Abstract
Methamphetamine (MA), as one kind of overflowed synthetic illicit drugs, has posed severe threats to health and social security. However, the on-site and visible fluorescence detection to MA remains limited. Herein, through covalently coupling diphenylacridine (DPA) and dimethylacridine (DMA) with pyridine, two ratiometric fluorescence probes (PyDPA and PyDMA) are constructed, which present rapid response, bathochromic-shifts over 100 nm and visible fluorescence color changes from blue to cyan upon the exposure to methylphenethylamine (MPEA, a simulant of MA). And the similar responses are observed for MA in confiscated samples. Further, a smartphone-based quantitative detected system is established to provide the on-site trace detection of MPEA as ppb level. Specially, PyDPA or PyDMA can identify MPEA from its interferences according to the unique ratiometric response based on their dual-emission-enhancement. Here, we show two ratiometric fluorescence probes to MA and MPEA with high potential for on-site application.
Similar content being viewed by others
Introduction
Synthetic illegal drugs, including ecstasy, methamphetamine (MA), 3,4-Methylenedioxymethamphetamine (MDMA) and Magu, are often utilized as medicine for their psychoactive effects. However, their highly addictive nature has posed serious threats to public health and social stability1. Among them, MA is the most representative. According to the World Drug Report 2022, there has been a five-fold increase in MA seizures over the past decade, reaching an unprecedented level2. Till now, some methods have been used for MA analysis in laboratories with high sensitivity, but they are usually time-consuming and not suitable for the on-site detection3,4,5,6,7,8,9,10. And though some portable detection methods such as immunoassay11,12 and Marquis reagent test13,14 have been already commercialized, they primarily focus on screening drug addicts15 rather than monitoring illegal drugs at their source. Therefore, to prevent drug circulation effectively, a portable, rapid and accurate detection method for MA is urgently needed.
Electrochemical16,17,18 and optical sensors19,20 are promising tools for monitoring MA for executing on-site detection and without substantive expertise. Compared to electrochemical detection that plays a primary role in terminal drug metabolism detection, the optical-based vapor detection has been expected to seize MA at the source or transportation21,22. Among the known optical methods, fluorescent sensors have shown to be an excellent option owing to the prominent features of non-invasive, easy operation23 and designability24 and great reusability25,26. Up to now, more and more research has been devoted to exploring fluorescent materials for MA or methylphenethylamine (MPEA, a simulant of MA) detection, and some results have been achieved, including fluorene-based fluorescent material27,28, quantum dots29, naphthalimide-based material30,31,32, boronate ester oxidizing reaction33 and so on. However, these probes usually encounter two problems: (1) MA or MPEA has a distinctive structure combining the characteristics of both aliphatic amine and aromatic amine (Fig. 1a). But it lacks the high nucleophilicity of aliphatic amines or the high energy level of aromatic amines. Thus, it is rather difficult to eliminate interference from these analogsamines. (2) Most existing probes rely on the photo-induced electron transfer (PET) mechanism and exhibit a single signal response as fluorescence on/off, and this kind of fluorescence response is easily affected by various non-analyte factors and leads to false positive signals, which affects the accuracy and selectivity of the detection. Hereby, other responded type fluorescent probes are needed.
a Chemical structures of methamphetamine (MA), methylphenethylamine (MPEA) and their interferences. b The chemical structures of PyDPA and PyDMA. c The diagrammatic sketch to detect and identify MPEA in this work; The blue curves correspond to photoluminescence (PL) spectra of the fluorescent films in air, the red curves represent the response upon exposure to different vapors (aliphatic amine, MPEA, aromatic amine and organic solvents, respectively), the blocks represent PyDPA and PyDMA films and the colors represent the fluorescence emitted by films after exposure different vapors under the excitation of 365 nm.
To fill the above-mentioned gap, there are two promising methods. One is adopting ratiometric fluorescence probes, the other is applying the chemical sensor array. The ratiometric fluorescence probes usually present two or more alterations in photoluminescence (PL) spectra to the analyte, which can facilitate the improvement in detection accuracy by self-calibrating signal correction34 and show great potential as ideal optical sensors with high accuracy and selectivity of the detection. Additionally, the ratiometric fluorescence probes can show distinct fluorescent color response35. As we know, human eyes are commonly less sensitive to the change of fluorescence brightness but higher sensitive to the color changes36, while the ratiometric fluorescence probes can benefit the visual recognition of the analyte. However, there is only one report of ratiometric fluorescence probes for MPEA till now37. To solve the problem of low selectivity caused by similar structures to the interferences, the other method is applying the strategy of chemical sensor array, since the different probes in the array can present various pattern responses to identify MPEA38,39. But the constructed chemical sensor arrays usually adopt multiple fluorescent probes, which increases the complexity and cost of the detection. Thus, from the viewpoint of practical uses, a more compact, efficient and simply structured sensor is urgently needed.
In this work, with diphenylacridine (DPA) and dimethylacridine (DMA) as the donors, two fluorescent probes (PyDPA and PyDMA, Fig. 1b) are designed and synthesized with a donor-acceptor (D-A) structure. As we know, D-A type organic luminescent materials, featured by intramolecular charge transfer (ICT) properties, are widely employed to construct ratiometric fluorescence probes40,41. Along with the modulation of ICT induced by analysts, the charge separation of probe molecules could be changed and cause alterations in PL intensity and wavelength42,43. As can be seen from Fig. 1b, PyDPA and PyDMA share similar chemical structures but different substituents at the 9th site of dihydracridine. As construction units for luminescent materials, dihydroacridine derivatives have received much attention owing to their multiple chemical reactive sites, easy modification, high hydrogen bond binding ability44,45,46. Nevertheless, nearly no reports have utilized dihydroacridine derivatives to detect MA or MPEA till now. Thus, here, dihydroacridine is selected as the electron donor, with the pyridine unit serving as the electron acceptor to build a D-A type fluorescent probe for MPEA detection. To our delight, these two film-based ratiometric fluorescence probes showed rapid fluorescent responses, accompanied by adequate limits of detection (1.2 ppb and 4.1 ppb). Moreover, these film probes exhibited visible color changes, which have rarely studied and reported so far. What should be noted is that different from the above-mentioned chemical sensor arrays involving multiple probes, in this work, a single probe-based sensor array was constructed and realized the accurate identification of MPEA by different response patterns between MPEA and its interferences (Fig. 1c). By this design, the present fluorescence probes (PyDPA and PyDMA) exhibit not only visible detection but also high selectivity, providing a prospective tool for on-site and real-time detection of MPEA.
Result
Synthesis and characterization
The synthetic details for PyDPA and PyDMA were provided in Supplementary Fig. 1 and their structures were characterized by 1H NMR spectroscopy, 13C NMR spectroscopy, Fourier transform infrared (FTIR) spectra and matrix-assisted laser desorption ionization time-of-flight mass spectrometry (MALDI-TOF MS). Comprehensive informations were available in Supplementary Figs. 2–8.
To reveal the frontier orbital electron distributions, firstly, we performed theoretical calculations by density functional theory (DFT) at the basis set level of B3LYP/6–31 G (d, p) (Supplementary Fig. 9). It can be found that PyDPA and PyDMA have little differences in the charge distribution: the hole is mainly located on the dihydroacridine while the electron is located on pyridine units, which proves the charge separation properties as designed.
Photophysical properties
The absorption and PL spectra of PyDPA and PyDMA in tetrahydrofuran (THF) (c = 1.0 × 10−5 M) and spin-coated film are depicted in Fig. 2a, b. The absorption band at 350 nm of PyDPA and PyDMA in THF corresponds to the large electron-delocalization effect and the π → π* electronic transition. For the spin-coated films, the absorption band shows a bathochromic shift of 5 nm compared with that in THF. Upon excitation at 350 nm, PyDPA and PyDMA show blue emission both in solution and film state, with the maximum emission peak at 420 nm.
a, b The absorption and PL spectra of PyDPA and PyDMA in tetrahydrofuran (c = 1.0 × 10−5 M) and spin-coated films. c, d Linear correlation of the orientation polarization (Δf) of solvent media versus the Stokes shift (υa-υb) (υ: wave number, a: absorption and b: fluorescence) for PyDPA and PyDMA.
To investigate the ICT properties of PyDPA and PyDMA, the absorption and PL spectra in various solvents with different polarities were recorded. As displayed in Supplementary Fig. 10, with the solvent polarity increase, the PL spectra display an obvious bathochromic shift, indicating a large dipolar change at the excited state of PyDPA and PyDMA. Furthermore, the Lippert–Mataga model was adopted to understand the impact of solvent polarity on the excited state of PyDPA and PyDMA (Supplementary Tables 1 and 2). From the plots of the orientation polarizability (Δf) versus the Stokes shift (υa-υb), the excited state transitional dipole moment (μe) of PyDPA and PyDMA are calculated to be 11.70 Debye and 10.24 Debye (Fig. 2c, d, based on the linear fitting results of y = 7099.4x + 2190.6, y = 7303.3x + 2478.7), further proving the ICT properties of PyDPA and PyDMA as expected.
Further, the photostability, the most important factor for fluorescent probes was investigated by continuously irradiating PyDPA and PyDMA spin-coated films under the excitation of 350 nm. As depicted in Supplementary Fig. 11, the PL intensity retains over 80% of its initial fluorescence even after 30 min, which demonstrates their good resistance to photochemical bleaching. Moreover, the humidity and storage stability were also investigated (Supplementary Fig. 12). It is found that storage for a month led to a 20% quenching in the PL intensity both in black and light environment while different humidity led to <5% fluctuation in fluorescence intensity, providing a foundation for sensing applications.
Sensor performance
The optimized PyDPA and PyDMA films (Supplementary Fig. 13) were applied to detect MPEA vapor. As shown in Fig. 3a, MPEA 316 ppm) vapor induces an enhancement at 420 nm for PyDPA film, accompanied by the rise of another emission at 520 nm, and this dual-emission-enhancement ratiometric response gradually reaches stability within 1 min (the PL intensity ratio of I520/I420 is 0.36). And the fluorescent color changes from blue to cyan, which can be discerned by naked-eye (inset in Fig. 3a). Similar phenomena are also observed for the PyDMA film (Fig. 3b). It should be noted that the visible recognition of MPEA vapor is rarely reported (Supplementary Table 3). The scarcity of visible recognition underscores the advantage of PyDPA and PyDMA, making them highly potential alternatives for on-site MPEA vapor detection.
a, b Time-dependent fluorescent response of PyDPA and PyDMA films to MPEA vapor, the inset: photographs of the films before and after exposure to MPEA vapors for 60 s, under the irradiation of 365 nm UV lamps. c, d PL intensity ratios (I520/I420) of PyDPA and PyDMA films upon exposure to MPEA vapor with various concentrations, the insets: linear relationship between I520/I420 and MPEA vapor with various concentrations; error bars represent the standard deviation (n = 3 independent replicates). e, f Recovery test of PyDPA and PyDMA films after the exposure to MPEA vapor (316 ppm) and air blowing, the red solid lines represent the response process by exposing to MPEA vapor and the blue dashed lines represent the recovery process by air blowing, the insets: photographs of the films in the recovery test for two cycles, under the irradiation of 365 nm UV lamps; The size of quartz cell in the insets of (a, b) and (e, f) is 1.25 cm × 1.25 cm × 4.5 cm.
Encouragingly, the response behavior of PyDPA and PyDMA films to varying concentrations of MPEA vapor was studied. As shown in Fig. 3c, d, the films exhibit good sensing capability to MPEA vapor within a wide range from 40 ppb to 316 ppm. The limit of detection (LOD) is calculated to be 0.06 ppm for PyDPA and 0.17 ppm for PyDMA (S/N = 3) based on the linear fitting results (y = 0.25x + 0.05, y = 0.08x + 0.03).
Further, the recyclability of probes was assessed and depicted in Fig. 3e, f, PyDPA and PyDMA films can be reused by simple air blowing and the the recovery process costs <60 s (see Supplementary Movie 1 and 2). It is notable that even after fifteen consecutive sensing cycles, the films still exhibit reliable fluorescence signals, indicating their recyclability.
To investigate the specificity of PyDPA and PyDMA probes to MPEA vapor, some commonly used solvents and organic amines, including primary amines, secondary amines and tertiary amines with diverse alkyl chains, cyclic and benzene structures that may potentially interfere with MPEA vapor detection were employed as interferences. First, it can be found from Supplementary Fig. 14 that the fluorescence color of PyDPA and PyDMA films change from blue to cyan upon exposure to MPEA vapor, which is distinctly discernible from other interferences. Fig. 4 depicts the PL spectra of PyDPA upon exposure to partial amines and solvent vapor (the details are provided in Supplementary Figs. 15, 16), three distinct response patterns are found by investigating the PL intensity changes at 420 nm and 520 nm: aliphatic amines cause PL quenching at 420 nm and enhancement at 520 nm (Fig. 4a–c), aromatic amines and organic solvents (Fig. 4d, e) cause the enhancement at 420 nm solely, while MPEA displays a unique response pattern with enhanced PL intensity at both 420 nm and 520 nm (Fig. 4f), setting it apart from other amines and solvents. Thus, the accurate identification of MPEA by PyDPA and PyDMA becomes feasible based on the three fluorescence response patterns.
a Cadaverine; b Benzylamine; c Diethylamine; d Aniline; e Tetrahydrofuran and f MPEA; The insets: photographs of PyDPA films after exposure to different vapors for 60 s, under the irradiation of 365 nm UV lamps. The size of quartz cell in the insets is 1.25 cm × 1.25 cm × 4.5 cm.
Identification of MPEA by a single probe-based fluorescence sensor array
To further simplify the visual recognition process of the PyDPA and PyDMA toward MPEA and interference, pseudocolor images and radar maps were elucidated in Fig. 5a–c, in which the colors and radius transitions correspond to changes of PL intensity at 420 and 520 nm and MPEA is conspicuously discernible. Afterward, these response fluorescence signals exhibited by PyDPA and PyDMA are individually subjected to principal component analysis (PCA). A trivariate data matrix is constructed, incorporating the dimensions of probe specificity, twelve distinct chemical species, and quintuplicate experimental replications. This matrix yields three canonical factors (PC1, 52.36%; PC2, 45.21%; PC3, 2.43% for PyDPA and PC1, 56.29%; PC2, 43.42%; PC3, 0.29% for PyDMA). As shown in Fig. 5d and e, two prominent canonical factors (PC1 and PC2) are employed to obtain the score plot, where MPEA is stratified into separate elliptical clusters, each enveloped by a 95% confidence interval. Importantly, MPEA can be well isolated from closely related amines, such as benzylamine, aniline, and diethylamine. These results underscore the significant capability of PyDPA and PyDMA in accurate identification of MPEA.
a Pseudo-color images of PyDPA and PyDMA toward MPEA and various interferences, the numbers of the color scale bar correspond to min/max axis values of I/I0 at 420 nm and 520 nm, respectively. b, c Radar plots corresponding to the response of PyDPA and PyDMA films toward MPEA and seven representative interferences, the radial axis represent I/I0 at 420 nm and 520 nm. d, e Principal component analysis score plot of PyDPA and PyDMA against various interferences; Put putrescine, Cad cadaverine, BZA benzylamine, c-HA cyclohexylamine, PA n-propylamine, DEA diethylamine, DMEN N, N’-Dimethyl ethylene diamine, TEA triethylamine, AN aniline, THF tetrahydrofuran, EtOH ethyl alcohol, Tol toluene.
Moreover, to validate the practical applicability of the film probes, we conducted MPEA detection in complex interference vapors. After exposing films to the complex interference vapors, only a slight enhancement at 420 nm is observed. However, as depicted in Supplementary Fig. 17, the introduction of MPEA vapor can lead to obvious enhancements of PL intensity at both 420 nm and 520 nm even though its vapor content is calulated to be as low as 4.4% in the mixed interference vapor (according to Eq. (1) and (2) in Supplementary Note 4). The anti-interference capability of PyDPA and PyDMA facilitates a potential analysis of MPEA in intricate environments.
Smartphone-assisted system enabled on-site MPEA vapor detection
To facilitate on-site and real-time detection of MPEA vapor, the calibrated semi-quantitative fluorescence cards based on PyDPA and PyDMA films were prepared. The fluorescent response photographs of PyDPA and PyDMA films to MPEA vapor with varying concentrations were taken under the irradiation of 365 nm UV lamps (Fig. 6a, b). It can be seen that as the concentration increases, the fluorescence colors of the cards are gradually changed, and the color gradations can be distinguished at six levels with naked-eye. These semi-quantitative fluorescence cards enable users to rapidly perceive the MPEA levels with the naked eye, akin to reading a pH paper.
The fluorescence response photographs of PyDPA (a) and PyDMA (b) films upon exposure to MPEA vapors with different concentrations ranging from 0 – 316 ppm, under the irradiation of 365 nm UV lamps; Plots of the linear relationship between chromatic aberration (ΔE) of PyDPA (c) and PyDMA (d) photographs and MPEA concentrations. e Schematic illustration of the smartphone-assisted system, adapted from Ref. 58 with permission, © 2021 Wiley-VCH GmbH.
Considering the limitation of the human eye in identifying subtle color variations, and with the aim of improving reliability, a quantitative sensing system was developed. Fig. 6e illustrates the components, including a built-in LED, sensing films, a gas cell and a smartphone. With the assistance of the smartphone (equipped with an App named Color Analyzer), any change of the sensing film will be recorded as photographs and converted to digital chromatic aberration (ΔE) according to equation (1). The detailed ΔE values are summarized in Supplementary Table 4. Notably, there is a good linear correlation between ΔE and MPEA concentrations within the range of 0-0.32 ppm, as illustrated in Fig. 6c, d. When the sensing films are exposed to an unknown concentration of MPEA, the fluorescence color is recorded and digitized. Using a data processing module, ΔE of the corresponding photographs is substituted into the established linear correlation in Fig. 6c, d for analysis, and the MPEA content is ultimately displayed on the phone screen. In particular, when ΔE is equal to 1, which is a threshold value to be discriminable47, the corresponding concentration of MPEA vapor is 1.20 ppb for PyDPA and 4.14 ppb for PyDMA, indicating that a ppb level detection for MPEA vapor could be realized. There is no doubt that the adequate LODs and visual recognition of the quantitative sensing platform facilitate their capability for practical applications in sensing trace levels of MPEA vapor.
Mechanism research
A series of mechanism investigations were performed. Firstly, the 1H NMR spectra of PyDPA and PyDMA in CD2Cl2 were collected with the MPEA incrementally introduced (Fig. 7a and Supplementary Fig. 18a). As the MPEA concentration increases, the resonance peaks of Hα (linked to N in the 10th site of PyDPA and PyDMA) undergo a downfield shift with Δδ values of 0.08 and 0.06 ppm, respectively. And the resonance peak of Hβ (linked to N in MPEA) exhibits an observable upfield-shift from 2.14 ppm to 1.53 ppm. These results indicate the increased de-shielding effect around Hα and the shielding effect around Hβ, which is typically ascribed to the formation of hydrogen bonds between PyDPA/PyDMA and MPEA48,49. Then, DFT and time-dependent density functional theory (TD-DFT) calculations were carried. As can be seen from Fig. 7b, after being reacted with MPEA, a decreased energy gap is presented, which corresponds to the bathochromic shifted emission peak at 520 nm. And the same results are also found for PyDMA, which are shown in Supplementary Fig. 18b.
a 1H NMR titration spectra (CD2Cl2, 500 MHz) of PyDPA upon the addition of MPEA (0-9 equiv), the y-axis represents absolute scale of the stacked spectra. b Frontier orbitals and energy diagrams of PyDPA and PyDPA + MPEA, frontier orbitals are generated by DFT calculations and energy diagrams are generated by TD-DFT calculations; Pseudo-color map of the femtosecond transient absorption (fs-TA) spectra of PyDPA in CH2Cl2 under an excitation wavelength of 345 nm (c), fs-TA spectra under different delay times from −1ps to 6 ns (d), and the early relaxation dynamics (e); Pseudo-color map of the fs-TA spectra of PyDPA+ MPEA in CH2Cl2 under an excitation wavelength of 345 nm; (f), fs-TA spectra under different delay times from −1ps to 5 ns (g), and the early relaxation dynamics (h).
Further, the femtosecond transient absorption (fs-TA) spectroscopy was employed to analyze the excited state process before and after interacting with MPEA. Fig. 7c shows a representative pseudo-color two-dimensional-fs-TA spectrum of PyDPA detected in the range of 410-675 nm with a scan time delay between -1ps and 6.0 ns. The negative absorption band maximized at 415 nm is ascribed to the stimulated emission (SE). The positive signal centered at 500 nm is the excited state absorption (ESA) process caused by the absorption of photons by PyDPA in the excited state. Fig. 7d shows the fs-TA spectra at different delay times. The SE signal gradually increases until it reaches the maximum at about 100 ps, and then decreases with increasing delay time. While the ESA signal appears immediately after being excited by the laser and gradually increases within 1 ps, then weakens and eventually disappears. The observation of an isosbestic point at ∼440 nm indicates only locally excited state (LE) emission50. Fig. 7e shows the kinetic fitting of PyDPA, where a 26.67 ps decay of rise component for 500 nm is resolved, then undergoes an identical population decay of 2.5 ns.
In comparison, the fs-TA spectra of PyDPA + MPEA are much more complicated (Fig. 7f). In addition to the original SE band at 415 nm, a broad SE signal appears centered from 550 to 570 nm, which aligns with the steady-state PL spectra of PyDPA + MPEA. From the fs-TA spectra at different delay times (Fig. 7g) we can see that the ESA signal associated with the S1 → Sn transition (which centered at 500 nm in PyDPA) is interfered by a series of relaxations. The ESA signal appears at 500 nm within 1 ps, which subsequently increases accompanied by a blueshift rapidly, reaching a maximum at 100 ps with a positive signal at 470 nm. It is worth noting that a shoulder peak appears with the gradual decrease of ESA at 470 nm over time from 1.5 ns – 6 ns, which eventually results in a red-shifted ESA signal of PyDPA + MPEA maximized at 530 nm. In sharp contrast to PyDPA, the kinetic fitting of fs-TA spectra of PyDPA + MPEA (Fig. 7h) is rather complicated. This complexity arises from the combination of the absorbance of LE, intramolecular charge transfer (ICT) and their associated SE. Nevertheless, compared with the relaxation at 500 nm, the kinetic fitting at 470 nm shows relatively longer rise and decay components of 1.27 ns and 4.57 ns, respectively. These results firmly support the existence of an intense ICT process in PyDPA + MPEA51. Combined with the structure of PyDPA, we can conclude that the enhanced charge density caused by hydrogen bond formation and the released ICT relaxation can answer the bathochromic shifted emission peak at 520 nm. A similar conclusion can be drawn from the fs-TA spectra of PyDMA and PyDMA + MPEA (Supplementary Fig. 18).
It is important to note that above DFT calculations and fs-TA analysis are conducted in gas and solution phases, respectively, which introduces limitations as they do not fully account for intermolecular interactions in the solid state. To address this limitation, we carried out Fourier transform infrared (FTIR) spectroscopy to investigate the interactions between the probe and MPEA in the solid state (Supplementary Fig. 19). The FTIR spectra revealed a noticeable red-shift in the characteristic N-H stretching band after interacting with MPEA, showing wavenumber variations of 25 cm-1 (from 3250 cm-1 to 3225 cm-1 for PyDPA) and 23 cm-1 (from 3251 cm-1 to 3228 cm-1 for PyDMA). This result provides direct experimental evidence of the interaction between PyDPA/PyDMA with MPEA in the solid state, which is not accounted in the DFT calculations and fs-TA analysis, further validating the sensing mechanism proposed in this work.
Since the enhancement at 420 nm is only observed in the films, no similar enhancement is observed when MPEA is added to PyDPA/PyDMA solution (Supplementary Fig. 20), the microenvironment changes in the aggregation state of thin films might be a reasonable inference52,53,54. To further verify this inference, the fluorescence behavior of PyDPA and PyDMA in the aggregated state was studied. Firstly, concentration-dependent absorption and PL spectra of PyDPA and PyDMA films were collected (Supplementary Fig. 21). Upon increasing the concentration from 0.5 to 2.0 mg/ml, no significant change is observed in absorption spectra. However, the PL spectra display a diminish at 420 nm and the emergence of another peak, indicating an electronic excited-state interaction and excimer formation between PyDPA/PyDMA molecules55. Further, as the concentration increases, the time-resolved fluorescence spectra reveal a much slower decay process and a transition from single to double exponential decay (Supplementary Fig. 22 and Supplementary Table 6), further supporting the excimer formation in the aggregated state56.
Fig. 8 exhibits the fluorescence decay curves of PyDPA films before and after exposure to MPEA vapor (monitored at 520 nm), τ of PyDPA film is shortened from 2.16 to 1.49 ns upon the exposure of MPEA. According to the detailed fitting data (presented in Table 1 and Supplementary Table 7), τ1 remains almost unchanged while τ2 decreases from 4.31 ns to 2.21 ns, which are assigned to monomer decay and excimer decay, respectively. The apparent alteration demonstrated the destruction of the excimer and conversion to a monomer57. Further research conducted at 420 nm could confirm this view that PyDPA films exhibit consistent lifetimes after exposure to MPEA (Supplementary Fig. 23).
Time-resolved PL spectra of PyDPA (a) and PyDMA (b) films before and after the exposure to MPEA vapor, when monitored at 520 nm.
Overall, the detection mechanism for MPEA by PyDPA and PyDMA is concluded that the Hα on the dihydracridine attracts the lone pair electrons of -NH on MPEA to form hydrogen bond complexes, regulating the ICT property and ultimately realizing ratiometric fluorescence response. Meanwhile, MPEA might be favored its insert into the excimers, causing the destruction of excimer and conversion to monomer, thereby resulting in the PL intensity enhancement at 420 nm.
Validating the suitability of sensing films toward real MA in confiscated samples
To validate the practical applicability of the developed sensing films, it is essential to assess their suitability for confiscated samples containing MA, even though the films have performed well with MPEA (the simulant of MA). The evaluation of the performance was carried out by analyzing the real MA samples, which are subpackaged from the confiscated samples. The detected MA samples were analyzed by High-Performance Liquid Chromatography (HPLC) with 78% purity (Supplementary Table 8). To our delight, both PyDPA and PyDMA show similar ratiometric responses in the presence of varying masses of MA, ranging from 10 mg to 0.25 mg (Fig. 9). Further, as shown in the insets of Fig. 9, the fluorescence color change of sensing films could be discriminated by naked-eye when exposed to MA, which strongly supports the application potential of developed probes.
Mass-dependent fluorescent response of PyDPA (a) and PyDMA (b) films to MA vapor, volatilized by confiscated samples of different masses; The inset: photographs of the films before and after exposure to MA vapors, under the irradiation of 365 nm UV lamps, the size of quartz cell in the insets is 1.25 cm × 1.25 cm × 4.5 cm.
Further, since the real MA samples are usually adulterated with some else drugs (Heroin and some pharmaceutical drugs like phenacetin, phenobarbital, levamisole, and caffeine), ruling out their interference is the prerequisite for a successful detection. The response spectra of the films toward these individual drugs and toward the binary/ternary mixtures that contain MA were collected (Supplementary Figs. 24, 25). Fortunately, the developed films present no response to the pharmaceutical drugs themselves but exhibit a clear dual-emission-enhancement response to both binary and ternary mixtures, which contain equal mass of MA and phenobarbital or phenobarbital/caffeine. This indicates that MA could be detected in the tested mixtures.
Discussion
In summary, the ratiometric response of PyDPA and PyDMA films toward MPEA has broken the limitations of conventional fluorescent probes for illicit drugs, resulting in a visible color change. We have figured out that the arresting sensing performances are attributed to the enhancement of ICT, which is caused by the formation of hydrogen bond. And the proposed mechanism is further validated by the NMR data, DFT calculations and fs-TA spectra. Interestingly, our selectivity study data suggests that PyDPA or PyDMA exhibits three response patterns toward aliphatic amine, aromatic amine and MPEA, which is further applied as a single probe-based chemical sensor array to distinguish MPEA from other 11 kinds of amines. Moreover, the two probes were integrated into a quantitative sensing platform with the assistance of a smartphone serving as the color variation indicator. The platform exhibited a ppb level response and reversible detection (by air blowing) for MPEA vapor. Ultimately, the developed probes explored the potential for the detection of MA in seized samples, presenting similar analytical performance to MPEA, proving the applicability of this work for on-site screening of suspected MA samples. All in all, this work not only developed two ratiometric fluorescent probes for visual detection and accurate identification of illicit drugs but also offered insights into the design of highly applicable sensing materials for analyte detection.
Methods
Materials and apparatus
All the chemicals were reagent grade and used as received without further purification. Super-dry solvent including tetrahydrofuran (THF) with molecular sieves was purchased from Innochem. and used as received. Dry solvents were purchased from Beijing Chemical Works (Beijing, China) or Energy co., Ltd. The illicit drugs were provided by the Criminal Investigation Police University of China, and used directly without further purification. (Note: the illicit drug samples used are all seized drugs by the local police officers from the illicit market). The nuclear magnetic resonance (1H NMR) spectra were recorded on the Bruker AVANCEIII 500 spectrometer at 500 MHz at 298 K and tetramethyl silane (TMS) (δH = 0 ppm) as the internal standard. MALDI-TOF MS spectra were recorded on a Brucker Autoflex speed TOF/TOF mass spectrometer with DCTB as a matrix. Elemental analyses were performed on the flash EA 1112 spectrometer. Fourier transform infrared (FTIR) spectra were recorded on a Brucker VERTEX 80 V FTIR spectrometer. An Edinburgh fluorescence spectrometer (FLS980) was used for the fluorescence decay. The lifetime of the excited states was measured by the time-correlated single photon counting method (detected at the peak of the PL) under the excitation of a laser (375 nm) with a pulse width of 50 ps. UV–vis absorption spectra were recorded on the model UV–vis-2550 spectrophotometer. Fluorescence spectra were carried out using the model RF-5301PC Spectro-fluorophotometer.
synthesis of PyDPA and PyDMA
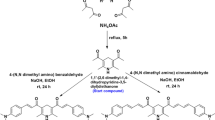
2,7-dibromo-9,9-diphenyl-9,10-dihydroacridine (300 mg, 0.61 mmol, 1.0 eq), pyridin-4-ylboronic acid (172 mg, 1.4 mmol, 2.3 eq), potassium carbonate aqueous solution (210 mg, 1.52 mmol, 2.5 eq) was dissolved in a mixture of THF (20 mL) and H2O (10 mL). After sealing, the raw material mixture was frozen for 8 min with liquid nitrogen, and then vacuumed for 5 min. Bis (triphenylphosphine) palladium (II) chloride (21.4 mg, 0.031 mmol, 5%) was added, and then frozen and vacuumed three times, oxygen was removed. The reaction mixture was heated to 80 °C and stirred for 24 h under argon. After removal of the solvent under reduced pressure, the residue was extracted with CH2Cl2 and H2O. The combined organic layer was dried over Na2SO4. After removal of the solvent under reduced pressure, the residue was purified by the neutral aluminum oxide column chromatography (MeOH/Petroleum ether = 1/15, Rf = 0.1). After purification, a yellow solid was obtained (118 mg, 40% yield). 1H NMR (500 MHz, DMSO) δ 9.76 (s, 1H), 8.74 (d, J = 6.0 Hz, 2H), 8.51 (d, J = 5.9 Hz, 4H), 7.84 (d, J = 4.8 Hz, 2H), 7.70 (d, J = 8.3 Hz, 2H), 7.35 (dd, J = 17.0, 6.9 Hz, 6H), 7.28 (t, J = 7.2 Hz, 2H), 7.15 – 7.07 (m, 3H), 6.99 (d, J = 7.6 Hz, 3H). 13C NMR (126 MHz, CD2Cl2) δ 148.69, 147.75, 144.02, 128.20, 127.36, 127.11, 126.12, 124.91, 124.45, 121.49, 112.70. MALDI-TOF: C35H25N3 calcd. [M+] 487.6060; found: 487.1903. Anal. calcd for C35H25N3: C 86.21, H 5.17, N 8.62; found: C 86.04, H 5.54, N 8.94.
PyDMA was synthesized by the same procedure as above with a yield of 65%. 1H NMR (500 MHz, DMSO) δ 9.49 (s, 1H), 8.57 (d, J = 6.1 Hz, 4H), 7.86 (d, J = 1.7 Hz, 2H), 7.72 (d, J = 1.4 Hz, 4H), 7.59 (d, J = 10.2 Hz, 2H), 6.95 (d, J = 8.3 Hz, 2H), 1.69 (s, 6H). 13C NMR (126 MHz, CD2Cl2) δ 150.13, 147.98, 138.76, 130.44, 129.63, 125.65, 124.54, 120.65, 114.34, 53.42, 31.10. MALDI-TOF: C25H21N3 calcd. [M+] 363.4640; found: 363.3029. Anal. calcd for C35H25N3: C 82.61, H 5.82, N 11.46; found: C 82.90, H 5.44, N 11.06.
Film preparation
The films were prepared by a spin-coating method. Briefly, 1 mg PyDPA/PyDMA and 19 mg polystyrene (PS) were dissolved in THF (1 mL). Then the films were obtained from their solution (60 μL) on quartz at 1500 rpm for 30 s. The films were vacuum dried for 30 min at 45oC.
Detection of gaseous analytes
Detection of MPEA vapor: 20 μL MPEA was added into the quartz cell bottom, then the quartz cell was sealed to get a saturated vapor (naturally volatilized at room temperature for 5 min). We adopt a static method for sensing measurement—1 mL MPEA was injected into a 20 mL bottle and sealed for 24 h at room temperature. Then different volumes of saturated vapor were injected into the sealed quartz cell with microliter syringes, and different vapor pressures were obtained by air dilution. The films were placed in a sealed quartz cell containing different concentrations of MPEA vapor, and after the films were allowed to interact with MPEA vapor for 60 s, The fluorescence spectrum and photographs were collected by spectrophotometer and iPhone 14 respectively.
Detection of MA vapor: The hydrochloride salts of the illicit drugs were converted to their free-base form using the following procedure: an equal amount of Na2CO3 and the drugs under tests were added to a bottle, then ground with a glass rod for 1 min and finally, the first hand vapor above the illicit drug was collected for tests.
Preparation of calibrated quantitative fluorescence cards
Preparing MPEA vapor standard samples with concentrations ranging from 0.04 ppm – 316 ppm using the static air dilution method. Subsequently, expose the prepared PyDPA and PyDMA fluorescence films to a series of MPEA vapor concentrations ranging from 0.04 ppm to 316 ppm, and wait for 60 s. Illuminate the fluorescence films with a 365 nm UV lamp and record the color changes on the fluorescence card. Calibrate the PyDPA and PyDMA fluorescence films based on the concentration of the MPEA standard samples to obtain quantitative fluorescence cards.
Photograph processing by smartphone
The color composition in CIELab color space of the photographs was obtained from the optical photographs by an openly accessible smartphone app named Color Analyzer (version 3.0.1, Copyright @ 2024 kamusoft, developed by Satoshi Nakamura). The chromatic aberration (Eq. 1) applied in CIELab color space represents the quantitative difference between two colors, being equal to the Euclidean distance (dC) between the two tristimulus values:
Femtosecond time-resolved transient absorption spectroscopy
A femtosecond transient absorption (fs-TA) spectroscopic setup was used for recording TA spectra. Ti: sapphire laser system (Coherent Astrella, 100 fs, 1 kHz) is the fundamental light source. The pump pulse was generated by an optical parametric amplifier (TOPAS-Prime). Then a supercontinuum white light was used as the probe pulse, which is produced by focusing the fundamental frequency light into a sapphire plate. The pump and probe pulse meet at the sample position. After passing through the sample, the probe pulse is focused into an optical fiber that couples the light to a spectrometer (Helios fire, Ultrafast System). In the current experiment, the excitation wavelength was set at 345 nm, the sample was placed into a 1-mm-thick fused silica flow cell in order to avoid sample damage. The decay kinetics was scanned at a temporal resolution of 14 fs with a time window of 8 ns. A chirp program corrected the group velocity dispersion effect of solvents. The pump-probe delay scan results for each measurement are the averages of three repeated data.
Global analysis was used to resolve the individual spectral components from the congested spectrum, the program was implemented based on Surface Xplorer, the method of singular value decomposition (SVD) was employed, and the principal spectral components can be obtained from global analysis directly.
Statistics and reproducibility
Experiments about sensing performance were performed from 3 independent replicates with similar results. For the plots in Fig. 5d, e, experiment was repeated 5 times independently, and the statistical analyses were performed using the Origin 2023b (principal component analysis v1.50), confidence level was chosen as 0.05 (95% confidence interval).
Data availability
The data that support the findings of this work are available within the article and its supplementary information. Source data are provided with this paper.
References
Amin-Esmaeili, M. et al. Patterns of reduced use and abstinence in multi-site randomized controlled trials of pharmacotherapies for cocaine and methamphetamine use disorders. Drug Alcohol Depend. 226, 108904 (2021).
United Nations Office on Drugs and Crime. World Drug Report 2022, United Nations Publication Press. https://www.unodc.org/unodc/en/data-and-analysis/world-drug-report-2022 (2022).
Shi, J. W., Zhou, J. F., He, X. & Zhang, Y. Rapid analysis of four amphetamines in urine by self-made pipette-tip solid-phase extraction followed by GC-MS/MS. J. Chromatogr. Sci. 58, 569–575 (2020).
Saar-Reismaa, P. et al. Determination of illegal drugs in oral fluid by portable capillary electrophoresis with deep UV excited fluorescence detection. Anal. Chem. 90, 6253–6258 (2018).
Peng, Y., Gautam, L. & Hall, S. W. The detection of drugs of abuse and pharmaceuticals in drinking water using solid-phase extraction and liquid chromatography-mass spectrometry. Chemosphere 223, 438–447 (2019).
Wang, S. et al. Rapid on-site detection of illegal drugs in complex matrix by thermal desorption acetone-assisted photoionization miniature ion trap mass spectrometer. Anal. Chem. 91, 3845–3851 (2019).
Xie, Y., Wu, S., Chen, Z., Jiang, J. & Sun, J. Rapid nanomolar detection of methamphetamine in biofluids via a reagentless electrochemical aptamer-based biosensor. Anal. Chim. Acta 1207, 339742 (2022).
Hong, Y. et al. Optoplasmonic hybrid materials for trace detection of methamphetamine in biological fluids through SERS. ACS Appl. Mater. Interfaces 12, 24192–24200 (2020).
Von Luders, L. et al. Functionalisation of graphene sensor surfaces for the specific detection of biomarkers. Angew. Chem. Int. Ed. Engl. 62, e202219024 (2023).
Rafiee, B., Fakhari, A. & Ghaffarzadeh, R. M. Impedimetric and stripping voltammetric determination of methamphetamine at gold nanoparticles-multiwalled carbon nanotubes modified screen printed electrode. Sens. Actuat. B-Chem. 218, 271–279 (2015).
Petrie, M. et al. Cross-reactivity studies and predictive modeling of “bath salts” and other amphetamine-type stimulants with amphetamine screening immunoassays. Clin. Toxicol. 51, 83–91 (2013).
Swortwood, M. J., Hearn, W. L. & DeCaprio, A. P. Cross-reactivity of designer drugs, including cathinone derivatives, in commercial enzyme-linked immunosorbent assays. Drug Test. Anal. 6, 716–727 (2014).
Bell, S. C. & Hanes, R. D. A microfluidic device for presumptive testing of controlled substances. J. Forensic Sci. 52, 884 (2007).
Prunty, S., Carmany, D., Dhummakupt, E. S. & Manicke, N. E. Combining presumptive color tests, pressure-sensitive adhesive-based collection, and paper spray-mass spectrometry for illicit drug detection. Analyst 148, 3274–3284 (2023).
Dong, R. L., Weng, S. Z., Yang, L. B. & Liu, J. H. Detection and direct readout of drugs in human urine using dynamic surface-enhanced raman spectroscopy and support vector machines. Anal. Chem. 87, 2937–2944 (2015).
Dragan, A. M. et al. Investigating the electrochemical profile of methamphetamine to enable fast on-site detection in forensic analysis. Talanta 255, 124208 (2023).
Schram, J. et al. Local conversion of redox inactive molecules into redox active ones: a formaldehyde based strategy for the electrochemical detection of illicit drugs containing primary and secondary amines. Electrochim. Acta 367, 137515 (2021).
Dokuzparmak, E., Brown, K. & Dennany, L. Electrochemiluminescent screening for methamphetamine metabolites. Analyst 146, 3336–3345 (2021).
Mao, K. et al. A novel colorimetric biosensor based on non-aggregated Au@Ag core–shell nanoparticles for methamphetamine and cocaine detection,. Talanta 175, 338–346 (2017).
Mao, K., Ma, J., Li, X. & Yang, Z. Rapid duplexed detection of illicit drugs in wastewater using gold nanoparticle conjugated aptamer sensors. Sci. Total Environ. 688, 771–779 (2019).
Ding, N. N. et al. Film-based fluorescent sensors: from sensing materials to hardware structures. Sci. Bull. 68, 546–548 (2023).
Li, Z. J. et al. Achieving gas pressure-dependent luminescence from an AIEgen-based metal-organic framework. Nat. Commun. 13, 2142 (2022).
Li, Z. et al. Accurate identification of cysteine by a nucleophilic-reaction-induced conversion of an open-shell luminescent radical into closed-shell molecule. Sens. Actuat. B Chem. 376, 132934 (2023).
Liu, W., Zheng, P., Xia, Y., Li, F. & Zhang, M. A simple AIE probe to pesticide trifluralin residues in aqueous phase: ultra-fast response, high sensitivity, and quantitative detection utilizing a portable platform. Talanta 269, 125352 (2023).
Zhu, Z. et al. Direct active site at the van der Waals heterostructure interface with synthetic drug analogue N-methylphenethylimine ultrasensitivity. ACS Sens. 8, 1318–1327 (2023).
Li, D. Y. et al. Ultra-sensitive, selective and repeatable fluorescence sensor for mthanol based on a highly emissive 0D hybrid lead-free perovskite. Angew. Chem. Int. Ed. Engl. 61, e202206437 (2022).
Vannoy, K. J., Tarolla, N. E., Kauffmann, P. J., Clark, R. B. & Dick, J. E. Electroanalysis-detecting methamphetamine in aerosols by electroanalysis in a soap bubble wall. Anal. Chem. 94, 6311–6317 (2022).
Loch, A. S., Burn, P. L. & Shaw, P. E. Fluorescent sensors for the detection of free-base illicit drugs-effect of tuning the electronic properties. Sens. Actuat. B-Chem. 387, 133766 (2023).
Elmizadeh, H., Bardajee, G. R. & Moaddeli, A. Ultrasensitive and rapid detection of methamphetamine in forensic biological fluids using fluorescent apta-nanobiosensors based on CdTe quantum dots. Microchem. J. 189, 108519 (2023).
Liu, K. et al. Non-contact identification and differentiation of illicit drugs using fluorescent films. Nat. Commun. 9, 1695 (2018).
Liu, K. et al. Unambiguous discrimination and detection of controlled chemical vapors by a film‐based fluorescent sensor array. Adv. Mater. Technol. 4, 1800644 (2019).
Chen, M. et al. Trifluoromethyl‐substituted perylene diimides for rapid and sensitive film‐based fluorescence detection of methamphetamine and cocaine hydrochloride. Adv. Funct. Mater. 34, 2311328 (2024).
Li, K. et al. Detecting methylphenethylamine vapor using fluorescence aggregate concentration quenching materials. Sens. Actuat. B-Chem. 334, 129629 (2021).
Chen, Y. C. et al. A ratiometric fluorescent probe for rapid detection of hydrogen sulfide in mitochondria. Angew. Chem. Int. Ed. 52, 1688–1691 (2013).
Jia, R. N. et al. Amine-responsive cellulose-based ratiometric fluorescent materials for real-time and visual detection of shrimp and crab freshness. Nat. Commun. 10, 795 (2019).
Shen, Y., Wei, Y., Zhu, C., Cao, J. & Han, D. M. Ratiometric fluorescent signals-driven smartphone-based portable sensors for onsite visual detection of food contaminants. Coord. Chem. Rev. 458, 214442 (2022).
Fan, T. C. et al. Naked-eye visible solid illicit drug detection at picogram level via a multiple-anchored fluorescent probe. ACS Sens. 1, 312–317 (2016).
Yen, Y. T. et al. Nanomaterial ‐ based sensor arrays with deep learning for screening of illicit drugs. Adv. Mater. Technol. 7, 2200243 (2022).
Yan, M. et al. Tri-probe fluorescent sensor array for a wide concentration range and high precision identification of aqueous organic amines. Sens. Actuat. B-Chem. 358, 131519 (2022).
Yan, C. X. et al. Fluorescence umpolung enables light-up sensing of acetyltransferases and nerve agents. Nat. Commun. 12, 3869 (2021).
Chen, Y. C., Cheng, Y. Q. & Sun, M. T. Nonlinear plexcitons: excitons coupled with plasmons in two-photon absorption. Nanoscale 14, 7269–7279 (2022).
Guliyev, R., Coskun, A. & Akkaya, E. U. Design strategies for ratiometric chemosensors: modulation of excitation energy transfer at the energy donor site. J. Am. Chem. Soc. 131, 9007–9013 (2009).
Miao, X. & Wu, C. X.& Li, F. & Zhang, M. Fast and visual detection of biogenic amines and food freshness based on ICT‐induced ratiometric fluorescent probes. Adv. Funct. Mater. 33, 2212980 (2023).
Guo, J. J. et al. Achieving high-performance nondoped OLEDs with extremely small efficiency roll-off by combining aggregation-induced emission and thermally activated delayed fluorescence. Adv. Funct. Mater. 27, 1606458 (2017).
Yang, H., Liang, Q. Q., Han, C. M., Zhang, J. & Xu, H. A phosphanthrene oxide host with close sphere packing for ultralow-voltage-driven eficient blue thermally activated delayed fluorescence diodes. Adv. Mater. 29, 1606458 (2017).
Wu, X. et al. Robust sky-blue aggregation-induced delayed fluorescence materials for high-performance top-emitting OLEDs and single emissive layer white OLEDs. Chem. Eng. J. 451, 138919 (2023).
Ke, Y. et al. Electronic tuning in reaction-based fluorescent sensing for instantaneous and ultrasensitive visualization of ethylenediamine. Angew. Chem. Int. Ed. Engl. 61, e202203358 (2022).
Wazio, J. A., Jimenez, V., Soparawalla, S., John, S. & Moehring, G. A. Hydrogen exchange of rhenium(VII) heptahydridobis(triphenylphosphine) with water, aniline, methanol, and itself. Inorg. Chim. Acta 362, 159 (2009).
Sutter, K., Aucar, G. A. & Autschbach, J. Analysis of proton NMR in hydrogen bonds in terms of lone-pair and bond orbital contributions. Chem. Eur. J. 21, 18138 (2015).
Ding, N. et al. Bi-ortho-carborane unit-riveted perylene monoimides: structure-tuned optical switches for electron transfer and robust thin film-based fluorescence sensors. CCS Chem. 12, 2922–2932 (2023).
Vazquez, R. J. et al. New direct approach for determining the reverse intersystem crossing rate in organic thermally activated delayed fluorescent (TADF) emitters. J. Am. Chem. Soc. 142, 8074–8079 (2020).
Liu, L., Liu, C., Wang, L., Shen, X. C. & Chen, H. A disaggregation-induced emission strategy to obtain dual-emission fluorescence-enhanced probe for visualization of SO2 fluctuation in living cells, tumor tissue and zebrafish. Sens. Actuat. B Chem. 371, 132542 (2022).
Zhai, D. T., Xu, W., Zhang, L. Y. & Chang, Y. T. The role of “disaggregation” in optical probe development. Chem. Soc. Rev. 43, 2402–2411 (2014).
Battistelli, G. et al. Local detection of pH-induced disaggregation of biocompatible micelles by fluorescence switch ON. Chem. Sci. 13, 4884–4892 (2022).
Förster, T. Excimers. Angew. Chem. Ed. Engl. 8, 333–343 (1969).
Yoshihara, K., Kasuya, T., Inoue, A. & Nagakura, S. Time resolved spectra of pyrene excimer and pyrene-dimethylaniline exciplex. Chem. Phys. Lett. 9, 469–472 (1971).
Qi, S. et al. Turn-off near-infrared fluorescent probe for free bilirubin detection constructed by enhanced excimer emission. Anal. Chim. Acta. 1238, 340657 (2023).
Wang, J. et al. A fluorescent metal–organic framework for food real-time visual monitoring. Adv. Mater. 15, 2008020 (2021).
Acknowledgements
This work was financially supported by the Science and Technology Development Program of Changchun City (Grant Number 23xxx19) and the National Natural Science Foundation of China (grant number 21875083) to M. Z.
Author information
Authors and Affiliations
Contributions
M.Z. and F.L. proposed the research direction, X.M. designed various experiments, performed the material characterizations, fluorescence testing, and data analysis with guidance from M.Z. and F.L. C.W. performed material synthesis, Y.X. provided the detection materials, S.Y. performed femtosecond spectroscopic experiments and helped to analyze, M.Z. and X.M. wrote the manuscript. All authors discussed the results and commented on the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Martin Baumgarten, Jinhong Guo and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Miao, X., Wu, C., Xia, Y. et al. Ratiometric fluorescence probes for visible detection and accurate identification of MPEA vapor. Nat Commun 15, 10641 (2024). https://doi.org/10.1038/s41467-024-55011-5
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-024-55011-5