Abstract
Oral corticosteroids represents the most prevalent treatment for idiopathic granulomatous mastitis. Ductal lavage with triamcinolone acetonide and antibiotics followed by observation (DL-OBS) has emerged as a novel strategy, but a comparison of them remains lacking. Here in this multicenter, open-label, non-inferiority, randomized trial (ClinicalTrials.gov identifier: NCT03724903), we assigned 140 patients to oral corticosteroids (N = 71) and DL-OBS (N = 69), stratified by baseline M-score. The primary outcome is complete Clinical Response rate at 1 year. The non-inferiority margin is −15%. The primary outcome is 85.5% in DL-OBS and 87.3% in oral corticosteroids (difference: −1.8%; 95%CI, 13.2 to 9.5; Pnon-inferiority = .01) in intention-to-treat population, and 92.6% vs 98.2% (difference −5.6%; 95%CI −13.4 to 2.2; Pnon-inferiority = .01) in per-protocol population, respectively. The most common (>15%) adverse events were Cushingoid, epigastric pain and arthralgia in oral corticosteroids, and irregular menstruation in DL-OBS, respectively. Here, we report that DL-OBS shows similar efficacy to oral corticosteroids but with better safety profile.
Similar content being viewed by others
Introduction
Idiopathic granulomatous mastitis (IGM), first reported by Kessler in 19721, is a benign inflammatory breast disease with self-limited nature2,3,4, and characterized by the presence of palpable mass, pain, erythema and/or fistula formation5,6. It often requires therapeutic intervention during flares up7,8. However, the underlying mechanism of its pathogenesis remains unclear, thus hindering the development of effective targeted treatment.
Several treatment strategies have been reported, including surgical procedure9,10, oral corticosteroids9,11,12, intralesional steroids injection13,14,15, methotrexate16,17, and bromocriptine12. Among these strategies, oral corticosteroids is the most extensively employed and recommended by some consensuses8,18. However, the well-established fact is that the administration of corticosteroids comes with a range of adverse events (AEs)19,20,21. In light of this, to seek a novel treatment strategy with better safety profiles is imperative.
Studies have shown that observation alone is an alternative option for IGM patients2,3. However, observation alone might be associated with compromised quality of life (QoL) and a longer time to achieve complete clinical response (cCR). Therefore, incorporating approaches that could be able to relief the symptoms might be a potential solution. Ductal lavage was proposed for detecting cellular atypia in women with a high risk of breast cancer at first and then extended to intraductal administration of anti-tumor drugs22,23,24,25. In our previous trial, we observed a rapid resolution of the Visual Analogue Scale (VAS) and Mastitis score (M-score) after completing a two-week medicated ductal lavage regimen containing triamcinolone acetonide and antibiotics, with an excellent safety profile, suggesting it might be a potential method for IGM symptom relief26. Thus, we hypothesized that ductal lavage, as a symptom relief approach, followed by observation, could be a feasible and safe approach for IGM patients.
Herein, we conducted this multicenter, open-label, non-inferiority, randomized trial to compare the efficacy and safety of ducal lavage followed by observation (DL-OBS) and oral corticosteroids for treating IGM. This trial aimed to provide high-quality evidences for clinical-decision making about management of IGM.
Results
Participants
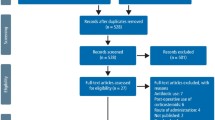
Between April 1, 2019, and April 22, 2023, we enrolled 140 eligible patients at 3 hospitals in China and randomly assigned them to DL-OBS (n = 69) and oral corticosteroids group (n = 71) (Supplementary Table 1). Among them, 129 received at least 1 dosage of treatment allocation as randomized and were included in the safety population (DL-OBS group: n = 67; oral corticosteroids group: n = 62). The intention-to-treat analysis included 140 patients, and the remaining 109 patients who completed the trial without protocol deviations were included in the per-protocol analysis (DL-OBS group: n = 54; oral corticosteroids group: n = 55). The reasons for exclusion are shown in Fig. 1. This study was completed on April 21, 2023.
Abbreviation: IGM, idiopathic granulomatous mastitis; DL-OBS, ductal lavage followed by observation. 1Some patients had more than one reason for screening failure, including: received pertinent treatment previously (n = 41), unsuitable physical or mental condition (n = 31), exclusion based on clinical or pathological evidence (n = 23), history of breast trauma (n = 7), M-score less than 2 (n = 7), bilateral mastitis (n = 6), ineligible age or gender (n = 6) and grade III inverted nipple (n = 4). 2Randomization was stratified by baseline M-score. 3Of those who had protocol violation: received oral corticosteroids (n = 6), received repeated ductal lavage (n = 2), and received surgery treatment (n = 1) in DL-OBS group; received ductal lavage (n = 2), received repeated oral corticosteroids (n = 1), and received surgery treatment (n = 1) in oral corticosteroids group before reaching complete Clinical Response.
Baseline characteristics were well balanced between the two groups (Table 1). The median age was 35 years old (IQR, 32–40), most of them were premenopausal (97.1%) and had a history of lactation (82.9%). The median time since the last lactation was 25.0 (IQR, 14.8–60.0) months, and the median time from onset to visit was 1.0 (IQR, 0.5–2.0) months. At the time of diagnosis, the median mass size was 6.3 (IQR, 4.5–10.0) cm, and less than half of them (30.0%) had lesion (s) located near the nipple-areolar complex. Mild-to-moderate pain caused by IGM, which delineated as VAS under 6 points, was the most frequent symptom (62.1%), slightly less than half of patients (45.7%) had erythema on the surface of the breast, and a minority of them (10.7%) presented with fistula formation. There were 36 (52.2%) and 37 (52.1%) of the patients with M-score\( < \)5 in the DL-OBS and oral corticosteroids group, respectively. The median number of the cannulated ducts in the DL-OBS group was 4 (IQR, 3–6) (Supplementary Table 2), while the median duration of taking corticosteroids was 87 (IQR, 42–137) days in the oral corticosteroids group.
Primary outcome
During 1 year after randomization, the proportions of patients with cCR at any visit follow-up were 59 (85.5%) and 62 (87.3%) in DL-OBS and oral corticosteroids group, respectively, in the ITT population. The absolute risk difference was − 1.8% (95%CI − 13.2 to 9.5), which did not cross the predefined margin of − 15% (Pnoninferiority = .01). The incidence of cCR was higher in the per-protocol population, with a between-group difference of − 5.6% (95%CI − 13.4 to 2.2, Pnoninferiority = .01), which also confirmed the non-inferiority (Table 2). There were no statistically significant interactions between treatment and prespecified subgroups. (Supplementary Fig. 1).
Secondary outcomes
No significant differences were found regarding the predefined secondary efficacy outcomes: median time to first cCR was 32.0 vs 32.5 days (P = 0.07), the treatment failure rate was 14.7% vs 12.3% (difference, 2.4%; [95%CI, − 9.2 to 14.0]; P = 0.88), the relapse rate was 10.1% vs 4.6% (difference, 5.5%; [95%CI, − 3.2 to 14.3]; P = 0.37), and protocol compliance rate was 82.6% vs 74.6% (difference, 8.0%; [95%CI, − 5.5 to 21.5]; P = 0.35) between DL-OBS and oral corticosteroids group (Table 2).
Safety
The incidence of adverse events of any grade was 34 (50.7%) and 57 (91.9%) in DL-OBS and oral corticosteroids, respectively. There was only 1 serious AE in oral corticosteroids and 3 surgical-related AEs in the DL-OBS group (Supplementary Table 2 and Fig. 2). In the initial ductal lavage session, procedure-related pain scores on the VAS reported by patients were distributed as follows: 38 (57.6%), 27 (40.9%), and 1 (1.5%) of them reported scores from 0 to 3, 4 to 6, and 7 to 10 separately. Notably, this discomfort improved gradually. By the fifth session, the distribution had shifted to 49 (77.8%), 14 (22.2%), and 0 (0.0%) (Fig. 3). The most frequent adverse events (> 10%) were Cushingoid (n = 49, 79.0%), epigastric pain (n = 11, 17.7%), arthralgia (n = 10, 16.1%), blurred vision (n = 9, 14.5%) and irregular menstruation (n = 8, 12.9%) in the oral corticosteroids group, and irregular menstruation (n = 31, 46.3%) in the DL-OBS group, respectively (Fig. 2). For patients who experienced Cushingoid symptoms in oral corticosteroids group, 33 (67.4%) of them had symptom alleviation within 6 months, whereas for patients in DL-OBS group who experienced irregular menstruation, 24 (77.4%) of them would be resolved spontaneously within 6 months (Supplementary Table 3). Dose de-escalate in early or discontinuation due to AEs were 2 (3.0%) and 18 (29.0%) in the DL-OBS and oral corticosteroids (Supplementary Table 4).
Abbreviations: DL-OBS, ductal lavage followed by observation. The data were reported as counts (percentage). Counts represent the number of patients who experienced at least one corresponding adverse event during the 1-year follow-up period. Besides, one patient could have more than one adverse event. In the DL-OBS group, there were 6 patients who experienced Cushingoid symptoms, 2 had irregular menstruation, and 1 developed rash after additional oral corticosteroids treatment. In the oral corticosteroids group, 1 patient experienced irregular menstruation following additional ductal lavage treatment, 1 reported headache and alopecia after ductal lavage and anti-tuberculosis treatment, 1 experienced back pain due to personal reasons, and 1 developed rash after discontinuing oral corticosteroids and undergoing surgery. Therefore, these adverse events were considered as having no definite association with the treatments assigned. All adverse events were graded according to the National Cancer Institute Common Terminology Criteria for Adverse Events, version 5.0. Source data are provided as a Source Data file1. Cushingoid included the following symptoms: moon face, buffalo hump obesity, weight gain, edema, hirsutism, skin striation, and acne2. Surgical-related disorders included bleeding, post-bleeding, allergy, nipple ischemia or necrosis, and thrombosis.
Over the total five ductal lavage sessions, the missing records were caused by the following reasons: rejected to receive randomized ductal lavage followed by observation (n = 2), suspicious allergy (no records at all, n = 1; only recorded session 1, n = 1), only received 2 sessions since feeling alleviation of symptoms (n = 1), only received 3 sessions due to personal reason (n = 1), and missing recording of session 2 (n = 1). Source data are provided as a Source Data file.
Exploratory analyses
The distribution of the visit time points at which patients first achieved cCR was also similar between the two groups (Supplementary Fig. 2). Prespecified exploratory analyses about the distribution of patients achieving cCR at different time points and maintaining cCR for various durations presented the non-inferiority also established at 6 months, with a cCR rate of 75.4% in DL-OBS and 74.7% in oral corticosteroids group (risk difference, 0.7%; 95%CI, − 13.6 to 15.1) (Supplementary Table 5). In addition, the explore analyses about inflammatory biomarkers showed that the higher levels of baseline erythrocyte sedimentation rate (ESR) (HR, 0.49 [95%CI, 0.30–0.79]; P < 0.01), C-reactive protein (CRP) (HR, 0.55 [95%CI, 0.34–0.92]; P = 0.02) and neutrophils-lymphocytes ratio (NLR) (HR, 0.64 [95%CI, 0.44-0.92]; P = 0.02) were associated with achieving cCR more difficultly in univariate analysis, while only the levels of NLR still remained significant in multivariate analysis (HR, 0.57 [95%CI, 0.34–0.94]; P = 0.03) (Supplementary Table 6). The baseline levels of ESR, CRP, and NLR were also significantly different in patients who achieved cCR within visits 1 to 2 or not (Fig. 4).
Abbreviations: ESR, erythrocyte sedimentation rate; CRP, C-reactive protein; NLR, neutrophils to lymphocytes ratio; cCR, complete Clinical Response. Box plot comparing (a) ESR, (b) CRP, and (c) NLR levels between patients who achieved their first complete clinical response within Visit 1/2 or after Visit 1/2. The boxplots show the interquartile range (IQR) for the values distribution of each group, with the middle lines of the boxes indicating the median values. The whiskers represent the 1.5\(\times\)IQR, and points beyond the upper or lower bounds of the box represent the outliers. Source data are provided as a Source Data file1. Given the disparities in laboratory testing equipment performance, specific CRP levels were unattainable in some testing sites when they fell within the normal range. Therefore, these results with approximate range were not included in the exploratory analysis (CRP < 5 mg/L: n = 2, CRP < 10 mg/L: n = 2).
Discussion
Recently, some researches have suggested IGM as self-limiting, recommending observation alone2,3,8. However, this strategy might be associated with a longer time to resolution, with an average resolution time of 5 (range 0–20) and 7.4 (range 0–20) months reported by Davis et al. and Bouton et al.2,3. Given its potential impact on quality of life, it is necessary to incorporate some treatment methods, such as pain control and inflammation relief, so as to relieve the symptoms, and, if possible, shorten the time to achieve cCR. We observed that the median time to first cCR was achieved in 32.5 days for oral corticosteroid groups, close to the previously reported average resolution time of 1.5 to 3 months27,28,29. Remarkably, the ductal lavage in the DL-OBS group, lasting only 2 weeks, presented comparable efficacy in symptom-relieving and median time to first cCR (32.0 days). Our findings indicate that the intraductal corticosteroid administration was as effective as its systemic counterpart. However, these findings should be interpreted with caution due to the inclusion of antibiotics in DL-OBS, while not in oral corticosteroids. The role of antibiotics is still under debate, while some evidence suggest its limited benefits for IGM12,30,31, others propose it may aids treatment18,32,33. We incorporated antibiotics into the DL-OBS regimen based on the effectiveness in our prior single-arm study26, and this efficacy might be attributed to Corynebacterium kroppenstedtii, a common skin flora8,34, which has been associated with IGM in several studies35,36,37,38 and known to be sensitive to ceftriaxone39. Nevertheless, neither the role of antibiotics nor that of Corynebacterium kroppenstedtii in IGM has been fully understood, highlighting the need for further investigation.
As for the long-term effective control of IGM, a lower relapse rate was observed in our trial when compared to previous studies (range in 5.0–46.2%)40. We acknowledged the absence of a standardized definition for recurrence at present, and various criteria may lead to diverse recurrence rate outcomes. In our study, we defined local recurrence as the re-emergence of symptoms after achieving cCR, provided that these symptoms surpass a predetermined threshold for a specific duration. This definition is rooted in the understanding that IGM is a self-limiting condition that may exhibit recurrent symptoms over an extended period. However, treatment to alleviate symptoms is only deemed necessary when they reached a certain level of severity. This rationale may differ somewhat from treatment for cancer, where any sign of recurrence mandates immediate therapeutic intervention.
IGM exhibits variable durations previously, with the factors determining its course remaining unclear. Our research suggests that the baseline levels of ESR, CRP, and NLR were significantly higher in patients who achieved cCR after visit 2 and those who experienced fistula or erythema formation. Since ESR, CRP, and NLR have been considered as systemic inflammation markers previously41,42,43, we hypothesized that there are two types of IGM: (1) Localized type with no skin erythema, fistula, and elevated CRP/ESR/NLR; (2) Systemic inflamed type with skin erythema, fistula or elevated CRP/ESR/NLR. The observed associations between the baseline levels of CRP/ESR and the treatment efficacy of two groups also confirmed this hypothesis. This could potentially provide clues for predicting the disease course and treatment efficacy.
Among all the treatment options for IGM, oral corticosteroids is the most widely applied, as recommended by expert consensus and guidelines8,18. However, the optimal dosage in IGM remains uncertain. Some studies suggested high-dose was superior to low-dose44, while others indicate contrary findings45. In this study, we adopted the low-dose regimen recommended by the Chinese consensus24. But even in this low-dose regimen, we still observed a significantly higher incidence of adverse events when compared to DL-OBS, particularly the Cushingoid symptoms, which aligns with the occurrence rate in previous studies using oral corticosteroids46,47. Thus, oral corticosteroids might not be the most appropriate first-line treatment and could be considered as a second-line option after DL-OBS.
Ductal lavage, which was originally designed for sampling ductal epithelium to evaluate the risk of breast cancer, has yielded successful cannulation rates varied from 72%–100% in prior studies48. In this trial, we achieved a 100% successful cannulation rate, which is crucial for therapeutic efficacy. We identified several steps to be of benefit for ensuring successful cannulation: Firstly, selecting the appropriate patients for this procedure, as we excluded those with severe (Grade III49) inverted nipples at the onset. Secondly, using lidocaine for intra-nipple anesthesia, which is also present in our lavage fluids, is vital for dilating the ducts48. Thirdly, encountering resistance during initial cannulation attempts should not be deemed as immediate failure. Continuing to inject a small number of lavage fluids, followed by pauses to allow fluids diffusion and dislodge obstructions within the ducts, helps prevent fluid extravasation and ensures therapeutic efficacy. In most such scenarios, this intermittent lavage approach is advisable according to our clinical practice. Lastly, while histological analysis indicates 10–15 nipple orifices could be cannulated50,51, successful cannulation usually occurs in about half, consistent with other findings52,53. Cannulating as many ducts as possible remains crucial for enhancing success rates.
Regarding the safety profile of DL-OBS, one concern is the tolerability of this procedure. The procedure-related VAS score was consistent with those reported in prior studies on ductal lavage54,55, while there was still an 8.7% (6/69) dropout rate during the ductal lavage treatment period, none of them was attributed to procedure-related pain or discomfort. Besides, the incidence was much lower than that of previous research55. This discrepancy may be ascribed to the distinct application settings of ductal lavage; while previous studies primarily used it for diagnostic purposes amidst various non-invasive and more accurate alternatives, we utilized it therapeutically to alleviate severe IGM symptoms. The comparative advantage in symptom relief might explain the higher adherence to ductal lavage among IGM patients.
Another concern is that we observed an unexpectedly high incidence of irregular menstruation in the DL-OBS group as an adverse event (46.3%) when compared to the corticosteroid group (12.9%), which merits further exploration. We propose two potential explanations: one center on nipple stimulation in the DL-OBS group, which may play a role, as it has been associated with increased levels of oxytocin (OT) and prolactin (PRL), which are known to affect the luteal phase56,57; the other on the differences in the corticosteroids used between the groups, could also be a contributing factor, as Mens et al. noted that intra-articular or periarticular injection of triamcinolone acetonide could lead to irregular menstruation in 50.6% of female patients58. However, this adverse event was rarely reported in patients treated with oral prednisone or methylprednisolone. Besides, local injections of triamcinolone acetonide have been found to induce a transient but significant reduction of estrogen and progesterone levels in females, which could also potentially lead to irregular menstruation59. Encouragingly, most patients with irregular menstruation harbored a resolution within six months in our study and required no medical consultation or intervention. Overall, the DL-OBS regimen is well tolerated and demonstrates a favorable safety profile than oral corticosteroids.
Application of ductal lavage would ensure a favorable benefit-to-risk ratio for IGM patients, however, the diverse incidence of IGM in varied regional and national contexts means that the cost-effectiveness of this method should be considered, particularly given the differences in health insurance systems. The procedure necessitates multiple clinic visits within a two-week span, potentially incurring significant costs in certain countries. In addition, despite its simplicity and ease of operation, it currently lacks other clinical applications. Consequently, it may appear less appealing to physicians in areas with a low incidence of IGM, who would need to surmount a learning curve of this procedure. Under that circumstance, intralesional steroid injections could be considered as an alternative local therapy13,14,15. Despite its potential, this technique is not recommended for cases presenting with abscesses or fistulas60,61, which somewhat limits its applicability. Furthermore, the current evidence for this procedure only stems from retrospective or small-scale studies, necessitating further research to validate its therapeutic effectiveness and safety. We also anticipate upcoming studies that compare ductal lavage with intralesional steroid injections, which could provide further clinical evidence for treating IGM in diverse scenarios.
Our study had several limitations. Firstly, we did not include an observation-only group as a control. When the trial was designed, there was limited evidence supporting the inclusion of such a group. Even now, international expert consensus recommends observation only for patients with mild symptoms8,28,62,63. Therefore, ethical concerns would arise about withholding necessary symptomatic relief, particularly for patients with severe symptoms. In addition, the definition of ‘observation’ varies, with some studies allowing the use of nonsteroidal anti-inflammatory drugs or antibiotics2,3, while others did not4,28,31,62. We acknowledged that the absence of an observation-only group would make it unclear whether oral corticosteroids or DL-OBS offer greater benefits, such as accelerating resolution and reducing the risk of recurrences, compared to observation alone. This represents a major limitation of our study, and prospective trials are needed to address this issue. Secondly, the open-label design might introduce some biases for objectively evaluating the severity of symptoms and adverse events. To reduce such biases, we utilized the M-score system previously reported by our team26, provided a relatively objective assessment of IGM severity and therapeutic effects, arranged all evaluators to undergo standardized training, and blinded them to treatment assignment when evaluating patients, largely ensuring reliable evaluations. Lastly, the M-score system only captures the on-site status of patients during clinic visits, leading to the symptom fluctuations that occur before the appointment might be overlooked. Thus, the development of a patient-reported outcome (PRO) scale is of great importance, allowing patients to record their symptom severity and changes in real-time, also providing a fuller, dynamic view of their condition beyond the clinical environment.
In summary, this multicenter randomized controlled clinical trial demonstrated that DL-OBS is non-inferior to upfront oral corticosteroid therapy in efficacy but with a better safety profile. This research provides a high-quality evidence for selecting the first-line treatment strategy for IGM.
Methods
Study design
This multicenter, open-label, non-inferiority, randomized trial was conducted in accordance with the Declaration of Helsinki at 3 hospitals in China64. The aim of this study is to compare the efficacy and safety between DL-OBS and oral corticosteroids. All patients provided written informed consent before enrollment. The enrollment dates of the first and last patients were April 1, 2019, and April 22, 2022. The study protocol and amendments were approved by the ethics committee at all trial sites: Sun Yat-sen Memorial Hospital of Sun Yat-sen University, Lianjiang People’s Hospital, and Jiang Men Maternity and Child Health Care Hospital. The study protocol has been published previously65, and provided in Supplement 2 along with the statistic analysis plan.
Participants
Females aged between 18–65 years old with IGM that occurred more than 1 month after the cessation of lactation were eligible for inclusion if they were pathologically confirmed, treatment-naïve, in good health condition, had M-score\(\ge\)2, and were willing to sign an informed consent and accept randomized treatment strategy. Exclusion criteria were clinically considered as having grade III inverted nipple49, lactating mastitis, bilateral mastitis, periductal mastitis, breast carcinoma, fungal infection of the breast, history of breast trauma, and foreign body retained in the breast. We also excluded patients with pregnant status, history of known allergy to the medications applied in this study, sepsis or severe inflammation caused by IGM, tuberculosis, and systemic autoimmune disease (systemic lupus erythematosus and rheumatic disorders, etc.). Details about the inclusion and exclusion criteria are provided in the protocol (Supplementary Fig. 2).
The reason why only female patients were included in this study is that IGM primarily occurs in females of reproductive age, with male cases being extremely rare66,67.
Randomization, stratification, and masking
A statistician who had no contact with the participants generated the randomization sequence using the computer. Eligible patients were randomly assigned to receive either DL-OBS or oral corticosteroids in a 1:1 ratio, stratified by their baseline M-score (< 5 or ≥ 5). The randomization process adopted block randomization with varying block sizes of 4 or 6. The Clinical Research Coordinator (CRC) at the principal center obtained the randomization number by accessing a centralized web-based randomization service called the Research Electronic Data Capture (REDCap) system68, which was maintained by Sun Yat-sen Memorial Hospital (principal center). Researchers at each center would then contact the CRC at the principal center to obtain the ID numbers and treatment allocations. This trial was open-label, meaning that both the researchers and patients were aware of the treatment allocation and unable to be masked.
Interventions and assessments
For patients assigned to the DL-OBS group, ductal lavage was performed as previously reported26. We used 1% lidocaine for local anesthesia and a lacrimal probe to localize 4–8 lactiferous ducts on the nipple, then cannulated the ducts using micro-catheters (Anhui JN Medical Device Co., Ltd, China.) (Supplementary Fig. 3). Then, the ducts were infused with 5 ml of 2% lidocaine mixed with 20 ml of normal saline containing 40 mg of triamcinolone acetonide and 1 g of ceftriaxone. If resistance is felt or fluid extravasation occurs during the lavage process (except for those extravasated from the fistula), it is advisable to administer the lavage fluids intermittently and slowly until all fluids have been infused. The next day, patients need to receive breast massage in the clinic. This process runs in cycles 5 times and is then followed by observation alone.
For patients assigned to the oral corticosteroids group, 25–50 mg prednisone or 20–40 mg methylprednisolone tablets were given per day initially based on the standard of each institution, with the dose decreased by 5 mg of prednisone or 4 mg of methylprednisolone every two weeks until the maintenance dose reached to 25 mg of prednisone or 20 mg of methylprednisolone, then maintained for at least one month and up to six months. The discontinuation is permitted if the patients achieve cCR after one month of treatment (Fig. 1 in Supplementary Fig. 2).
Aspiration of the abscess is allowed in both groups if indicated during this period. Once patients are considered as relapse or treatment failure, the standard-of-care management is allowed based on the standard of institutions where they belong, as well as the preference of patients in both arms. All patients were scheduled for follow-up at 2 weeks, 1, 2, 6, and 12 months. Here, we used the M-score to evaluate the severity of symptoms as well as the efficacy of treatment (Supplementary Fig. 2). The M-score considered the following aspects on a two-point scale, Mass: 0 (absence of a mass by palpation), 1 (mass\(\le\)3 cm by palpation), and 2 (mass\( > \)3 cm by palpation); Erythema: 0 (absence of skin erythema) and 2 (presence of skin erythema); Fistula: 0 (absence of fistula) and 2(presence of fistula); Pain: 0 (VAS 0 to 2), 1 (VAS 3 to 5) and 2 (VAS 6 to 10); Quality of life: 0 (absence of effects on QoL), 1 (mild effects on QoL) and 2 (serious effects on QoL). It ranged between 0 to 10, with a higher score indicating a more severe disease symptom that medical intervention was much more needed. All evaluators have received standardized training to ensure consistency to a great extent. Besides, the M-score would be calculated by evaluators who were blinded to the group assignment.
Outcomes
The primary outcome was a complete Clinical Response (cCR) rate, defined as the proportion of patients who achieved M-score\(\le\)1 at 1-year follow-up. Secondary outcomes were time to cCR (the median time to cCR after randomization), treatment failure rate (TF; TF evaluation was dependent on different baseline M-score as predefined65), relapse rate (had M-score > 4 among those patients who achieved cCR), and protocol compliance rate (received and completed the treatment protocol as allocated). Safety outcomes included predefined and any other unexpected AEs. Any AE would be collected and graded based on the National Cancer Institute Common Terminology Criteria Adverse Events (NCI-CTCAE) version 5.0.
Sample size calculation
The clinically acceptable non-inferiority margin for DL-OBS was defined as a 1-year cCR rate not lower than 15% compared to the oral corticosteroids, hypothesizing that the cCR rate of oral corticosteroids would be 90%. We estimated 140 patients were required to give 80% statistical power with a significant one-sided \(\alpha\) level of 2.5%, assuming 10% of patients may be lost to follow-up.
Statistical analysis
The aim of this trial was to assess the non-inferiority of DL-OBS compared to oral corticosteroids. The primary analysis was conducted on the intention-to-treat (ITT) and per-protocol populations, non-inferiority would be claimed if the lower limit of 95%CI was greater than − 15% in both analyses. The ITT population included all patients who underwent randomization, while the per-protocol population excluded patients with protocol violations. The risk difference and corresponding 95%CI of cCR rate between the two groups were calculated by the Wald ratio method. In the primary analysis, no adjustment was implemented. Missing data were treated as missing without any imputation for all efficacy analyses but the primary endpoint in the ITT population, which would be imputed as not reaching cCR for missing. Prespecified subgroup analyses of the primary outcome were performed by conducting the Cox proportional hazards model to explore if there exist any interactions between treatment and covariates.
For secondary outcomes, \(\chi\)2 tests or Fisher’s exact test was used to compare the efficacy based on specific conditions, except for time to cCR, which would be estimated by log-rank test. No imputation was performed for missing data in secondary outcomes analyses. Safety analyses were conducted on patients who received at least one dosage of ductal lavage or oral corticosteroids. Additional exploratory analysis was performed by using the Wilcox test to compare the difference in baseline levels of the hematological index in different cCR statuses and time to cCR.
All two-sided p-values less than 0.05 were considered to be statistically significant. Analyses were performed with R version 4.2.1(R Foundation) and SAS version 9.4 (SAS Institute). This trial was prospectively registered at ClinicalTrials.gov (NCT03724903).
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
The original individual patient-level clinical data cannot be publicly available due to privacy restrictions. The de-identified clinical data, including data dictionaries, will be made available on request to the corresponding authors one year after publication of the manuscript with a detailed proposal for research purposes via email (K.C: chenkai23@mail.sysu.edu.cn and S.Li: lishr@mail.sysu.edu.cn). Requests will be subject to the review and approval of Sun Yat-sen Memorial Hospital. The study protocol, including the statistical analysis plan, has been uploaded as Supplement 2 in the Supplementary Information files. The remaining data are available within the Article, Supplementary Information, or Source Data File. Source data are provided in this paper.
References
Kessler, E. & Wolloch, Y. Granulomatous mastitis: a lesion clinically simulating carcinoma. Am. J. Clin. Pathol. 58, 642–646 (1972).
Bouton, M. E., Jayaram, L., O’Neill, P. J., Hsu, C.-H. & Komenaka, I. K. Management of idiopathic granulomatous mastitis with observation. Am. J. Surg. 210, 258–262 (2015).
Davis, J. et al. Re-evaluating if observation continues to be the best management of idiopathic granulomatous mastitis. Surgery 166, 1176–1180 (2019).
Çetinkaya, G., Kozan, R., Emral, A. C. & Tezel, E. Granulomatous mastitis, watch and wait is a good option. Ir. J. Med. Sci. 190, 1117–1122 (2021).
Martinez-Ramos, D. et al. Idiopathic granulomatous mastitis: A systematic review of 3060 patients. Breast J. 25, 1245–1250 (2019).
Steuer, A. B. et al. Clinical characteristics and medical management of idiopathic granulomatous mastitis. JAMA Dermatol. 156, 460–464 (2020).
Joseph, K.-A., Luu, X. & Mor, A. Granulomatous mastitis: A New York public hospital experience. Ann. Surg. Oncol. 21, 4159–4163 (2014).
Yuan, Q.-Q. et al. Management of granulomatous lobular mastitis: an international multidisciplinary consensus (2021 edition). Mil. Med. Res. 9, 20 (2022).
Yabanoğlu, H. et al. A comparative study of conservative versus surgical treatment protocols for 77 patients with idiopathic granulomatous mastitis. Breast J. 21, 363–369 (2015).
Akcan, A. et al. Idiopathic granulomatous mastitis: Comparison of wide local excision with or without corticosteroid therapy. Breast Care 9, 111–115 (2014).
Néel, A. et al. Long-term outcome in idiopathic granulomatous mastitis: a western multicentre study. QJM Mon. J. Assoc. Physicians 106, 433–441 (2013).
Aghajanzadeh, M. et al. Granulomatous mastitis: Presentations, diagnosis, treatment and outcome in 206 patients from the north of Iran. Breast Edinb. Scotl. 24, 456–460 (2015).
Manst, D., Ganschow, P., Marcus, E., Holden, C. & Datta, S. Abstract P3-14-10: Intralesional steroid injection: A novel method to treat the symptoms of idiopathic granulomatous mastitis. Cancer Res. 79, P3-14–P3-110 (2019).
Yildirim, E. et al. Comparison of the efficiency of systemic therapy and intralesional steroid administration in the treatment of idiopathic granulomatous Mastitis. The novel treatment for Granulomatous Mastitis. Ann. Ital. Chir. 92, 234–241 (2021).
Moldoveanu, D., Lee, C. & Hesley, G. Framework and guide for intralesional steroid injections in idiopathic granulomatous mastitis. Eur. J. Radiol. 168, 111118 (2023).
Kehribar, D. Y., Duran, T. I., Polat, A. K. & Ozgen, M. Effectiveness of methotrexate in idiopathic granulomatous mastitis treatment. Am. J. Med. Sci. 360, 560–565 (2020).
Kafadar, M. T., Bahadır, M. V. & Girgin, S. Low-dose methotrexate use in idiopathic granulomatous mastitis: An alternative treatment method. Breast Care 16, 402–407 (2021).
Zhou, F. et al. Expert consensus on diagnosis and treatment of non-lactating mastitis(in Chinese). Chin. J. Pract. Surg. 36, 755–758 (2016).
Fardet, L., Kassar, A., Cabane, J. & Flahault, A. Corticosteroid-induced adverse events in adults: frequency, screening and prevention. Drug Saf. 30, 861–881 (2007).
Fardet, L. et al. Corticosteroid-induced clinical adverse events: frequency, risk factors and patient’s opinion. Br. J. Dermatol. 157, 142–148 (2007).
Volmer, T., Effenberger, T., Trautner, C. & Buhl, R. Consequences of long-term oral corticosteroid therapy and its side-effects in severe asthma in adults: a focused review of the impact data in the literature. Eur. Respir. J. 52, 1800703 (2018).
Dooley, W. C. et al. Ductal lavage for detection of cellular atypia in women at high risk for breast cancer. J. Natl. Cancer Inst. 93, 1624–1632 (2001).
Evron, E. et al. Detection of breast cancer cells in ductal lavage fluid by methylation-specific PCR. Lancet Lond. Engl. 357, 1335–1336 (2001).
Stearns, V. et al. Preclinical and clinical evaluation of intraductally administered agents in early breast cancer. Sci. Transl. Med. 3, 106ra108 (2011).
Murata S. et al. Ductal access for prevention and therapy of mammary tumors. Cancer Res. 66, 638–645 (2006).
Chen, K. et al. Ductal lavage for patients with nonlactational mastitis: A single-arm, proof-of-concept trial. J. Surg. Res. 235, 440–446 (2019).
Deng, J. Q. et al. Steroids administered after vacuum-assisted biopsy in the management of idiopathic granulomatous mastitis. J. Clin. Pathol. 70, 827–831 (2017).
Kaviani, A. et al. Idiopathic granulomatous mastitis: Looking for the most effective therapy with the least side effects according to the severity of the disease in 374 patients in Iran. Breast J. 25, 672–677 (2019).
Tan, Q.-W. et al. Methylprednisolone for idiopathic granulomatous mastitis: a prospective observational cohort study. Gland Surg. 11, 1538–1545 (2022).
Troxell, M. L. et al. Cystic neutrophilic granulomatous mastitis: Association with gram-positive bacilli and corynebacterium. Am. J. Clin. Pathol. 145, 635–645 (2016).
Bhattarai, P. et al. Idiopathic granulomatous mastitis: experience at a New York hospital. Ann. R. Coll. Surg. Engl. 104, 543–547 (2022).
Yin, Y. et al. Idiopathic granulomatous mastitis: Etiology, clinical manifestation, diagnosis and treatment. J. Investig. Surg. J. Acad. Surg. Res. 35, 709–720 (2022).
Li, J. Diagnosis and treatment of 75 patients with idiopathic lobular granulomatous mastitis. J. Investig. Surg. J. Acad. Surg. Res. 32, 414–420 (2019).
Dilaveri, C. et al. Idiopathic granulomatous mastitis. Breast J. https://doi.org/10.1155/2024/6693720 (2024).
Bi, J. et al. Etiology of granulomatous lobular mastitis based on metagenomic next-generation sequencing. Int. J. Infect. Dis. IJID. Publ. Int. Soc. Infect. Dis.113, 243–250 (2021).
Yu, H.-J. et al. Clinical metagenomic analysis of bacterial communities in breast abscesses of granulomatous mastitis. Int. J. Infect. Dis. 53, 30–33 (2016).
Ong, S. S. et al. Profiling microbial communities in idiopathic granulomatous mastitis. Int. J. Mol. Sci. 24, 1042 (2023).
Taylor, G. B., Paviour, S. D., Musaad, S., Jones, W. O. & Holland, D. J. A clinicopathological review of 34 cases of inflammatory breast disease showing an association between corynebacteria infection and granulomatous mastitis. Pathology 35, 109–119 (2003).
Dobinson, H. C. et al. Antimicrobial treatment options for granulomatous mastitis caused by corynebacterium species. J. Clin. Microbiol. 53, 2895–2899 (2015).
Lei, X. et al. Treatments for idiopathic granulomatous mastitis: Systematic review and meta-analysis. Breastfeed. Med. J. Acad. Breastfeed. Med. 12, 415–421 (2017).
Henriksen, M. et al. C-reactive protein: a predictive factor and marker of inflammation in inflammatory bowel disease. Results from a prospective population-based study. Gut 57, 1518–1523 (2008).
Savioli, F. et al. Prognostic role of preoperative circulating systemic inflammatory response markers in primary breast cancer: meta-analysis. Br. J. Surg. 109, 1206–1215 (2022).
Zhao, Y., Yin, L., Patel, J., Tang, L. & Huang, Y. The inflammatory markers of multisystem inflammatory syndrome in children (MIS-C) and adolescents associated with COVID-19: A meta-analysis. J. Med. Virol. 93, 4358–4369 (2021).
Montazer, M., Dadashzadeh, M. & Moosavi Toomatari, S. E. Comparison of the outcome of low dose and high-dose corticosteroid in the treatment of idiopathic granulomatous mastitis. Asian Pac. J. Cancer Prev. APJCP 21, 993–996 (2020).
Shojaee, L., Rahmani, N., Moradi, S., Motamedi, A. & Godazandeh, G. Idiopathic granulomatous mastitis: challenges of treatment in iranian women. BMC Surg. 21, 206 (2021).
Griggs, R. C. et al. Efficacy and safety of deflazacort vs prednisone and placebo for Duchenne muscular dystrophy. Neurology 87, 2123–2131 (2016).
Zhu, H.-M. et al. Safety and effectiveness of oral methylprednisolone therapy in comparison with intramuscular adrenocorticotropic hormone and oral prednisolone in children with infantile spasms. Front. Neurol. 12, 756746 (2021).
Tondre, J., Nejad, M., Casano, A., Mills, D. & Love, S. Technical enhancements to breast ductal lavage. Ann. Surg. Oncol. 15, 2734–2738 (2008).
Han, S. & Hong, Y. G. The inverted nipple: its grading and surgical correction. Plast. Reconstr. Surg. 104, 389–395 (1999).
Townsend, C. M., Beauchamp, R. D., Evers, B. M. & Mattox, K. L. Sabiston Textbook of Surgery: The Biological Basis of Modern Surgical Practice. (Elsevier Health Sciences, 2016).
Teboul, M. & Halliwell, M. Atlas of Ultrasound and Ductal Echography of the Breast: The Introduction of Anatomic Intelligence into Breast Imaging. (1995).
Sartorius, O. W., Smith, H. S., Morris, P., Benedict, D. & Friesen, L. Cytologic evaluation of breast fluid in the detection of breast disease. J. Natl. Cancer Inst. 59, 1073–1080 (1977).
Love, S. M. & Barsky, S. H. Anatomy of the nipple and breast ducts revisited. Cancer 101, 1947–1957 (2004).
Loud, J. T. et al. Tolerability of breast ductal lavage in women from families at high genetic risk of breast cancer. BMC Women’s Health 9, 20 (2009).
Visvanathan, K. et al. The reliability of nipple aspirate and ductal lavage in women at increased risk for breast cancer–a potential tool for breast cancer risk assessment and biomarker evaluation. Cancer Epidemiol. Biomark. Prev. 16, 950–955 (2007).
Noel, G. L., Suh, H. K. & Frantz, A. G. Prolactin release during nursing and breast stimulation in postpartum and nonpostpartum subjects. J. Clin. Endocrinol. Metab. 38, 413–423 (1974).
Amico, J. A. & Finley, B. E. Breast stimulation in cycling women, pregnant women and a woman with induced lactation: pattern of release of oxytocin, prolactin and luteinizing hormone. Clin. Endocrinol. 25, 97–106 (1986).
Mens, J. M., Nico de Wolf, A., Berkhout, B. J. & Stam, H. J. Disturbance of the menstrual pattern after local injection with triamcinolone acetonide. Ann. Rheum. Dis. 57, https://doi.org/10.1136/ard.57.11.700 (1998).
Weitoft, T., Larsson, A. & Rönnblom, L. Serum levels of sex steroid hormones and matrix metalloproteinases after intra-articular glucocorticoid treatment in female patients with rheumatoid arthritis. Ann. Rheum. Dis. 67, 422–424 (2008).
Ren, Y., Zhang, J., Zhang, J. & Guo, R. Combining intralesional steroid injection with oral steroids in patients with idiopathic granulomatous mastitis. Medicine 102, e34055 (2023).
Toktas, O. et al. A novel first-line treatment alternative for noncomplicated idiopathic granulomatous mastitis: Combined intralesional steroid injection with topical steroid administration. Breast Care 16, 181–187 (2021).
Kapoor, N. S. et al. Presentation and management of granulomatous mastitis in the United States: results of an American Society of Breast Surgeons Registry Study. Ann. Surg. Oncol. https://doi.org/10.1245/s10434-024-15714-x (2024).
Ma, X., Min, X. & Yao, C. Different treatments for granulomatous lobular mastitis: A systematic review and meta-analysis. Breast Care 15, 60–66 (2020).
World Medical Association. World Medical Association Declaration of Helsinki: ethical principles for medical research involving human subjects. JAMA 310, 2191–2194 (2013).
Hu, T. et al. Multicentre, randomised, open-label, non-inferiority trial comparing the effectiveness and safety of ductal lavage versus oral corticosteroids for idiopathic granulomatous mastitis: a study protocol. BMJ Open 10, e036643 (2020).
Kawashima, K. et al. Granulomatous mastitis in a male breast: A case report and review of literature. Clin. Case Rep. 11, e7048 (2023).
Cui, L.-Y., Sun, C.-P., Li, Y.-Y. & Liu, S. Granulomatous mastitis in a 50-year-old male: A case report and review of literature. World J. Clin. Cases 12, 451–459 (2024).
Harris, P. A. et al. Research electronic data capture (REDCap)–a metadata-driven methodology and workflow process for providing translational research informatics support. J. Biomed. Inform. 42, 377–381 (2009).
Acknowledgements
We appreciate the assistance from the Disease Registry Department, the Artificial Intelligence Lab, and the Big Data Center of Sun Yat-sen Memorial Hospital, Sun Yat-sen University, and the support from the REDCap development team and research teams of Vanderbilt University Medical Center. We also sincerely appreciate the guidance and supervision from Prof. Erwei Song from Sun Yat-sen Memorial Hospital of Sun Yat-sen University and the technical and financial support from Dr. Qian Li from the Guangzhou Women and Children’s Medical Center. This work was supported by grants from the National Natural Science Foundation of China (#82271650 [K.C]), and the Yat-sen Scholarship of Young Scientist program of Sun Yat-sen Memorial Hospital of Sun Yat-sen University [K.C]. This work was also supported by grant from the Sun Yat-Sen University Clinical Research 5010 Program (#2018022 [K.C]), and the grants from the China Anti-aging Promoting Association [S.L]. The sponsors had no role in design and conduct of the study; collection, management, analysis, and interpretation of the data; preparation, review, and approval of the manuscript, and the decision to submit for publication.
Author information
Authors and Affiliations
Contributions
All authors contributed extensive efforts to the study. K.C., Heng.H., and Hui.H. were principal investigators. K.C., S.Li, and L.Z. contributed to the study design and development of the concept. K.C., X.C., and J.Zhang wrote the study protocol, all authors approved the study protocol. K.C., S.Li, Heng.H., Hui.H., L.Z., F.Y., Y.W., Jianli.Z, T.H., X.G., Y.L., W.W., L.D., N.R., Jinghua.Z., Y.Y., Y.Z., and L.T. enrolled and treated the patients. L.Y., Y.X., Y.J., L.S., Q.X., and X.C. performed the ductal lavage. Q.C., Q.L., and S.Liu collected patient data. J.Y. and C.T. conducted the pathological diagnosis of enrolled patients. K.C., X.C., and J.Zhang conducted the statistical analyses. K.C., X.C., and J.Zhang wrote the first draft of the manuscript. All authors reviewed, edited, proofed, and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Anurag K Srivastava and the other anonymous reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Chen, X., Huang, H., Huang, H. et al. Ductal lavage followed by observation versus oral corticosteroids in idiopathic granulomatous mastitis: A randomized trial. Nat Commun 15, 9144 (2024). https://doi.org/10.1038/s41467-024-53143-2
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-024-53143-2