Abstract
Internal combustion engine fueled by carbon-free hydrogen (H2-ICE) offers a promising alternative for sustainable transportation. Herein, we report a facile and universal strategy through the physical mixing of Pt catalyst with zeolites to significantly improve the catalytic performance in the selective catalytic reduction of nitrogen oxides (NOx) with H2 (H2-SCR), a process aiming at NOx removal from H2-ICE. Via the physical mixing of Pt/TiO2 with Y zeolite (Pt/TiO2 + Y), a remarkable enhancement of NOx reduction activity and N2 selectivity was simultaneously achieved. The incorporation of Y zeolite effectively captured the in-situ generated water, fostering a water-rich environment surrounding the Pt active sites. This environment weakened the NO adsorption while concurrently promoting the H2 activation, leading to the strikingly elevated H2-SCR activity and N2 selectivity on Pt/TiO2 + Y catalyst. This study provides a unique, easy and sustainable physical mixing approach to achieve proficient heterogeneous catalysis for environmental applications.
Similar content being viewed by others
Introduction
The transportation sector has a considerable impact on global climate change1, being responsible for nearly 24% of the world’s CO2 emissions stemming from fossil fuel combustion2. Consequently, it is imperative to prioritize substantial CO2 reduction within this sector. While electric powertrains powered by renewable energy hold promise, their environmental cost and limited energy capacity for heavy-duty vehicles pose significant challenges for widespread application3. There is another viable avenue lies in the adoption of internal combustion engines (ICE) operating on carbon-free hydrogen (H2), which presents a promising alternative for sustainable transportation3,4. During the H2 combustion process, nitrogen oxides (NOx) are the primary environmental pollutants5,6. Selective catalytic reduction (SCR) of NOx is one of the most efficient and widely used technologies for NOx abatement in excess oxygen6,7. For H2-ICE applications, H2 extracted from the fuel tank can serve directly as a reducing agent for the SCR of NOx (H2-SCR)8. This approach may offer significant economic and environmental benefits. However, to make this technique viable, the key issue to be solved is the development of robust H2-SCR catalyst systems, which can demonstrate excellent low-temperature NOx reduction activity and N2 selectivity simultaneously.
Supported platinum (Pt) and palladium (Pd) catalysts have been extensively investigated for H2-SCR reaction6,9,10. Notably, Pt catalysts have shown great promise with their superior low-temperature (<150 oC) activity comparing to Pd catalysts11,12, although there is urgent need for significant improvement in N2 selectivity13. H2 activation was considered as one of the most critical factors on Pt catalysts that could profoundly influence the H2-SCR performance14. Improving H2 activation and sustaining abundant *H species on Pt catalysts could positively promote the NO dissociation15,16,17, which has been reported as the rate-determining step for the H2-SCR reaction18,19. Additionally, this enhancement could also facilitate the formation of NHx species, which, in some cases, have been found beneficial for the H2-SCR reaction14,20,21,22,23. Currently, substantial efforts have been dedicated towards increasing the presence of metallic Pt species24,25, as it plays a crucial role in H2 activation. Studies have reported that specific additives could substantially enhance the NOx reduction activity by reducing the Pt valence. For instance, the addition of Mo and Na to Pt/SiO226 and the introduction of Ti species into Pt/MCM-4113 resulted in the lowered Pt valence state, leading to widened temperature window for NOx conversion. Additionally, the acidity or basicity of supports also strongly influenced the dispersion and chemical state of Pt27,28, with acidic supports being beneficial for the formation of metallic Pt species therefore promoting the H2-SCR performance27,29. Such strategies involving the chemical modification of Pt catalysts to form more metallic Pt species were mainly intent to enhance the H2 activation. However, it was observed that the presence of metallic Pt species usually favored the NO adsorption over H2 adsorption, inevitably resulting in a reduced *H coverage during H2-SCR reaction30. Moreover, these chemical modification strategies were found to be effective only for specific Pt catalyst systems, and in most cases the enhancement was only restricted to NOx reduction activity but not to N2 selectivity. Therefore, there is urgent need to design a simple, effective and universal strategy to boost the H2 activation while reducing the NO adsorption on Pt active sites, thus improving the low-temperature activity and N2 selectivity in the H2-SCR reaction on Pt-based catalysts accordingly.
Different from the sophisticated chemical modification strategies as previously reported, in this work, we successfully developed a simple, sustainable physical mixing strategy of oxide-supported Pt catalysts (e.g., Pt/TiO2, Pt/Al2O3, or Pt/SiO2) with various zeolites (e.g., H-Y, H-ZSM-5, H-chabazite (CHA), H-ferrierite (FER), or H-Beta) to significantly promote the H2-SCR reaction. Using this facile approach that is easy to scale up in industry, a universal increase in both the H2-SCR activity and N2 selectivity was achieved. Focusing on a typical physically mixed catalyst system involving the extensively studied Pt/TiO231,32 and commercial H-Y zeolite (i.e., Pt/TiO2 + Y), in-depth mechanistic studies were performed through the combined experimental and theoretical approaches. It was clearly revealed that the introduction of Y zeolite facilitated the formation of water-enriched micro-environment on Pt/TiO2, which played a crucial role in mitigating the over-strong adsorption of NO while promoting the H2 activation on Pt sites. As a result, the disassociation of NO, a crucial step in the H2-SCR reaction, was substantially promoted, leading to the drastic enhancement in the catalytic performance.
Results
Physical mixing of Pt catalysts and zeolites to promote the H2-SCR reaction
The Pt/TiO2 catalyst was prepared via a conventional incipient wetness impregnation (IWI) method using colloidal Pt precursor and a commercial TiO2 support. In the H2-SCR reaction under typical given condition, the Pt/TiO2 catalyst showed NOx conversion above 11% (Fig. 1a) and N2 selectivity above 17% (Fig. 1b) below 250 °C. When physically mixing the Pt/TiO2 catalyst with an inactive commercial H-Y zeolite (SiO2/Al2O3 molar ratio = 30) (Fig. 1a, b), within the investigated temperature range, the Pt/TiO2 + Y catalyst system showed substantially improved catalytic performance, with NOx conversion above 59% and N2 selectivity above 58% below 250 °C. In addition, this Pt/TiO2 + Y catalyst showed much higher reaction rates and N₂ selectivity at 100 and 200 °C compared to most reported Pt and Pd catalysts (Supplementary Table 1). Such a broad operation temperature window (100–250 °C) and excellent catalytic performance from Pt/TiO2 + Y system are highly desirable for the practical H2-SCR application33. In the presence of both H2 and O2, NO can either be reduced by H2 to form N2/N2O or be oxidized by O2 to form NO2 (Supplementary Fig. 1). Therefore, it is reasonable that the NOx conversion and N2 selectivity could hardly achieve 100% under the high space velocity H2-SCR testing conditions with H2O and CO2 (500 ppm NO, 1% H2, 10% O2, 5% CO2, and 5% H2O; WHSV = 461,540 mL·gPt/TiO2–1·h–1). Comparing to Pt/TiO2, the Pt/TiO2 + Y system consistently showed higher selectivity towards NO reduction and lower selectivity towards NO oxidation during the H2-SCR reaction (Fig. 1c), particularly at high temperatures. The results clearly demonstrated that the presence of Y significantly promoted the NO reduction by H2 on Pt/TiO2 + Y system.
a NOx conversion, and (b) N2 selectivity in H2-SCR reaction; c NO selective conversion (i.e., NO reacting with H2 or O2) in H2-SCR reaction over Pt/TiO2 and Pt/TiO2 + Y catalysts (see Methods section for detailed calculation); d Correlation between N2 selectivity and NOx conversion in H2-SCR reaction over Pt/TiO2, Pt/TiO2 + TiO2, and Pt/TiO2 + Y catalysts. Reaction conditions: 26 mg of Pt/TiO2 catalyst, or a physical mixture containing 26 mg of Pt/TiO2 and 26 mg of Y or TiO2; steady-state testing; 500 ppm NO, 1% H2, 10% O2, 5% CO2, and 5% H2O; weight hourly space velocity (WHSV) = 461,540 mL·gPt/TiO2–1·h–1.
To verify if there was synergy effect and how it worked between Pt/TiO2 and Y components, we investigated the different physical mixing methods (Supplementary Fig. 2a) and see how the H2-SCR performance was impacted. It was demonstrated that, in clear contrast to the similar catalytic performance (i.e., low NOx conversion and low N2 selectivity) obtained on Pt/TiO2 + Y-front and Pt/TiO2 + Y-rear systems, much more excellent H2-SCR performance was achieved on the Pt/TiO2 + Y system, where Pt/TiO2 and Y powders were thoroughly physically mixed with appropriate contact (Supplementary Fig. 2b, c). To achieve even closer contact between Pt/TiO2 and Y, we further physically mixed the Pt/TiO2 and Y powders in the presence of water, referred as (Pt/TiO2 + Y)_H2O, and loaded Pt onto a pre-prepared 50% TiO2/Y support (denoted as Pt/TiO2/Y). It was observed that the (Pt/TiO2 + Y)_H2O catalyst showed slightly higher activity (Supplementary Fig. 3), and the Pt/TiO2/Y catalyst exhibited lower activity compared to the Pt/TiO2 + Y catalyst. However, both catalysts demonstrated lower N2 selectivity than Pt/TiO2 + Y catalyst. These results evidently suggest the critical role of establishing an appropriate contact between Pt/TiO2 and Y zeolite in enhancing the overall H2-SCR performance. As shown in Supplementary Fig. 4, the optimal content of Y in the Pt/TiO2 + Y mixture system was determined as 50 wt%, and this formulation was simply denoted as Pt/TiO2 + Y thereafter. To better understand this system, we also physically mixed the Pt/TiO2 catalyst with additional TiO2, and the Pt/Y catalyst (prepared by IWI method) with additional TiO2 or Y, and used them as reference catalysts. As presented in Supplementary Fig. 5, the physical mixing of Pt/TiO2 and TiO2 showed no obvious impact on the NOx conversion and N2 selectivity. However, the physical mixing of Pt/Y with TiO2 or Y resulted in considerable enhancement of the H2-SCR performance. It was worth noting that the Pt/TiO2 + Y formulation outperformed all other catalysts in terms of H2-SCR activity and showed reasonable N2 selectivity. To gain a deeper insight into the Y promotion effect, the relationship between N2 selectivity and NOx conversion in the H2-SCR reaction on selected catalysts was established, as depicted in Fig. 1d. Interestingly, the N2 selectivity versus NOx conversion on all catalysts adhered to the same linear relationship, suggesting that the addition of Y or TiO2 did not alter the overall H2-SCR reaction mechanism on Pt/TiO2 catalyst (yet the Y addition might have changed the N2 formation pathway leading to lower N2O production, which can be verified by the subsequent experimental results and theoretical calculations).
In addition to Y, the use of other types of zeolites for physical mixing with Pt/TiO2 has also been explored in the H2-SCR reaction (Supplementary Fig. 6). Remarkably, the incorporation of different zeolites such as ZSM-5, CHA, FER, and Beta also yielded substantial benefit, significantly enhancing the H2-SCR performance. Considering both the H2-SCR activity and N2 selectivity in the investigated temperature range, it is evident that Y stands out as the optimal zeolite for promoting the Pt/TiO2 catalyst. To simulate the status of catalysts for H2-ICE exhaust purification after prolonged operation, hydrothermal aging on Pt/TiO2 and Pt/TiO2 + Y catalysts was conducted at 650 °C for 50 h under 10% H2O and 10% O2. As shown in Supplementary Fig. 7, not only before but also after the hydrothermal aging, the inclusion of Y in Pt/TiO2 + Y system consistently exhibited remarkable enhancement on the H2-SCR performance, with notably higher NOx conversion and N2 selectivity achieved than those by the zeolite-free Pt/TiO2 catalyst. To further verify the universality of this physical mixing strategy, the H2-SCR testing on the hydrothermally aged Pt/Al2O3 and Pt/SiO2 catalysts with and without Y addition was also performed, and the results are shown in Supplementary Fig. 8. Evidently, the aged Pt/Al2O3 + Y and Pt/SiO2 + Y systems demonstrated significantly enhanced activity and N2 selectivity across the entire spectrum of reaction temperatures when contrasted with their Y-absent counterparts. It is clear that physically mixing the conventional Pt/oxide catalysts with zeolites represents a simple yet universally effective strategy for boosting the H2-SCR performance, particularly tailored for the efficient NOx removal from vehicle exhaust at low temperatures.
Structural characterization of Pt/TiO2 before and after physical mixing with Y zeolite
It might be expected that the physical mixing with Y could have modified the physicochemical properties of Pt/TiO2 leading to the distinguishable catalytic performance. We excluded this hypothesis by systematically characterizing the Pt/TiO2 and Pt/TiO2 + Y catalysts using multiple techniques. X-ray diffraction (XRD) (Supplementary Fig. 9) and N2 adsorption-desorption experiments (Supplementary Fig. 10 and Supplementary Table 2) revealed that the physical mixing showed negligible impact on the crystal structure and textual properties including surface area and porosity of both Pt/TiO2 and Y. It was observed that the Pt/TiO2 + Y system exhibited a similar TiO2 grain size (20.6 nm) to that of Pt/TiO2 (20.0 nm), and its surface area (349 m2/g) and total pore volume (0.318 cm3/g) were approximately the mathematical average of the values for Pt/TiO2 (81 m2/g, 0.178 cm3/g) and Y (709 m2/g, 0.513 cm3/g), respectively. Additionally, the Pt/TiO2 + Y system demonstrated structural stability, with no apparent changes in crystal structure or textural properties after reaction at 300 °C under testing conditions with H2O. In addition to the presence of micropores with the average diameter of 0.6 nm, Y zeolite also displayed significant mesopore defects with the average diameter of 3.8 nm that were probably formed during the dealumination process for Y zeolite production. These defects could potentially offer a substantial number of special Brønsted acidic sites (i.e., hydroxyls associated to extra-framework Al enriched on the inner pore surface), which might play a crucial role in facilitating the adsorption of H2O molecules to occupy the mesopore structures34. The change of the H2O adsorption behavior induced by Y zeolite might have altered the H2-SCR reaction pathway on Pt/TiO2, which will be thoroughly discussed in later sections.
As expected, the Pt particles within both Pt/TiO2 and Pt/TiO2 + Y catalysts showed very similar average sizes (6.0 nm vs. 6.2 nm), Pt dispersions (8.9% vs. 8.5%), CO adsorption features on Pt particle, and Pt-Pt coordination numbers (11.4 vs. 10.7), as evidenced by the characterization results of transmission electron microscopy (TEM), CO pulse titration, in situ diffuse reflectance infrared Fourier transform spectroscopy (DRIFTS) of CO adsorption, and X-ray absorption spectroscopy (XAS) (Fig. 2a, b, Supplementary Figs. 11, 12 and 13, Supplementary Table 3). Furthermore, the linear combination fitting results of X-ray absorption near-edge structure (XANES) for Pt L3-edge demonstrated that the averaged oxidation states of Pt were 0.21 and 0.48 in Pt/TiO2 before and after the Y addition. These values closely resembled the metallic Pt, a finding further supported by the X-ray photoelectron spectroscopy (XPS) analysis of Pt 4d (Supplementary Fig. 14, Supplementary Table 4). These results clearly demonstrated that the physical mixing with Y zeolite did not change the structure of Pt/TiO2, and this conclusion was further supported by the observation of almost identical H2 temperature-programed reduction (H2-TPR) profiles on Pt/TiO2 and Pt/TiO2 + Y (Supplementary Fig. 15). Additionally, the energy dispersive spectroscopy (EDS) mapping results of Pt/TiO2 + Y revealed that the Pt/TiO2 components were surrounded by Y zeolite particles, without obvious direct interaction between Pt species and Y, before and after H2-SCR reaction (Fig. 2c, Supplementary Fig. 16). Therefore, different from the chemical modifications as reported previously24,25,26,27,28,29, the substantial enhancement in H2-SCR performance on Pt/TiO2 by physically mixing with Y was unequivocally attributable to the factors other than the active site modification.
a CO pulse titration results (with the Pt metal dispersion data inserted); b in situ DRIFTS of CO adsorption at 25 °C on Pt/TiO2 and Pt/TiO2 + Y catalysts; c EDS mapping images for Pt/TiO2 + Y system.
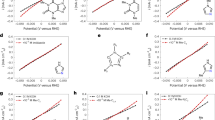
Understanding on the Y promotion effect in Pt/TiO2 + Y system
To determine the role of each reactant and the promotion effect of Y, the reaction orders of NO, H2 and O2 were measured for the H2-SCR reaction. It was found that the reaction orders of NO on both Pt/TiO2 and Pt/TiO2 + Y were dependent on the NO partial pressure (Supplementary Fig. 17a). On Pt/TiO2, at NO partial pressure below 25.3 Pa, the NO reaction order was determined as 0.95, while this value decreased to 0.63 at NO partial pressure above 25.3 Pa. Meanwhile, the reaction orders of H2 and O2 on Pt/TiO2 were determined as 0.48 and –0.13 (Supplementary Fig. 17b, c), respectively. After physical mixing with Y, there was no evident change in the O2 reaction order on Pt/TiO2 + Y (only from –0.13 to –0.08). However, a notable increase in the NO reaction order (from 0.95 to 1.09 at lower NO partial pressure, and from 0.63 to 0.80 at higher NO partial pressure) and an obvious decrease in the H2 reaction order (from 0.48 to 0.32) were observed on Pt/TiO2 + Y. These results suggest that the introduction of Y probably decreased the NO adsorption, and at the same time promoted the H2 activation because of the potential increase in H2 coverage on catalyst surface. The enhanced H2 activation was further supported by the experimental findings presented in Supplementary Figs. 17d and 18. The Pt/TiO₂ + Y system demonstrated superior H₂ oxidation activity compared to the Pt/TiO₂ reference, with the H₂ oxidation activity promoted further as the Y content in the Pt/TiO₂ + Y system increased (Supplementary Fig. 18). This additional increase in H₂ activation could improve the low-temperature activity and reduce the high-temperature activity, as observed on Pt/TiO₂ + Y-67% compared to that on Pt/TiO₂ + Y-50% (Supplementary Fig. 4)14,25. Under our testing conditions (500 ppm NO, 1% H2, and 10% O2), the H2-SCR reaction rates can be expressed as: r(Pt/TiO2) = k1⋅[NO]0.63⋅[H2]0.48⋅[O2]−0.13 and r(Pt/TiO2 +Y) = k2⋅[NO]0.80⋅[H2]0.32⋅[O2]−0.08, where k1 and k2 are constants. Notably, the NO and H₂ reaction orders on both Pt/TiO₂ (0.63 and 0.48, respectively) and Pt/TiO₂ + Y (0.80 and 0.32, respectively) are lower than 1. This suggests that the H₂-SCR reaction on both catalysts involved adsorbed NO and dissociated H* species, following the Langmuir-Hinshelwood (L-H) mechanism. Without changing the L-H mechanism, the enhanced H2 activation could contribute to the improved H2-SCR activity of Pt/TiO₂ + Y.
The effect of the possibly present NO2 or NH3 in the reaction atmosphere on H2-SCR activity was also studied. In separate NO oxidation testing, the Pt/TiO2 + Y system displayed noticeably lower NO oxidation activity comparing to Pt/TiO2 (Supplementary Fig. 19a), and the presence of NO2 in the H2-SCR reaction atmosphere drastically decreased the low-temperature NOx conversion on both Pt/TiO2 and Pt/TiO2 + Y (Supplementary Fig. 19b). Therefore, the presence of any NO2, generated during H2-SCR, was not responsible for the enhanced H2-SCR activity on Pt/TiO2 + Y. In addition, the potential promotional effect of NH3 (possibly formed in situ through the reduction of NOx by H2) on H2-SCR activity was ruled out on both Pt/TiO2 and Pt/TiO2 + Y. This was evident from the decrease in the H2-SCR activity observed upon the introduction of NH3 into the reaction stream at different temperatures (Supplementary Fig. 20).
To understand the impact of Y addition on NO adsorption behavior, the in situ DRIFTS of NO desorption at different temperatures and NO-temperature programmed desorption (NO-TPD) were conducted on Pt/TiO2 and Pt/TiO2 + Y catalysts. As shown in Fig. 3a, the NO adsorption on Pt/TiO2 at 100 °C showed three distinctive bands, corresponding to bridging nitrates (1618 cm−1), bidentate nitrates (1586 cm−1), and monodentate nitrates (1521 cm−1)35,36. In clear contrast, the NO adsorption on Pt/TiO2 + Y exhibited a significantly lower intensity, with the disappearance of monodentate nitrates (Fig. 3b). As the temperature elevated, the nitrate species on both Pt/TiO2 and Pt/TiO2 + Y catalysts decreased in intensity, following the sequence of monodentate nitrates > bidentate nitrates > bridging nitrates (Fig. 3a and b). An initial upswing in the bridging nitrates on Pt/TiO2 was noted, attributed to the intrinsic transformation within the different types of nitrate species37. To assess the NO adsorption affinity, the normalized intensities of bidentate nitrates were presented at different temperatures (Fig. 3c). A much more rapid nitrate desorption from Pt/TiO2 + Y was observed comparing to that from Pt/TiO2. Such results unequivocally demonstrated the substantial inhibitory effect of Y zeolite on the NO adsorption onto Pt/TiO2, concurrently fostering the desorption of NO from Pt/TiO2 + Y. These findings were further supported by the NO-TPD results (Fig. 3d), revealing that the Pt/TiO2 + Y system indeed exhibited notably reduced NO desorption intensity and lowered desorption temperature (242 oC) comparing to Pt/TiO2 (278 oC).
In situ DRIFTS of NO desorption on (a) Pt/TiO2 and (b) Pt/TiO2 + Y catalysts, and (c) normalized peak intensity (at 1586 cm–1) for NO adsorption on Pt/TiO2 and Pt/TiO2 + Y catalysts at different temperatures; (d) NO-TPD profiles, (e) in situ DRIFTS of H2O adsorption at 120 oC, and (f) H2O-TPD profiles on Pt/TiO2 and Pt/TiO2 + Y catalysts.
Considering that H2O is the primary product in the H2-SCR reaction, the impact of Y addition on H2O adsorption property was also investigated. The in situ DRIFTS of H2O adsorption on both Pt/TiO2 and Pt/TiO2 + Y at 120 oC clearly showed distinct peaks at ca. 1630 cm−1, indicative of adsorbed H2O molecules38. Additionally, broad peaks at ca. 3200 cm−1 were observed, corresponding to the hydroxyl species derived from adsorbed H2O with bending feature38,39,40. Comparing to the case on Pt/TiO2, H2O adsorption on Pt/TiO2 + Y displayed more prominent peaks (Fig. 3e), suggesting the enhanced H2O adsorption due to the presence of Y. This enhancement was also confirmed by the H2O-TPD results (Fig. 3f), where more pronounced H2O desorption peaks were observed on Pt/TiO2 + Y. Other than the H2O desorption peak observed at 223 oC on both catalysts, an additional desorption peak at 164 oC was detected only on Pt/TiO2 + Y. This low-temperature peak could be attributed to the physically adsorbed H2O on the Y zeolite.
To reveal the effect of H2O adsorption on the H2-SCR performance, the catalysts were either pre-dehydrated or pre-adsorbed with H2O prior to the H2-SCR testing. Under the testing condition without H2O, Pt/TiO2 + Y always outperformed Pt/TiO2 in terms of NOx conversion and N2 selectivity (Fig. 4a, Supplementary Fig. 21a). Comparing to the situation with pre-dehydration, interestingly, the pre-adsorption of H2O on both catalysts improved their H2-SCR performance. This improvement was particularly significant regarding the NOx conversion on Pt/TiO2 catalyst, which exhibited relatively weaker H2O adsorption capacity as confirmed earlier. The gas phase H2O formation was monitored during the H2-SCR reaction (Supplementary Fig. 21b). As expected, much faster increase in H2O concentration was observed over both catalysts subjected to the pre-adsorption of H2O comparing to those subjected to the pre-dehydration. It was noticeable that, as shown in Fig. 4b, a discernible correlation emerged between the elevation in NOx conversion and the concurrent rise in gas phase H2O concentration over Pt/TiO2 catalyst. However, over Pt/TiO2 + Y system, the rise in gas phase H2O concentration exhibited a delay compared to the progression of NOx conversion, suggesting the capture of in situ formed H2O due to the presence of Y. To further confirm the promotion effect of in situ generated H2O and to verify the effect of NO adsorption on the H2-SCR activity, transient H2-SCR testing was conducted at 100 oC (Fig. 4c). Using the NOx concentrations when switching from Ar flow to H2-SCR flow as baselines, significant decrease in NOx concentrations was observed when switching from H2 + O2 flow to H2-SCR flow, while obvious increase in NOx concentrations was observed when switching from NO + O2 flow to H2-SCR flow, on both catalysts. Clearly, initiating a pre-flow of H2 + O2 yielded benefit on improving the H2-SCR activity, while pre-flowing NO + O2 inhibited the H2-SCR reaction to a certain extent. Such inhibition caused by the NO + O2 flow could be due to the extensive coverage of Pt sites by NO, impeding the activation of H230. At 100 oC, the complete oxidation of H2 to H2O could be achieved on both catalysts (Supplementary Fig. 18). Consequently, the benefit of pre-flowing H2 + O2 should be originated from the adsorption of in situ formed H2O. The presence of H2O could strongly inhibit the NO adsorption, as confirmed by the in situ DRIFTS (Fig. 4d) and NO-TPD (Fig. 4e) analyses conducted on Pt/TiO2 catalyst. The physical mixing of Pt/TiO2 with Y could further promote the adsorption of in situ generated H2O (Fig. 3e, f), creating a H2O-rich environment around the Pt sites and facilitating the formation of a H2O-covered Pt surface. This surface could reduce the NO coverage on Pt sites, thereby improving H₂ activation and H₂-SCR performance (Fig. 4a). However, introducing 5% external H2O into the reaction flow could significantly inhibit the diffusion of NO and H2 (NO/H2/H2O molar ratio = 1/20/100) to the catalyst surface. Despite this inhibition resulting in the decreased activity for both catalysts, the Pt/TiO₂ + Y catalyst still exhibited significantly higher activity compared to the Pt/TiO₂ catalyst (Fig. 1a).
a NOx conversion and (b) gas phase H2O formation during the H2-SCR reaction over Pt/TiO2 and Pt/TiO2 + Y catalysts with pre-dehydration at 300 oC or pre-adsorption of H2O at 30 oC. Reaction conditions: 26 mg of Pt/TiO2 catalyst, or a mixture containing 26 mg of Pt/TiO2 and 26 mg of Y; transient-state light-off testing; 500 ppm NO, 1% H2, and 10% O2; WHSV = 461,540 mL·gPt/TiO2–1·h–1. c Time-resolved NOx concentration after switching from different flows (Ar; or 1% H2 + 10% O2; or 500 ppm NO + 10% O2) to H2-SCR flow (500 ppm NO + 1% H2 + 10% O2) on Pt/TiO2 and Pt/TiO2 + Y catalysts at 100 oC. Testing conditions: 10 mg of Pt/TiO2, or a mixture containing 10 mg of Pt/TiO2 and 10 mg of Y; WHSV = 1,200,000 mL·gPt/TiO2–1·h–1. d In situ DRIFTS of NO adsorption at 100 oC, and (e) NO-TPD profiles on Pt/TiO2 catalyst under the NO adsorption conditions with and without 5% H2O.
To elucidate the intrinsic promotion effect of Y addition to Pt/TiO2 on the H2-SCR performance, systematic density functional theory (DFT) calculations were performed. The Pt (111) surface was selected to represent the Pt active site in Pt/TiO2 catalyst due to its high thermodynamic stability (Supplementary Fig. 22). As previously confirmed, the Y zeolite in Pt/TiO2 + Y system possessed high ability to capture the in situ generated H2O, creating the H2O-rich environment around Pt sites. In light of this, a stable H2O/Pt (111) interface was constructed (Supplementary Fig. 23), featuring a hydrogen bonding network with half of H2O molecules dissociated on Pt with 2/3 monolayer (ML) coverage41,42. Such configuration was denoted as the H2O/Pt (111) surface to represent the Pt active site in Pt/TiO2 + Y system.
As shown in Supplementary Fig. 24, the Pt (111) surface was found more favorable for the NO adsorption with much higher free adsorption energy (–1.77 eV) comparing to that for H2 adsorption (–0.87 eV) at the low coverage limit. Consequently, the optimal NO coverage on Pt (111) was firstly investigated by calculating the total Gibbs free adsorption energies, which was determined as 7/12 ML at relative low temperatures (T = 320-470 K and PNO = 50 Pa) and in line with previous study30. With the highest total Gibbs free adsorption energy, the 7 NO/Pt (111) structure emerged as the most stable configuration (Fig. 5a, Supplementary Fig. 25), which was adopted as the starting point for studying the H2 activation and reaction mechanism on Pt/TiO2. On the stable H2O/Pt (111) surface, the presence of a repulsive hydrogen bonding network led to a significant decline in the averaged free NO adsorption energies. Consequently, a notably reduced NO coverage (1/3 ML) was observed on the H2O/Pt (111) surface (Fig. 5a, Supplementary Fig. 26), in comparison to the NO overage (7/12 ML) on the Pt (111) surface. Considering that the weakly-bonded *NO (T = 373 K, average Gads = ~0.5 eV) was highly active and unstable, the H2O/Pt (111) surface without *NO was used as the starting point for studying the H2 activation and reaction mechanism on Pt/TiO2 + Y. Accordingly, the H2 adsorption and activation energies on 7 NO/Pt (111) and H2O/Pt (111) surfaces were calculated, and the results are shown in Fig. 5b. Comparing to the endergonic process of H2 adsorption (ΔG = 0.30 eV) and high H2 activation barrier (Ga = 0.75 eV) observed on 7 NO/Pt (111) surface, an exergonic process of H2 adsorption (ΔG = –0.63 eV) and much lower H2 activation barrier (Ga = 0.30 eV) was found on H2O/Pt (111) surface. Evidently, the H2O/Pt (111) surface benefited the H2 adsorption and activation, well aligned with the experimental results showing that Pt/TiO2 + Y system exhibited superior H2 activation ability comparing to Pt/TiO2 (Supplementary Fig. 18).
a Total Gibbs free adsorption energy of NO molecules on Pt (111) and H2O/Pt (111) surfaces. The reference state is the gas phase NO at 50 Pa. b Gibbs free energy diagram of H2 activation on the 7 NO/Pt (111) surface and the H2O/Pt (111) surface. The reference state is the gas phase H2 at 1 atm. Color code: Pt (silver), O (red), N (blue), and H (yellow).
The theoretical calculations of H2-SCR reaction mechanism on Pt (111) and H2O/Pt (111) surfaces were conducted to further elucidate the promotion effect of Y zeolite in Pt/TiO2 + Y system. On Pt (111) surface, as shown in Supplementary Fig. 27 and Supplementary Table 5, the dissociation of *HNOH species into *NH and *OH was found to be the rate-determining step (RDS) for NO reduction with an activation energy (Ea) of 1.20 eV. Due to the high NO coverage, the inhibited H2 activation further hindered the selective reduction of N-containing species to N2, resulting in the high N2O formation and low N2 selectivity. In clear contrast, on H2O/Pt (111) surface (Fig. 6, Supplementary Table 6), the *HNOH species could be readily dissociated into *NH and *OH with a lower activation energy of 0.24 eV (image viii to ix). Once the *NH species was formed, the gas phase NO could facilely couple with it to generate *HNNO, involving a substantial exothermicity of 2.31 eV (image ix to x). Interestingly, rather than releasing N2O (with Ea = 1.23 eV from image x to ii), the *HNNO species remained until an OH vacancy was facilely created in the hydrogen bonding network following the H2O formation (Ea = 0.40 eV from image x to xi) and desorption (ΔE = 0.32 eV from image xi to xii). Subsequently, the *HNNO species was activated and dissociated, selectively producing N2 with a barrier of 0.54 eV (image xii to xiii). Therefore, the RDS for NO reduction on H2O/Pt (111) surface included both the creation of OH vacancy in the hydrogen bonding network and N2 formation, with an overall activation energy of 0.86 eV (from image xi to xiii). Such activation energy for the RDS of H2-SCR reaction on H2O/Pt (111) surface was much lower than that on Pt (111) surface (1.20 eV). These simulation results well explained the significant promotion effect of Y zeolite in Pt/TiO2 + Y system for H2-SCR in terms of both enhanced NO removal efficiency and elevated N2 selectivity.
The reaction was proposed to proceed on the H2O/Pt (111) surface representing the structure of Pt/TiO2 + Y catalyst under reaction conditions. The reaction energies and activation energies are indicated in eV in the diagram. Color code: Pt (silver), O (red), N (blue), and H (yellow). Corresponding energies are given in Supplementary Table 6.
Discussion
A facile, universal and sustainable strategy of physically mixing Pt/oxide catalysts with zeolites has been successfully developed to improve the H2-SCR performance of Pt-based catalysts for low-temperature NOx removal. The Pt/TiO2 + Y system exhibited superior H2-SCR performance consistently in terms of NOx conversion and N2 selectivity, both before and after hydrothermal aging, as well as across various testing conditions. This catalyst system shows immense potential in H2-SCR applications, particularly for H2-ICE emission control. It was discovered that the incorporation of Y zeolite effectively promoted H2O adsorption and the formation of H2O-rich environment surrounding Pt active sites in Pt/TiO2 + Y system. This consequently led to the reduction in excessive NO coverage and the improvement in H2 activation, yielding substantial advantages for boosting both H2-SCR efficiency and N2 selectivity. In contrast to modifying the active sites through chemical methods, this study underscores the crucial importance of fine tuning the surrounding environment of active sites through an easy, sustainable physical mixing approach to achieve proficient heterogeneous catalysis.
Methods
Catalyst preparation
The Pt/oxide catalysts used in this study, including Pt/TiO2, Pt/Al2O3, and Pt/SiO2, were prepared using incipient wetness impregnation (IWI) method. A solution of colloidal Pt (2-6 nm) with 1 wt% Pt was added dropwise onto commercial anatase TiO2 (surface area = 90 m2/g), γ-Al2O3 (surface area = 150 m2/g), or SiO2 (surface area = 180 m2/g) under stirring, followed by drying at 120 °C for 1 h. After calcination in air at 550 °C for 2 h with the temperature ramp of 5 oC/min, the catalysts were obtained and denoted as Pt/TiO2, Pt/Al2O3, and Pt/SiO2, respectively. As a reference, Pt/Y catalyst was also prepared by the same method using H-Y zeolite (SiO2/Al2O3 molar ratio = 30) as support.
For physical mixing with Pt/oxide catalysts, commercial zeolites including H-Y (SiO2/Al2O3 molar ratio = 30), H-ZSM-5 (SiO2/Al2O3 molar ratio = 30), H-chabazite (CHA, SiO2/Al2O3 molar ratio = 29), H-ferrierite (FER, SiO2/Al2O3 molar ratio = 30), and H-Beta (SiO2/Al2O3 molar ratio = 25) were used. TiO2 or H-Y was also used to dilute Pt/TiO2 or Pt/Y, respectively, for comparison. The content of additional zeolite/oxide was typically controlled at 50 wt% in the physically mixed samples, except for the Pt/TiO2 + Y system with different Y contents of 33, 50, and 67 wt%. These mixed samples were denoted as Pt/oxide + zeolite or oxide. To simulate the catalyst throughout its operational lifespan in heavy-duty vehicles powered by diesel or hydrogen fuel, accelerated aging treatment under hydrothermal conditions of 550–650 °C for 50–100 h should be conducted. In this study, an aging treatment under 10% H2O and 10% O2 at 650 °C for 50 h was performed, and the resulting catalysts were labeled with “-Aged”.
Catalyst characterizations
X-ray diffraction (XRD) measurement was performed on a PANalytical Empyrean diffractometer using a Cu Kα radiation source (λ = 0.15406 nm). The measurement covered the 5o to 80o range with a scan mode of 6 o/min and a scan step of 0.067o.
N2 physisorption was used to determine the surface area, pore volume, and pore size distribution, which was performed on a Quantachrome Autosorb-iQ instrument at liquid nitrogen temperature (77 K). Prior to measurement, all samples were degassed at 300 °C for 2 h under vacuum. The N2 adsorption-desorption isotherm was measured with 40 adsorption and 40 desorption points for Y and Pt/TiO2 + Y samples, and with 20 adsorption and 20 desorption points for Pt/TiO2 using the pressure intervals of 0 < P/P0 < 1. The surface area was calculated using the Brunauer–Emmett–Teller (BET) method based on the adsorption points in the relative pressure range between 0.05 and 0.3. The Horvath-Kawazoe (HK) method and non-local density functional theory (DFT) method were used to determine the pore volume and pore size distribution.
Transmission electron microscopy (TEM) and energy dispersive X-ray spectroscopy (EDS) mapping images were collected on a field emission FEI Tecnai F-30 with HAADF/ADF/BF STEM and EDS detectors operated at 200 kV.
The CO chemisorption measurement was performed on a Quantachrome Autosorb-iQ instrument. Before each measurement, the sample was first exposed to flowing He from room temperature to 150 °C at the ramp rate of 5 °C/min, and then held at 150 °C for 10 min. Next, the system was purged with 10% H2/Ar, and the temperature was ramped to 400 °C at the rate of 5 °C/min and kept for 30 min. It is important to note that a certain degree of Pt sintering might occur during this reduction treatment, potentially resulting in a lower-estimated Pt dispersion value. The system was then switched back to He, while maintaining the temperature at 400 °C for 30 min. The final step involved cooling the system down to 35 °C in He at the rate of 20 °C/min, holding at 35 °C for 30 min, and injecting multiple CO pulses (5% CO/He) using thermal conductivity detector (TCD) to monitor the gas phase CO.
The X-ray absorption near-edge structure (XANES) and extended X-ray absorption fine structure (EXAFS) of Pt L3-edge were measured at room temperature in fluorescent mode at beamline 7-BM QAS of the National Synchrotron Light Source II (NSLS-II), Brookhaven National Laboratory. Pt foil was measured during data collection for energy calibration and drift correction of the monochromator. Data analysis was conducted using Athena and Artemis from the Demeter software package. The processed EXAFS, χ(k), was weighted by k2 to amplify the high-k oscillations. For Fourier-transformed (FT) spectra, the k range between 3.0 and 12.0 Å was used, and the curve fitting was performed using the Artemis software.
X-ray photoelectron spectroscopy (XPS) was measured on a Thermo Scientific ESCALAB 250Xi photoelectron spectrometer using Al K-α (hν = 1486.68 eV) as the X-ray source in ultrahigh vacuum condition (10−7 Pa). The binding energy (BE) of Pt 4d spectra was corrected using the C 1s signal at 284.6 eV as reference.
H2 temperature-programmed reduction (H2-TPR) was performed on the Quantachrome Autosorb-iQ instrument. Prior to testing, the samples were pretreated in a flow of 5% O2/He at 300 °C for 1 h. After cooling down to 40 oC, a flow of 10% H2/Ar was used, and the temperature was raised linearly from 40 to 700 oC at the ramp rate of 10 oC/min. The H2 consumption was monitored on-line using TCD.
In situ DRIFTS experiments were performed on a Nicolet iS50 FTIR spectrometer equipped with a liquid nitrogen-cooled mercury-cadmium-telluride (MCT) detector and an in situ IR cell with ZnSe windows (DiffusIR, PIKE Technologies). Prior to measurements, all samples were pretreated in Ar flow at 300 oC for 1 h. The background spectra at different temperatures (e.g., 25, 100, 125, 150, 175, 200, 225, 250, 275, and 300 oC) were collected in Ar flow using 100 scans with a resolution of 4 cm−1. For in situ DRIFTS of CO adsorption at 25 oC, 1% CO/Ar was introduced into the IR cell and kept for 30 min. Then, the samples were purged by Ar for 30 min to remove the weakly adsorbed CO, followed by spectra collection. For in situ DRIFTS of NO adsorption/desorption, the feed stream of 1000 ppm NO, 10% O2, and 5% H2O (when used) in Ar was introduced into the cell with a flow rate of 50 mL/min and kept for 60 min to achieve the saturated NO adsorption at 100 oC. The NO flow was then discontinued while Ar (50 mL/min) was kept flowing for 30 min to remove the gaseous and weakly adsorbed NO. Afterwards, the desorption experiments were carried out in Ar flow with the temperature elevated from 100 to 300 °C with an interval of 25 oC, and the spectra were collected under steady state accordingly. For in situ DRIFTS of H2O adsorption, a feed stream of 5% H2O in Ar was introduced into the cell at a flow rate of 50 mL/min and kept for 60 min, achieving saturated H2O adsorption at 120 oC. Then, the sample was purged with Ar for 30 min at 120 oC to remove the weakly adsorbed H2O, and a background spectrum was collected. The sample was finally treated in Ar flow at 500 °C for 2 h and cooled down to 120 oC, followed by the spectrum collection.
NO temperature-programmed desorption (NO-TPD) and H2O temperature-programmed desorption (H2O-TPD) were conducted on a continuous flow fixed-bed system. A quartz tubular microreactor with an internal diameter of 4.0 mm was used, and a Hidden Analytical mass spectrometer (MS) was employed as detector. Typically, a feed stream of 1000 ppm NO, 10% O2, and 5% H2O (when used) in Ar was introduced into the reactor at a flow rate of 40 mL/min and kept for 60 min, achieving saturated NO adsorption at 50 oC. Afterwards, the sample was purged with Ar (40 mL/min) for 120 min at 50 oC to remove the weakly adsorbed molecules. The temperature was then elevated linearly from 50 to 600 oC at a ramp rate of 10 oC/min. For H2O-TPD, a feed stream of 5% H2O in Ar was introduced into the reactor at a flow rate of 40 mL/min and kept for 60 min, achieving saturated H2O adsorption at 50 oC. The sample was then purged with Ar (40 mL/min) for 120 min at 50 oC to remove the weakly adsorbed H2O. Subsequently, the temperature was elevated linearly from 50 to 600 oC at a ramp rate of 10 oC/min. The NO or H2O desorption was monitored on-line using m/z of 30 or 18, respectively.
Catalytic performance evaluation
The catalytic activity evaluation for the H2-SCR of NOx over all catalysts was conducted using a continuous flow fixed-bed quartz tubular microreactor with an internal diameter of 4.0 mm. In each test, the catalyst or physical mixture containing 26 mg of Pt/oxide catalyst (40–60 mesh) was diluted with 0.25 g of inert SiC (40-60 mesh) to minimize the effect of hot spots. The reaction atmosphere comprised of 500 ppm NO, 1% H2, 10% O2, 5% CO2 (when used) and 5% H2O (when used), using Ar as balance. The total flow rate was controlled at 200 mL/min, resulting in a weight hourly space velocity (WHSV) of 461,540 mL·gPt/oxide–1·h–1. During the steady-state testing, the catalyst was held at each temperature for a duration of 30 min. Reactants and products were analyzed online by a MultiGas 2030 CEM-Cert FTIR spectrometer. The reactant conversion was defined as (cinlet – coutlet)/cinlet × 100%, where cinlet and coutlet were the inlet and outlet NOx concentration in the feed stream, respectively. The N2 selectivity was defined as ([NO]inlet + [NO2]inlet – [NO]outlet – [NO2]outlet – 2 × [N2O]outlet)/([NO]inlet + [NO2]inlet – [NO]outlet – [NO2]outlet) × 100%. Under the H2-SCR testing conditions with 1% H2 and 10% O2, NO could be either selectively reduced by H2 to form N2/N2O or oxidized by O2 to form NO2. The NO selective conversion attributed to the NO reduction by H2 (NO + H2) under the H2-SCR condition was defined as ([NO]inlet + [NO2]inlet – [NO]outlet – [NO2]outlet)/([NO]inlet – [NO]outlet) × 100%, and the NO conversion attributed to the NO oxidation by O2 (NO + O2) under the H2-SCR condition was defined as ([NO2]outlet – [NO2]inlet)/([NO]inlet – [NO]outlet) × 100%. To avoid the significant heat or mass transfer limitation, the kinetics study was performed at 100 oC under the WHSV of 2,400,000 mL·gPt/TiO2–1·h–1 to determine the NO, H2, and O2 reaction orders on Pt/TiO2 and Pt/TiO2 + Y catalysts. The catalytic performance evaluations for separate NO oxidation, H2-SCR in the presence of NO2, H2-SCR in the presence of NH3, separate H2 oxidation, as well as the H2-SCR reaction on the catalysts with pre-dehydration and pre-adsorption of H2O, were also conducted. The detailed information can be found in Supplementary Text 1.
DFT calculations
Periodic non-spin-polarized DFT calculations were performed using the Vienna Ab-initio Simulation Package (VASP) and the Perdew-Burke-Ernzerhof functional within generalized gradient approximation (GGA). The valence electrons were described by projector augmented wave pseudopotentials with an energy cutoff of 400 eV for all the calculations. The Methfessel-Paxton smearing scheme was used with a width of 0.15 eV and the precision was set to “accurate”. The convergence criteria for energies and forces in structure optimizations were set as 10−5 eV and 0.02 eV Å−1, respectively. The van der Waals (vdW) interactions were included via using Grimme’s DFT-D3 method. The Brillouin zone for periodic slab calculations was sampled on Γ-centered Monkhorst-Pack type 2 × 3 × 1 k-point grid. Transition states of surface reactions were searched by the nudged elastic band (NEB) together with the dimer method. Further vibrational analysis was adopted to confirm the transition states. Only one imaginary frequency mode along the reaction trajectory represented the true saddle point.
The reaction energy (ΔE) of each elementary step was computed by the difference between the DFT energy of the final state (EFS) and that of the corresponding initial state (EIS), with ΔE = EFS – EIS. Similarly, the activation energy was calculated using the equation, Ea = ETS – EIS, where ETS was the DFT energy of corresponding transition state (TS). H binding energy (Eb(H)) was computed by the equation, Eb(H) = EH/support – Esupport – 0.5EH2, where EH/support, Esupport and EH2 were the DFT energies of support with the *H adsorbate, the support, and gas phase H2, respectively. Gibbs free energy of each species was calculated by
in which G was the Gibbs free energy, and E, EZPE, Cp and S were the DFT energy, zero point energy, heat capacity and entropy of each gas-phase species or surface intermediates, respectively. The EZPE, Cp, and S were calculated within the harmonic approximation. The Atomic Simulation Environment (ASE) package was employed to calculate the Gibbs free energy of gas and adsorbed species at certain temperatures and pressures.
The Gibbs free formation energies of adsorbates on corresponding surface were calculated via the following equation:
in which G(NxOyHz/surface), G(surface), G(NO), G(H2O), and G(H2) were the Gibbs free energies of the surface with adsorbates, the clean surface, and gas phase NO, H2O, and H2 under relevant temperatures and pressures, respectively. The partial pressures of gas phase NO, H2, and H2O were set as 50, 1000, and 5000 Pa, which were within the range of experimental operation conditions.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
Source data are provided with this paper.
References
Raza, S. A., Shah, N. & Sharif, A. Time frequency relationship between energy consumption, economic growth and environmental degradation in the United States: evidence from transportation sector. Energy 173, 706–720 (2019).
Solaymani, S. CO2 emissions patterns in 7 top carbon emitter economies: the case of transport sector. Energy 168, 989–1001 (2019).
Stępień, Z. A comprehensive overview of hydrogen-fueled internal combustion engines: achievements and future challenges. Energies 14, 6504 (2021).
Onorati, A. et al. The role of hydrogen for future internal combustion engines. Int. J. Engine Res. 23, 529–540 (2022).
Luo, Q. H. et al. Experimental investigation of combustion characteristics and NOx emission of a turbocharged hydrogen internal combustion engine. Int. J. Hydrog. 44, 5573–5584 (2019).
Hu, Z. & Yang, R. T. 110th Anniversary: Recent progress and future challenges in selective catalytic reduction of NO by H2 in the presence of O2. Int. J. Engine Res. 58, 10140–10153 (2019).
Liu, Z., Li, J. & Woo, S. I. Recent advances in the selective catalytic reduction of NOx by hydrogen in the presence of oxygen. Energy Environ. Sci. 5, 8799 (2012).
Borchers, M., Keller, K., Lott, P. & Deutschmann, O. Selective catalytic reduction of NOx with H2 for cleaning exhausts of hydrogen engines: Impact of H2O, O2, and NO/H2 ratio. Ind. Eng. Chem. Res. 60, 6613–6626 (2021).
Xie, S. et al. Silica modulated palladium catalyst with superior activity for the selective catalytic reduction of nitrogen oxides with hydrogen. Appl. Catal. B Environ. 327, 122437 (2023).
Yang, S., Wang, X., Chu, W., Song, Z. & Zhao, S. An investigation of the surface intermediates of H2-SCR of NOx over Pt/H-FER. Appl. Catal. B Environ. 107, 380–385 (2011).
Duan, K., Chen, B., Zhu, T. & Liu, Z. Mn promoted Pd/TiO2–Al2O3 catalyst for the selective catalytic reduction of NO by H2. Appl. Catal. B Environ. 176-177, 618–626 (2015).
Park, D. C. et al. Widening the operating window of Pt/ZSM-5 catalysts for efficient NOx removal in H2-SCR: Insights from thermal aging. Catal. Today 425, 114318 (2024).
Li, L., Wu, P., Yu, Q., Wu, G. & Guan, N. Low temperature H2-SCR over platinum catalysts supported on Ti-containing MCM-41. Appl. Catal. B Environ. 94, 254–262 (2010).
Zhang, X. et al. An investigation on N2O formation route over Pt/HY in H2-SCR. Chem. Eng. J. 252, 288–297 (2014).
Dhainaut, F., Pietrzyk, S. & Granger, P. Kinetics of the NO+H2 reaction over supported noble metal based catalysts: support effect on their adsorption properties. Appl. Catal. B Environ. 70, 100–110 (2007).
Dhainaut, F., Pietrzyk, S. & Granger, P. Kinetics of the NO/H2 reaction on Pt/LaCoO3: a combined theoretical and experimental study. J. Catal. 258, 296–305 (2008).
Machida, M., Ikeda, S., Kurogi, D. & Kijima, T. Low temperature catalytic NOx–H2 reactions over Pt/TiO2-ZrO2 in an excess oxygen. Appl. Catal. B Environ. 35, 107–116 (2001).
Costa, C. N. & Efstathiou, A. M. Mechanistic aspects of the H2-SCR of NO on a novel Pt/MgO-CeO2 catalyst. J. Phys. Chem. C 111, 3010–3020 (2007).
Costa, C. N. & Efstathiou, A. M. Low-temperature H2-SCR of NO on a novel Pt/MgO-CeO2 catalyst. Appl. Catal. B Environ. 72, 240–252 (2007).
Park, S. M., Kim, M. Y., Kim, E. S., Han, H. S. & Seo, G. H2-SCR of NO on Pt–MnOx catalysts: reaction path via NH3 formation. Appl. Catal. A Gen. 395, 120–128 (2011).
Yu, Q. et al. The promotional effect of Cr on catalytic activity of Pt/ZSM-35 for H2-SCR in excess oxygen. Catal. Commun. 11, 955–959 (2010).
Komatsubara, M., Koga, A., Tanaka, M., Hagiwara, R. & Iwamoto, M. Three pathways to selective catalytic reduction of NO over Pt/Nb-AlMCM-41 under H2 with excess O2. Catal. Sci. Technol. 6, 7398–7407 (2016).
Yu, Q. et al. Selective catalytic reduction of NO by hydrogen over Pt/ZSM-35. Catal. Today 158, 452–458 (2010).
Zhang, X. et al. Promotion effect of tungsten on the activity of Pt/HZSM-5 for H2-SCR. Chem. Eng. J. 260, 419–426 (2015).
Wang, X., Wang, X., Yu, H. & Wang, X. The functions of Pt located at different positions of HZSM-5 in H2-SCR. Chem. Eng. J. 355, 470–477 (2019).
Burch, R. & Coleman, M. D. An investigation of promoter effects in the reduction of NO by H2 under lean-burn conditions. J. Catal. 208, 435–447 (2002).
Li, X., Zhang, X., Xu, Y., Liu, Y. & Wang, X. Influence of support properties on H2 selective catalytic reduction activities and N2 selectivities of Pt catalysts. Chin. J. Catal. 36, 197–203 (2015).
Yazawa, Y. et al. The support effect on propane combustion over platinum catalyst: control of the oxidation-resistance of platinum by the acid strength of support materials. Appl. Catal. A Gen. 233, 103–112 (2002).
Shibata, J. et al. Factors controlling activity and selectivity for SCR of NO by hydrogen over supported platinum catalysts. J. Phys. Chem. B 108, 18327–18335 (2004).
Farberow, C. A., Dumesic, J. A. & Mavrikakis, M. Density functional theory calculations and analysis of reaction pathways for reduction of nitric oxide by hydrogen on Pt(111). ACS Catal. 4, 3307–3319 (2014).
Liu, Z., Jia, B., Zhang, Y. & Haneda, M. Engineering the metal–support interaction on Pt/TiO2 catalyst to boost the H2-SCR of NOx. Ind. Eng. Chem. Res. 59, 13916–13922 (2020).
Kim, S. S. & Hong, S. C. Relationship between the surface characteristics of Pt catalyst and catalytic performance on the H2-SCR. J. Ind. Eng. Chem. 16, 992–996 (2010).
Sterlepper, S., Fischer, M., Claßen, J., Huth, V. & Pischinger, S. Concepts for hydrogen internal combustion engines and their implications on the exhaust gas aftertreatment system. Energies 14, 8166 (2021).
Halasz, I. & Agarwal, M. Hydrophobic nano-layer on surface prevents H2O adsorption in moderately aluminum deficient Y zeolite crystals. Microporous Mesoporous Mater. 310, 110621 (2021).
Yao, X. et al. Selective catalytic reduction of NOx by NH3 over CeO2 supported on TiO2: comparison of anatase, brookite, and rutile. Appl. Catal. B Environ. 208, 82–93 (2017).
Xie, S. et al. Copper single atom-triggered niobia–ceria catalyst for efficient low-temperature reduction of nitrogen oxides. ACS Catal. 12, 2441–2453 (2022).
Meng, D. et al. A highly effective catalyst of Sm-MnOx for the NH3-SCR of NOx at low temperature: promotional role of Sm and its catalytic performance. ACS Catal. 5, 5973–5983 (2015).
Kipreos, M. D. & Foster, M. Water interactions on the surface of 50 nm rutile TiO2 nanoparticles using in situ DRIFTS. Surf. Sci. 677, 1–7 (2018).
Wu, J. et al. Breaking through water-splitting bottlenecks over carbon nitride with fluorination. Nat. Commun. 13, 6999 (2022).
Rieth, A. J., Hunter, K. M., Dinca, M. & Paesani, F. Hydrogen bonding structure of confined water templated by a metal-organic framework with open metal sites. Nat. Commun. 10, 4771 (2019).
Tripković, V., Skúlason, E., Siahrostami, S., Nørskov, J. K. & Rossmeisl, J. The oxygen reduction reaction mechanism on Pt(111) from density functional theory calculations. Electrochim.Acta 55, 7975–7981 (2010).
Ogasawara, H. et al. Structure and bonding of water on Pt(111). Phys. Rev. Lett. 89, 276102 (2002).
Acknowledgements
This work was supported by a research fund from BASF Environmental Catalyst and Metal Solutions and the Startup Fund (F.L.) from the University of California, Riverside (UCR). S.X., D.K. and X.Z. thank the support from the Preeminent Postdoctoral Program (P3) at the University of Central Florida (UCF). L.L. and H.X. thank the support from NSF CDS&E program (CBET-2245402). F.L. sincerely thanks Mr. Franck Thibaut and Ms. Corinne Lehaut from Tronox Inc., Dr. Marcos Schöneborn from Sasol, and Dr. Chris Bauer from Evonik for providing raw materials in catalyst synthesis. F.L. and S.X. thank Dr. Tangyuan Li and Prof. Liangbing Hu from the University of Maryland for their assistance with N2 physisorption testing. This research used beamline 7-BM (QAS) of the National Synchrotron Light Source II, a U.S. Department of Energy (DOE) Office of Science User Facility operated for the DOE Office of Science by Brookhaven National Laboratory under Contract No. DE-SC0012704. H.X. acknowledges the computational resource provided by the advanced research computing at Virginia Polytechnic Institute and State University.
Author information
Authors and Affiliations
Contributions
F.L. and Y.L. conceived the idea and directed the project. S.X. designed the experiments, performed the experiments, and analyzed the data. K.Y., D.K. and X.Z. assisted with the catalyst testing and characterization. L.M. and S.E. conducted XAS measurements. L.L. and H.X. performed DFT calculations and analysis. S.X. and L.L. wrote the manuscript. F.L. and H.X. mentored the manuscript writing and revision. All authors discussed the results and commented on the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Unai De-La-Torre and the other, anonymous, reviewers for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Xie, S., Liu, L., Li, Y. et al. Zeolite-promoted platinum catalyst for efficient reduction of nitrogen oxides with hydrogen. Nat Commun 15, 7988 (2024). https://doi.org/10.1038/s41467-024-52382-7
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-024-52382-7