Abstract
Electronically mismatched Diels−Alder reaction between two electron-deficient components is synthetically useful and yet underdeveloped under thermal conditions. Herein, a photoinduced formal [4 + 2] cycloaddition of enone with a variety of electron-deficient dienes is described. Key to the success of this stepwise methodology relies on a C − C bond cleavage/rearrangement of the cyclobutane based overbred intermediate via diversified mechanistic pathways. Based on this annulation method, total synthesis of lucidumone is achieved in nine steps.
Similar content being viewed by others
Introduction
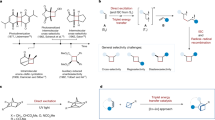
Target-oriented synthesis of biologically important natural products with ever-increasing efficiency has attracted significant research interest in the synthetic community1,2,3,4,5,6,7,8. In this context, new transformations have always been in high demand for computer-aided synthesis planning, providing diverse annulation strategies to facilitate efficient access to molecular complexity. Among the most efficient annulation strategies, Diels–Alder cycloaddition is of considerable importance and has been widely used in natural product synthesis9,10,11,12,13. To secure a synthetically useful reaction efficiency, the state-of-the-art reaction partners are usually an electron-rich diene and an electron-deficient dienophile, while the electronically mismatched variants require harsh conditions under thermal conditions. In 2011, Yoon and co-workers reported a radical cation Diels−Alder reaction of anethole derivatives with electron-rich diene under visible light irradiation14. Using an inverse design, they also realized the intramolecular radical anion hetero-Diels−Alder cycloaddition of tethered bis(enones)15. Nonetheless, the [4 + 2] cycloaddition between two electron-deficient components is largely underdeveloped (Fig. 1a)16,17.
a Four types of Diels-Alder cycloaddition. b This work: formal [4 + 2] photocycloaddition of two electron-deficient olefins and its synthetic application to lucidumone. EDG electron-donating group, EWG electron-withdrawing group.
The inherent challenge within this closed-shell process under thermal conditions lies in their energetically mismatched frontier molecular orbitals and the nature of an entropy reduction event, as well as the elusive control of reaction selectivity. In this regard, an open-shell radical path would render a reliable choice for the highest-occupied molecular orbital (HOMO) activation of dienophiles18,19,20,21. Through direct photoexcitation or triplet-triplet energy transfer under visible light irradiation, the overbred intermediate (usually the [2 + 2] cycloadduct) is formed, which we speculate would undergo a strain-release-driven rearrangement22,23 to deliver the [4 + 2] cycloadduct in a stepwise manner (Fig. 1b). In this work, we describe an operationally easy protocol, namely, the formal [4 + 2] cycloaddition of indenone and electron-deficient dienes for the rapid access to 6/5/6 indanone fused skeleton, which exists widely in bioactive natural products, such as cephanolide A24, lucidumone25, dysambiol26, etc.
Results
Reaction development
At first, the exploration of this intermolecular [4 + 2] cycloaddition employing indenone 1 and diene 2 as two electron-deficient components was investigated (Table 1). As expected, the [2 + 2] cycloadduct 3 was generated in one hour with a 94% yield. Interestingly, the diastereomeric ratio (d.r.) of 3 increased as the extension of reaction time (Table 1, entries 1–5), indicating a reversible cleavage and formation of C4’ − C5’ bond in 3 under irradiation. In fact, the homolytic opening of this cyclobutane-based overbred intermediate 3 through direct photoexcitation could lead to the formation of diradical species A (Fig. 2, path a) and subsequent cyclization via the connection of C5’ and C7’ to deliver 4, albeit with low efficiency (Table 1, entries 2–5). Given a vinylogous donor-acceptor cyclobutane character of 3, we then turned to an alternative pathway for the C − C bond cleavage/rearrangement27,28,29. Through the introduction of a catalytic amount of Lewis acid, such as LiI, LiBF4, Cu(ClO4)2 and Ce(OTf)3 (Table 1, entries 6–9), a heterolytic cleavage of C4’ − C5’ in 3 was realized through a dipolar, zwitterionic pathway (Fig. 2, path b), affording the exo-selective [4 + 2] cycloadduct 4 with moderate yields (21% to 59%). The relative configuration of 3 and 4 was determined by their X-ray diffraction analysis, respectively. To our delight, 4 was obtained in 85% yield when Mg(ClO4)2 was used (Table 1, entry 10). A slight decrease in the reaction efficiency was observed when an alternative LED source centered at 455 nm (430–485 nm wavelength) was employed (Table 1, entry 11), presumably because of the relatively weaker absorption of 1 at the longer wavelength. Moreover, control experiments showed that light was essential for this cycloaddition reaction since neither 3 nor 4 could be detected when the reaction was conducted in the dark or even at higher temperatures (Table 1, entries 12–13).
p-TsOH•H2O p-toluenesulfonic acid monohydrate.
Mechanistic studies
To probe the crucial rearrangement mechanism, UV-vis experiments and control experiments were first investigated (See Supplementary Information for more details). Both indenone substrate 1 and the overbred intermediate 3 were able to harvest photons upon illumination with a narrow-band LED centered at 395 nm (380–420 nm wavelength), allowing secondary excitation a feasible path for the homolytic cleavage of C4’ − C5’ bond. Experimentally, the pure intermediate 3 was subjected to irradiation in the absence of Lewis acid (Fig. 2, eq 1). While the cycloadduct 4 was formed in 16% yield, a byproduct 5, identified as the dimer of indenone 1, was detected simultaneously. In addition, the cross experiment showed [2 + 2] cycloadduct 6 was obtained in 17% yield (Fig. 2, eq 2). Consequently, the in-situ generated diradical species A triggered the desired rearrangement to form the C5’ − C7’ bond, along with the cleavage of C2’ − C3’ as a competitive path to regenerate indenone 1. On the other hand, trapping of the zwitterionic intermediate B in the presence of external Bronsted acid was realized as the fact that the demethylation product 7 was isolated in 20% yield (Fig. 2, eq 3). In light of these findings, the diversified cleavage of C4’ − C5’ in both paths contributes together to the rearrangement of a cyclobutane-based overbred intermediate.
Reaction scope
Next, we explored the substrate scope of this Lewis acid-assisted photocycloaddition. As shown in Fig. 3, a negligible electronic effect of the C6-substituents on the phenyl ring was observed, since the corresponding [4 + 2] cycloadducts (8–13) were obtained in good yields (78%–91%). Notably, the functional group prone to single electron reduction, such as acyl-substituted aryl bromide (10), stayed inert in this photocycloaddition protocol. Removal of the methyl enol ether functionality of the enone substrate resulted in a dramatic decline of reaction efficiency, affording the cycloadducts (14–22) in 55%–81% yields. In these cases, an alternative LED centered at 370 nm (355–385 nm wavelength) was used instead to facilitate the efficient secondary excitation of the overbred intermediate. In addition, β-arylated or alkylated indenones were able to undergo the photocycloaddition smoothly, affording the [4 + 2] cycloadducts (23–25) in moderate to good yields. In the case where chemoselectivity is an issue, the [4 + 2] cycloaddition occurred predominantly at the enone motif (26), leaving the phenylacetylene functionality intact. The reaction was also found to be compatible with several sensitive groups, such as phenol ester (27) that could be easily hydrolyzed, and α-TMS-substituted ethers (28) that fragment with cleavage of the silyl group via photoinduced electron transfer (PET)30,31,32,33. Moreover, 2-methylidene and even fully substituted indenone could be compatible with this photocycloaddition, delivering the cycloadducts 29 and 30 in 56% and 77% yield, respectively. Besides indenone, the cycloaddition of acyclic enones, such as chalcone (31) and its derivatives (32–35), could also proceed smoothly with excellent exo selectivity.
Reaction conditions: enone (0.2 mmol), 10 mol% Mg(ClO4)2, and a diene (0.6 mmol) in MeCN (2 mL) in the presence of 395 nm LEDs at room temperature under argon, a single isomer was detected unless noted otherwise. a Diene 2 was used. b 370 nm LEDs were used. c 10 mol% Cu(NTf2)2 was used instead. d Enone 1 was used. Cu(NTf2)2 Copper(II) bis(trifluoromethanesulfonyl)imide.
Pleasingly, this mild and easy-handling cycloaddition protocol proved to bear good tolerance to a variety of electron-deficient dienes, rendering rather robust access to 6/5/6 indanone fused scaffolds. For example, alternating the steric hindrance at C3 of the 1,1-diester substituted 1,3-dienes by changing Me to H, Et, iPr, and OTBS, no significant decrease of the yield for 36–39 was observed. Moreover, the reaction showcased perfect diastereoselectivity control when a non-terminal diene was used, yielding 40 as a single diastereomer. We reasoned the diastereoselectivity was determined prior in the kinetically controlled [2 + 2] photocycloaddition process, which usually occurred regioselectively at the more electron-rich and less hindered terminus of the diene. Although as many as three stereocenters were formed, the cycloaddition of 1,2-disubstituted diene with aryl functionality could also provide the thermodynamically more stable cycloadducts (41–46) predominantly as elucidated by the X-ray crystal structure of 41. Aside from linear dienes, 1,3-cyclohexadiene bearing electron-withdrawing groups (47–50) could also work well when indenone 1 was used, enabling a rapid assembly of the rigid [2.2.2] ring system. Different from the state-of-the-art Diels-Alder reaction, the exo-cycloadduct becomes predominant in all cases of this photoinduced process. Notably, the reaction of the 2-pyrone derivative (47) was also realized at ambient temperature, circumventing the loss of CO2 upon vigorous heating conditions34,35,36, and accordingly making this formal [4 + 2] photocycloaddition protocol amendable for developing cascade Diels-Alder reactions.
Synthetic application
To demonstrate the synthetic potential of this simple and photocatalyst-free protocol, total synthesis of lucidumone37,38,39,40,41, a unique polycyclic meroterpenoid isolated from the fruiting bodies of Ganoderma lucidum, was achieved as depicted in Fig. 4. As expected, the [4 + 2] photocycloaddition of indenone 51 with diene 52 proceeded smoothly, furnishing 53 in 86% yield, albeit a dramatic decline of the exo/endo selectivity to 2.5:1 (See supplementary information for more optimization details). The configuration of the major isomer was confirmed by X-ray crystallography. We reasoned in this odd case that a dipole-dipole interaction of the carbonyl groups caused the poor diastereoselectivity (1.2:1 d.r.) of the corresponding overbred intermediate and, accordingly, a moderate exo selectivity for the [4 + 2] cycloadduct. Importantly, no significant decrease in the yield was observed when this photocycloaddition was performed on a gram scale in a 250 mL quartz glass apparatus, facilitating the accumulation of compound 53 for further manipulations. Inspired by the independent work of Dong42,43 and Martin44, attempts using an aromatization-promoted deacylation strategy were first made to remove the extra acetyl group at C6’. However, the acetyl group at this position was left intact under the condensation conditions with the excess amount of 2-aminobenzamide or 2-pyridyl hydrazine due to the large steric hindrance. To solve this problem, site-selective deacylation at C6’ was performed via a three-step process. Firstly, selective enol silylation was realized through indiscriminate protection with the excess amount of TMSOTf and release of the less hindered acyl group at C3’ using potassium fluoride. Next, a successive oxidative cleavage of the enol silyl ether 54 using m-CPBA and Pb(OAc)4 in one pot succeeded in providing the acid 55 in 63% yield. Finally, the advanced intermediate 55 was decarboxylated45 in the presence of disulfide 56 and tributylphosphine, affording the desired compound 57 in 85% yield. After the successful assembly of the indanone fused tetracyclic ring system, a photocatalytic oxidative alkoxymethylation46 was designed to install the ether ring and C4’-OH in a single operation. Thus, annulation of 57 was achieved in the presence of oxygen as a radical trapping agent, providing lucidumone in 68% yield. Notably, only five steps were required leveraging this photoinduced annulation relay from 51 (9 steps as the longest linear sequence from succinic anhydride).
Reagent and conditions: (a) Mg(ClO4)2 (0.1 equiv.), diene 52 (3 equiv.), MeCN, 395 nm LEDs, 35 °C, 86%, exo/endo = 2.5:1. b TMSOTf (3 equiv.), Et3N (3 equiv.), DCM, rt; then KF (1.2 equiv.), acetone, rt, 67%. c m-CPBA (1.2 equiv.), NaHCO3 (2.5 equiv.), 0 °C, DCM; then Pb(OAc)4 (3 equiv.), K2CO3 (6 equiv.), toluene, 0 °C to rt, 63%. d 56 (2 equiv.), nBu3P (2.2 equiv.), tBuSH (5 equiv.), tungsten lamp, THF, rt, 85%. e TPP (0.1 equiv.), Co(acac)2 (3 equiv.), CF3SO3H (3 equiv.), MeCN, 455 nm LEDs; then 6 M aq. HCl, MeOH, 60 °C, 68%, single isomer at C4’. TMSOTf trimethylsilyl trifluoromethanesulfonate, m-CPBA 3-chloroperbenzoic acid, Pb(OAc)4 Lead(IV) acetate, nBu3P tributyl phosphine, tBuSH tert-butyl mercaptan.
In summary, a formal [4 + 2] photocycloaddition between two electron-deficient olefins has been developed. A strain-release-driven rearrangement of the overbred intermediate occurred via diversified pathways to deliver the corresponding [4 + 2] cycloadduct with excellent exo selectivity. Given the flexible substrate scope, this photocatalyst-free protocol provided a robust cycloaddition paradigm that would otherwise be difficult to achieve. With the assistance of two other photoinduced reactions featuring a Barton decarboxylation and an oxidative alkoxymethylation of a spatially proximate double bond, the application of this photocycloaddition methodology to the facile synthesis of lucidumone is accomplished. More synthetic applications and the enantioselective variant of this reaction are under investigated in our lab.
Methods
General photoreaction procedure
In an 8 mL vial, a solution of enone (0.2 mmol, 1.0 equiv.), diene (0.6 mmol, 3 equiv.) and Mg(ClO4)2 (0.02 mmol, 0.1 equiv.) in MeCN (2.0 mL) was degassed by Argon sparging for 10 min, then irradiated with 395 nm LED at ~ 10 cm away from the light source (to maintain the reaction temperature below 35 oC). After the enone substrate was fully consumed, the LED lamp was turned off, and the reaction mixture was concentrated under reduced pressure. The residue was purified by flash column chromatography on silica gel to afford the desired [4 + 2] cycloadducts.
Data availability
The X-ray crystallographic data for structures reported in this study have been deposited at the Cambridge Crystallographic Data Center (CCDC) under deposition numbers 2344341 (for 53), 2290150 (for 3), 2290151 (for 4), 2346629 (for 32), 2290149 (for 47), 2290148 (for 41), and 2357505 (for 5). Copies of these data can be accessed free of charge via https://www.ccdc.cam.ac.uk/structures/. All other data supporting the findings of this study are available within this article and its Supplementary Information file. Data supporting the findings of this manuscript are also available from the authors upon request. The experimental procedures and characterization of all new compounds are provided in Supplementary Information.
References
Schreiber, S. L. Target-oriented and diversity-oriented organic synthesis in drug discovery. Science 287, 1964–1969 (2000).
Roche, S. P. & Porco, J. A. Jr. Dearomatization strategies in the synthesis of complex natural products. Angew. Chem. Int. Ed. 50, 4068–4093 (2011).
Kang, B., Jakubec, P. & Dixon, D. J. Strategies towards the synthesis of calyciphylline A-type daphniphyllum alkaloids. Nat. Prod. Rep. 31, 550–562 (2014).
Shen, X., Thach, D. Q., Ting, C. P. & Maimone, T. J. Annulative methods in the synthesis of complex meroterpene natural products. Acc. Chem. Res. 54, 583–594 (2021).
Kanda, Y. et al. Two-phase synthesis of taxol. J. Am. Chem. Soc. 142, 10526–10533 (2020).
He, W., Zhang, Z. & Ma, D. A scalable total synthesis of the antitumor agents Et-743 and lurbinectedin. Angew. Chem. Int. Ed. 58, 3972–3975 (2019).
Woo, S. & Shenvi, R. A. Synthesis and target annotation of the alkaloid GB18. Nature 606, 917–921 (2022).
George, J. H. Biomimetic dearomatization strategies in the total Synthesis of meroterpenoid natural products. Acc. Chem. Res. 54, 1843–1855 (2021).
Nicolaou, K. C., Snyder, S. A., Montagnon, T. & Vassilikogiannakis, G. The Diels–Alder reaction in total synthesis. Angew. Chem. Int. Ed. 41, 1668–1698 (2002).
Corey, E. J. Catalytic Enantioselective Diels–Alder reactions: methods, mechanistic fundamentals, pathways, and applications. Angew. Chem. Int. Ed. 41, 1650–1667 (2002).
Takao, K.-I., Munakata, R. & Tadano, K.-I. Recent advances in natural product synthesis by using intramolecular Diels−Alder reactions. Chem. Rev. 105, 4779–4807 (2005).
Li, J.-L., Liu, T.-Y. & Chen, Y.-C. Aminocatalytic asymmetric Diels–Alder reactions via HOMO activation. Acc. Chem. Res. 45, 1491–1500 (2012).
Xu, G., Bai, X. & Dang, Q. Aromatic heterocycles as productive dienophiles in the inverse electron-demand Diels–Alder reactions of 1,3,5-triazines. Acc. Chem. Res. 53, 773–781 (2020).
Lin, S., Ischay, M. A., Fry, C. G. & Yoon, T. P. Radical cation Diels–Alder cycloadditions by visible light photocatalysis. J. Am. Chem. Soc. 133, 19350–19353 (2011).
Hurtley, A. E., Cismesia, M. A., Ischay, M. A. & Yoon, T. P. Visible light photocatalysis of radical anion hetero-Diels–Alder cycloadditions. Tetrahedron 67, 4442–4448 (2011).
Liu, S. et al. Construction of alkyl-substituted 7-norbornenones through Diels−Alder cycloaddition of electron-deficient olefins and a cyclopentadienone derivative generated in situ. Chin. Chem. Lett. 33, 2041–2043 (2022).
Feng, J. et al. Enantioselective and collective total syntheses of xanthanolides. Angew. Chem. Int. Ed. 56, 16323–16327 (2017).
Alonso, R. & Bach, T. A chiral thioxanthone as an organocatalyst for enantioselective [2+2] photocycloaddition reactions induced by visible light. Angew. Chem. Int. Ed. 53, 4368–4371 (2014).
Daub, M. E. et al. Enantioselective [2+2] cycloadditions of cinnamate esters: generalizing Lewis acid catalysis of triplet energy transfer. J. Am. Chem. Soc. 141, 9543–9547 (2019).
Murray, P. R. D. et al. Intermolecular crossed [2 + 2] cycloaddition promoted by visible-light triplet photosensitization: expedient access to polysubstituted 2-oxaspiro[3.3]heptanes. J. Am. Chem. Soc. 143, 4055–4063 (2021).
Sicignano, M., Rodríguez, R. I. & Alemán, J. Recent visible light and metal free strategies in [2+2] and [4+2] photocycloadditions. Eur. J. Org. Chem. 2021, 3303–3321 (2021).
Xiang, J.-C., Wang, Q. & Zhu, J. Radical-cation cascade to aryltetralin cyclic ether lignans under visible-light photoredox catalysis. Angew. Chem. Int. Ed. 59, 21195–21202 (2020).
Zhu, M., Xu, H., Zhang, X., Zheng, C. & You, S.-L. Visible-light-induced intramolecular double dearomative cycloaddition of arenes. Angew. Chem. Int. Ed. 60, 7036–7040 (2021).
Fan, Y.-Y. et al. Cephanolides A–J, cephalotane-type diterpenoids from cephalotaxus sinensis. J. Nat. Prod. 80, 3159–3166 (2017).
Yan, Y.-M. et al. (+/−)-Lucidumone, a COX-2 inhibitory caged fungal meroterpenoid from Ganoderma lucidum. Org. Lett. 21, 8523–8527 (2019).
Jiao, W.-H. et al. Dysambiol, an anti-inflammatory secomeroterpenoid from a dysidea sp. marine sponge. Org. Lett. 25, 6391–6395 (2023).
Parsons, A. T. & Johnson, J. S. Formal [4 + 2] cycloaddition of donor−acceptor cyclobutanes and aldehydes: stereoselective access to substituted tetrahydropyrans. J. Am. Chem. Soc. 131, 14202–14203 (2009).
Allart, E. A., Christie, S. D. R., Pritchard, G. J. & Elsegood, M. R. J. Preparation of highly substituted tetrahydropyrans via a metal assisted dipolar cycloaddition reaction. Chem. Commun. 47, 7339–7341 (2009).
Ahmed, A., Christie, S. D. R. & Pritchard, G. J. A comparison of the benzylic and the allylic group as a donor in the formal [4+2] cycloaddition to tetrahydropyrans using donor-acceptor cyclobutanes. Tetrahedron Lett. 58, 3028–3031 (2017).
Gutenberger, G., Steckhan, E. & Blechert, S. α-Silyl ethers as hydroxymethyl anion equivalents in photoinduced radical electron transfer additions. Angew. Chem. Int. Ed. 37, 660–662 (1998).
Khatun, N., Kim, M. J. & Woo, S. K. Visible-light photoredox-catalyzed hydroalkoxymethylation of activated alkenes using α-silyl ethers as alkoxymethyl radical equivalents. Org. Lett. 20, 6239–6243 (2018).
Ahn, D. K., Kang, Y. W. & Woo, S. K. Oxidative deprotection of p-methoxybenzyl ethers via metal-free photoredox catalysis. J. Org. Chem. 84, 3612–3623 (2019).
Gontala, A., Huh, H. & Woo, S. K. Photoredox-catalyzed synthesis of β-amino alcohols: hydroxymethylation of imines with α-silyl ether as hydroxymethyl radical precursor. Org. Lett. 25, 21–26 (2023).
Cai, Q. The [4 + 2] cycloaddition of 2-pyrone in total synthesis. Chin. J. Chem. 37, 946–976 (2019).
Liang, X.-W. et al. Enantioselective synthesis of arene cis-dihydrodiols from 2-pyrones. Angew. Chem. Int. Ed. 58, 14562–14567 (2019).
Huang, G., Kouklovsky, C. & de la Torre, A. Inverse-electron-demand Diels–Alder reactions of 2-pyrones: bridged lactones and beyond. Chem. Eur. J. 27, 4760–4788 (2021).
Ma, S. et al. Construction of the skeleton of lucidumone. Org. Lett. 24, 5541–5545 (2022).
Huang, G., Kouklovsky, C. & de la Torre, A. Gram-scale enantioselective synthesis of (+)-lucidumone. J. Am. Chem. Soc. 144, 17803–17807 (2022).
Huang, G., Kouklovsky, C. & de la Torre, A. Retro-[4+2]/intramolecular Diels–Alder cascade allows a concise total synthesis of lucidumone. Synlett 34, 1195–1199 (2023).
Kawamoto, Y., Noguchi, N., Kobayashi, T. & Ito, H. Total synthesis of lucidumone through convenient one-pot preparation of the tetracyclic skeleton by Claisen rearrangement and subsequent intramolecular aldol reaction. Angew. Chem. Int. Ed. 62, e202304132 (2023).
Liao, X.-Z., Wang, R., Wang, X. & Li, G. Enantioselective total synthesis of (‒)-lucidumone enabled by tandem prins cyclization/cycloetherification sequence. Nat. Commun. 15, 2647 (2024).
Xu, Y. et al. Deacylative transformations of ketones via aromatization-promoted C–C bond activation. Nature 567, 373–378 (2019).
Zhou, X., Xu, Y. & Dong, G. Deacylation-aided C–H alkylative annulation through C–C cleavage of unstrained ketones. Nat. Catal. 4, 703–710 (2021).
Cong, F., Mega, R. S., Chen, J., Day, C. S. & Martin, R. Trifluoromethylation of carbonyl and unactivated olefin derivatives by C(sp3)−C bond cleavage. Angew. Chem. Int. Ed. 62, e202214633 (2023).
Barton, D. H. R. & Samadi, M. The invention of radical reactions. Part XXV. A convenient method for the synthesis of the acyl derivatives of N-hydroxypyridine-2-thione. Tetrahedron 48, 7083–7090 (1992).
Jackl, M. K., Legnani, L., Morandi, B. & Bode, J. W. Continuous flow synthesis of morpholines and oxazepanes with silicon amine protocol (SLAP) reagents and Lewis acid facilitated photoredox catalysis. Org. Lett. 19, 4696–4699 (2017).
Acknowledgements
We are grateful for financial support from the National Natural Science Foundation of China (Grants 22071086, 21801165, 22201113, and 22371106), The Science and Technology Major Program of Gansu Province of China (22ZD6FA006, 23ZDFA015), and FRFCU (lzujbky-2020-ct01, lzujbky-2023-ct02). We also thank Prof. Chun-an Fan (Lanzhou University), Prof. Fumin Zhang (Lanzhou University), and Prof. Zhiwei Zuo (Shanghai Institute of Organic Chemistry) for helpful discussions and advice during the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
Z.X., J.J.G., and A.H. designed and conceived the project. Z.X. and W.P. conducted all the synthetic reactions. Z.X., W.P., J.H., J.S., and A.H. analyzed and interpreted the experiment data. W.P., J.J.G., and A.H. designed and performed the mechanistic experiments. Z.X. performed the HRMS experiments and analyzed the data. A.H. prepared the manuscript. Z.X. and J.J.G. prepared the Supplementary Information.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Aurelien de la Torre, and the other anonymous reviewers for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Xu, Z., Peng, W., Huang, J. et al. Photoinduced formal [4 + 2] cycloaddition of two electron-deficient olefins and its application to the synthesis of lucidumone. Nat Commun 15, 9748 (2024). https://doi.org/10.1038/s41467-024-54117-0
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-024-54117-0
This article is cited by
-
Photoinduced benzene ring contraction of arylhydrazines for the synthesis of fused pyridines
Nature Synthesis (2026)