Abstract
Developing highly active materials that efficiently utilize solar spectra is crucial for photocatalysis, but still remains a challenge. Here, we report a new donor-acceptor (D-A) covalent organic framework (COF) with a wide absorption range from 200 nm to 900 nm (ultraviolet-visible-near infrared light). We find that the thiophene functional group is accurately introduced into the electron acceptor units of TpDPP-Py (TpDPP: 5,5’-(2,5-bis(2-ethylhexyl)−3,6-dioxo-2,3,5,6-tetrahydropyrrolo [3,4-c]pyrrole-1,4-diyl)bis(thiophene-2-carbaldehyde), Py: 1,3,6,8-tetrakis(4-aminophenyl)pyrene) COFs not only significantly extends its spectral absorption capacity but also endows them with two-photon and three-photon absorption effects, greatly enhancing the utilization rate of sunlight. The selective coupling of benzylamine as the target reactant is used to assess the photocatalytic activity of TpDPP-Py COFs, showing high photocatalytic conversion of 99% and selectivity of 98% in 20 min. Additionally, the TpDPP-Py COFs also exhibit the universality of photocatalytic selective coupling of other imine derivatives with ~100% conversion efficiency. Overall, this work brings a significant strategy for developing COFs with a wide absorption range to enhance photocatalytic activity.
Similar content being viewed by others
Introduction
The development of efficient photocatalysts is crucial for solar-driven photocatalysis benefit owing to its clean, eco-friendly, and renewable advantages1,2,3. Conventional inorganic photocatalysts, such as TiO24,5, ZnO6,7, and CdS8,9 encounter constraints in modulating their broad-band gaps, thereby limiting their capability to harness sunlight. Organic small-molecule photocatalyst, on the other hand, grapple with stability concerns, which complicates the processes of separation and recycling, impacting their overall sustainability10. Covalent organic frameworks (COFs), one of new crystalline porous materials, exhibiting enormous advantages, including high porosity11, extended π conjugation12,13, adjustable bandgaps14, and good stability15. Significantly, the Pt-doped COFs reported by Lotch’s group for continuous hydrogen production exhibit excellent structure and chemical stability, which has ignited extensive research on COFs-based photocatalysts16. Recently, COFs have been widely studied and applied in photocatalysts fields, including carbon dioxide reduction17,18, photocatalytic hydrogen production19,20, hydrogen peroxide synthesis21,22, and organic synthesis23,24. The noteworthy importance of imines as crucial intermediates in chemical synthesis, pharmaceuticals, and biology has prompted extensive investigations into the use of COFs for achieving efficient photocatalytic imine production25,26,27,28. In this pursuit, researchers have consistently focused on designing and synthesizing COFs with high photocatalytic active.
The common effective strategies for enhancing photocatalytic active are to promote the separation of photogenerated electron-hole, such as metal doping, heterojunctions, regulating band gap or the position of conduction and valence band29. Recently, the triazine-structured monomers30,31, porphyrins32,33, or phenolic functional group-containing monomers such as 2,4,6-triformyl resorcinol have been used to synthesize COFs for promoting the charge separation efficiency27,34. However, most COFs have a narrow absorption range typically between 200 nm and 550 nm, leading to reduced photocatalytic efficiency. To solve this bottleneck, developing novel strategies to expand the absorption range of COFs is key for enhancing their photocatalytic efficiency, but it still remains grand challenges.
Herein, we developed a novel strategy to precisely incorporate thiophene into the electron acceptor units of TpDPP-Py COFs (TpDPP: 5,5’-(2,5-bis(2-ethylhexyl)−3,6-dioxo-2,3,5,6-tetrahydropyrrolo [3,4-c]pyrrole-1,4-diyl)bis(thiophene-2-carbaldehyde), Py: 1,3,6,8-tetrakis(4-aminophenyl)pyrene), which extends its π-conjugated framework structure, significantly increasing the sunlight harvesting capabilities. In addition, the extended π-conjugated framework structure facilitates substantial interactions among non-adjacent chromophores without inducing aggregation, resulting in multi-photon absorption35. The fabricated TpDpp-Py COFs exhibit a wide absorption wavelength range of 200–900 nm and two/three-photon absorption effects, which enormous enhance the utilization rate of solar energy, thus exhibiting excellent performance of photocatalytic benzylamine (BA) coupling with conversion of 99% and selectivity of 98% in 20 min. In addition, the TpDPP-Py COFs also exhibit the universality of photocatalytic selective coupling of other imine derivatives with ~100% photocatalytic conversion efficiency. Overall, this work presents a significant strategy for the development of COFs with a broad absorption range to enhance photocatalytic activity.
Results and discussion
Design and synthesis of TpDPP-Py COFs molecular with D-A structure
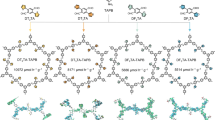
Diketopyrrolopyrrolopyrrole dialdehyde is widely recognized as a favorable candidate for the construction of donor‒acceptor (D‒A) structured COFs with enhanced light absorption performance owing to its well-established chromophore36. In the synthesis pathway, the thiophene functional groups with strong aromaticity and enhanced photocatalytic activity were selected as the side chain electron acceptor unit37,38,39 (Fig. 1a, Supplementary Fig. 1–3). For comparison, the DPP (4,4’-(2,5-bis(2-ethylhexyl)−3,6-dioxo-2,3,5,6-tetrahydropyrrolo[3,4-c]pyrrole-1,4 diyl)dibenzaldehyde) molecule with phenyl groups as the side chain was also synthesized (Fig. 1b, Supplementary Fig. 4, 5). Further, the Py (Py: 1,3,6,8-tetrakis(4-aminophenyl)pyrene) molecular with abundant conjugated electrons was chosen as the electronic donor unit40,41. A novel TpDPP-Py COFs with large π-conjugated system was synthesized by employing TpDPP as the electron acceptor and Py as the electron donor (Fig. 1c). The reference DPP-Py COFs were synthesized by employing DPP as the electron acceptor and Py as the electron donor (Fig. 1d).
a, b Synthetic route of TpDPP and DPP organic molecules. c, d Molecular structure of TpDPP-Py COFs and DPP-Py COFs.
Characterization of TpDPP-Py COFs
To confirm the chemical bonding properties, Fourier transform infrared (FT-IR) and Raman spectroscopy were carried out. The FT-IR spectroscopy showed the disappearance of the amino group of Py (νN-H = 3370‒3346 cm–1) and aldehyde group of TpDPP (νHC=O = 1693 cm–1), accompanied by the presence of a new characteristic peak located at 1571 cm–1 divided to C = N imine bonds, indicating the formation of TpDPP-Py COFs connected by imine bonds (Fig. 2a). Similarly, Raman spectroscopy displayed pronounced peaks at 1571 cm–1, consistent with the presence of imine bonds observed in the FT-IR analysis (Fig. 2b). X-ray Photoelectron Spectroscopy (XPS) data further confirmed the formation of imine bonds in TpDPP-Py COFs (Supplementary Fig. 6). Additionally, the solid-state 13C NMR (ss NMR) spectroscopy was performed to reveal the molecular framework structure of TpDPP-Py COFs, showing a chemical shift at 150 ppm corresponding to the imine bond (C = N) (Fig. 2c). In addition, the 15N ssNMR spectroscopy showed that the chemical shift located 162 ppm could be attributed to imine bond (Supplementary Fig. 7). These results provided additional evidence for the formation of COFs42,43,44,45,46,47. Overall, the above results provided powerful evidence for the formation of imine bonded TpDPP-Py COFs. Further, The FT-IR spectra and 13C ssNMR also demonstrated the formation of DPP-Py COFs (Supplementary Fig. 8).
a FT-IR spectra of TpDPP, Py and TpDPP-Py COFs. b Raman spectra of Py, TpDPP and TpDPP-Py COFs. c Solid-state 13C CP-MAS NMR spectrum of TpDPP-Py COFs. d Experimental and simulated XRD pattern of TpDPP-Py COFs (purple). e Simulated TpDPP-Py COFs molecular structure. f Temperature dependent XRD pattern of TpDPP-Py COFs.
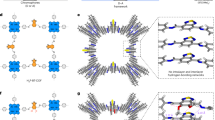
To further determine the ordered porous structure of TpDPP-Py COFs, N2 adsorption and desorption curves, X-ray diffraction (XRD) analysis and Cryogenic transmission electron microscopy (cryo-TEM) were conducted. N2 adsorption and desorption analysis revealed that the Brunauer-Emmett-Teller (BET) surface area and pore size of TpDPP-Py COFs were 408 m2 g–1 and 2.35 nm respectively, which was consistent with the theoretical simulations (Supplementary Fig. 9). The cell parameters of TpDPP-Py COFs and DPP-Py COFs are shown in Supplementary Tables 1, 2, respectively. The XRD pattern showed prominent peaks at 2.83°, 3.61°, 4.35° and 5.69°, corresponding to the crystal facets (110), (210), (120) and (310) respectively, with low-profile R-values of 5.2% and weighted-profile R-values of 6.9% (Fig. 2d, Supplementary Fig. 10). Compared to the A‒B stacking arrangement observed in TpDPP-Py COFs (Supplementary Fig. 11), the A‒A stacking arrangement aligns better with the experimental results (Fig. 2e). Additionally, temperature dependent XRD, Thermo-Gravimetric Analysis (TGA) and Differential Scanning Calorimetry (DSC) results demonstrate the excellent structural stability of TpDPP-Py COFs (Fig. 2f, Supplementary Fig. 12). Furthermore, SEM and TEM images were used to study the surface morphology of the TpDPP-Py COFs and DPP-Py COFs (Supplementary Fig. 13, Fig. 14). TEM images revealed the nanosheet morphology of TpDPP-Py COFs, displaying an average particle size of 650 nm (Supplementary Fig. 14). Additionally, cryo-TEM provided in-depth insights into the microstructural and lattice structure of the COF material (Fig. 3a). The crystal spacing of 3.11 nm was corresponded to the (110) crystal plane (Fig. 3b). The Fast Fourier Transform (FFT) patterns also exhibited a tetragonal structure, which was a match to the simulated molecular structure (Fig. 3c). Elemental mapping showed the uniform distribution of sulfur (S), oxygen (O), nitrogen (N), and carbon (C) on TpDPP-Py COFs (Fig. 3d). Thus, the above results confirmed the successful synthesis of TpDPP-Py COFs with ordered porous structure.
a Cryo-TEM images of TpDPP-Py COFs (scale bar, 50 nm). b High resolution Cryo-TEM of the TpDPP-Py COFs (scale bar, 20 nm). c FFT patterns of TpDPP-Py COFs. d HAADF-STEM image and the corresponding STEM-EDS elemental mapping images of TpDPP-Py COFs (scale bar, 100 nm) (d1, merge; d2, S; d3, O; d4, N; d5, C).
Optical characteristics and DFT theoretical calculations of TpDPP-Py COFs
The optical band gap of TpDPP-Py COFs was calculated as 1.38 eV through the equation Eg (eV) = 1240 / λ (nm) (Fig. 4a). The UV-vis spectrum showed that TpDPP-Py COFs exhibited broad absorption peaks range from 200 to 900 nm, indicating a narrower optical bandgap and higher solar energy utilization efficiency compared to DPP-Py COFs (Fig. 4b, Supplementary Fig. 15). The photoluminescence fluorescence spectrum exhibited that TpDPP-Py COFs could be excited within the wavelength range from 300 nm to 700 nm, with an emission peak observed at 810 nm, signifying favorable absorption properties in the visible and near-infrared regions (Fig. 4c, Supplementary Fig. 16). The fluorescence decay of TpDPP-Py COFs exhibited a dual exponential decay model, with fluorescence decay times of 0.0595 ns and 0.299 ns (Fig. 4d, Supplementary Fig. 17), which were shorter than DPP-Py COFs (0.168 ns and 0.862 ns) (Supplementary Fig. 18), indicating more efficient charge separation efficiency and transport dynamics48,49.
a Kubelka-Munk function analysis of UV/Vis absorption spectra for TpDPP-Py COFs, TpDPP, and Py precursor materials, along with diffuse reflection spectra. b Kubelka-Munk Function UV/Vis Absorption Spectra of TpDPP-Py COFs and DPP-Py COFs. c Solid-state fluorescence spectra of TpDPP-Py COFs under different excitation wavelengths. d Time-resolved photoluminescence spectra of TpDPP-Py COFs (λex = 1200 nm). e Molecular orbital amplitude plots of HOMO and LUMO of the ligand and p-extended ligand calculated at the ωb97xd/6-31 G* basis set. f Two-photon induced solid-state fluorescence emission of TpDPP-Py COFs at different units (λex = 1200 nm). g three-photon induced solid-state fluorescence emission of TpDPP-Py COFs at different units (λex = 2100 nm). h transient photocurrent responses of the TpDPP-Py COFs and DPP-Py COFs.
The HOMO and LUMO energy levels of TpDPP Py COFs were calculated to be −4.67 eV and −3.39 eV, respectively (Fig. 4e), yielding a theoretical band gap of 1.28 eV. Moreover, compared with DPP-Py COFs, TpDPP-Py COFs exhibited excellent two-photon and three-photon absorption characteristics (Fig. 4f, g, Supplementary Fig. 19), implying more effectively utilization of sunlight. Figure 4h shows the photocurrent response curves of TpDPP-Py COFs and DPP-Py COFs. The TpDPP-Py COFs exhibited a photocurrent density of 0.054 μA cm‒2, which was significantly higher than that of DPP-Py COFs (0.035 μA cm‒2). The high photocurrent density of TpDPP-Py COFs mainly benefits from its excellent light absorption characteristics.
Photocatalytic benzylamine (BA) coupling and reaction mechanism
To assess the photocatalytic properties, TpDPP-Py COFs were used for the selective photocatalytic coupling of benzylamine (BA) to N-benzylidenebenzylamine. The gas chromatogram (GC) was carried out to detect the conversion rate of N-benzylidenebenzylamine (Supplementary Fig. 20, 21). Figure 5a shows the activity and selectivity of converting BA to N-benzylidenebenzylamine over TpDPP-Py COFs. The TpDPP-Py COFs exhibit a high photocatalytic coupling conversion of BA of 99% and high selectivity of 98% in 20 min, which is significantly better than DPP-Py COFs (Fig. 5b). Meanwhile, the performance of TpDPP-Py COFs photocatalytic conversion of BA to N-benzylidenebenzylamine is also better than most reported photocatalysts (Fig. 5c, Supplementary Table 3). Figure 5d shows the activity and selectivity of different batches TpDPP-Py COFs for photocatalytic conversion of BA to N-benzylidenebenzylamine. No obvious attenuation indicates the excellent performance stability of TpDPP-Py COFs. Furthermore, XRD and FT-IR spectra confirm that the crystal structure of TpDPP-Py COFs does not undergo significant changes after the photocatalytic reaction (Supplementary Fig. 22), validating its excellent structural stability.
a Activity and selectivity of TPDPP-Py COFs photocatalytic conversion of BA to N-benzylidenebenzylamine. b Activity and selectivity of DPP-Py COFs photocatalytic conversion of BA to N-benzylidenebenzylamine. c performance comparison of TpDPP-Py COFs with reported photocatalyst. d Repeatability measurement of TpDPP-Py COFs photocatalytic conversion of BA to N-benzylidenebenzylamine. e EPR detection of the 1O2 trapped by TEMP over TpDPP-Py COFs under dark and light irradiation. f Comparison of EPR intensity of 1O2 between TpDPP-Py COFs and DPP-Py COFs. g EPR detection of the ∙O2– anion radical trapped by DMPO over TpDPP-Py COFs under dark and light irradiation. h Comparison of EPR intensity of ∙O2–anion radical between TpDPP-Py COFs and DPP-Py COFs. i EPR detection of the hole over TpDPP-Py COFs with and without BA.
Understanding the reaction mechanism was very necessary for both fundamental research and application research. We found that BA could not be converted to N-benzylidenebenzylamine without photocatalyst or dark conditions (Supplementary Table 4), implying the necessary of photocatalyst and light irradiation for the selective photocatalytic coupling of BA. Besides, oxygen also played an important role in the selective photocatalytic coupling of BA (Supplementary Table 4). Thus, another important problem to be solved is how oxygen plays a role in selective photocatalysis coupling of BA. Electron paramagnetic resonance (EPR) was carried out to study the active intermediates of O2. Figure 5e shows that the EPR detection of the 1O2 can be detected under light irradiation, while it disappears under dark condition, confirming that the triplet state oxygen is converted into singlet oxygen under light irradiation. The 1O2 intensity over TpDPP-Py COFs is higher than that of DPP-Py COFs (Fig. 5f and Supplementary Fig. 23), implying its higher efficiency of oxygen conversion, which is mainly benefited from the high utilization of sunlight of TpDPP-Py COFs. Further, Fig. 5g shows the EPR signal of the ∙O2– anion radical over TpDPP-Py COFs. No obvious signal can be detected under dark condition, while EPR signal of ∙O2– anion radical is obtained under light irradiation, confirming that 1O2 can capture the photogenerated electrons to form ∙O2– anion radical. The EPR intensity of ∙O2– anion radical over TpDPP-Py COFs is higher than that of DPP-Py COFs (Fig. 5h and Supplementary Fig. 24), indicating that more 1O2 are converted into ∙O2– anion radical on TpDPP-Py COFs. In addition, the photogenerated hole intensity of TpDPP-Py COFs with the presence of BA is significantly lower than that without BA (Fig. 5i), indicating that BA consumes photogenerated holes in the system.
Based on the above discussion, the mechanism of selective photocatalytic coupling of BA to generate N-benzylidenebenzylamine can be explained (Supplementary Fig. 25). Firstly, TpDPP-Py COFs are excited to produce photogenerated electron-hole pairs. And then, the 1O2 captures photogenerated electrons and is reduced to ∙O2– anion radical. Meanwhile, the α-carbon of BA is activated by the photogenerated hole. Further, the ∙O2– anion radical is used to react with activated BA molecule to generate benzaldehyde intermediate. Finally, the unstable benzaldehyde intermediates rapidly react with BA to synthesize N-benzylidenebenzylamine (Supplementary Fig. 25).
Study on the universality of photocatalytic amine coupling
The TpDPP-Py COFs photocatalyst was also used to explore selective photocatalytic coupling of amine derivatives (2-ethoxyaniline, 3-ethoxyaniline, 4-ethoxyaniline, 2-thiophene methylamine, 4-fluoroaniline, 4-chlorobenzylamine) under the visible light irradiation. As shown in Fig. 6, with an increase in reaction time, the conversion of all amine derivatives increases and then stabilizes, indicating complete conversion of the amine derivatives and confirming the universality of TpDPP-Py COFs for photocatalytic amine coupling. Furthermore, the yield of all amine conversion to corresponding coupling products is higher than 97%, and the conversion efficiency of 2-thiophene methylamine approaching 100% within 1 h (Fig. 6c), which indicates the high photocatalytic activity of TpDPP-Py COFs. The excellent photocatalytic performance is benefited from the wide absorption properties and multi-photon absorption effect.
a 2-ethoxyaniline, b 3-ethoxyaniline, c 4-ethoxyaniline, d 2-thiophene methylamine, e 4-fluoroaniline, and f 4-Chlorobenzylamine.
In summary, we present a novel TpDPP-Py COFs for the first time by integrating thiophene into DPP organic ligands, resulting in a broad absorption range spanning from 200 nm to 900 nm (ultraviolet-visible-near infrared light). Our study illustrates that the extended π-conjugated framework structure not only enhances the absorption capacity but also confers multi-photon absorption effects, significantly improving sunlight utilization and photocatalytic activity. Utilizing the TpDPP-Py COFs for the selective photocatalytic coupling of benzylamine (BA) resulted in a high BA coupling conversion rate of 99% and a selectivity of 98% within a 20-min timeframe, surpassing that of most reported active materials. Furthermore, the TpDPP-Py COFs demonstrate a general applicability for the photocatalytic coupling of various amine derivatives. This work introduces a new strategy for the development of innovative COFs photocatalysts, which is of great significance for promoting the practical application of COFs in the field of photocatalysis.
Methods
Synthesis of TpDPP-Py COFs
Synthesis of TpDPP-Py-COFs: 5,5’-(2,5-bis(2-ethylhexyl)−3,6-dioxo-2,3,5,6-tetrahydropyrrolo [3,4-c]pyrrole-1,4-diyl)bis(thiophene-2-carbaldehyde) (TpDpp) (12 mg, 0.02 mmol), and 1,3,6,8-tetrakis(4-aminophenyl)pyrene (Py) (6 mg, 0.01 mmol) were weighed into a 10 mL glass tube. Afterwards, o-DCB: mesitylene = 4:1 = 1.0 mL was added and the mixture was sonicated for 5 min. After addition of 0.1 mL AcOH (6 M, aqueous), the tube was degassed by the three freeze-pump-thaw cycles and sealed with flame. Upon warming to room temperature, the sealed tube was heated at 120 °C for 72 h. The formed precipitate was collected by filtration, washed with methanol and tetrahydrofuran (THF) and further activated by Soxhlet extraction using methanol and THF for 72 h. Finally, the solid was collected and dried at 120 °C for 12 h under vacuum to give TpDPP-Py COFs as the dark purple powder (15 mg, 60% yield).
Synthesis of DPP-Py COFs
Synthesis of DPP-Py COFs: 4,4’-(2,5-bis(2-ethylhexyl)−3,6-dioxo-2,3,5,6-tetrahydropyrrolo[3,4-c]pyrrole-1,4-diyl)dibenzaldehyde (DPP) (12 mg, 0.02 mmol), and1,3,6,8-tetrakis(4-aminophenyl)pyrene (Py) (6 mg, 0.01 mmol) were weighed into a 10 mL glass tube. Afterwards, 1.4-dioxane: mesitylene = 1:1 = 1.0 mL was added and the mixture was sonicated for 5 min. After addition of 0.1 mL AcOH (6 M, aqueous), the tube was degassed by the three freeze-pump-thaw cycles and sealed with flame. Upon warming to room temperature, the sealed tube was heated at 120 °C for 72 h. The formed precipitate was collected by filtration, washed with methanol and tetrahydrofuran (THF) and further activated by Soxhlet extraction using methanol and THF for 72 h. Finally, the solid was collected and dried at 120 °C for 12 h under vacuum to give DPP-Py COFs as shiny red powder (10 mg, 50% yield). The synthesis route of TpDPP and DPP is in Supplementary Fig. S1–5.
Selective photocatalysis coupling of benzylamine (BA)
5 mg photocatalyst was added into acetonitrile solution containing 1% benzylamine in the Pyrex vessel reactor. the mixed solution was bubbled with oxygen for 30 min before light irradiation. And then, the suspension solution was irradiated by a 300 W Xe lamp (Microsolar300, Beijing Perfectlight) equipped with AM1.5 G solar intensity. The gas chromatography (GC-7890B) with a flame ionization detector (FID) detector was carried out to detect the conversion rate of N-benzylidenebenzylamine. The photocatalytic coupling of amine derivatives (2-ethoxyaniline, 3-ethoxyaniline, 4-ethoxyaniline, 2-thiophene methylamine, 4-fluoroaniline, 4-chlorobenzylamine) is similar to that of BA.
Characterization
Powder X-ray diffraction (PXRD) patterns were recorded on PANalytical Empyrean diffractometer for Cu/Kα radiation (λ = 1.5416 nm) The samples were spread on the square recess of XRD sample holder as a thin layer. Variable temperature PXRD was recorded at a rate of 10 °C min‒1 and held for 2 min. Themis 300 (cryo-TEM) at an accelerating voltage of 200 KV. Imaging was conducted using low-dose techniques and the dose rate was <5.0 e/Å2/s. Disperse 2 mg of COF into 1 ml of ethanol and add dropwise in the microgrid. Nanosheets were observed at JEM-2100 (200KV). Scanning Electron Microscopy (SEM) images were recorded using a Zeiss Gemini 300 scanning electron microscopy. Infrared spectra were measured on Lambda 1050+ under vacuum. All spectra were background corrected. Thermogravimetric analysis (TGA) measurements were carried out on a PerkinElmer series 7 thermal analysis system under N2 at a heating rate of 10 °C min−1. 13C cross polarization magic angle spinning nuclear magnetic resonance (13C CP/MAS NMR) spectra was recorded on a Bruker NEO 600 WB spectrometer. Samples were packed in 4 mm ZrO2 rotors, which were spun at 8 kHz in a double resonance MAS probe. All spectra were background corrected. Absorption spectra were recorded by PerkinElmer Lambda 1050 + UV-vis-NIR. UV-vis spectrophotometer at room temperature. The absorption spectra of solid powder employed integrating sphere mode with Ba2SO4 background. Fluorescence spectra of COF powders was using fluorescence spectrometer FLS1000, and the transient photoluminescence decay lifetime profiles of COF powder was using Astrella.
Data availability
The authors declare that the experimental data supporting the results of this study can be found in the paper and its Supplementary Information file. The experimental results of the study along with other simulation data are provided in the Supplementary Information file. The detailed simulation files for the study are available from the corresponding author upon request. Source data are provided with this paper.
References
Wang, E. et al. Solar-driven photocatalytic reforming of lignocellulose into H2 and value-added biochemicals. ACS Catal. 12, 11206–11215 (2022).
Wang, Y. et al. Current understanding and challenges of solar-driven hydrogen generation using polymeric photocatalysts. Nat. Energy 4, 746–760 (2019).
Wang, Z. et al. Recent developments in heterogeneous photocatalysts for solar-driven overall water splitting. Chem. Soc. Rev. 48, 2109–2125 (2019).
Belkessa, N. et al. Gaseous ethylbenzene removal by photocatalytic TiO2 nanoparticles immobilized on glass fiber tissue under real conditions: evaluation of reactive oxygen species contribution to the photocatalytic process. Environ. Sci. Pollut. Res. Int. 30, 35745–35756 (2023).
Jia Z.-M., et al. Adsorption kinetics of the photocatalytic reaction of nano-TiO2 cement-based materials: A review. Constr. Build. Mater. 370, 130462 (2023).
Dhull, P. et al. An overview on ZnO-based sonophotocatalytic mitigation of aqueous phase pollutants. Chemosphere 333, 138873 (2023).
Li, Z. et al. Blocking the reverse reactions of overall water splitting on a Rh/GaN–ZnO photocatalyst modified with Al2O3. Nat. Catal. 6, 80–88 (2023).
Dong, F. et al. A novel pathway toward efficient improvement of the photocatalytic activity and stability of CdS-based photocatalyst for light driven H2 evolution: The synergistic effect between CdS and SrWO4. Int. J. Hydrog. Energy 48, 13877–13889 (2023).
Xu, Z. et al. Rational synthesis of Au-CdS composite photocatalysts for broad-spectrum photocatalytic hydrogen evolution. ACS Nano 17, 11655–11664 (2023).
Prier, C. K. et al. Visible light photoredox catalysis with transition metal complexes: Applications in organic synthesis. Chem. Rev. 113, 5322–5363 (2013).
Glaser, F. et al. Multi-photon excitation in photoredox catalysis: concepts, applications, methods. Angew. Chem. Int. Ed. 59, 10266–10284 (2020).
Deng, M. et al. Extending the π-conjugation system of covalent organic frameworks for more efficient photocatalytic H2O2 production. Green. Chem. 25, 3069–3076 (2023).
Fan, Y. et al. Enhanced energy transfer in a π-conjugated covalent organic framework facilitates excited-state nickel catalysis. Angew. Chem. Int. Ed. 62, e202218908 (2023).
Li, Z. et al. Three-dimensional covalent organic framework with ceq topology. J. Am. Chem. Soc. 143, 92–96 (2021).
Lyu, H. et al. Porous crystalline olefin-linked covalent organic frameworks. J. Am. Chem. Soc. 141, 6848–6852 (2019).
Stegbauer, L. et al. A hydrazone-based covalent organic framework for photocatalytic hydrogen production. Chem. Sci. 5, 2789–2793 (2014).
Gao, S. et al. Ingenious artificial leaf based on covalent organic framework membranes for boosting CO2 photoreduction. J. Am. Chem. Soc. 145, 9520–9529 (2023).
Zhang, Y. et al. Engineering single Cu sites into covalent organic framework for selective photocatalytic CO2 reduction. Small 19, e2300035 (2023).
Li, Z. et al. Three-component donor-π-acceptor covalent-organic frameworks for boosting photocatalytic hydrogen evolution. J. Am. Chem. Soc. 145, 8364–8374 (2023).
Liu, Y. et al. Cu2O/2D COFs core/shell nanocubes with antiphotocorrosion ability for efficient photocatalytic hydrogen evolution. ACS Nano 17, 5994–6001 (2023).
Luo, Y. et al. Sulfone-modified covalent organic frameworks enabling efficient photocatalytic hydrogen peroxide generation via one-Step two-electron O2 Reduction. Angew. Chem. Int. Ed. 62, e202305355 (2023).
Chen, D. et al. Covalent organic frameworks containing dual O2 reduction centers for overall photosynthetic hydrogen peroxide production. Angew. Chem. Int. Ed. 62, e202217479 (2023).
Liu, Y. et al. A universal room‐temperature approach to large‐area continuous COFs film for photocatalytic coupling of amines. Adv. Funct. Mater. 33, 2302874 (2023).
Liu, X. et al. Triazine-porphyrin-based hyperconjugated covalent organic framework for high-performance photocatalysis. J. Am. Chem. Soc. 144, 23396–23404 (2022).
Kumar, N. et al. Selective aerobic coupling of amines to imines using solar spectrum-responsive flower-like Nen-graphene quantum dots (GQDs) decorated with 2,4-dinitrophenylhydrazine (PH) as a photocatalyst. Chemosphere 341, 139697 (2023).
Liu, H. et al. Photocatalytic hydrogen production coupled with selective benzylamine oxidation over MOF composites. Angew. Chem. Int. Ed. 57, 5379–5383 (2018).
Yang, S. et al. Modulating the stacking model of covalent organic framework isomers with different generation efficiencies of reactive oxygen species. ACS Appl. Mater. Interfaces 13, 29471–29481 (2021).
Xiong, K. et al. Linker length-dependent photocatalytic activity of β-ketoenamine covalent organic frameworks. Appl. Catal. B Environ. 322, 122135 (2023).
Wang, H. et al. Covalent organic framework photocatalysts: structures and applications. Chem. Soc. Rev. 49, 4135–4165 (2020).
Qi, W. et al. Fluorinated covalent organic framework materials for photocatalytically driven benzylamine coupling and azo dyes degradation. J. Photochem. Photobiol. A Chem. 437, 114502 (2023).
Li, Q. et al. Structural and morphological engineering of benzothiadiazole-based covalent organic frameworks for visible light-driven oxidative coupling of amines. ACS Appl. Mater. Interfaces 13, 39291–39303 (2021).
Wu, S. et al. Hydrazone-linked 2D porphyrinic covalent organic framework photocatalysis for visible light-driven aerobic oxidation of amines to imines. J. Colloid Interface Sci. 610, 446–454 (2022).
Shi, J. L. et al. 2D sp2 Carbon-conjugated porphyrin covalent organic framework for cooperative photocatalysis with TEMPO. Angew. Chem. Int. Ed. 59, 9088–9093 (2020).
Wu, Z. et al. Covalent-organic frameworks with keto-enol tautomerism for efficient photocatalytic oxidative coupling of amines to imines under visible light. Sci. Chi. Chem. 64, 2169–2179 (2021).
Nath, S. et al. Covalent organic frameworks as emerging nonlinear optical materials. Angew. Chem. Int. Ed. 62, e202218974 (2023).
Jiang, W. et al. New synthetic approaches to N-aryl and π-expanded diketopyrrolopyrroles as new building blocks for organic optoelectronic materials. Angew. Chem. Int. Ed. 60, 10700–10708 (2021).
Yue, J. Y. et al. Thiophene-containing covalent organic frameworks for overall photocatalytic H2O2 synthesis in water and seawater. Angew. Chem. Int. Ed. 62, e202309624 (2023).
Shen, L. et al. Effects of aromatic substituents on the electronic structure and excited state energy levels of diketopyrrolopyrrole derivatives for singlet fission. Phys. Chem. Chem. Phys. 20, 22997 (2018).
Leenaers, P. J. et al. Impact of π‑conjugated linkers on the effective exciton binding energy of diketopyrrolopyrrole−dithienopyrrole copolymers. J. Phys. Chem. C. 124, 27403–27412 (2020).
Ascherl, L. et al. Solvatochromic covalent organic frameworks. Nat. Commun. 9, 3802 (2018).
Auras, F. et al. Synchronized offset stacking: a concept for growing large-domain and highly crystalline 2D covalent organic frameworks. J. Am. Chem. Soc. 138, 16703–16710 (2016).
Xu, J. et al. A Highly conductive COF@CNT electrocatalyst boosting polysulfide conversion for Li–S chemistry. ACS Energy Lett. 6, 3053–3062 (2021).
Gole, B. et al. Microtubular self-assembly of covalent organic frameworks. Angew. Chem. Int. Ed. 57, 846–850 (2018).
Mohata, S. et al. Selective metal-free CO2 photoreduction in water using porous nanostructures with internal molecular free volume. J. Am. Chem. Soc. 145, 23802–23813 (2023).
Majumder, P. et al. Proximity-enabled photochemical C−H functionalization using a covalent organic framework-confined Fe2IV−μ−oxo species in water. J. Am. Chem. Soc. 145, 18855–18864 (2023).
Bag, S. et al. Covalent organic framework thin-film photodetectors from solution-processable porous nanospheres. J. Am. Chem. Soc. 145, 1649–1659 (2023).
Koner, K. et al. Bottom-up synthesis of crystalline covalent organic framework nanosheets, nanotubes, and kippah vesicles: an odd−even effect induction. J. Am. Chem. Soc. 145, 14475–14483 (2023).
Tan, H. et al. Photocatalysis of water into hydrogen peroxide over an atomic Ga-N5 site. Nat. Synth. 2, 557–563 (2023).
Wei, P.-F. et al. Benzoxazole-linked ultrastable covalent organic frameworks for photocatalysis. J. Am. Chem. Soc. 140, 4623–4631 (2018).
Acknowledgements
This work was financially supported by the Strategic Priority Research Program of CAS (XDB0520101), the National Natural Science Foundation of China (U22A6002, 22173109, 52303363), the National Key R&D Program of China (2018YFA0703200), the CAS Project for Young Scientists in Basic Research (YSBR-053), the CAS-Croucher Scheme for Joint Laboratories, Lu Jiaxi international team (GJTD-2020-02), the CAS Cooperation Project (121111KYSB20200036), and the Beijing Nova Program (20220484173). The authors gratefully acknowledge the assistance of Ningning Wu in data collection for nuclear magnetic analysis. We also extend our appreciation to Jiling Yue and Kaiang Liu for their valuable contributions in characterizing the cryo-TEM experiments. Additionally, we would like to thank Meirong Liu for her help in operating the femtosecond laser spectrometer.
Author information
Authors and Affiliations
Contributions
Y.F. and Yo.L contributed equally to this work. Y.G., Q.T. and Yu.L proposed and supervised the project. Y.F., Yo.L conceived the idea and designed the experiments. H.H. conducted DFT calculations. J.S. conducted Raman spectroscopy characterization. J.H. conducted transient photocurrent response. W.G. and M.S. assisted in the synthesis of materials. Q.T., F.Z., X.W. and Y.G. assisted in the writing of the article. X.W. assisted in the spectral experiment. Y.F. and Yo.L wrote the manuscript and all authors reviewed it.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Rahul Banerjee, Zhiguo Zhang and Xianjun Lang for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Fang, Y., Liu, Y., Huang, H. et al. Design and synthesis of broadband absorption covalent organic framework for efficient artificial photocatalytic amine coupling. Nat Commun 15, 4856 (2024). https://doi.org/10.1038/s41467-024-49036-z
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-024-49036-z
This article is cited by
-
Diketopyrrolopyrrole-based two-dimensional poly(arylene vinylene)s with high charge carrier mobility
Nature Communications (2026)
-
Exciton tuning and charge steering in donor-acceptor covalent triazine frameworks toward boosted photocatalytic oxidation
Science China Materials (2026)
-
Nitrogen-atom skeletal editing of β-ketoenamine covalent organic frameworks enables photogenerated charge transfer via TEMPO
Science China Chemistry (2026)
-
Straightforward construction of functionalized γ-lactams via conjugated-engineered covalent organic framework photocatalysed cascade reactions
Nature Communications (2025)
-
Regulating the electronic structure of covalent organic frameworks via heterocyclic isomers for highly efficient photocatalytic H2O2 generation
Nature Communications (2025)