Abstract
Deposition of monosodium urate and calcium pyrophosphate (MSU and CPP) micro-crystals is responsible for painful and recurrent inflammation flares in gout and chondrocalcinosis. In these pathologies, the inflammatory reactions are due to the activation of macrophages responsible for releasing various cytokines including IL-1β. The maturation of IL-1β is mediated by the multiprotein NLRP3 inflammasome. Here, we find that activation of the NLRP3 inflammasome by crystals and concomitant production of IL-1β depend on cell volume regulation via activation of the osmo-sensitive LRRC8 anion channels. Both pharmacological inhibition and genetic silencing of LRRC8 abolish NLRP3 inflammasome activation by crystals in vitro and in mouse models of crystal-induced inflammation. Activation of LRRC8 upon MSU/CPP crystal exposure induces ATP release, P2Y receptor activation and intracellular calcium increase necessary for NLRP3 inflammasome activation and IL-1β maturation. We identify a function of the LRRC8 osmo-sensitive anion channels with pathophysiological relevance in the context of joint crystal-induced inflammation.
Similar content being viewed by others
Introduction
Gout due to monosodium urate (MSU) crystal deposition, which occurs after chronic elevation of serum urate level, and calcium pyrophosphate (CPP) crystal deposition (also called chondrocalcinosis) are 2 major crystal-related diseases responsible for recurrent interleukin 1β (IL-1β)-orchestrated inflammation flares. Despite the availability of efficient urate-lowering drugs, gout remains active in more than 70% of patients, with continued gout flares1,2. Moreover, current treatments of CPP disease are only symptomatic and not able to dissolve CPP crystals. IL-1β blockade is effective in reducing MSU and CPP crystal-induced inflammation, but alternative therapies are still needed because prolonged IL-1β blockade is associated with severe side effects such as infections and MSU and CPP crystal-induced inflammation can be resistant to IL-1β blockade3.
MSU and CPP crystals induce the release of IL-1β by resident macrophages4. Production of active IL-1β occurs via the activation of the multiprotein complex nucleotide-binding domain and leucine-rich repeat pyrin 3 domain (NLRP3) inflammasome. NLRP3 oligomerizes and assembles with Apoptosis-associated Speck-like protein containing a caspase activation and recruitment domain (CARD) (ASC) to form an active inflammasome leading to caspase-1 activation, which in turn cleaves pro–IL-1β into mature IL-1β that is released from the cell5. NLRP3 inflammasome can be activated by different stimuli including sterile molecules such as extracellular adenosine triphosphate (ATP) or nanoparticles (for review see ref. 6). In macrophages, a decrease in extracellular osmolarity (hypotonic challenge) and the subsequent regulatory volume decrease (RVD) phenomenon also leads to NLRP3 inflammasome activation7,8. In mammal cells, the RVD mechanism counteracts the decrease in extracellular osmolarity by a compensatory mechanism leading to the loss of K+/Cl− ions and osmotically obliged water. This massive loss of osmolytes results from the simultaneous activation of potassium channels and volume-regulated anion channels (VRACs). The ubiquitously expressed leucine-rich repeat-containing 8 (LRRC8) anion channel family has been identified as the main molecular component of the VRAC9,10. These channels are activated during low osmolarity exposure and trigger the RVD process by extruding Cl− and osmolytes11, thus allowing for recovery of normal cell volume. The LRRC8 family contains 5 subunits, LRRC8A to E, which form heteromeric channels12,13,14. The LRRC8A subunit is the mandatory subunit to form functional channels10. Recently, in macrophages, LRRC8 channels were demonstrated to be essential for hypotonic-induced NLRP3 activation and IL-1β secretion15.
In macrophages, MSU and CPP crystals induce NLRP3 activation via several cellular disturbances including reactive oxygen species production, autophagy alterations and metabolism modifications16. Previous works suggested that phagocytosed MSU crystals can be dissolved in phagolysosomes, thereby leading to an intracellular increase in osmolarity and subsequent osmolarity-driven water influx responsible for the decrease in intracellular potassium concentration.
Despite these data, the role of LRRC8A in MSU crystal-induced IL-1β production is unknown. Here we show in humans and in mouse models that cell volume changes play a central role in inflammasome activation upon macrophage exposure to MSU/CPP crystals. Crystal exposure triggers LRRC8 activation and subsequent ATP efflux, both necessary for NLRP3 inflammasome activation via P2Y purinergic receptor and intracellular calcium signalling.
Results
In humans, high IL-1β concentration is associated with decreased synovial fluid (SF) osmolarity during gout and CPP flares
The identification of MSU and CPP crystals on compensated polarized light microscopy of human SF with high cell concentration (>2000/mm3) established the diagnosis of gout and CPP flares, respectively. The diagnosis of osteoarthritis (OA) was based on clinical, SF with low cell concentration (<1000/mm3) and radiography patterns (Supp. Fig. 1a). We excluded OA associated with CPP crystals identified in SF. The osmolarity of SFs was lower during gout (MSU) and chondrocalcinosis (CPP) flares than that for OA (Fig. 1a). Crystal (Cryst.) refers to all values for patients with gout- and CPP-mediated diseases. Levels of IL-1β (Fig. 1b) and IL-8 (Fig. 1c) were higher in MSU and CPP than OA SFs. We found a negative correlation between the osmolarity of SF and concentration of IL-1β and IL-8 (Fig. 1d, e), with IL-1β and IL-8 level increased with decreasing osmolarity. There was also a mild negative correlation between SF osmolarity and SF cell concentration (Sup. Fig. 1b). All these results suggest that SF osmolarity modulates MSU and CPP crystal-induced IL-1β/IL-8 production.
a–c osmolarity (a, n = 15 OA, 10 MSU, 9 CPP) IL-1β level (b, n = 17 OA, 13 MSU, 11 CPP) and IL-8 (c, n = 10 OA, 6 MSU, 8 CPP) level measured in the SF of osteoarthritis (OA), MSU and CPP disease. d, e Correlation between IL-1β (d), IL-8 (e) level and SF osmolarity (n = 25 paired samples). Crystal (Cryst.) refers to all values obtained for gout and CPP- diseases. Values are mean ± SEM. Multiple comparisons by one-way ANOVA (a–c). R2 coefficients were calculated with the Spearman test (d, e).
Microcrystal-induced inflammation depends on osmolarity and cell water flux in mouse model
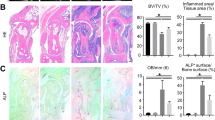
To explore the putative contribution of osmolarity changes in the modulation of the microcrystal-induced inflammation, we used the in vivo mouse air pouch model (Fig. 2a). The air pouch cavity mimics a joint cavity with a lining structure resembling the synovial membrane, formed as early as 6 days after air pouch creation17. Crystal-induced inflammation in this model reproduces the different phases of MSU and CPP flares observed in patients with a self-limited reaction18. Using this model, we artificially increased mouse plasma osmolarity by intraperitoneal injection of mannitol 20% (~1 ml, Pos ~1050 mOsm.l−1), a non-charged osmolyte. After 3 consecutive days of mannitol injection, plasma osmolarity increased to 321 mOsm.l−1. Mice receiving phosphate buffered saline (PBS) showed normal plasma osmolarity, 298 mOsm.l−1 (Fig. 2b). Mannitol treatment strongly reduced microcrystal-induced inflammation (with both MSU and CPP crystals), as evidenced by the decrease in air pouch membrane inflammation score, number of cells collected and IL-1β level measured in air pouch lavage (Fig. 2c–f).
a Air pouch was created by injection of 3 ml sterile air in subcutaneous dorsal area on days 0 and 3. On day 6, sterile MSU or CPP crystals (1 mg in 1 ml PBS) or PBS were injected into the air pouch and inflammation was assessed 6 h later by quantifying cytokine levels and cell infiltration in the lavage solution. b Plasma osmolarity (in mOsm.l−1) measured in mice receiving intraperitoneal injection of PBS (isotonic, 300 mOsm.l−1, n = 11 mice) or 1 ml mannitol 20 % (hypertonic 1050 mOsm.l−1, n = 10 mice) every day from day 4 to 6. c, d Representative micrographs (c, HE staining) and semi-quantitative inflammation score (d) of air pouch membranes obtained from control (n = 4) and mannitol-treated mice receiving PBS, or MSU or CPP crystal injection (n = 8 mice/group). Scale bar, 40 μm. e, f cell infiltration numbers (e) (n = 8 mice PBS, 6 mannitol, 7 MSU and MSU+mannitol, 8 CPP and CPP+mannitol) and IL-1β concentrations (f, ELISA quantification) (n = 8 mice in PBS, MSU, CPP and CPP+mannitol, 6 in mannitol, 7 MSU+mannitol) measured in the air pouch lavage. g, h Representative micrographs (g) and semi-quantitative inflammation score (h) of the air pouch membranes obtained from control and intraperitoneal mercury (HgCl2)-treated mice receiving PBS, or MSU or CPP crystal injection (n = 4 mice in HgCl2, 9 PBS, 10 in MSU and CPP crystal groups). Scale bar, 20 μm. i, j Cell infiltration numbers (i) and IL-1β level (j) measured in air-pouch lavage collected from the mouse groups treated or not with mercury (n = 10 mice/per group). Values are mean ± SEM. Two-tailed unpaired t-test (b), two-way ANOVA with Sidak’s multiple comparisons test (c–j).
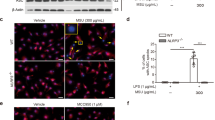
To balance osmolarity challenge and cellular swelling, cells facilitate water exchange through aquaporin channels. Therefore, we used mercury chloride (IP injection, HgCl2, 10 µg/g) to block aquaporin channels19. Aquaporin inhibition completely abolished inflammation induced by MSU and CPP crystals by reducing the inflammation score, number of cells and level of IL-1β collected in lavage (Fig. 2g–j). Aquaporin blockade also reversed the ratio of macrophages to neutrophils to the same proportion as observed in PBS-treated mice (supp. Fig. 2a, b, supp Fig. 7a, b).
Next, we confirmed in vitro that aquaporin inhibition (HgCl2) reduced microcrystal-induced IL-1β production in both the monocytic primed THP-1 cell line and primed bone marrow-derived macrophages (BMDMs) (supp. Fig. 2c, d). In THP-1 cells, HgCl2 treatment also prevented microcrystal-induced speck formation by ASC (supp. Fig. 2e) and NF-κB activation (supp. Fig. 2f). We also found reduced release of 2 cytokines independent of NLRP3 activation, IL-8 and tumor necrosis factor α (TNF) (supp. Fig. 2g, h). These results support that the anti-inflammatory effects of HgCl2 were both inflammasome-dependent and -independent. To determine whether crystal-induced NF-κB activation is independent of NLRP3, we treated THP-1 cells with the NLRP3 inflammasome inhibitor MCC950 and assessed NF-κB activity and IL-1β production under crystal stimulation. MCC950 did not affect NF-κB activation while effectively inhibited IL-1β production (supp. Fig. 2i, j). These findings suggest that NF-κB activation is independent of NLRP3 inflammasome activation and that NF-κB activation by crystals is not a paracrine effect of IL-1β secretion induced by crystals.
Cell volume changes and LRRC8 channels mediate NLRP3 inflammasome activation and IL-1β secretion during crystal exposure
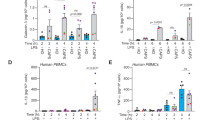
We next deciphered the mechanisms underlying the effect of osmolarity on crystal-induced IL-1β inflammation in monocytic THP-1 cells and the contribution of the osmo-sensitive LRRC8 channels in this process. LRRC8 are activated by a decrease in extracellular osmolarity (hypotonicity, <300 mOsm.l−1) and conversely inhibited by an increase in extracellular osmolarity (hypertonicity; >300 mOsm.l−1). As a result, a hypertonic solution obtained by adding 100 mOsm.l−1 of mannitol to isotonic PBS medium (Mannitol, osmolarity increased to ~400 mOsm.l−1 without modification of ion concentrations) greatly decreased MSU and CPP crystal-induced IL-1β production (200 µg/ml, 3 h, Fig. 3a). Similarly, the addition of DCPIB (20 µM) to isotonic PBS medium, a known and specific inhibitor of LRRC8 channels20,21, inhibited the production of IL-1β induced by both MSU and CPP crystals (Fig. 3b), which suggests that crystal exposure activated LRRC8 channels. To ensure that adding crystals (MSU or CPP) did not alter the osmotic pressures of the bath solutions, we measured the osmolarity after a 6-hours incubation with or without THP-1 cells. The results showed no change in osmolarity across hypotonic, isotonic, or hypertonic solutions, regardless of crystal addition or cell presence (supp. Fig. 3a).
a, b IL−1β release in supernatants of primed THP−1 cells stimulated with crystals in isotonic (PBS) or hypertonic solutions (mannitol) (a, n = 4 independent experiments) and in isotonic with/without DCPIB (b, n = 6 independent experiments). c, d Cellular volume change measured in THP−1 cells induced by PBS alone or PBS containing MSU (c) or CPP (d) with/without DCPIB, n = 4 independent experiments. e, f LRRC8A protein expression level in control and LRRC8A-KD THP-1 cells (n = 5 independent preparations). g Chloride whole-cell currents in control (upper panel) and LRRC8A-KD (lower panel) THP-1 cells recorded successively with isotonic and hypotonic solution (5 min) and in the presence of DCPIB. Pipette and bath solutions contained NMDG-Cl. The corresponding I/V curves calculated with isotonic solution (black), hypotonic solution (green) and with DCPIB (light green) are depicted on the right. (n = 22 and 9 independent recordings for control and LRRC8A-KD THP-1 cells, respectively). h, i Micrographs illustrating the ASC speck formation (h) and percentage of cells containing ASC speck (i, n = 4 independent experiments) detected in control and LRRC8A-KD cells maintained in isotonic or hypotonic solutions. Red arrows indicate the ASC specks. Scale bar 30 µm. j Western blot analysis of NLRP3 inflammasome components and supernatant IL-1β release in control and LRRC8A-KD THP-1 cells after exposure to a hypotonic solution or a Poly(dA:dT) stimulation. k Quantification of IL-1β release in supernatant of control and LRRC8A-KD cells after hypotonic exposure (n = 3 independent experiments). l, m IL-1β release (l) and percentage of cells containing ASC speck (m) measured in control and LRRC8A-KD cells after MSU and CPP exposure (n = 6 and 3 independent experiments). n Cellular volume change measured in control and LRRC8A-KD cells after MSU crystal exposure. (n = 9 independent experiments). All values are mean ± SEM. Two-tailed unpaired t-test (e, f, k), two-way ANOVA, Sidak’s multiple comparisons test (a, b, i, l, m).
Measurement of cell volume changes (using calcein self-quenching fluorescence method20) showed that the addition of MSU or CPP crystals induced rapid cell swelling followed by decreased cell volume that was faster with CPP than MSU crystals (Fig. 3c, d), whereas under iso-osmotic conditions and in the absence of crystals, the cell size remained constant and stable for 120 min (Fig. 3c). As expected, DCPIB (20 µM) treatment did not prevent the cell swelling but significantly reduced the cell shrinkage (Fig. 3c, d and supp. Fig. 3b, c). DCPIB also greatly decreased MSU and CPP crystal-induced NF-κB activation as well as TNF production and ASC speck formation (supp. Fig. 3d–f).
All these results support a contribution of LRRC8 in microcrystal-induced inflammation. To test this hypothesis, we stably knocked down the expression of LRRC8A, the mandatory subunit of VRAC channels10, in THP-1 cells by using a lentiviral-based shRNA strategy, silencing LRRC8A expression by more than 80% as compared with parental control cells (Fig. 3e, f). Patch-clamp whole-cell recordings performed using NMDGCl pipette and bath solutions (Fig. 3g) revealed that in LRRC8A-KD cells, the volume-activated chloride currents evoked by hypotonic conditions (220 mOsm.l−1) were decreased by 85% as compared with control cells (current densities measured at -100 mV were -64 ± 13 pA/pF and -9.7 ± 1 pA/pF, for control and LRRC8A-KD, respectively). DCPIB (20 µM) treatment completely abolished the hypotonic-activated chloride currents in both control and LRRC8A-KD cells. Cell volume measurement confirmed that RVD evoked by hypotonic challenge was impaired in LRRC8A-KD cells (supp. Fig. 3g). Hypotonicity-induced ASC speck formation (Fig. 3h, i, 100 mOsm.l−1, 1 h), as well as hypotonic-mediated IL-1β processing and release (Fig. 3j, k, 100 mOsm.l−1, 1 h), were also decreased in LRRC8A-KD cells. Importantly, LRRC8A silencing did not affect the cellular expression of ASC, NLRP3 and pro-IL-1β proteins (Fig. 3j) nor the activation of AIM2 inflammasome induced by Poly(dA:dT). Similarly, in BMDMs, pharmacological inhibition of LRRC8 (with DCPIB) impaired volume-activated chloride currents, cell volume change and IL-1β release after hypotonic challenge (supp. Fig. 4a–c). Expressions of Lrrc8 and Tmem16 subunits were not modified by LPS treatment of BMDMs (supp. Fig. 4a–d), while knockdown of Lrrc8A significantly decreased IL-1β release after hypotonic challenge without affecting the expression of the other Lrrc8 and Tmem16f subunits (supp. Fig. 4d–f).
Finally, in THP-1 cells, we explored the contribution of the LRRC8 channels in crystal-induced NLRP3 inflammasome activation. After MSU or CPP crystal exposure, both IL-1β release (Fig. 3l), number of ASC speck-positive cells (Fig. 3m) and RVD (Fig. 3n and supp. Fig. 5a–c) were strongly impaired in LRRC8A-KD cells. These data support that NLRP3 inflammasome activation and IL-1β secretion during crystal exposure require LRRC8 anion channels.
Lrrc8A ablation in monocyte/macrophage lineage prevents microcrystal-induced inflammation in vivo and in vitro
To further investigate the implication of LRRC8 channels in microcrystal-induced inflammation, Lrrc8A was specifically invalidated in the monocyte/macrophage lineage in Cx3Cr1CreERT2/Lrrc8Afl/fl mice22,23. This conditional model contains a Cre recombinase fused to a mutant estrogen ligand binding domain (CreERT2) that is activated by the estrogen antagonist tamoxifen. Upon tamoxifen injection, Cx3Cr1-expressing cells (i.e., the monocyte/macrophage lineage) invalidate Lrrc8A expression. In this model microcrystal-evoked inflammation was induced in the air pouch model after tamoxifen treatment. FACs analysis of air pouch lavage showed that LRRC8A expression was lower in macrophages from tamoxifen-treated Cx3Cr1CreERT2/Lrrc8Afl/fl mice (hereafter Lrrc8A∆Mo mice) than tamoxifen-treated Cx3Cr1CreERT2 mice (hereafter Lrrc8Acont mice) (Fig. 4a). Inflammation induced by MSU and CPP microcrystals in the air pouch was greatly inhibited in Lrrc8A∆Mo mice as compared with Lrrc8Acont mice as demonstrated by reduced IL-1β level, cell infiltration and inflammation score (Fig. 4b–g). Finally, in BMDMs isolated from Cx3Cr1CreERT2/Lrrc8Afl/fl mice, tamoxifen treatment decreased LRRC8A expression (Fig. 4h) and IL-1β production induced by MSU and CPP crystal exposure (Fig. 4i). These results identified the LRRC8A subunit as a major actor in MSU and CPP crystal-induced inflammation.
a Percentage of macrophages expressing LRRC8A assessed by FACs (left, representative expression of each group; middle and right, mean ± SEM). Cells collected from air pouch lavage from Lrrc8cont and Lrrc8∆Mo mice were stained with antibody against F4/80 and LRRC8A (n = 10 Lrrc8cont, and n = 14 Lrrc8∆Mo mice). b–e Lrrc8A deletion in macrophage linage reduced inflammation in air pouch model: IL-1β level quantified by ELISA (b, n = 7/9/10 WT mice PBS/MSU/CPP, 8/10/11 Lrrc8∆Mo PBS/MSU/CPP); cell infiltration number (c, n = 7/10/14 WT mice PBS/MSU/CPP, 8/20/16 Lrrc8∆Mo PBS/MSU/CPP); percentage of macrophages (d) and neutrophils (e) assessed by FACs measured in air pouch lavage (n = 7/9/6 WT mice PBS/MSU/CPP, 7/9/9 Lrrc8∆Mo PBS/MSU/CPP). f Representative air pouch membrane histology after HE staining and (g) air pouch membrane semi-quantitative inflammation score (n = 8/9/8 WT mice PBS/MSU/CPP, 7/9/14 Lrrc8∆Mo PBS/MSU/CPP). Scale bar, 20 μm. h, i Lrrc8A deletion in BMDMs reduced LRRC8 expression (h) and IL-1β production (i), quantified by FACs and ELISA, respectively, in supernatant of BMDM isolated from Cx3Cr1CreERT2 or Cx3Cr1CreERT2/Lrrc8Afl/fl mice, treated by tamoxifen from day 1 to 21, then stimulated with MSU or CPP (200 µg/ml, 6 h, n = 5 and 6 independent experiments). Values are mean ± SEM. Two-tailed unpaired t-test (a, h), two-way ANOVA, Sidak’s multiple comparisons test (b–g, i).
Hypotonic and crystal activation of LRRC8 mediates ATP release by macrophages
We next investigated the mechanisms by which LRRC8 can regulate NLRP3 inflammasome activation. We focused on ATP signaling because purinergic receptors have been found involved in crystal-evoked inflammation24. Control THP-1 cells exposed to a hypotonic challenge (100 mOsm.l−1) showed a significant release of ATP in the supernatant (15 min, Fig. 5a) as compared to isotonic condition (300 mOsm.l−1). This ATP release was strongly inhibited by DCPIB (20 µM) or in LRRC8A-KD THP-1 cells, which suggests a major role of LRRC8 in hypotonic-mediated ATP release (Fig. 5a). To temporally characterize this ATP release in the THP-1 cell population, we used a recently developed ATP fluorescent biosensor called PNG625 (Fig. 5b). We first confirmed that this biosensor was functional. THP-1 cells expressing PNG6-P2A-Scarlet sensed extracellular ATP dose-dependently, whereas those expressing PKG6, a dead mutant of the sensor, did not show any fluorescence upon ATP stimulation (supp. Fig. 5d, e). In THP-1 cells expressing PNG6-P2A-Scarlet hypotonic challenge (160 mOsm.l−1) induced a transient and massive release of ATP that peaked at ~10 min and decreased to the basal level within 20 min (Fig. 5c). DCPIB (20 µM) or LRRC8A-KD abolished ATP-evoked fluorescence without affecting biosensor activation, as assessed by a final stimulation with 10 µM ATP (Fig. 5c, supp. Fig. 5f). Because LRRC8 channels are permeant to small anionic molecules, we evaluated whether they might be permeable to ATP. To that aim, we used patch-clamp whole-cell recording in THP-1 cells with Na2ATP in both bath and pipette solutions. In these conditions, exposure to hypotonic solution rapidly (<5 min; 260 mOsml.l−1) induced ATP-driven currents at negative and positive holding potentials, corresponding to an efflux and influx of negative charges, respectively (Fig. 5d, e). The mean ATP current densities evoked by the hypotonic condition were -20.0 ± 2.0 pA/pF (n = 6) and −8.1 ± 2.1 pA/pF (measured at −100 mV, Fig. 5f) in the absence or presence of DCPIB (20 µM), respectively, supporting the idea that LRRC8 channels exhibit ATP conductance.
a ATP levels in supernatants of control and LRRC8A-KD THP-1 cells under isotonic or hypotonic solutions ± DCPIB (n = 6–15 from 6 independent experiments). b Illustration of the PNG6 biosensor fluorescence change upon extracellular ATP binding. c Kinetics of ATP release evoked by hypotonic solution in control and LRRC8A-KD THP-1 cells expressing the PNG6 biosensor with/without DCPIB. Maximum fluorescence was evoked by ATP application at the end of the recordings (n = 5 recordings from 3 independent experiments). d–f ATP whole-cell currents in control THP-1 cells in isotonic and hypotonic ATP bath solutions with/without DCPIB (d). I/V curves (e) and mean currents at -100 mV (f) in isotonic (black) and hypotonic solution (green, 5 min) and with DCPIB (light green). n = 6 independent recordings. g Lentiviral construction expressing PNG6-P2A-Scarlet and images showing ATP release (green PNG6) and surface/volume decrease (red mScarlet) in control THP-1 cells under hypotonic solution. Yellow and blue arrows identified cells with different time responses. Scale bar 20 μm. n = 3 independent experiments. h Single-cell video recording of surface change and ATP release in control and LRRC8A-KD THP-1 cells expressing PNG6. Colored lines represent individual cells; black lines show mean surface change and ATP-evoked fluorescence. i Surface change (upper) and ATP release (lower) in 3 cells during a hypotonic challenge. j Time correlation between ATP release event and RVD induction (n = 32 cells from 2 independent experiments). k ATP levels in supernatants of control and LRRC8A-KD THP-1 cells exposed to MSU or CPP crystals with/without DCPIB. n = 8–23 from 8 independent experiments (control THP-1) and n = 9–21 from 7 independent experiments (LRRC8A-KD cells). Values are mean ± SEM. One-way ANOVA (f), 2-way ANOVA, Sidak’s multiple comparisons test (a, k). Spearman correlation test (j).
We next investigated at the single cell level a possible relation between hypotonic-evoked ATP release and RVD. Using time-lapse microscopy on THP-1 cells stably expressing PNG6-P2A-Scarlet, we simultaneously recorded ATP release and cell surface changes during hypotonic challenge (100 mOsm.l−1) by measuring the Scarlet signal as a readout of cellular volume (Fig. 5g). Most of the cells initiated their RVD phase and released ATP during the first 10 min after the hypotonic challenge (Fig. 5h). Importantly, we observed only one event of ATP release per cell during the recording period. Genetic silencing of LRRC8A (LRRC8A-KD) completely prevented ATP release as well as RVD at the single cell level (Fig. 5h). We next analyzed the occurrence of the ATP release event and the initiation of RVD. The ATP release event occurred only during the swelling phase and concomitantly with the initiation of the RVD phase (Fig. 5i). We found a high correlation between the 2 events, with a correlation coefficient > 0.97 (Fig. 5j). These results suggest that ATP release via LRRC8 is the triggering event leading to RVD.
Finally, we tested the putative contribution of LRRC8 in ATP release on crystal exposure in THP-1 cells. ATP release in the supernatants was significantly increased after MSU or CPP crystals exposure (200 µg/ml, 3 h) and was blunted by DCPIB or in LRRC8A-KD THP-1 cells (Fig. 5k). These results support that MSU and CPP crystals trigger LRRC8-dependent extracellular ATP release.
LRRC8-evoked ATP release governs the RVD and NLRP3 inflammasome activation via P2YR signaling
We next analyzed the contribution of purinergic receptor signaling to RVD and inflammasome activation in THP-1 cells. Incubation with 20 units/mL apyrase, an extracellular ATP/ADP-hydrolyzing enzyme, prevented hypotonicity-induced RVD (Fig. 6a) and IL-1β production (Fig. 6b). This effect was dose-dependent and observed with different grades of apyrase (Fig. 6b, c). Using BMDM from P2X7−/− mice we showed that MSU and CPP crystal-evoked IL-1β production was not impaired, ruling out the contribution of P2X7 receptors (supp. Fig. 6). Because P2X receptors are not involved in hypotonicity-evoked inflammasome activation7, we tested a potential contribution of different P2Y receptors to RVD and IL-1β release. The P2Y2 receptor antagonist ARC118925 inhibited RVD to almost the same extent as LRRC8A-KD, whereas antagonists of P2Y1, P2Y6, and P2Y11 had no effect (Fig. 6d). Both P2Y2 and P2Y6 antagonists inhibited hypotonic-induced IL-1β release (Fig. 6e, f), yet without affecting hypotonic-evoked ATP release (supp. Fig. 5g). P2Y1 and P2Y11 antagonists had no effect (Fig. 6f). Accordingly, in THP-1 cells, MSU and CPP crystal-induced IL-1β release was strongly reduced with P2Y2 and P2Y6 antagonist application (Fig. 6g). Finally, using the air pouch model, in vivo inhibition of P2Y6 greatly decreased the MSU and CPP crystal-induced inflammation score, immune cell infiltration and IL-1β release (Fig. 6h–k).
a Monitored surface changes (mScarlet fluorescence) in control THP-1 cells under hypotonic conditions (100 mOsm.l−1) with and without apyrase (A6535, 20U/ml). n = 3 independent experiments with 30-66 cells per experiment. b, c Western blot analysis and quantification of IL-1β release in control THP-1 cells under hypotonic conditions with varying apyrase levels and grades. Values normalized to the hypotonic condition without apyrase, mean ± SEM from 3-5 experiments. d Surface changes in control and LRRC8A-KD THP-1 cells during hypotonic challenge with various P2Y receptor antagonists (10 µM). n = 3 independent experiments with 30-66 cells per experiment. e Western blot analysis of mature IL−1β in supernatants and pro-IL-1β and LRRC8A expression in cell extracts from control and LRRC8A-KD cells under hypotonic conditions with/without P2Y2 antagonist ARC118925 (10 µM). f Quantification of IL-1β release in control THP-1 cells under hypotonic stimulation with/without 10 µM of each antagonist ARC118925 (P2Y2), MRS2500 (P2Y1), MRS2578 (P2Y6), or NF340 (P2Y11). Results normalized to conditions without antagonists, n = 4–5 independent experiments. g ELISA measurement of IL-1β levels in the supernatant of control THP−1 cells exposed to MSU or CPP crystals (200 µg/ml, 3 h) with/without antagonists ARC118925 (10 µM) or MRS2578 (10 µM). n = 4 independent experiments. h, i Micrographs and inflammation scores for air pouch membranes from control and MRS2578-treated mice injected with PBS, MSU, or CPP crystal (n = 8 mice/group). Scale bar, 40 μm. j, k Cell infiltration (j, n = 9 PBS and MSU, 8 CPP, 7 MRS, 8 MRS + MSU/CPP) and IL-1β (k, n = 9 PBS and MSU, 8 CPP, 7 MRS, 9/8 MRS + MSU/CPP) levels in air pouch lavage from mice treated with/without MRS2578. Values are mean ± SEM. Two-tailed unpaired t-test (c, f), two-way ANOVA, Sidak’s multiple comparisons test (g, i–k).
P2YR signaling evokes intracellular calcium increase and IL-1β release
We next investigated the intracellular signaling pathways downstream of P2Y receptor activation, focusing on intracellular calcium, which is involved in NLRP3 inflammasome activation26. To address this, we used single cell time-lapse microscopy on human THP-1 cells stably expressing the cytoplasmic calcium indicator GCaMP6s and the fluorescent reporter mScarlet (Fig. 7a). Our recordings of hypotonicity-evoked intracellular calcium variations revealed that hypotonicity induced a significant burst in intracellular calcium levels, peaking approximately 10 minutes after the reduction of extracellular osmolarity (Fig. 7a). This major calcium increase was abolished by silencing LRRC8A or inhibiting the P2Y2 receptor with 10 µM ARC118925 (Fig. 7a, b). Though, inhibition of P2Y2 by ARC118925, P2Y6 by MRS2578 or NLRP3 by MCC950, did not significantly affect the hypotonic-induced VRAC conductance in control THP-1 cells (Fig. 7c). Together these data support that intracellular calcium signaling evoked by hypotonicity results from VRAC activation and subsequent ATP release and P2YR activation. Single-cell analysis of both intracellular calcium signals and cell surface dynamics indicated that most cells exhibited at least one burst of intracellular calcium concomitant with the initiation of the RVD phase following hypotonicity challenge (Fig. 7d, e), in accordance with ATP release events shown in Fig. 5i. A significant correlation was observed between the time to RVD and the time to maximal calcium peak (R2 > 0.5, p < 0.001, n = 70 paired, Fig. 7f) suggesting a role of intracellular calcium signaling in RVD initiation.
a Video-microscopy recording of intracellular calcium during hypotonic challenge in control and LRRC8A-KD THP-1 cells expressing GCaMP6-P2A-mScarlet, with or without P2Y2 antagonist ARC118925 (10 µM). b Quantification from (a) showing area under the peaks averaged and normalized to control. Data are mean ± SEM from n = 5 experiments for control and LRRC8A-KD, n = 2 experiments for ARC118925. 30–60 cells per experiment. c Chloride currents in control THP-1 cells in isotonic (300 mOsm.l−1) and hypotonic (220 mOsm.l-1) NMDGCl solutions, with/without ARC118925 (10 µM). I/V curves show mean current density under hypotonic conditions with ARC118925, MRS2578, or MCC950 (10 µM). Pipette solution contained NMDGCl. Data are mean ± SEM from 4 to 15 cells, n = 4–15 experiments. d–f Simultaneous intracellular calcium signals and cell surface analysis during hypotonic challenge at single-cell level. d Combined recording of cell surface variation (mScarlet) and intracellular calcium (GCaMP6). Scale bar 20 µm. e Single-cell analysis shows RVD initiation coincides with a burst of intracellular calcium release. f Time correlation between RVD initiation and peak of intracellular calcium signal. d–f: n = 2 experiments, analysis of 70 cells. g Left panel: cartoon of PLC pathway activation by Gq-coupled P2YR leading to intracellular calcium mobilization. Middle and right panels: ELISA results of IL-1β levels in supernatants of control THP-1 cells exposed to MSU or CPP crystals (200 μg/ml, 4 h) with/without PLC inhibitor (U73122, 5 μM) or its inactive analog (U73343, 5 μM). n = 3 independent experiments. Values are mean ± SEM. Two Way ANOVA with Tukey’s multiple comparison post hoc test. Panel g Created with BioRender.com released under a Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International license https://creativecommons.org/licenses/by-nc-nd/4.0/deed.en.
P2Y2 and P2Y6 are Gq-coupled GPCRs that both activate the phospholipase C (PLC) pathway, resulting in Ca2+ release from intracellular stores (Fig. 7g). We found that, in THP-1 cells, U73122, a PLC inhibitor, suppressed MSU and CPP crystal-induced IL-1β release (Fig. 7g), whereas U73343, an inactive analog of U73122, did not. Altogether, these data support that LRRC8A-evoked ATP release promotes autocrine and/or paracrine activation of P2Y2 and P2Y6 receptors, leading to subsequent intracellular calcium signaling and inflammasome activation.
Discussion
In this study, we identified LRRC8 channels and cell volume regulation as central orchestrators of MSU and CPP crystal-induced inflammation in both human and mouse macrophages (Fig. 8). MSU and CPP crystals induce cell swelling followed by a LRRC8A-dependent RVD response, a cellular phenomenon mimicking hypotonic challenge. Crystal-mediated LRRC8 activation leads to ATP release which in turn activates P2Y2 and P2Y6 purinergic receptors and the subsequent activation of PLC-calcium pathway, eventually contributing to NLRP3 inflammasome activation and IL-1β production.
Scheme illustrating that microcrystal-induced production of proinflammatory cytokine IL-1β relies on cell volume regulation. During microcrystal-induced inflammation, both the decrease of synovial fluid osmolarity and the presence of microcrystals induce resident macrophage swelling, which activates the volume regulatory anion channels LRRC8 leading to ATP outflow. ATP in an autocrine and/or paracrine way activates purinergic P2Y2 and P2Y6 receptors that, through a PLC and intracellular calcium-dependent pathways contribute to NLRP3 activation and subsequent pro-IL-1β maturation and release. Created with BioRender.com released under a Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International license https://creativecommons.org/licenses/by-nc-nd/4.0/deed.en.
Osmolarity changes occur in multiple physiological and pathological conditions such as cell proliferation, transepithelial transport, tissue damage, necrosis or inflammatory diseases27,28. Here we reported that SF osmolarities of patients with gout (MSU) and CPP inflammatory flares are decreased as compared to patients suffering from osteoarthritis (OA). Hypotonic SF is also a characteristic of rheumatoid arthritis, a different joint-inflammation disease also associated with high production of inflammatory cytokines29,30. We observed that extracellular osmolarity modulated crystal-induced inflammation (MSU and CPP): hypertonic solution prevents MSU and CPP-mediated IL-1β secretion. Therefore, in patients with gout and CPP inflammatory flares, both reduced SF osmolarity and crystal exposure may be responsible for inflammatory cytokines production. Our present results and those of others support that increasing plasma osmolarity reduces the inflammatory responses encountered during gout and chondrocalcinosis flares, intracerebral hemorrhage or acute distress respiratory syndrome31,32. Here, we had similar observations in rodents by using crystal-evoked inflammation in an air pouch model, and we found that crystal-induced inflammation was reduced by artificially increasing plasma osmolarity with mannitol. The identification of this osmotherapeutic effect paves the way for the development of alternative therapeutic strategies in humans to dampen inflammation, especially in the context of microcrystal-mediated joint inflammation.
In our study, blocking aquaporin channels by HgCl2 completely abolished MSU and CPP crystal-induced inflammation both in vivo and in vitro. Water influx through aquaporins has been proposed to occur following the increase of intracellular osmolarity provoked by intra-lysosomal dissolution of phagocytosed MSU crystals33 and to be responsible for decreased intracellular potassium level and subsequent inflammasome activation. Similarly, aquaporin 1 was identified as contributing to silica-induced lung inflammation34 and aquaporin 3 to NLRP3 inflammasome activation under various inflammatory conditions35. These findings further support the implication of aquaporin and change in cell volume in microcrystal-induced inflammation.
Our data also demonstrate that in the THP-1 cell line, the knockdown of LRRC8A (the mandatory subunit of VRAC) is sufficient to reduce crystal-evoked RVD, ASC speck formation, and IL-1β release. Moreover, conditional inactivation of LRRC8A in macrophages reduced crystal-evoked inflammation in the air pouch mouse model. Cell swelling and LRRC8 activation were previously found responsible for NLRP3 inflammasome activation on macrophage exposure to hypotonic challenge7,15,36. Our results support that LRRC8-mediated NLRP3 activation is not limited to hypotonic conditions but can be triggered by crystal exposure. This observation is consistent with our previous results showing that MSU crystal-induced inflammasome activation is impaired by saline hypertonic solution in vitro7. Our data support that MSU and CPP crystal-activated NLPR3 inflammasome depends on LRRC8 activation and RVD initiation.
Although our results point to altered intracellular osmolarity caused by crystal exposure, we cannot exclude that crystals could activate specific intracellular pathways leading to LRRC8 activation. Nor can we exclude the involvement of additional anion channels in NLRP3 activation. For example, chloride intracellular channel proteins 1-6 (CLICs 1-6) play a major role in LPS-, ATP-, nigericin- and MSU crystal-induced IL-1β production and NLRP3 inflammasome activation37,38. Notably, genetic deletion of CLIC 1, 4, and 5 partially abolished MSU crystal-activated NLPR3 inflammasome37.
MSU, silica or alum crystal exposure induced ATP and IL1-β release in both BMDMs and THP-1 cells24. From pharmacological approaches (carbenoxolone and flufenamic acid), ATP release was thought to be mediated by the connexin/pannexin pathways. Yet these drugs also affect LRRC8 activity10,20. Several studies support the permeability of LRRC8 to chloride ions but also to negatively charged molecules such as glutamate, cGAMP, or glutathione11,39,40,41. By combining several approaches, including electrophysiology and real-time extracellular ATP imaging, we show that LRRC8 channels are also permeant to ATP and that LRRC8 activation evoked by crystal exposure or hypotonic challenge results in ATP release. An osmolarity-dependent permeability to ATP was previously described in oocytes following overexpression of both LRRC8A/8E subunits42. Yet, ATP could be secreted by other mechanisms dependent on LRRC8 expression and activation.
We also provide evidence that LRRC8-evoked ATP release is essential to RVD and NLRP3 inflammasome activation. Implication of purinergic signaling in inflammasome activation has already been reported. For instance, P2X7 receptor is a well-known activator of NLRP3 inflammasome in macrophages43,44 and the contribution of P2X and P2Y receptors in nanoparticle-induced IL-1β release has been reported45. Here, we observed that P2X7 receptor is not involved in MSU and CPP crystal-induced IL-1β production as previously reported by others24. Using apyrase or specific inhibitors of P2Y2 or P2Y6, we showed that impairing purinergic signaling blocked IL-1β release induced by hypotonic challenge or crystal exposure. In the air pouch model, P2Y6 inhibition was sufficient to inhibit MSU and CPP crystal-induced IL-1β release and reduced the inflammation score. These data are in agreement with previous studies showing that administration of MRS2578, a potent P2Y6 inhibitor, affected MSU crystal-induced neutrophil influx and activation in vivo46,47. In this study, we observed that P2Y receptors acted downstream of LRRC8 activation and ATP release. Yet, P2Y2 and P2Y6 seem to act through different pathways. Indeed, P2Y2 antagonist is able to inhibit RVD in the hypotonic condition, whereas P2Y6 has no effect. Involvement of multiple purinergic receptors to crystal-evoked inflammation has already been proposed24. In particular, P2Y6 inhibition blocks phagocytosis of MSU crystals by polymorphonuclear leukocytes and neutrophil extracellular trap46. P2Y2 and P2Y6 receptors, through their coupling with Gq proteins and subsequent activation of phospholipase C (PLC), play crucial roles in calcium mobilization. An intracellular Ca2+ increase has been reported to be essential for the activation of the NLRP3 inflammasome by multiple stimuli26,48,49. Additionally, we found that blocking the PLC pathway inhibited inflammasome activation induced by MSU and CPP crystals. While the exact mechanisms by which increased intracellular calcium leads to inflammasome activation remain to be fully elucidated, it has been proposed that calcium could directly promote NLRP3–ASC association50, causes mitochondrial Ca2+ overload and mitochondrial dysfunction leading to mitochondrial reactive oxygen species (mtROS) production26,51,52. In any case, P2Y2 and P2Y6 are necessary, but other key cellular events, such as K+ and Cl− efflux, are also required to trigger inflammasome activation in coordination with purinergic signaling and calcium mobilization.
Collectively, our findings suggest that osmotherapy targeting the LRRC8 channels or directly inhibiting this osmo-sensitive channels could represent innovative therapeutic strategies to dampen microcrystal-related inflammation.
Methods
Ethics statement, quantification of osmolarity, IL-1β content in patient synovial fluid
Synovial fluid (SF) was collected from patients during the management of gout, CPP deposition disease flares and osteoarthritis joint effusion. Patients gave written informed consent for use of these waste samples for research. Ethical approval was obtained from the French national ethical committee Comité de protection des personnes participant à la recherche (DC 2020–3976). Gout and CPP deposition diagnoses were established by the identification of MSU and CPP crystals, respectively, on SF analysis by compensated polarized microscopy. Osteoarthritis diagnosis was based on clinical and plain radiography signs. We separated inflammatory SF related to microcrystal flare (defined by cell concentration > 2000/mm3 and presence of crystals detected by polarized light microscopy) from mechanical OA synovial fluids (defined by cell concentration <1000/mm3) and we excluded mechanical SFs containing CPP crystals detected by polarized light microscopy. Cell count was performed manually with a Malassez cell counting chamber. When cell count exceeded 500/mm3, May-Grunwald-Giemsa staining was performed, and the cells were classified as erythrocytes, neutrophils, lymphocytes and other types.
SF from patients were centrifuged to eliminate cells, crystals and debris. Supernatants were collected and stored at -80 °C. Osmolarities of the centrifugated SF or of cell culture media were measured by using the freezing point technique (Roebling Osmometer automatik) and IL-1β concentration was quantified with an ELISA commercial kit (Invitrogen).
Ethics statement and mouse air pouch model
This study was approved by the French national ethical committee Le comité d’éthique en expérimentation animale Lariboisière/Villemin n°9 (APAFIS#28637). Mouse studies were carried out in accordance with European Union directives (2010/63/UE) and French ethical laws guidelines (act no. R214; 87-137; Ministère de l’Agriculture) and followed the ARRIVE guidelines (Animal Research: Reporting of In vivo Experiments; http://www.nc3rs.org.uk). Each animal was humanely handled throughout the experiment in accordance with internationally accepted ethical principles for laboratory animal use and care, and all efforts were made to minimize animal suffering.
Male 8-week-old wild-type (WT) C57Bl/6 J mice were provided by the Janvier Lab. An air pouch was created by dorsal subcutaneous injections of 3 ml sterile air on days 0 and 3. On day 6, crystals (MSU or CPP, 1 mg/ml, 1 ml) or phosphate buffered saline (PBS) (1 ml) were injected into the air pouch. To artificially increase plasma osmolarity, we used intraperitoneal (IP) injection of mannitol 20 % (1 ml) for 3 consecutive days before crystal injection. To inhibit aquaporin channels, we treated mice with 10 µg/g mercury chloride (HgCl2; Sigma) by IP injection 30 min before crystal stimulation. Untreated mice received an IP injection of the same volume of saline (PBS). Inflammation was studied 6 h after crystal or PBS stimulation, assessing in air pouch lavages the concentration of inflammatory mediators by using an ELISA commercial kit (IL-1β, Invitrogen) and cell infiltration by manual counting (absolute number) and FACs analysis (quantification of the proportion of neutrophils and macrophages). Air pouch membranes were collected and fixed with paraformaldehyde (PFA) 4%. Tissue inflammation was histologically estimated on collected membranes after paraffin embedding and hematoxylin-eosin (HE) staining by using a semi-quantitative score16.
Conditional deletion of Lrrc8A in monocyte/macrophage lineage in vivo
Male 8-week-old Cx3cr1CreERT2/Lrrc8Aflox/flox C57Bl/6 J mice (kind gift from Pr. T Jentsch) and their control Cx3cr1CreERT2 C57Bl/6 J mice underwent IP injection of 6-OH-tamoxifen (0.1 mg/g, Sigma T5648) for 5 consecutive days to specifically inactivate the Lrrc8A gene in the monocyte/macrophage lineage and were called Lrrc8∆Mo and Lrrc8cont mice, respectively. One day after tamoxifen treatment, the inflammation induced by crystals was assessed with the air pouch model. Deletion of Lrrc8A was controlled by FACs analysis with macrophages isolated from air pouch lavage and bone marrow-derived macrophages (BMDMs).
Histology
To measure the inflammatory infiltrate, mouse air pouches were fixed in PFA 4%, embedded in paraffin, and serial 5 µm-thick sections were obtained. Sections were then stained with HE. Inflammation was assessed with a semi-quantitative score as we previously described16. In brief, the inflammatory score of each membrane was the highest score observed in 10 random sections.
THP-1 cell culture, transduction and differentiation
THP-1 cells were maintained in suspension culture in RPMI 1640 medium (Invitrogen), 10% fetal bovine serum (FBS, Thermo Fisher) and 1% penicillin-streptomycin (PS, 10000 U/mL, Thermo Fisher). Cells were grown in a humidified atmosphere of 95% air/ 5% CO2 at 37 °C in a cell culture incubator. Cells were split 1/10 every 3 to 4 days, then cells at 50% confluence were plated (1 million cells) in a 6-well plate 24 h before lentiviral transduction and transduced with 6 µL virus encoding the PNG6-P2A-Scarlet biosensor25 or GCamp6-P2A-Scarlet mixed with 6 µL LentiBlast solution (3 µL LentiBlast A solution and 3 µL LentiBlast B solution, Ozbiosciences). Lentiviral production was previously described25. One week after transduction, most cells stably expressed the transgene and were cultivated in RPMI 1640 + 10 % FBS + 1 % PS. Differentiation of THP-1 cells in macrophages involved 40-min incubation in media supplemented with phorbol 12-myristate 13-acetate (PMA) 200 nM (Sigma). Treated cells were then seeded in 96-well plates (140,000 cells/well) until imaging experiments, 24 h later for time lapse imaging or 48 h later for plate reader experiments.
For experiments related to cytokine quantification in the supernatant, primed THP-1 cells were seeded in 24-well dishes at 300,000 cells/well and left overnight in complete media. Then, cells were stimulated with sterile MSU or mCPP crystals (200 µg/ml) in isotonic serum-free media (PBS, 300 mOsm.l−1), or hypertonic solution (addition of 100 mM Mannitol, 400 mOsm.l−1) for 3 h, or with hypotonic solution (dilution 1/3 with water, 100 mOsm.l−1) for 60 min as described7. For some experiments, THP-1 cells were pre-incubated with HgCl2 (Sigma M1132), 4-(2-Butyl-6,7-dichloro-2-cyclopentyl-indan-1-on-5-yl) oxobutyric acid (DCPIB; Tocris 82749-70-0), U73122 (Tocris 1268), U73343 (Tocris 4133) and D-mannitol (Sigma, M9647) 30 min before stimulation. Cell culture supernatants were collected and stored at -20 °C until cytokine quantification.
THP-1 knock-down cells (LRRC8A-KD) were generated with lentiviral shRNA (TRCN0000118623 from Sigma, Sequence: CCGGCCGCAACAAGATCGAGAAGATCTCGAGATCTTCTCGATCTTGTTGCGGTTTTTG) and were selected by culturing cells in the presence of puromycin (3 µg/ml) for 7 days.
BMDM isolation, culture and differentiation
Bone-marrow cells were recovered from tibia and femoral bones of 8-week-old mice (WT, homozygous Cx3cr1CreERT2 and Cx3cr1CreERT2/Lrrc8Aflox/flox)22,23 and cultured in 24-well plates at 2 million cells/ml in 10%-L929 conditioned RPMI1640 media supplemented with 10% FBS, HEPES (5 mM), L-glutamine (2 mM), penicillin (100 U/ml), and streptomycin (100 µg/ml) until complete differentiation of BMDMs. To delete Lrrc8A expression, bone-marrow cells isolated from Cx3cr1CreERT2 and Cx3cr1CreERT2/Lrrc8Aflox/flox mice were treated with tamoxifen (Sigma, H7904) on days 1, 3, 4, 7, and 11 and cultured for 21 days. BMDMs were primed overnight with ultrapure lipopolysaccharide (LPS; 20 ng/ml) before stimulation with microcrystals or PBS in serum-free media for 6 h. Supernatants were collected and frozen at -20 °C until cytokine quantification.
Monosodium urate (MSU) and calcium pyrophosphate (CPP) crystals
MSU and monoclinic CPP crystals were synthesized, characterized and sterilized as described16. MSU and mCPP crystal specific surface were 1.4 ± 0.1 m2/g and 2.8 ± 0.1 m2/g, respectively. Both mCPP and MSU were needle-like crystals, and their average length/width were 16.0/1.5 μm and 32.5/2.9 µm, respectively.
Cytokine quantification
Cytokine production in supernatants or air pouch lavage was measured by using ELISA kits for IL-1β, IL-8 and tumor necrosis factor α (TNF) (Invitrogen, Thermo Fisher).
Quantification of NF-KB activation THP−1-Lucia
THP−1 cells containing an NF-κB-inducible Luc reporter construct (THP-1-Lucia NF-κB Cells, Invivogen) were used to assess NF-κB activation. THP-1-Lucia cells were cultured in RPMI 1640 media supplemented with pyruvate 1 mM (Gibco) and the selection antibiotic zeocin (Invivogen).
PMA-primed THP-1-Lucia cells were washed twice and stimulated for 8 h with MSU or CPP crystals (200 µg/mL) or heat-killed Listeria monocytogenes (HKLM, Invivogen: positive control of NF-κB induction) in serum-free medium. Absolute luciferase activity was determined in the supernatants with a Xenius luminometer (SAFAS Monaco) according to the manufacturer’s instructions.
Flow cytometry
A total of 0.1 million cells from air pouch lavages were stained with anti-F4/80-APC (clone REA126; Miltenyi Biotechnology) and anti-Ly6G-PE mouse monoclonal antibodies for 20 min at 4 °C, washed in PBS, and analyzed with the BD FACS Canto II cytometer (BD Bioscience).
Anti-LRRC8A antibody (Sigma SAB2108060) was used to stain 0.1 million pouch infiltrate cells and BMDMs incubated overnight at 1/1000 dilution to determine the efficiency of the knockdown. The expression of LRRC8A was analyzed the following day for 60 min by using a rabbit secondary antibody conjugated to FITC. Data were analyzed by using BDFACS Diva software (BD Biosciences).
Electrophysiological studies
The ruptured whole-cell configuration of the patch-clamp technique was used to assess the chloride and adenosine triphosphate (ATP) current permeabilities of the LRRC8/VRAC. Cell currents and cell capacitances were recorded by using an EPC 10 amplifier (HEKA Elektronik, Germany). Cells were held at −40 mV, and 400-ms pulses from −100 to +100 mV were applied in 20-mV increments. The offset potentials between both electrodes were zeroed before sealing and corrected for liquid junction potentials as described53. Original whole-cell traces were depicted after use of a 3-kHz Bessel filter. Currents were measured 10 ms after the onset pulse, and I/V curves were corrected by the capacitance of the cells (pA/pF, current density). Patch pipettes (3-4 MOhm resistance) were created from borosilicate capillary tubes (1.5-mm OD, 1.1-mm ID, Vitrex, Danemark) by using a two-stage vertical puller (model PP 830, Narishige, Japan).
For chloride current recordings, the pipette solution contained (in mM) 140 NMDGCl, 10 HEPES (pH 7.4, HCl), 5 EGTA and 5 MgATP (~290 mOsm.l−1). For ATP current recordings, the pipette solution contained (in mM) 100 Na2ATP, 5 MgATP, 5 HEPES (pH 7.4, NaOH) and 5 EGTA (~290 mOsm.l−1).
The normal NMDGCl bath solution contained (in mM) 120 NMDGCl, 10 HEPES (pH 7.4, HCl), 1 CaCl2, 1 MgCl2, 80 mannitol (300-310 mOsm.l−1). The normal ATP bath solution contained (in mM) 100 Na2ATP, 5 HEPES (pH 7.4, NaOH), 70 mannitol (320-330 mOsm.l−1). This bath isotonic solution was designed to avoid spontaneous activation of LRRC8/VRAC currents. Hypotonic NMDGCl solution (220 mOsm.l−1) or Na2ATP (260 mOsm.l−1) solution was obtained by removing the mannitol from the normal NMDGCl or ATP bath solution.
Measurements of cell volume change with the calcein self-quenching method
Measurements of changes in cell volume in THP-1 cells or BMDMs involved the calcein fluorescence self-quenching method. Briefly, calcein at high concentration undergoes spontaneous self-quenching, and its fluorescence intensity decreases with increasing intracellular calcein concentration, which fluctuates according to cell volume variations20,54. Macrophages were cultured on poly-lysine-coated 24-well plates for 2 days. Cells were then loaded for 40 min with a solution calcein-AM (Sigma–Aldrich). Cells were then washed 3 times and incubated with an isotonic medium containing (in mM) 145 NaCl, 5 KCl, 1 MgCl2, 0.4 CaCl2, 10 glucose, and 10 HEPES, pH 7.4 (300 mOsm.l−1). After a baseline recording period (10 min), the medium was replaced with hypo-osmotic bath solution (100 mOsm.l−1) or isotonic solution containing MSU or CPP crystals (200 µg/ml). The fluorescence was measured every 60 or 90 s for 120 min by using a plate reader (Ex: 460 nm, Em: 520 nm; Synergy HT, Biotek). Values (representing the cell volume) were normalized to the fluorescence measured at the beginning of the experiment (first 10 min) in isotonic bath solution (F/F0).
Western blot analysis
Cells were washed with PBS and scraped in lysis buffer (20 mM HEPES [pH 7.4], 100 mM NaCl, 5 mM EDTA, 1% TX-100) supplemented with a mixture of protease and phosphatase inhibitors (Thermo Fisher). After 30 min at 4 °C, lysates were centrifuged (15,900 × g, 10 min, 4 °C) and the supernatant was collected and mixed with Sample Buffer (NuPAGE, Thermo Fisher). Cell supernatants were clarified by brief centrifugation, concentrated by using 10-kDa nominal molecular mass cutoff filters (Millipore), then mixed with Sample Buffer, as described above. Proteins were separated on 4–12% Bis-Tris gels (NuPage Novex, Thermo Fisher) and transferred to nitrocellulose membranes (iBlot2, Thermo Fisher). Proteins were detected by using the primary antibodies for IL-1β (Santa Cruz Biotechnology, sc-7884); LRRC8A (Sigma, HPA016811); Apoptosis-associated Speck-like protein containing a caspase activation and recruitment domain (CARD) (ASC; Santa Cruz Biotechnology, sc-22514-R); NLRP3 (Adipogen, AG-20B-0014); tubulin (DSHB clone 12G10); and GAPDH (Cell Signaling, 3683), all diluted at 1/1000), and appropriate HRP-coupled secondary antibodies and Clarity Western ECL substrate (Biorad).
Immunolabelling of ASC speck
A total of 250,000 PMA-primed control and LRRC8A-KD THP-1 cells were plated on glass coverslips; cells were preincubated with PBS, HgCl2 or DCPIB and stimulated with crystals for 3 h or hypotonic solutions for 1 h, then fixed in 4% PFA. The membrane was permeabilized (5 min at room temperature, 0.1% Triton X-100 in PBS), washed 3 times, incubated overnight at 4 °C with ASC antibody (1/200, Santa Cruz Biotechnology, sci22514-R), then revealed with DyLight-550-conjugated anti-IgG secondary antibody (Pierce). DAPI was used as a counterstain for nuclei. ASC speck formation was analysed by confocal microscopy with a 63x magnification lens or an Apotome and 40x magnification lens.
Plate reader imaging for ATP extracellular measurement
At 48 h after differentiation, primed THP-1 cells expressing PNG6 biosensor were seeded in 96-well plates, washed and incubated for 20 min in Flex buffer (HBSS (Thermo Fisher) supplemented with 20 mM HEPES, pH 7.4, 1 mM MgSO4, 3 mM Na2CO3, and 0.4 mM CaCl2) at 37 °C. For hypotonicity experiments, H20 Milli-Q water + 0.4 mM CaCl2 were added to the well to obtain 160 mOsm.l−1 solutions. The osmotic concentration of each solution was verified by using an osmometer. For pharmacological experiments, cells were pre-incubated for 15 min with drugs diluted in FLEX buffer. Fluorescence (λexc/em 485/540) was acquired with an INFINITY 500 (TECAN) plate reader. The recording was performed at 0.15 Hz. All experiments were performed at 37 °C.
Time-lapse imaging
Primed THP-1 cells seeded in 96-well plate were imaged 24 h after differentiation. At this time, the shape of macrophages is round and allows for automatic detection of the cell surface. THP-1 cells were washed and incubated for 20 min in Flex buffer (HBSS supplemented with 20 mM HEPES, pH 7.4, 1 mM MgSO4, 3 mM Na2CO3 and 0.4 mM CaCl2) at 37 °C. For hypotonicity experiments, H20 Milli-Q water + 0.4 mM CaCl2 was added to the well to obtain 100 mOsm solutions. The osmotic concentration of each solution was verified by using an osmometer. For pharmacological experiments, cells were pre-incubated for 15 min with drugs diluted in FLEX buffer. All cells were imaged by using a motorized inverted epifluorescence microscope (ZEISS AXIO OBSERVER 7) equipped with an incubator (5% CO2, 37 °C) and a CMOS Hamamatsu ORCA camera controlled via ZEN software. Acquisitions involved using a 20X objective lens at 2 frames/min (λexc/em 485/540, 530/590). For image analysis, an automatic ROI-based analysis was developed on IMARIS that allowed for simultaneously detecting GCaMP6s signals (λexc/em 485/540) and cell surface (λexc/em 530/590).
RT-qPCR analysis in BMDMs
RNA from BMDMs was extracted by using the Qiagen RNeasy Plus Mini kit (Qiagen, #74136) following the manufacturer’s protocol. Total RNA quality was checked by using the 2100 Bioanalyzer (Agilent); All RNAs had RNA Integrity Numbers (RINs) > 8.0. Reverse transcription of total RNA involved using the iScript kit (BioRad). Quantitative PCR was performed in 384-well plates in a final volume of 10 µl by using SYBR Green dye detection on the LightCycler480 system (Roche-Diagnostic).
Primer sequences were designed by using Primer Express software (Thermo Fisher) or Primer-355 and tested for specificity and efficiency (LightCycler 480, Roche). Expression of selected mouse genes was quantified by the comparative -ΔCt method using Cox6b1 as a normalizing gene. In shRNA-mediated knockdown experiments, gene expression was further normalized to that of control virus-treated cultures and expressed as -ΔΔCt. Primer sequences were as follows:
Lrrc8a: s- GGGTTGAACCATGATTCCGGTGAC as- GAAGACGGCAATCATCAGCATGAC
Lrrc8b: s- ACCTGGATGGCCCACAGGTAATAG as- ATGCTGGTCAACTGGAACCTCTGC
Lrrc8c: s- ACAAGCCATGAGCAGCGAC as- GGAATCATGTTTCTCCGGGC
Lrrc8d: s- ATGGAGGAGTGAAGTCTCCTGTCG as- CTTCCGCAAGGGTAAACATTCCTG
Lrrc8e: s- ACCGTGGCCATGCTCATGATTG as- ATCTTGTCCTGTGTCACCTGGAG
Cftr: s- GCAGCCTTACTTTGAAACTC as- AACAGCAATGAAGAAGATGAC
Tmem16a: s- GGCATATTCCAGAGGAGTCAA as- TCCATGTCAGCTTCACTTTGTC
Tmem16b: s- GCCAGGGATCCATCTTTGT as- CCTGCTTTGATCTCGTACATTTT
Tmem16c: s- GCAGAGAGGCTGAATATCAGGA as- GCATCCTGCCCATTGATT
Tmem16d: s- TGACTGGGATTTGATAGACTGG as- GCTTCAAACTGGGGTCGTAT
Tmem16e: s- TGGAAACATTAAAGAAGCCATTT as- GAGTTTGTCCGAGCTTTTCG
Tmem16f: s- AGGAATGTTTTGCTACAAATGGA as- GTCCAAGGTTTTCCAACACG
Tmem16g: s- GCTCTGTGGTGATCGTGGT as- GGCACGGTACAGGATGATAGA
Tmem16h: s- GGAGGACCAGCCAATCATC as- TGCTCGTGGACAGGGAAC
Tmem16j: s- CGGAAGTCAGGTAGGAGCAC as- ATCCGGAGGCTCTCTTCG
Tmem16k s- TTGTATCCAGGAAAATCATTGTTG as- AAGCTTCTTCAGGGCTTCACT
Cox6b1: s- CTCCGGGACAATCTTTAGGA as-GGGGGCAGTTTTGTAGTTCTT.
Quantification of ATP release in supernatant
WT and LRRC8A-KD THP-1 cells were cultured for 24-48 h after PMA priming. Culture medium was replaced with a isotonic bath solution for 1 h and cells were exposed to isotonic solutions (PBS-like containing 138 mM NaCl, 2.7 mM KCl, 1.4 mM KH2PO4, 8 mM Na2HPO4, 10 mM HEPES, 2 mM MgCl2, 0.4 mM CaCl2, Pos ~300 mOsm.l−1), hypotonic solutions (46 mM NaCl, 2.7 mM KCl, 0.5 mM KH2PO4, 3 mM Na2HPO4, 10 mM HEPES, 2 mM MgCl2, 0.4 mM CaCl2, Pos ~100 mOsm.l−1) or isotonic solutions containing MSU or CPP crystals (200 µg/ml) in the presence or absence of different inhibitors. Supernatants were collected at the end of the experiments (4 °C), centrifuged (10 min, 300 g), and ATP content was evaluated by using luciferine/luciferase ATP assays (ATPlite; PerkinElmer, Waltham, MA) according to the manufacturer’s instructions. The luminescence (LUM) was measured on a microplate reader (Synergy HT, BioTek, US). Values of ATP levels measured in the supernatant were normalized to protein content of each individual cultures by using DC Bio-Rad protein assays.
Data analysis
Graphics and data analysis involved using GraphPad Prism 7.0. Quantitative data are presented as mean ± SEM. After verification of Gaussian distribution and homogenous variance of each group, two-tailed unpaired t test was used to compare two experimental conditions or one-way and two-way ANOVA with Sidak’s multiple comparisons test to compare more than 2 experimental conditions involving 1 or 2 independent variables. For parametric or non-parametric tests, linear correlation was determined by the non-parametric Spearman test. The significance level was set at p < 0.05.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
All data supporting the findings of this study are available in the article and its supplementary files or from the corresponding authors on request. Source data are provided with this paper.
References
Dalbeth, N., Gosling, A. L., Gaffo, A. & Abhishek, A. Gout. Lancet 397, 1843–1855 (2021).
Doherty, M. et al. Efficacy and cost-effectiveness of nurse-led care involving education and engagement of patients and a treat-to-target urate-lowering strategy versus usual care for gout: a randomised controlled trial. Lancet 392, 1403–1412 (2018).
Latourte, A. et al. Tocilizumab in symptomatic calcium pyrophosphate deposition disease: a pilot study. Ann. Rheum. Dis. 79, 1126–1128 (2020).
Martin, W. J., Walton, M. & Harper, J. Resident macrophages initiating and driving inflammation in a monosodium urate monohydrate crystal–induced murine peritoneal model of acute gout. Arthritis Rheumatism 60, 281–289 (2009).
Martinon, F., Pétrilli, V., Mayor, A., Tardivel, A. & Tschopp, J. Gout-associated uric acid crystals activate the NALP3 inflammasome. Nature 440, 237–241 (2006).
Broz, P. & Dixit, V. M. Inflammasomes: mechanism of assembly, regulation and signalling. Nat. Rev. Immunol. 16, 407–420 (2016).
Compan, V. et al. Cell volume regulation modulates NLRP3 inflammasome activation. Immunity 37, 487–500 (2012).
Ip, W. K. E. & Medzhitov, R. Macrophages monitor tissue osmolarity and induce inflammatory response through NLRP3 and NLRC4 inflammasome activation. Nat. Commun. 6, 6931 (2015).
Qiu, Z. et al. SWELL1, a plasma membrane protein, is an essential component of volume-regulated anion channel. Cell 157, 447–458 (2014).
Voss, F. K. et al. Identification of LRRC8 heteromers as an essential component of the volume-regulated anion channel VRAC. Science 344, 634–638 (2014).
Lutter, D., Ullrich, F., Lueck, J. C., Kempa, S. & Jentsch, T. J. Selective transport of neurotransmitters and modulators by distinct volume-regulated LRRC8 anion channels. J. Cell Sci. 130, 1122–1133 (2017).
Syeda, R. et al. LRRC8 proteins form volume-regulated anion channels that sense ionic strength. Cell 164, 499–511 (2016).
Takahashi, H., Yamada, T., Denton, J. S., Strange, K. & Karakas, E. Cryo-EM structures of an LRRC8 chimera with native functional properties reveal heptameric assembly. Elife 12, e82431 (2023).
Liu, H. et al. Structural insights into anion selectivity and activation mechanism of LRRC8 volume-regulated anion channels. Cell Rep. 42, 112926 (2023).
Green, J. P. et al. LRRC8A is essential for hypotonicity-, but not for DAMP-induced NLRP3 inflammasome activation. Elife 9, e59704 (2020).
Renaudin, F. et al. Gout and pseudo-gout-related crystals promote GLUT1-mediated glycolysis that governs NLRP3 and interleukin-1β activation on macrophages. Ann. Rheum. Dis. 79, 1506–1514 (2020).
Edwards, J. C., Sedgwick, A. D. & Willoughby, D. A. The formation of a structure with the features of synovial lining by subcutaneous injection of air: an in vivo tissue culture system. J. Pathol. 134, 147–156 (1981).
Campillo-Gimenez, L. et al. Inflammatory potential of four different phases of calcium pyrophosphate relies on NF-κB Activation and MAPK Pathways. Front Immunol. 9, 2248 (2018).
Savage, D. F. & Stroud, R. M. Structural basis of aquaporin inhibition by mercury. J. Mol. Biol. 368, 607–617 (2007).
Friard, J. et al. Comparative effects of chloride channel inhibitors on LRRC8/VRAC-mediated chloride conductance. Front. Pharmacol. 8, 328 (2017).
Decher, N. et al. DCPIB is a novel selective blocker of I(Cl,swell) and prevents swelling-induced shortening of guinea-pig atrial action potential duration. Br. J. Pharm. 134, 1467–1479 (2001).
Lück, J. C., Puchkov, D., Ullrich, F. & Jentsch, T. J. LRRC8/VRAC anion channels are required for late stages of spermatid development in mice. J. Biol. Chem. 293, 11796–11808 (2018).
Stuhlmann, T., Planells-Cases, R. & Jentsch, T. J. LRRC8/VRAC anion channels enhance β-cell glucose sensing and insulin secretion. Nat. Commun. 9, 1974 (2018).
Riteau, N. et al. ATP release and purinergic signaling: a common pathway for particle-mediated inflammasome activation. Cell Death Dis. 3, e403 (2012).
Ollivier, M. et al. P2X-GCaMPs as versatile tools for imaging extracellular ATP signaling. eNeuro 8, ENEURO.0185-20.2020 (2021).
Murakami, T. et al. Critical role for calcium mobilization in activation of the NLRP3 inflammasome. Proc. Natl Acad. Sci. 109, 11282–11287 (2012).
Hoffmann, E. K., Lambert, I. H. & Pedersen, S. F. Physiology of cell volume regulation in vertebrates. Physiol. Rev. 89, 193–277 (2009).
Jentsch, T. J. VRACs and other ion channels and transporters in the regulation of cell volume and beyond. Nat. Rev. Mol. Cell Biol. 17, 293–307 (2016).
Shanfield, S., Campbell, P., Baumgarten, M., Bloebaum, R. & Sarmiento, A. Synovial fluid osmolality in osteoarthritis and rheumatoid arthritis. Clin. Orthop. Relat. Res. 235, 289–295 (1988).
Bertram, K. L. & Krawetz, R. J. Osmolarity regulates chondrogenic differentiation potential of synovial fluid derived mesenchymal progenitor cells. Biochem Biophys. Res Commun. 422, 455–461 (2012).
Petroni, R. C. et al. Hypertonic saline (NaCl 7.5%) reduces lps-induced acute lung injury in rats. Inflammation 38, 2026–2035 (2015).
Schreibman, D. L. et al. Mannitol and hypertonic saline reduce swelling and modulate inflammatory markers in a rat model of intracerebral hemorrhage. Neurocrit Care 29, 253–263 (2018).
Schorn, C. et al. Sodium overload and water influx activate the NALP3 inflammasome. J. Biol. Chem. 286, 35–41 (2011).
Rabolli, V. et al. Critical role of aquaporins in interleukin 1β (IL-1β)-induced inflammation. J. Biol. Chem. 289, 13937–13947 (2014).
da Silva, I. V. et al. Aquaporin-3 is involved in NLRP3-inflammasome activation contributing to the setting of inflammatory response. Cell Mol. Life Sci. 78, 3073–3085 (2021).
Burow, P., Klapperstück, M. & Markwardt, F. Activation of ATP secretion via volume-regulated anion channels by sphingosine-1-phosphate in RAW macrophages. Pflug. Arch. 467, 1215–1226 (2015).
Tang, T. et al. CLICs-dependent chloride efflux is an essential and proximal upstream event for NLRP3 inflammasome activation. Nat. Commun. 8, 202 (2017).
Domingo-Fernández, R., Coll, R. C., Kearney, J., Breit, S. & O’Neill, L. A. J. The intracellular chloride channel proteins CLIC1 and CLIC4 induce IL-1β transcription and activate the NLRP3 inflammasome. J. Biol. Chem. 292, 12077–12087 (2017).
Friard, J. et al. LRRC8/VRAC channels exhibit a noncanonical permeability to glutathione, which modulates epithelial-to-mesenchymal transition (EMT). Cell Death Dis 10, 925 (2019).
Zhou, C. et al. Transfer of cGAMP into bystander cells via LRRC8 volume-regulated anion channels augments sting-mediated interferon responses and anti-viral immunity. Immunity 52, 767–781.e6 (2020).
Hyzinski-García, M. C., Rudkouskaya, A. & Mongin, A. A. LRRC8A protein is indispensable for swelling-activated and ATP-induced release of excitatory amino acids in rat astrocytes. J. Physiol. 592, 4855–4862 (2014).
Gaitán-Peñas, H. et al. Investigation of LRRC8-mediated volume-regulated anion currents in xenopus oocytes. Biophys. J. 111, 1429–1443 (2016).
Ferrari, D. et al. Extracellular ATP triggers IL-1 beta release by activating the purinergic P2Z receptor of human macrophages. J. Immunol. 159, 1451–1458 (1997).
Di Virgilio, F. Liaisons dangereuses: P2X7 and the inflammasome. Trends Pharmacol. Sci. 28, 465–472 (2007).
Baron, L. et al. The NLRP3 inflammasome is activated by nanoparticles through ATP, ADP and adenosine. Cell Death Dis. 6, e1629 (2015).
Sil, P. et al. P2Y6 receptor antagonist MRS2578 inhibits neutrophil activation and aggregated neutrophil extracellular trap formation induced by gout-associated monosodium urate crystals. J. Immunol. 198, 428–442 (2017).
Uratsuji, H. et al. P2Y6 receptor signaling pathway mediates inflammatory responses induced by monosodium urate crystals. J. Immunol. 188, 436–444 (2012).
Lee, G.-S. et al. The calcium-sensing receptor regulates the NLRP3 inflammasome through Ca2+ and cAMP. Nature 492, 123–127 (2012).
Triantafilou, K., Hughes, T. R., Triantafilou, M. & Morgan, B. P. The complement membrane attack complex triggers intracellular Ca2+ fluxes leading to NLRP3 inflammasome activation. J. Cell Sci. 126, 2903–2913 (2013).
Gong, T., Yang, Y., Jin, T., Jiang, W. & Zhou, R. Orchestration of NLRP3 inflammasome activation by ion fluxes. Trends Immunol. 39, 393–406 (2018).
Yaron, J. R. et al. K+ regulates Ca2+ to drive inflammasome signaling: dynamic visualization of ion flux in live cells. Cell Death Dis. 6, e1954–e1954 (2015).
Camello-Almaraz, C., Gomez-Pinilla, P. J., Pozo, M. J. & Camello, P. J. Mitochondrial reactive oxygen species and Ca2+ signaling. Am. J. Physiol.-Cell Physiol. 291, C1082–C1088 (2006).
Duranton, C., Huber, S. M. & Lang, F. Oxidation induces a Cl(-)-dependent cation conductance in human red blood cells. J. Physiol. 539, 847–855 (2002).
Capó-Aponte, J. E., Iserovich, P. & Reinach, P. S. Characterization of regulatory volume behavior by fluorescence quenching in human corneal epithelial cells. J. Membr. Biol. 207, 11–22 (2005).
Untergasser, A. et al. Primer3–new capabilities and interfaces. Nucleic Acids Res. 40, e115 (2012).
Acknowledgements
We thank Pr. T.J. Jentsch (Leibniz-Forschungsinstitut für Molekulare Pharmakologie and Max-Delbrück-Centrum für Molekulare Medizin, Berlin, Germany) for providing the Cx3cr1CreERT2 and Cx3cr1CreERT2/Lrrc8Aflox/flox mice22,23 and his constructive comments during the writing of the manuscript. We thank Pr. P. Bobé (Université Paris-Saclay, Paris, France) for providing P2X7-/- BMDM. This work was supported by the Laboratory of Excellence: Ion Channel Science and Therapeutics (LabEx ICST, FR), and by grants from the French society of rheumatology (Société française de rhumatologie), from Novartis (DREAMER), and from the French National Research Agency (ANR) #ANR-22-CE14-0020 (CRISTAL-in). Figure 7 panel g and Fig. 8 were created with BioRender.com, released under a Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International license https://creativecommons.org/licenses/by-nc-nd/4.0/deed.en.
Author information
Authors and Affiliations
Contributions
Collection of the human samples (HK.E., F.L, P.R.); crystal synthesis and characterization (C.C.); measurement of human synovial fluid parameters (TW.C., M.K., C.C., C.D.); in vivo mouse experiments (TW.C., CN.P., HK.E.); histology studies (TW.C., CN.P., M. Z., HK.E.); generation of THP-1 cell line (V.C., N.L., M.O.); patch-clamp whole cell experiments (M.K., J.F., C.D.); measurement of ATP and IL-1β by enzymatic method (I.R., M.K. J.F., C.D.); measurement of IL-1β concentration (M.K., CN.P., TW.C., V.C.); western blot analysis, qPCR experiments, generation of cellular clone (LRRC8AKD) and analysis (H.H., N.L., V.C., M.O.); measurement of ATP release using ATP biosensor and time lapse imaging (M.O., F.R., V.C.); data analysis and interpretation (F.R. HK.E., V.C., C.D.); drafting manuscript (F.R. HK.E., V.C., C.D.); editing manuscript (F.R. HK.E., V.C., C.D.).
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Junya Masumoto and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Chirayath, T.W., Ollivier, M., Kayatekin, M. et al. Activation of osmo-sensitive LRRC8 anion channels in macrophages is important for micro-crystallin joint inflammation. Nat Commun 15, 8179 (2024). https://doi.org/10.1038/s41467-024-52543-8
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-024-52543-8
This article is cited by
-
Updated insights into the molecular networks for NLRP3 inflammasome activation
Cellular & Molecular Immunology (2025)
-
State of the art indicators for imaging purinergic dynamics in vitro and in vivo
Purinergic Signalling (2025)
-
Osmo-sensitive anion channels reduce crystal-induced inflammation
Nature Reviews Rheumatology (2024)