Abstract
Metal promotion is the most widely adopted strategy for enhancing the hydrogenation functionality of an oxide catalyst. Typically, metal nanoparticles or dopants are located directly on the catalyst surface to create interfacial synergy with active sites on the oxide, but the enhancement effect may be compromised by insufficient hydrogen delivery to these sites. Here, we introduce a strategy to promote a ZnZrOx methanol synthesis catalyst by incorporating hydrogen activation and delivery functions through optimized integration of ZnZrOx and Pd supported on carbon nanotube (Pd/CNT). The CNT in the Pd/CNT + ZnZrOx system delivers hydrogen activated on Pd to a broad area on the ZnZrOx surface, with an enhancement factor of 10 compared to the conventional Pd-promoted ZnZrOx catalyst, which only transfers hydrogen to Pd-adjacent sites. In CO2 hydrogenation to methanol, Pd/CNT + ZnZrOx exhibits drastically boosted activity—the highest among reported ZnZrOx-based catalysts—and excellent stability over 600 h on stream test, showing potential for practical implementation.
Similar content being viewed by others

Introduction
Aiming to mitigate anthropogenic CO2 and utilize it as a valuable product, CO2 hydrogenation to methanol has received extensive interest from academia and industry1,2,3,4,5. Carbon Recycling International established the world’s first large-scale CO2-to-methanol plant using Cu/ZnO/Al2O3, a classical catalyst originally developed and optimized for syngas-to-methanol conversion6. Numerous efforts are still being undertaken to elucidate the active site and optimize catalyst activity7,8,9,10. Nevertheless, one of the critical limitations of Cu/ZnO/Al2O3 is deactivation due to the unsatisfactory stability of the catalyst under exposure to heat and moisture, hampering long-term process operation11,12,13,14. Consequently, several Cu-free metal oxides have been reported as new classes of catalysts for CO2 hydrogenation to methanol15,16,17,18,19, among which, ZnO-ZrO2 solid solution catalyst (ZnZrOx) reported by Li and co-workers18 is undergoing pilot-scale testing20. One distinctive feature of ZnZrOx is its superior long-term stability; however, a downside is its lower activity compared to Cu/ZnO/Al2O3. Achieving both high activity and long-term stability is a challenging mission for developing next-generation CO2 hydrogenation catalysts.
Since H2 activation over metal oxide catalysts is often kinetically limited16,21,22, the metal promotion has been widely explored for improving methanol formation activity. In particular, for ZnZrOx, various metals such as Cu23, Pt23, Ga24, and Pd22,23,25 have been reported as effective metal promoters. It is a general belief that the metal promoter and oxide support should be placed close to each other to maximize interfacial synergy (including strong metal-support interaction), and thus, various synthetic techniques were developed to anchor metal promotors directly on the oxide catalyst surface26,27,28,29,30,31,32,33,34,35,36,37,38. Previously, we studied Pd-promoted ZnZrOx catalyst (Pd/ZnZrOx) for methanol formation22, revealing that the major role of Pd is to activate and split H2 into H atoms that are subsequently transferred to adjacent Zn sites for methanol synthesis. Thus, the rate of H supply to the active sites was identified as a key factor governing the rate of CO2 reduction to methanol. However, at present, there are limited ways to enhance the interfacial area between the Pd promoter and ZnZrOx catalyst for traditional metal/oxide nanocomposite catalysts.
Herein, we address this challenge by combining ZnZrOx with Pd supported on carbon nanotubes (CNT): Pd/CNT + ZnZrOx. CNT is a well-known material for hydrogen storage39,40,41, and doping CNT with metal- or metal oxide- greatly enhances its H storage capacity, hinting at swift hydrogen transfer between CNT and the dopants40,41,42,43,44. Moreover, Crossley and co-workers conducted an elegant study of the nature of active sites on Pd/TiO2 and Cu/TiO2 catalysts in furfural and anisole hydrogenations by separating metal from the support by a controlled distance using CNT, further substantiating that CNT may act as a bridge between metal and oxide45. Torrente-Murciano and co-workers reported the superiority of graphitized CNT as a support for Ru catalysts in NH3 cracking, due to the electronic modification of Ru particles by CNT46. Based on these previous reports, and the unique property of CNT, we presumed that Pd/CNT can deliver hydrogen to ZnZrOx via hydrogen activation on Pd and spillover via CNT. Our experiments showed that Pd/CNT + ZnZrOx features drastically improved methanol formation activity from CO2 compared to not only ZnZrOx, but also Pd/ZnZrOx with the same Pd content. The Pd/CNT + ZnZrOx additionally exhibits excellent long-term stability, surpassing the performance of the state-of-the-art Cu/ZnO/Al2O3 industrial catalysts during the on-stream test from 200 h onwards.
Results
Preparation and characterization of the catalysts
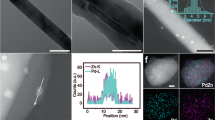
The structures of ZnZrOx, Pd/ZnZrOx, and Pd/CNT + ZnZrOx catalysts were characterized using various techniques. According to TEM and SEM analyses, ZnZrOx is composed of ca. 10–15 nm crystallites, which are present as agglomerated ~µm-scale particles (Supplementary Fig. 1). Pd/CNT contains uniformly dispersed Pd nanoparticles (ca. 5 nm) inside and outside the carbon tubes. (Supplementary Fig. 2). XRD analysis proves that ZnZrOx has a t-ZrO2 phase-like crystalline solid solution structure18. The crystallinity of ZnZrOx is well-preserved after Pd-impregnation as well as physical mixing with Pd/CNT (Fig. 1a). The TEM image of Pd/ZnZrOx shows the presence of ca. 5 nm Pd nanoparticles on the surface of ZnZrOx (Fig. 1b). In Pd/CNT + ZnZrOx, the majority of Pd particles are present far away from the ZnZrOx surface (Fig. 1c). As Pd/CNT and ZnZrOx entangle with each other (Fig. 1c, d), EDX elemental mapping in the 2D-projection shows a uniform distribution of Pd over the catalyst domain (Fig. 1e). H2- and CO-chemisorption analysis confirms that Pd/ZnZrOx and Pd/CNT have a similar Pd dispersion of 18–20%, which is consistent with TEM analysis. Of note, Pd dispersion is not altered during physical mixing (Supplementary Table 1). Based on these results, we present an illustration of the three catalysts in Fig. 1f. Furthermore, Pd/GNP (GNP: graphene nanoplatelets), Pd/AC (AC: activated carbon), Pd/SiO2 and Pd/TiO2 were prepared and mixed with ZnZrOx as control samples. The characterizations of these catalysts are included in Supplementary Figs. 3, 4 and Supplementary Table 1. The general observations about their structural features are very similar to those of Pd/CNT and Pd/CNT + ZnZrOx.
a Powder XRD patterns of ZnZrOx, Pd/ZnZrOx, and Pd/CNT + ZnZrOx. The peak shift to higher 2θ of ZnZrOx compared to pure ZrO2 indicates the substitution of Zr atoms to Zn atoms (i.e., solid solution formation). The XRD pattern of pure t-ZrO2 is obtained from Materials Data on ZrO2 by Materials Project, ID: mp-2574. b TEM image of Pd/ZnZrOx. c TEM image of Pd/CNT + ZnZrOx. d SEM image of Pd/CNT + ZnZrOx. e EDX elemental maps of Pd/CNT + ZnZrOx during TEM analysis. f Illustration of the structures for ZnZrOx, Pd/ZnZrOx, and Pd/CNT + ZnZrOx catalysts (o.d. outer diameter).
Unique CO2 hydrogenation performance of Pd/CNT + ZnZrOx
The activities of ZnZrOx and various Pd-promoted ZnZrOx catalysts for CO2 hydrogenation to methanol (MeOH) were investigated under the reaction kinetics-controlled region (conditions: 533 K, 5 MPa total pressure with CO2/H2/Ar = 19/76/5 (molar), conversion: 0.64–1.36%). The exclusion of external and internal mass transfer limitation is described in Section 3 in Supplementary Information.
CO2 conversion and product selectivity of the catalysts are shown in Supplementary Table 2. For all catalysts, the major products are methanol and CO. Only Pd/SiO2 + ZnZrOx and Pd/TiO2 + ZnZrOx produced CH4 (≤3.40%) and dimethyl ether (DME, ≤0.14%) as side products. Since standalone Pd catalysts have negligible methanol formation activity compared to ZnZrOx (Supplementary Fig. 5), the methanol formation rate (rMeOH) of ZnZrOx-containing catalysts in Fig. 2a was represented based on the mass of ZnZrOx. Pd/ZnZrOx exhibits higher rMeOH (3.96 mmol gZnZrOx−1 h−1) than ZnZrOx (1.69 mmol gZnZrOx−1 h−1), illustrating the promotional effect from surface Pd nanoparticles. Unexpectedly, a higher rMeOH is observed over Pd/CNT + ZnZrOx (13.44 mmol gZnZrOx−1 h−1) compared to Pd/ZnZrOx, which is an exceptional finding, since the metal promoter (Pd) induces a significantly more pronounced effect when away from the active sites on ZnZrOx. Considering that Pd/ZnZrOx and Pd/CNT + ZnZrOx possess similar Pd dispersion and identical Pd loading with respect to ZnZrOx (Supplementary Table 1), and that no promotion was observed when ZnZrOx is mixed with CNT (CNT + ZnZrOx), both Pd and CNT are concluded to be essential for the superior catalytic performance of Pd/CNT + ZnZrOx. A strong promoting effect is also observed when Pd/ZnZrOx is mixed with CNT (i.e., rMeOH = 7.63 mmol gZnZrOx−1 h−1 for CNT + Pd/ZnZrOx) as well as when ZnZrOx is mixed with other carbon-supported Pd catalysts (i.e., Pd/GNP + ZnZrOx, Pd/AC + ZnZrOx). In contrast, when ZnZrOx is mixed with Pd/oxide catalysts such as Pd/SiO2 and Pd/TiO2 (i.e., Pd/SiO2 + ZnZrOx, Pd/TiO2 + ZnZrOx), there is only slight promotion, demonstrating the unique role of carbon supports. Although TiO2 is a well-known reducible oxide for fast hydrogen spillover, Pd/TiO2 has an order of magnitude higher CO2 hydrogenation activity toward CO compared to other Pd/support catalysts (Supplementary Fig. 5). Thus, Pd/TiO2 converts CO2 and H2 by itself rather than acting as a ‘hydrogen dispenser’. These observations indicate promoting performance is related to hydrogen spillover efficiency, which strongly depends on the type of support as hydrogen moves as an H+/e− pair47,48,49.
a Methanol formation rate (rMeOH) over ZnZrOx-based catalysts (Condition: 533 K, 5 MPa, CO2/H2/Ar = 19/76/5 (molar), gas hourly space velocities (GHSV) are shown in Supplementary Table 2). b Reaction orders with respect to H2 and CO2 for methanol formation. c Apparent activation energy for methanol formation. The values were determined by the Arrhenius plot shown in Supplementary Fig. 7. d Effect of catalyst mixing configurations on methanol formation in the combination of Pd/CNT and ZnZrOx. Inset shows the illustration of each mixing configuration. The green dashed line in Fig. a, d indicates the activity of Pd/ZnZrOx.
In reaction kinetic analyses (Fig. 2b and Supplementary Fig. 6), the reaction orders with respect to H2 (nH2) and CO2 (nCO2) for methanol synthesis over ZnZrOx are ca. 1.0 and 0, respectively. The results imply that the surface coverage of H2 is much lower than that of CO2. The nH2 = ca. 1.0 suggests that H2 activation may be a rate-determining step. Pd/ZnZrOx catalyst shows similar nH2 and nCO2 values to those of pristine ZnZrOx. In contrast, there is a substantial decrease of nH2 (0.84) alongside a simultaneous increase of nCO2 (0.21) for Pd/CNT + ZnZrOx. The result indicates the coverage of hydrogen on the ZnZrOx surface increased considerably through combination with Pd/CNT. As shown in Fig. 2c and Supplementary Fig. 7, Pd/CNT + ZnZrOx exhibits drastically decreased apparent activation energy (Ea = 0.50 eV) compared to ZnZrOx (Ea = 1.18 eV), confirming the immense contribution of physical mixing with Pd/CNT on accelerating reaction kinetics.
For the ZnZrOx and Pd/CNT combination, the effect of mixing configuration was studied to gain insight into the origin of promotion (Fig. 2d). Four different mixing configurations were prepared by varying the mixing method, namely separated bed, large pellet mix (size: 450–850 µm), small pellet mix (size: 180–450 µm), and mortar grind mix (size: 0.2–20 µm). The separated bed configuration gives a detrimental effect on methanol formation, implying the promoting effect in Pd/CNT + ZnZrOx is not originated from the transport of intermediates in the gas-phase. On the other hand, a higher activity compared to pristine ZnZrOx was detected for all other configurations. The promoting effect is strengthened as the contact between the two components (Pd/CNT and ZnZrOx) increases, reaching the maximum in the mortar grind mix configuration. It is noteworthy that even when the two components are mixed in 180–850 μm-scale, the promoting effect (rMeOH = 4.81–6.42 mmol gZnZrOx−1 h−1) is stronger than that of Pd/ZnZrOx (Fig. 2a, rMeOH = 3.96 mmol gZnZrOx−1 h−1). This result firmly demonstrates the effectiveness of the strategy for hydrogen delivery; hydrogen transfer efficiency is no longer limited by the number of Pd-ZnZrOx interface sites, but rather by the closeness of ZnZrOx particles and CNT.
We further tested the performance of Pd/CNT + ZnZrOx under industry-relevant reaction conditions (5 MPa, GHSV = 24,000 cm3STP gcat.−1 h−1) to evaluate the practical potential of this catalyst. First, CO2 conversion and selectivity of products at different temperatures (513–633 K) are summarized in Supplementary Table 3. The result indicates that methanol and CO are the major products under these reaction conditions. The selectivity of Pd/CNT + ZnZrOx is lower than that of ZnZrOx and Pd/ZnZrOx due to the CO formation activity of Pd/CNT itself (Supplementary Fig. 5). Nevertheless, the MeOH selectivity gap between Pd/CNT + ZnZrOx and ZnZrOx is less than 10% at the same CO2 conversion level (Supplementary Fig. 8).
Methanol yield and corresponding space-time yield of methanol (STYMeOH) were monitored while varying reaction temperature (Fig. 3a). The STYMeOH for Pd/CNT + ZnZrOx is 0.780 g gcat.−1 h−1 at 593 K, the highest value among all reported ZnZrOx-based catalysts (Supplementary Table 4). Moreover, the long-term stability of each catalyst was monitored at 593 K while a commercial Cu/ZnO/Al2O3 catalyst was simultaneously introduced as a control sample. As shown in Supplementary Fig. 9, the optimal reaction temperature for Cu/ZnO/Al2O3 catalyst that shows the maximum STYMeOH is 533 K (thermodynamic equilibrium MeOH yield = 24%), thus the long-term test was conducted at this temperature for Cu/ZnO/Al2O3. Not unexpectedly, Cu/ZnO/Al2O3 exhibits relatively high activity initially but shows rapid deactivation from the beginning (Fig. 3b); after 600 h of reaction, 46% of initial activity has been lost. In contrast, Pd/CNT + ZnZrOx catalyst shows superior long-term performance; the methanol yield over Pd/CNT + ZnZrOx reaches 13.5% (STYMeOH = 0.90 g gcat.−1 h−1) at 600 h time-on-stream, very close to the value at thermodynamic equilibrium (MeOH yield = 14.0% at 593 K). XANES and EXAFS analyses after the reaction reveal that there are no substantial changes in the chemical state of either Zn or Zr in Pd/CNT + ZnZrOx, validating the superior stability of Pd/CNT + ZnZrOx (Supplementary Figs. 10–11). Both catalysts were additionally tested at a higher GHSV (80,000 cm3STP gcat.−1 h−1) to shift conversion further away from thermodynamic equilibrium. Again, Cu/ZnO/Al2O3 exhibited continued deactivation while the Pd/CNT + ZnZrOx system kept increasing its methanol productivity during a 104 h test. (Supplementary Fig. 12). Compared to ZnZrOx and Pd/ZnZrOx, distinct increases in conversion and methanol selectivity are observed for Pd/CNT + ZnZrOx, particularly during the initial stage of reaction (Fig. 3c). Under the steady state (time-on-stream >400 h), Pd/CNT + ZnZrOx shows 66% methanol selectivity at 20% CO2 conversion, higher than the methanol selectivity of Cu/ZnO/Al2O3 at comparable CO2 conversion (50–52% at 23–20% CO2 conversion). As a result of the opposite activity variation trend, cumulative methanol production (QMeOH) of Pd/CNT + ZnZrOx overwhelms that of commercial Cu/ZnO/Al2O3 after 360 h reaction (Fig. 3b, inset). At 600 h, Pd/CNT + ZnZrOx offered a 1.44-fold higher methanol yield over the commercial Cu/ZnO/Al2O3 catalyst, and its STYMeOH (g gcat.−1 h−1) compare favorably with state-of-the-art catalysts, that were scrutinized in long-term stability tests in the literature (Supplementary Table 5), further validating the superior performance of the Pd/CNT + ZnZrOx catalyst. In addition, Pd/CNT + ZnZrOx contains 33 wt% CNT, which can act as a catalyst binder/diluent due to its high mechanical strength50 and thermal conductivity51. Therefore, Pd/CNT + ZnZrOx may not require further use of an extra binder/diluent, providing additional benefits for practical implementation.
a Methanol yield and STYMeOH of the ZnZrOx-based catalysts as a function of reaction temperature. The dashed line indicates the methanol yield at thermodynamic equilibrium. b Long-term catalytic tests of ZnZrOx-based catalysts (593 K), and commercial Cu/ZnO/Al2O3 (533 K). The inset shows cumulative methanol production (QMeOH, g gcat.−1) as a function of time-on-stream (Condition: 5 MPa, CO2/H2/Ar = 19/76/5, GHSV = 24,000 cm3STP gcat.−1 h−1). c CO2 conversion and methanol selectivity changes during long-term reaction test over ZnZrOx, Pd/ZnZrOx, Pd/CNT + ZnZrOx, and commercial Cu/ZnO/Al2O3 catalysts.
Characterization of the promotion effect in Pd/CNT + ZnZrOx
The origin of promotion was elucidated by quantitative gas adsorption-desorption analyses. CO2-TPD profiles reveal ZnZrOx, Pd/ZnZrOx, and Pd/CNT + ZnZrOx have comparable adsorption strengths (Supplementary Fig. 13) and capacity of CO2 (Fig. 4a), indicating CO2 adsorption properties of the catalysts are similar. H2–D2 isotope exchange experiments were then conducted at 373 K (Fig. 4b) to determine their H2 activation function. ZnZrOx shows a low H–D exchange rate (8.93 mmol gZnZrOx−1 min−1), while Pd/ZnZrOx and Pd/CNT + ZnZrOx show accelerated rates at 42.9 and 49.8 mmol gZnZrOx−1 min−1, respectively. We draw two major conclusions from these isotope exchange experiments. First, Pd species easily activate H2, regardless of whether Pd nanoparticles are directly supported on ZnZrOx, or on CNT. Second, the H2 activation function alone is not the critical factor in determining methanol formation activity, since the H–D exchange rates on Pd/ZnZrOx and Pd/CNT + ZnZrOx are comparable despite their pronounced difference in methanol production.
a CO2 adsorption capacity of catalysts determined by CO2-TPD. b H–D exchange rate of catalysts at 373 K. c H2-TPD profile and quantification of reversible H2 of catalysts. The quantification of reversible H2 was conducted by integrating the peak area in the range of 323–533 K which is below the temperature of our reaction kinetic analysis. d Zn 2p3/2 XPS of ZnZrOx and Pd/CNT + ZnZrOx catalysts before and after sequential gas treatments (gas treatment condition: H2 at 673 K, CO2 at 533 K, and CO2 + H2 (1:4) at 533 K).
Next, H2-TPD profiles of various catalysts were examined to differentiate their ability in delivering activated hydrogen (H*) (Fig. 4c). ZnZrOx shows an H2 desorption peak above 423 K, while Pd-containing catalysts exhibit a peak starting from 343 K due to more facile desorption of H2 from the Pd surface. The amounts of adsorbed H2 on ZnZrOx and Pd/ZnZrOx were 2.3 and 4.1 µmol gcat.−1, respectively. In sharp contrast, Pd/CNT + ZnZrOx shows an order of magnitude increase in H2 adsorption (39 µmol gcat.−1). Considering Pd/CNT alone only possesses 7.1 µmol H2 gcat.−1, and that Pd/CNT accounts for one-third of the mass in the mixture catalyst, over 90% of the adsorbed hydrogen in the Pd/CNT + ZnZrOx mixture is stored at the interfaces between ZnZrOx and CNT, which undergo reverse spillover during TPD analysis (Fig. 4c, right panels). In the case of Pd/SiO2 and Pd/TiO2, there are only marginal increments in the H2 adsorption capacity after physical mixing with ZnZrOx (Supplementary Fig. 14), confirming the unique property of CNT as a hydrogen delivery system. Band gap analysis suggests carbonaceous materials have much smaller band gap values (0.74–0.75 eV) compared to metal oxides (≥3.09 eV) including ZnZrOx (Supplementary Fig. 15), suggesting higher electrical conductivity results in faster migration kinetics of hydrogen (H+/electron pair) on the carbon surface versus oxides such as ZnZrOx.
X-ray photoelectron spectroscopy (XPS) after sequential gas treatments (without sample exposure to air) for ZnZrOx and Pd/CNT + ZnZrOx were carried out to provide more insight into the hydrogen spillover. The spectra of each element are demonstrated in Supplementary Figs. 16–20, while the XPS of Zn 2p3/2 region is represented in Fig. 4d. Before gas treatment, ZnZrOx and Pd/CNT + ZnZrOx show identical symmetric Zn 2p3/2 signals at 1022 eV which represent Zn2+ bound to lattice oxygen (Zn2+–O2−). After H2 treatment at 673 K, ZnZrOx shows no signal changes, whereas Pd/CNT + ZnZrOx exhibits the formation of a shoulder peak at ~1 eV lower binding energy region. The transformation of Zn2+–O2− to reduced Zn species (Znδ-)52,53 is a result of electron transfer from spilt over hydrogen to the Zn site of the ZnZrOx surface. Both H2-reduced catalysts were further treated with CO2 and CO2 + H2 after which a shoulder peak at ~1 eV higher binding energy region appears, indicative of CO2 chemisorption on Zn–O pair (Zn2+–CO32−)53. Notably, the existence of Znδ- species is still pronounced in Pd/CNT + ZnZrOx after consecutive treatments with CO2 and CO2 + H2. The portion of Znδ- is slightly decreased after CO2 treatment (from 63 to 57%), but it recovers the original fraction when treated with CO2 + H2 (65%), providing evidence of effective hydrogen spillover in the Pd/CNT + ZnZrOx system under CO2 hydrogenation condition.
Computational studies on hydrogen spillover in Pd/CNT + ZnZrOx
Density functional theory calculations were applied to provide an atomistic description of hydrogen spillover in the Pd/CNT + ZnZrOx nanocomposite. After identifying the most stable model for ZnZrOx (Supplementary Figs. 21–24), we studied the adsorption and movement of H on the bare ZnZrOx surface (Fig. 5 and Supplementary Fig. 25). H2 dissociation on pure ZrO2 is kinetically limited54, which can be improved by doping the oxide with Zn atoms18,55. We calculated Zn–O pairs in the ZnZrOx surface to have the lowest dissociative adsorption energies of H2 among all surface sites (Supplementary Fig. 25b). Since our ZnZrOx supercell contains two Zn sites (Fig. 5a), we are able to perform simulations for the sequential dissociation of two H2 molecules. The dissociation of the first H2 molecule is a barrierless step (Fig. 5b) with reaction energy of ΔG = − 0.31 eV (Fig. 5c). Yet, the dissociation of the second H2 molecule is impaired by a higher activation barrier (ΔGa = 0.71 eV) and reaction energy of ΔG = 0.17 eV (Fig. 5c), indicating limited H coverage on unmodified ZnZrOx under reaction conditions.
a Top view of four employed ZnZrOx supercells illustrating 4 H atoms adsorbed per unit cell on Zn–O pairs, formed after successive dissociation of two H2 molecules. b Potential energy profile along the reaction coordinate for barrierless dissociation of the first H2 molecule (TS transition state, FS final state). c Potential energy profile of successive H2 dissociation at ZnZrOx for 2 H2 molecules. d Energy profile for H movement from Zn to Zr, O to O, and Zr to Zr on the ZnZrOx surface. The inset images show the transition state configurations. Turquoise, purple, red, and yellow colors represent Zr, Zn, O, and H, respectively.
Moreover, H atoms are calculated to have negligible mobility on the ZnZrOx surface with barriers up to ΔGa = 1.18 eV for diffusion between O sites and ΔGa = 1.51 eV for migration of H atoms from Zn to Zr atoms (Fig. 5d). Although a much lower barrier of ΔGa = 0.34 eV was calculated for H diffusion between Zr sites, these sites were calculated to be up to 2.22 eV less stable for H than O sites on ZnZrOx surface based on the adsorption energy of one H atom. These suggest that directly supported Pd nanoparticles cannot deliver reactive H atoms to the active sites on ZnZrOx since long-distance H spillover is hindered by high diffusion barriers of H atoms on the oxide surface.
In contrast to the bare ZnZrOx, our calculations show that H can be fluently supplied to the active sites on the ZnZrOx surface via spillover from Pd/CNT (Fig. 6). We created models of physisorbed CNT on ZnZrOx using an adapted lattice matching algorithm, which ensured the minimal strain of −2.33% along the CNT length (Fig. 6a). According to our simulations, a single H atom bind stronger to ZnZrOx than to CNT, which creates a significant thermodynamic driving force for the spillover and makes this process essentially barrierless (Fig. 6c). However, the spillover barrier increases when more H atoms are adsorbed on CNT. For example, the Gibbs barrier for the spillover of an H atom from CNT to ZnZrOx increases to 0.43 eV when another H atom is adsorbed in the most stable position on the vicinal site on the inner side of the nanotube (Fig. 6d). In turn, H spillover from CNT to ZnZrOx becomes endothermic by about ΔG ~ 2.5 eV when every C atom of CNT is hydrogenated. In conclusion, low H coverage on CNT can be regarded as the critical structural factor enabling hydrogen spillover from C to the oxide. Low H coverage on CNTs is also suggested by our XPS studies showing mostly sp2 hybridization of C atoms in the CNT under reaction conditions (Supplementary Fig. 19).
a Atomic models of CNT(5,5) on ZnZrOx and Pd on carbon (a graphene sheet) for H spillover. b Energy profile for movement of one H from hydrogen-saturated Pd to the graphene sheet with a pre-adsorbed H atom (IS initial state, TS transition state, and FS final state). c Energy profile for spillover of the single H atom from CNT(5,5) to ZnZrOx. d Energy profile for spillover of one H atom from CNT(5,5) with another H atom adsorbed on it to ZnZrOx.
To evaluate the rate of H spillover from Pd to sp2 carbon, we simulated the movement of H from a 1.56 nm large Pd127 fcc nanocrystallite to a graphene sheet representing a broad nanotube. We chose the shape of the Pd127 particle in line with TEM data and identified the most stable structure of the Pd127/graphene interface by screening all high-symmetry interfaces (Supplementary Fig. 26). Our calculations show that the barriers for H spillover from Pd to graphene are essentially governed by the endothermicity of this process (Fig. 6b), which depends on the H coverage. Namely, the spillover of the first H atom from Pd127 to graphene is endothermic by ΔG = 1.57 eV, which decreases to ΔG = 0.37 eV when another H atom is present on the graphene sheet. Since the Gibbs adsorption energy per H atom of one (two) H on the freestanding CNT(5,5) is 1.04 eV (0.70 eV/H atom), which is 0.84 eV (0.37 eV/H atom) stronger than on graphene, the curvature of the CNT should further favor the transfer of H from Pd to the carbon domain. Thus, the barriers for H spillover from Pd to CNT are estimated to be much lower than the activation energy of H2 dissociation on ZnZrOx (ΔGa = 0.71 eV). In turn, CNTs are well known to be excellent transport channels for H atoms45, whose movement over graphene was shown to be enabled by quantum tunneling56. Thus, our simulations suggest a much faster H supply to the active sites on Pd/CNT + ZnZrOx nanocomposite compared to the pristine ZnZrOx.
Effect of catalyst structure on catalytic results in CO2-to-methanol
A number of studies using metal/support catalysts (e.g., Cu or Pd on ZnO, ZrO2, or ZnZrOx) refer to hydrogen spillover as an important process for methanol synthesis from CO/CO223,57,58,59,60,61,62,63, but it is difficult to disentangle catalytic activity generated by sites adjacent to metals from that generated by sites farther away64. In Fig. 7, we present the effect of the catalyst structure on CO2 hydrogenation to methanol over Pd-promoted ZnZrOx catalysts. ZnZrOx is an excellent material to chemisorb CO2, but methanol formation is limited due to low H2 activation and H* delivery capability (Fig. 7a). In turn, Pd particles supported on ZnZrOx significantly enhance H2 activation, but the delivery of H* toward the active sites is hampered due to slow H-migration kinetics within ZnZrOx surface (Fig. 7b). It is estimated the contact area between Pd particles and ZnZrOx is about 0.5 m2 gcat.−1, only 1% of the entire catalyst surface area (estimation procedure available in Section 4 in Supplementary Information). Our experimental and computational studies support Prins’s rigorous critique that hydrogen may not travel farther than the immediate interface between metal and basic oxide support47, and Li et al.’s hypothesis that the rate-determining step of CO2 hydrogenation to methanol over various catalysts (i.e., ZnZrOx, Cu/ZnO/Al2O3, and Pd/ZnO) is the migration of H* on the catalyst surface65. In the case of Pd/CNT + ZnZrOx, CNT acts as a hydrogen dispenser, effectively delivering activated H* from Pd particles to the ZnZrOx surface (Fig. 7c). Since the promotion range is determined by the contact area between CNT and ZnZrOx, a ten-times larger promotion area is generated (as determined by H2-TPD result) under mortar grind mode compared to traditional metal doping on the catalyst surface, leading to a drastically enhanced methanol formation rate. In the literature, non-contact integration of multiple catalytically active components, especially physical mixing, has been adopted in catalyst development for bifunctional- or cascade catalytic reactions for which each catalyst is responsible for one step of the entire transformation66,67,68,69,70,71,72,73,74. Our work, in contrast, reports a bicomponent catalyst in which one component provides active sites for the formation of a target product while the other serves as an activation and delivery system for a kinetically hindered intermediate, which to our knowledge, has not been demonstrated for CO2 hydrogenation.
a ZnZrOx catalyst without any metal promotion. b Pd-supported ZnZrOx catalyst which represents the traditional metal/oxide catalyst harnessing metal promotion. c Pd/CNT + ZnZrOx catalyst which represents a conceptually new catalyst system harnessing metal promotion with a hydrogen dispenser.
Furthermore, our preliminary tests of physical mixing of commercial Cu/ZnO/Al2O3 with CNT results in 1.4 to 2.0-times higher methanol formation activity versus pristine Cu/ZnO/Al2O3 in the pressure range of 0.1–1.0 MPa (Supplementary Fig. 27), suggesting this promotion strategy is not limited to ZnZrOx. For the successful implementation of the CO2-to-MeOH process on large scales, recycling of the product stream (including unconverted CO2, H2, and produced CO) is necessary to intensify process economics75,76. Future research should be directed to studying the effect of CO on nanoscale H supply in newly developed systems.
Discussion
Insufficient H2 activation has long been known to limit the activity of oxide catalysts, such as ZnZrOx, for CO2 reduction to methanol. In this work, we report CNT as a hydrogen dispenser for enhancing hydrogen delivery to active sites on ZnZrOx. The combination of Pd/CNT and ZnZrOx leads to an exceptional catalytic system for methanol formation, far better than Pd/ZnZrOx catalyst, as CNT effectively delivers Pd-activated hydrogen to a broad range of ZnZrOx surface sites. The Pd/CNT + ZnZrOx shows the highest STYMeOH among reported non-Cu/ZnO-based catalysts to date and is durable over 600 h continuous operation, demonstrating its potential for practical implementation; concurrently, the physical mixing strategy offers a robust and transferable method for material integration to design better catalytic systems. This study provides an example in oxide catalyst design for which the transition metal promoter in non-contact mode induces a greater effect than the transition metal promoter directly attached to the surface of the catalytically active phase, expanding the concept of metal promotion beyond the traditional focus of metal-support interfaces.
Methods
Catalysts preparation
ZnO-ZrO2 (Zn/Zr = 1/5) solid solution catalyst (ZnZrOx) was prepared via a coprecipitation method with reference to the earlier literature22. An 1 wt% Pd/ZnZrOx catalyst was prepared by wet impregnation using Pd(NO3)2·xH2O as Pd precursor. To make the Pd/CNT + ZnZrOx catalyst, 2 wt% Pd was first supported on CNT by wet impregnation followed by reduction at 673 K, and then Pd/CNT was physically mixed with ZnZrOx with Pd:ZnZrOx = 1:100 mass ratio. Physical mixing was carried out by mortar grinding, unless otherwise stated. For comparison, various other physically mixed catalysts (Pd/support + ZnZrOx) were also prepared in a similar manner while varying the support material (support = type of support used; AC: activated carbon, GNP: graphene nanoplatelet, SiO2: silica, TiO2: titania P25). CNT + ZnZrOx and CNT + Pd/ZnZrOx were also prepared by physical mixing of CNT and ZnZrOx or Pd/ZnZrOx. Prior to the characterizations and catalytic reactions, the catalysts were mortar ground. As shown in Section 5 in Supplementary Information, the mortar grinding itself does not significantly affect the surface area and catalytic activity of ZnZrOx.
Catalyst characterization
The structures of catalysts were characterized using transmission electron microscopy (TEM) coupled with energy dispersive X-ray (EDX) elemental analysis, scanning electron microscopy (SEM), powder X-ray diffraction (XRD), N2 physisorption, H2 and CO chemisorption, CO2- and H2-temperature-programmed desorption (TPD), H2–D2 isotope exchange, UV-Vis-NIR spectroscopy, and X-ray photoelectron spectroscopy (XPS).
Catalytic test
CO2 hydrogenation was carried out using an automatic multi-channel high-pressure flow reactor designed by PID Eng & Tech. Prior to the reaction, the catalysts were pressed, cracked, and sieved to make pellets. The sieved catalyst pellets (450–850 µm) were loaded into the reactor, and pretreated with H2 (40 cm3STP min−1) at 673 K for 2 h (For Cu/ZnO/Al2O3 catalysts, pretreatments were conducted at 573 K). After the pretreatment, the reactor was cooled down to a target temperature (493–633 K), and was pressurized with a reaction gas mixture (CO2/H2/Ar, typically 19/76/5). The outlet gas line was kept at 453 K to prevent condensation of products, and products were analyzed by online gas chromatography (Agilent 8890) equipped with a flame ionization detector (FID) and a thermal conductivity detector (TCD). Agilent HP-PoraplotQ and Restek ShinCarbon were used as columns. Reported data are given as values of time-on-stream at 3 h unless otherwise stated.
Computational studies
The computational approach was based on spin-unpolarized density functional theory within the Perdew-Burke-Ernzerhof77 exchange-correlation approximation as implemented in the Vienna Ab initio simulation package (VASP)78,79. The zero-damping D3 correction was added to improve the description of dispersive interactions80. To treat strong correlation effects for d electrons, the simplified implementation of Hubbard corrections81 was adopted with U − J = 4 eV on Zr atoms. Atomic charges were assigned using Bader charge analysis82,83. The Gibbs adsorption energies of H were calculated as \({G}_{{ad}}=G[{{{{{\rm{H}}}}}}/{{{{{\rm{cat}}}}}}]-E[{{{{{\rm{cat}}}}}}]-0.5\,G[{{{\mbox{H}}}}_{2}]\), where, \(G[{{{{{\rm{H}}}}}}/{{{{{\rm{cat}}}}}}]\) is the Gibbs total energy of the system with the adsorbate, while \(E[{{{{{\rm{cat}}}}}}]\) and \(G[{{{{{{\rm{H}}}}}}}_{2}]\) are the total and Gibbs total energy of the isolated catalyst model and H2 molecule in the gas phase. All energetic properties except for the relative energies in Figs. 5b and 6b–d, include Gibbs corrections calculated at 533 K and 4 MPa of H2 pressure under either the ideal gas or harmonic approximations84,85. In Fig. 6, the H movement coordinate is the quadratic distance of each structure with respect to the corresponding initial state. The k-point sampling was performed using Monkhorst-Pack meshes yielding converged adsorption energies of H, namely, the k-mesh parameters were 8 × 1 × 1 for all systems containing CNT(5,5), 2 × 2 × 1 for the 10×10 supercell of graphene, while the gamma point was sufficient for calculations involving only the ZnZrOx supercell. All models had a vacuum region of about 11 Å between adjacent slabs and all lateral distances between periodic images were at least 6 Å to keep interactions between periodic images minimal. All systems were optimized with the convergence criteria of 0.03 eV/Å and 10−5 eV for the force on each atom and total electronic energy. To optimize transition states, the climbing image nudged elastic band86,87 and dimer88 methods were employed and the optimized first-order saddle points were confirmed to have one imaginary vibrational frequency leading to the reported local minima.
Additional details of the methods for catalyst preparation, catalyst characterization, catalytic test, and computational studies are provided in the Supplementary Information.
Data availability
Relevant data supporting the key findings of this study are available within the article and the Supplementary Information file. All raw data generated during the current study are available from the corresponding authors upon request.
References
Olah, G. A., Goeppert, A. & Prakash, G. K. S. Chemical recycling of carbon dioxide to methanol and dimethyl ether: from greenhouse gas to renewable, environmentally carbon neutral fuels and synthetic hydrocarbons. J. Org. Chem. 74, 487–498 (2009).
Olah, G. A. Towards oil independence through renewable methanol chemistry. Angew. Chem. Int. Ed. 52, 104–107 (2013).
Alvarez, A. et al. Challenges in the greener production of formates/formic acid, methanol, and DME by heterogeneously catalyzed CO2 hydrogenation processes. Chem. Rev. 117, 9804–9838 (2017).
Jiang, X., Nie, X., Guo, X., Song, C. & Chen, J. G. Recent advances in carbon dioxide hydrogenation to methanol via heterogeneous catalysis. Chem. Rev. 120, 7984–8034 (2020).
Innovation Outlook: Renewable Methanol. (IRENA AND METHANOL INSTITUTE, International Renewable Energy Agency, 2021).
Richter, C. World’s First Commercial CO2 to Methanol Plant. https://smartech.gatech.edu/handle/1853/61845 (2019).
Kattel, S., Ramírez, P. J., Chen, J. G., Rodriguez, J. A. & Liu, P. Active sites for CO2 hydrogenation to methanol on Cu/ZnO catalysts. Science 355, 1296–1299 (2017).
Zabilskiy, M. et al. The unique interplay between copper and zinc during catalytic carbon dioxide hydrogenation to methanol. Nat. Commun. 11, 2409 (2020).
Divins, N. J. et al. Operando high-pressure investigation of size-controlled CuZn catalysts for the methanol synthesis reaction. Nat. Commun. 12, 1435 (2021).
Beck, A. et al. Drastic events and gradual change define the structure of an active copper-zinc-alumina catalyst for methanol synthesis. Angew. Chem. Int. Ed. 61, e202200301 (2022).
Sun, J. T., Metcalfe, I. S. & Sahibzada, M. Deactivation of Cu/ZnO/Al2O3 methanol synthesis catalyst by sintering. Ind. Eng. Chem. Res. 38, 3868–3872 (1999).
Twigg, M. V. & Spencer, M. S. Deactivation of copper metal catalysts for methanol decomposition, methanol steam reforming and methanol synthesis. Top. Catal. 22, 191–203 (2003).
Fichtl, M. B. et al. Kinetics of deactivation on Cu/ZnO/Al2O3 methanol synthesis catalysts. Appl. Catal. A Gen. 502, 262–270 (2015).
Liang, B. et al. Investigation on deactivation of Cu/ZnO/Al2O3 catalyst for CO2 hydrogenation to methanol. Ind. Eng. Chem. Res. 58, 9030–9037 (2019).
Martin, O. et al. Indium oxide as a superior catalyst for methanol synthesis by CO2 hydrogenation. Angew. Chem. Int. Ed. 55, 6261–6265 (2016).
Frei, M. S. et al. Role of zirconia in indium oxide-catalyzed CO2 hydrogenation to methanol. ACS Catal. 10, 1133–1145 (2020).
Dang, S. et al. Rationally designed indium oxide catalysts for CO2 hydrogenation to methanol with high activity and selectivity. Sci. Adv. 6, eaaz2060 (2020).
Wang, J. et al. A highly selective and stable ZnO-ZrO2 solid solution catalyst for CO2 hydrogenation to methanol. Sci. Adv. 3, e1701290 (2017).
Wang, J. et al. High-performance MaZrOx (Ma = Cd, Ga) solid-solution catalysts for CO2 hydrogenation to methanol. ACS Catal. 9, 10253–10259 (2019).
Thousand-ton scale demonstration of solar fuel synthesis starts operation in Lanzhou, China. EurekaAlrert (17 January 2020).
Frei, M. S. et al. Atomic-scale engineering of indium oxide promotion by palladium for methanol production via CO2 hydrogenation. Nat. Commun. 10, 3377 (2019).
Lee, K. et al. Atomic Pd-promoted ZnZrOx solid solution catalyst for CO2 hydrogenation to methanol. Appl. Catal. B Environ. 304, 120994 (2022).
Xu, D., Hong, X. & Liu, G. Highly dispersed metal doping to ZnZr oxide catalyst for CO2 hydrogenation to methanol: insight into hydrogen spillover. J. Catal. 393, 207–214 (2021).
Sha, F. et al. The promoting role of Ga in ZnZrOx solid solution catalyst for CO2 hydrogenation to methanol. J. Catal. 404, 383–392 (2021).
Huang, C. et al. CO2 hydrogenation to methanol over PdZnZr solid solution: effects of the PdZn alloy and oxygen vacancy. ACS Appl. Energy Mater. 4, 9258–9266 (2021).
Behrens, M. et al. The active site of methanol synthesis over Cu/ZnO/Al2O3 industrial catalysts. Science 6083, 893–897 (2012).
Li, S. et al. Tuning the selectivity of catalytic carbon dioxide hydrogenation over iridium/cerium oxide catalysts with a strong metal-support interaction. Angew. Chem. Int. Ed. 56, 10761–10765 (2017).
Meng, C. et al. Oxygen-deficient metal oxides supported nano-intermetallic InNi3C0.5 toward efficient CO2 hydrogenation to methanol. Sci. Adv. 7, eabi6012 (2021).
Liu, K. et al. Strong metal-support interaction promoted scalable production of thermally stable single-atom catalysts. Nat. Commun. 11, 1263 (2020).
Parastaev, A. et al. Boosting CO2 hydrogenation via size-dependent metal–support interactions in cobalt/ceria-based catalysts. Nat. Catal. 3, 526–533 (2020).
Chen, K. et al. CO2 hydrogenation to methanol over Cu catalysts supported on La-modified SBA-15: The crucial role of Cu–LaOx interfaces. Appl. Catal. B Environ. 251, 119–129 (2019).
Palomino, R. M. et al. Hydrogenation of CO2 on ZnO/Cu(100) and ZnO/Cu(111) catalysts: role of copper structure and metal-oxide interface in methanol synthesis. J. Phys. Chem. B 122, 794–800 (2018).
Graciani, J. et al. Highly active copper-ceria and copper-ceria-titania catalysts for methanol synthesis from CO2. Science 345, 546–550 (2014).
Rodriguez, J. A. et al. Hydrogenation of CO2 to methanol: importance of metal–oxide and metal–carbide interfaces in the activation of CO2. ACS Catal. 5, 6696–6706 (2015).
Wu, C., Cheng, D., Wang, M. & Ma, D. Understanding and application of strong metal–support interactions in conversion of CO2 to methanol: a review. Energy Fuels 35, 19012–19023 (2021).
Zabilskiy, M. et al. Mechanistic study of carbon dioxide hydrogenation over Pd/ZnO-based catalysts: the role of palladium-zinc alloy in selective methanol synthesis. Angew. Chem. Int. Ed. 60, 17053–17059 (2021).
Li, M. M. et al. Methanol synthesis at a wide range of H2/CO2 ratios over a Rh-In bimetallic catalyst. Angew. Chem. Int. Ed. 59, 16039–16046 (2020).
Wu, C. et al. Inverse ZrO2/Cu as a highly efficient methanol synthesis catalyst from CO2 hydrogenation. Nat. Commun. 11, 5767 (2020).
Lee, S. M. & Lee, Y. H. Hydrogen storage in single-walled carbon nanotubes. Appl. Phys. Lett. 76, 2877–2879 (2000).
Zacharia, R., Rather, S.-U., Hwang, S. W. & Nahm, K. S. Spillover of physisorbed hydrogen from sputter-deposited arrays of platinum nanoparticles to multi-walled carbon nanotubes. Chem. Phys. Lett. 434, 286–291 (2007).
Reyhani, A. et al. Hydrogen storage in decorated multiwalled carbon nanotubes by Ca, Co, Fe, Ni, and Pd nanoparticles under ambient conditions. J. Phys. Chem. C. 115, 6994–7001 (2011).
Lueking, A. & Yang, R. T. Hydrogen spillover from a metal oxide catalyst onto carbon nanotubes—implications for hydrogen storage. J. Catal. 206, 165–168 (2002).
Yoo, E. et al. Atomic hydrogen storage in carbon nanotubes promoted by metal catalysts. J. Phys. Chem. B 108, 18903–18907 (2004).
Mu, S.-C., Tang, H.-L., Qian, S.-H., Pan, M. & Yuan, R.-Z. Hydrogen storage in carbon nanotubes modified by microwave plasma etching and Pd decoration. Carbon 44, 762–767 (2006).
Briggs, N. M. et al. Identification of active sites on supported metal catalysts with carbon nanotube hydrogen highways. Nat. Commun. 9, 3827 (2018).
Hill, A. K. & Torrente-Murciano, L. Low temperature H2 production from ammonia using ruthenium-based catalysts: synergetic effect of promoter and support. Appl. Catal. B Environ. 172–173, 129–135 (2015).
Prins, R. Hydrogen spillover. facts and fiction. Chem. Rev. 112, 2714–2738 (2012).
Karim, W. et al. Catalyst support effects on hydrogen spillover. Nature 541, 68–71 (2017).
Roland, U., Braunschweig, T. & Roessner, F. On the nature of spilt-over hydrogen. J. Mol. Catal. A Chem. 127, 61–84 (1997).
Yu, M.-F. et al. Strength and breaking mechanism of multiwalled carbon nanotubes under tensile load. Science 287, 637–640 (2000).
Kumanek, B. & Janas, D. Thermal conductivity of carbon nanotube networks: a review. J. Mater. Sci. 54, 7397–7427 (2019).
Diler, E. et al. Initial formation of corrosion products on pure zinc and MgZn2 examinated by XPS. Corros. Sci. 79, 83–88 (2014).
Dake, L. S., Baer, D. R. & Zachara, J. M. Auger parameter measurements of zinc compounds relevant to zinc transport in the environment. Surf. Interface Anal. 14, 71–75 (1989).
Ruiz Puigdollers, A., Illas, F. & Pacchioni, G. Reduction of hydrogenated ZrO2 nanoparticles by water desorption. ACS Omega 2, 3878–3885 (2017).
Tada, S. et al. Active sites on ZnxZr1–xO2–x solid solution catalysts for CO2-to-methanol hydrogenation. ACS Catal. 12, 7748–7759 (2022).
Han, E., Fang, W., Stamatakis, M., Richardson, J. O. & Chen, J. Quantum tunnelling driven H2 formation on graphene. J. Phys. Chem. Lett. 13, 3173–3181 (2022).
Burch, R., Golunski, S. E. & Spencer, M. S. The role of copper and zinc oxide in methanol synthesis catalysts. J. Chem. Soc. Faraday Trans. 86, 2683–2691 (1990).
Sahibzada, M., Chadwick, D. & Metcalfe, I. S. Hydrogenation of carbon dioxide to methanol over palladium-promoted Cu/ZnO/Al2O3 catalysts. Catal. Today 29, 367–372 (1996).
Jung, K.-D. & Bell, A. T. Role of hydrogen spillover in methanol synthesis over Cu/ZrO2. J. Catal. 193, 207–223 (2000).
Meliancabrera, I. Pd-modified Cu-Zn catalysts for methanol synthesis from CO2/H2 mixtures: catalytic structures and performance. J. Catal. 210, 285–294 (2002).
Collins, S. E., Chiavassa, D. L., Bonivardi, A. L. & Baltanás, M. A. Hydrogen spillover in Ga2O3–Pd/SiO2 catalysts for methanol synthesis from CO2/H2. Catal. Lett. 103, 83–88 (2005).
Liang, X.-L., Dong, X., Lin, G.-D. & Zhang, H.-B. Carbon nanotube-supported Pd–ZnO catalyst for hydrogenation of CO2 to methanol. Appl. Catal. B Environ. 88, 315–322 (2009).
Hu, B. et al. Hydrogen spillover enabled active Cu sites for methanol synthesis from CO2 hydrogenation over Pd doped CuZn catalysts. J. Catal. 359, 17–26 (2018).
Im, J., Shin, H., Jang, H., Kim, H. & Choi, M. Maximizing the catalytic function of hydrogen spillover in platinum-encapsulated aluminosilicates with controlled nanostructures. Nat. Commun. 5, 3370 (2014).
Tang, C. et al. Insights into the selectivity determinant and rate-determining step of CO2 hydrogenation to methanol. J. Phys. Chem. C. 126, 10399–10407 (2022).
Zecevic, J., Vanbutsele, G., de Jong, K. P. & Martens, J. A. Nanoscale intimacy in bifunctional catalysts for selective conversion of hydrocarbons. Nature 528, 245–248 (2015).
Kasipandi, S. & Bae, J. W. Recent advances in direct synthesis of value-added aromatic chemicals from syngas by cascade reactions over bifunctional catalysts. Adv. Mater. 31, e1803390 (2019).
Zhou, W. et al. New horizon in C1 chemistry: breaking the selectivity limitation in transformation of syngas and hydrogenation of CO2 into hydrocarbon chemicals and fuels. Chem. Soc. Rev. 48, 3193–3228 (2019).
Ye, R. P. et al. CO2 hydrogenation to high-value products via heterogeneous catalysis. Nat. Commun. 10, 5698 (2019).
Pan, X., Jiao, F., Miao, D. & Bao, X. Oxide-zeolite-based composite catalyst concept that enables syngas chemistry beyond Fischer-Tropsch synthesis. Chem. Rev. 121, 6588–6609 (2021).
Wang, D., Xie, Z., Porosoff, M. D. & Chen, J. G. Recent advances in carbon dioxide hydrogenation to produce olefins and aromatics. Chem 7, 2277–2311 (2021).
Liu, S., Kots, P. A., Vance, B. C., Danielson, A. & Vlachos, D. G. Plastic waste to fuels by hydrocracking at mild conditions. Sci. Adv. 7, eabf8283 (2021).
Huang, X. et al. Au–Pd separation enhances bimetallic catalysis of alcohol oxidation. Nature 603, 271–275 (2022).
Cheng, K. et al. Maximizing noble metal utilization in solid catalysts by control of nanoparticle location. Science 377, 204–208 (2022).
Zachopoulos, A. & Heracleous, E. Overcoming the equilibrium barriers of CO2 hydrogenation to methanol via water sorption: a thermodynamic analysis. J. CO2 Util. 21, 360–367 (2017).
Leonzio, G., Zondervan, E. & Foscolo, P. U. Methanol production by CO2 hydrogenation: Analysis and simulation of reactor performance. Int. J. Hydrog. Energy 44, 7915–7933 (2019).
Perdew, J. P., Burke, K. & Ernzerhof, M. Generalized gradient approximation made simple. Phys. Rev. Lett. 77, 3865 (1996).
Kresse, G. & Hafner, J. Ab initio molecular dynamics for open-shell transition metals. Phys. Rev. B 48, 13115 (1993).
Kresse, G. & Furthmüller, J. Efficient iterative schemes for ab initio total-energy calculations using a plane-wave basis set. Phys. Rev. B 54, 11169 (1996).
Grimme, S., Antony, J., Ehrlich, S. & Krieg, H. A consistent and accurate ab initio parametrization of density functional dispersion correction (DFT-D) for the 94 elements H-Pu. J. Chem. Phys. 132, 154104 (2010).
Dudarev, S. L., Botton, G. A., Savrasov, S. Y., Humphreys, C. & Sutton, A. P. Electron-energy-loss spectra and the structural stability of nickel oxide: An LSDA+U study. Phys. Rev. B 57, 1505 (1998).
Bader, R. F. The zero-flux surface and the topological and quantum definitions of an atom in a molecule. Theor. Chem. Acc. 105, 276–283 (2001).
Henkelman, G., Arnaldsson, A. & Jónsson, H. A fast and robust algorithm for Bader decomposition of charge density. Comput. Mater. Sci. 36, 354–360 (2006).
Bahn, S. R. & Jacobsen, K. W. An object-oriented scripting interface to a legacy electronic structure code. Comput. Sci. Eng. 4, 56–66 (2002).
Larsen, A. H. et al. The atomic simulation environment—a Python library for working with atoms. J. Phys. Condens. Matter 29, 273002 (2017).
Henkelman, G. & Jónsson, H. Improved tangent estimate in the nudged elastic band method for finding minimum energy paths and saddle points. J. Chem. Phys. 113, 9978–9985 (2000).
Henkelman, G., Uberuaga, B. P. & Jónsson, H. A climbing image nudged elastic band method for finding saddle points and minimum energy paths. J. Chem. Phys. 113, 9901–9904 (2000).
Henkelman, G. & Jónsson, H. A dimer method for finding saddle points on high dimensional potential surfaces using only first derivatives. J. Chem. Phys. 111, 7010–7022 (1999).
Acknowledgements
This work was supported by the National University of Singapore Flagship Green Energy Program (A-0005323-11-00) and the National Research Foundation under the NRF Fellowship NRFF13-2021-0126 and NRF investigatorship NRF-NRFI07-2021-0006. Computational work was performed using resources of the National Supercomputing Centre, Singapore. T.-C.Y. and C.-M.Y. gratefully acknowledge the financial support by the Ministry of Science and Technology, Taiwan under contract No. MOST 109-2113-M-007-018-MY3 and MOST 111-2634-F-007-007 and by the Frontier Research Center on Fundamental and Applied Sciences of Matters from The Featured Areas Research Center Program within the framework of the Higher Education Sprout Project by the Ministry of Education (MOE) in Taiwan. Ms. Wang Zhan for the quasi in situ XPS experiment.
Author information
Authors and Affiliations
Contributions
K.L. and N.Y. conceived the project. N.Y. supervised the project and revised the manuscript. K.L. carried out most experiments, conducted data analysis, and drafted the initial manuscript. M.P.D. participated in catalyst synthesis and reaction testing. P.C.D.M., Y.S., and U.A. conducted DFT calculations and analyses, prepared the DFT draft, and revised the manuscript, P.C.D.M. also wrote the computer scripts, oversaw the methodology and draft for the DFT part, S.M.K. conceptualized, supervised, revised the draft and manuscript, and conducted data analysis for the DFT part. H.J. and M.C. conducted and analyzed H2/CO chemisorption, CO2-TPD, H2-TPD, and H2-D2 exchange experiments. L.C., K.L., and N.Y. conducted XPS experiments and analyzed data. T.-C.Y. and C.-M.Y. conducted and analyzed XAFS experiments. All authors participated in the discussions, commented, and edited the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks the anonymous reviewers for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Lee, K., Mendes, P.C.D., Jeon, H. et al. Engineering nanoscale H supply chain to accelerate methanol synthesis on ZnZrOx. Nat Commun 14, 819 (2023). https://doi.org/10.1038/s41467-023-36407-1
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-023-36407-1
This article is cited by
-
Economical biogas direct methanation to pipeline grade natural gas via structured Ni based inverse catalyst
Nature Communications (2026)
-
Advances in hydrogenation catalysis by porous materials supported palladium nanoparticles
Frontiers of Materials Science (2026)
-
Unlocking cathodic potential dependent Pd deactivation for energy efficient CO2 electroreduction to formate
Nature Communications (2025)
-
Electron and proton storage on separate Ru and BaO domains mediated by conductive low-work-function carbon to accelerate ammonia synthesis
Nature Catalysis (2025)
-
A dual-site Fe-based catalyst for efficient ammonia synthesis under mild conditions
Science China Chemistry (2025)