www.liwenbianji.cn/ac), for editing the English text of a draft of this paper.
Funding
This work was supported in part by the General Project of Liaoning Provincial Science and Technology Department (20170540844) and by the General Project of Liaoning Education Department (2017LQN16).
Author information
Authors and Affiliations
Contributions
CX and YY designed the research. YY, L-SX, YW, and F-FS performed the research. CX contributed new reagents or analytical tools. X-MZ analyzed the data. YY and CX wrote the paper.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Yu, Y., Xu, LS., Wu, Y. et al. The antihypertensive effect of MK on spontaneously hypertensive rats through the AMPK/Akt/eNOS/NO and ERK1/2/Cx43 signaling pathways. Hypertens Res 44, 781–790 (2021). https://doi.org/10.1038/s41440-021-00638-w
Received:
Revised:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41440-021-00638-w
Keywords
This article is cited by
-
Not a small frog in a big pond: targeting bradykinin receptor B2 signaling in vascular smooth muscle cells for treatment of hypertension
Hypertension Research (2023)
-
Maximakinin reduced intracellular Ca2+ level in vascular smooth muscle cells through AMPK/ERK1/2 signaling pathways
Hypertension Research (2023)