Abstract
Physisorption presents a promising alternative to cryogenic distillation for capturing the most potent greenhouse gas, SF6, but existing adsorbents face challenges in meeting diverse chemical and engineering concerns. Herein, with insights into in-pore chemistry and industrial process design, we report a systematic investigation that constructed two low-cost composites pellets (Al(fum)@2%HPC and Al(fum)@5%Kaolin) coupled with an innovative two-stage Vacuum Temperature Swing Adsorption (VTSA) process for the ultra-efficient recovery of low-concentration SF6 from N2. Record-high selectivities (> 2×104) and SF6 dynamic capacities (~ 2.7 mmol/g) were achieved, while exceptional SF6 productivities (~ 58.7 L/kg), yields (~ 96.8%), and recyclability (~ 1000 cycles) were demonstrated in fixed-bed adsorption-desorption experiments under mild regeneration conditions. 2D solid-state NMR/in-situ FTIR, DFT-D binding/diffusion simulation analyses revealed the multi-site binding mode and the ultra-fast diffusion of SF6 within the channels. The proposed VTSA processes successfully met the dual stringent requirements of both environmental protection and electricity equipment operation: the SF6 recovery of 99.91% accompanied with a SF6 purity/working capacity of 99.91%/2.1 mmol/g, which significantly outperformed the industrial employed adsorbent zeolite 13X and showed only 18.7% the energy consumption of the cryogenic distillation.
Similar content being viewed by others
Introduction
Global warming and climate change have become major challenges facing the world today, and reducing greenhouse gas (GHG) emissions has become a key issue in addressing these challenges. Among the various GHGs, sulfur hexafluoride (SF6) has the highest global warming potential (GWP) ― 23,900 times that of carbon dioxide (CO2) ― and the longest atmospheric lifetime of up to 3200 years, and is therefore listed as one of the regulated GHGs requiring urgent attention in the 1997 Kyoto Protocol1. However, SF6, with its excellent dielectric and arc extinguishing properties, has always been the most important insulating gas in the power industry, and it is still difficult to replace with other gases, especially in high-voltage power equipment2. In recent years, in order to balance environmental protection and power safety, the industry has favored the use of SF6/N2 gas mixtures (10% to 30% SF6 by volume) as a substitute for pure SF6, which can greatly reduce the consumption of SF6, but how to efficiently recover the low concentration SF6 from the end-of-life equipment and exhaust gases has become an important issue from both environmental and economic aspects3,4,5. This recovery process is very challenging because it requires not only a high recovery of SF6 (typically ≥ 99.9%) to meet increasingly stringent emission standards, but also a high purity of the recovered SF6 (also ≥ 99.9%) to meet the operational requirements of the power equipment.
Liquefaction is a classical separation method that relies on the difference in boiling points between SF6 (209 K) and N2 (77 K), respectively, at a standard atmospheric pressure. However, the pressure required in this process increases significantly when the concentration of SF6 is relatively low6. For example, while the pressure required to liquefy pure SF6 is 2 MPa at 293 K, liquefying SF6 from a 10/90 SF6/N2 mixture requires 20 MPa7. Although cryogenic distillation technology can reduce operating pressure, it still relies on the phase change process and consumes a large amount of energy, especially when reducing SF6 concentration to trace levels (< 1000 ppm)8,9. In comparison, physisorption using porous solid adsorbents is widely recognized as a promising energy-saving alternative to liquefaction and distillation technologies due to the absence of a phase change, particularly for separating low-concentration components10,11,12,13,14. However, the potential of traditional porous adsorbents, like active carbon and zeolite, for SF6 capture and separation is hindered by their insufficient capacity and selectivity (Supplementary Tables 10 and 11)15,16.
Metal-organic frameworks (MOFs), as a new generation of porous materials in this century, have received increasing attention for gas capture and separation due to their attractive diversity in pore structures together with on-demand tunable surface properties17,18,19,20,21,22,23,24. Notably, more than twenty MOFs have been designed for SF6 adsorption (from N2), and some of them have indeed exhibited enhanced performance, such as higher uptake capacity or selectivity, compared to zeolites4,5,25,26,27,28,29,30,31,32,33,34,35,36,37,38,39. However, from both a chemical and engineering point of view, current research is still a long way from practical application. Chemically, the adsorption capacity and selectivity of SF6 still fall short of the stringent “dual > 99.9%” requirements mentioned above due to limited precision in matching the pore structure to the SF6 molecule, and there is an obvious trade-off between them. From an engineering point of view, more issues have not yet received sufficient attention: First, the reported studies almost exclusively use powdered polycrystalline MOFs as adsorbents, leading to a number of problems such as bed pressure drop and dust formation40; Second, an often overlooked point is that, unlike the continuous processes that are essential in industrial practice, the evaluation of adsorbents in the reported studies is based solely on laboratory-scale batch experiments (static adsorption and fixed-bed breakthrough), which not only makes it difficult to accurately assess the feasibility of solving challenging separation problems through adsorption processes but may also limit the evaluation and selection of different adsorbents. In addition, there are always concerns about the cost and stability of adsorbents. Only when all these issues are adequately addressed can the transition of MOFs from research to practice be truly realized.
Herein, we report a coherent “from pore to process” research to address the cutting-edge challenge of recovering low-concentration SF6 from SF6/N2 mixtures using surface-matched MOF composites as low-cost robust adsorbents coupled with a two-stage continuous Vacuum Temperature Swing Adsorption (VTSA) process. Based on the insight into the different electrostatic potential distributions between SF6 and N2 molecular surfaces, it was expected that an adsorbent with widely positive surface ESP and precisely matched pore size, i.e., the Al(fum) in this work, would have the ability to maximize SF6 affinity while weakening N2 binding via the competitive attraction-repulsion effects (Fig. 1A). As expected, the adsorption experiments have indeed achieved a super-high SF6/N2 selectivity (> 50000, together with record SF6 dynamic capacity, moderate heat of adsorption, mild regeneration temperature and rapid in-pore diffusion, and the adsorption mechanism was verified by a group of advanced technologies such as 2D solid-state NMR analysis, in situ FTIR analysis and DFT-D binding/diffusion simulation. To overcome the various shortcomings of MOF powders, two composite adsorbents based on the scale-up synthesis of Al(fum) via anti-tearing and sticky binders, Al(fum)@2%HPC and Al(fum)@5%Kaolin, were successfully prepared, showing negligible loss of separation efficiency, good mechanical stability and much lower bed pressure drop compared to the original powders. Furthermore, to illustrate the promising prospect of the composite adsorbents in practical applications, a rational process design and thorough exploration of process parameters were carried out, resulting in a powerful two-stage VTSA process that achieved both low emission and high recovery as well as high purity of SF6. Significantly, this process not only meets the stringent environmental protection standards but also meets the stringent electrical safety requirements, with an obvious (> 80%) reduction in energy consumption compared to simulated cryogenic distillation.
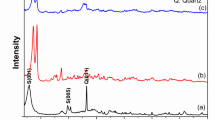
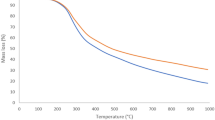
A The crystal structure of Al(fum) with 1D channels, the surface electrostatic potential of the Al(fum) framework, and the SF6 and N2 molecules with a scale spanning − 0.001 (red) to 0.005 a.u. (blue). B The process of shaping Al(fum) powder with Kaolin or HPC binder. C Powder X-ray diffraction patterns and the (D), TGA curves of Al(fum), Al(fum)@2%HPC and Al(fum)@5%Kaolin. The SEM images of Al(fum) powder (E), Al(fum)@2%HPC (F), and Al(fum)@5%Kaolin (G), respectively.
Results
Al(fum) was readily synthesized by a simple stirring in water, without the use of any organic solvents, at a relatively mild temperature of 333 K41, which can be easily scaled up (e.g., 35.58 g Al(fum) powder with a high yield of about 87% in this work). The crystal structure of Al(fum) showed that the single Al metal node acts as a six-coordination center connecting four O atoms in carboxylate groups from independent fumarate ligands and two O atoms in μ-OH groups from orthogonal orientation, forming an octahedral AlO6 structure which is linked by fumarate ligands to form a three-dimensional (3D) framework with infinite periodic one-dimensional (1D) square channels (Fig. 1A). Interestingly, the ESP analysis of the Al(fum) framework showed that the surface of the pore wall structure, surrounded by fumarate ligands, exhibits widely-positive ESP, and at the same time, the local ESP of the μ-OH groups in the orthogonal direction is also positive and reaches into the interior of the pore channel as the “corner”. Thus, the widely positive ESP distribution covering almost the entire interior of the pore channels makes Al(fum) a promising ESP trap for SF6, as it is conducive to interacting uniformly with the SF6 molecule whose outer surface is covered with negative ESP. Furthermore, the static pore size of Al(fum) was estimated to be 5.7 × 6.0 Å2, closer to the kinetic diameter of the SF6 molecule (5.15 Å) rather than N2 (3.64 Å), which can maximally improve the host-guest match/interaction to enhance the selective binding of SF6.
The experimental powder X-ray diffraction (PXRD) pattern of Al(fum) is shown in Fig. 1C. The positions and relative intensities of the characteristic peaks of the experimental results are in agreement with the simulated spectra, and the sharpness of the peaks indicates the excellent crystallinity of Al(fum). In addition, no loss of crystallinity was observed for Al(fum) during the exposure to air or soaking in water, suggesting that the Al(fum) is quite robust (Supplementary Fig. 1). Al(fum) also exhibited excellent thermal stability, as shown by the thermogravimetric analysis (TGA) curve (Fig. 1D), in which the solvent molecules in the pore channels can be completely removed at 393 K and a stable bulk structure can be maintained up to 673 K. To characterize the permanent porosity of the synthesized Al(fum), the adsorption of N2 on the activated sample was determined at 77 K, and the isotherm curve (Supplementary Fig. 5) revealed the saturation of the pore system at very low pressure (0.01 P/P0), from which we derived the Brunauer–Emmett–Teller (BET) surface area of 1092 m2/g.
In spite of the high porosity and excellent stability of the Al(fum) powder samples, the many disadvantages of the powder-form materials, including excessive bed pressure drop, dust pollution, pipeline blockage, and transportation difficulties, will greatly hinder its industrialization. To overcome these drawbacks, a wet granulation technology was employed to produce shaped Al(fum) pellets (which are actually composites), using strongly anti-tearing and viscous substances, hydroxypropyl cellulose (HPC) and Kaolin, as binders. The Al(fum) powder shows versatile formability, allowing the production of handmade pellets by incorporating as little as 2 wt% HPC binder (referred to as Al(fum)@2%HPC). Alternatively, mechanized production becomes feasible by utilizing 5 wt% Kaolin binders (designated as Al(fum)@5%Kaolin). Both variants of Al(fum) composite pellets exhibit good morphology and smooth surface (Fig. 1B). Meanwhile, they exhibited only a slight mass loss within 0.4% under oscillation at 25 °C and 200 rpm for 120 h (Supplementary Figs. 11 and 12), proving the good mechanical strength. Furthermore, the stress-strain tests demonstrated the excellent compressive strength of Al(fum)@5%Kaolin, measuring 7.2 MPa, which is 35.8% higher than that of the commercial zeolite 13X pellet (5.3 MPa) (Supplementary Fig. 13). X-ray photoelectron spectroscopy (XPS) tests verified that the element contents of the characterized elements in Al(fum)@2%HPC and Al(fum)@5%Kaolin were basically in agreement with those of Al(fum) powder (Supplementary Fig. 38 and Table 14), while the slightly higher O/C content in Al(fum)@2%HPC and O/Al contents in Al(fum)@5%Kaolin were due to the addition of a small amount of HPC and Kaolin binders, respectively. The PXRD pattern results revealed the almost lossless crystallinity of Al(fum)@2%HPC and Al(fum)@5%Kaolin composites compared with the pristine powders, even when exposed to air for one month, indicating the negligible influence of the shaping process on the crystal structure (Supplementary Figs. 1–3). The TGA curve of Al(fum)@2%HPC and Al(fum)@5%Kaolin (Fig. 1D) was also in accord with Al(fum), with a solvent loss at around 393 K while remaining stable up to 693 K, confirming the thermal stability of the shaped composites that can well meet the majority of industrial applications. It is noteworthy that a reported long-standing problem in adsorbent shaping by wet granulation is pore-blocking which will seriously reduce the adsorption performance42. However, we found that Al(fum)@2%HPC and Al(fum)@5%Kaolin showed merely a subtle decrease of porosity compared to Al(fum), with their BET surface area (Supplementary Figs. 5–7) of 999 m2/g (Al(fum)@2%HPC), 1087 (Al(fum)@5%Kaolin) and 1092 m2/g (Al(fum)), respectively, implying that the original pore structure was well preserved after shaping. From the scanning electron microscope (SEM) results, the original morphology of Al(fum) crystal (Fig. 1E) showed negligible change in Al(fum)@2%HPC (Fig. 1F) and Al(fum)@5%Kaolin (Fig. 1G) with the addition of a small amount of binder. The Al(fum) crystals were observed to be adhered by the HPC binder in clusters in Al(fum)@2%HPC, while in Al(fum)@5%Kaolin they were adhered by the flake-shaped Kaolin binder in a point-to-face format.
The adsorption behaviors of SF6 and N2 were explored by conducting the single-component adsorption isotherms on Al(fum) powder and pellets. As shown in Fig. 2A, it is noted that Al(fum) displayed a type I (the Langmuir isotherm) curve on SF6 with a considerable uptake of 3.79 mmol/g at 298 K and 1 bar. Meanwhile, the SF6 isotherm curve exhibited a marked steepness at low pressures, implying a strong SF6 affinity that is consistent with the prediction from the structural analysis for Al(fum). Al(fum) achieved an ultra-high SF6 uptake of 3 mmol/g at 10 kPa and 298 K, which is higher than most of the leading porous adsorbents at the same conditions (Fig. 2C) and only slightly lower than Cu-MOF-NH25. At the same time, an approximate molecular exclusion effect is observed in N2 adsorption, with the N2 uptake being only 0.27 mmol/g at 1 bar. The isosteric heat of adsorption (Qst) of SF6 and N2 on Al(fum) at zero loading was calculated by the Virial equation based on the single-component adsorption isotherms to be 35.9 kJ/mol and 7.5 kJ/mol, respectively (Supplementary Figs. 17 and 20). The notable Qst difference between the two gases provided strong support for their thermodynamic discrepancy in adsorption, and the moderate Qst value of SF6 is anticipated to enable lower energy consumption in adsorbent regeneration (Supplementary Fig. 41). Importantly, in accord with the 77 K N2 adsorption results, the shaped composite pellets Al(fum)@2%HPC and Al(fum)@5%Kaolin maintained the excellent adsorption performance compared to Al(fum). As shown in Fig. 2A, the isotherm profiles of SF6 and N2 on Al(fum)@2%HPC and Al(fum)@5%Kaolin were well consistent with that on Al(fum). The Qst of gases on Al(fum)@2%HPC and Alfum@5%Kaolin were also close to that on Al(fum) (Supplementary Figs. 18, 19, 21 and 22).
A The single-component isotherms of SF6 and N2 on Al(fum), Al(fum)@2%HPC, and Al(fum)@5%Kaolin at 298 K, respectively. B The SF6 and N2 adsorption isotherms of Al(fum)@5%Kaolin after immersed in water or exposure to air for a month at 298 K. C Comparison of the SF6 uptakes at 10 kPa and the IAST selectivities of SF6/N2 mixture (10/90, v/v) at 1 bar on different adsorbents at 298 K. D Breakthrough tests on Al(fum) columns for the SF6/N2 mixture (10/90, v/v) at 298 K. E Comparison of the dynamic SF6 uptake for the SF6/N2 mixture (10/90, v/v) among representative porous materials. F Desorption curve of the saturated Al(fum)@2%HPC and Al(fum)@5%Kaolin column for SF6/N2 mixture (10/90, v/v) at 313 K. G The adsorption-desorption cycles of the Al(fum)@2%HPC column by 1000 times pulse injection at 373 K.
The adsorption selectivities on Al(fum), Al(fum)@2%HPC and Al(fum)@5%Kaolin were predicted using ideal adsorbed solution theory (IAST) at 298 K for SF6/N2 gas mixtures (10/90, v/v), upon fitting the single-component adsorption isotherms to the dual-site Langmuir-Freundlich (DSLF) equations (the fitting parameters are provided in Supplementary Tables 1–3). Strikingly, these powders and pellets displayed excellent IAST selectivities among all reported porous materials (Fig. 2C and Supplementary Table 10). For example, at 298 K and 100 kPa, the IAST selectivity of Al(fum) reaches an exceptional value of 50139, which is significantly higher than the previous benchmark (Ni(adc)(dabco)0.5, 948)39 and other top-performing literature materials such as Ni(NDC)(TED)0.5 (750)27 and Ni(ina)2 (375)33, while the Al(fum)@2%HPC and Al(fum)@5%Kaolin also maintained the excellent selectivities of 28978 and 34409 in the same order of magnitude, respectively (Supplementary Figs. 30 and 31). In general, the physisorption mechanism in MOFs driving gas separations often leads to a notable trade-off between adsorption selectivity and capacity, which significantly affects the productivity and separation efficiency in practice. Nevertheless, Al(fum), Al(fum)@2%HPC, and Al(fum)@5%Kaolin have successfully achieved the balance between high SF6/N2 selectivity and SF6 adsorption capacity (Fig. 2C).
To evaluate the separation performance of the SF6/N2 mixture, experimental dynamic breakthrough tests mimicking actual conditions (the typical 10/90 (v/v) SF6/N2 gas mixture) were conducted on Al(fum), Al(fum)2%HPC and Al(fum)@5%Kaolin, respectively. As shown in Fig. 2D, the N2 gas immediately flew out of the Al(fum) column at 298 K and quickly reached equilibrium due to its weak affinity, agreeing with the negligible adsorption uptake in the static isotherm. In contrast, the retention time of SF6 on Al(fum) was as long as 201.4 min/g. The dynamic adsorption capacity of SF6 on Al(fum) was calculated as a record-high value of 2.76 mmol/g, which is exactly close to its static uptake at 10 kPa and higher than the previously reported top-performing adsorbents (Fig. 2E), and productivity of high-purity (> 99.999%) N2 was calculated to be 564 L/kg. Al(fum)@2%HPC and Al(fum)@5%Kaolin also maintained the high dynamic SF6 uptake (Fig. 2E). For example, the dynamic SF6 uptake of Al(fum)@2%HPC was 2.68 mmol/g at 298 K, which was 97.10% of that of Al(fum) powder; and the dynamic SF6 uptake of Al(fum)@5%Kaolin also reached 2.74 mmol/g, which was 99.27% of that of Al(fum). Importantly, despite the close packing density of Al(fum) powder and Al(fum)@2%HPC handmade pellet columns (0.18 g/cm3 and 0.2 g/cm3, respectively), the bed pressure drop of Al(fum)@2%HPC column was significantly lower than that of Al(fum) column as shown in Supplementary Fig. 45, verifying the significance of adsorbent shaping process on dynamic separation from an engineering perspective. The regeneration performance of gas-saturated Al(fum) composite pellets was determined to demonstrate the adsorbent recyclability and to obtain purified SF6. For the gas-saturated Al(fum)@2%HPC in SF6/N2 gas mixture (10/90, v/v), the N2 was immediately desorbed with only a negligible amount of 0.02 L/kg, contributing to a cumulative purity of 99.97% of recovered SF6. The desorbed amount of SF6 was calculated to be 2.61 mmol/g, corresponding to a high yield (97.39%) and high productivity (58.46 L/kg) of SF6 from desorption (Fig. 2F). In addition, the excellent separation performance of Al(fum)@2%HPC was hardly changed even after 1000 adsorption-desorption cycles (Fig. 2G). Similarly, the Al(fum)@5%Kaolin also exhibited excellent regeneration performance and recyclability (Supplementary Fig. 46). The desorbed amount of SF6 was calculated to be 2.64 mmol/g, corresponding to a high yield (96.35%) and high productivity (59.08 L/kg) of SF6 from desorption (Fig. 2F).
To gain a deeper insight into the different adsorption behavior of SF6 and N2 and the separation mechanism on Al(fum), both the first-principles dispersion-corrected density functional theory (DFT-D) calculation and in situ spectral experiments were further conducted. As shown in Fig. 3A, DFT-D calculation showed that SF6 and N2 molecules with different surface ESP distributions were located in a similar position inside the channel. However, large differences were noted in their interaction mode and binding energy with the framework. Specifically, it is observed that all the F atoms of the SF6 molecule were captured by the H atoms from fumarate ligands and μ-OH group through multiple van der Waals and hydrogen bonding interactions (the F···H distances were 2.84 ~ 3.91 Å), which is well agreeing with their ESP distribution (the negative surface ESP covers the F atom, while the positive surface ESP covers the -CH and μ-OH group). As a result, a relatively high binding energy (ΔE) of 51.3 kJ/mol was realized for SF6, which confirms the exceptional adsorption ability of SF6 and supported the above experimental data. In contrast, the numerical count of binding sites for N2 was significantly lower than that of SF6 (2 vs. 6), while the electrostatic interactions between the fumaric ligands and N2 were notably weaker (the N···H distances were 2.95 and 3.52 Å), leading to a considerably lower ΔE of 13.0 kJ/mol when compared to SF6. The experimental in situ Fourier transform infrared spectroscopy (FT-IR), and 2D solid-state nuclear magnetic resonance (2D solid-state NMR) were employed to delve into the host-guest interactions between the SF6 molecule and the Al(fum) framework. Notably, the FT-IR spectrum shown in Fig. 3B of Al(fum) exhibited peak characteristic differences corresponding to μ-OH and C-H during SF6 adsorption at room temperature, indicating the multiple host-guest interactions between Al(fum) and SF6 molecules. Furthermore, the in situ FT-IR measurement revealed a rapid SF6 equilibrium time (within 3 minutes), consistent with the relatively low diffusion energy barrier of SF6 derived from the DFT-D diffusion simulation (2.81 kJ/mol) (Fig. 3D). The 2D 1H-19F MAS solid-state NMR spectra of the SF6-loaded Al(fum) also verified the μ-OH···F and C-H···F interactions (Fig. 3C), consistent with the above DFT-D and in situ FT-IR results.
A N2 and SF6 binding sites on Al(fum) and binding energies from DFT-D calculation, with presentation of the molecular surface ESP for the pore surface and gas molecules. B In situ FT-IR spectra of the activated Al(fum) sample exposed to SF6 gas at room temperature. C 2D 1H-19F solid-state NMR spectra of SF6 adsorbed on Al(fum). D The minimum energy path (MEP) profile and the snapshots for SF6 in the Al(fum).
According to the above results, it is exciting that the shaped composite pellets Al(fum)@2%HPC and Al(fum)@5%Kaolin have shown highly promising SF6 capture and separation performance and have now met various essential criteria for an industrial adsorbent, including adsorption uptake (Fig. 2C and Supplementary Table 10), selectivity (Fig. 2C), stability (Fig. 2B), cost (Supplementary Table 15), etc. In order to evaluate the performance of the adsorbents in an industrializable manner, a rational design of a continuous adsorption-separation process is essential. The Al(fum)@5%Kaolin pellet, produced through mechanized mean, was employed in a single-stage Vacuum Temperature Swing Adsorption (VTSA) process to evaluate the actual SF6/N2 separation performance. The simulation was conducted at 1.5 bar and 298 K with 30 NL/min of SF6/N2 mixture (10/90, v/v), and its corresponding cycle sequence is shown in the right of Fig. 4A (the optimization of operational parameters was provided in Supplemental Information, Supplementary Table 8). Accordingly, a high purity (99.94%) SF6 was obtained with a high yield (1.21 kg/h) in a single-stage VTSA using two beds of 15 cm ID × 45 cm loaded with 6.44 kg Al(fum)@5%Kaolin. However, it was noted that ~ 12% SF6 was lost in the Adsorption (AD) and Heating co-current blowdown (PP) steps and the SF6 concentration in the light product was 1.22%, so the SF6 recovery was 88.43% which is far away from meeting the stringent requirements of environmental protection (Fig. 4C). To solve this issue, a two-stage VTSA process was established with the identical feed conditions (temperature, pressure and feed flow rate) to those of the single-stage VTSA. The schematic diagram and model of the two-stage VTSA are shown on the left of Fig. 4A, B. As shown in Supplementary Fig. 53, after 4 cycles, the first stage of the two-stage VTSA process reached the Cyclic Steady State (CSS) and the SF6 working capacity of the Al(fum)@5%Kaolin was 2.10 mmol/g, which was significantly larger than the SF6 uptake at 10 kPa of the conventional adsorbent zeolite 13X (0.99 mmol/g)30. Notably, the mixed light product, which combined gas from the top of the bed in the first-stage VTSA and the second-stage VTSA, has an N2 content of 99.91% which meets the environmental protection requirements. Moreover, due to the fact that SF6 was desorbed by heating and displaced the N2 in the bulk phase porosity of the shaped pellets during the PP step, the obtained cyclic average SF6 purity was 99.91% when the process reached CSS (Fig. 4C). The potential of the two-stage VTSA for recovering SF6 was further evaluated by an energy consumption analysis with conventional cryogenic distillation as the fundamental scheme (Supplementary Table 9). As shown in Fig. 4C, the energy consumption of the two-stage VTSA was only 1/5 of that of the cryogenic distillation, where cryogenic distillation was 0.13 equivalent standard oil kg/kg SF6 while the overall energy consumption of the two-stage VTSA was 0.025 equivalent standard oil kg/kg SF6. In addition, since the VTSA regeneration process for Al(fum)@5%Kaolin involves heating, and the previously reported leading MOFs have demonstrated the ability to achieve SF6 recovery and regeneration at ambient temperature in laboratory tests, it is important to assess whether Al(fum)@5%Kaolin has a disadvantage in terms of process energy consumption compared to these MOFs. To address this, the Vacuum Pressure Swing Adsorption (VPSA) process based on a leading MOF was established, and a preliminary estimation of its energy consumption was conducted. The results indicate that the energy consumption of VPSA may not necessarily be lower than VTSA, as the gas compressor required for feed gas handling incurs substantial electrical energy consumption (Supplementary Table 18).
A Schematic diagram of the VTSA cycle, left, two-stage VTSA, right, single-stage VTSA with bed utilization − 0% (yellow) to 100% (green), (AD, Adsorption; ED, Equalization depressurization; VU, Vacuum desorption; VU Purge, Vacuum desorption with Purge; ER, Equalization pressurization; CR, Pressurization with cooling; PP, Heating co-current blowdown; color code: blue, cooling; red, heating). B Schematic model of the two-stage VTSA process. C Simulation results of single-stage and two-stage VTSA and energy consumption for two-stage VTSA and cryogenic distillation.
Discussion
Adsorption technologies based on porous adsorbents are widely expected to replace energy-intensive phase-change gas separation processes, the key to which is to overcome the trade-off between adsorption capacity and selectivity while possessing various properties required for industrialization at the same time. With a focus on the general challenge of GHG gas capture, this work has conducted a comprehensive study on the adsorptive separation of low-concentration SF6 from both chemical and engineering perspectives, including pore chemistry analysis, materials synthesis, adsorption experiments, 2D solid-state NMR/in situ FTIR, DFT-D binding/diffusion simulation, process design and optimization, and has demonstrated powerful adsorbents and a continuous separation process with high potential for industrialization. As a highly efficient ESP trap for SF6 molecules with well-matched pore surface ESP and pore size, the Al(fum) showed a strong affinity and selective recognition ability to SF6, resulting in record-high SF6/N2 selectivity and SF6 dynamic adsorption capacity. Furthermore, Al(fum)@2%HPC and Al(fum)@5%kaolin pellets were prepared, which not only retained all the excellent properties of Al(fum) powder but also exhibited good mechanical strength and significantly reduced pressure drop of the adsorption bed. In addition, a combination of the following advantages, all of which are essential for a practical adsorption separation process was accompanied: low cost of synthesis precursors (expected to be similar to commercial zeolites); scale-up feasibility and green nature of the synthesis method; rapid mass transfer; easy regeneration; good thermal and air stability; exceptional recyclability. Importantly, the two-stage VTSA system presented in this work achieved SF6 purity of 99.91% and 99.91% SF6 recovery, successfully meeting the dual requirements of electrical equipment safety and environmental protection, with an energy consumption of only 18.7% that of cryogenic distillation. Overall, this study not only sets a benchmark for the capture and separation of SF6 by adsorption technology but also provides a reference for accelerating MOFs-based adsorption technologies from laboratory research to practical applications.
Methods
Fumaric acid (C4H4O4, 98%, Aladdin), Aluminum sulfate octadecahydrate (Al2(SO4)3·18H2O, 98%, Aladdin) and sodium hydroxide (NaOH, 98%, Aladdin), ethanol (C2H5OH, 99%, Sigma-Aldrich), Hydroxypropyl cellulose (HPC, 99%, Macklin), Kaolin (Kaolin, Aladdin) were commercially available and used as supplied without further purification. N2 (99.999%), SF6 (99.999%), He (99.999%), and SF6/N2 mixing gas (10/90, v/v) were purchased form JuLun Company (China).
Synthesis of Al(fum) (powder)
[Al(OH)(O2C-C2H2-CO2)]n (Al(fum)) was synthesized using the scaled-up method. Al2(SO4)3·18H2O (70.0 g, 0.105 mol) was dissolved in 300 mL DI water at 333 K to make solution A, meanwhile, a specific amount of fumaric acid (24.3 g, 0.209 mol) and NaOH (25.2 g, 0.63 mol) were dissolved in 360 mL DI water at 333 K to make solution. Next, solutions were mixed for 10 min with constant stirring at 333 K. The produced solution was filtered, replaced with DI water 3 times, then washed with ethanol and dried for 12 h overnight (29.53 g, 89% yield based on Al).
Synthesis of Al(fum)@2%HPC (Pellet)
The Alfum@2%HPC was prepared using the handmade wet granulating method. 980 mg dried Al(fum) powder was mixed with 20 mg HPC, and then 2 mL deionized water was added to the powder mixture several times while grinding the powder mixture continuously to form a paste. The semi-dry paste was formed into Al(fum)@2%HPC pellets of ~ 1 mm diameter.
Synthesis of Al(fum)@5%Kaolin (Pellet)
The Alfum@5%Kaolin was prepared using the mechanical wet granulating method. 9.5 g dried Al(fum) powder was mixed with 0.5 g Kaolin as a binder using a mixing pelletizer (CML, Caleva). The mixed powder with a small account of DI water was slowly added into the pelletiser extruder, when extruded into a strip, it was added into a rolling part and cut into small pellets. Then, the Al(fum)@5%Kaolin pellet was dried at 373 K overnight to remove DI water from the granule. To ensure uniform size distribution, 0.8–1.2 mm pellets were sieved through 16 and 20 mesh sieves.
Characterization
Powder X-ray diffraction (PXRD) data were collected at room temperature on an X’Pert diffractometer (Cu Kαλ = 1.540598 Ǻ) with an operating power of 40 kV. The collected range of 2θ is 5°–60°. The thermal gravimetric analysis (TGA) was collected on SDT 650 with a platinum pan under nitrogen flow with a heating rate of 10 °C/min from 30 °C to 800 °C. The scanning electron microscope (SEM) was analyzed by Hitachi SU-8100. 2D 1H-19F solid-state NMR spectra were collected on a Bruker Avance NEO 600WB spectrometer at ambient temperature. The in situ Fourier Transform Infrared Spectrum (in situ FT-IR) was collected on Thermo Fisher Scientific FTIR-650 at ambient temperature.
Gas adsorption measurements
Degassing, BET surface area, and gas adsorption measurements were conducted on a Micromeritics 2460 surface area analyzer. The Al(fum), Al(fum)@2%HPC, and Al(fum)@5%Kaolin samples were degassed at 393 K and 5 µmHg for 24 h before analysis. The N2 BET surface area of the Al(fum), Al(fum)@2%HPC, and Al(fum)@5%Kaolin were measured at 77 K. The SF6 and N2 adsorption isotherms were measured at 298 K ~ 363 K on activated samples, respectively.
IAST selectivity calculations
Before estimating the selectivity for binary gas mixture, the single-component adsorption isotherms of SF6 and N2 were fitted with the Dual-sites Langmuir-Freundlich model (DSLF).
Dual-sites Langmuir-Freundlich model (DSLF):
Where N is the amount of adsorption, mmol/g, p is the pressure, kPa, q is the amount of adsorption, mmol/g, b is the Langmuir constant, kPa−1, n is the uniformity of the adsorption site.
The adsorption selectivity of binary gas mixtures was then estimated by the Ideal Adsorbed Solution Theory (IAST)43, the IAST selectivity (Sads) has been defined as:
Where, \({{{\boldsymbol{q}}}}_{{{\boldsymbol{1}}}}\) and \({{{\boldsymbol{q}}}}_{{{\boldsymbol{2}}}}\) are the molar loadings in the adsorbed phase in equilibrium with the bulk gas phase with partial pressures \({{{\boldsymbol{p}}}}_{{{\boldsymbol{1}}}}\) and \({{{\boldsymbol{p}}}}_{{{\boldsymbol{2}}}}\).
Breakthrough experiments
The breakthrough tests for SF6/N2 (10/90, v/v) mixing gas were conducted at 298 K and 100 kPa on the dynamic gas breakthrough equipment. The experiments were conducted using a stainless-steel bed packed with the activated Al(fum), Al(fum)@2%HPC, and Al(fum)@5%Kaolin, respectively. Before the experiments, the He steam was introduced to the bed to purge the adsorbents at 373 K for 24 h to avoid channeling. The outlet gas was detected by GC-2030 pro (SHIMADZU) with a thermal conductivity detector (TCD). After each breakthrough experiment, the column was regenerated with a He flow rate of 6 mL/min at 313 K for 24 h.
The actual gas uptake capacity was calculated using the following equation:
where \({{{\boldsymbol{n}}}}_{{{\boldsymbol{adsi}}}}\) was the uptake capacity of the gas component \({{\boldsymbol{i}}}\), \({{{\boldsymbol{F}}}}_{{{\boldsymbol{i}}}}\) was the flow rate of gas \({{\boldsymbol{i}}}\) in the inlet, \({{{\boldsymbol{t}}}}_{{{\boldsymbol{1}}}}\) was the start time, \({{{\boldsymbol{t}}}}_{{{\boldsymbol{2}}}}\) was the end time, \({{\boldsymbol{F}}}\) was total flow rate, \({{{\boldsymbol{C}}}}_{{{\boldsymbol{i}}}}\) was the molar fraction of gas \({{\boldsymbol{i}}}\) in the outlet gas mixture.
The productivity of the high-purity of N2 was calculated using the following equation:
Where \(p\) was gas productivity, \(F\) was total flow rate, \({C}_{i}\) was the molar fraction of gas \(i\) in the outlet gas mixture, t1 was the breakthrough time of component i, t2 was the breakthrough time of SF6.
The captured SF6 during the breakthrough experiment can be recovered by desorption. When the breakthrough experiments were finished, the column was heated to 313 K, a flow rate of 6 mL/min was introduced, and the outlet gas from the column was monitored using gas chromatography. The SF6 productivity from desorption was calculated as Eq. (5), and the cumulative purity \(p\) of SF6 obtained by desorption process was calculated using the Eq. (6):
where \({{{\boldsymbol{n}}}}_{{{\boldsymbol{i}}}}\) was the eluted amount of the gas component \({{\boldsymbol{i}}}\), \({{\boldsymbol{F}}}\) was the total flow rate, \({{{\boldsymbol{C}}}}_{{{\boldsymbol{i}}}}\) was the molar fraction of gas \({{\boldsymbol{i}}}\) in the outlet gas mixture.
where \(p\) was gas purity, \({n}_{1}\) and \({n}_{2}\) were calculated from the given time period corresponding to the desorption curves.
Simulation method
Density-functional theory (DFT) calculations
The static binding energy between gas and adsorbent was calculated using the combination of first-principal density functional theory (DFT) and plane-wave ultrasoft implemented in the Materials Studio, CASTEP code44. A semi-empirical addition of dispersive forces to conventional DFT was included in the calculation to account for van der Waals interactions. Calculations were performed under the generalized gradient approximation (GGA) with Perdew-Burke-Ernzerhof (PBE) exchange correlation43. A cutoff energy of 544 eV and a 4 × 2 × 2 k point mesh with smearing 0.2 eV were found to be enough for the total energy to converge within 1 × 10−6 ev/atom, the calculation errors are within 0.15 Å. The structure of Al(fum) would be first optimized by the UFF force field implemented in the Materials Studio. Various guest gas molecules, including SF6 and N2, were finally introduced to different locations of the channel pore, followed by a full structural relaxation. The static binding energy was calculated:
Electrostatic potential calculations
The electrostatic potential of the Al(fum) was calculated using the DMol3 code in the Materials Studio. The Al(fum) framework was first undergoing geometry optimization based on DFT calculations. The single-point energy calculation was then performed on the optimized framework to obtain the periodic surface electrostatic potential distribution. The electrostatic potentials of the SF6 and N2 were obtained from the quantum chemistry calculations performed by the Gaussian 09 program based on DFT at the 6–31 G* basis set. The ESP analysis was performed by Multiwfn and VMD software package45,46.
Process simulation in Aspen adsorption
The single-stage and two-stage temperature vacuum swing adsorption (VTSA) were simulated by Aspen Adsorption to evaluate the separation performance of Al(fum)@5%Kaolin for SF6/N2 (10/90, v/v) mixtures. Aspen adsorption uses the first-order Upwind Differencing Scheme (UDS1) with 50 nodes. The mathematical models are a combination of the mass balance, energy balance, momentum balance, adsorption equilibrium and the adsorption kinetic model.
Mass balance
For Lumped kinetics, the mass transfer model in the gas phase can be seen in the following equation, which consists of convection accumulation and gas-solid mass transfer.
Where \({v}_{g}\) is the superficial gas velocity, m/s, \({c}_{{bi}}\) is the gas-phase concentration of component i, kmol/kg, \({\rho }_{s}\) is the gas loading, kmol/kg, \({\varepsilon }_{B}\) is the bulk phase porosity, m3/m3.
Momentum balance
The pressure drop across the adsorption bed can be described using the Ergun equation, which considers laminar and turbulent flow effects.
Where \({M}_{w}\) is the molecular weight of the gas phase, kg/kmol, \(\mu\) is the dynamic viscosity, kg/(m s), \(\varPsi\) is the particle sphericity, \({V}_{g}\) is the superficial gas velocity, m/s.
The energy balance
gas-phase energy balance includes terms of thermal conduction, convection of energy, accumulation of heat, compression, heat transfer from gas to solid, heat transfer from gas to the internal wall, and heat exchange between gas and jacket exchanger.
Where \({k}_{g}\) is the axial gas phase thermal conductivity, MW/(m K), \({C}_{{vg}}\) is the specific gas phase heat capacity at constant volume, MJ/(kmol K), \({HTC}\) is the constant for heat transfer coefficient between gas and adsorbent, MW/(m2 k).
Adsorption isotherm model
The extended Langmuir 2 model is used to describe the equilibrium loading of SF6, N2 on the Al(fum)@5%Kaolin, which gives a good fit to the experimental isotherm data as shown in the following.
Where \({W}_{i}\) is the equilibrium loading of component i on granule, kmol/g, P is the partial pressure of component i, bar, Ts is the temperature, K, \({{IP}}_{i}\) is the isotherm parameter of component i, i = 1,2 is for SF6 and N2 respectively. Isotherm model parameters for Al(fum)@5%Kaolin in Aspen Adsorption can be found in Supplementary Table 5.
Adsorption kinetic model
The adsorption kinetic model is used to describe the mass transfer resistance in the adsorption process, which is influenced by the external film resistance, the macropore diffusion, and the micropore diffusion. The Linear Driving Force (LDF) equation is defined as the most common model that can be estimated in the equation:
Where \({k}_{i}\) is the mass transfer coefficient (MTC), s−1, \({w}_{i}\) is the adsorption loading of Al(fum)@5%Kaolin, kmol/kg, \({{w}_{i}}^{*}\) is the equilibrium loading, kmol/kg, t is the time, s, i = 1,2 is for SF6 and N2 respectively.
Simulation cyclic sequence
Sensitivity analysis was utilized to investigate the intricate relationship between cycle performance and operational parameters, providing theoretical support for process design. The optimized simulation cyclic sequences were as follows.
Single-stage VTSA (2 beds and 4 steps)
Step 1: AD. The compressed feed mixture flew into bed 1, where SF6 was adsorbed.
Step 2: PP. N2 in the bulk phase porosity was replaced by SF6 and extracted from bed 1 to N2 tank. The adsorbent in bed 1 was indirectly heated by a hot stream in the jacket.
Step 3: VU. The adsorbent in bed 1 was indirectly heated by hot steam in the jacket. As SF6 was desorbed and released from the bottom of the bed, the pressure gradually decreased.
Step 4: CR. The pressure of bed 1 was increased, ultimately returning bed 1 to its original state. The adsorbent in bed 1 was indirectly cooling by a cool stream in the jacket.
Two-stage VTSA (4 beds and 10 steps)
Step 1: AD. Similar to step 1 of single-stage VTSA, bed 1 has undergone adsorption.
Step 2: ED. Bed 1 at the end of adsorption was connected to bed 2 at the end of the vacuum desorption purge.
Step 3: VU. Similar to step 3 of single-stage VTSA.
Step 4: VU Purge. The light product from the N2 tank was introduced to purge bed 1 at low pressure.
Step 5: ER. Similar to the step 2. Bed 1 was connected to bed 2 and pressurized by bed 2.
Step 6: CR. The bed 1 was pressurized by light product while the adsorbent in the bed 1 was indirectly cooling by a cool stream in the jacket.
Performance parameters for evaluation of VTSA process
While evaluating the performance of the process, the key parameters are working capacity, purity, recovery and yield.
Working capacity:
Where \({n}_{i,{ads}}\) is the adsorption capacity after AD step, \({n}_{i,{des}}\) is the adsorption capacity after VU or VU Purge step, i = 1,2 is for SF6 and N2 respectively.
Purity
Where \({F}_{{product}}\) is the molar flow rate of product steam, mol/s, \({y}_{{product},i}\) is molar fraction of i in product steam, i = 1,2 is for SF6 and N2 respectively.
SF6 recovery:
N2 recovery:
Yield:
Overall energy consumption analysis for VTSA and cryogenic distillation
The electrical energy consumption was calculated by following equations.
Where \({E}_{{ele}}\) is the electric energy of the compressor or pump in a cycle, kJ/mol SF6, \(\gamma\) is the heat capacity ratio (Cp/Cv), Tsteam is the temperature of steam, K, \({P}_{1}\) and \({P}_{2}\) are the outlet and inlet pressure of equipment, bar, \(F\) is the molar flow rate of steam, kmol/s, \(\eta\) is the mechanical efficiency and the value is selected as 0.95.
It is assumed that the bed jacket did not transfer energy with the surroundings, only with the medium and so heat duty can be calculated by following equations.
Where \(H\) is the heat duty, kJ/kg SF6, \({T}_{L}\) is the adsorption temperature, K, \({T}_{H}\) is the desorption temperature, K, \({W}_{i}\) is the working capacity for adsorbent, kJ/(kg K), \({C}_{{pw}}\) is the bed wall capacity, kJ/(kg K), \({{DH}}_{{{SF}}_{6}}\) is the isosteric heat of adsorption, kJ/mol, i = 1,2 is for SF6 and N2 respectively.
By converting to standard oil according to PRC National Standard (GB/T50441-2016)47, the overall energy consumption of two-stage VTSA and cryogenic distillation was calculated by following.
Where e is the total energy consumption, equivalent standard oil kg/kg SF6 \(E\) is the thermal and electrical energy consumption, MJ/kg SF6 \({C}_{i}\) is the conversion of utilities into standard oil.
Data availability
The authors declare that the main data supporting the findings of this study are available within the article and its Supplementary Information files. Source data that support the findings of this study are provided as a source data file. Source data are provided in this paper.
References
Ravishankara, A. R., Solomon, S., Turnipseed, A. A. & Warren, R. F. Atmospheric lifetimes of long-lived halogenated species. Science 259, 194–199 (1993).
Fang, X. et al. Sulfur hexafluoride (SF6)emission estimates for China: An inventory for 1990-2010 and a projection to 2020. Environ. Sci. Technol. 47, 3848–3855 (2013).
Hasell, T. et al. Porous organic cages for sulfur hexafluoride separation. J. Am. Chem. Soc. 138, 1653–1659 (2016).
Kim, M. et al. Highly selective adsorption of SF6 over N2 in a bromine-functionalized zirconium-based metal-organic framework. Chem. Eng. J. 339, 223–229 (2018).
Ren, J. et al. Computer-aided discovery of MOFs with calixarene-analogous microenvironment for exceptional SF6 capture. Chem. Mater. 33, 5108–5114 (2021).
Builes, S., Roussel, T. & Vega, L. F. Optimization of the separation of sulfur hexafluoride and nitrogen by selective adsorption using monte carlo simulations. AIChE J. 57, 962 (2011).
Toyoda, M. et al. Application of pressure swing adsorption to SF6 separation and liquefaction from SF6/N2 mixtures. In IEEE Power Engineering Society Winter Meeting Singapore 2156–2161 (IEEE, 2000).
Kim, K. et al. Status of SF6 separation/refining technology development for electric industry in Korea. Sep. Purif. Technol. 200, 29–35 (2018).
Xu, G. et al. An improved CO2 separation and purification system based on cryogenic separation and distillation theory. Energies 7, 3484–3502 (2014).
Zhou, J. et al. Highly efficient separation of C8 aromatic isomers by rationally designed nonaromatic metal–organic frameworks. J. Am. Chem. Soc. 144, 21417–21424 (2022).
Zhou, J. et al. Tunable confined aliphatic pore environment in robust MOFs for efficient separation of gases with comparable size and property. J. Am. Chem. Soc. 144, 14322–14329 (2022).
Shen, J. et al. Simultaneous interlayer and intralayer space control in two-dimensional metal-organic frameworks for acetylene/ethylene separation. Nat. Commun. 11, 6259 (2020).
Ke, T. et al. Molecular sieving of C2-C3 alkene from alkyne with tuned threshold pressure in robust layered metal-organic frameworks. Angew. Chem. Int. Ed. 59, 12725–12730 (2020).
Zhu, X. et al. Vertex strategy in layered 2D MOFs: Simultaneous improvement of thermodynamics and kinetics for record C2H2/CO2 separation performance. J. Am. Chem. Soc. 145, 9254–9263 (2023).
Chuah, C. Y., Yu, S., Na, K. & Bae, T.-H. Enhanced SF6 recovery by hierarchically structured MFI zeolite. J. Ind. Eng. Chem. 62, 64–71 (2018).
Matito-Martos, I. et al. Zeolites for the selective adsorption of sulfur hexafluoride. Phys. Chem. Chem. Phys. 17, 18121–18130 (2015).
Chen, K. et al. Synergistic sorbent separation for one-step ethylene purification from a four-component mixture. Science 366, 241–246 (2019).
Zeng, H. et al. Orthogonal-array dynamic molecular sieving of propylene/propane mixtures. Nature 595, 542–548 (2021).
Nugent, P. et al. Porous materials with optimal adsorption thermodynamics and kinetics for CO2 separation. Nature 495, 80–84 (2013).
Gu, C. et al. Design and control of gas diffusion process in a nanoporous soft crystal. Science 363, 387–391 (2019).
Xie, X. et al. Gate-opening effect in a flexible metal-organic framework for sieving acetylene. Chem. Bio. Eng. 1, 150–156 (2024).
Matsuda, R. et al. Highly controlled acetylene accommodation in a metal-organic microporous material. Nature 436, 241–248 (2005).
Li, H., Eddaoudi, M., O’Keeffe, M. & Yaghi, O. M. Design and synthesis of an exceptionally stable and highly porous metal-organic framework. Nature 402, 276–279 (1999).
Cadiau, A., Adil, K., Bhatt, P. M., Belmabkhout, Y. & Eddaoudi, M. A metal-organic framework-based splitter for separating propylene from propane. Science 353, 137–140 (2016).
Kim, M., Lee, S., Lee, C. & Bae, Y. High SF6 selectivities and capacities in isostructural metal-organic frameworks with proper pore sizes and highly dense unsaturated metal sites. Micropor. Mesopor. Mat. 190, 356–361 (2014).
Kim, M. et al. Efficient SF6/N2 separation at high pressures using a zirconium-based mesoporous metal-organic framework. J. Ind. Eng. Chem. 84, 179–184 (2020).
Yang, M., Chang, M., Yan, T. & Liu, D. A nickel-based metal-organic framework for efficient SF6/N2 separation with record SF6 uptake and SF6/N2 selectivity. Sep. Purif. Technol. 295, 121340 (2022).
Wang, T. et al. Calcium-based metal-organic framework for efficient capture of sulfur hexafluoride at low concentrations. Ind. Eng. Chem. Res. 60, 5976–5983 (2021).
Skarmoutsos, I., Eddaoudi, M. & Maurin, G. Highly tunable sulfur hexafluoride separation by interpenetration control in metal-organic frameworks. Micropor. Mesopor. Mat. 281, 44–49 (2019).
Kim, M. et al. High SF6/N2 selectivity in a hydrothermally stable zirconium-based metal-organic framework. Chem. Eng. J. 276, 315–321 (2015).
Chuah, C., Goh, K. & Bae, T. Hierarchically structured HKUST-1 nanocrystals for enhanced SF6 capture and recovery. J. Phys. Chem. C 121, 6748–6755 (2017).
Kim, P. et al. Separation of SF6 from SF6/N2 mixture using metal-organic framework MIL-100 (Fe) granule. Chem. Eng. J. 262, 683–690 (2015).
Wang, S. et al. Pore-structure control in metal-organic frameworks (MOFs) for capture of the greenhouse gas SF6 with record separation. Angew. Chem. Int. Ed. 61, e202207066 (2022).
Wanigarathna, D., Gao, J. & Liu, B. Metal organic frameworks for adsorption-based separation of fluorocompounds: A review. Mater. Adv. 1, 310 (2020).
Köppen, M. et al. Synthesis, transformation, catalysis, and gas sorption investigations on the bismuth metal-organic framework CAU‐17. Eur. J. Inorg. Chem. 2018, 3496–3503 (2018).
Yan, L. et al. Methyl-functionalized flexible ultra-microporous MOF for efficient SF6/N2 mixture separation. Chem. Eng. J. 472, 145145 (2023).
Yan, J. et al. An In(III)-MOF based on pore engineering for efficient capture SF6 from SF6/N2 mixture. Sep. Purif. Technol. 327, 124929 (2023).
Hu, Y. et al. Pore engineering in cost-effective and stable Al-MOFs for efficient capture of the greenhouse gas SF6. Chem. Eng. J. 471, 144851 (2023).
Chang, M. et al. Metal-organic framework-based single-molecule SF6 trap for record SF6 capture. Chem. Mater. 34, 9134–9143 (2022).
Yeskendir, B. et al. From metal-organic framework powders to shaped solids: recent developments and challenges. Adv. Mater. 2, 7139–7186 (2021).
Alvarez, E. et al. The structure of the aluminum fumarate metal-organic framework A520. Angew. Chem. Int. Ed. 54, 3664–3668 (2015).
Song, Y. et al. Shaped layered two-dimensional fluorinated metal-organic frameworks for highly efficient acetylene/ethylene separation. Sep. Purif. Technol. 323, 124377 (2023).
Myers, A. & Prausnitz, J. Thermodynamics of mixed-gas adsorption. AIChE J. 11, 121–127 (1965).
Segall, M. et al. First-principles simulation: ideas, illustrations and the CASTEP code. J. Phys. Condens. Matter. 14, 2717–2744 (2002).
Lu, T. & Chen, F. Multiwfn. A multifunctional wavefunction analyzer. J. Comput. Chem. 33, 580592 (2012).
Humphrey, W., Dalke, A. & Schulten, K. VMD: visual molecular dynamics. J. Mol. Graph. 14, 33–38 (1996).
Ministry of Housing and Urban-Rural Development of the People's Republic of China. PRC National Standard: Standard for calculation of energy consumption in petrochemical engineering design. https://www.chinesestandard.net/PDF/English.aspx/GBT50441-2016 (2016).
Acknowledgements
This work was supported by the National Natural Science Foundation of China (No. 22288102, 22178305, 22225802, 22308312) and the Zhejiang Provincial Natural Science Foundation of China (LR21B060002).
Author information
Authors and Affiliations
Contributions
Q.Y. designed the conceptual approach for research; J.L. and Y.C. carried out the synthesis experimental tests and conducted the computational simulations; T.K. contributed to the synthesis, separation test, and discussion. Y.J. contributed to the breakthrough separation tests; R.F. and G.X. contributed to the synthesis and experimental tests; L.Y. contributed to equipment preparation and data acquisition; Z.B., Z.Z., Q.R., and Q.Y. offered supervision and discussion; J.L., T.K. and Q.Y. wrote and revised the manuscript; All authors reviewed and approved the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Xiufeng Xu and the other anonymous reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Li, J., Chen, Y., Ke, T. et al. Efficient continuous SF6/N2 separation using low-cost and robust metal-organic frameworks composites. Nat Commun 16, 632 (2025). https://doi.org/10.1038/s41467-025-56031-5
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-025-56031-5
This article is cited by
-
Probing the thermal decomposition mechanism of CF3SO2F by deep learning molecular dynamics
Communications Chemistry (2025)