Abstract
Testosterone replacement therapy (TRT) remains controversial in men with a history of prostate cancer due to historical concerns regarding oncologic safety. This scoping review aimed to systematically map existing evidence on the safety and efficacy of TRT in men following definitive treatment for prostate cancer. A systematic search of PubMed, CENTRAL, and Embase identified 447 records, from which 12 studies met inclusion criteria. Most were retrospective cohort studies, with sample sizes ranging from 10 to 152 men. TRT was not associated with an increased risk of biochemical recurrence or cancer progression in any included study. Reported PSA kinetics remained within expected post-treatment parameters, and several studies showed lower recurrence rates in TRT groups compared to controls. TRT consistently increased total and/or free testosterone and improved hypogonadal symptoms. However, the evidence base is limited by retrospective designs, small sample sizes, heterogeneous outcome reporting, and a lack of long-term data. Despite these limitations, findings suggest TRT may be cautiously considered in selected men with stable disease and confirmed hypogonadism. High-quality prospective studies are needed to clarify safety in diverse and high-risk populations and inform future clinical guidelines.
Similar content being viewed by others
Introduction
Hypogonadism is a common condition among ageing men, affecting up to 30% of those aged 40–79 years-old [1]. It is marked by a reduction in serum testosterone levels, which may lead patients to present with fatigue, reduced libido and/or erectile dysfunction, as well as changes in body composition such as loss of lean muscle mass and increased fat accumulation [2, 3]. Beyond these effects, hypogonadism is linked to adverse health outcomes such as insulin resistance, type 2 diabetes, cardiovascular disease, and depression, collectively reducing quality of life and increasing long-term morbidity and mortality risks [4,5,6,7].
Testosterone replacement therapy (TRT) is a well-established treatment for men with symptomatic hypogonadism [8]. It has been shown to improve muscle mass, reduce fat mass, and improve cardiovascular and mental health parameters [8,9,10]. While lifestyle interventions remain the first-line approach, adherence and effectiveness can be limited, making TRT an important therapeutic option for appropriately selected individuals [8, 11].
However, the use of TRT in men with a history of prostate cancer remains controversial. Historically, this concern stemmed from early work by Huggins and Hodges, who demonstrated that reducing testosterone levels led to regression of metastatic prostate cancer, implying a stimulatory role of androgens in tumour progression [12]. This interpretation, drawn from limited data, led to longstanding caution around TRT in this population. Similarly, Fowler and Whitmore’s 1981 series of 67 men with metastatic prostate cancer treated with exogenous testosterone observed rapid disease progression primarily in those previously treated with androgen deprivation therapy (ADT), further reinforcing this conservative stance [13].
Given the increasing prevalence of prostate cancer survivors [14], many of whom experience hypogonadism either due to age [2] or prior ADT [15] there is a growing need to understand the safety and efficacy of TRT in this context. While several individual studies have reported on this issue, the evidence remains heterogeneous and methodologically varied.
Objective
The objective of this scoping review is to systematically map the existing literature on the safety and efficacy of TRT in men following definitive treatment for prostate cancer (i.e., radical prostatectomy or radiotherapy). Specifically, this review aims to:
-
1.
Identify the nature and extent of available evidence;
-
2.
Summarise oncological safety outcomes (e.g., biochemical recurrence [BCR], PSA kinetics, progression); and
-
3.
Describe therapeutic efficacy outcomes (e.g., symptom improvement, hormonal normalisation, quality of life).
This review is conducted in accordance with the PRISMA-ScR (Preferred Reporting Items for Systematic Reviews and Meta-Analyses extension for Scoping Reviews) guidelines [16], to provide a comprehensive overview of the current state of research and inform future investigations and clinical practice.
Materials and methods
Eligibility criteria
Studies were eligible for inclusion if they met the following criteria:
-
Peer-reviewed, human studies examining TRT in men post-radical prostatectomy or radiotherapy for prostate cancer.
-
Reported at least one oncological outcome (e.g., PSA kinetics, BCR, metastasis, or cancer-specific mortality) or one efficacy outcome (e.g., hormonal response, symptom relief, or quality of life).
-
Study types included RCTs, prospective or retrospective cohort studies, or case series with ≥10 participants.
-
Studies were excluded if they were reviews, editorials, letters, conference abstracts, lacked a clearly defined TRT intervention, involved men with untreated or metastatic disease, or did not report relevant outcomes.
Information sources and search strategy
A systematic search was performed on June 23, 2025, in three databases: PubMed, CENTRAL (Cochrane Central Register of Controlled Trials), and Embase. An initial pilot search was conducted, via PubMed, to refine search terms. The final search strategy, developed iteratively, was applied across all databases and included terms related to TRT and definitive prostate cancer treatments. The full PubMed strategy is included in Appendix A to allow for full replication.
-
4.
PubMed: Initial search returned 491 results. A refined strategy resulted in 220 results, which were narrowed to 82 after filters for human studies, adult male populations, and English language.
-
5.
CENTRAL: 69 results (17 duplicates).
-
6.
Embase: 342 results (29 duplicates).
After removing 46 duplicates across all databases, 447 titles and abstracts were screened.
Selection of sources of evidence
Titles and abstracts were independently screened by two reviewers to determine eligibility based on the predefined inclusion and exclusion criteria. Any disagreements were resolved through discussion and consensus. Full texts of potentially relevant articles were then retrieved and assessed for inclusion. Of the 447 screened studies, 38 full-text articles were reviewed, and 12 studies met the eligibility criteria and were included in the final synthesis. The study selection process is illustrated in the PRISMA-ScR flow diagram (Fig. 1).
Page MJ, et al. BMJ 2021;372:n71. https://doi.org/10.1136/bmj.n71. This work is licensed under CC BY 4.0. To view a copy of this license, visit https://creativecommons.org/licenses/by/4.0/.
Synthesis of results
Data were synthesised narratively, with findings grouped by oncological and hormonal outcomes. Given heterogeneity in study design, outcome measures, and follow-up durations, no quantitative synthesis was performed. Key results were tabulated to facilitate cross-study comparison (Tables 1 and 2).
Results
A total of 12 studies met the inclusion criteria and were included in this scoping review, published between 2005 and 2025. Most studies were retrospective cohort designs, with one study being a prospective case series, and sample sizes for TRT groups ranged from 10 to 152 men. Radical prostatectomy was the primary treatment in 6 studies, while 6 studies included patients primarily treated with radiotherapy. Patients receiving TRT post- definitive treatment for prostate cancer had reported mean ages ranging from 60.8 to 64.3 years, and median ages from 59 to 77 years across studies. The age range across all studies was 49 to 83 years.
The interval between definitive treatment for prostate cancer and TRT initiation varied widely across studies. Reported median times ranged from 12.3 to 45 months, with individual ranges extending from 2.6 to 170.9 months. Mean initiation times ranged from 14 to 36 months. One study stratified timing by treatment modality, with a median of 15 months post-prostatectomy and 45 months post-radiotherapy [17]. In 2 studies, timing of TRT post definitive treatment was not precisely specified [18, 19].
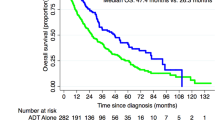
Across the 12 included studies, TRT was generally not associated with an increased risk of BCR or prostate cancer progression. Reported BCR rates in TRT groups were low, often lower than in non-TRT comparators [18, 20,21,22]. Ahlering et al. reported a BCR rate of 7.2% in the TRT treatment group compared to 12.6% in the matched control group [18], while Pastuszak et al. [20] observed BCR rates of 15.4% in the TRT treatment group versus 53.3% in the non-TRT treatment group, with all patients experiencing BCR in the high-risk prostate cancer groups [20]. Shahine et al. also reported favourable BCR outcomes in the TRT cohort with a rate of 6.4% compared to 12.6% in untreated controls [21]. PSA kinetics remained within expected post-treatment ranges [23]; Pastuszak et al. [20] reported PSA Velocity (PSAV) in the treatment group was 0.002 ng/mL/y [20] and Ory et al. [17] found the median PSAV in the treatment group was 0.0175 ng/mL/yr [17]. Several studies observed small PSA increases, especially in high-risk patients – e.g., a rise from 0.10 to 0.36 ng/mL in Gleason ≥8 patients [24], or from 0.004 to 0.007 ng/mL (p < 0.0001) [20], but these were not indicative of recurrence. Importantly, no study demonstrated a statistically significant increase in oncologic risk attributable to TRT use. Please see Table 1 for all oncological outcomes in each included study.
Of the 11 studies reporting hormonal outcomes, significant increases in total testosterone (TT) and/or free testosterone (FT) following TRT, with TT typically rising from hypogonadal to eugonadal levels were observed. Reported TT increases ranged from median 188 to 591 ng/dL. From the included studies, 7 noted improvements in sexual function, energy, and other hypogonadal symptoms including, where measured, validated instruments such as the Sexual Health Inventory for Men (SHIM) and Expanded Prostate Cancer Index Composite (EPIC) scores reflecting symptomatic benefit. Overall, TRT was effective in restoring testosterone levels and improving quality-of-life indicators in this post-treatment population. Please see Table 2 for full TRT efficacy outcomes in each included study.
Definitions of BCR varied across studies and follow-up duration ranged from 6 to 189 months. Studies differed in TRT modality, dosage, and monitoring protocols, introducing significant heterogeneity. Most included studies were retrospective and no studies focused on ethnically diverse populations or long-term recurrence outcomes. Only a minority of included studies stratified outcomes by Gleason score or cancer risk group.
Discussion
The findings from this scoping review systematically map the available evidence on the safety and efficacy of TRT in men following definitive treatment for prostate cancer. Historically, TRT has been contraindicated in this population due to concerns about oncological safety [12, 13]. However, the findings from the 12 included studies suggest that, in appropriately selected individuals, TRT may be both effective and oncologically safe.
11 of the included studies were retrospective cohort designs with small to moderate sample sizes, and one was a prospective case series. Despite methodological limitations, a consistent signal of oncologic safety was observed. BCR rates in men receiving TRT were low and, in several studies, even lower than in non-TRT comparators [18, 20, 21]. Where PSA kinetics were reported, increases were minor and align with established non-recurrence benchmarks [20, 23,24,25]. Importantly, no study demonstrated a statistically significant increase in oncologic risk attributable to TRT use. These findings are particularly notable given the historical hesitancy to use TRT in this setting, which stemmed from early studies by Huggins and Hodges suggesting that androgens promote prostate cancer growth [12]. However, emerging literature now challenges this paradigm; evidence supports the saturation model, which theorises that prostate androgen receptors become fully activated at low testosterone levels, beyond which additional testosterone does not stimulate further prostate cancer growth [26]. A schematic representation of the saturation model was tailored, providing a visual framework that supports the mechanistic insights discussed (Fig. 2) [26]. Furthermore, studies in both post-prostatectomy and post-radiotherapy patients demonstrate no increased risk of BCR, and in some cases, TRT may even confer a protective effect [18, 20, 22, 25].
T testosterone.
TRT was effective in addressing hypogonadism in this post-treatment population. All studies reporting hormonal outcomes observed significant increases in TT and/or FT levels, often from hypogonadal to eugonadal ranges [18, 20, 21, 24, 25, 27,28,29]. Symptomatic improvements were also noted, particularly in libido, energy levels, and sexual function [18, 24, 28,29,30]. While some studies used validated instruments such as the SHIM and EPIC questionnaires [21, 28, 29], others reported subjective improvements without standardised assessment tools [18, 20, 24, 27, 30], highlighting variability in outcome measurement.
Timing of TRT initiation varied substantially, ranging from as early as 2.6 months to over 14 years post-treatment. Some studies stratified outcomes by treatment modality, with slightly earlier initiation post-prostatectomy compared to radiotherapy [17]. However, in some cases, the rationale for TRT timing and criteria for initiation were not explicitly reported. This reflects a broader lack of consensus on optimal timing and patient selection for TRT in prostate cancer survivors. It is also important to note that two studies included patients treated not only with traditional TRT but also with other testosterone-targeted hormonal therapies such as clomiphene citrate [17, 19]. However, in this review, patients from these studies who were not treated with TRT were excluded from data extraction to preserve consistency with the review objectives.
While this review excluded studies with fewer than 10 participants, a small case series by Kadomoto et al. reported favourable outcomes among six men with high-risk prostate cancer treated with injectable TRT following brachytherapy [31]. No BCR occurred with TRT therapy and five out of six patients had only minor increases in PSA levels, with one experiencing a transient PSA rise to 0.843 ng/mL which later declined to 0.338 ng/mL without the need for TRT cessation [31]. Although not eligible for data extraction, this study adds anecdotal support to the emerging safety profile of TRT in patients post definitive treatment for prostate cancer and underscores the need for larger, prospective studies in this subgroup.
Despite the promising findings, the evidence base mapped in this scoping review is limited by several important factors. Most included studies were retrospective in design, which introduces potential for selection bias, incomplete data capture, and confounding. Sample sizes were generally small to moderate, limiting the statistical power to detect rare oncologic outcomes such as metastasis or prostate cancer-specific mortality. Additionally, definitions of BCR varied across studies, and follow-up durations ranged widely from 6 months to over 15 years, contributing to significant heterogeneity. Variability in TRT regimens (formulation, dosage, and monitoring protocols) further complicates interpretation. Quality-of-life outcomes were often reported using non-validated or subjective measures, and few studies stratified outcomes by risk group, Gleason score, or other prognostic factors. Furthermore, most study populations lacked ethnic diversity, limiting generalisability. Finally, this review included only English-language, peer-reviewed studies and excluded case reports with fewer than 10 participants, which may have led to the omission of relevant but limited evidence.
Nonetheless, across the reviewed studies, a consistent finding of oncologic safety and therapeutic benefit was observed. These preliminary findings suggest that, in carefully selected men with stable disease, TRT may be both effective and safe. However, prospective trials with rigorous methodology, standardised outcome measures, and long-term follow-up are essential to validate these observations and better inform clinical decision-making.
Conclusion
This scoping review systematically mapped the available evidence on TRT in men following definitive treatment for prostate cancer. Across 12 studies published between 2005 and 2025, TRT was not associated with increased risk of BCR or cancer progression. Instead, TRT demonstrated consistent efficacy in restoring serum testosterone levels and alleviating hypogonadal symptoms.
These findings challenge the historical belief that TRT poses an inherent oncologic risk to prostate cancer survivors. However, limitations in the existing evidence, such as small sample sizes, retrospective study designs, heterogeneous outcome reporting, and underrepresentation of high-risk and diverse patient groups, highlight the need for high-quality prospective research. Until such data become available, TRT may be cautiously considered in men with documented hypogonadism and stable post-treatment disease, provided that treatment decisions are individualised and closely monitored.
Data availability
No new primary data were generated for this study. All data supporting the conclusions of this review are derived from previously published studies, which are fully referenced in the manuscript. For further details or to access the original datasets, readers are encouraged to consult the cited publications.
References
Swartz C, Young M. Low serum testosterone and myocardial infarction in geriatric male inpatients. J Am Geriatr Soc. 1987;35:39–44. https://doi.org/10.1111/j.1532-5415.1987.tb01317.x
Wu FC, Tajar A, Beynon JM, Pye SR, Silman AJ, Finn JD, et al. Identification of late-onset hypogonadism in middle-aged and elderly men. N Engl J Med. 2010;363:123–35. https://doi.org/10.1056/NEJMoa0911101
Tajar A, Forti G, O’Neill T, Lee D, Silman A, Finn J, et al. Characteristics of secondary, primary, and compensated hypogonadism in aging men: evidence from the European Male Ageing Study. J Clin Endocrinol Metab. 2010;95:1810–8. https://doi.org/10.1210/jc.2009-1796
Schipf S, Haring R, Friedrich N, Nauck M, Lau K, Alte D, et al. Low total testosterone is associated with increased risk of incident type 2 diabetes mellitus in men: results from the Study of Health in Pomerania (SHIP). Aging Male. 2011;14:168–75. https://doi.org/10.3109/13685538.2010.524955
Haring R, John U, Völzke H, Nauck M, Dörr M, Felix S, et al. Low testosterone concentrations in men contribute to the gender gap in cardiovascular morbidity and mortality. Gend Med. 2012;9:557–68. https://doi.org/10.1016/j.genm.2012.10.001
Joshi D, Van Schoor N, De Ronde W, Schaap L, Comijs H, Beekman A, et al. Low free testosterone levels are associated with prevalence and incidence of depressive symptoms in older men. Clin Endocrinol (Oxf). 2010;72:232–40. https://doi.org/10.1111/j.1365-2265.2009.03641.x
Yeo S, Holl K, Peñaherrera N, Wissinger U, Anstee K, Wyn R. Burden of male hypogonadism and major comorbidities, and the clinical, economic, and humanistic benefits of testosterone therapy: a narrative review. Clin Outcomes Res. 2021;13:31–8. https://doi.org/10.2147/CEOR.S282047
Bhasin S, Cunningham G, Hayes F, Matsumoto A, Snyder P, Swerdloff R, et al. Testosterone therapy in adult men with androgen deficiency syndromes: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2006;91:1995–2010. https://doi.org/10.1210/jc.2005-2847
Barbonetti A, D’Andrea S, Francavilla S. Testosterone replacement therapy. Andrology. 2020;8:1551–66. https://doi.org/10.1111/andr.12774
Traish A. Benefits and health implications of testosterone therapy in men with testosterone deficiency. Sex Med Rev. 2018;6:86–105. https://doi.org/10.1016/j.sxmr.2017.03.003
Hackett G. Type 2 diabetes and testosterone therapy. World J Mens Health. 2019;37:1 https://doi.org/10.5534/wjmh.180027
Huggins C, Hodges C. Studies on prostatic cancer: I. The effect of castration, of estrogen and of androgen injection on serum phosphatases in metastatic carcinoma of the prostate. CA Cancer J Clin. 1972;22:232–40. https://doi.org/10.3322/canjclin.22.4.232
Fowler JE, Whitmore WF. The response of metastatic adenocarcinoma of the prostate to exogenous testosterone. J Urol. 1981;126:372–5. https://doi.org/10.1016/S0022-5347(17)54531-0
Wagle NS, Nogueira L, Devasia TP, Mariotto AB, Yabroff KR, Islami F, et al. Cancer treatment and survivorship statistics, 2025. CA Cancer J Clin. 2025;75:308–40. https://doi.org/10.3322/caac.70011
Alibhai SMH, Duong-Hua M, Sutradhar R, Fleshner NE, Warde P, Cheung AM, et al. Impact of androgen deprivation therapy on cardiovascular disease and diabetes. J Clin Oncol. 2009;27:3452–8. https://doi.org/10.1200/JCO.2008.20.0923
Tricco AC, Lillie E, Zarin W, O’Brien KK, Colquhoun H, Levac D, et al. PRISMA extension for scoping reviews (PRISMA-ScR): checklist and explanation. Ann Intern Med. 2018;169:467–73. https://doi.org/10.7326/M18-0850
Ory J, Flannigan R, Lundeen C, Huang JG, Pommerville P, Goldenberg SL. Testosterone therapy in patients with treated and untreated prostate cancer: impact on oncologic outcomes. J Urol. 2016;196:1082–9. https://doi.org/10.1016/j.juro.2016.04.074
Ahlering TE, My Huynh L, Towe M, See K, Tran J, Osann K, et al. Testosterone replacement therapy reduces biochemical recurrence after radical prostatectomy. BJU Int. 2020;126:91–6. https://doi.org/10.1111/bju.15042
Flores JM, Vertosick EA, Salter CA, Liso N, Teloken P, Berookhim B, et al. Testosterone therapy in men after radical prostatectomy for low-intermediate organ-confined prostate cancer. J Urol. 2025;213:27–33.
Pastuszak AW, Pearlman AM, Lai WS, Godoy G, Sathyamoorthy K, Liu JS, et al. Testosterone replacement therapy in patients with prostate cancer after radical prostatectomy. J Urol. 2013;190:639–44. https://doi.org/10.1016/j.juro.2013.02.002
Shahine H, Zanaty M, Zakaria AS, Nguyen D-D, Couture F, Sadri I, et al. Oncological safety and functional outcomes of testosterone replacement therapy in symptomatic adult-onset hypogonadal prostate cancer patients following robot-assisted radical prostatectomy. World J Urol. 2021;39:3223–9.
Pastuszak AW, Pearlman AM, Godoy G, Miles BJ, Lipshultz LI, Khera M. Testosterone replacement therapy in the setting of prostate cancer treated with radiation. Int J Impot Res. 2013;25:24–8.
Koulikov D, Mohler MC, Mehedint DC, Attwood K, Wilding GE, Mohler JL. Low detectable prostate specific antigen after radical prostatectomy—treat or watch? J Urol. 2014;192:1390–6. https://doi.org/10.1016/j.juro.2014.05.088
Pastuszak AW, Khanna A, Badhiwala N, Morgentaler A, Hult M, Conners WP, et al. Testosterone therapy after radiation therapy for low, intermediate and high risk prostate cancer. J Urol. 2015;194:1271–6. https://doi.org/10.1016/j.juro.2015.05.084
Aglan M, Kong Y, Connell B. Testosterone therapy in patients with locoregional prostate cancer treated with prior androgen deprivation therapy and radiation: a retrospective single center review. Transl Androl Urol. 2025;14:250–7.
Morgentaler A, Traish AM. Shifting the paradigm of testosterone and prostate cancer: the saturation model and the limits of androgen-dependent growth. Eur Urol. 2009;55:310–21. https://doi.org/10.1016/j.eururo.2008.09.024
Khera M, Grober ED, Najari B, Colen JS, Mohamed O, Lamb DJ, et al. Testosterone replacement therapy following radical prostatectomy. J Sex Med. 2009;6:1165–70.
Agarwal PK, Oefelein MG. Testosterone replacement therapy after primary treatment for prostate cancer. J Urol. 2005;173:533–6.
Balbontin FG, Moreno SA, Bley E, Chacon R, Silva A, Morgentaler A. Long-acting testosterone injections for treatment of testosterone deficiency after brachytherapy for prostate cancer. BJU Int. 2014;114:125–30.
Sarosdy MF. Testosterone replacement for hypogonadism after treatment of early prostate cancer with brachytherapy. Cancer. 2007;109:536–41.
Kadomoto S, Shigehara K, Iwamoto H, Yaegashi H, Izumi K, Kadono Y, et al. Testosterone replacement therapy for patients with hypogonadism after high dose-rate brachytherapy for high-risk prostate cancer: a report of six cases and literature review. World J Mens Health. 2020;38:132 https://doi.org/10.5534/wjmh.180130
Author information
Authors and Affiliations
Consortia
Contributions
JG was responsible for development of the search strategy, study selection criteria, screening of studies, data charting, consensus resolution, data curation, visualisation, writing of the original draft, and overall project administration. MG contributed to manuscript review, editing, and visualisation. PG assisted with study screening and participated in consensus resolution during the selection and data charting processes. AM, IP, and TS contributed to critical review and editing of the manuscript. VM provided supervision, guidance on methodology, and oversight of the review process. All authors reviewed and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical considerations
This review was conducted using previously published data and did not involve direct patient contact or the use of unpublished data. As such, ethics committee approval was not required.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Gibson, J., George, M., Grice, P. et al. Testosterone replacement therapy following definitive treatment for prostate cancer: a scoping review of safety and efficacy. Int J Impot Res (2025). https://doi.org/10.1038/s41443-025-01206-3
Received:
Revised:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41443-025-01206-3