Abstract
This study presents a copper-catalyzed, substrate-controlled regio- and enantioselective intermolecular hydrosilylation method capable of accommodating a broad scope of alkenes and prochiral silanes. The approach offers an efficient and versatile pathway to generate enantioenriched linear and branched alkyl-substituted Si-stereogenic silanes. Key features of this reaction include mild reaction conditions, simple catalytic systems, compatibility with diverse substrates, high yields and enantioselectivities.
Similar content being viewed by others
Introduction
Silicon, like carbon, belongs to Group IVA in the periodic table and is the second most abundant element in the Earth’s crust1,2. Comparatively, while chiral molecules containing a stereogenic carbon atom have been extensively studied and practically utilized, there has also been substantial interest in synthesizing organic silicon molecules with Si-stereogenic centers3. These molecules play a pivotal role in various fields such as organic synthesis, functional materials, and biomedicines4,5,6,7. Traditionally, the creation of silicon-centered chirality has heavily relied on stoichiometric chiral reagents or auxiliaries8,9,10,11,12,13,14,15,16,17,18. To improve efficiency and minimize waste, novel strategies for synthesizing Si-stereogenic silanes have been developed through both transition-metal-catalyzed19,20,21,22,23,24,25,26,27,28,29,30 and metal-free organocatalyzed31,32,33,34,35,36,37 desymmetrization of prochiral organosilanes. Additionally, significant advancements have been made recently in constructing silicon-stereogenic silanes from racemic starting materials through dynamic kinetic asymmetric transformation (DYKAT)38,39,40 and metal-catalyzed kinetic resolution (KR)41,42. Among various Si-stereogenic organosilanes, monohydrosilanes have attracted considerable attention due to their unique reactivity and promising applications (Fig. 1a)7.
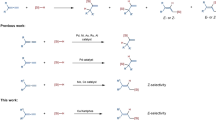
a Catalytic asymmetric synthesis of Si-stereogenic monohydrosilanes. b Metal-catalyzed intermolecular enantioselective hydrosilylation of C-C unsaturated bonds for synthesis of chiral monohydrosilanes (previous work). c Copper-catalyzed intermolecular regiodivergent and stereoselective hydrosilylation of alkenes (this work).
Within the realm of strategies developed for the synthesis of chiral monohydrosilanes, metal-catalyzed asymmetric intermolecular hydrosilylation of C–C unsaturated bonds emerges as an efficient approach (Fig. 1b)19,20,21,22,23,24,25,26,27,28,29,30,43,44,45,46,47,48,49,50,51. It is noteworthy that while enantioselective hydrosilylation of alkenes has been extensively studied for creating a C-stereogenic center with a C-Si bond52,53,54,55,56,57,58,59,60,61,62,63,64,65,66,67,68,69,70,71,72,73, only limited progress has been made in constructing a Si-stereogenic center via this strategy. To date, there have only been two reported instances of hydrosilylation of terminal olefins for the preparation of linear alkyl-substituted Si-stereogenic silanes49,50. In 2018, Hou and colleagues reported a groundbreaking, catalytic, and enantioselective method for synthesizing Si-stereogenic silanes through Sc-catalyzed intermolecular hydrosilylation of terminal alkenes49. Subsequently, He and co-workers demonstrated an example using Rh-catalysis, yielding Si-stereogenic compounds with moderate enantioselectivities50. In contrast, rare protocols have successfully enabled the simultaneous creation of both a C- and a Si-stereocenter41,48,51,74,75,76,77,78. Only few examples of the simultaneous construction of a C- and a Si-stereogenic center in acyclic systems through intermolecular reactions have been reported via noble transition metal-catalyzed enantioselective Si–H bond insertion of carbene species77,78 or Co-catalyzed intermolecular hydrosilylation of 1,3-dienes48. However, the catalytic asymmetric synthesis of chiral monohydrosilanes with both a C- and a Si-stereogenic center, using readily available alkenes as substrates through the hydrosilylation process, remains unexplored. Therefore, the development of highly efficient and robust methodologies catalyzed by abundant metals to access chiral hydrosilanes with a Si-stereogenic center or with both a C- and a Si-stereogenic center is highly desirable. Copper-catalyzed transformations are pivotal in advancing cutting-edge organic synthesis, owing to the widespread availability of copper metal, as well as the low cost, reduced toxicity, and easy commercial accessibility of copper catalysts79,80,81. Recently, a great deal of progress has been made in the study of copper-catalyzed asymmetric hydrosilylation of unsaturated C–C bonds47,59,69,76,82,83,84,85,86,87,88. However, there is a notable absence of copper-catalyzed enantioselective hydrosilylation of alkenes for the construction of Si-stereogenic monohydrosilanes in acyclic systems.
Herein, we present a Cu-catalyzed intermolecular regio- and enantioselective hydrosilylation of alkenes with prochiral silanes for the synthesis of diverse enantioenriched hydrosilane products (Fig. 1c).
Results
Evaluation of reaction conditions for the copper-catalyzed hydrosilylation of allylbenzene
Using allylbenzene (1a) as the model substrate, the Cu-catalyzed hydrosilylation of alkenes was first investigated by assessing the steric effects of different dihydrosilanes, including PhMeSiH2 (2a), mesityl(methyl)silane (2b), and Ph(tBu)SiH2 (2c). Various copper precursors and ligands were also examined for the model reaction. The results of selected experiments are summarized in Table 1. The reactions between allylbenzene and the dihydrosilanes R1R2SiH2 were conducted using 4 mol% Cu(OAc)2 and 8 mol% (R,R)-Ph-BPE (L1) at 40 °C (entries 1−3). Notably, it was observed that the reaction of allylbenzene with mesityl(methyl)silane (2b) produced the anti-Markovnikov product at 40 °C with moderate conversion and a 94:6 enantiomeric ratio (entry 2). Despite PhMeSiH2 (2a) exhibiting a higher conversion rate compared to mesityl(methyl)silane (2b) and Ph(tBu)SiH2 (2c), the latter demonstrated better enantioselectivities. A slight enhancement in enantioselectivity was noted upon transitioning from Cu(OAc)2 to CuOAc as the copper precursor (entry 4). Conversely, the use of CuCl and Cu(Etacac)2 as the catalysts did not yield any reaction (entries 5−6). By increasing the catalyst loading to 8 mol%, the yield of 3ab could be elevated to 58% (entry 7). Encouragingly, incorporating a secondary ligand proved advantageous for this reaction (entries 8−11)89, with CyJohnPhos delivering the optimal outcome (95:5 enantiomeric ratio; entry 11). This may be attributed to the role of PAr3 as a secondary ligand in stabilizing the CuH species. Ultimately, the best result was obtained when conducting the reaction of 1a with 2b (3.0 equiv) in the presence of CuOAc (10 mol%), (R,R)-Ph-BPE (L1, 11 mol%), and CyJohnPhos (11 mol%) at 40 °C for 2 days, resulting in a 75% yield and a 95:5 enantiomeric ratio (entry 12).
Scope of linear-selective hydrosilylation of alkenes
Following the identification of an active catalyst and optimized conditions for the anti-Markovnikov hydrosilylation of allylbenzene (entry 11 in Table 1), we proceeded to explore the substrate scope. Key findings from this investigation are outlined in Fig. 2. Initially, we examined the hydrosilylation of various alkenes using 2b or 2c. Allylbenzene derivatives containing both electron-donating and electron-withdrawing groups could efficiently undergo reactions with mesityl(methyl)silane (2b) to yield the desired chiral linear products, typically achieving moderate to excellent yields with good enantioselectivities (3ab–3fb).
aConditions: 1 (0.2 mmol, 1.0 equiv), 2 (3.0 equiv), CuOAc (10 mol%), (R,R)-Ph-BPE (11 mol%) and CyJohnPhos (11 mol%) were stirred at 40 °C for 2 d under N2 atmosphere. bIsolated yields. cThe er values were determined by chiral HPLC analysis. d4 d. e2c (6.0 equiv) was added. fThe dr value was determined by chiral HPLC analysis. gCu(Etacac)2 (10 mol%) and (R,R)-Ph-BPE (11 mol%) were used. Cu(Etacac)2 = copper(II) ethylacetoacetate.
In general, electron-withdrawing allylbenzene proved to be a superior substrate (3db) that yielded the hydrosilylation product with higher efficiency. The efficiency and enantioselectivity of the desired products were slightly influenced as the carbon chain prolonged, as observed in 3hb and 3ib. Additionally, heteroaryl-substituted alkenes served as suitable substrates, producing chiral silanes with high efficiencies and enantioselectivities (3jb–3lb). Various functional groups such as amino (3 mb), phenoxy (3nb), thioether (3ob), silyloxy (3pb), halogens (3qb) were all compatible. These reactions proceeded smoothly, yielding the corresponding tertiary silane products with good yields (57%–91%) and high enantioselectivities (91:9 to 96:4 er). In cases where the substrate contained both terminal and internal olefin units, the reaction selectively occurred at the less sterically hindered terminal olefin, leaving the internal olefin moiety intact (3rb). When tert-butyl group-substituted silane was utilized, the desired products were obtained with improved efficiency and enantioselectivity (3cc–3uc). The absolute configuration of 3tc was determined through X-ray diffraction analysis (CCDC: 2358701). Furthermore, 1,4-diallylbenzene was selectively hydrosilylated, resulting in the bis-silane product 3vc with a yield of 71%, an enantiomeric ratio of 98:2, and a diastereomeric ratio of 88:12. Prochiral silanes were examined under identical conditions (Fig. 2). The efficiency and enantioselectivity of the product were significantly influenced by the steric hindrance of the silane. Reactions involving silanes bearing various bulky aryl or alkyl groups exhibited high enantioselectivities, albeit with a slight decrease in efficiencies (3ac–3af). Fortunately, when Cu(Etacac)2 was used as the catalyst, good yields and enantioselectivities were also achieved for less sterically bulky silanes (3aa, 3ak). A variety of readily available alkylphenylsilanes proved to be suitable substrates (3ah–3aj). Although the desired product was obtained when the arylmethylsilane contained electron-withdrawing group (3ag), the efficiency and enantioselectivity were negatively affected.
Evaluation of reaction conditions for the copper-catalyzed hydrosilylation of styrene
Based on the aforementioned investigation, we aimed to expand the substrate scope by incorporating aryl alkenes (Table 2). Despite compound (R,R)-5ak demonstrating excellent efficiency and a favorable enantiomeric ratio, the diastereomeric ratio achieved under the optimized conditions (entry 11, Table 1) was only moderately satisfactory (entry 1). It is worth noting that introducing a second ligand in this reaction didn’t have a positive effect (entry 2). When switching from CuOAc to Cu(OAc)2 as the copper precursor (entry 3), a slight increase in diastereoselectivity was observed. Although different chiral ligands were tested, the desired outcomes were not achieved (entries 4–6). Encouragingly, increasing the amounts of the chiral ligand yielded positive results (entries 7–8). Reducing the reaction time to 36 h did not significantly alter the results, resulting in the target product (R,R)-5ak being obtained in a 91% isolated yield, with a 98:2 er and up to a 95:5 dr ratio (entry 9).
Scope of branched-selective hydrosilylation of alkenes
Under the optimized conditions, we explored the substrate scope as illustrated in Fig. 3. Enantioenriched branched silanes were successfully synthesized, incorporating halogenated (5bk), electron-rich (5ck), electron-deficient (5fk) aryl groups, or a fused aromatic ring (5gk). Yields varied between 70% and 94%, with diastereomeric ratios ranging from 86:14 to 95:5, and enantiomeric ratios reaching up to 98:2. Notably, the reaction did not accommodate aryl bromide and iodide substrates. A styrene derivative carrying a methylthio group (5ek) was obtained in moderate yield but displayed poor diastereo- and enantioselectivity. Of significance was the selective transformation of aryl alkenes bearing trisubstituted olefin moieties with the silane reagent resulting in high efficiency and stereoselectivity (5dk). Furthermore, a wide array of heteroaryl-substituted alkenes proved to be suitable substrates for the production of chiral silanes exhibiting both C- and Si-stereogenic centers efficiently, enantioselectively, and with moderate diastereoselectivities (5ik–5lp). The absolute configuration of the chiral branched alkylsilane product was conclusively determined through X-ray crystallographic analysis of compound 5lp (CCDC: 2358711).
aConditions: 4 (0.2 mmol, 1.0 equiv), 2 (3.0 equiv), Cu(OAc)2 (4.0 mol%) and (R,R)-Ph-BPE (8.0 mol%) were stirred at 40 °C for 36 h under N2 atmosphere. bIsolated yields. cThe er values were determined by chiral HPLC analysis. dThe dr values were determined by GC analysis or 1H NMR of the crude reaction mixture. e72 h. fIn extra dry cyclohexane (2.0 M).
Next, the investigation focused on the scope of prochiral silanes. Reactions with readily available arylmethylsilanes exhibited remarkable efficiencies, diastereoselectivities, and enantioselectivities (5ca, 5cl–5cp). Another alkylarylsilane (5ci) was converted to the target product with a yield of 92%, a diastereomeric ratio (dr) of 94:6, and an enantiomeric ratio (er) of 97:3. However, diarylsilanes and sterically hindered silanes such as mesityl(methyl)silane (2b) and Ph(tBu)SiH2 (2c) were found to be unsuitable substrates for these reactions.
Gram-scale reactions and synthetic applications
The reaction was successfully conducted on a gram scale (Fig. 4a). This led to the production of enantioenriched linear alkylsilane 3db in a 82% yield, with a 94:6 er ratio. Additionally, the enantioenriched branched alkylsilane 5ck was formed in a 99% yield, with a 91:9 dr ratio and a 98:2 er ratio. To showcase the versatility of chiral monohydrosilanes further, a series of stereospecific transformations involving the stereogenic Si-H bond were performed. In the presence of Pt(PPh3)4, 3tc could be transformed into silicon-stereogenic optically active silyl-borane 6 with moderate yield and without any loss in enantioselectivity. Subsequently, Pt-catalyzed hydrosilylation of 3sa with 5-phenyl−1-penten furnished enantioenriched silane 7 in 79% yield with 93:7 dr.
a Gram-scale synthesis of linear and branched alkylsilanes. b Derivatizations of chiral silanes.
The branched alkylsilanes are important synthetic intermediates and can undergo various transformations. Subjecting 5ck to hydrosilylation conditions with 4-fluorostyrene unexpectedly yielded the dehydrocoupling product 8 with a satisfactory yield of 76%, 98:2 er, and 91:9 dr. Furthermore, by dehydrogenative coupling of 5dk with benzyl alcohol, the corresponding chiral silyl-ether 9 was obtained in an impressive yield of 89% without any compromise in diastereopurity and enantiopurity using CuCl/Ph-BPE as a catalyst.
Mechanistic investigation
Deuterium labeling experiments were conducted as part of the investigation into the reaction mechanism. Initially, the isotopically labeled substrate methyl(phenyl)silane-d2 (2a-d2) was subjected to standard conditions, leading to the formation of deuterated products 3aa-d2 and 5aa-dn, illustrated in Fig. 5a. To enhance our comprehension of the reversibility factors that influence the reaction steps, multiple control experiments were performed. When styrene (4a), 2a-d2, and (4-methoxyphenyl)(methyl)silane (2m) were simultaneously employed under standard conditions, the integration of the Si- H/D peaks of 5aa-dm and 5am-dm give a ratio of approximately 1:1. The obtained result provides further evidence for the reversibility of migratory insertion and β-hydride elimination90. Another crossover experiment was performed using 1.0 equiv 2a-d2 and 2m reacting without an alkene under standard conditions. The presence of deuterium crossover was confirmed through 1H NMR analysis. These findings strengthen our conclusion that the generation of copper hydride species, migratory insertion, and β-hydride elimination display reversibility (Fig. 5b). To gain further insight into the rate-determining step (RDS), a kinetic isotope effect (KIE) was measured by comparing the rates of hydrosilylation of 1a with 2c or 2c-d2 at 40 °C. Using the initial rates method, the KIE was found to be 2.09 ± 0.09. Similarly, the KIE for branched-selective hydrosilylation was determined to be 1.64 ± 0.14. This can be attributed to the primary kinetic isotope effect associated with Si–H bond cleavage in the rate-determining step, which involves a cyclic four-center transition state. This observation is consistent with both the calculated and measured values for related models91,92. Based on these data, we propose a potential reaction mechanism for the copper-catalyzed intermolecular regiodivergent and stereoselective hydrosilylation of alkenes (Fig. 5c).
a Deuterium labeling experiments. b Crossover study. c Proposed reaction pathway.
High-resolution mass spectrometry analysis of the CuH mixture solution revealed major peaks attributed to a monomeric [Ph-BPE]Cu species (for details, see Supplementary Information, Figs. S8 and S9). Nonlinear effect studies on the enantiomeric composition of the chiral ligand (R,R)-Ph-BPE (L1) and the product 3ab or 5ca indicated a linear relationship, supporting the monomeric nature of the active catalysts.
Computational studies
To elucidate the origin of the regio- and stereoselectivities observed in the reaction, we resorted to DFT studies on the hydride-insertion and the subsequent metathesis steps (for details, see Supplementary Data 1). The potential energy surface leading to the linear products was explored with 1-butene as the model substrate (Fig. 6a). The migratory insertion step from S1 to TS1 has a barrier of 10.9 kcal/mol, generating the linear alkyl Cu(I) P1, with an energy downhill of 23.1 kcal/mol. Subsequent silane association leads to further energy downhill of 3.3 kcal/mol. From S2, the conformation space of the metathesis step was mapped systematically and three representative conformers (conf-A ~ conf-C) were located. Our calculation shows that the most energetically favored pathway proceeds through transition structure TS2_conf-B_R (in favor of the R-product) with a barrier of only 13.4 kcal/mol, which is much more favored (by 13.0 kcal/mol) than the backward β-H elimination event (26.4 kcal/mol), in congruent with the absence of H/D scrambling observed experimentally (Fig. 5a). The second lowest-energy TS is TS2_conf-A_S which is 3.9 kcal/mol higher, in good agreement of the sense and degree of enantiocontrol observed experimentally.
DFT studies on the migratory insertion and the subsequent metathesis steps for the reaction with (a) 1-butene and (b) styrene as starting materials, respectively. Energies are in kcal/mol and bond lengths in Å. Level of theory: M06/def2-tzvpp//M06 /6-31 G(d)/SDD(Cu). Distortion Interaction analysis (DIAS) of the stereo-determining TSs of the reactions using 1-butene (c) and styrene (d).
For the reaction with styrene as starting material, sampling of the conformational space of the hydrocapration in the branched product forming transition structures located four representative conformers (re/si pair of conf-a, conf-b_re and conf-c_si), where the relative energies of the re-configured conformers are lower than those of the si-conterparts, and TS1’_conf-b_re was the one with the lowest energy (Fig. 6b). The Cu–H insertion TS forming terminal C–Cu bond (TS1’_r.r.) is 9.7 kcal/mol higher than TS1’_conf-b_re, consistent with the exclusive regioselectivity experimentally observed. The most stable conformation TS1’_conf-b_re leads to the lowest energy benzyl-Cu(I) intermediate P1’_conf-b_re, which then associates with the phenyl silane to give the σ-complex S2’ with a 3.9 kcal/mol energy drop. Subsequently, the conformational space of the metathesis with Ph(Me)SiH2 was mapped. Of these transition structures, TS2’_conf-B_RR and TS2’_conf-D_RR converged to the same structure which was found to be lowest in energy. Notably, there is only a small energy difference of 2.0 kcal/mol between the β-H elimination of the benzylic Cu(I) S2’ and its metathesis (26.9 vs 24.9 kcal/mol), suggesting of a partially reversible hydrometallation step before the rate-determining metathesis. This can explain the isotope scrambling observed in the mechanistic experiments (Fig. 5a, b). According to distortion interaction analysis (DIAS) of these TSs, it can be concluded that for both substrates, the configuration established at the silicon atom are largely a result of differences of the distortion of the silane moiety (47.1 vs 51.9 kcal/mol in Fig. 6c, and 52.1 vs ca. 58.9 kcal/mol, Fig. 6d).
Discussion
In summary, we have successfully developed a copper-catalyzed, highly selective hydrosilylation method for alkenes. Various monosubstituted alkenes with aromatic and aliphatic groups efficiently reacted with hydrosilanes to produce enantiomerically enriched alkyl-substituted silanes with a Si-stereogenic center or Si/C two stereogenic centers under substrate influence. Control experiments suggested that metathesis likely plays a crucial role as the rate-determining step. The outstanding regioselectivity and enantioselectivity were further confirmed through DFT calculations. Additionally, the unreacted Si−H bond in the chiral silane products provides opportunities for further derivatization.
Methods
General procedure for copper-catalyzed enantioselective linear-selective hydrosilylation of alkenes: an oven-dried 10 mL Schlenk tube equipped with a stirring bar was charged with CuOAc (10 mol%), (R,R)-Ph-BPE (L1, 11 mol%) and CyJohnPhos (11 mol %). Then the silane (0.6 mmol, 3.0 equiv) was added under nitrogen atmosphere and the mixture was stirred at 40 °C for 15 min. Subsequently, the alkene (0.2 mmol, 1.0 equiv) was added by syringe at ambient temperature under nitrogen atmosphere. The reaction mixture was stirred at 40 °C for 2 days. The resultant solution was concentrated, and the crude product was purified by column chromatography.
General procedure for copper-catalyzed enantioselective branched-selective hydrosilylation of alkenes: an oven-dried 10 mL Schlenk tube equipped with a stirring bar was charged with Cu(OAc)2 (4 mol%), (R,R)-Ph-BPE (L1, 8 mol%). Then the silane (0.6 mmol, 3.0 equiv) was added under nitrogen atmosphere and the mixture was stirred at 40 °C for 15 min. Subsequently, the alkene (0.2 mmol, 1.0 equiv) was added by syringe at ambient temperature under nitrogen atmosphere. The reaction mixture was stirred at 40 °C for 36 h. The resultant solution was concentrated, and the crude product was purified by column chromatography.
Data availability
The authors declare that all other data supporting the findings of this study are available within the article and Supplementary Information files, and also are available from the corresponding author on request. The crystallographic data of compounds 3tc and 5lp are available at Cambridge Crystallographic Data Centre under the deposition number CCDC: 2358701 (3tc) and 2358711 (5lp). These data can be obtained free of charge from The Cambridge Crystallographic Data Center via www.ccdc.cam.ac.uk/data_request/cif.
References
Tréguer, P. et al. The Silica Balance in the World Ocean: a Rees-timate. Science 268, 375 (1995).
Struyf, E., Smis, A., Van Damme, S., Meire, P. & Conley, D. J. The Global biogeochemical silicon cycle. Silicon 1, 207 (2009).
Schwarz, J. Atypical Elements in Drug Design, (Springer, Heidelberg, 2016).
Igawa, K. & Tomooka, K. Chiral Silicon Molecules, in Organosilicon Chemistry: Novel Approaches and Reactions, (eds Hiyama, T. & Oestreich, M.) (Wiley, 2019).
Xu, L.-W., Li, L., Lai, G.-Q. & Jiang, J.-X. The recent synthesis and application of silicon-stereogenic silanes: a renewed and significant challenge in asymmetric synthesis. Chem. Soc. Rev. 40, 1777–1790 (2011).
Bauer, J. O. & Strohmann, C. Recent progress in asymmetric synthesis and application of difunctionalized silicon-stereogenic silanes. Eur. J. Inorg. Chem. 2016, 2868–2881 (2016).
Wu, Y. & Wang, P. Silicon-stereogenic monohydrosilane: synthesis and applications. Angew. Chem. Int. Ed. 61, e202205382 (2022).
Sommer, L. H. Stereochemistry, Mechanism and Silicon; an Introduction to the Dynamic Stereochemistry and Reaction Mechanisms of Silicon Centers (McGraw-Hill, 1965).
Kipping, F. S. XXII. Organic Derivatives of Silicon. Part II. The Synthesis of Benzylethylpropylsilicol, its Sulphonation, and the Resolution of the Dl-sulphonic Derivative into its Optically Active Components. J. Chem. Soc. 91, 209–240 (1907).
Sommer, L. H. & Frye, C. L. Optically active organosilicon compounds having reactive groups bonded to asymmetric silicon. displacement reactions at silicon with pure retention and pure inversion of configuration. J. Am Chem. Soc. 81, 1013 (1959).
Yamamoto, K., Kawanami, Y. & Miyazawa, M. Kinetic resolution of (±)-cyclohex−1-enylsilanols by the sharpless asymmetric epoxidation. J. Chem. Soc. Chem. Commun. 5, 436–437 (1993).
Strohmann, C., Hörnig, J. & Auer, D. Synthesis of a highly enantiomerically enriched silyllithium compound. Chem. Commun. 2, 766–767 (2002).
Trzoss, M., Shao, J. & Bienz, S. Preparation of a ‘Si-centered’ chiral auxiliary by resolution. Tetrahedron Asymmetry 15, 1501–1505 (2004).
Rendler, S., Auer, G. & Oestreich, M. Kinetic resolution of chiral secondary alcohols by Dehydrogena-tive Coupling with recyclable silicon-stereogenic silanes. Angew. Chem. Int. Ed. 44, 7620–7624 (2005).
Rendler, S., Auer, G., Keller, M. & Oestreich, M. Preparation of a privileged silicon-stereogenic silane: classical versus kinetic resolution. Adv. Synth. Catal. 348, 1171–1182 (2006).
Rendler, S., Oestreich, M., Butts, C. P. & Lloyd-Jones, G. C. Intermolecular chirality transfer from silicon to carbon: interrogation of the two-silicon cycle for Pd-catalyzed hydrosilylation by stereoisotopochemical crossover. J. Am. Chem. Soc. 129, 502–503 (2007).
Igawa, K., Takada, J., Shimono, T. & Tomooka, K. Enantioselective synthesis of Silanol. J. Am. Chem. Soc. 130, 16132–16133 (2008).
Igawa, K., Kokan, N. & Tomooka, K. Asymmetric synthesis of chiral silacarboxylic acids and their ester derivatives. Angew. Chem. Int. Ed. 49, 728–731 (2010).
Xu, L.-W. Desymmetrization catalyzed by transition-metal complexes: enantioselective formation of silicon-stereogenic silanes. Angew. Chem. Int. Ed. 51, 12932–12934 (2012).
Shintani, R. Recent advances in the transition‐metal‐catalyzed Enantioselective synthesis of Silicon-Stereogenic Organosilanes. Asian J. Org. Chem. 4, 510–514 (2015).
Cui, Y.-M., Lin, Y. & Xu, L.-W. Catalytic synthesis of chiral organoheteroatom compounds of silicon, phosphorus, and sulfur via asymmetric transition metal-catalyzed C–H functionalization. Coord. Chem. Rev. 330, 37–52 (2017).
Shintani, R. Recent progress in catalytic enantioselective desymmetrization of prochiral organosilanes for the synthesis of silicon-stereogenic compounds. Synlett 29, 388–396 (2018).
Ye, F. & Xu, L.-W. A glimpse and perspective of current organosilicon Chem-istry from the view of hydrosilylation and synthesis of silicon-stereogenic silanes. Synlett 32, 1281–1288 (2021).
Zheng, L., Nie, X.-X., Wu, Y. & Wang, P. Construction of Si-Stereogenic Silanes through C−H activation approach. Eur. J. Org. Chem. 2021, 6006–6014 (2021).
He, C. & Yuan, W. Enantioselective C–H functionalization toward Silicon-Stereogenic Silanes. Synthesis 54, 1939–1950 (2022).
Ge, Y., Huang, X., Ke, J. & He, C. Transition-metal-catalyzed Enantioselective C-H silylation. Chem. Catal. 2, 2898–2928 (2022).
Xu, L.-W., Huang, W.-S., Wang, Q. & Yang, H. State-of-the-art advances in enantioselective transition-metal-mediated reactions of Silacyclobutanes. Synthesis 54, 5400–5408 (2022).
Zeng, Y. & Ye, F. Research progress on new catalytic reaction systems for asymmetric synthesis of Silicon-Stereogenic center containing compounds. Chin. J. Org. Chem. 43, 3388–3413 (2023).
Li, L., Huang, W., Xu, Z. & Xu, L. Catalytic asymmetric Silicon-carbon bond-forming transformations based on Si-H functionalization. Sci. China Chem. 66, 1654–1687 (2023).
Zhao, J., Ge, Y. & He, C. Construction of Silicon-Stereogenic center via catalytic asymmetric Si-H/X-H dehydrogenative coupling. Chin. J. Org. Chem. 43, 3352–3366 (2023).
Zhou, M. et al. Construction of Tetrasubstituted Silicon-Stereogenic Silanes via conformational isomerization and N-Heterocyclic Carbene-Catalyzed desymmetrization. ACS Catal. 12, 7781–7788 (2022).
Liu, H. et al. Stereoselective access to Silicon-Stereogenic Silacycles via the carbene-catalyzed desymmetric benzoin reaction of Siladials. ACS Catal. 12, 9864–9871 (2022).
Liu, H., Zhou, H., Chen, X. & Xu, J. N-Heterocyclic Carbene-Catalyzed desymmetrization of siladials to access Silicon-Stereogenic organosilanes. J. Org. Chem. 87, 16127–16137 (2022).
Zhou, H. et al. Organocatalytic asymmetric synthesis of Si-Stereogenic Silyl ethers. J. Am. Chem. Soc. 144, 10156–10161 (2022).
Lu, X. et al. Catalytic synthesis of functional Silicon-Stereogenic Silanes through Candida antarctica Lipase B catalyzed remote desymmetrization of Silicon-Centered Diols. Eur. J. Org. Chem. 2013, 5814–5819 (2013).
Zhang, X.-X., Gao, Y., Zhang, Y.-X., Zhou, J. & Yu, J.-S. Highly enantioselective construction of multifunctional Silicon-Stereogenic Silacycles by asymmetric enamine catalysis. Angew. Chem. Int. Ed. 62, e202217724 (2023).
Liao, X., Zhou, H., Chen, X. & Xu, J. Isothiourea-Catalyzed Acylative desymmetrization of Silicon-Centered Bisphenols. Org. Lett. 25, 3099–3103 (2023).
Zeng, Y. et al. Rhodium-catalyzed dynamic kinetic asymmetric Hydrosi-lylation to Access Silicon-Stereogenic Center. Angew. Chem. Int. Ed. 61, e202214147 (2022).
Zhou, H. et al. Organocatalytic DYKAT of Si-Stereogenic Silanes. J. Am. Chem. Soc. 145, 4994–5000 (2023).
Hu, T. et al. Enantioconvergent construction of Stereogenic Silicon via Lewis Base-Catalyzed Dynamic Kinetic Silyletherification of Racemic Chlorosilanes. Nat. Commun. 14, 4900 (2023).
Gou, F.-H., Ren, F., Wu, Y.-C. & Wang, P. Catalytic kinetic resolution of monohydrosilanes via Rhodium-catalyzed enantioselective intramolecular hydrosilylation. Angew. Chem. Int. Ed. 63, e202404732 (2024).
Gan, W.-E. et al. Copper-catalyzed asymmetric synthesis of Silicon-Stereogenic Benzoxasiloles. Angew. Chem. Int. Ed. 63, e202317973 (2024).
Igawa, K., Yoshihiro, D., Ichikawa, N., Kokan, N. & Tomooka, K. Catalytic enantioselective synthesis of Alkenylhydrosilanes. Angew. Chem. Int. Ed. 51, 12745–12748 (2012).
Wen, H., Wan, X. & Huang, Z. Asymmetric synthesis of Silicon-Stereogenic Vinylhydrosilanes by Cobalt-Catalyzed Regio- and Enantioselective Alkyne Hydrosilylation with Dihydrosilanes. Angew. Chem. Int. Ed. 57, 6319–6323 (2018).
Xie, J.-L. et al. Palladium-catalyzed Hydrosilylation of Ynones to Access Silicon-stereogenic Silylenones by Stereospecific Sromatic Interaction-assisted Si-H Activation. Sci. China Chem. 64, 761–769 (2021).
Ling, F. et al. An unusual autocatalysis with an air-stable Pd complex to promote enantioselective synthesis of Si-stereogenic Enynes. Chem. Sci. 14, 1123–1131 (2023).
Xu, J.-L., Wang, Z.-L., Zhao, J.-B. & Xu, Y.-H. Enantioselective construction of Si-stereogenic Linear Alkenylhydrosilanes via copper-catalyzed Hydrosilylation of Alkynes. Chem. Catal. 4, 100887 (2024).
Wang, L., Lu, W., Zhang, J., Chong, Q. & Meng, F. Cobalt-Catalyzed Regio-, Diastereo- and Enantioselective Intermolecular Hydrosilylation of 1,3-Dienes with Prochiral Silanes. Angew. Chem. Int. Ed. 61, e202205624 (2022).
Zhan, G. et al. Enantioselective construction of Silicon-Stereogenic Silanes by Scandium-Catalyzed Intermolecular Alkene Hydrosilylation. Angew. Chem. Int. Ed. 57, 12342–12346 (2018).
He, T. et al. Enantioselective construction of Si-Stereogenic center via Rhodium-Catalyzed intermolecular hydrosilylation of alkene. Chem. Eur. J. 26, 17011–17015 (2020).
Zhao, Z.-Y. et al. Enantioselective rhodium-catalyzed desymmetric hydrosilylation of cyclopropenes. ACS Catal. 9, 9110–9116 (2019).
Gibson, S. E. & Rudd, M. The role of secondary interactions in the asymmetric palladium-catalysed hydrosilylation of olefins with monophosphane ligands. Adv. Synth. Catal. 349, 781–795 (2007).
Uozumi, Y. & Hayashi, T. Catalytic asymmetric synthesis of optically Active 2-alkanols via Hydrosilylation of 1-alkenes with a Chiral Monophosphine-palladium Catalyst. J. Am. Chem. Soc. 113, 9887–9888 (1991).
Jensen, J. F., Svendsen, B. Y., Ia Cour, T. V., Pedersen, H. L. & Jo-hannsen, M. Highly Enantioselective Hydrosilylation of Aromatic Alkenes. J. Am. Chem. Soc. 124, 4558–4559 (2002).
Guo, X.-X. et al. Asymmetric Palladium-catalyzed hydrosilylation of styrenes using efficient chiral spiro phosphoramidite ligands. Tetrahedron. Asymmetry 15, 2231–2234 (2004).
Junge, K., Wendt, B., Enthaler, S. & Beller, M. Palladium-Catalyzed Enantioselective Hydrosilylation of Aromatic Olefins. ChemCatChem 2, 453–458 (2010).
Weber, I. & Jones, G. B. Bidentate Planar Chiral η6-arene Tricarbonyl Chromium(0) Complexes: Ligands for Catalytic Asymmetric Alkene Hydrosilylation. Tetrahedron Lett. 42, 6983–6986 (2001).
Zhang, F. & Fan, Q.-H. Synthesis and application of bulky phosphoramidites: highly effective monophosphorus ligands for asymmetric hydrosilylation of styrenes. Org. Biomol. Chem. 7, 4470–4474 (2009).
Gribble, M. W. Jr, Pirnot, M. T., Bandar, J. S., Liu, R. Y. & Buchwald, S. L. Asymmetric Copper Hydride-Catalyzed Markovnikov Hydrosilylation of Vinylarenes and Vinyl Heterocycles. J. Am. Chem. Soc. 139, 2192–2195 (2017).
Naito, T., Yoneda, T., Ito, J. & Nishiyama, H. Enantioselective hydrosilylation of aromatic alkenes catalyzed by Chiral Bis(oxazolinyl)phenyl–Rhodium Acetate Complexes. Synlett 23, 2957–2960 (2012).
Kitayama, K., Uozumi, Y. & Hayashi, T. Palladium-catalysed Asymmetric Hydrosilylation of Styrenes with a New Chiral Monodentate Phosphine Ligand. J. Chem. Soc. Chem. Commun. 15, 1533–1534 (1995).
Cheng, B., Liu, W. & Lu, Z. Iron-catalyzed highly enantioselective hydrosilylation of unactivated terminal alkenes. J. Am. Chem. Soc. 140, 5014–5017 (2018).
Cheng, B., Lu, P., Zhang, H., Cheng, X. & Lu, Z. Highly enantioselective cobalt-catalyzed hydrosilylation of alkenes. J. Am. Chem. Soc. 139, 9439–9442 (2017).
Chen, J., Cheng, B., Cao, M. & Lu, Z. Iron-catalyzed asymmetric hydrosilylation of 1,1-Disubstituted Alkene. Angew. Chem. Int. Ed. 54, 4661–4664 (2015).
Wen, H., Wang, K., Zhang, Y., Liu, G. & Huang, Z. Cobalt-Catalyzed Regio- and Enantioselective Markovnikov 1,2-Hydrosilylation of Conjugated Dienes. ACS Catal. 9, 1612–1618 (2019).
Chen, C. et al. Iron-catalyzed asymmetric hydrosilylation of Vinyl-cyclopropanes via Stereospecific C-C Bond Cleavage. iScience 23, 100985 (2020).
You, Y. & Ge, S. Asymmetric Cobalt-Catalyzed Regiose-lective Hydrosilylation/Cyclization of 1,6-Enynes. Angew. Chem. Int. Ed. 60, 12046–12052 (2021).
Gu, X.-W. et al. Stereospecific Si-C coupling and remote control of axial chirality by enantioselective palladium-catalyzed hydrosilylation of Maleimides. Nat. Commun. 11, 2904 (2020).
Zhang, M., Ji, Y., Zhang, Z. & Zhang, C. Copper-catalyzed highly selective hydrosilylation of Silyl or Boryl Alkene: a method for preparing chiral geminated Disilyl and Borylsilyl Reagents. Org. Lett. 24, 2756–2761 (2022).
Bai, D. et al. Highly Regio- and Enantioselective Hydrosilylation of gem-Difluoroalkenes by Nickel Catalysis. Angew. Chem. Int. Ed. 61, e202114918 (2022).
Zhang, W.-W. & Li, B.-J. Enantioselective hydrosilylation of β,β-Disubstituted Enamides to Construct α-Aminosilanes with Vicinal Stereocenters. Angew. Chem. 135, e202214534 (2023).
Zhang, W.-R., Zhang, W.-W. & Li, B.-J. Amide-Directed, Rhodium-Catalyzed Enantioselective Hydrosilylation of unactivated internal alkenes. Org. Lett. 25, 1667–1672 (2023).
Zhu, C., Zhou, Y., Yang, J., Feng, X. & Dong, S. Lanthanide-catalyzed Regio- and Enantioselective Hydrosilylation of Aryl-substituted Terminal and Internal Alkenes. Org. Chem. Front. 10, 1263–1269 (2023).
Huang, Y.-H. et al. Enantioselective synthesis of Silicon-Stereogenic Monohydrosilanes by Rhodium-Catalyzed intramolecular hydrosilylation. Angew. Chem. Int. Ed. 61, e202113052 (2022).
Lu, W., Zhao, Y. & Meng, F. Cobalt-Catalyzed Sequential Site- and Stereoselective Hydrosilylation of 1,3- and 1,4-Enynes. J. Am. Chem. Soc. 144, 5233–5240 (2022).
Fu, B. et al. Enantioselective Copper-Catalyzed Sequential Hydrosilylation of Arylmethylenecyclopropanes. Angew. Chem. Int. Ed. 63, e202407391 (2024).
Yasutomi, Y., Suematsu, H. & Katsuki, T. Iridium(III)-Catalyzed Enantioselective Si−H Bond Insertion and Formation of an Enanti-oenriched Silicon Center. J. Am. Chem. Soc. 132, 4510–4511 (2010).
Jagannathan, J. R., Fettinger, J. C., Shaw, J. T. & Franz, A. K. Enantioselective Si–H Insertion Reactions of Diarylcarbenes for the Synthesis of Silicon-Stereogenic Silanes. J. Am. Chem. Soc. 142, 11674–11679 (2020).
Guo, X.-X., Gu, D.-W., Wu, Z. & Zhang, W. Copper-Catalyzed C–H functionalization reactions: efficient synthesis of heterocycles. Chem. Rev. 115, 1622–1651 (2015).
Allen, S. E., Walvoord, R. R., Padilla-Salinas, R. & Kozlowski, M. C. Aerobic copper-catalyzed organic reactions. Chem. Rev. 114, 899 (2014).
Gawande, M. B. et al. Cu and Cu-Based Nanoparticles: synthesis and applications in catalysis. Chem. Rev. 116, 3722–3811 (2016).
Nishino, S., Hirano, K. & Miura, M. Cu-Catalyzed Reductive gem-Difunctionalization of Terminal Alkynes via Hydrosilylation/ Hydroamination Cascade: concise Synthesis of α-Aminosilanes. Chem. Eur. J. 26, 8725–8728 (2020).
Wang, H. et al. Copper-catalyzed non-directed hydrosilylation of cyclopropenes: highly diastereoselective synthesis of fully substituted Cyclopropylsilanes. Chem. Commun. 56, 1819–1822 (2020).
Xu, Q.-F., Yang, P., Zhang, X. & You, S.-L. Enantioselective Synthesis of 4-Silyl-1,2,3,4-tetrahydroquinolines via Copper(I) Hydride Catalyzed Asymmetric Hydrosilylation of 1,2-Dihydroquinolines. Synlett 32, 505–510 (2021).
Xu, J.-L. et al. Copper-catalyzed Regiodivergent and enantioselective hydrosilylation of allenes. J. Am. Chem. Soc. 144, 5535–5542 (2022).
Li, S., Xu, J.-L. & Xu, Y.-H. Copper-catalyzed enantioselective hydrosilylation of allenes to access axially chiral (Cyclohexylidene)ethyl Silanes. Org. Lett. 24, 6054–6059 (2022).
Wang, Z.-L. et al. Regio- and Enantioselective CuH-catalyzed 1,2- and 1,4-Hydrosilylation of 1,3-Enynes. Nat. Commun. 14, 5048 (2023).
Jin, C. et al. Axial chirality reversal and enantioselective access to Si-stereogenic silylallene. Chem 9, 2956–2970 (2023).
Lipshutz, B. H., Noson, K., Chrisman, W. & Lower, A. Asymmetric hydrosilylation of Aryl Ketones catalyzed by copper hydride complexed by nonracemic Biphenyl Bis-phosphine Ligands. J. Am. Chem. Soc. 125, 8779 (2003).
Rix, F. C., Brookhart, M. & White, P. S. Electronic effects on the β-Alkyl migratory insertion reaction of Para-substituted Styrene Methyl Palladium Complexes. J. Am. Chem. Soc. 118, 2436–2448 (1996).
More O’Ferrall, R. A. Model calculations of hydrogen isotope effects for non-linear transition states. J. Chem. Soc. B 785–790 https://doi.org/10.1039/J29700000785 (1970).
Doherty, N. M. & Bercaw, J. E. Kinetics and Mechanism of the Insertion of Olefins into Transition Metal-hydride Bonds. J. Am. Chem. Soc. 107, 2670–2682 (1985).
Acknowledgements
We gratefully acknowledge research support of this work by the funding of the National Natural Science Foundation of China (22371269, (Y.H.X.), 22471024, (J.B.Z.)), the State Key Laboratory of Elemento-organic Chemistry Nankai University (202001, (Y.H.X.)), the Strategic Priority Research Program of the Chinese Academy of Sciences (XDB0450301, (Y.H.X.)), the Open Project of Key Laboratory of Organosilicon Chemistry, and Material Technology of Ministry of Education, Hangzhou Normal University (KFJJ2022013, (Y.H.X.)).
Author information
Authors and Affiliations
Contributions
Y.H.X. directed the project and composed the manuscript with revisions provided by the other authors. X.Y.Z., J.L.X., and Z.L.W. performed the experiments. W.G. and J.B.Z. performed the DFT calculations. All the authors were involved the analysis of results and discussions of the project.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Li-Wen Xu and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Zhu, XY., Gao, W., Xu, JL. et al. Copper-catalyzed intermolecular Regio- and Enantioselective Hydrosilylation of Alkenes with Prochiral Silanes. Nat Commun 16, 378 (2025). https://doi.org/10.1038/s41467-024-55592-1
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-024-55592-1
This article is cited by
-
Desymmetric sulfonylation of prochiral siladiols and related tandem sequences to multifunctional Si-chiral platform molecules
Science China Chemistry (2025)
-
Crystallization-mediated clusterization-induced ultralong room-temperature phosphorescence in bio-based polyamide 56
Science China Chemistry (2025)